Bioactivities of Lyngbyabellins from Cyanobacteria of Moorea and Okeania Genera
Abstract
1. Introduction
2. Results and Discussion
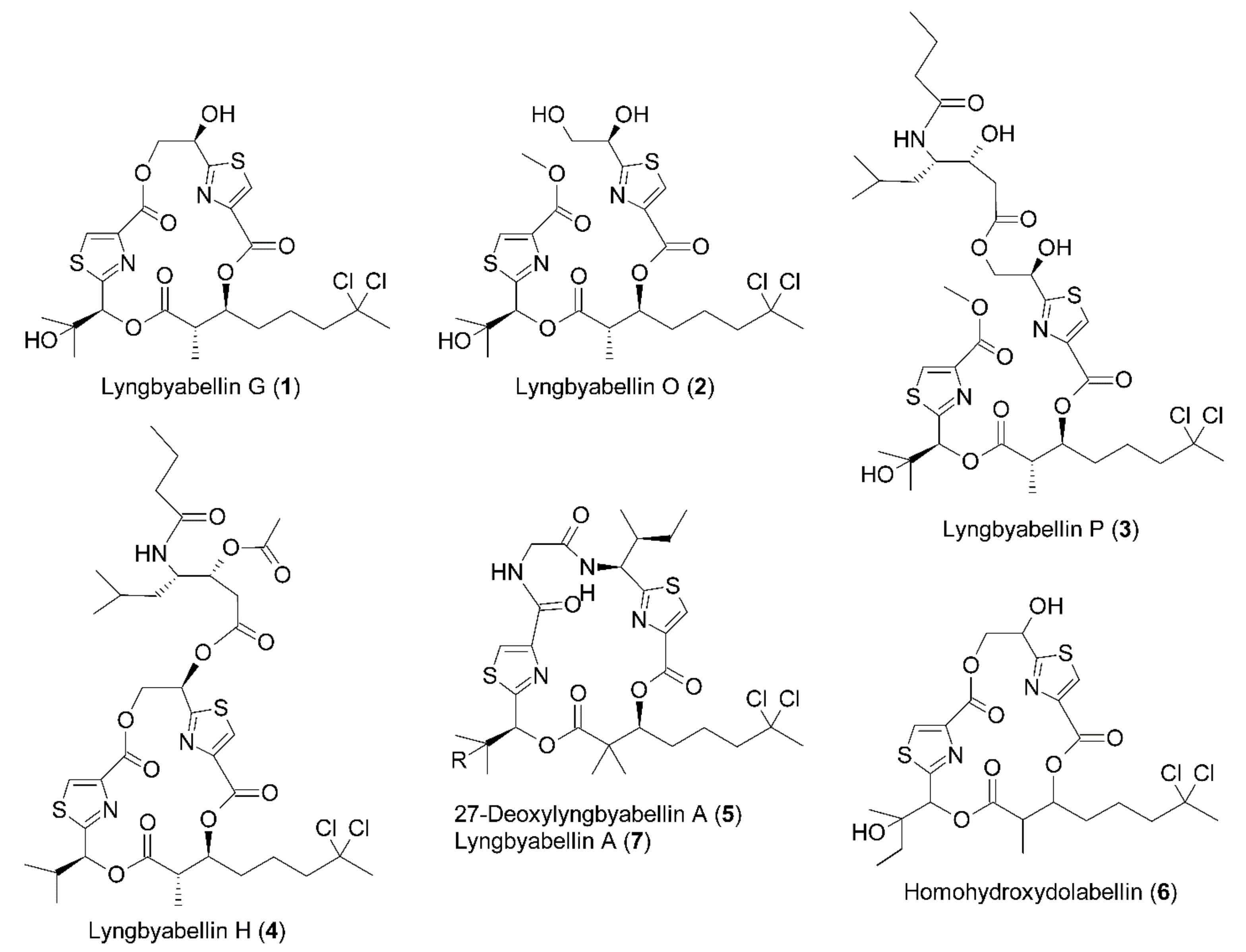
2.1. Isolation and Characterization of Compounds
2.2. Antifouling Activity
2.3. Cytotoxicity
2.4. Antiplasmodial Activity
3. Materials and Methods
3.1. Cyanobacteria Collection
3.2. Identification of Cyanobacteria
3.3. Extraction and Isolation of Compounds
3.4. Antifouling Assay on A. amphitrite
3.5. Cytotoxicity Assay on MCF7 Breast Cancer Cells
3.6. Antiplasmodial Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tan, L.T.; Phyo, M.Y. Marine cyanobacteria: A source of lead compounds and their clinically-relevant molecular targets. Molecules 2020, 25, 2197. [Google Scholar] [CrossRef] [PubMed]
- Al-Awadhi, F.H.; Luesch, H. Targeting eukaryotic proteases for natural products-based drug development. Nat. Prod. Rep 2019, 37, 827. [Google Scholar] [CrossRef] [PubMed]
- Tse, E.G.; Korsik, M.; Todd, M.H. The past, present and future of anti-malarial medicines. Malar. J. 2019, 18, 93. [Google Scholar] [CrossRef] [PubMed]
- Moo-Puc, R.; Robledo, D.; Freile-Pelegrin, Y. Evaluation of selected tropical seaweeds for in vitro anti-trichomonal activity. J. Ethnopharmacol. 2008, 120, 92–97. [Google Scholar] [CrossRef]
- Fred, M.S.; Ghee, T.T. Natural Products with Antimalarial Activity. In Phytochemistry and Pharmacognosy; Encyclopedia of Life Support Systems (EOLSS); Pezzutto, J.M., Kato, M.J., Eds.; Eolss Publishers: Oxford, UK, 2011; pp. 1–54. [Google Scholar]
- Qi, S.; Ma, X. Antifouling compounds from marine invertebrates. Mar. Drugs. 2017, 15, 263. [Google Scholar] [CrossRef]
- Oguri, Y.; Watanabe, M.; Ishikawa, T.; Kamada, T.; Vairappan, C.S.; Matsuura, H.; Kaneko, K.; Ishii, T.; Suzuki, M.; Yoshimura, E.; et al. New marine antifouling compounds from the red alga Laurencia sp. Mar. Drugs 2017, 15, 267. [Google Scholar] [CrossRef]
- Johnson, T.J.; Katuwal, S.; Anderson, G.A.; Gu, L.; Zhou, R.; Gibbons, W.R. Photobioreactor Cultivation Strategies for Microalgae and Cyanobacteria. Biotechnol. Prog. 2018, 34, 811–827. [Google Scholar] [CrossRef]
- Engene, N.; Rottacker, E.C.; Choi, H.; Byrum, T.; Kaštovský, J.H.; Ellisman, M.H.; Komárek, J.; Gerwick, W.H. Moorea producens gen. nov., sp. nov. and Moorea bouillonii comb. nov., tropical marine cyanobacteria rich in bioactive secondary metabolites. Int. J. Syst. Evol. Microbiol. 2012, 62, 1171–1178. [Google Scholar] [CrossRef]
- Eugene, N.; Paul, V.P.; Byrum, T.; Gerwick, W.H.; Thor, A.; Ellisman, M.H. Five chemically rich species of tropical marine cyanobacteria of the genus Okeania gen. nov. (oscillatoriales, cyanoprokaryota). J. Phycol. 2013, 49, 1095–1106. [Google Scholar] [CrossRef]
