Sephadex® LH-20, Isolation, and Purification of Flavonoids from Plant Species: A Comprehensive Review
Abstract
1. Introduction
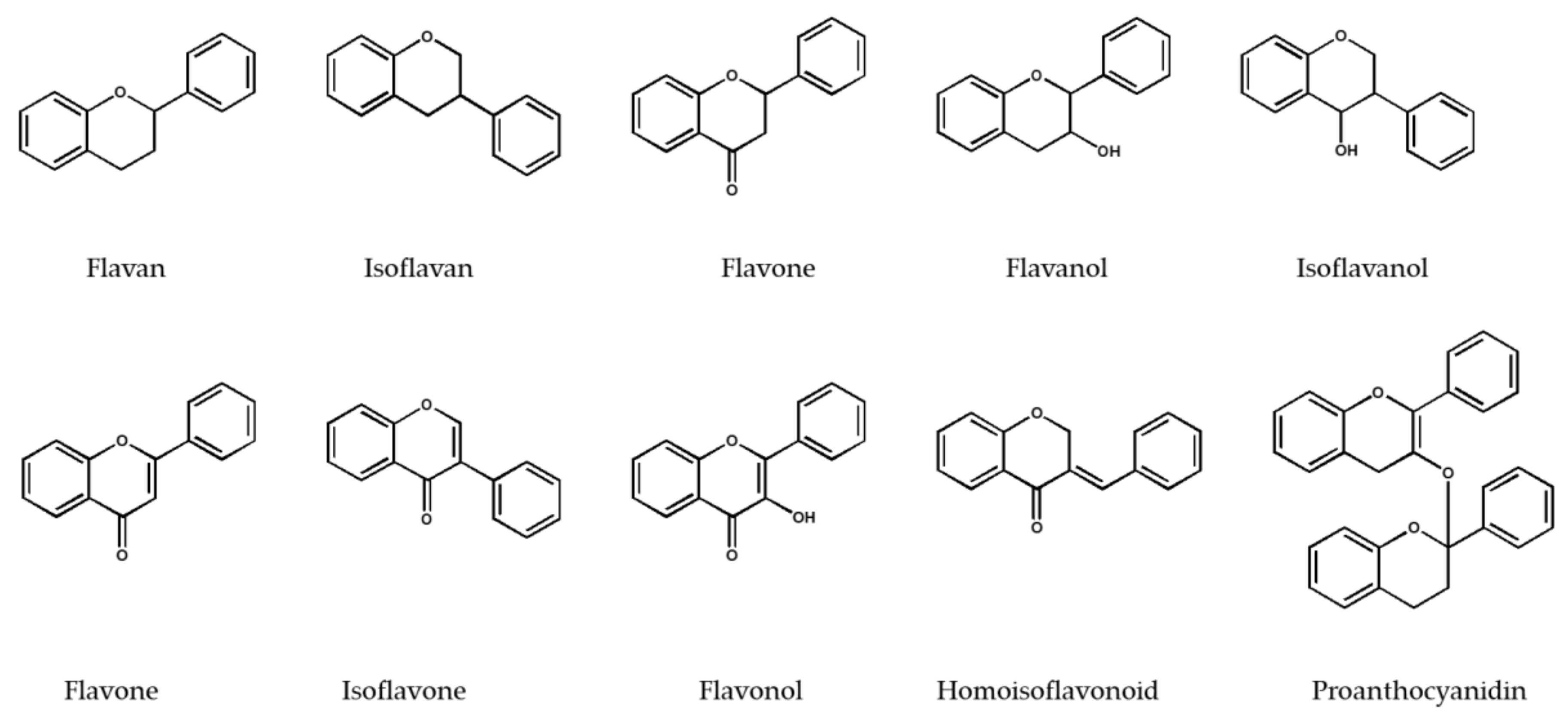
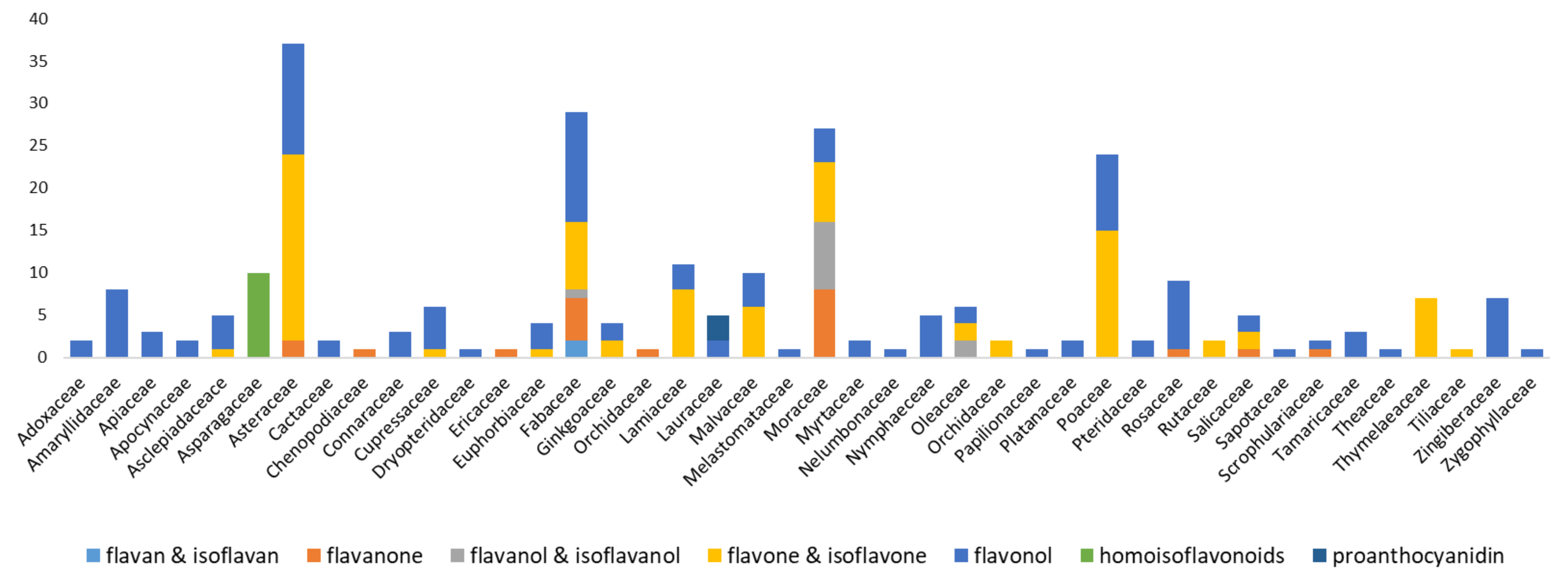
2. Isolation of Various Flavonoid Classifications by Sephadex® LH-20
2.1. Flavan and Isoflavan Derivatives
2.2. Flavanone Derivatives
2.3. Flavanol and Isoflavanol Derivatives
2.4. Flavone and Isoflavone Derivatives
2.5. Flavonol Derivatives
2.6. Homoisoflavonoid Derivatives
2.7. Proanthocyanidins
3. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Feng, W.; Hao, Z.; Li, M. Isolation and structure identification of flavonoids. In Flavonoids—From Biosynthesis to Human Health; InTech: London, UK, 2017. [Google Scholar]
- Amarowicz, R.; Kolodziejczyk, P.P.; Pegg, R.B. Chromatographic separation of phenolic compounds from rapeseed by a Sephadex LH-20 column with ethanol as the mobile phase. J. Liq. Chromatogr. Relat. Technol. 2003, 26, 2157–2165. [Google Scholar] [CrossRef]
- Markham, K.R. Isolation techniques for flavonoids. In The Flavonoids; Springer US: Boston, MA, USA, 1975; pp. 1–44. [Google Scholar]
- Chávez-González, M.L.; Sepúlveda, L.; Verma, D.K.; Luna-García, H.A.; Rodríguez-Durán, L.V.; Ilina, A.; Aguilar, C.N. Conventional and emerging extraction processes of flavonoids. Processes 2020, 8, 434. [Google Scholar] [CrossRef]
- Putnik, P.; Kovačević, D.B.; Radojčin, M.; Dragović-Uzelaca, V. Influence of acidity and extraction time on the recovery of flavonoids from grape skin pomace optimized by Response Surface Methodology. Chem. Biochem. Eng. Q. J. 2017, 30, 455–464. [Google Scholar] [CrossRef]
- Roselló-Soto, E.; Martí-Quijal, F.; Cilla, A.; Munekata, P.; Lorenzo, J.; Remize, F.; Barba, F. Influence of temperature, solvent and pH on the selective extraction of phenolic compounds from tiger nuts by-products: Triple-TOF-LC-MS-MS characterization. Molecules 2019, 24, 797. [Google Scholar] [CrossRef] [PubMed]
- Hameed, B.S.; Bhatt, C.S.; Nagaraj, B.; Suresh, A.K. Chromatography as an efficient technique for the separation of diversified nanoparticles. In Nanomaterials in Chromatography; Elsevier: Amsterdam, The Netherlands, 2018; pp. 503–518. [Google Scholar]
- Lindqvist, B.; Storgårds, T. Molecular-sieving properties of starch. Nature 1955, 175, 511–512. [Google Scholar] [CrossRef]
- Hong, P.; Koza, S.; Bouvier, E.S.P. Size-exclusion chromatography for the analysis of protein biotherapeutics and their aggregates. J. Liq. Chromatogr. Relat. Technol. 2012, 35, 2923–2950. [Google Scholar] [CrossRef]
- Flodin, P.G.M. Process for Preparing Hydrophilic Copolymerization and Product Obtained Thereby. U.S. Patent 3208994A, 28 September 1965. [Google Scholar]
- Liu, R.; Wen, X.; Shao, F.; Zhang, P.; Huang, H.; Zhang, S. Flavonoids from heartwood of Dalbergia cochinchinensis. Chinese Herb. Med. 2016, 8, 89–93. [Google Scholar] [CrossRef]
- Bergeron, C.; Marston, A.; Hakizamungu, E.; Hostettmann, K. Antifungal constituents of Chenopodium procerum. Int. J. Pharmacogn. 1995, 33, 115–119. [Google Scholar] [CrossRef]
- Si, C.L.; Lu, Y.Y.; Qin, P.P.; Sun, R.C.; Ni, Y.H. Phenolic extractives with chemotaxonomic significance from the bark of Paulownia tomentosa var. Tomentosa. BioResources 2011, 6, 5086–5098. [Google Scholar]
- Si, C.-L.; Xu, Q.; Li, S.-M.; Liu, Z. Phenolic compounds from Populus davidiana wood. Chem. Nat. Compd. 2009, 45, 634–636. [Google Scholar] [CrossRef]
