It Is Our Turn to Get Cannabis High: Put Cannabinoids in Food and Health Baskets
Abstract
1. Introduction
2. Cannabis Botany and Its Constituents
3. Cannabis Secondary Metabolites
4. Phenolic Compounds: Flavonoids, Stilbenoids, Lignamides, and Phenolic Amides
5. Terpenoids
6. Alkaloids
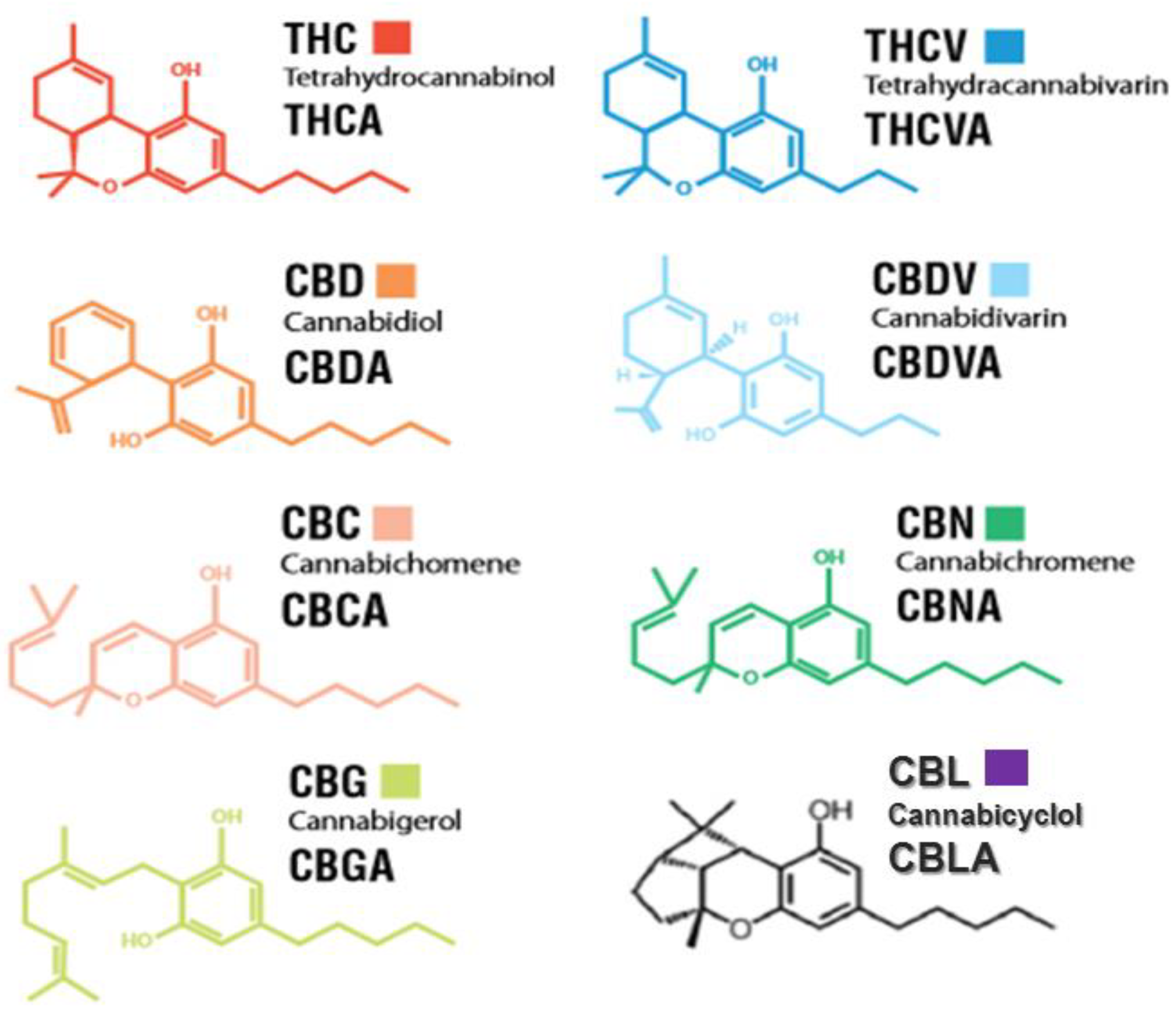
7. Phytocannabinoids
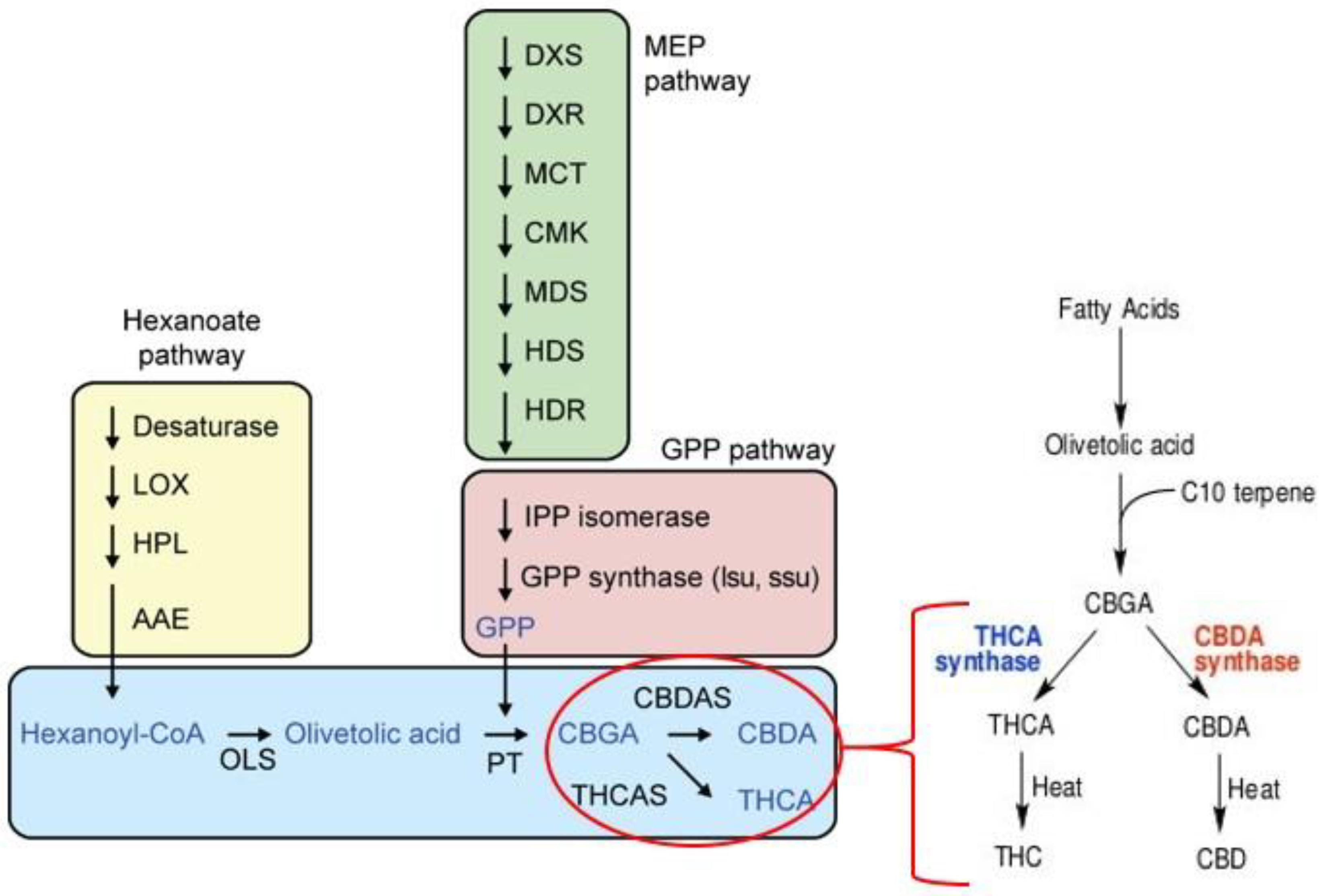
Phytocannabinoids Biosynthesis and Its Gene Networks
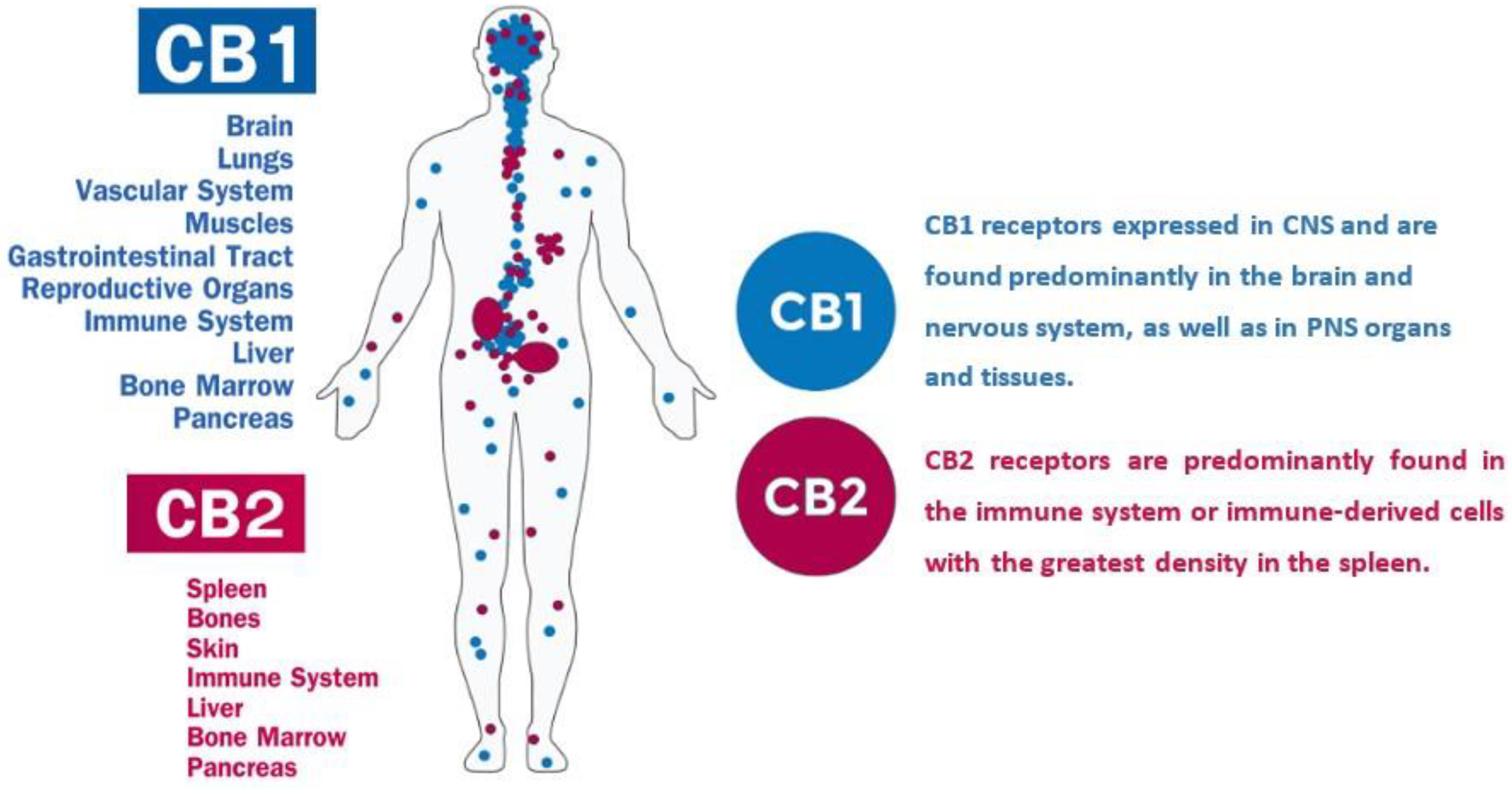
8. Medicinal Properties of Cannabinoids
8.1. Cannabis Flos Variety Bedrocan®
8.2. Cannabis Flos Variety Bedrobinol®
8.3. Marinol®
8.4. Syndros® (Dronabinol® Oral Solution)
8.5. Namisol®
8.6. Cesamet™
8.7. Sativex®
8.8. Epidiolex®
8.9. Arvisol®
9. Quality for Health and Nutrition: Cannabis as Food and Feed
10. Current and Future Trends for Cannabis/Hemp Products
11. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hazekamp, A.; Bastola, K.; Rashidi, H.; Bender, J.; Verpoorte, R. Cannabis tea revisited: A systematic evaluation of the cannabinoid composition of cannabis tea. J. Ethnopharmacol. 2007, 113, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, P.; Okigbo, R. Effects of plants and medicinal plant combinations as anti-infectives. Afr. J. Pharm. Pharmacol. 2008, 2, 130–135. [Google Scholar]
- Vines, G. Herbal Harvests with a Future: Towards Sustainable Sources for Medicinal Plants; Plantlife International: Salisbury, UK, 2004. [Google Scholar]
- Russo, E. History of cannabis as a medicine. In Medicinal Uses of Cannabis and Cannabinoids; Pharmaceutical Press: London, UK, 2004; pp. 1–16. [Google Scholar]
- Jiang, H.-E.; Li, X.; Zhao, Y.-X.; Ferguson, D.K.; Hueber, F.; Bera, S.; Wang, Y.-F.; Zhao, L.-C.; Liu, C.; Li, C.-S. A new insight into Cannabis sativa (Cannabaceae) utilization from 2500-year-old Yanghai Tombs, Xinjiang, China. J. Ethnopharmacol. 2006, 108, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Soorni, A.; Fatahi, R.; Haak, D.C.; Salami, S.A.; Bombarely, A. Assessment of Genetic Diversity and Population Structure in Iranian Cannabis Germplasm. Sci. Rep. 2017, 7, 15668. [Google Scholar] [CrossRef]
- Li, H.-L. An archaeological and historical account of cannabis in China. Econ. Bot. 1973, 28, 437–448. [Google Scholar] [CrossRef]
- Touw, M. The religious and medicinal uses of Cannabis in China, India and Tibet. J. Psychoact. Drugs 1981, 13, 23–34. [Google Scholar] [CrossRef]
- Oomah, B.; Busson, M.; Godfrey, D.V.; Drover, J.C.G. Characteristics of hemp (Cannabis sativa L.) seed oil. Food Chem. 2002, 76, 33–43. [Google Scholar] [CrossRef]
- Anwar, F.; Latif, S.; Ashraf, M. Analytical characterization of hemp (Cannabis sativa) seed oil from different agro-ecological zones of Pakistan. J. Am. Oil Chem. Soc. 2006, 83, 323–329. [Google Scholar] [CrossRef]
- Cohen, K.; Weinstein, A.M. The Effects of Cannabinoids on Executive Functions: Evidence from Cannabis and Synthetic Cannabinoids—A Systematic Review. Brain Sci. 2018, 8, 40. [Google Scholar] [CrossRef]
- Van Bakel, H.; Stout, J.; Cote, A.G.; Tallon, C.M.; Sharpe, A.G.; Hughes, T.R.; Page, J.E. The draft genome and transcriptome of Cannabis sativa. Genome Boil. 2011, 12, R102. [Google Scholar] [CrossRef] [PubMed]
- Devinsky, O.; Cilio, M.R.; Cross, H.; Fernandez-Ruiz, J.; French, J.A.; Hill, C.; Katz, R.; Di Marzo, V.; Jutras-Aswad, D.; Notcutt, W.G.; et al. Cannabidiol: Pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders. Epilepsia 2014, 55, 791–802. [Google Scholar] [CrossRef]