- Sone, H.; Kondo, T.; Kiryu, M.; Ishiwata, H.; Okika, M.; Yamada, K. Dolabellin, a cytotoxic bisthiazole metabolite from the sea hare Dolabella auricularia: Structural determination and synthesis. J. Org. Chem. 1995, 60, 4774–4781. [Google Scholar] [CrossRef]
- Han, B.; McPhail, K.L.; Gross, H.; Goeger, D.E.; Mooberry, S.L.; Gerwick, W.H. Isolation and structure of five lyngbyabellin derivatives from a Papua New Guinea collection of the marine cyanobacterium Lyngbya majuscula. Tetrahedron 2005, 61, 11723–11729. [Google Scholar] [CrossRef]
- Sweeney-Jones, A.M.; Gagaring, K.; Antonova-Koch, J.; Zhou, H.; Mojib, N.; Soapi, K.; Skolnick, J.; McNamara, C.W.; Kubanek, J. Antimalarial peptide and polyketide natural products from the Fijian marine cyanobacterium Moorea producens. Mar. Drugs 2020, 18, 167. [Google Scholar] [CrossRef] [PubMed]
- Pettit, G.R. The dolastatins. In Fortschritte der Chemie Organischer Naturstoffe Progress in the Chemistry of Organic Natural Products; Herz, W., Kirby, G.W., Moore, R.E., Steglich, W., Tamm, C., Eds.; Springer: Vienna, Austria, 1997; pp. 1–79. [Google Scholar] [CrossRef]
- Fennell, B.J.; Carolan, S.; Pettit, G.R.; Bell, A. Effects of the Antimitotic Natural Product Dolastatin 10, and Related Peptides, on the Human Malarial Parasite Plasmodium falciparum. J. Antimicrob. Chemother. 2003, 51, 833–841. [Google Scholar] [CrossRef] [PubMed]
- Linington, R.G.; Clark, B.R.; Trimble, E.E.; Almanza, A.; Luis-David, U.E.; Kyle, E.D.; Gerwick, W.H. Antimalarial peptides from marine cyanobacteria: Isolation and structural elucidation of gallinamide A. J. Nat. Prod. 2009, 72, 14–17. [Google Scholar] [CrossRef]
- Iwasaki, A.; Tadenuma, T.; Sumimoto, S.; Shiota, I.; Matsubara, T.; Saito-Nakano, Y.; Nozaki, T.; Sato, T.; Suenaga, K. Hoshinoamides A and B, acyclic lipopeptides from the marine cyanobacterium Caldora penicillate. J. Nat. Prod. 2018, 81, 2545–2552. [Google Scholar] [CrossRef]
- McPhail, K.L.; Correa, J.; Linington, R.G.; Gonzalez, J.; Ortega-Barría, E.; Capson, T.L.; Gerwick, W.H. Antimalarial linear lipopeptides from a panamanian strain of the marine cyanobacterium Lyngbya majuscula. J. Nat. Prod. 2007, 70, 984–988. [Google Scholar] [CrossRef]
- Linington, R.G.; González, J.; Ureña, L.-D.; Romero, L.I.; Ortega-Barría, E.; Gerwick, W.H. Venturamides A and B: Antimalarial constituents of the panamanian marine cyanobacterium Oscillatoria sp. J. Nat. Prod. 2007, 70, 397–401. [Google Scholar] [CrossRef]
- Vining, O.B.; Medina, R.A.; Mitchell, E.A.; Videau, P.; Li, D.; Serrill, J.D.; Kelly, J.X.; Gerwick, W.H.; Proteau, P.J.; Ishmael, J.E.; et al. Depsipeptide companeramides from a panamanian marine cyanobacterium associated with the coibamide producer. J. Nat. Prod. 2015, 78, 413–420. [Google Scholar] [CrossRef]
