Investigation on the Interaction between Cellulosic Paper and Organic Acids Based on Molecular Dynamics
Abstract
1. Introduction
2. Results and Discussion
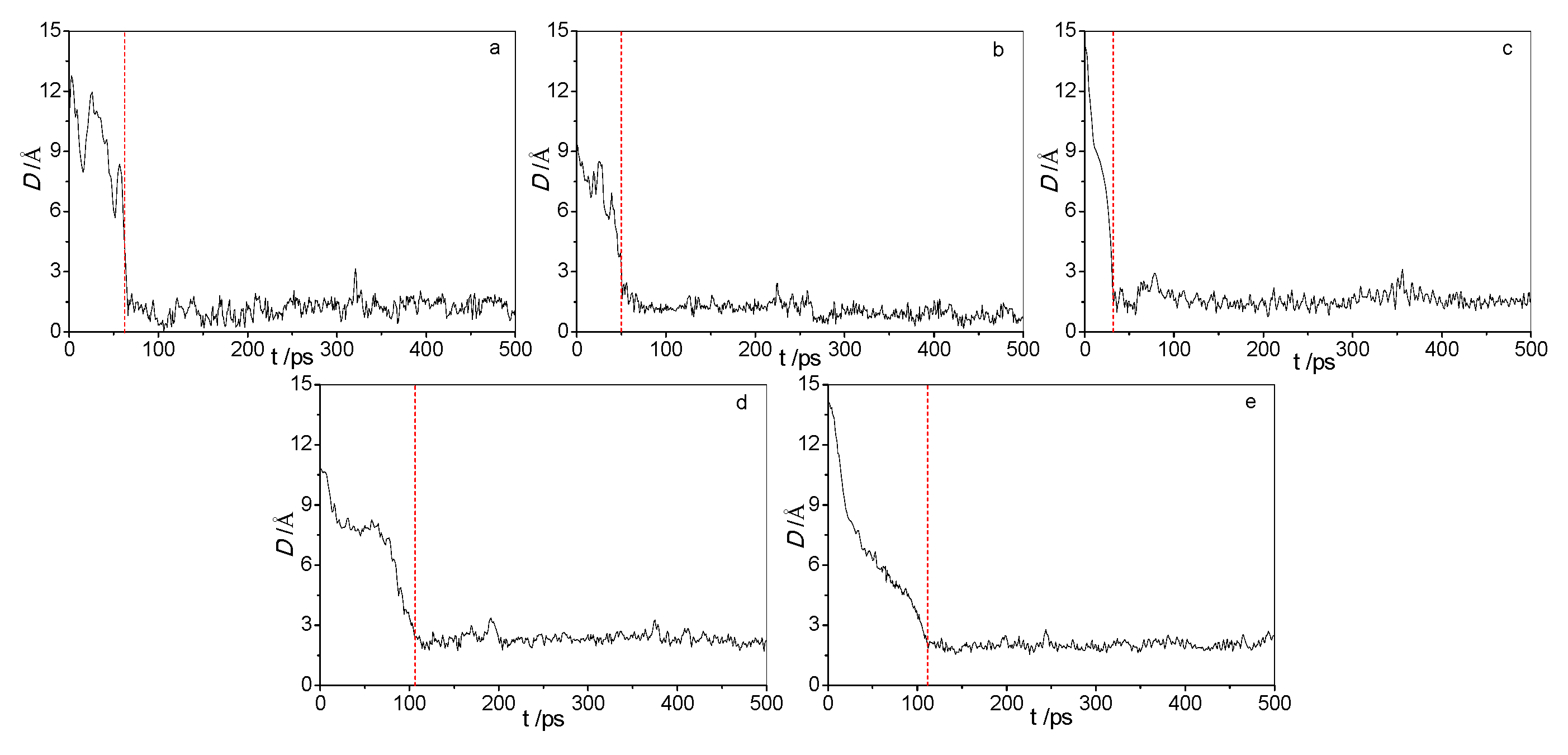
2.1. Adsorption of Acid Molecules
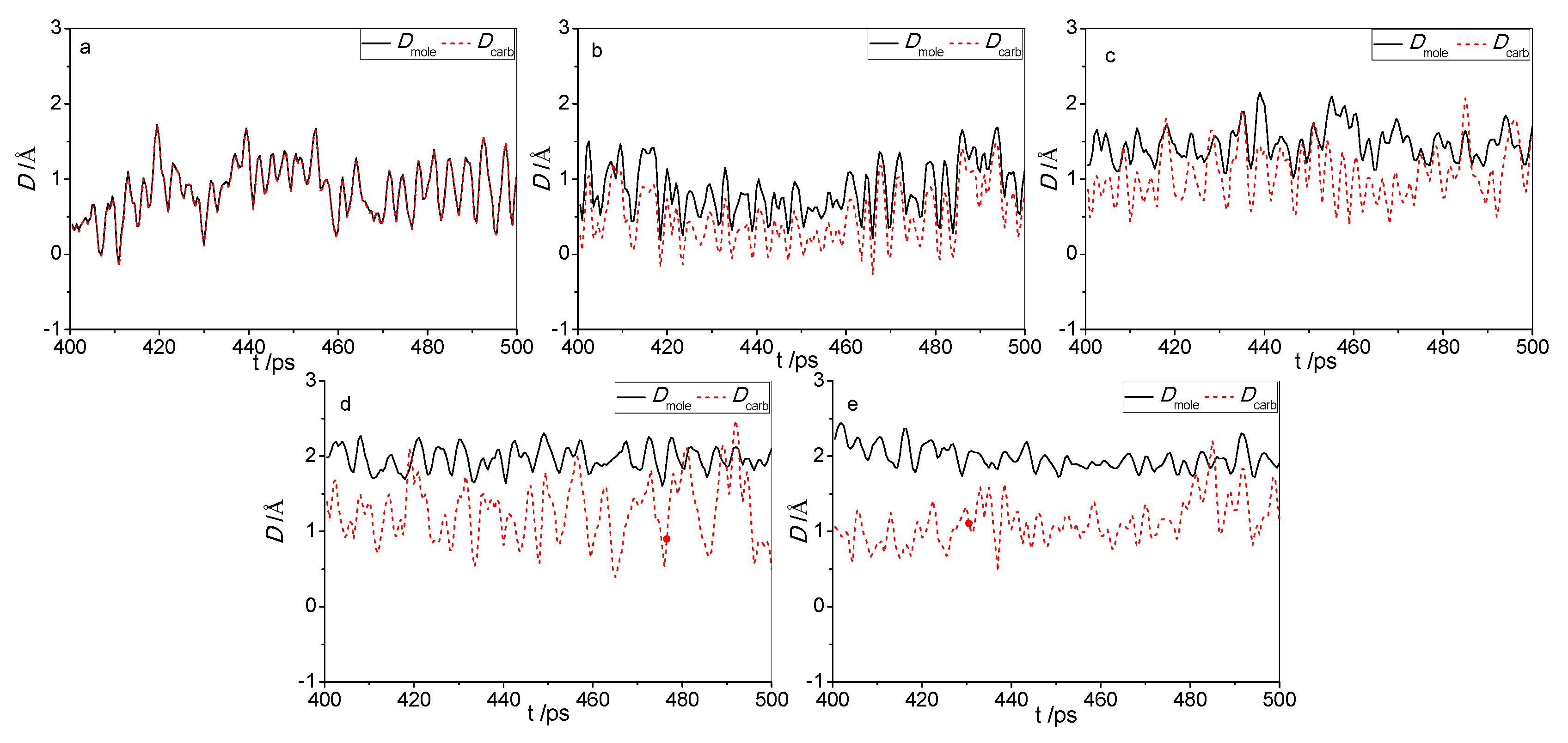
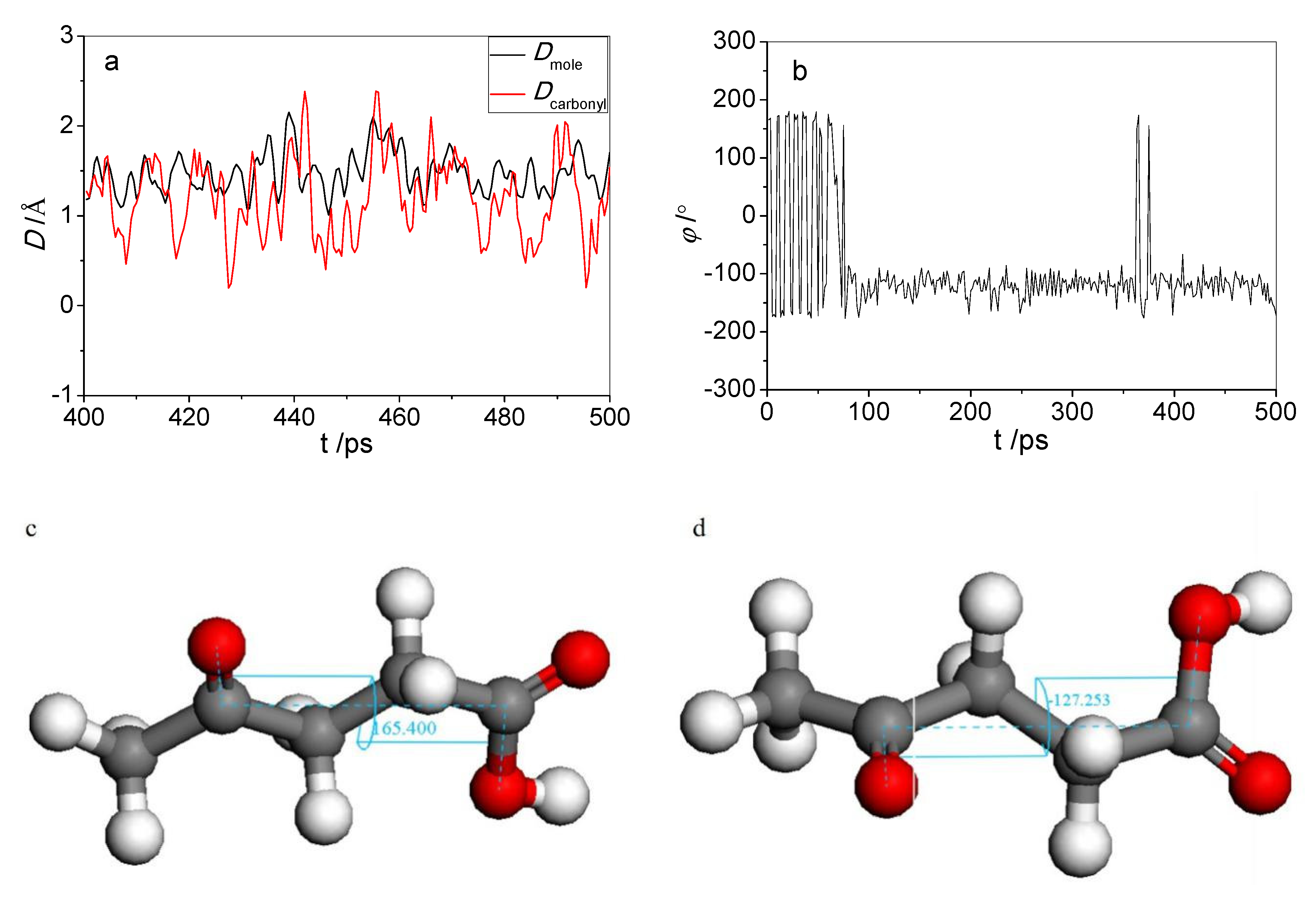
2.2. Morphologies of the Adsorbed Acid Molecules
2.2.1. Deformation Energy
2.2.2. Adsorption Energy
2.3. Solubility Parameter
3. Calculation Methods
3.1. Modeling
3.2. Simulated Details
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Christina, A.J.; Salam, M.A.; Rahman, Q.M.; Wen, F.; Ang, S.P.; Voon, W. Causes of transformer failures and diagnostic methods—A review. Renew. Sust. Energ. Rev. 2018, 82, 1442–1456. [Google Scholar]
- Vasovic, V.; Lukic, J.; Mihajlovic, D.; Pejovic, B.; Radakovic, Z.; Radoman, U.; Orlovic, A. Aging of transformer insulation—experimental transformers and laboratory models with different moisture contents: Part I—DP and furans aging profiles. IEEE Trans. Dielectr. Electr. Insul. 2019, 26, 1840–1846. [Google Scholar] [CrossRef]
- Zheng, H.; Zhang, C.; Zhang, Y.; Liu, J.; Zhang, E.; Shi, Z.; Shao, G.; Shi, Z.; Guo, J.; Zhang, C. Optimization of Ethanol Detection by Automatic Headspace Method for Cellulose Insulation Aging of Oil-immersed Transformers. Polymers 2020, 12, 1567. [Google Scholar] [CrossRef] [PubMed]
