Design, Synthesis and Biological Evaluation of Novel Triazole N-acylhydrazone Hybrids for Alzheimer’s Disease
Abstract
1. Introduction
2. Results and Discussion
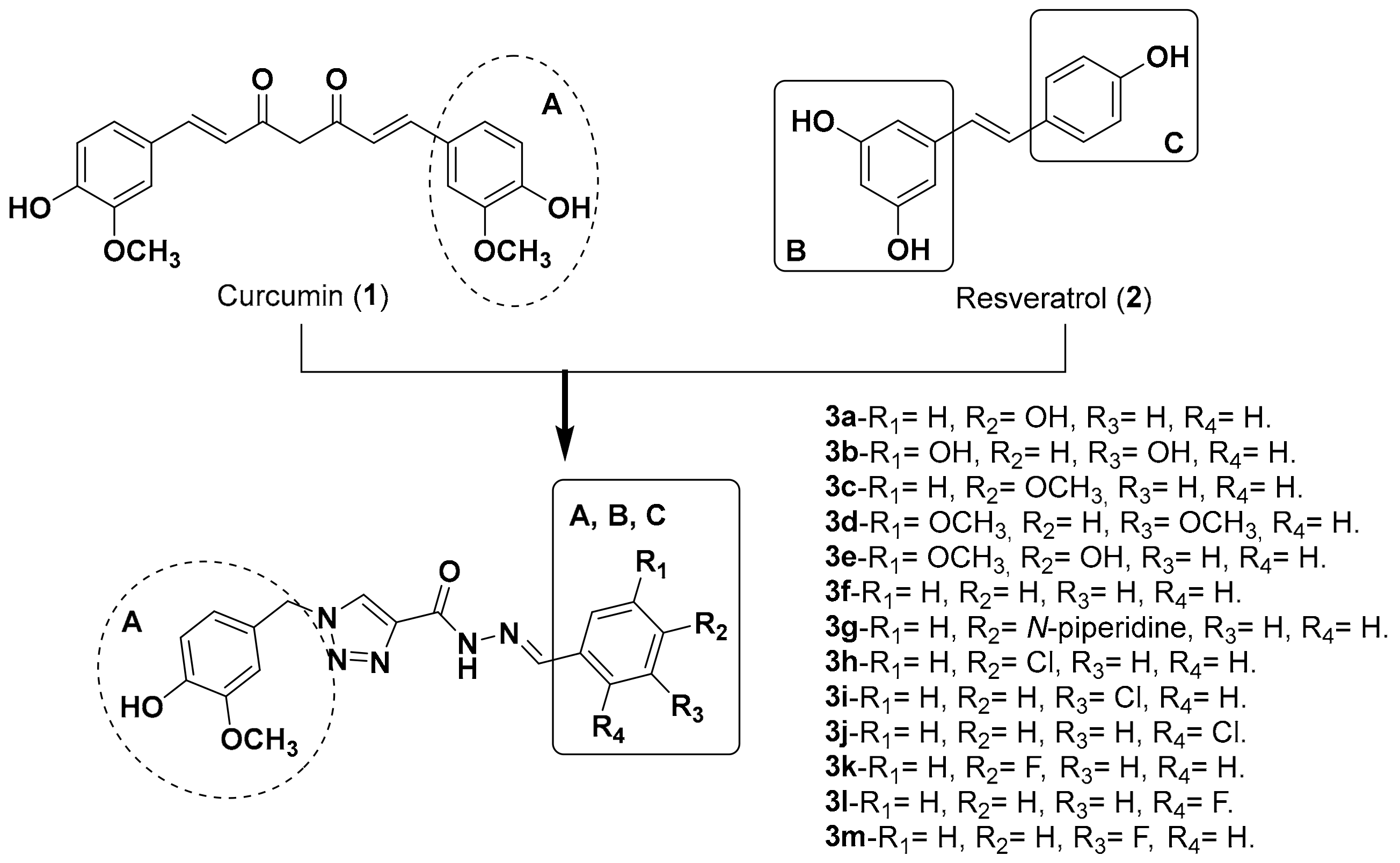
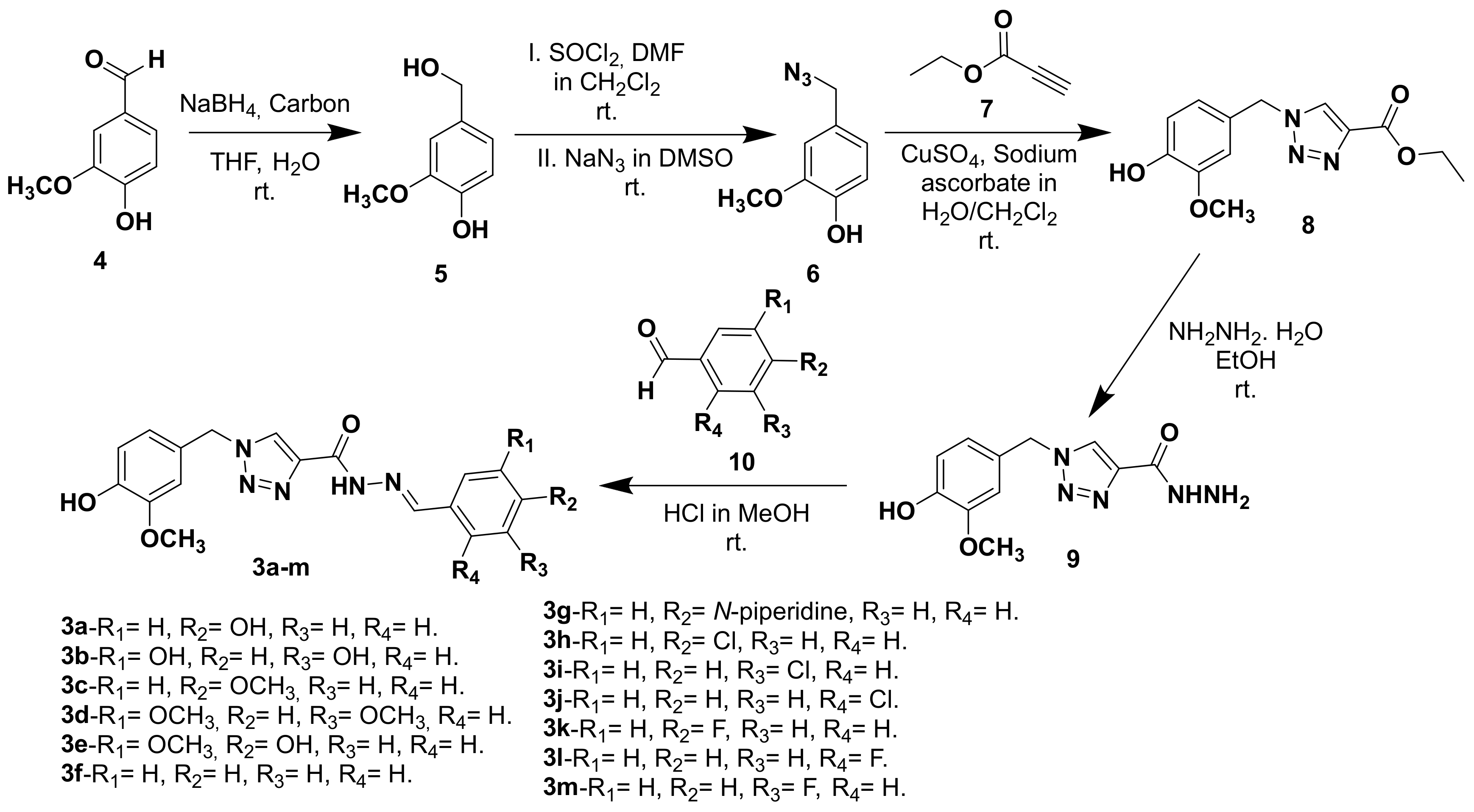
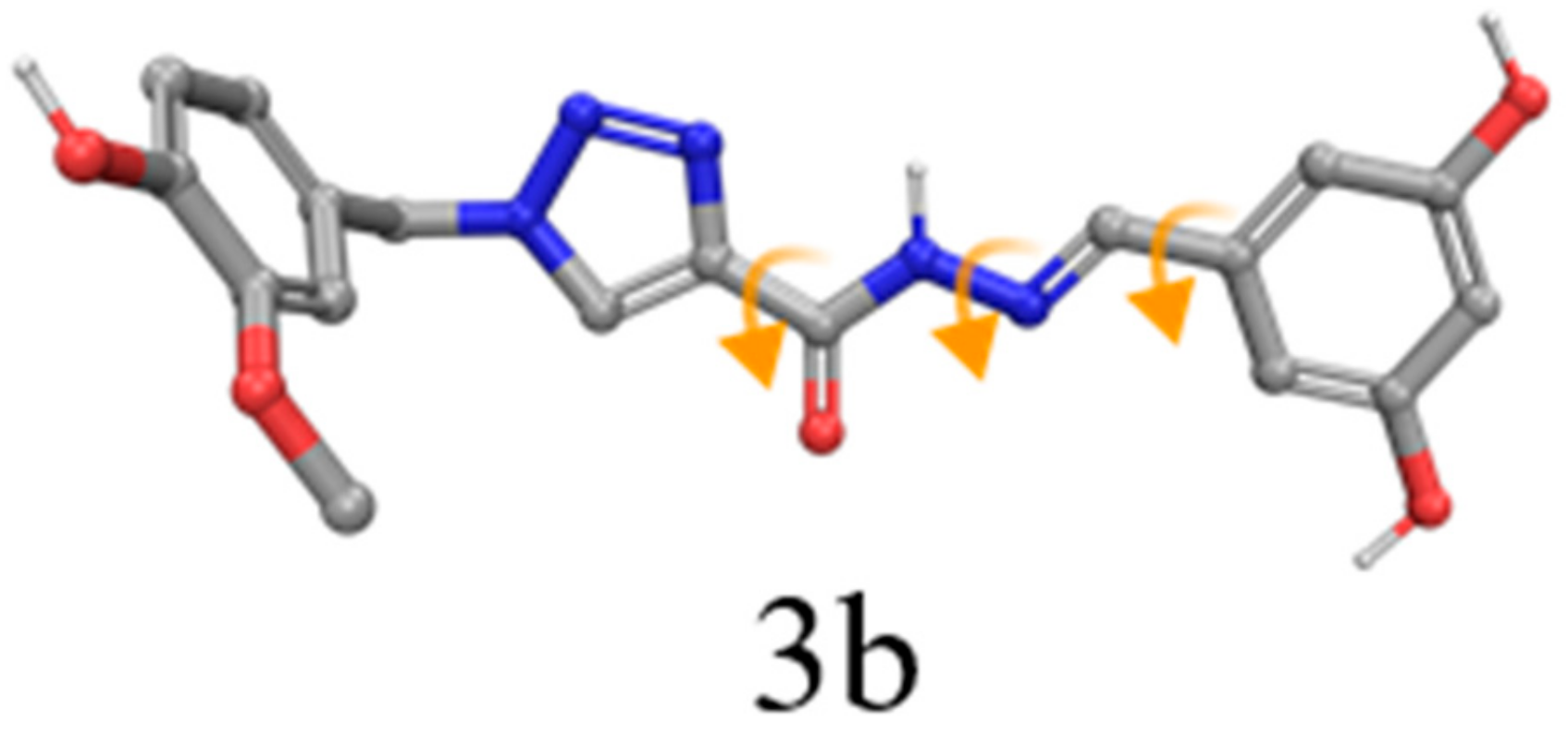
2.1. Chemistry
2.2. Biological Evaluation and Computational Study
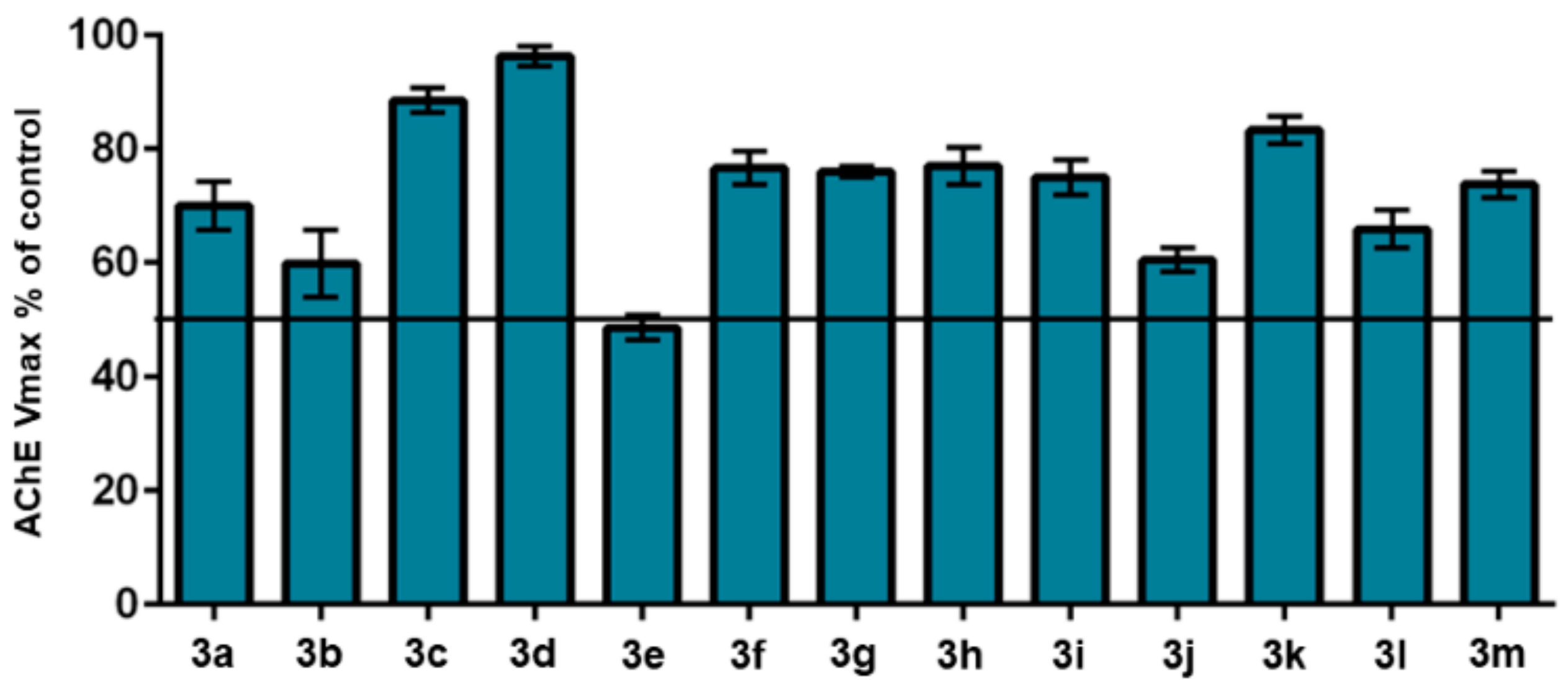
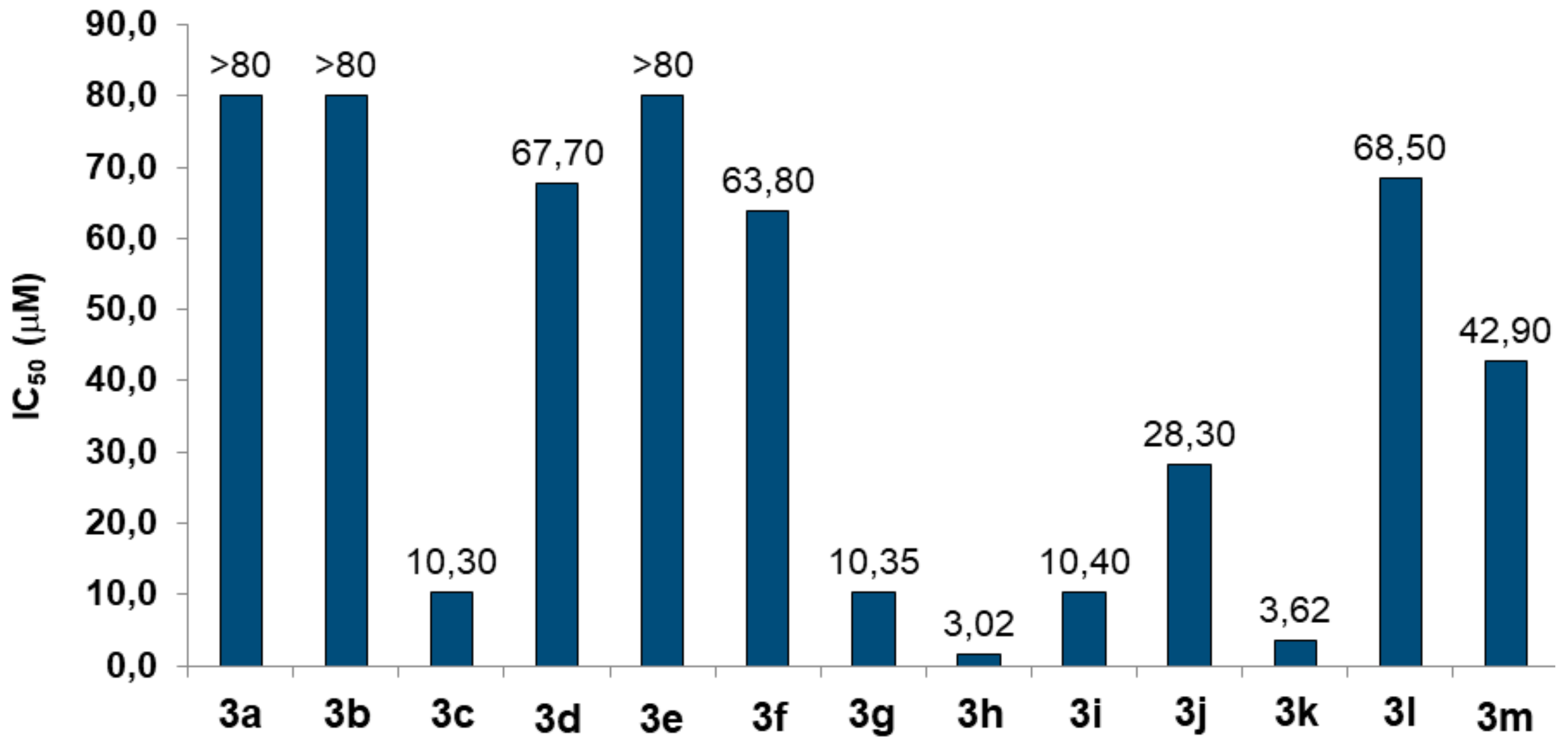
2.2.1. Evaluation of AChE Inhibition
2.2.2. Toxicity
2.2.3. In Silico ADME Properties
2.2.4. Antioxidant Activity
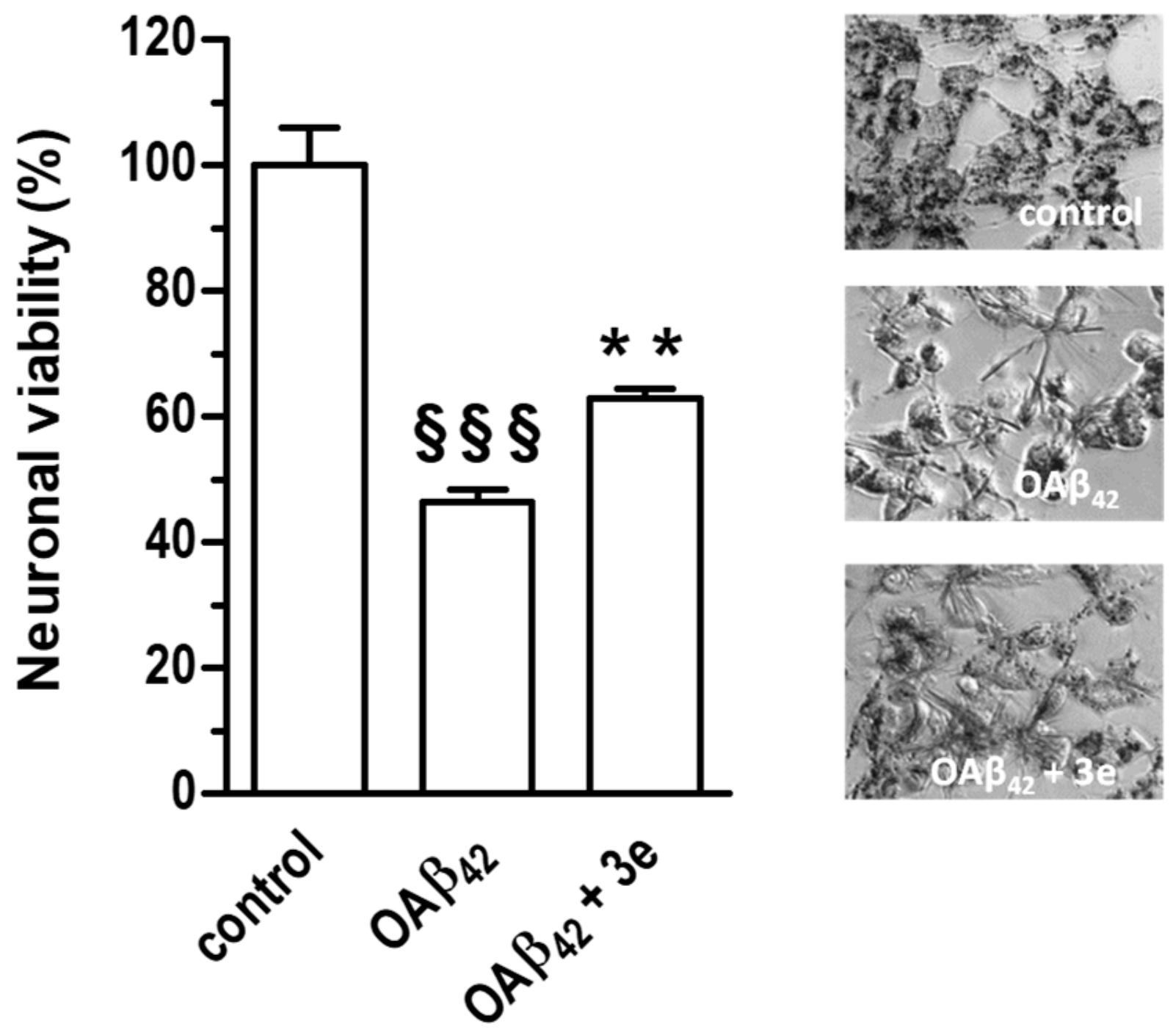
2.2.5. Neuroprotective Activity
3. Conclusions
4. Experimental Section
4.1. General Information
4.2. Chemistry
4.2.1. 4-(Hydroxymethyl)-2-methoxyphenol (5)
4.2.2. 4-(Azidomethyl)-2-methoxyphenol (6)
4.2.3. Ethyl 1-(4-hydroxy-3-methoxybenzyl)-1H-1,2,3-triazole-4-carboxylate (8)
4.2.4. 1-(4-Hydroxy-3-methoxybenzyl)-1H-1,2,3-triazole-4-carbohydrazide (9)
4.2.5. General procedure for obtaining the final triazoles 3a–m
4.3. Pharmacological Activity
4.3.1. Evaluation of AChE Inhibition
4.3.2. Determination of DPPH Scavenging Ability
4.3.3. Cell Cultures
4.3.4. Determination of Neuronal Viability
4.3.5. Determination of Intracellular GSH Levels
4.3.6. Determination of Intracellular ROS Formation
4.3.7. Determination of Neuroprotective Activity Against Aβ42 Oligomers
4.4. In Silico Studies
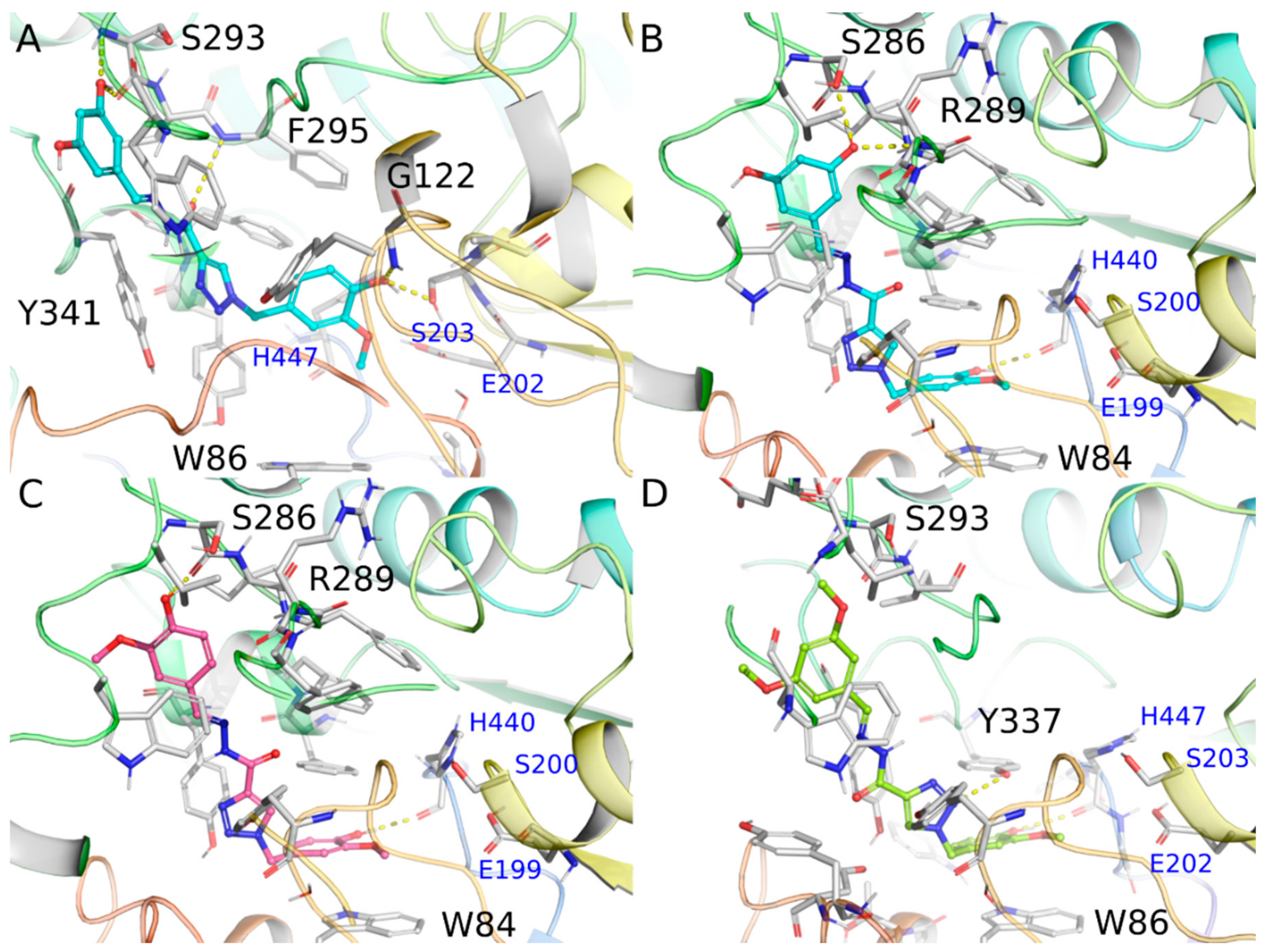
Ensemble Docking Validation
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Viegas, C.; Bolzani, V.D.S.; Barreiro, E.J.; Fraga, C.A.M. New anti-Alzheimer drugs from biodiversity: the role of the natural acetylcholinesterase inhibitors. Mini Rev. Med. Chem. 2005, 5, 915–926. [Google Scholar] [CrossRef] [PubMed]
- Rosini, M.; Simoni, E.; Bartolini, M.; Cavalli, A.; Ceccarini, L.; Pascu, N.; McClymont, D.W.; Tarozzi, A.; Bolognesi, M.L.; Minarini, A.; et al. Inhibition of Acetylcholinesterase, β-Amyloid Aggregation, and NMDA Receptors in Alzheimer’s Disease: A Promising Direction for the Multi-target-Directed Ligands Gold Rush. J. Med. Chem. 2008, 51, 4381–4384. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.M.; Gurung, A.B.; Bhattacharjee, A.; Aguan, K.; Mitra, S. Human serum albumin reduces the potency of acetylcholinesterase inhibitor based drugs for Alzheimer’s disease. Chem. Biol. Interact. 2016, 249, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Barai, P.; Raval, N.; Acharya, S.; Borisa, A.; Bhatt, H.; Acharya, N. Neuroprotective effects of bergenin in Alzheimer’s disease: Investigation through molecular docking, in vitro and in vivo studies. Behav. Brain Res. 2018, 356, 18–40. [Google Scholar] [CrossRef] [PubMed]