- Ozaki, K.; Iwasaki, A.; Sezawa, D.; Fujimura, H.; Nozaki, T.; Saito-Nakano, Y.; Suenaga, K.; Teruya, T. Isolation and total synthesis of mabuniamide, a lipopeptide from an Okeania sp. marine cyanobacterium. J. Nat. Prod. 2019, 82, 2907–2915. [Google Scholar] [CrossRef]
- Iwasaki, K.; Iwasaki, A.; Sumimoto, S.; Matsubara, T.; Sato, T.; Nozaki, T.; Saito-Nakano, Y.; Suenaga, K. Ikoamide, an antimalarial lipopeptide from an Okeania sp. marine cyanobacterium. J. Nat. Prod. 2020, 83, 481–488. [Google Scholar] [CrossRef]
- Shao, C.; Linington, R.G.; Balunas, M.J.; Centeno, A.; Boudreau, P.; Zhang, C.; Engene, N.; Spadafora, C.; Mutka, T.S.; Kyle, D.E.; et al. Bastimolide A, a potent antimalarial polyhydroxy macrolide from the marine cyanobacterium Okeania hirsute. J. Org. Chem. 2015, 80, 7849–7855. [Google Scholar] [CrossRef] [PubMed]
- Shao, C.; Mou, X.; Cao, F.; Spadafora, C.; Glukhov, E.; Gerwick, L.; Wang, C.; Gerwick, W.H. Bastimolide B, an antimalarial 24-membered marine macrolide possessing a tert-butyl group. J. Nat. Prod. 2018, 81, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Petitbois, J.G.; Casalme, L.O.; Lopez, J.A.V.; Alarif, W.M.; Abdel-Lateff, A.; Al-Lihaibi, S.S.; Yoshimura, E.; Nogata, Y.; Umezawa, T.; Matsuda, F.; et al. Serinolamides and lyngbyabellins from an Okeania sp. cyanobacterium collected from the red sea. J. Nat. Prod. 2017, 80, 2708–2715. [Google Scholar] [CrossRef] [PubMed]
- Luesch, H.; Yoshida, W.Y.; Moore, R.E.; Paul, V.J.; Mooberry, S.L. Isolation, structure determination, and biological activity of lyngbyabellin A from the marine cyanobacterium Lyngbya majuscula. J. Nat. Prod. 2000, 63, 611–615. [Google Scholar] [CrossRef] [PubMed]
- Matthew, S.; Salvador, L.A.; Schupp, P.J.; Paul, V.J.; Luesch, V.J. Cytotoxic halogenated macrolides and modified peptides from the apratoxin-producing marine cyanobacterium Lyngbya bouillonii from Guam. J. Nat. Prod. 2010, 73, 1544–1552. [Google Scholar] [CrossRef] [PubMed]
- Luesch, H.; Yoshida, W.Y.; Moore, R.E.; Paul, V.J. Structurally diverse new alkaloids from Palauan collections of the apratoxin-producing marine cyanobacterium Lyngbya sp. Tetrahedron 2002, 58, 58–7959. [Google Scholar] [CrossRef]
- Peatey, C.L.; Leroy, D.; Gardiner, D.L.; Trenholme, K.R. Anti-malarial drugs: How effective are they against Plasmodium falciparum gametocytes? Malar. J. 2012, 11, 34. [Google Scholar] [CrossRef]
- Lopez, J.A.V.; Al-Lihaibi, S.S.; Alarif, W.M.; Abdel-Lateff, A.; Nogata, Y.; Washio, K.; Morikawa, M.; Okino, T. Wewakazole B, a cytotoxic cyanobactin from the Cyanobacterium Moorea producens Collected in the Red Sea. J. Nat. Prod. 2016, 79, 1213–1218. [Google Scholar] [CrossRef]
- Aydin-Schmidt, B. Giemsa staining of thick or thin blood films. In Methods in Malaria Research, 6th ed.; Moll, K., Kaneko, A., Scherf, A., Wahlgren, M., Eds.; EVIMalaR: Glasgow, UK, 2013; pp. 23–24. [Google Scholar]