- Russo, E.B. Cannabis Therapeutics and the Future of Neurology. Front. Integr. Neurosci. 2018, 12, 51. [Google Scholar] [CrossRef]
- Ellis, R.J.; Toperoff, W.; Vaida, F.; Brande, G.V.D.; Gonzales, J.; Gouaux, B.; Bentley, H.; Atkinson, J.H. Smoked Medicinal Cannabis for Neuropathic Pain in HIV: A Randomized, Crossover Clinical Trial. Neuropsychopharmacology 2008, 34, 672–680. [Google Scholar] [CrossRef] [PubMed]
- Ranalli, P.; Di Candilo, M.; Mandolino, G.; Grassi, G.; Carboni, A. Hemp for sustainable agricultural systems. Agro Food Industry Hi Tech 1999, 10, 33–38. [Google Scholar]
- Pane, A.; Cosentino, S.L.; Copani, V.; Cacciola, S.O. First Report of Southern Blight Caused by Sclerotium rolfsii on Hemp (Cannabis sativa) in Sicily and Southern Italy. Plant Dis. 2007, 91, 636. [Google Scholar] [CrossRef]
- Orhan, I.; Sener, B. Fatty acid content of selected seed oils. J. Herb. Pharmacother. 2002, 2, 29–33. [Google Scholar] [CrossRef] [PubMed]
- Callaway, J.; Pate, D.W. Hempseed Oil. In Gourmet and Health-Promoting Specialty Oils; Moreau, R., Kamal-Eldin, A., Eds.; Academic Press and AOCS Press: Urbana, IL, USA, 2009; pp. 185–213. [Google Scholar]
- Rubin, G. Tattoo Ink Containing Cannabis or Hemp Derived Cannabinoids or Mixture of Both. U.S. Patent 2020/0069567 A1, 5 March 2020. [Google Scholar]
- Flores-Sanchez, I.J.; Verpoorte, R. Secondary metabolism in cannabis. Phytochem. Rev. 2008, 7, 615–639. [Google Scholar] [CrossRef]
- Grant, S.; Houben, A.; Vyskot, B.; Siroky, J.; Pan, W.-H.; Macas, J.; Saedler, H. Genetics of sex determination in flowering plants. Dev. Genet. 1994, 15, 214–230. [Google Scholar] [CrossRef]
- McPartland, J.M.; Clarke, R.C.; Watson, D.P. Hemp Diseases and Pests: Management and Biological Control—An Advanced Treatise; CABI: Wallingford, UK, 2000. [Google Scholar] [CrossRef]
- Solymosi, K.; Köfalvi, A.; Köfalvi, K.S.A.A. Cannabis: A Treasure Trove or Pandora’s Box? Mini Rev. Med. Chem. 2017, 17, 1223–1291. [Google Scholar] [CrossRef]
- Andre, C.M.; Hausman, J.-F.; Guerriero, G. Cannabis sativa: The Plant of the Thousand and One Molecules. Front. Plant Sci. 2016, 7, 19. [Google Scholar] [CrossRef]
- Scheau, C.; Badarau, I.A.; Mihai, G.L.; Scheau, A.-E.; Costache, D.O.; Constantin, C.; Calina, D.; Caruntu, C.; Costache, R.S.; Caruntu, A. Cannabinoids in the Pathophysiology of Skin Inflammation. Molecules 2020, 25, 652. [Google Scholar] [CrossRef] [PubMed]
- ElSohly, M.A.; Radwan, M.M.; Gul, W.; Chandra, S.; Galal, A. Phytochemistry of Cannabis sativa L. In The Chemistry of Mycotoxins; Springer Science and Business Media LLC: Cham, Switzerland, 2017; Volume 103, pp. 1–36. [Google Scholar]
- Ross, S.A.; ElSohly, M.A. The Volatile Oil Composition of Fresh and Air-Dried Buds of Cannabis sativa. J. Nat. Prod. 1996, 59, 49–51. [Google Scholar] [CrossRef] [PubMed]
- Keller, A.; Leupin, M.; Mediavilla, V.; Wintermantel, E. Influence of the growth stage of industrial hemp on chemical and physical properties of the fibres. Ind. Crop. Prod. 2001, 13, 35–48. [Google Scholar] [CrossRef]
- Pate, D.W. The Phytochemistry of Cannabis: Its Ecological and Evolutionary Implications. In Advance in Hemp Research; Food Product Press: New York, NY, USA, 1999; pp. 21–42. [Google Scholar]
- Jalali, S.; Salami, S.A.; Sharifi, M.; Sohrabi, S. Signaling compounds elicit expression of key genes in cannabinoid pathway and related metabolites in cannabis. Ind. Crop. Prod. 2019, 133, 105–110. [Google Scholar] [CrossRef]