- Viegas, F.P.D.; Simões, M.C.R.; da Rocha, M.D.; Castelli, M.R.; Moreira, M.S.; Viegas, C., Jr. Alzheimer’s Disease: Characterization, Evolution and Implications of the Neuroinflammatory Process. Rev. Virtual Química 2011, 3, 286–306. [Google Scholar] [CrossRef]
- Tarozzi, A.; Morroni, F.; Merlicco, A.; Bolondi, C.; Teti, G.; Falconi, M.; Cantelli-Forti, G.; Hrelia, P. Neuroprotective effects of cyanidin 3-O-glucopyranoside on amyloid beta (25–35) oligomer-induced toxicity. Neurosci. Lett. 2010, 473, 72–76. [Google Scholar] [CrossRef]
- Haass, C.; Selkoe, D.J. Soluble protein oligomers in neurodegeneration: lessons from the Alzheimer’s amyloid beta-peptide. Nat. Rev. Mol. Cell Biol. 2007, 8, 101–112. [Google Scholar] [CrossRef]
- Tarozzi, A.; Merlicco, A.; Morroni, F.; Bolondi, C.; Di Iorio, P.; Ciccarelli, R.; Romano, S.; Giuliani, P.; Hrelia, P. Guanosine protects human neuroblastoma cells from oxidative stress and toxicity induced by Amyloid-beta peptide oligomers. J. Biol. Regul. Homeost. Agents 2010, 24, 297–306. [Google Scholar]
- Zhan, Q.; Shi, X.; Wang, T.; Hu, J.; Zhou, J.; Zhou, L.; Wei, S. Design and synthesis of thymine modified phthalocyanine for Aβ protofibrils photodegradation and Aβ peptide aggregation inhibition. Talanta 2019, 191, 27–38. [Google Scholar] [CrossRef]
- Walsh, S.; Aisen, P.S. Inflammatory processes and Alzheimer’s disease. Expert Rev. Neurother. 2004, 4, 793–798. [Google Scholar] [CrossRef]
- Reale, M.; Kamal, M.A.; Velluto, L.; Gambi, D.; Di Nicola, M.; Greig, N.H. Relationship between inflammatory mediators, Abeta levels and ApoE genotype in Alzheimer disease. Curr. Alzheimer Res. 2012, 9, 447–457. [Google Scholar] [CrossRef] [PubMed]
- Dias Viegas, F.P.; de Freitas Silva, M.; Divino da Rocha, M.; Castelli, M.R.; Riquiel, M.M.; Machado, R.P.; Vaz, S.M.; Simões de Lima, L.M.; Mancini, K.C.; Marques de Oliveira, P.C.; et al. Design, synthesis and pharmacological evaluation of N -benzyl-piperidinyl-aryl-acylhydrazone derivatives as donepezil hybrids: Discovery of novel multi-target anti-alzheimer prototype drug candidates. Eur. J. Med. Chem. 2018, 147, 48–65. [Google Scholar] [CrossRef] [PubMed]
- Cavalli, A.; Bolognesi, M.L.; Minarini, A.; Rosini, M.; Tumiatti, V.; Recanatini, M.; Melchiorre, C. Multi-target-Directed Ligands To Combat Neurodegenerative Diseases. J. Med. Chem. 2008, 51, 347–372. [Google Scholar] [CrossRef] [PubMed]