- Fathoni, I.; Poerwanto, S.H.; Astuti, R.R.U.N.W. Bioactivity study of moss Dumortiera hirsuta (swaegr.) nees ethanolic extract on parasitemic level of Plasmodium berghei vincke and lips, 1948 in mice (Mus musculus L.). Asian J. Pharm. Clin. Res. 2017, 10, 5–8. [Google Scholar] [CrossRef][Green Version]
- Marquez, B.L.; Watts, K.S.; Yokochi, A.; Roberts, M.A.; Verdier-Pinard, P.; Jimenez, J.I.; Hamel, E.; Scheuer, P.J.; Gerwick, W.H. Structure and absolute stereochemistry of hectochlorin, a potent stimulator of actin assembly. J. Nat. Prod. 2002, 65, 866–871. [Google Scholar] [CrossRef]
- Williams, P.G.; Luesch, H.; Yoshida, W.Y.; Moore, R.E.; Paul, V.J. Continuing studies on the cyanobacterium Lyngbya sp.: Isolation and structure determination of 15-norlyngbyapeptin A and lyngbyabellin D. J. Nat. Prod. 2003, 66, 595–598. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Mevers, E.; Byrun, T.; Valeriote, F.A.; Gerwick, W.H. Lyngbyabellins K–N from two Palmyra atoll collections of the marine cyanobacterium Moorea bouillonii. Eur. J. Org. Chem. 2012, 27, 5141–5150. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
| Compounds | Sources | Structural Class | IC50 (µM) | Assay Method | References |
|---|---|---|---|---|---|
| Dolastatin 10 | Dolabella auricularia | peptide | 0.0001 | pLDH | [15] |
| Dolastatin 15 | Symploca sp. | peptide | 0.0024 | pLDH | [15] |
| Gallinamide A | Schizotrix sp. | depsipeptide | 8.4 | Pico-Green | [16] |
| Hoshinoamide A | Caldora penicillate | lipopeptides | 5.2 | pLDH | [17] |
| Carmabin A | Lyngbya. majuscula | lipopeptides | 4.3 | Pico-Green | [18] |
| Dragomabin | L. majuscula | lipopeptides | 6.0 | Pico-Green | [18] |
| Venturamide A | Oscillatoria sp. | hexapeptides | 8.2 | Pico-Green | [19] |
| Companeramide A | Leptolyngbya | depsipeptide | 5.7 | Sybr Green | [20] |
| Mabunamide | Okeania sp. | lipopeptides | 1.4 | pLDH | [21] |
| Ikoamide | Okeania sp. | lipopeptides | 0.14 | pLDH | [22] |
| Kakeromamide B | Moorea producens | pentapeptides | 8.9 | Sybr Green | [13] |
| Ulongamide A | M. producens | depsipeptides | 0.99 | Sybr Green | [13] |
| Lyngbyabellin A | M. producens | depsipeptides | 0.0015 | Sybr Green | [13] |
| Bastimolide A | Okeania hirsuta | polyhydroxy macrolide | 0.27 | Sybr Green | [23] |
| Bastimolide B | O. hirsuta | macrolide | 5.7 | Pico-Green | [24] |
| Compounds | Bioactivities | IC50 (µM) | EC50(µg/mL) | Cells Lines/Organisms | References |
|---|---|---|---|---|---|
| Lyngbyabellin A | Antimalarial, anticancer | 0.021–0.345 | HT29, HeLA, KB, LoVo | [13,26,27], current study | |
| 0.0015 | P. falciparum (liver-stage) | ||||
| 0.3 | P.falciparum (intraerythrocytic stage) | ||||