- Rayyan, S.; Fossen, T.; Nateland, H.S.; Andersen, Ø.M. Isolation and identification of flavonoids, including flavone rotamers, from the herbal drug ‘crataegi folium cum flore’ (hawthorn). Phytochem. Anal. 2005, 16, 334–341. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.-W.; Ko, W.-M.; Park, J.-H.; Seo, K.-H.; Oh, E.-J.; Lee, D.-Y.; Lee, D.-S.; Kim, Y.-C.; Lim, D.-W.; Han, D.; et al. Isoprenylated flavonoids from the root bark of Morus alba and their hepatoprotective and neuroprotective activities. Arch. Pharm. Res. 2015, 38, 2066–2075. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.-W.; Park, J.-H.; Lee, Y.-G.; Seo, K.-H.; Oh, E.-J.; Lee, D.-Y.; Lim, D.-W.; Han, D.; Baek, N.-I. Three new isoprenylated flavonoids from the root bark of Morus alba. Molecules 2016, 21, 1112. [Google Scholar] [CrossRef]
- Liu, B.; Yan, T.; Xiao, J.; Wang, X. α-Glucosidase inhibitors and antioxidants from root bark of Morus alba. Chinese Herb. Med. 2018, 10, 331–335. [Google Scholar] [CrossRef]
- Lan, X.; Wang, W.; Li, Q.; Wang, J. The natural flavonoid pinocembrin: Molecular targets and potential therapeutic applications. Mol. Neurobiol. 2016, 53, 1794–1801. [Google Scholar] [CrossRef]
- Lan, X.; Han, X.; Li, Q.; Li, Q.; Gao, Y.; Cheng, T.; Wan, J.; Zhu, W.; Wang, J. Pinocembrin protects hemorrhagic brain primarily by inhibiting toll-like receptor 4 and reducing M1 phenotype microglia. Brain. Behav. Immun. 2017, 61, 326–339. [Google Scholar] [CrossRef]
- Shi, S.; Zhao, Y.; Zhou, H.; Zhang, Y.; Jiang, X.; Huang, K. Identification of antioxidants from Taraxacum mongolicum by high-performance liquid chromatography–diode array detection–radical-scavenging detection–electrospray ionization mass spectrometry and nuclear magnetic resonance experiments. J. Chromatogr. A 2008, 1209, 145–152. [Google Scholar] [CrossRef]
- Tanagornmeatar, K.; Chaotham, C.; Sritularak, B.; Likhitwitayawuid, K.; Chanvorachote, P. Cytotoxic and anti-metastatic activities of phenolic compounds from Dendrobium ellipsophyllum. Anticancer Res. 2014, 34, 6573–6580. [Google Scholar]
- Lee, Y.-G.; Lee, H.; Jung, J.-W.; Seo, K.-H.; Lee, D.Y.; Kim, H.-G.; Ko, J.-H.; Lee, D.-S.; Baek, N.-I. Flavonoids from Chionanthus retusus (Oleaceae) flowers and their protective effects against glutamate-induced cell toxicity in HT22 cells. Int. J. Mol. Sci. 2019, 20, 3517. [Google Scholar] [CrossRef]
- Zheng, Z.-P.; Liang, J.-Y.; Hu, L.-H. Water-soluble constituents of Cudrania tricuspidata (Carr.) Bur. J. Integr. Plant Biol. 2006, 48, 996–1000. [Google Scholar] [CrossRef]
- Park, J.-H.; Jung, Y.-J.; Jung, J.-W.; Shrestha, S.; Lim, D.W.; Han, D.; Baek, N.-I. A new flavonoid glycoside from the root bark of Morus alba L. Nat. Prod. Res. 2014, 28, 1859–1863. [Google Scholar] [CrossRef] [PubMed]
- Awouafack, M.D.; Tchuenguem, R.T.; Ito, T.; Dzoyem, J.P.; Tane, P.; Morita, H. A new isoflavanol from the fruits of Kotschya strigosa (Fabaceae). Helv. Chim. Acta 2016, 99, 321–324. [Google Scholar] [CrossRef]
- Xuan, L.; Bin-Feng, Z.; Li, Y.; Gui-Xin, C.; Zheng-Tao, W. Two new chromones and a new flavone glycoside from Imperata cylindrica. Chin. J. Nat. Med. 2013, 11, 77–80. [Google Scholar]
- Jung, M.J.; Chung, H.Y.; Kang, S.S.; Choi, J.H.; Bae, K.S.; Choi, J.S. Antioxidant activity from the stem bark of Albizzia julibrissin. Arch. Pharm. Res. 2003, 26, 458–462. [Google Scholar] [CrossRef] [PubMed]
- Mavundza, E.J.; Tshikalange, T.E.; Lall, N.; Hussein, A.A.; Mudau, F.N.; Meyer, J.J.M. Antioxidant activity and cytotoxicity effect of flavonoids isolated from Athrixia phylicoides. J. Med. Plants Res. 2010, 4, 2584–2587. [Google Scholar]
- Imran, M.; Rauf, A.; Abu-Izneid, T.; Nadeem, M.; Shariati, M.A.; Khan, I.A.; Imran, A.; Orhan, I.E.; Rizwan, M.; Atif, M.; et al. Luteolin, a flavonoid, as an anticancer agent: A review. Biomed. Pharmacother. 2019, 112, 108612. [Google Scholar] [CrossRef]
- Luo, Y.; Shang, P.; Li, D. Luteolin: A flavonoid that has multiple cardio-protective effects and its molecular mechanisms. Front. Pharmacol. 2017, 8, 692. [Google Scholar] [CrossRef]
- Lin, Y.; Shi, R.; Wang, X.; Shen, H.-M. Luteolin, a flavonoid with potential for cancer prevention and therapy. Curr. Cancer Drug Targets 2008, 8, 634–646. [Google Scholar] [CrossRef]