- Dias, K.S.T.; Viegas Jr., C.; Simone Tranches Dias, K.; Viegas, C.; Dias, K.S.T.; Viegas, C. Multi-Target Directed Drugs: A Modern Approach for Design of New Drugs for the treatment of Alzheimer ’ s Disease. Curr. Neuropharmacol. 2014, 12, 239–255. [Google Scholar] [CrossRef] [PubMed]
- Villaflores, O.B.; Chen, Y.J.; Chen, C.P.; Yeh, J.M.; Wu, T.Y. Curcuminoids and resveratrol as anti-Alzheimer agents. Taiwan J. Obstet. Gynecol. 2012, 51, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Repossi, G.; Dain, A.; Tarres, M.C.; Das, U.N.; Eynard, A.R. Beneficial action of resveratrol: How and why? Nutrition 2015, 32, 1–5. [Google Scholar] [CrossRef]
- Li, S.Y.; Wang, X.B.; Kong, L.Y. Design, synthesis and biological evaluation of imine resveratrol derivatives as multi-targeted agents against Alzheimer’s disease. Eur. J. Med. Chem. 2014, 71, 36–45. [Google Scholar] [CrossRef]
- Yin, W.; Li, Y. Curcumin Upregulate Expression of HO-1 and Nrf-2 in SHSY5Y Cells. In Proceedings of the 2010 4th International Conference on Bioinformatics and Biomedical Engineering, Chengdu, China, 10–12 June 2010; IEEE: Piscataway, NJ, USA, 2010; pp. 1–4. [Google Scholar]
- De Freitas Silva, M.; Coelho, L.F.; Guirelli, I.M.; Pereira, R.M.; Ferreira-Silva, G.; Graravelli, G.Y.; Horvath, R.D.O.; Caixeta, E.S.; Ionta, M.; Viegas, C. Synthetic resveratrol-curcumin hybrid derivative inhibits mitosis progression in estrogen positive MCF-7 breast cancer cells. Toxicol. Vitr. 2018, 50, 75–85. [Google Scholar] [CrossRef]
- Cao, Z.; Yang, J.; Xu, R.; Song, Q.; Zhang, X.; Liu, H.; Qiang, X.; Li, Y.; Tan, Z.; Deng, Y. Design, synthesis and evaluation of 4′-OH-flurbiprofen-chalcone hybrids as potential multifunctional agents for Alzheimer’s disease treatment. Bioorg. Med. Chem. 2018, 26, 1102–1115. [Google Scholar] [CrossRef]
- Zheng, Y.; Qiang, X.; Xu, R.; Song, Q.; Tian, C.; Liu, H.; Li, W.; Tan, Z.; Deng, Y. Design, synthesis and evaluation of pterostilbene β-amino alcohol derivatives as multifunctional agents for Alzheimer’s disease treatment. Bioorg. Chem. 2018, 78, 298–306. [Google Scholar] [CrossRef]
- Setamdideh, D.; Zeynizadeh, B. Mild and Convenient Method for Reduction of Carbonyl Compounds with the NaBH 4/Charcoal System in Wet THF. Z. Naturforsch. B 2006, 61, 1275–1281. [Google Scholar] [CrossRef]
- Martinu, T.; Dailey, W.P. Facile One-Pot Preparation of 3-Chloro-2-(chloromethyl)propene and an ab Initio Study of the Deamination Reaction of Nitrosoaziridine. J. Org. Chem. 2000, 65, 6784–6786. [Google Scholar] [CrossRef] [PubMed]