- Ferguson, L.R. Role of plant polyphenols in genomic stability. Mutat. Res. Mol. Mech. Mutagen. 2001, 475, 89–111. [Google Scholar] [CrossRef]
- Clark, M.N.; Bohm, B.A. Flavonoid variation in Cannabis L. Bot. J. Linn. Soc. 1979, 79, 249–257. [Google Scholar] [CrossRef]
- Vanhoenacker, G.; Van Rompaey, P.; De Keukeleire, D.; Sandra, P. Chemotaxonomic features associated with flavonoids of cannabinoid-free cannabis (Cannabis sativa subsp. sativa L.) in relation to hops (Humulus lupulus L.). Nat. Prod. Lett. 2002, 16, 57–63. [Google Scholar] [CrossRef]
- Ross, S.A.; ElSohly, M.A.; Sultana, G.N.N.; Mehmedic, Z.; Hossain, C.F.; Chandra, S. Flavonoid glycosides and cannabinoids from the pollen of Cannabis sativa L. Phytochem. Anal. 2005, 16, 45–48. [Google Scholar] [CrossRef]
- Chiron, H.; Drouet, A.; Lieutier, F.; Payer, H.-D.; Ernst, D.; Sandermann, H. Gene Induction of Stilbene Biosynthesis in Scots Pine in Response to Ozone Treatment, Wounding, and Fungal Infection. Plant Physiol. 2000, 124, 865–872. [Google Scholar] [CrossRef]
- Jeandet, P.; Douillet-Breuil, A.-C.; Bessis, R.; Debord, S.; Sbaghi, M.; Adrian, M. Phytoalexins from the Vitaceae: Biosynthesis, Phytoalexin Gene Expression in Transgenic Plants, Antifungal Activity, and Metabolism. J. Agric. Food Chem. 2002, 50, 2731–2741. [Google Scholar] [CrossRef]
- Gorham, J. The stilbenoids. Prog. Phytochem. 1980, 6, 203–252. [Google Scholar]
- Hillis, W.; Inoue, T. The formation of polyphenols in trees—IV The polyphenols formed in Pinus radiata after Sirex attack. Phytochemistry 1968, 7, 13–22. [Google Scholar] [CrossRef]
- Stivala, L.A.; Savio, M.; Carafoli, F.; Perucca, P.; Bianchi, L.; Maga, G.; Forti, L.; Pagnoni, U.M.; Albini, A.; Prosperi, E.; et al. Specific Structural Determinants Are Responsible for the Antioxidant Activity and the Cell Cycle Effects of Resveratrol. J. Boil. Chem. 2001, 276, 22586–22594. [Google Scholar] [CrossRef] [PubMed]
- Vastano, B.C.; Chen, Y.; Zhu, N.; Ho, C.-T.; Zhou, Z.; Rosen, R.T. Isolation and Identification of Stilbenes in Two Varieties ofPolygonumcuspidatum. J. Agric. Food Chem. 2000, 48, 253–256. [Google Scholar] [CrossRef] [PubMed]
- Kostecki, K.; Engelmeier, D.; Pacher, T.; Hofer, O.; Vajrodaya, S.; Greger, H. Dihydrophenanthrenes and other antifungal stilbenoids from Stemona cf. Pierrei. Phytochemistry 2004, 65, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.; ElSohly, M. Constituents of Cannabis sativa L. XXVIII—A review of the natural constituents: 1980–1994. Zagazig J. Pharm. Sci. 1995, 4, 1–10. [Google Scholar]
- Guo, T.; Liu, Q.; Hou, P.; Li, F.; Guo, S.; Song, W.; Zhang, H.; Liu, X.; Zhang, S.; Zhang, J.; et al. Stilbenoids and cannabinoids from the leaves of Cannabis sativa f. sativa with potential reverse cholesterol transport activity. Food Funct. 2018, 9, 6608–6617. [Google Scholar] [CrossRef]
- Crombie, L.; Tuchinda, P.; Powell, M.J. Total synthesis of the spirans of Cannabis: Cannabispiradienone, cannabispirenone-A and-B, cannabispirone, α-and β-cannabispiranols and the dihydrophenanthrene cannithrene-1. J. Chem. Soc. 1982, 1477–1484. [Google Scholar] [CrossRef]
- Recueil des Travaux Chimiques des Pays-Bas. 2018, Volume 97, pp. 221–222, ISSN 0165-0513. Available online: https://en.wikipedia.org/wiki/Recueil_des_Travaux_Chimiques_des_Pays-Bas (accessed on 24 July 2020).