- Abou-Zeid, A.H.; Farag, M.A.; Hamed, M.A.A.; Kandil, Z.A.A.; El-Akad, R.H.; El-Rafie, H.M. Flavonoid chemical composition and antidiabetic potential of Brachychiton acerifolius leaves extract. Asian Pac. J. Trop. Biomed. 2017, 7, 389–396. [Google Scholar] [CrossRef]
- Erenler, R.; Sen, O.; Yildiz, I.; Aydın, A. Antiproliferative activities of chemical constituents isolated from Thymus praecox subsp. grossheimii (ronniger) jalas. Rec. Nat. Prod. 2016, 10, 766–770. [Google Scholar]
- Hyun, S.K.; Jung, H.A.; Chung, H.Y.; Choi, J.S. In vitro peroxynitrite scavenging activity of 6-hydroxykynurenic acid and other flavonoids from Gingko biloba yellow leaves. Arch. Pharm. Res. 2006, 29, 1074–1079. [Google Scholar] [CrossRef] [PubMed]
- Hyun, H.B.; Shrestha, S.; Boo, K.H.; Cho, S.K. Evaluation of antioxidant potential of ethyl acetate fraction of Rosmarinus officinalis L. and its major components. J. Korean Soc. Appl. Biol. Chem. 2015, 58, 715–722. [Google Scholar] [CrossRef]
- Sándor, Z.; Mottaghipisheh, J.; Veres, K.; Hohmann, J.; Bencsik, T.; Horváth, A.; Kelemen, D.; Papp, R.; Barthó, L.; Csupor, D. Evidence supports tradition: The in vitro effects of roman chamomile on smooth muscles. Front. Pharmacol. 2018, 9, 323. [Google Scholar] [CrossRef]
- Taiwo, B.J.; Obuotor, E.; Onawunmi, G.O.; Ogundaini, A.O. Radical scavenging compounds from the aerial parts of Solenostemon monostachys briq (Lamiaceae). African J. Tradit. Complement. Altern. Med. 2015, 12, 140. [Google Scholar] [CrossRef]
- Sajjadi, S.; Delazari, Z.; Aghaei, M.; Ghannadian, M. Flavone constituents of Phlomis bruguieri Desf. with cytotoxic activity against MCF-7 breast cancer cells. Res. Pharm. Sci. 2018, 13, 422. [Google Scholar] [PubMed]
- Si, C.-L.; Lu, Y.-Y.; Zhang, Y.; Xu, J.; Qin, P.-P.; Sun, R.-C.; Ni, Y.-H. Antioxidative low molecular weight extractives from triploid Populus tomentosa xylem. BioResources 2011, 6, 232–242. [Google Scholar]
- Abdallah, W.E.; Elsayed, W.M.; Hassanein, H.D.; Shams, K.A.; Ismail, S.I.; Ali, S.M. Phytochemical and biological investigations of Onopordum alexandrinum seeds. Res. J. Pharm. Biol. Chem. Sci. 2015, 6, 361–374. [Google Scholar]
- Tan, J.; Shenghua, L.; Zeng, J.; Wu, X. Anti-oxidant and hepatoprotective activities of total flavonoids of Indocalamus latifolius. Bangladesh J. Pharmacol. 2015, 10, 779. [Google Scholar] [CrossRef]
- Cui, H.-X.; Zhang, L.-S.; Yan, H.-G.; Yuan, K.; Jin, S.-H. Constituents of flavonoids from Tridax procumbens L. and antioxidant activity. Pharmacogn. Mag. 2020, 16, 201. [Google Scholar]
- Gohari, A.R.; Ebrahimi, H.; Saeidnia, S.; Foruzani, M.; Ebrahimi, P.; Ajani, Y. Flavones and flavone glycosides from Salvia macrosiphon Boiss. Iran. J. Pharm. Res. IJPR 2011, 10, 247–251. [Google Scholar] [PubMed]
- Komaki, E.; Yamaguchi, S.; Maru, I.; Kinoshita, M.; Kakehi, K.; Ohta, Y.; Tsukada, Y. Identification of anti-α-amylase components from olive leaf extracts. Food Sci. Technol. Res. 2003, 9, 35–39. [Google Scholar] [CrossRef]
- Hwang, J.-H.; Yang, H.-S.; Ra, K.S.; Park, S.S.; Yu, K.-W. Intestinal immune system-modulating activity through peyer’s patch of flavonoid glycoside purified from Citrus unshiu peel. J. Food Biochem. 2013, 37, 151–160. [Google Scholar] [CrossRef]
- Salehi, B.; Venditti, A.; Sharifi-Rad, M.; Kręgiel, D.; Sharifi-Rad, J.; Durazzo, A.; Lucarini, M.; Santini, A.; Souto, E.; Novellino, E.; et al. The therapeutic potential of apigenin. Int. J. Mol. Sci. 2019, 20, 1305. [Google Scholar] [CrossRef]
- Shankar, E.; Goel, A.; Gupta, K.; Gupta, S. Plant flavone apigenin: An emerging anticancer agent. Curr. Pharmacol. Reports 2017, 3, 423–446. [Google Scholar] [CrossRef]
- Ali, F.; Rahul-Naz, F.; Jyoti, S.; Siddique, Y.H. Health functionality of apigenin: A review. Int. J. Food Prop. 2017, 20, 1197–1238. [Google Scholar] [CrossRef]