- Bai, S.; Li, S.; Xu, J.; Peng, X.; Sai, K.; Chu, W.; Tu, Z.; Zeng, C.; Mach, R.H. Synthesis and Structure–Activity Relationship Studies of Conformationally Flexible Tetrahydroisoquinolinyl Triazole Carboxamide and Triazole Substituted Benzamide Analogues as σ 2 Receptor Ligands. J. Med. Chem. 2014, 57, 4239–4251. [Google Scholar] [CrossRef] [PubMed]
- Romeiro, N.C.; Aguirre, G.; Hernández, P.; González, M.; Cerecetto, H.; Aldana, I.; Pérez-Silanes, S.; Monge, A.; Barreiro, E.J.; Lima, L.M. Synthesis, trypanocidal activity and docking studies of novel quinoxaline-N-acylhydrazones, designed as cruzain inhibitors candidates. Bioorg. Med. Chem. 2009, 17, 641–652. [Google Scholar] [CrossRef] [PubMed]
- Dias, K.S.T.; de Paula, C.T.; dos Santos, T.; Souza, I.N.O.; Boni, M.S.; Guimarães, M.J.R.; da Silva, F.M.R.; Castro, N.G.; Neves, G.A.; Veloso, C.C.; et al. Design, synthesis and evaluation of novel feruloyl-donepezil hybrids as potential multitarget drugs for the treatment of Alzheimer’s disease. Eur. J. Med. Chem. 2017, 130, 440–457. [Google Scholar] [CrossRef]
- Sugimoto, H.; Yamanish, Y.; Iimura, Y.; Kawakami, Y. Donepezil Hydrochloride (E2020) and Other Acetylcholinesterase Inhibitors. Curr. Med. Chem. 2000, 7, 303–339. [Google Scholar] [CrossRef]
- Ripoll, D.R.; Faerman, C.H.; Axelsen, P.H.; Silman, I.; Sussman, J.L. An electrostatic mechanism for substrate guidance down the aromatic gorge of acetylcholinesterase. Proc. Natl. Acad. Sci. USA 1993, 90, 5128–5132. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Phaniendra, A.; Jestadi, D.B.; Periyasamy, L. Free Radicals: Properties, Sources, Targets, and Their Implication in Various Diseases. Indian J. Clin. Biochem. 2015, 30, 11–26. [Google Scholar] [CrossRef]
- Andersen, J.K. Oxidative stress in neurodegeneration: Cause or consequence? Nat. Rev. Neurosci. 2004, 10, 18. [Google Scholar] [CrossRef]
- Awasthi, M.; Upadhyay, A.K.; Singh, S.; Pandey, V.P.; Dwivedi, U.N. Terpenoids as promising therapeutic molecules against Alzheimer’s disease: amyloid beta- and acetylcholinesterase-directed pharmacokinetic and molecular docking analyses. Mol. Simul. 2018, 44, 1–11. [Google Scholar] [CrossRef]
- Perrin, D.D.; Armarengo, W.L.F. Purification of Laboratory Chemicals, 4th ed.; Butterworth-Heinemann: Canberra, Australia, 1997. [Google Scholar]