- El-Feraly, F.S.; El-Sherei, M.M.; Al-Muhtadi, F.J. Spiro-indans from Cannabis sativa. Phytochemistry 1986, 25, 1992–1994. [Google Scholar] [CrossRef]
- Sakakibara, I.; Ikeya, Y.; Hayashi, K.; Okada, M.; Maruno, M. Three acyclic bis-phenylpropane lignanamides from fruits of Cannabis sativa. Phytochemistry 1995, 38, 1003–1007. [Google Scholar] [CrossRef]
- Heblinski, M.; Santiago, M.; Fletcher, C.; Stuart, J.; Connor, M.; McGregor, I.S.; Arnold, J.C. Terpenoids Commonly Found in Cannabis sativa Do Not Modulate the Actions of Phytocannabinoids or Endocannabinoids on TRPA1 and TRPV1 Channels. Cannabis Cannabinoid Res. 2020. [Google Scholar] [CrossRef]
- ElSohly, M.A.; Slade, D. Chemical constituents of marijuana: The complex mixture of natural cannabinoids. Life Sci. 2005, 78, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Slatkin, D.J.; Doorenbos, N.J.; Harris, L.S.; Masoud, A.N.; Quimby, M.W.; Schiff, P.L. Chemical Constituents of Cannabis sativa L. Root. J. Pharm. Sci. 1971, 60, 1891–1892. [Google Scholar] [CrossRef]
- Hendriks, H.; Malingre, T.M.; Batterman, S.; Bos, R. The essential oil of Cannabis sativa L. Pharm. Weekbl. 1978, 113, 413–424. [Google Scholar]
- Malingré, T.; Hendriks, H.; Batterman, S.; Bos, R.; Visser, J. The Essential Oil of Cannabis sativa. Planta Med. 1975, 28, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Eguchi, R.; Ono, N.; Morita, A.H.; Katsuragi, T.; Nakamura, S.; Huang, M.; Amin, A.-U.-A.; Kanaya, S. Classification of alkaloids according to the starting substances of their biosynthetic pathways using graph convolutional neural networks. BMC Bioinform. 2019, 20, 380. [Google Scholar] [CrossRef]
- Yang, L.; Stöckigt, J. Trends for diverse production strategies of plant medicinal alkaloids. Nat. Prod. Rep. 2010, 27, 1469. [Google Scholar] [CrossRef]
- Waller, G.R.; Nowacki, E.K. The Role of Alkaloids in Plants. In Alkaloid Biology and Metabolism in Plants; Springer Science and Business Media LLC: Boston, MA, USA, 1978; pp. 143–181. [Google Scholar]
- Zheng, X.-Q.; Nagai, C.; Ashihara, H. Pyridine nucleotide cycle and trigonelline (N-methylnicotinic acid) synthesis in developing leaves and fruits of Coffea arabica. Physiol. Plant. 2004, 122, 404–411. [Google Scholar] [CrossRef]
- Rhodes, D.; Hanson, A. Quaternary ammonium and tertiary sulfonium compounds in higher plants. Annu. Rev. Plant Biol. 1993, 44, 357–384. [Google Scholar] [CrossRef]
- Paris, M.; Boucher, F.; Cosson, L. The constituents of Cannabis sativa pollen. Econ. Bot. 1975, 29, 245–253. [Google Scholar] [CrossRef]
- El-Feraly, F.S.; Turner, C.E. Alkaloids of Cannabis sativa leaves. Phytochemistry 1975, 14, 2304. [Google Scholar] [CrossRef]
- ElSohly, M.A.; Turner, C.E.; Phoebe, C.H.; Knapp, J.E.; Schiff, P.L.; Slatkin, D.J. Anhydrocannabisativine, a New Alkaloid from Cannabis sativa L. J. Pharm. Sci. 1978, 67, 124. [Google Scholar] [CrossRef] [PubMed]
- Wahby, I.; Arráez-Román, D.; Segura-Carretero, A.; Ligero, F.; Caba, J.M.; Fernández-Gutiérrez, A. Analysis of choline and atropine in hairy root cultures of Cannabis sativa L. by capillary electrophoresis-electrospray mass spectrometry. Electrophoresis 2006, 27, 2208–2215. [Google Scholar] [CrossRef] [PubMed]
- Kutchan, T.M. Alkaloid Biosynthesis[mdash]The Basis for Metabolic Engineering of Medicinal Plants. Plant Cell 1995, 7, 1059–1070. [Google Scholar] [CrossRef] [PubMed]
- Fischedick, J.; Van Der Kooy, F.; Verpoorte, R. Cannabinoid receptor 1 binding activity and quantitative analysis of Cannabis sativa L. smoke and vapor. Chem. Pharm. Bull. 2010, 58, 201–207. [Google Scholar] [CrossRef]
- Lafaye, G.; Karila, L.; Blecha, L.; Benyamina, A. Cannabis, cannabinoids, and health. Dialog Clin. Neurosci. 2007, 19, 309–316. [Google Scholar]
- Sutipatanasomboon, A.; Panvisavas, N. Discrimination of ‘fiber-type’ and ‘drug-type’ Cannabis sativa L. by fluorescent duplex PCR. Forensic Sci. Int. Genet. Suppl. Ser. 2011, 3, e522–e523. [Google Scholar] [CrossRef]
- Niesink, R.J.; van Laar, M.W. Does cannabidiol protect against adverse psychological effects of THC? Front. Psychiatry 2013, 4, 130. [Google Scholar] [CrossRef]
- Borna, T.; Salami, S.A.; Shokrpour, M. High resolution melting curve analysis revealed SNPs in major cannabinoid genes associated with drug and non-drug types of cannabis. Biotechnol. Biotechnol. Equip. 2017, 31, 1–7. [Google Scholar] [CrossRef]
- Small, E.; Beckstead, H.D. Common cannabinoid phenotypes in 350 stocks of Cannabis. Lloydia 1973, 36, 144–165. [Google Scholar]
- Fournier, G.; Richez-Dumanois, C.; Duvezin, J.; Mathieu, J.-P.; Paris, M. Identification of a New Chemotype in Cannabis sativa: Cannabigerol—Dominant Plants, Biogenetic and Agronomic Prospects. Planta Med. 1987, 53, 277–280. [Google Scholar] [CrossRef] [PubMed]
- De Meijer, E.P.M.; Bagatta, M.; Carboni, A.; Crucitti, P.; Moliterni, V.M.C.; Ranalli, P.; Mandolino, G. The inheritance of chemical phenotype in Cannabis sativa L. Genetics 2003, 163, 335–346. [Google Scholar] [PubMed]
- Mandolino, G.; Carboni, A. Potential of marker-assisted selection in hemp genetic improvement. Euphytica 2004, 140, 107–120. [Google Scholar] [CrossRef]
- Pacifico, D.; Miselli, F.; Micheler, M.; Carboni, A.; Ranalli, P.; Mandolino, G. Genetics and Marker-assisted Selection of the Chemotype in Cannabis sativa L. Mol. Breed. 2006, 17, 257–268. [Google Scholar] [CrossRef]
- Guo, T.-T.; Zhang, J.-C.; Zhang, H.; Liu, Q.; Zhao, Y.; Hou, Y.-F.; Bai, L.; Zhang, L.; Liu, X.-Q.; Zhang, S.-Y.; et al. Bioactive spirans and other constituents from the leaves of Cannabis sativa f. sativa. J. Asian Nat. Prod. Res. 2016, 19, 793–802. [Google Scholar] [CrossRef]
- Appendino, G.B.; Chianese, G.; Taglialatela-Scafati, O. Cannabinoids: Occurrence and Medicinal Chemistry. Curr. Med. Chem. 2011, 18, 1085–1099. [Google Scholar] [CrossRef]
- Morales, P.; Hurst, D.; Reggio, P.H. Molecular Targets of the Phytocannabinoids: A Complex Picture. Prog. Chem. Org. Nat. Prod. 2017, 103, 103–131. [Google Scholar] [CrossRef]
- Izzo, A.A.; Borrelli, F.; Capasso, R.; Di Marzo, V.; Mechoulam, R. Non-psychotropic plant cannabinoids: New therapeutic opportunities from an ancient herb. Trends Pharm. Sci. 2009, 30, 515–527. [Google Scholar] [CrossRef]
- Hill, A.J.; Williams, C.M.; Whalley, B.J.; Stephens, G.J. Phytocannabinoids as novel therapeutic agents in CNS disorders. Pharmacol. Ther. 2012, 133, 79–97. [Google Scholar] [CrossRef]
- Wang, M.; Wang, Y.-H.; Avula, B.; Radwan, M.M.; Wanas, A.S.; Van Antwerp, J.; Parcher, J.F.; ElSohly, M.A.; Khan, I.A. Decarboxylation Study of Acidic Cannabinoids: A Novel Approach Using Ultra-High-Performance Supercritical Fluid Chromatography/Photodiode Array-Mass Spectrometry. Cannabis Cannabinoid Res. 2016, 1, 262–271. [Google Scholar] [CrossRef]
- Hill, T.D.M.; Cascio, M.-G.; Romano, B.; Duncan, M.; Pertwee, R.G.; Williams, C.M.; Whalley, B.J.; Hill, A.J. Cannabidivarin-rich cannabis extracts are anticonvulsant in mouse and rat via a CB1 receptor-independent mechanism. Br. J. Pharm. 2013, 170, 679–692. [Google Scholar] [CrossRef] [PubMed]