- Yan, X.; Qi, M.; Li, P.; Zhan, Y.; Shao, H. Apigenin in cancer therapy: Anti-cancer effects and mechanisms of action. Cell Biosci. 2017, 7, 50. [Google Scholar] [CrossRef]
- Zhou, X.; Wang, F.; Zhou, R.; Song, X.; Xie, M. Apigenin: A current review on its beneficial biological activities. J. Food Biochem. 2017, 41, e12376. [Google Scholar] [CrossRef]
- Sun, J.; He, X.-M.; Zhao, M.-M.; Li, L.; Li, C.-B.; Dong, Y. Antioxidant and nitrite-scavenging capacities of phenolic compounds from sugarcane (Saccharum officinarum L.) tops. Molecules 2014, 19, 13147–13160. [Google Scholar] [CrossRef]
- Luo, P.; Zhang, Z.; Yi, T.; Zhang, H.; Liu, X.; Mo, Z. Anti-inflammatory activity of the extracts and fractions from Erigeron multiradiatus through bioassay-guided procedures. J. Ethnopharmacol. 2008, 119, 232–237. [Google Scholar] [CrossRef]
- Zielińska-Pisklak, M.A.; Kaliszewska, D.; Stolarczyk, M.; Kiss, A.K. Activity-guided isolation, identification and quantification of biologically active isomeric compounds from folk medicinal plant Desmodium adscendens using high performance liquid chromatography with diode array detector, mass spectrometry and multidiment. J. Pharm. Biomed. Anal. 2015, 102, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Aderogba, M.A.; McGaw, L.J.; Bezabih, M.; Abegaz, B.M. Isolation and characterisation of novel antioxidant constituents of Croton zambesicus leaf extract. Nat. Prod. Res. 2011, 25, 1224–1233. [Google Scholar] [CrossRef] [PubMed]
- CHEN, D.; BI, D.; SONG, Y.-L.; TU, P.-F. Flavanoids from the stems of Aquilaria sinensis. Chin. J. Nat. Med. 2012, 10, 287–291. [Google Scholar] [CrossRef]
- Lee, E.H. Flavonoids from the leaves of Thuja orientalis inhibit the aldose reductase and the formation of advanced glycation endproducts. J. Korean Soc. Appl. Biol. Chem. 2009, 52, 448–455. [Google Scholar] [CrossRef]
- Lu, M.; Kong, Q.; Xu, X.; Lu, H.; Lu, Z.; Yu, W.; Zuo, B.; Su, J.; Guo, R. Pectolinarigenin—A flavonoid compound from Cirsium Japonicum with potential anti-proliferation activity in MCF- 7 breast cancer cell. Trop. J. Pharm. Res. 2014, 13, 225. [Google Scholar] [CrossRef]
- Mirzahosseini, G.; Manayi, A.; Khanavi, M.; Safavi, M.; Salari, A.; Madjid-Ansari, A.; San’ati, H.; Vazirian, M. Bio-guided isolation of Centaurea bruguierana subsp. belangerana cytotoxic components. Nat. Prod. Res. 2019, 33, 1687–1690. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.-M.; Ibrahim, R.K. Tricin—A potential multifunctional nutraceutical. Phytochem. Rev. 2010, 9, 413–424. [Google Scholar] [CrossRef]
- Matsuta, T.; Sakagami, H.; Satoh, K.; Kanamoto, T.; Terakubo, S.; Nakashima, H.; Kitajima, M.; Oizumi, H.; Oizumi, T. Biological activity of luteolin glycosides and tricin from Sasa senanensis Rehder. In Vivo 2011, 25, 757–762. [Google Scholar]
- Mu-Xin, Z.; LIU, Y.-Y.; Wei, S.; Xiao-Hong, Y.; Guang-Shu, W. Isolation and identification of novel flavonoids from the bract of Zea mays L. Chem. J. Chin. Univ. 2011, 32, 2554–2557. [Google Scholar]
- ZHANG, W.-K.; XU, J.-K.; ZHANG, L.; DU, G.-H. Flavonoids from the bran of Avena sativa. Chin. J. Nat. Med. 2012, 10, 110–114. [Google Scholar] [CrossRef]
- Sala, A.; Recio, M.C.; Giner, R.M.; Manez, S.; Rios, J.L. Anti-phospholipase A(2) and anti-inflammatory activity of Santolina chamaecyparissus. LIFE Sci. 2000, 66, PL35–PL40. [Google Scholar] [CrossRef]
- Ahmed, A.S.; Moodley, N.; Eloff, J.N. Bioactive compounds from the leaf extract of Bauhinia galpinii (Fabaceae) used as antidiarrhoeal therapy in southern Africa. S. Afr. J. Bot. 2019, 126, 345–353. [Google Scholar] [CrossRef]
- Youssef-Moustafa, A.M.; Khodair, A.I.; Saleh, M.A. Isolation, structural elucidation of flavonoid constituents from Leptadenia pyrotechnica and evaluation of their toxicity and antitumor activity. Pharm. Biol. 2009, 47, 539–552. [Google Scholar] [CrossRef]