- Ellman, G.L.; Courtney, K.D.; Andres, V., Jr.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Castro, N.G.; Costa, R.S.; Pimentel, L.S.B.; Danuello, A.; Romeiro, N.C.; Viegas, C.; Barreiro, E.J.; Fraga, C.A.M.; Bolzani, V.S.; Rocha, M.S. CNS-selective noncompetitive cholinesterase inhibitors derived from the natural piperidine alkaloid (-)-spectaline. Eur. J. Pharmacol. 2008, 580, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Lemes, L.F.N.; de Andrade Ramos, G.; de Oliveira, A.S.; da Silva, F.M.R.; de Castro Couto, G.; da Silva Boni, M.; Guimarães, M.J.R.; Souza, I.N.O.; Bartolini, M.; Andrisano, V.; et al. Cardanol-derived AChE inhibitors: Towards the development of dual binding derivatives for Alzheimer’s disease. Eur. J. Med. Chem. 2016, 108, 687–700. [Google Scholar] [CrossRef] [PubMed]
- Gontijo, V.S.; De Souza, T.C.; Rosa, I.A.; Soares, M.G.; Da Silva, M.A.; Vilegas, W.; Viegas, C.; Dos Santos, M.H. Isolation and evaluation of the antioxidant activity of phenolic constituents of the Garcinia brasiliensis epicarp. Food Chem. 2012, 132, 1230–1235. [Google Scholar] [CrossRef] [PubMed]
- Rampa, A.; Tarozzi, A.; Mancini, F.; Pruccoli, L.; Di Martino, R.M.C.; Gobbi, S.; Bisi, A.; De Simone, A.; Palomba, F.; Zaccheroni, N.; et al. Naturally inspired molecules as multifunctional agents for Alzheimer’s disease treatment. Molecules 2016, 21, 643. [Google Scholar] [CrossRef] [PubMed]
- Rampa, A.; Montanari, S.; Pruccoli, L.; Bartolini, M.; Falchi, F.; Feoli, A.; Cavalli, A.; Belluti, F.; Gobbi, S.; Tarozzi, A.; et al. Chalcone-based carbamates for Alzheimer’s disease treatment. Future Med. Chem. 2017, 9, 749–764. [Google Scholar] [CrossRef] [PubMed]
- Tarozzi, A.; Bartolini, M.; Piazzi, L.; Valgimigli, L.; Amorati, R.; Bolondi, C.; Djemil, A.; Mancini, F.; Andrisano, V.; Rampa, A. From the dual function lead AP2238 to AP2469, a multi-target-directed ligand for the treatment of Alzheimer’s disease. Pharmacol. Res. Perspect. 2014, 2, 1–14. [Google Scholar] [CrossRef]
- Johnson, G.; Moore, S.W. The peripheral anionic site of acetylcholinesterase: structure, functions and potential role in rational drug design. Curr. Pharm. Des. 2006, 12, 217–225. [Google Scholar] [CrossRef]
- Ceschi, M.A.; da Costa, J.S.; Lopes, J.P.B.; Câmara, V.S.; Campo, L.F.; de Amorim Borges, A.C.; Gonçalves, C.A.S.; de Souza, D.F.; Konrath, E.L.; Karl, A.L.M.; et al. Novel series of tacrine-tianeptine hybrids: Synthesis, cholinesterase inhibitory activity, S100B secretion and a molecular modeling approach. Eur. J. Med. Chem. 2016, 121, 758–772. [Google Scholar] [CrossRef]
- Lopes, J.P.B.; Da Costa, J.S.; Ceschi, M.A.; Gonçalves, C.A.S.; Konrath, E.L.; Karl, L.M.; Guedes, I.A.; Dardenne, L.E. Chiral bistacrine analogues: Synthesis, cholinesterase inhibitory activity and a molecular modeling approach. J. Braz. Chem. Soc. 2017, 28, 2218–2228. [Google Scholar] [CrossRef]