- Pertwee, R.G.; Howlett, A.C.; Abood, M.E.; Alexander, S.P.; Di Marzo, V.; Elphick, M.R.; Greasley, P.J.; Hansen, H.S.; Kunos, G.; Mackie, K.; et al. International Union of Basic and Clinical Pharmacology. LXXIX. Cannabinoid Receptors and Their Ligands: Beyond CB1and CB2. Pharm. Rev. 2010, 62, 588–631. [Google Scholar] [CrossRef] [PubMed]
- Giuffrida, A.; Parsons, L.; Kerr, T.; De Fonseca, F.R.; Navarro, M.; Piomelli, D. Dopamine activation of endogenous cannabinoid signaling in dorsal striatum. Nat. Neurosci. 1999, 2, 358–363. [Google Scholar] [CrossRef] [PubMed]
- Velasco, G.; Galve-Roperh, I.; Sanchez, C.; Blázquez, C.; Haro, A.; Guzmán, M. Cannabinoids and ceramide: Two lipids acting hand-by-hand. Life Sci. 2005, 77, 1723–1731. [Google Scholar] [CrossRef] [PubMed]
- Di Marzo, V.; Bisogno, T.; De Petrocellis, L. Endocannabinoids and Related Compounds: Walking Back and Forth between Plant Natural Products and Animal Physiology. Chem. Boil. 2007, 14, 741–756. [Google Scholar] [CrossRef] [PubMed]
- Horvath, B.; Mukhopadhyay, P.; Haskó, G.; Pacher, P. The Endocannabinoid System and Plant-Derived Cannabinoids in Diabetes and Diabetic Complications. Am. J. Pathol. 2012, 180, 432–442. [Google Scholar] [CrossRef]
- Sledzinski, P.; Zeyland, J.; Słomski, R.; Nowak, A. The current state and future perspectives of cannabinoids in cancer biology. Cancer Med. 2018, 7, 765–775. [Google Scholar] [CrossRef]
- DeVane, W.; Hanus, L.; Breuer, A.; Pertwee, R.G.; Stevenson, L.; Griffin, G.; Gibson, D.; Mandelbaum, A.; Etinger, A.; Mechoulam, R. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 1992, 258, 1946–1949. [Google Scholar] [CrossRef]
- Pacher, P.; Bátkai, S.; Kunos, G. The Endocannabinoid System as an Emerging Target of Pharmacotherapy. Pharmacol. Rev. 2006, 58, 389–462. [Google Scholar] [CrossRef]
- Pertwee, R.G. (Ed.) Cannabinoids. In Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2005; Volume 168. [Google Scholar] [CrossRef]
- Straiker, A.; Mackie, K. Depolarization-induced suppression of excitation in murine autaptic hippocampal neurones. J. Physiol. 2005, 569, 501–517. [Google Scholar] [CrossRef]
- Wu, D.-F.; Yang, L.-Q.; Goschke, A.; Stumm, R.; Brandenburg, L.-O.; Liang, Y.-J.; Höllt, V.; Koch, T. Role of receptor internalization in the agonist-induced desensitization of cannabinoid type 1 receptors. J. Neurochem. 2008, 104, 1132–1143. [Google Scholar] [CrossRef] [PubMed]
- Herkenham, M.; Lynn, A.; Johnson, M.R.; Melvin, L.; De Costa, B.; Rice, K. Characterization and localization of cannabinoid receptors in rat brain: A quantitative in vitro autoradiographic study. J. Neurosci. 1991, 11, 563–583. [Google Scholar] [CrossRef] [PubMed]
- Marsicano, G.; Kuner, R. Anatomical Distribution of Receptors, Ligands and Enzymes in the Brain and in the Spinal Cord: Circuitries and Neurochemistry. In Cannabinoids and the Brain; Springer Science and Business Media LLC: New York, NY, USA, 2008; pp. 161–201. [Google Scholar]
- Kendall, D.A.; Yudowski, G.A. Cannabinoid Receptors in the Central Nervous System: Their Signaling and Roles in Disease. Front. Cell. Neurosci. 2017, 10, 294. [Google Scholar] [CrossRef] [PubMed]
- Condie, R.; Herring, A.; Koh, W.S.; Lee, M.; Kaminski, N.E. Cannabinoid Inhibition of Adenylate Cyclase-mediated Signal Transduction and Interleukin 2 (IL-2) Expression in the Murine T-cell Line, EL4.IL-2. J. Boil. Chem. 1996, 271, 13175–13183. [Google Scholar] [CrossRef]
- Klein, T.W. Cannabinoid-based drugs as anti-inflammatory therapeutics. Nat. Rev. Immunol. 2005, 5, 400–411. [Google Scholar] [CrossRef]
- Galiègue, S.; Mary, S.; Marchand, J.; Dussossoy, D.; Carrière, D.; Carayon, P.; Bouaboula, M.; Shire, D.; Fur, G.; Casellas, P. Expression of Central and Peripheral Cannabinoid Receptors in Human Immune Tissues and Leukocyte Subpopulations. JBIC J. Boil. Inorg. Chem. 1995, 232, 54–61. [Google Scholar] [CrossRef]
- Munro, S.P.; Thomas, K.L.; Abu-Shaar, M. Molecular characterization of a peripheral receptor for cannabinoids. Nature 1993, 365, 61–65. [Google Scholar] [CrossRef]
- André, C.; Vercruysse, A. Histochemical Study of the Stalked Glandular Hairs of The Female Cannabis Plants, Using Fast Blue Salt. Planta Med. 1976, 29, 361–366. [Google Scholar] [CrossRef]
- Petri, G.; Oroszlán, P.; Fridvalszky, L. Histochemical detection of hemp trichomes and their correlation with the THC content. Acta Boil. Hung. 1988, 39, 59–73. [Google Scholar]
- Lanyon, V.S.; Turner, J.C.; Mahlberg, P.G. Quantitative Analysis of Cannabinoids in the Secretory Product from Capitate-Stalked Glands of Cannabis sativa L. (Cannabaceae). Int. J. Plant Sci. 1981, 142, 316–319. [Google Scholar] [CrossRef]
- Kim, E.-S.; Mahlberg, P.G. Immunochemical localization of Tetrahydrocannabinol (THC) in Cryofixed Glandular Trichomes of Cannabis (Cannabaceae). Am. J. Bot. 1997, 84, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.A.; Mehmedic, Z.; Murphy, T.P.; ElSohly, M.A. GC-MS Analysis of the Total 9-THC Content of Both Drug- and Fiber-Type Cannabis Seeds. J. Anal. Toxicol. 2000, 24, 715–717. [Google Scholar] [CrossRef] [PubMed]
- Petrović, M.; Debeljak, Ž.; Kezić, N.; Džidara, P. Relationship between cannabinoids content and composition of fatty acids in hempseed oils. Food Chem. 2015, 170, 218–225. [Google Scholar] [CrossRef]
- Potter, D. Growth and morphology of medicinal cannabis. In Medicinal Uses of Cannabis and Cannabinoids; Pharmaceutical Press: London, UK, 2004; pp. 17–54. [Google Scholar]
- Stout, J.; Boubakir, Z.; Ambrose, S.J.; Purves, R.W.; Page, J.E. The hexanoyl-CoA precursor for cannabinoid biosynthesis is formed by an acyl-activating enzyme in Cannabis sativa trichomes. Plant J. 2012, 71, 353–365. [Google Scholar] [CrossRef]
- Tanaka, H.; Shoyama, Y. Monoclonal antibody against tetrahydrocannabinolic acid distinguishes Cannabis sativa samples from different plant species. Forensic Sci. Int. 1999, 106, 135–146. [Google Scholar] [CrossRef]
- Fischedick, J.T.; Hazekamp, A.; Erkelens, T.; Choi, Y.H.; Verpoorte, R.; Verpoorte, R. Metabolic fingerprinting of Cannabis sativa L., cannabinoids and terpenoids for chemotaxonomic and drug standardization purposes. Phytochemistry 2010, 71, 2058–2073. [Google Scholar] [CrossRef]
- Shoyama, Y.; Yagi, M.; Nishioka, I.; Yamauchi, T. Biosynthesis of cannabinoid acids. Phytochemistry 1975, 14, 2189–2192. [Google Scholar] [CrossRef]
- Fellermeier, M.; Eisenreich, W.; Bacher, A.; Zenk, M.H. Biosynthesis of cannabinoids. Incorporation experiments with (13)C-labeled glucoses. JBIC J. Boil. Inorg. Chem. 2001, 268, 1596–1604. [Google Scholar] [CrossRef]
- Gagne, S.J.; Stout, J.; Liu, E.; Boubakir, Z.; Clark, S.M.; Page, J.E. Identification of olivetolic acid cyclase from Cannabis sativa reveals a unique catalytic route to plant polyketides. Proc. Natl. Acad. Sci. USA 2012, 109, 12811–12816. [Google Scholar] [CrossRef]
- Page, J.; Boubakir, Z. Aromatic Prenyltransferase from Cannabis. PCT Patent Application WO/2011/017798, 17 February 2011. [Google Scholar]