- Lundgaard, L.E.; Hansen, W.; Linhjell, D.; Painter, T.J. Aging of oil-impregnated paper in power transformers. IEEE Trans. Power Deliv. 2004, 19, 230–239. [Google Scholar] [CrossRef]
- Huang, Z.; Wang, F.; Wang, Q.; Yao, W.; Sun, K.; Zhang, R.; Zhao, J.; Lou, Z.; Li, J. Significantly Enhanced Electrical Performances of Eco-Friendly Dielectric Liquids for Harsh Conditions with Fullerene. Nanomaterials 2019, 9, 989. [Google Scholar] [CrossRef]
- Lundgaard, L.E.; Hansen, W.; Ingebrigtsen, S. Ageing of mineral oil impregnated cellulose by acid catalysis. IEEE Trans. Dielectr. Electr. Insul. 2008, 15, 540–546. [Google Scholar] [CrossRef]
- Azis, N.; Liu, Q.; Wang, Z.D. Ageing assessment of transformer paper insulation through post mortem analysis. IEEE Trans. Dielectr. Electr. Insul. 2014, 21, 845–853. [Google Scholar] [CrossRef]
- Ingebrigtsen, S.; Dahlund, M.; Hansen, W.; Linhjell, D.; Lundgaard, L.E. Solubility of carboxylic acids in paper (Kraft)-oil insulation systems. In Proceedings of the 17th Annual Meeting of the IEEE Lasers and Electro-Optics Society, Boulder, CO, USA, 20–20 October 2004; pp. 253–257. [Google Scholar]
- Kouassi, K.D.; Fofana, I.; Cissé, L.; Hadjadj, Y.; Yapi, K.M.L.; Diby, K.A. Impact of Low Molecular Weight Acids on Oil Impregnated Paper Insulation Degradation. Energies 2018, 11, 1465. [Google Scholar] [CrossRef]