- Lopes, J.P.B.; Silva, L.; Ceschi, M.A.; Lüdtke, D.S.; Zimmer, A.R.; Ruaro, T.C.; Dantas, R.F.; de Salles, C.M.C.; Silva, F.P., Jr.; Senger, M.R.; et al. Synthesis of new lophine–carbohydrate hybrids as cholinesterase inhibitors: cytotoxicity evaluation and molecular modeling. Medchemcomm 2019, 10, 2089–2101. [Google Scholar] [CrossRef]
- Cheung, J.; Rudolph, M.J.; Burshteyn, F.; Cassidy, M.S.; Gary, E.N.; Love, J.; Franklin, M.C.; Height, J.J. Structures of human acetylcholinesterase in complex with pharmacologically important ligands. J. Med. Chem. 2012, 55, 10282–10286. [Google Scholar] [CrossRef] [PubMed]
- Craig, I.R.; Essex, J.W.; Spiegel, K. Ensemble docking into multiple crystallographically derived protein structures: An evaluation based on the statistical analysis of enrichments. J. Chem. Inf. Model. 2010, 50, 511–524. [Google Scholar] [CrossRef] [PubMed]
- Guedes, I.A.; de Magalhães, C.S.; Dardenne, L.E. Receptor-ligand molecular docking. Biophys. Rev. 2014, 6, 75–87. [Google Scholar] [CrossRef]
- Guedes, I.A.; Pereira, F.S.S.; Dardenne, L.E. Empirical scoring functions for structure-based virtual screening: Applications, critical aspects, and challenges. Front. Pharmacol. 2018, 9, 1–18. [Google Scholar] [CrossRef]
- Haviv, H.; Wong, D.M.; Greenblatt, H.M.; Carlier, P.R.; Pang, Y.P.; Silman, I.; Sussman, J.L. Crystal packing mediates enantioselective ligand recognition at the peripheral site of acetylcholinesterase. J. Am. Chem. Soc. 2005, 127, 11029–11036. [Google Scholar] [CrossRef]
- Rydberg, E.H.; Brumshtein, B.; Greenblatt, H.M.; Wong, D.M.; Shaya, D.; Williams, L.D.; Carlier, P.R.; Pang, Y.P.; Silman, I.; Sussman, J.L. Complexes of Alkylene-linked tacrine dimers with Torpedo californica acetylcholinesterase: Binding of bis(5)-tacrine produces a dramatic rearrangement in the active-site gorge. J. Med. Chem. 2006, 49, 5491–5500. [Google Scholar] [CrossRef]
- Bourne, Y.; Kolb, H.C.; Radić, Z.; Sharpless, K.B.; Taylor, P.; Marchot, P. Freeze-frame inhibitor captures acetylcholinesterase in a unique conformation. Proc. Natl. Acad. Sci. USA 2004, 101, 1449–1454. [Google Scholar] [CrossRef]
- Madhavi Sastry, G.; Adzhigirey, M.; Day, T.; Annabhimoju, R.; Sherman, W. Protein and ligand preparation: Parameters, protocols, and influence on virtual screening enrichments. J. Comput. Aided. Mol. Des. 2013, 27, 221–234. [Google Scholar] [CrossRef]
- Greenwood, J.R.; Calkins, D.; Sullivan, A.P.; Shelley, J.C. Towards the comprehensive, rapid, and accurate prediction of the favorable tautomeric states of drug-like molecules in aqueous solution. J. Comput. Aided. Mol. Des. 2010, 24, 591–604. [Google Scholar] [CrossRef] [PubMed]