- Zhao, H.-Y.; Yang, L.; Wei, J.; Huang, M.; Jiang, J.-G. Bioactivity evaluations of ingredients extracted from the flowers of Citrus aurantium L. var. amara Engl. Food Chem. 2012, 135, 2175–2181. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.B.; Hwang, S.H.; Wang, Z.; Yu, J.M.; Lim, S.S. Rapid identification and isolation of inhibitors of rat lens aldose reductase and antioxidant in Maackia amurensis. Biomed Res. Int. 2017, 2017, 1–10. [Google Scholar]
- Anand-David, A.V.; Arulmoli, R.; Parasuraman, S. Overviews of biological importance of quercetin: A bioactive flavonoid. Pharmacogn. Rev. 2016, 10, 84–89. [Google Scholar]
- Formica, J.V.; Regelson, W. Review of the biology of quercetin and related bioflavonoids. Food Chem. Toxicol. 1995, 33, 1061–1080. [Google Scholar] [CrossRef]
- Rauf, A.; Imran, M.; Khan, I.A.; Ur-Rehman, M.-; Gilani, S.A.; Mehmood, Z.; Mubarak, M.S. Anticancer potential of quercetin: A comprehensive review. Phyther. Res. 2018, 32, 2109–2130. [Google Scholar] [CrossRef]
- Deng, Q.; Li, X.X.; Fang, Y.; Chen, X.; Xue, J. Therapeutic potential of quercetin as an antiatherosclerotic agent in atherosclerotic cardiovascular disease: A review. Evidence-Based Complement. Altern. Med. 2020, 2020, 1–12. [Google Scholar] [CrossRef]
- Baek, Y.-S.; Song, N.-Y.; Nam, T.-G.; Kim, D.-O.; Kang, H.-C.; Kwon, O.-K.; Baek, N.-I. Flavonoids from Fragaria ananassa calyx and their antioxidant capacities. J. Korean Soc. Appl. Biol. Chem. 2015, 58, 787–793. [Google Scholar] [CrossRef]
- Cao, S.; Wan, C.; Yu, Y.; Zhou, S.; Tian, S. Isolation and identification of phenolic compounds from Gynura divaricata leaves. Pharmacogn. Mag. 2011, 7, 101. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Wan, C.; Zhou, S. Quercetin - a flavonoid compound from Sarcopyramis bodinieri var. delicate with potential apoptotic activity in HepG2 liver cancer cells. Trop. J. Pharm. Res. 2013, 12, 529–533. [Google Scholar]
- Liao, J.; Tang, Y.; Tan, C.; Ni, H.; Wu, X.; Li, N.; Jia, X.; Xing, Y.; Zhang, P. ACE and platelet aggregation inhibitors from Tamarix hohenackeri Bunge (host plant of Herba Cistanches) growing in Xinjiang. Pharmacogn. Mag. 2014, 10, 111. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Huang, X.; Lv, Z. Isolation and identification of flavonoids components from Pteris vittata L. Springerplus 2016, 5, 1649. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Gao, H.; Zhao, J.; Wang, Q.; Zhou, L.; Han, J.; Yu, Z.; Yang, F. Preparative separation of phenolic compounds from Halimodendron halodendron by high-speed counter-current chromatography. Molecules 2010, 15, 5998–6007. [Google Scholar] [CrossRef] [PubMed]
- Ahmadu, A.A.; Hassan, H.S.; Abubakar, M.; Akpulu, I.N. Flavonoid glycosides from Byrsocarpus coccineus leaves. Schum and Thonn (Connaraceae). African J. Tradit. Complement. Altern. Med. 2007, 4, 257–260. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lim, J.P.; Song, Y.C.; Kim, J.W.; Ku, C.H.; Eun, J.S.; Leem, K.H.; Kim, D.K. Free radical scavengers from the heartwood of Juniperus chinensis. Arch. Pharm. Res. 2002, 25, 449–452. [Google Scholar] [CrossRef]
- Jarial, R.; Shard, A.; Thakur, S.; Sakinah, M.; Zularisam, A.W.; Rezania, S.; Kanwar, S.S.; Singh, L. Characterization of flavonoids from fern Cheilanthes tenuifolia and evaluation of antioxidant, antimicrobial and anticancer activities. J. King Saud Univ. Sci. 2018, 30, 425–432. [Google Scholar] [CrossRef]
- Kassem, M.E.S.; Ibrahim, L.F.; Hussein, S.R.; El-Sharawy, R.; El-Ansari, M.A.; Hassanane, M.M.; Booles, H.F. Myricitrin and bioactive extract of Albizia amara leaves: DNA protection and modulation of fertility and antioxidant-related genes expression. Pharm. Biol. 2016, 54, 2404–2409. [Google Scholar] [CrossRef]