- Sirikantaramas, S.; Morimoto, S.; Shoyama, Y.; Ishikawa, Y.; Wada, Y.; Shoyama, Y.; Taura, F. The gene controlling marijuana psychoactivity molecular cloning and heterologous expression of Δ1-tetrahydrocannabinolic acid synthase from Cannabis sativa L. J. Biol. Chem. 2004, 279, 39767–39774. [Google Scholar] [CrossRef]
- Taura, F.; Dono, E.; Sirikantaramas, S.; Yoshimura, K.; Shoyama, Y.; Morimoto, S. Production of Δ1-tetrahydrocannabinolic acid by the biosynthetic enzyme secreted from transgenic Pichia pastoris. Biochem. Biophys. Res. Commun. 2007, 361, 675–680. [Google Scholar] [CrossRef] [PubMed]
- Mechoulam, R.; Gaoni, Y. A Total Synthesis of dl-Δ1-Tetrahydrocannabinol, the Active Constituent of Hashish1. J. Am. Chem. Soc. 1965, 87, 3273–3275. [Google Scholar] [CrossRef] [PubMed]
- D’Souza, D.C.; Perry, E.; MacDougall, L.; Ammerman, Y.; Cooper, T.; Wu, Y.-T.; Braley, G.; Gueorguieva, R.V.; Krystal, J.H. The Psychotomimetic Effects of Intravenous Delta-9-Tetrahydrocannabinol in Healthy Individuals: Implications for Psychosis. Neuropsychopharmacology 2004, 29, 1558–1572. [Google Scholar] [CrossRef] [PubMed]
- Pertwee, R.G. Emerging strategies for exploiting cannabinoid receptor agonists as medicines. Br. J. Pharm. 2009, 156, 397–411. [Google Scholar] [CrossRef]
- Avraham, Y.; Latzer, Y.; Hasid, D.; Berry, E.M. The Impact of Δ9-THC on the Psychological Symptoms of Anorexia Nervosa: A Pilot Study. Isr. J. Psychiatry Relat. Sci. 2017, 54, 44–51. [Google Scholar]
- Kramer, J.L. Medical marijuana for cancer. Ca A Cancer J. Clin. 2014, 65, 109–122. [Google Scholar] [CrossRef]
- De La Ossa, D.H.P.; Gil-Alegre, M.E.; Ligresti, A.; Aberturas, M.D.R.; Molpeceres, J.; Torres-Suárez, A.I.; Di Marzo, V. Preparation and characterization of Δ9-tetrahydrocannabinol-loaded biodegradable polymeric microparticles and their antitumoral efficacy on cancer cell lines. J. Drug Target. 2013, 21, 710–718. [Google Scholar] [CrossRef]
- Colasanti, B.K.; Powell, S.R.; Craig, C.R. Intraocular pressure, ocular toxicity and neurotoxicity after administration of Δ9-Tetrahydrocannabinol or cannabichromene. Exp. Eye Res. 1984, 38, 63–71. [Google Scholar] [CrossRef]
- Rock, E.M.; Parker, L.A. Cannabinoids as Potential Treatment for Chemotherapy-Induced Nausea and Vomiting. Front. Pharm. 2016, 7, 221. [Google Scholar] [CrossRef]
- Salazar-Roa, M.; Carracedo, A.; Salanueva, I.J.; Hernandez-Tiedra, S.; Lorente, M.; Egia, A.; Vázquez, P.; Blázquez, C.; Torres, S.; Garcia, S.; et al. Cannabinoid action induces autophagy-mediated cell death through stimulation of ER stress in human glioma cells. J. Clin. Investig. 2009, 119, 1359–1372. [Google Scholar] [CrossRef]
- Nabissi, M.; Morelli, M.B.; Offidani, M.; Amantini, C.; Gentili, S.; Soriani, A.; Cardinali, C.; Leoni, P.; Santoni, G. Cannabinoids synergize with carfilzomib, reducing multiple myeloma cells viability and migration. Oncotarget 2016, 7, 77543–77557. [Google Scholar] [CrossRef] [PubMed]
- Blessing, E.; Steenkamp, M.M.; Manzanares, J.; Marmar, C.R. Cannabidiol as a Potential Treatment for Anxiety Disorders. Neurotherapeutics 2015, 12, 825–836. [Google Scholar] [CrossRef] [PubMed]
- Katona, I.; Freund, T.F. Endocannabinoid signaling as a synaptic circuit breaker in neurological disease. Nat. Med. 2008, 14, 923–930. [Google Scholar] [CrossRef] [PubMed]
- MacIntyre, J.; Dong, A.; Straiker, A.; Zhu, J.; Howlett, S.E.; Bagher, A.; Denovan-Wright, E.; Yu, D.-Y.; Kelly, M.E.M. Cannabinoid and lipid-mediated vasorelaxation in retinal microvasculature. Eur. J. Pharm. 2014, 735, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Joseph, J.; Niggemann, B.; Zaenker, K.S.; Entschladen, F. Anandamide is an endogenous inhibitor for the migration of tumor cells and T lymphocytes. Cancer Immunol. Immunother. 2004, 53, 723–728. [Google Scholar] [CrossRef]
- Javid, F.A.; Phillips, R.M.; Afshinjavid, S.; Verde, R.; Ligresti, A. Cannabinoid pharmacology in cancer research: A new hope for cancer patients? Eur. J. Pharm. 2016, 775, 1–14. [Google Scholar] [CrossRef]
- Velasco, G.; Sanchez, C.; Guzmán, M. Anticancer mechanisms of cannabinoids. Curr. Oncol. 2016, 23, S23–S32. [Google Scholar] [CrossRef]
- Paton, W.D.; Pertwee, R.G. The actions of cannabis in man. In Marijuana: Chemistry, Pharmacology, Metabolism and Clinical Effects; Academic Press: New York, NY, USA, 1973; pp. 287–333. [Google Scholar]
- Matsuda, L.A.; Lolait, S.J.; Brownstein, M.J.; Young, A.C.; Bonner, T.I. Structure of a cannabinoid receptor and functional expression of the cloned cDNA. Nature 1990, 346, 561–564. [Google Scholar] [CrossRef]
- Mechoulam, R.; Fride, E.; Di Marzo, V. Endocannabinoids. Eur. J. Pharm. 1998, 359, 1–18. [Google Scholar] [CrossRef]
- Ameri, A. The effects of cannabinoids on the brain. Prog. Neurobiol. 1999, 58, 315–348. [Google Scholar] [CrossRef]
- Mechoulam, R.; Ben-Shabat, S. From gan-zi-gun-nu to anandamide and 2-arachidonoylglycerol: The ongoing story of cannabis. Nat. Prod. Rep. 1999, 16, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Carchman, R.A.; Harris, L.S.; Munson, A.E. The inhibition of DNA synthesis by cannabinoids. Cancer Res. 1976, 36, 95–100. [Google Scholar] [PubMed]
- Massi, P.; Vaccani, A.; Ceruti, S.; Colombo, A.; Abbracchio, M.P.; Parolaro, D. Antitumor Effects of Cannabidiol, a Nonpsychoactive Cannabinoid, on Human Glioma Cell Lines. J. Pharm. Exp. 2003, 308, 838–845. [Google Scholar] [CrossRef] [PubMed]
- Eisohly, H.N.; Turner, C.E.; Clark, A.M.; Eisohly, M.A.; Elsohly, H.N.; Elsohly, M.A. Synthesis and Antimicrobial Activities of Certain Cannabichromene and Cannabigerol Related Compounds. J. Pharm. Sci. 1982, 71, 1319–1323. [Google Scholar] [CrossRef] [PubMed]
- Formukong, E.A.; Evans, A.T.; Evans, F.J. Analgesic and antiinflammatory activity of constituents of Cannabis sativa L. Inflammation 1988, 12, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Hampson, A.; Grimaldi, M.; Axelrod, J.; Wink, D. Cannabidiol and (−) Δ9-tetrahydrocannabinol are neuroprotective antioxidants. Proc. Natl. Acad. Sci. USA 1998, 95, 8268–8273. [Google Scholar] [CrossRef]
- Tamba, B.-I.; Stanciu, G.; Urîtu, C.M.; Rezuș, E.; Stefanescu, R.; Mihai, C.; Luca, A.; Rusu-Zota, G.; Leon-Constantin, M.M.; Cojocaru, E.; et al. Challenges and Opportunities in Preclinical Research of Synthetic Cannabinoids for Pain Therapy. Medicina 2020, 56, 24. [Google Scholar] [CrossRef]
- Perisetti, A.; Rimu, A.H.; Khan, S.A.; Bansal, P.; Goyal, H. Role of cannabis in inflammatory bowel diseases. Ann. Gastroenterol. 2020, 33, 134–144. [Google Scholar] [CrossRef]
- Grotenhermen, F.; Russo, E. Cannabis and Cannabinoids: Pharmacology, Toxicology, and Therapeutic Potential; Psychology Press: New York, NY, USA, 2002. [Google Scholar]
- Musty, R.E. Natural Cannabinoids: Interactions and Effects; Pharm Press: London, UK, 2004. [Google Scholar]
- Tomida, I.; Pertwee, R.G.; Azuara-Blanco, A. Cannabinoids and glaucoma. Br. J. Ophthalmol. 2004, 88, 708–713. [Google Scholar] [CrossRef]
- Eagleston, L.R.M.; Kalani, N.K.; Patel, R.R.; Flaten, H.K.; Dunnick, C.A.; Dellavalle, R.P. Cannabinoids in dermatology: A scoping review. Dermatol. Online J. 2018, 24, 1–17. [Google Scholar]