- Shelley, J.C.; Cholleti, A.; Frye, L.L.; Greenwood, J.R.; Timlin, M.R.; Uchimaya, M. Epik: A software program for pKa prediction and protonation state generation for drug-like molecules. J. Comput. Aided. Mol. Des. 2007, 21, 681–691. [Google Scholar] [CrossRef] [PubMed]
- Friesner, R.A.; Banks, J.L.; Murphy, R.B.; Halgren, T.A.; Klicic, J.J.; Mainz, D.T.; Repasky, M.P.; Knoll, E.H.; Shelley, M.; Perry, J.K.; et al. Glide: A New Approach for Rapid, Accurate Docking and Scoring. 1. Method and Assessment of Docking Accuracy. J. Med. Chem. 2004, 47, 1739–1749. [Google Scholar] [CrossRef]
- Friesner, R.A.; Murphy, R.B.; Repasky, M.P.; Frye, L.L.; Greenwood, J.R.; Halgren, T.A.; Sanschagrin, P.C.; Mainz, D.T. Extra precision glide: Docking and scoring incorporating a model of hydrophobic enclosure for protein-ligand complexes. J. Med. Chem. 2006, 49, 6177–6196. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |

| Compounds | Ensemble Docking | Top-Scored | ||||
|---|---|---|---|---|---|---|
| 1ZGC | 1Q84 | 2CKM | 4EY7 | Score | PDB ID | |
| 3b | −10.67 | −9.63 | −8.19 | −10.69 | −10.69/−10.67 | 4EY7/1ZGC |
| 3d | −8.79 | −9.15 | −8.47 | −8.83 | −9.15 | 1Q84 |
| 3e | −10.20 | −9.39 | −8.10 | −8.78 | −10.20 | 1ZGC |
| Donepezil | −11.14 | −10.06 | −12.57 | −17.94 | −17.94 | 4EY7 |
| Compounds | QPlogP o/w | HBA | HBD | PSA | % HOA | QPlogS | QPPCaco | QPlogBB |
|---|---|---|---|---|---|---|---|---|
| 3a | 1.73 | 7.3 | 3 | 124.8 | 72.28 | -3.595 | 92.7 | −2.09 |
| 3b | 1.07 | 8.0 | 4 | 147.3 | 61.12 | -3.282 | 36.2 | −2.57 |
| 3c | 2.48 | 7.3 | 2 | 111.2 | 85.85 | -3.911 | 303.7 | −1.56 |
| 3d | 2.54 | 8.0 | 2 | 120.1 | 86.24 | -4.056 | 303.7 | −1.65 |
| 3e | 1.79 | 8.0 | 3 | 133.0 | 73.14 | -3.708 | 98.7 | −2.15 |
| 3f | 2.45 | 6.5 | 2 | 102.4 | 85.70 | -3.817 | 303.2 | −1.48 |
| 3g | 3.58 | 7.5 | 2 | 104.4 | 92.35 | -5.709 | 303.2 | −1.63 |
| 3h | 2.92 | 6.5 | 2 | 102.4 | 88.48 | -4.519 | 303.2 | −1.34 |
| 3i | 2.84 | 6.5 | 2 | 102.4 | 87.96 | -4.287 | 303.2 | −1.34 |
| 3j | 2.68 | 6.5 | 2 | 100.7 | 88.97 | -3.839 | 388.8 | −1.30 |
| 3k | 2.67 | 6.5 | 2 | 102.4 | 86.99 | -4.154 | 303.2 | −1.38 |
| 3l | 2.47 | 6.5 | 2 | 102.1 | 85.90 | -3.837 | 305.7 | −1.46 |
| 3m | 2.62 | 6.5 | 2 | 102.4 | 86.73 | -4.045 | 303.2 | −1.39 |
| Compound | DPPH EC50 (mg/L) | Direct Antioxidant Activity in SH-SY5Y Cells b | Indirect Antioxidant Activity in SH-SY5Y Cells c |
|---|---|---|---|
| 3a | >200 | In d | 20.58 |
| 3b | >200 | In | 14.07 |
| 3c | >200 | - | - |
| 3d | >200 | - | - |
| 3e | 30.44 | 29.79 | 35.16 |
| 3f | >200 | - | - |