| 27-deoxylyngbyabellin A | Antifouling, anticancer | 0.0073–0.31 | HT29, HeLA, MCF7 | [27], current study | |
| 0.09 (but toxic) | A.amphitrite barnacle cypris | ||||
| Lyngbyabellin B | Anticancer | 0.1–1.1 | HT29, HeLA, CA46, PtK2 | [27,33] | |
| Lyngbyabellin C | Anticancer | 2.1–5.3 | KB, LoVo | [28] | |
| Lyngbyabellin D | Anticancer | 0.1 | KB | [34] | |
| Lyngbyabellin E | Anticancer | 0.4–1.2 | H460, Neuro-2a | [12] | |
| Lyngbyabellin F | Anticancer | 1.0–1.8 | H460, Neuro-2a | [12] | |
| Lyngbyabellin G | Antimalarial, anticancer, antifouling | 2.2–120 | H460, Neuro-2a, MCF7 | [12,25], current study | |
| 4.41 | A.amphitrite barnacle cypris | ||||
| P.falciparum (intraerythrocytic stage) | |||||
| Lyngbyabellin H | Anticancer | 0.07–1.4 | H460, Neuro-2a, MCF7 | [12], current study | |
| Inactive | A.amphitrite barnacle cypris | [12] | |||
| Lyngbyabellin I | Anticancer | 0.7–1.0 | H460, Neuro-2a | [12] | |
| Lyngbyabellin J | Anticancer | 0.041–0.054 | HT29, HeLA | [12] | |
| Lyngbyabellin K | Inactive | H460 | [35] | ||
| Lyngbyabellin L | Inactive | H460 | [35] | ||
| Lyngbyabellin M | Inactive | H460 | [35] | ||
| Lyngbyabellin N | Anticancer | 0.0048–1.8 | H460, HCT116 | [35] | |
| Lyngbyabellin O | Antifouling | Inactive | MCF7 | [25] | |
| 0.24 | A.amphitrite barnacle cypris | ||||
| Lyngbyabellin P | Anticancer, antifouling | 9.0 | MCF7 | [25] | |
| 0.62 | A.amphitrite barnacle cypris |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fathoni, I.; Petitbois, J.G.; Alarif, W.M.; Abdel-Lateff, A.; Al-Lihaibi, S.S.; Yoshimura, E.; Nogata, Y.; Vairappan, C.S.; Sholikhah, E.N.; Okino, T. Bioactivities of Lyngbyabellins from Cyanobacteria of Moorea and Okeania Genera. Molecules 2020, 25, 3986. https://doi.org/10.3390/molecules25173986
Fathoni I, Petitbois JG, Alarif WM, Abdel-Lateff A, Al-Lihaibi SS, Yoshimura E, Nogata Y, Vairappan CS, Sholikhah EN, Okino T. Bioactivities of Lyngbyabellins from Cyanobacteria of Moorea and Okeania Genera. Molecules. 2020; 25(17):3986. https://doi.org/10.3390/molecules25173986
Chicago/Turabian StyleFathoni, Imam, Julie G. Petitbois, Walied M. Alarif, Ahmed Abdel-Lateff, Sultan S. Al-Lihaibi, Erina Yoshimura, Yasuyuki Nogata, Charles S. Vairappan, Eti Nurwening Sholikhah, and Tatsufumi Okino. 2020. "Bioactivities of Lyngbyabellins from Cyanobacteria of Moorea and Okeania Genera" Molecules 25, no. 17: 3986. https://doi.org/10.3390/molecules25173986
APA StyleFathoni, I., Petitbois, J. G., Alarif, W. M., Abdel-Lateff, A., Al-Lihaibi, S. S., Yoshimura, E., Nogata, Y., Vairappan, C. S., Sholikhah, E. N., & Okino, T. (2020). Bioactivities of Lyngbyabellins from Cyanobacteria of Moorea and Okeania Genera. Molecules, 25(17), 3986. https://doi.org/10.3390/molecules25173986