- Marks, D.H.; Friedman, A. The Therapeutic Potential of Cannabinoids in Dermatology. Skin Ther. Lett. 2018, 23, 1–5. [Google Scholar]
- Sheriff, T.; Lin, M.J.; Dubin, D.; Khorasani, H. The potential role of cannabinoids in dermatology. J. Derm. Treat. 2019, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.J.; Weston, S.E.; Jones, N.A.; Smith, I.; Bevan, S.A.; Williamson, E.M.; Stephens, G.J.; Williams, C.M.; Whalley, B.J. Δ9-Tetrahydrocannabivarin suppresses in vitro epileptiform and in vivo seizure activity in adult rats. Epilepsia 2010, 51, 1522–1532. [Google Scholar] [CrossRef]
- García, C.; Palomo-Garo, C.; García-Arencibia, M.; Ramos, J.; Pertwee, R.G.; Fernández-Ruiz, J. Symptom-relieving and neuroprotective effects of the phytocannabinoid Δ9-THCV in animal models of Parkinson’s disease. Br. J. Pharm. 2011, 163, 1495–1506. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.; Mercier, M.; Hill, T.; Glyn, S.; Jones, N.; Yamasaki, Y.; Futamura, T.; Duncan, M.; Stott, C.; Stephens, G.; et al. Cannabidivarin is anticonvulsant in mouse and rat. Br. J. Pharm. 2012, 167, 1629–1642. [Google Scholar] [CrossRef] [PubMed]
- Scorza, F.A.; Cysneiros, R.M.; Cavalheiro, E.A.; Arida, R.M.; De Albuquerque, M. Progress in neuro-psychopharmacology and biological psychiatry Re.: Omega-3 fatty acids and sudden unexpected death in epilepsy: What does the evidence tell us? Prog. Neuro Psychopharmacol. Boil. Psychiatry 2007, 31, 972–973. [Google Scholar] [CrossRef]
- Tubaro, A.; Giangaspero, A.; Sosa, S.; Negri, R.; Grassi, G.; Casano, S.; Della Loggia, R.; Appendino, G. Comparative topical anti-inflammatory activity of cannabinoids and cannabivarins. Fitoterapia 2010, 81, 816–819. [Google Scholar] [CrossRef]
- Bolognini, D.; Costa, B.; Maione, S.; Comelli, F.; Marini, P.; Di Marzo, V.; Parolaro, D.; Ross, R.A.; Gauson, L.A.; Cascio, M.G.; et al. The plant cannabinoid Δ9-tetrahydrocannabivarin can decrease signs of inflammation and inflammatory pain in mice. Br. J. Pharm. 2010, 160, 677–687. [Google Scholar] [CrossRef]
- Riedel, G.; Fadda, P.; McKillop-Smith, S.; Pertwee, R.G.; Platt, B.; Robinson, L. Synthetic and plant-derived cannabinoid receptor antagonists show hypophagic properties in fasted and non-fasted mice. Br. J. Pharm. 2009, 156, 1154–1166. [Google Scholar] [CrossRef]
- Booz, G.W. Cannabidiol as an emergent therapeutic strategy for lessening the impact of inflammation on oxidative stress. Free Radic. Boil. Med. 2011, 51, 1054–1061. [Google Scholar] [CrossRef]
- Deng, L.; Ng, L.; Ozawa, T.; Stella, N. Quantitative Analyses of Synergistic Responses between Cannabidiol and DNA-Damaging Agents on the Proliferation and Viability of Glioblastoma and Neural Progenitor Cells in Culture. J. Pharm. Exp. 2016, 360, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Fraguas-Sánchez, A.I.; Fernández-Carballido, A.; Torres-Suárez, A.I. Phyto-, endo- and synthetic cannabinoids: Promising chemotherapeutic agents in the treatment of breast and prostate carcinomas. Expert Opin. Investig. Drugs 2016, 25, 1311–1323. [Google Scholar] [CrossRef] [PubMed]
- Parker, L.A.; Rock, E.M.; Limebeer, C.L. Regulation of nausea and vomiting by cannabinoids. Br. J. Pharm. 2011, 163, 1411–1422. [Google Scholar] [CrossRef] [PubMed]
- Perucca, E. Cannabinoids in the Treatment of Epilepsy: Hard Evidence at Last? J. Epilepsy Res. 2017, 7, 61–76. [Google Scholar] [CrossRef]
- McGuire, P.; Robson, P.; Cubała, W.J.; Vasile, D.; Morrison, P.D.; Barron, R.; Taylor, A.; Wright, S. Cannabidiol (CBD) as an Adjunctive Therapy in Schizophrenia: A Multicenter Randomized Controlled Trial. Am. J. Psychiatry 2018, 175, 225–231. [Google Scholar] [CrossRef]
- Deiana, S. Medical use of cannabis. Cannabidiol: A new light for schizophrenia? Drug Test. Anal. 2012, 5, 46–51. [Google Scholar] [CrossRef]
- Fernández-Ruiz, J.; Sagredo, O.; Pazos, M.R.; García, C.; Pertwee, R.G.; Mechoulam, R.; Martínez-Orgado, J. Cannabidiol for neurodegenerative disorders: Important new clinical applications for this phytocannabinoid? Br. J. Clin. Pharm. 2012, 75, 323–333. [Google Scholar] [CrossRef]
- Rajesh, M.; Mukhopadhyay, P.; Bátkai, S.; Patel, V.; Saito, K.; Matsumoto, S.; Kashiwaya, Y.; Horvath, B.; Mukhopadhyay, B.; Becker, L.; et al. Cannabidiol Attenuates Cardiac Dysfunction, Oxidative Stress, Fibrosis, and Inflammatory and Cell Death Signaling Pathways in Diabetic Cardiomyopathy. J. Am. Coll. Cardiol. 2010, 56, 2115–2125. [Google Scholar] [CrossRef]
- Shinjyo, N.; Di Marzo, V. The effect of cannabichromene on adult neural stem/progenitor cells. Neurochem. Int. 2013, 63, 432–437. [Google Scholar] [CrossRef]
- Prenderville, J.A.; Kelly, Á.M.; Downer, E.J. The role of cannabinoids in adult neurogenesis. Br. J. Pharm. 2015, 172, 3950–3963. [Google Scholar] [CrossRef]
- Bouquié, R.; Deslandes, G.; Mazaré, H.; Cogné, M.; Mahé, J.; Grégoire, M.; Jolliet, P. Cannabis and anticancer drugs: Societal usage and expected pharmacological interactions—A review. Fundam. Clin. Pharm. 2018, 32, 462–484. [Google Scholar] [CrossRef] [PubMed]
- Van De Donk, T.; Niesters, M.; Kowal, M.A.; Olofsen, E.; Dahan, A.; Van Velzen, M. An experimental randomized study on the analgesic effects of pharmaceutical-grade cannabis in chronic pain patients with fibromyalgia. Pain 2019, 160, 860–869. [Google Scholar] [CrossRef] [PubMed]
- Hazekamp, A.; Fischedick, J.T. Cannabis—from cultivar to chemovar. Drug Test. Anal. 2012, 4, 660–667. [Google Scholar] [CrossRef] [PubMed]
- Krcevski-Skvarc, N.; Wells, C.; Häuser, W. Availability and approval of cannabis-based medicines for chronic pain management and palliative/supportive care in Europe: A survey of the status in the chapters of the European Pain Federation. Eur. J. Pain 2017, 22, 440–454. [Google Scholar] [CrossRef]
- Schwilke, E.W.; Schwope, D.M.; Karschner, E.L.; Lowe, R.H.; Darwin, W.D.; Kelly, D.L.; Goodwin, R.S.; Gorelick, D.A.; Huestis, M.A. Δ9-Tetrahydrocannabinol (THC), 11-Hydroxy-THC, and 11-Nor-9-carboxy-THC Plasma Pharmacokinetics during and after Continuous High-Dose Oral THC. Clin. Chem. 2009, 55, 2180–2189. [Google Scholar] [CrossRef]
- Klumpers, L.; Beumer, T.L.; Van Hasselt, J.G.C.; Lipplaa, A.; Karger, L.B.; Kleinloog, D.; Freijer, J.I.; De Kam, M.L.; Van Gerven, J.M.A. Novel Δ9-tetrahydrocannabinol formulation Namisol® has beneficial pharmacokinetics and promising pharmacodynamic effects. Br. J. Clin. Pharm. 2011, 74, 42–53. [Google Scholar] [CrossRef]
- Berlach, D.M.; Shir, Y.; Ware, M.A. Experience with the Synthetic Cannabinoid Nabilone in Chronic Noncancer Pain. Pain Med. 2006, 7, 25–29. [Google Scholar] [CrossRef]
- Perras, C. Sativex for the management of multiple sclerosis symptoms. Issues Emerg. Health Technol. 2005, 72, 1–4. [Google Scholar]
- Abu-Sawwa, R.; Stehling, C. Epidiolex (Cannabidiol) Primer: Frequently Asked Questions for Patients and Caregivers. J. Pediatr. Pharm. 2020, 25, 75–77. [Google Scholar] [CrossRef]