- Mahmoud, A.H.; Foda, D.S.; Ibrahim, N.E.; El-Hagrassi, A.M.; Yousef, O.M.H. Secondary metabolites and biological activities of Allium porrum L. attack Ehrlich ascites carcinoma in mice. Egypt. J. Chem. 2019, 62, 211–227. [Google Scholar]
- Yuenyongsawad, S.; Bunluepuech, K.; Wattanapiromsakul, C.; Tewtrakul, S. Anti-cancer activity of compounds from Bauhinia strychnifolia stem. J. Ethnopharmacol. 2013, 150, 765–769. [Google Scholar] [CrossRef]
- Gullón, B.; Lú-Chau, T.A.; Moreira, M.T.; Lema, J.M.; Eibes, G. Rutin: A review on extraction, identification and purification methods, biological activities and approaches to enhance its bioavailability. Trends Food Sci. Technol. 2017, 67, 220–235. [Google Scholar] [CrossRef]
- Al-Dhabi, N.A.; Arasu, M.V.; Park, C.H.; Park, S.U. An up-to-date review of rutin and its biological and pharmacological activities. EXCLI J. 2015, 14, 59–63. [Google Scholar]
- Ganeshpurkar, A.; Saluja, A.K. The pharmacological potential of rutin. Saudi Pharm. J. 2017, 25, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Jayaprakasha, G.K.; Ohnishi-Kameyama, M.; Ono, H.; Yoshida, M.; Jaganmohan-Rao, L. Phenolic constituents in the fruits of Cinnamomum zeylanicum and their antioxidant activity. J. Agric. Food Chem. 2006, 54, 1672–1679. [Google Scholar] [CrossRef] [PubMed]
- Erhirhie, E.O.; Emeghebo, C.N.; Ilodigwe, E.E.; Ajaghaku, D.L.; Umeokoli, B.O.; Eze, P.M.; Ngwoke, K.G.; Okoye, F.B.C. Dryopteris filix-mas (L.) Schott ethanolic leaf extract and fractions exhibited profound anti-inflammatory activity. Avicenna J. Phytomed. 2019, 9, 396–409. [Google Scholar] [PubMed]
- Jiang, C.-L.; Tsai, S.-F.; Lee, S.-S. Flavonoids from Curcuma longa leaves and their NMR assignments. Nat. Prod. Commun. 2015, 10, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Taiwo, B.; Igbeneghu, O. Antioxidant and antibacterial activities of flavonoid glycosides from Ficus Exasperata Vahl-Holl (Moraceae) leaves. Afr. J. Tradit. Complement. Altern. Med. 2014, 11, 97. [Google Scholar] [CrossRef] [PubMed]
- Yesilada, E.; Gürbüz, İ.; Toker, G. Anti-ulcerogenic activity and isolation of the active principles from Sambucus ebulus L. leaves. J. Ethnopharmacol. 2014, 153, 478–483. [Google Scholar] [CrossRef] [PubMed]
- Nawwar, M.A.; Hashem, A.N.; Hussein, S.A.; Swilam, N.F.; Becker, A.; Haertel, B.; Lindequist, U.; El-Khatib, A.; Linscheid, M.W. Phenolic profiling of an extract from Eugenia jambos L. (Alston)—The structure of three flavonoid glycosides—Antioxidant and cytotoxic activities. Pharmazie 2016, 71, 162–168. [Google Scholar]
- Lim, S.S.; Jung, Y.J.; Hyun, S.K.; Lee, Y.S.; Choi, J.S. Rat lens aldose reductase inhibitory constituents of Nelumbo nucifera stamens. Phyther. Res. 2006, 20, 825–830. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.A.; Park, J.C.; Chung, H.Y.; Kim, J.; Choi, J.S. Antioxidant flavonoids and chlorogenic acid from the leaves of Eriobotrya japonica. Arch. Pharm. Res. 1999, 22, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Liu, Y.; Yi, Y.; Miao, Q.; Liu, S.; Zhao, F.; Cong, W.; Wang, C.; Xia, C. Purification of quercetin-3-O-sophoroside and isoquercitrin from Poacynum hendersonii leaves using macroporous resins followed by Sephadex LH-20 column chromatography. J. Chromatogr. B 2017, 1048, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-Y.; Shrestha, S.; Seo, W.D.; Lee, M.-H.; Jeong, T.-S.; Cho, J.H.; Song, Y.C.; Kang, H.W.; Rho, Y.-D.; Baek, N.-I. Structural and quantitative analysis of antioxidant and low-density lipoprotein-antioxidant flavonoids from the grains of sugary rice. J. Med. Food 2012, 15, 399–405. [Google Scholar] [CrossRef]
- Azuma, C.M.; Santos, F.C.S.; Lago, J.H.G. Flavonoids and fatty acids of Camellia japonica leaves extract. Rev. Bras. Farmacogn. 2011, 21, 1159–1162. [Google Scholar] [CrossRef]