- Lelekakis, N.; Wijaya, J.; Martin, D.; Susa, D. The effect of acid accumulation in power-transformer oil on the aging rate of paper insulation. IEEE Electr. Insul. Mag. 2014, 30, 19–26. [Google Scholar] [CrossRef]
- Leach, A.R. Molecular Modelling: Principles and Applications, 2nd ed.; Pearson Education: Harlow, UK, 2001. [Google Scholar]
- Xiao, X.; Yang, W.; Li, L.; Zhong, T.; Zhang, X. Application of molecular simulation in transformer oil-paper insulation. J. Eng. 2019, 16, 1324–1327. [Google Scholar] [CrossRef]
- Wang, Y.; Fan, P.; Tian, M.; Chen, B. Molecular simulation for the effect of electric fields on the yield behaviour and cracking process of insulation paper. Mol. Simul. 2015, 41, 1137–1142. [Google Scholar] [CrossRef]
- Yaws, C.L. Thermophysical Properties of Chemicals and Hydrocarbons; William Andrew: Norwich, NY, USA, 2008; pp. 312–400. [Google Scholar]
- Hildebrand, J.H.; Scott, R.L. The Solubility of Nonelectrolytes, 3rd ed.; Reinhold Press: New York, NY, USA, 1950. [Google Scholar]
- Karasawa, N.; Goddard III, W.A. Force fields, structures, and properties of poly (vinylidene fluoride) crystals. Macromolecules 2002, 25, 7268–7281. [Google Scholar] [CrossRef]