- Yeshurun, M.; Shpilberg, O.; Herscovici, C.; Shargian, L.; Dreyer, J.; Peck, A.; Israeli, M.; Levy-Assaraf, M.; Gruenewald, T.; Mechoulam, R.; et al. Cannabidiol for the Prevention of Graft-versus-Host-Disease after Allogeneic Hematopoietic Cell Transplantation: Results of a Phase II Study. Boil. Blood Marrow Transpl. 2015, 21, 1770–1775. [Google Scholar] [CrossRef]
- Haney, M.; Malcolm, R.J.; Babalonis, S.; Nuzzo, P.A.; Cooper, Z.D.; Bedi, G.; Gray, K.M.; McRae-Clark, A.; Lofwall, M.R.; Sparenborg, S.; et al. Oral Cannabidiol does not Alter the Subjective, Reinforcing or Cardiovascular Effects of Smoked Cannabis. Neuropsychopharmacology 2015, 41, 1974–1982. [Google Scholar] [CrossRef] [PubMed]
- Morgan, C.J.A.; Das, R.K.; Joye, A.; Curran, H.V.; Kamboj, S.K. Cannabidiol reduces cigarette consumption in tobacco smokers: Preliminary findings. Addict. Behav. 2013, 38, 2433–2436. [Google Scholar] [CrossRef] [PubMed]
- Arndt, D.L.; De Wit, H. Cannabidiol Does Not Dampen Responses to Emotional Stimuli in Healthy Adults. Cannabis Cannabinoid Res. 2017, 2, 105–113. [Google Scholar] [CrossRef]
- Atsmon, J.; Heffetz, D.; Deutsch, L.; Deutsch, F.; Sacks, H. Single-Dose Pharmacokinetics of Oral Cannabidiol Following Administration of PTL101: A New Formulation Based on Gelatin Matrix Pellets Technology. Clin. Pharm. Drug Dev. 2017, 7, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Cerino, P.; Buonerba, C.; Cannazza, G.; D’Auria, J.; Ottoni, E.; Fulgione, A.; Di Stasio, A.; Pierri, B.; Gallo, A. A Review of Hemp as Food and Nutritional Supplement. Cannabis Cannabinoid Res. 2020. [Google Scholar] [CrossRef]
- Bonn-Miller, M.O.; Loflin, M.J.; Thomas, B.F.; Marcu, J.P.; Hyke, T.; Vandrey, R. Labeling Accuracy of Cannabidiol Extracts Sold Online. JAMA 2017, 318, 1708–1709. [Google Scholar] [CrossRef]
- Farinon, B.; Molinari, R.; Costantini, L.; Merendino, N. The seed of industrial hemp (Cannabis sativa L.): Nutritional Quality and Potential Functionality for Human Health and Nutrition. Nutrients 2020, 12, 1935. [Google Scholar] [CrossRef]
- Callaway, J.C. Hempseed as a nutritional resource: An overview. Euphytica 2004, 140, 65–72. [Google Scholar] [CrossRef]
- Irakli, M.; Tsaliki, E.; Kalivas, A.; Kleisiaris, F.; Sarrou, E.; Cook, C.M. Effect οf Genotype and Growing Year on the Nutritional, Phytochemical, and Antioxidant Properties of Industrial Hemp (Cannabis sativa L.) Seeds. Antioxidants 2019, 8, 491. [Google Scholar] [CrossRef]
- Saini, R.K.; Keum, Y.-S. Omega-3 and omega-6 polyunsaturated fatty acids: Dietary sources, metabolism, and significance—A review. Life Sci. 2018, 203, 255–267. [Google Scholar] [CrossRef]
- Antonelli, M.; Benedetti, B.; Cannazza, G.; Cerrato, A.; Citti, C.; Montone, C.M.; Piovesana, S.; Laganà, A. New insights in hemp chemical composition: A comprehensive polar lipidome characterization by combining solid phase enrichment, high-resolution mass spectrometry, and cheminformatics. Anal. Bioanal. Chem. 2019, 412, 413–423. [Google Scholar] [CrossRef]
- Cerrato, A.; Cannazza, G.; Capriotti, A.L.; Citti, C.; La Barbera, G.; Laganà, A.; Montone, C.M.; Piovesana, S.; Cavaliere, C. A new software-assisted analytical workflow based on high-resolution mass spectrometry for the systematic study of phenolic compounds in complex matrices. Talanta 2020, 209, 120573. [Google Scholar] [CrossRef] [PubMed]
- Menezes, R.; Rodriguez-Mateos, A.; Kaltsatou, A.; González-Sarrías, A.; Greyling, A.; Giannaki, C.D.; Andres-Lacueva, C.; Milenkovic, D.; Gibney, E.R.; Dumont, J.; et al. Impact of Flavonols on Cardiometabolic Biomarkers: A Meta-Analysis of Randomized Controlled Human Trials to Explore the Role of Inter-Individual Variability. Nutrients 2017, 9, 117. [Google Scholar] [CrossRef]
- Stevanovic, Z.D.; Bošnjak-Neumüller, J.; Pajić-Lijaković, I.; Raj, J.; Vasiljević, M. Essential Oils as Feed Additives—Future Perspectives. Molecules 2018, 23, 1717. [Google Scholar] [CrossRef] [PubMed]
- Lakhani, N.; Lakhani, P. Plant secondary metabolites as a potential source to inhibit methane production and improve animal performance. Int. J. Chem. Stud. 2018, 6, 3375–3379. [Google Scholar]
- Steiner, T.; Syed, B. Phytogenic Feed Additives in Animal Nutrition. In Medicinal and Aromatic Plants of the World; Springer Science and Business Media LLC: Dordrecht, The Netherlands, 2015; Volume 1, pp. 403–423. [Google Scholar]
| Effects | THC | AEA | 2-AG | CBD | CBC | CBDV | CBG | THCV | References |
|---|---|---|---|---|---|---|---|---|---|
| Suppress Seizure | √ | [149] | |||||||
| Reduce Parkinson’s Disease symptoms | √ | [150] | |||||||
| Anti-inflammatory | √ | √ | √ | √ | √ | √ | √ | [127,128,151,152,153,154] | |
| Reduce weight | √ | [155] | |||||||
| Anticonvulsant | √ | [156] | |||||||
| Anxiolytic | √ | [119,125] | |||||||
| Effective against cancer | √ | √ | √ | √ | √ | [120,123,124,127,128,157,158] | |||
| Chemotherapy induced emesis | √ | √ | [117,122,159] | ||||||
| Effective against glaucoma | √ | √ | √ | [121,127,128] | |||||
| Epilepsy | √ | √ | [151,160] | ||||||
| Oxidative injury | √ | [156] | |||||||
| Schizophrenia | √ | √ | [161,162] | ||||||
| Neuro-degeneration | √ | [163] | |||||||
| Diabetic retinopathy | √ | [164] | |||||||
| Colitis | √ | [77] | |||||||
| Exhibits Neuron protection | √ | [165,166] | |||||||
| Antiemetic, Analgesic activity | √ | √ | [119] | ||||||
| Energy and fat metabolism | √ | √ | [127,128] | ||||||
| Insulin sensitivity | √ | √ | [127,128] | ||||||
| Anorexia | √ | [117,118] | |||||||
| Bowel disease | √ | [165] |
| Product | Possible Therapeutic Effects | Component | References |
|---|---|---|---|
| Bedrocan® |
HIV medication and anorexia
| (>22% Δ9-THC and <1% CBD) | [167,170] |
| Bedrobinol® |
| 13% Δ9-THC and 1% CBD | [167,170] |
| Marinol® |
| Dronabinol in doses 2.5, 5, and 10 mg | [167,171] |
| Namisol® |
| Purified Δ9-THC (>98%) | [172] |
| Cesamet™ |
| THC in the form of oral capsules which contain 1 mg (2.7 µmol) nabilone | [167,173] |
| Sativex® |
| Nabiximols contains 2.7 mg of Δ9-THC and 2.5 mg of CBD in a 1:1 ratio. Sativex® | [167,174] |
| Epidiolex® |
| Purified pharmaceutical grade cannabidiol (CBD), | [175] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salami, S.A.; Martinelli, F.; Giovino, A.; Bachari, A.; Arad, N.; Mantri, N. It Is Our Turn to Get Cannabis High: Put Cannabinoids in Food and Health Baskets. Molecules 2020, 25, 4036. https://doi.org/10.3390/molecules25184036
Salami SA, Martinelli F, Giovino A, Bachari A, Arad N, Mantri N. It Is Our Turn to Get Cannabis High: Put Cannabinoids in Food and Health Baskets. Molecules. 2020; 25(18):4036. https://doi.org/10.3390/molecules25184036
Chicago/Turabian StyleSalami, Seyed Alireza, Federico Martinelli, Antonio Giovino, Ava Bachari, Neda Arad, and Nitin Mantri. 2020. "It Is Our Turn to Get Cannabis High: Put Cannabinoids in Food and Health Baskets" Molecules 25, no. 18: 4036. https://doi.org/10.3390/molecules25184036
APA StyleSalami, S. A., Martinelli, F., Giovino, A., Bachari, A., Arad, N., & Mantri, N. (2020). It Is Our Turn to Get Cannabis High: Put Cannabinoids in Food and Health Baskets. Molecules, 25(18), 4036. https://doi.org/10.3390/molecules25184036