| 3g | 81.51 | - | - |
| 3h | 131.5 | - | - |
| 3i | 190.9 | - | - |
| 3j | >200 | - | - |
| 3k | 59.00 | - | - |
| 3l | 138.5 | - | - |
| 3m | 62.65 | - | - |
| Ascorbic acid | 14.92 | - | - |
| Trolox | 27.76 | - | - |
| Water | 1ZGC | 1Q84 | 2CKM | 4EY7 |
|---|---|---|---|---|
| Wat1 | 1468 | 1708 | 2062 | 729 |
| Wat2 | 1481 | 1755 | 2054 | 737 |
| Wat3 | 1489 | 1715 | 2061 | 722 |
| Wat4 | 1531 | 1735 | 2035 | 731 |
| Reference Ligand | 1ZGC | 1Q84 | 2CKM | 4EY7 | ||||
|---|---|---|---|---|---|---|---|---|
| Score | RMSD | Score | RMSD | Score | RMSD | Score | RMSD | |
| 1ZGC | −18.873 | 1.411 | −18.553 | 1.515 | −18.989 | 1.618 | −17.273 | 12.382 |
| 1Q84 | −18.246 | 1.439 | −19.085 | 0.833 | −12.208 | 4.773 | −11.546 | 6.852 |
| 2CKM | −17.954 | 4.255 | −16.969 | 3.608 | −20.525 | 1.234 | −13.255 | 5.905 |
| 4EY7 | −11.136 | 2.436 | −10.064 | 3.250 | −12.575 | 4.235 | −17.941 | 0.594 |
| Success Rate | 50% | 50% | 50% | 25% | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Freitas Silva, M.; Tardelli Lima, E.; Pruccoli, L.; Castro, N.G.; Guimarães, M.J.R.; da Silva, F.M.R.; Fonseca Nadur, N.; de Azevedo, L.L.; Kümmerle, A.E.; Guedes, I.A.; et al. Design, Synthesis and Biological Evaluation of Novel Triazole N-acylhydrazone Hybrids for Alzheimer’s Disease. Molecules 2020, 25, 3165. https://doi.org/10.3390/molecules25143165
de Freitas Silva M, Tardelli Lima E, Pruccoli L, Castro NG, Guimarães MJR, da Silva FMR, Fonseca Nadur N, de Azevedo LL, Kümmerle AE, Guedes IA, et al. Design, Synthesis and Biological Evaluation of Novel Triazole N-acylhydrazone Hybrids for Alzheimer’s Disease. Molecules. 2020; 25(14):3165. https://doi.org/10.3390/molecules25143165
Chicago/Turabian Stylede Freitas Silva, Matheus, Ellen Tardelli Lima, Letizia Pruccoli, Newton G. Castro, Marcos Jorge R. Guimarães, Fernanda M. R. da Silva, Nathalia Fonseca Nadur, Luciana Luiz de Azevedo, Arthur Eugen Kümmerle, Isabella Alvim Guedes, and et al. 2020. "Design, Synthesis and Biological Evaluation of Novel Triazole N-acylhydrazone Hybrids for Alzheimer’s Disease" Molecules 25, no. 14: 3165. https://doi.org/10.3390/molecules25143165
APA Stylede Freitas Silva, M., Tardelli Lima, E., Pruccoli, L., Castro, N. G., Guimarães, M. J. R., da Silva, F. M. R., Fonseca Nadur, N., de Azevedo, L. L., Kümmerle, A. E., Guedes, I. A., Dardenne, L. E., Gontijo, V. S., Tarozzi, A., & Viegas, C. (2020). Design, Synthesis and Biological Evaluation of Novel Triazole N-acylhydrazone Hybrids for Alzheimer’s Disease. Molecules, 25(14), 3165. https://doi.org/10.3390/molecules25143165