- Steinbach, P.J.; Brooks, B.R. New spherical-cutoff methods for long-range forces in macromolecular simulation. J. Comput. Chem. 1994, 15, 667–683. [Google Scholar] [CrossRef]
- Ewald, P.P. Die Berechnung optischer und elektrostatischer Gitterpotentiale. Ann. Phys. 1921, 369, 253–287. [Google Scholar] [CrossRef]
- Nymand, T.M.; Linse, P. Ewald summation and reaction field methods for potentials with atomic charges, dipoles, and polarizabilities. J. Chem. Phys. 2000, 112, 6152–6160. [Google Scholar] [CrossRef]
- Prevost, T.A.; Oommen, T.V. Cellulose insulation in oil-filled power transformers: Part I-history and development. IEEE Electr. Insul. Mag. 2006, 22, 28–35. [Google Scholar] [CrossRef]
- Oommen, T.V.; Prevost, T.A. Cellulose insulation in oil-filled power transformers: Part II maintaining insulation integrity and life. IEEE Electr. Insul. Mag. 2006, 22, 5–14. [Google Scholar] [CrossRef]
- Nishiyama, Y.; Langan, P.; Chanzy, H. Crystal structure and hydrogen-bonding system in cellulose Iβ from synchrotron X-ray and neutron fiber diffraction. J. Am. Chem. Soc. 2002, 124, 9074–9082. [Google Scholar] [CrossRef]
- Besombes, S.; Mazeau, K. The cellulose/lignin assembly assessed by molecular modeling. Part 1: Adsorption of a threo guaiacyl β-O-4 dimer onto a Iβ cellulose whisker. Plant. Physiol. Biochem. 2005, 43, 299–308. [Google Scholar] [CrossRef]