- Delnavazi, M.R.; Hadjiakhoondi, A.; Delazar, A.; Ajani, Y.; Tavakoli, S.; Yassa, N. Phytochemical and antioxidant investigation of the aerial parts of Dorema glabrum fisch. & C.A. mey. Iran. J. Pharm. Res. 2015, 14, 925–931. [Google Scholar]
- Lam, S.-H.; Wang, C.-Y.; Chen, C.-K.; Lee, S.-S. Chemical investigation of Phyllanthus reticulatus by HPLC-SPE-NMR and conventional methods. Phytochem. Anal. 2007, 18, 251–255. [Google Scholar] [CrossRef]
- Mottaghipisheh, J.; Mahmoodi-Sourestani, M.; Kiss, T.; Horváth, A.; Tóth, B.; Ayanmanesh, M.; Khamushi, A.; Csupor, D. Comprehensive chemotaxonomic analysis of saffron crocus tepal and stamen samples, as raw materials with potential antidepressant activity. J. Pharm. Biomed. Anal. 2020, 184, 113183. [Google Scholar] [CrossRef]
- Ashrafizadeh, M.; Tavakol, S.; Ahmadi, Z.; Roomiani, S.; Mohammadinejad, R.; Samarghandian, S. Therapeutic effects of kaempferol affecting autophagy and endoplasmic reticulum stress. Phyther. Res. 2020, 34, 911–923. [Google Scholar] [CrossRef]
- Rasouli, H.; Farzaei, M.H.; Khodarahmi, R. Polyphenols and their benefits: A review. Int. J. Food Prop. 2017, 1–42. [Google Scholar] [CrossRef]
- Yang, C.; Yang, W.; He, Z.; He, H.; Yang, X.; Lu, Y.; Li, H. Kaempferol improves lung ischemia-reperfusion injury via antiinflammation and antioxidative stress regulated by SIRT1/HMGB1/NF-κB Axis. Front. Pharmacol. 2020, 10, 1635. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.Y.; Chen, Y.C. A review of the dietary flavonoid, kaempferol on human health and cancer chemoprevention. Food Chem. 2013, 138, 2099–2107. [Google Scholar] [CrossRef]
- Ren, J.; Lu, Y.; Qian, Y.; Chen, B.; Wu, T.; Ji, G. Recent progress regarding kaempferol for the treatment of various diseases (review). Exp. Ther. Med. 2019, 18, 2759–2776. [Google Scholar] [CrossRef] [PubMed]
- Calderon-Montano, J.M.; Burgos-Moron, E.; Perez-Guerrero, C.; Lopez-Lazaro, M. A review on the dietary flavonoid kaempferol. Mini Rev. Med. Chem. 2011, 11, 298–344. [Google Scholar] [CrossRef]
- Imran, M.; Salehi, B.; Sharifi-Rad, J.; Aslam-Gondal, T.; Saeed, F.; Imran, A.; Shahbaz, M.; Tsouh-Fokou, P.V.; Umair-Arshad, M.; Khan, H.; et al. Kaempferol: A key emphasis to its anticancer potential. Molecules 2019, 24, 2277. [Google Scholar] [CrossRef] [PubMed]
- Ouf, S.A.; Abdel-Hady, F.K.; El-Gamal, M.H.; Shaker, K.H. Isolation of antifungal compounds from some Zygophyllum species and their bioassay against two soil-borne plant pathogens. Folia Microbiol. 1994, 39, 215–221. [Google Scholar] [CrossRef]
- Erosa-Rejón, G.; Peña-Rodríguez, L.; Sterner, O. Isolation of kaempferol-3-rutinoside from the leaf extract of Sideroxylon foetidissimum subsp. Gaumeri. Rev. Latinoam. Química 2010, 38, 7–11. [Google Scholar]
- Ahmed, M.S.; El-Tanbouly, N.D.; Islam, W.T.; Sleem, A.A.; El-Senousy, A.S. Antiinflammatory flavonoids from Opuntia dillenii (Ker-Gawl) Haw. flowers growing in Egypt. Phyther. Res. 2005, 19, 807–809. [Google Scholar] [CrossRef]
- Kaouadji, M.; Morand, J.-M.; Garcia, J. Further acylated kaempferol rhamnosides from Platanus acerifolia buds. J. Nat. Prod. 1993, 56, 1618–1621. [Google Scholar] [CrossRef]
- Jung, H.A.; Kim, J.E.; Chung, H.Y.; Choi, J.S. Antioxidant principles of Nelumbo nucifera stamens. Arch. Pharm. Res. 2003, 26, 279–285. [Google Scholar] [CrossRef]
- al Musayeib, N.M.; Amina, M.; Mohamed, G.A.; Ibrahim, S.R.M. Plectranol A, a new sesquiterpene from Plectranthus cylindraceus growing in Saudi Arabia. Lett. Org. Chem. 2017, 14, 4–7. [Google Scholar] [CrossRef]
- Cho, J.-G.; Song, N.-Y.; Nam, T.-G.; Shrestha, S.; Park, H.-J.; Lyu, H.-N.; Kim, D.-O.; Lee, G.; Woo, Y.-M.; Jeong, T.-S.; et al. Flavonoids from the grains of C1/R-S transgenic rice, the transgenic Oryza sativa spp. japonica, and their radical scavenging activities. J. Agric. Food Chem. 2013, 61, 10354–10359. [Google Scholar] [PubMed]