- Besombes, S.; Mazeau, K. The cellulose/lignin assembly assessed by molecular modeling. Part 2: Seeking for evidence of organization of lignin molecules at the interface with cellulose. Plant. Physiol. Biochem. 2005, 43, 277–286. [Google Scholar] [CrossRef]
- Baker, A.A.; Helbert, W.; Sugiyama, J.; Miles, M.J. New insight into cellulose structure by atomic force microscopy shows the Iα crystal phase at near-atomic resolution. Biophys. J. 2000, 79, 1139–1145. [Google Scholar] [CrossRef]
- Kuutti, L.; Peltonen, J.; Pere, J.; Teleman, O. Identification and surface structure of crystalline cellulose studied by atomic force microscopy. J. Microsc. 1995, 178, 1–6. [Google Scholar] [CrossRef]
- Baker, A.A.; Helbert, W.; Sugiyama, J.; Miles, M.J. High-resolution atomic force microscopy of nativeValoniacellulose I microcrystals. J. Struct. Biol. 1997, 119, 129–138. [Google Scholar] [CrossRef]
- Baker, A.A.; Helbert, W.; Sugiyama, J.; Miles, M.J. Surface structure of native cellulose microcrystals by AFM. Appl. Phys. A 1998, 66, S559–S563. [Google Scholar] [CrossRef]
- Borghesani, A.F.; Aitken, F. Molecular dynamics simulations of the O2− ion mobility in dense Ne gas at low temperature: Influence of the repulsive part of the ion-neutral interaction potential. IEEE Trans. Dielectr. Electr. Insul. 2018, 25, 1992–1998. [Google Scholar] [CrossRef]
- Tang, C.; Zhang, S.; Xie, J.; Lv, C. Molecular simulation and experimental analysis of Al2O3-nanoparticle-modified insulation paper cellulose. IEEE Trans. Dielectr. Electr. Insul. 2017, 24, 1018–1026. [Google Scholar] [CrossRef]
- Tang, C.; Zhang, S.; Li, X.; Xiong, B.; Xie, J. Experimental analyses and molecular simulation of the thermal aging of transformer insulation paper. IEEE Trans. Dielectr. Electr. Insul. 2015, 22, 3608–3616. [Google Scholar] [CrossRef]
- Du, B.X.; Su, J.G.; Han, T. Compressive stress dependence of electrical tree growth characteristics in EPDM. IEEE Trans. Dielectr. Electr. Insul. 2018, 25, 13–20. [Google Scholar] [CrossRef]
- Du, B.X.; Su, J.G.; Han, T. Effects of mechanical stretching on electrical treeing characteristics in EPDM. IEEE Trans. Dielectr. Electr. Insul. 2018, 25, 84–93. [Google Scholar] [CrossRef]
- Liao, R.; Zhu, M.; Zhou, X.; Zhang, F.; Yan, J.; Zhu, W.; Gu, C. Molecular dynamics study of the disruption of H-bonds by water molecules and its diffusion behavior in amorphous cellulose. Mod. Phys. Lett. B 2012, 26, 1250088. [Google Scholar] [CrossRef]
- Mazeau, K.; Heux, L. Molecular dynamics simulations of bulk native crystalline and amorphous structures of cellulose. J. Phys. Chem. B 2003, 107, 2394–2403. [Google Scholar] [CrossRef]

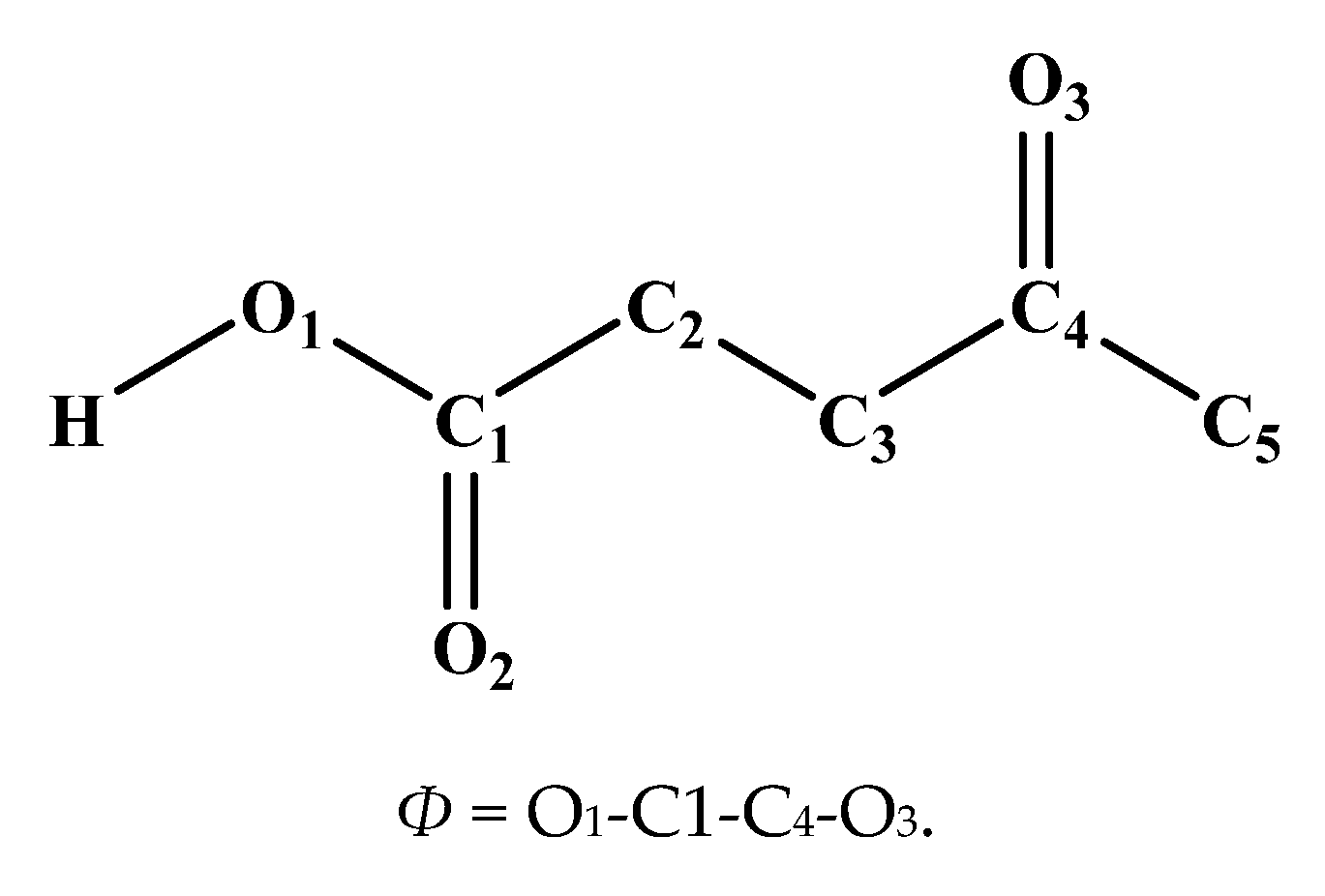
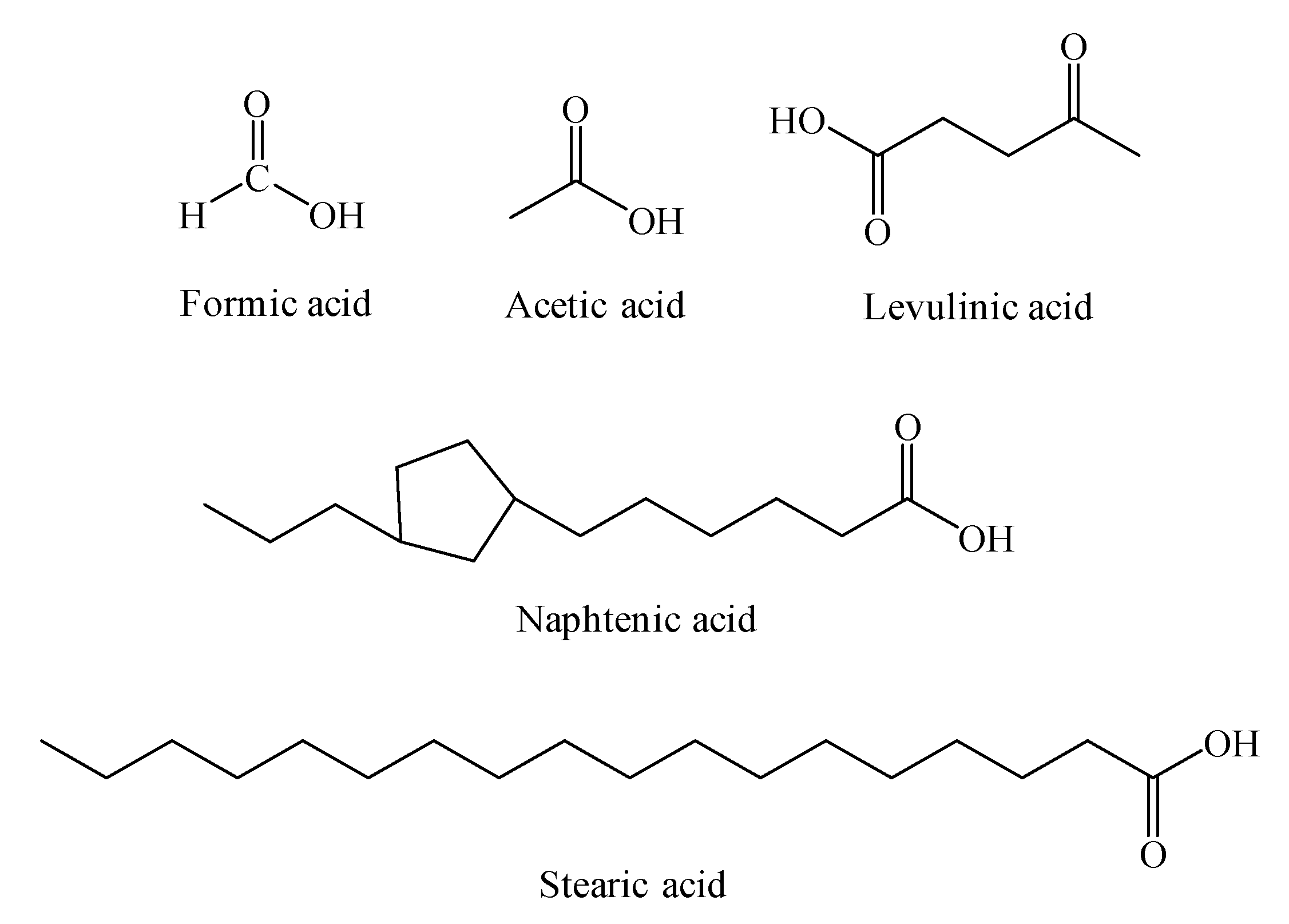