- Hartati, S.; Angelina, M.; Meilawati, L.; Dewijanti, I.D. Isolation and characterization of compounds from the leaves of Pterocarpus indicus Willd and their antioxidant activity. Ann. Bogor. Trop. Gen. Bot. 2016, 20, 13–18. [Google Scholar]
- Leem, H.H.; Lee, G.Y.; Lee, J.S.; Lee, H.; Kim, J.H.; Kim, Y.H. Soluble epoxide hydrolase inhibitory activity of components from Leonurus japonicus. Int. J. Biol. Macromol. 2017, 103, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Yang, H.; Cho, N.; Kim, B.; Kim, Y.; Sung, S. Hepatoprotective constituents of Firmiana simplex stem bark against ethanol insult to primary rat hepatocytes. Pharmacogn. Mag. 2015, 11, 55. [Google Scholar]
- Jung, H.A.; Jin, S.E.; Park, J.-S.; Choi, J.S. Antidiabetic complications and anti-alzheimer activities of sophoflavescenol, a prenylated flavonol from Sophora flavescens, and its structure-activity relationship. Phyther. Res. 2011, 25, 709–715. [Google Scholar] [CrossRef]
- Li, L.; Yang, Y.; Hou, X.; Gu, D.; Ba, H.; Abdulla, R.; Wu, G.; Xin, X.; Aisa, H.A. Bioassay-guided separation and purification of water-soluble antioxidants from Carthamus tinctorius L. by combination of chromatographic techniques. Sep. Purif. Technol. 2013, 104, 200–207. [Google Scholar] [CrossRef]
- Castelli, M.V.; López, S.N. Homoisoflavonoids: Occurrence, biosynthesis, and biological activity. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2017; pp. 315–354. [Google Scholar]
- Abegaz, B.M.; Kinfe, H.H. Naturally occurring homoisoflavonoids: Phytochemistry, biological activities, and synthesis (Part II). Nat. Prod. Commun. 2019, 14. [Google Scholar] [CrossRef]
- Zhou, X.; Yuping, Z.; Zhao, H.; Liang, J.; Zhang, Y.; Shi, S. Antioxidant homoisoflavonoids from Polygonatum odoratum. Food Chem. 2015, 186, 63–68. [Google Scholar] [CrossRef]
- Rauf, A.; Imran, M.; Abu-Izneid, T.; Iahtisham-Ul, H.; Patel, S.; Pan, X.; Naz, S.; Sanches-Silva, A.; Saeed, F.; Rasul-Suleria, H.A.; et al. Proanthocyanidins: A comprehensive review. Biomed. Pharmacother. 2019, 116, 108999. [Google Scholar] [CrossRef]
- Cos, P.; Bruyne, T.; Hermans, N.; Apers, S.; Berghe, D.; Vlietinck, A. Proanthocyanidins in health care: Current and new trends. Curr. Med. Chem. 2004, 11, 1345–1359. [Google Scholar] [CrossRef] [PubMed]
- Huh, G.-W.; Park, J.-H.; Kang, J.-H.; Jeong, T.-S.; Kang, H.C.; Baek, N.-I. Flavonoids from Lindera glauca Blume as low-density lipoprotein oxidation inhibitors. Nat. Prod. Res. 2014, 28, 831–834. [Google Scholar] [CrossRef] [PubMed]
| Flavonoid Classes | Eluent Systems | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | |
| Flavan & Isoflavan | 2 | ||||||||||||||||
| Flavanone | 1 | 5 | 4 | 9 | |||||||||||||
| Flavanol & Isoflavanol | 2 | 7 | |||||||||||||||
| Flavone & Isoflavone | 1 | 1 | 8 | 9 | 1 | 2 | 28 | 22 | 4 | 2 | |||||||
| Flavonol | 3 | 3 | 3 | 8 | 1 | 37 | 26 | 2 | |||||||||
| Homoisoflavonoids | 10 | ||||||||||||||||
| Proanthocyanidin | 3 | ||||||||||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mottaghipisheh, J.; Iriti, M. Sephadex® LH-20, Isolation, and Purification of Flavonoids from Plant Species: A Comprehensive Review. Molecules 2020, 25, 4146. https://doi.org/10.3390/molecules25184146
Mottaghipisheh J, Iriti M. Sephadex® LH-20, Isolation, and Purification of Flavonoids from Plant Species: A Comprehensive Review. Molecules. 2020; 25(18):4146. https://doi.org/10.3390/molecules25184146
Chicago/Turabian StyleMottaghipisheh, Javad, and Marcello Iriti. 2020. "Sephadex® LH-20, Isolation, and Purification of Flavonoids from Plant Species: A Comprehensive Review" Molecules 25, no. 18: 4146. https://doi.org/10.3390/molecules25184146
APA StyleMottaghipisheh, J., & Iriti, M. (2020). Sephadex® LH-20, Isolation, and Purification of Flavonoids from Plant Species: A Comprehensive Review. Molecules, 25(18), 4146. https://doi.org/10.3390/molecules25184146