| Formic | Acetic | Levulinic | Naphtenic | Stearic | |
|---|---|---|---|---|---|
| Dmole | 15.16 | 15.16 | 15.77 | 16.27 | 16.28 |
| Dcarb | 15.14 | 14.79 | 15.36 | 15.56 | 15.42 |
| Ddiff | 0.01 | 0.37 | 0.41 | 0.71 | 0.86 |
| Formic | Acetic | Levulinic | Naphtenic | Stearic | |
|---|---|---|---|---|---|
| Efree | −33.7215 | −56.9919 | −49.7096 | −66.4094 | −94.1720 |
| Ebind | −29.2497 | −51.8050 | −35.5326 | −20.9013 | −15.4935 |
| Edef | 4.4718 | 5.18689 | 14.1770 | 45.5081 | 78.6786 |
| Formic | Acetic | Levulinic | Naphtenic | Stearic | |
|---|---|---|---|---|---|
| Eads | 11.4667 | 13.9769 | 19.4414 | 23.4770 | 26.7302 |
| EVdW | 4.8227 | 5.9117 | 9.9447 | 17.6817 | 23.3993 |
| Eelec | 8.0650 | 6.6441 | 9.4967 | 5.7953 | 3.3309 |
| Formic | Acetic | Levulinic | Naphtenic | Stearic | |
|---|---|---|---|---|---|
| V (cm3/mol) | 39.42 | 57.10 | 113.40 | 264.85 | 350.76 |
| V ([26]) | 37.912 | 57.577 | 103.573 | -- | 313.898 |
| D (kcal/cm3) | 0.2909 | 0.2448 | 0.1714 | 0.0886 | 0.0762 |
| ∆E (kcal/mol) | 6.995 | 8.7898 | 5.2644 | −22.0311 | −51.9484 |
| Cellulose | Formic | Acetic | Levulinic | Naphtenic | Stearic | Oil | |
|---|---|---|---|---|---|---|---|
| δ | 32 | 33.2134 | 28.6448 | 27.0623 | 19.7663 | 18.5987 | 16–19 * |
| δE | -- | 23.4952 | 19.1791 | 15.9908 | 9.07736 | 7.81325 | -- |
| δV | -- | 23.4702 | 21.2716 | 21.8312 | 17.5577 | 16.8772 | -- |
| δ ([26]) | 32 | 24.242 | 20.613 | 25.314 | 16.344 | 17.975 | -- |
| Name | Molecular Formula 1 | Molecular Formula 2 | Molecular Weight |
|---|---|---|---|
| Formic | HCOOH | CH2O2 | 46 |
| Acetic | CH3COOH | C2H4O2 | 60 |
| Levulinic | CH3COCH2CH2COOH | C5H8O3 | 116 |
| Naphtenic | CnH2n-1COOH a | Cn+1H2nCO2 | 226 |
| Stearic | CH3(CH2)16COOH | C18H36O2 | 284 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, M.; Gu, C.; Zhu, W. Investigation on the Interaction between Cellulosic Paper and Organic Acids Based on Molecular Dynamics. Molecules 2020, 25, 3938. https://doi.org/10.3390/molecules25173938
Zhu M, Gu C, Zhu W. Investigation on the Interaction between Cellulosic Paper and Organic Acids Based on Molecular Dynamics. Molecules. 2020; 25(17):3938. https://doi.org/10.3390/molecules25173938
Chicago/Turabian StyleZhu, Mengzhao, Chao Gu, and Wenbing Zhu. 2020. "Investigation on the Interaction between Cellulosic Paper and Organic Acids Based on Molecular Dynamics" Molecules 25, no. 17: 3938. https://doi.org/10.3390/molecules25173938
APA StyleZhu, M., Gu, C., & Zhu, W. (2020). Investigation on the Interaction between Cellulosic Paper and Organic Acids Based on Molecular Dynamics. Molecules, 25(17), 3938. https://doi.org/10.3390/molecules25173938





