Second-Generation Pharmacological Chaperones: Beyond Inhibitors
Abstract
1. Introduction
2. Second-Generation Pharmacological Chaperones against LSDs
2.1. Gaucher Disease
2.2. Fabry Disease
2.3. Pompe Disease
2.4. Krabbe Disease
3. Non-Inhibitory Pharmacological Chaperones for Miscellaneous Conformational Diseases
3.1. Phenylketonuria
3.2. Congenital Erythropoietic Porphyria
3.3. Methylmalonic Aciduria (MMA) Cdlb Type
3.4. Phosphomannomutase 2 Deficiency
4. Conclusions
Funding
Conflicts of Interest
References
- Valastyan, J.S.; Lindquist, S. Mechanisms of protein-folding diseases at a glance. Dis. Model. Mech. 2014, 7, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Labbadia, J.; Morimoto, R.I. The biology of proteostasis in aging and disease. Annu. Rev. Biochem. 2015, 84, 435–464. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.X.; Conn, P.M. Pharmacoperones as novel therapeutics for diverse protein conformational diseases. Physiol. Rev. 2018, 98, 697–725. [Google Scholar] [CrossRef] [PubMed]
- Yerbury, J.J.; Stewart, E.M.; Wyatt, A.R.; Wilson, M.R. Quality control of protein folding in extracellular space. EMBO Rep. 2005, 6, 1131–1136. [Google Scholar] [CrossRef]
- Muntau, A.C.; Leandro, J.; Staudigl, M.; Mayer, F.; Gersting, S.W. Innovative strategies to treat protein misfolding in inborn errors of metabolism: Pharmacological chaperones and proteostasis regulators. J. Inherit. Metab. Dis. 2014, 37, 505–523. [Google Scholar] [CrossRef] [PubMed]
- Liguori, L.; Monticelli, M.; Allocca, M.; Hay Mele, B.; Lukas, J.; Cubellis, M.V.; Andreotti, G. Pharmacological chaperones: A therapeutic approach for diseases caused by destabilizing missense mutations. Int. J. Mol. Sci. 2020, 21, 489. [Google Scholar] [CrossRef]
- Bernier, V.; Lagacé, M.; Bichet, D.G.; Bouvier, M. Pharmacological chaperones: Potential treatment for conformational diseases. Trends Endocrinol. Metab. 2004, 15, 222–228. [Google Scholar] [CrossRef]
- Soto, C. Unfolding the role of protein misfolding in neurodegenerative diseases. Nat. Rev. Neurosci. 2003, 4, 49–60. [Google Scholar] [CrossRef]
- Convertino, M.; Das, J.; Dokholyan, N.V. Pharmacological chaperones: Design and development of new therapeutic strategies for the treatment of conformational diseases. ACS Chem. Biol. 2016, 11, 1471–1489. [Google Scholar] [CrossRef]
- Pereira, D.M.; Valentão, P.; Andrade, P.B. Tuning protein folding in lysosomal storage diseases: The chemistry behind pharmacological chaperones. Chem. Sci. 2018, 9, 1740–1752. [Google Scholar] [CrossRef]
- Zeitlin, P.L.; Diener-West, M.; Rubenstein, R.C.; Boyle, M.P.; Lee, C.K.K.; Brass-Ernst, L. Evidence of CFTR function in cystic fibrosis after systemic administration of 4-phenylbutyrate. Mol. Ther. 2002, 6, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.-Q. A contradictory treatment for lysosomal storage disorders: Inhibitors enhance mutant enzyme activity. Trends Pharmacol. Sci. 2003, 24, 355–360. [Google Scholar] [CrossRef]
- Fan, J.-Q. A counterintuitive approach to treat enzyme deficiencies: Use of enzyme inhibitors for restoring mutant enzyme activity. Biol. Chem. 2008, 389, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Beerepoot, P.; Nazari, R.; Salahpour, A. Pharmacological chaperone approaches for rescuing GPCR mutants: Current state, challenges, and screening strategies. Pharmacol. Res. 2017, 117, 242–251. [Google Scholar] [CrossRef]
- Vauthier, V.; Housset, C.; Falguières, T. Targeted pharmacotherapies for defective ABC transporters. Biochem. Pharmacol. 2017, 136, 1–11. [Google Scholar] [CrossRef]
- Moran, N. FDA approves Galafold, a triumph for Amicus. Nat. Biotechnol. 2018, 36, 913. [Google Scholar] [CrossRef]
- Parenti, G.; Andria, G.; Valenzano, K.J. Pharmacological chaperone therapy: preclinical development, clinical translation, and prospects for the treatment of lysosomal storage disorders. Mol. Ther. 2015, 23, 1138–1148. [Google Scholar] [CrossRef]
- Losada Díaz, J.C.; Cepeda del Castillo, J.; Rodriguez-López, E.A.; Alméciga-Díaz, C.J. Advances in the Development of pharmacological chaperones for the mucopolysaccharidoses. Int. J. Mol. Sci. 2019, 21, 232. [Google Scholar] [CrossRef]
- Hammarström, P.; Wiseman, R.L.; Powers, E.T.; Kelly, J.W. Prevention of transthyretin amyloid disease by changing protein misfolding energetics. Science 2003, 299, 713–716. [Google Scholar] [CrossRef]
- Graziano, A.C.E.; Pannuzzo, G.; Avola, R.; Cardile, V. Chaperones as potential therapeutics for Krabbe disease. J. Neurosci. Res. 2016, 94, 1220–1230. [Google Scholar] [CrossRef]
- Newton, C.L.; Anderson, R.C. Pharmacoperones for Misfolded Gonadotropin Receptors. In Targeting Trafficking in Drug Development; Ulloa-Aguirre, A., Tao, Y.-X., Eds.; Springer: Cham, Switzerland, 2017; Volume 245, pp. 111–134. ISBN 978-3-319-74163-5. [Google Scholar]
- Hill, C.H.; Viuff, A.H.; Spratley, S.J.; Salamone, S.; Christensen, S.H.; Read, R.J.; Moriarty, N.W.; Jensen, H.H.; Deane, J.E. Azasugar inhibitors as pharmacological chaperones for Krabbe disease. Chem. Sci. 2015, 6, 3075–3086. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.H.; Lim, H.-S. Screening methods for identifying pharmacological chaperones. Mol. Biosyst. 2017, 13, 638–647. [Google Scholar] [CrossRef]
- Winchester, B.; Vellodi, A.; Young, E. The molecular basis of lysosomal storage diseases and their treatment. Biochem. Soc. Trans. 2000, 28, 5. [Google Scholar] [CrossRef] [PubMed]
- Parkinson-Lawrence, E.J.; Shandala, T.; Prodoehl, M.; Plew, R.; Borlace, G.N.; Brooks, D.A. Lysosomal storage disease: Revealing lysosomal function and physiology. Physiology 2010, 25, 102–115. [Google Scholar] [CrossRef] [PubMed]
- Ballabio, A.; Gieselmann, V. Lysosomal disorders: From storage to cellular damage. Biochim. Biophys. Acta BBA Mol. Cell Res. 2009, 1793, 684–696. [Google Scholar] [CrossRef] [PubMed]
- Kaminsky, P.; Lidove, O. Stratégies thérapeutiques actuelles dans les maladies lysosomales. Presse Médicale 2014, 43, 1174–1184. [Google Scholar] [CrossRef] [PubMed]
- Schueler, U.H.; Kolter, T.; Kaneski, C.R.; Zirzow, G.C.; Sandhoff, K.; Brady, R.O. Correlation between enzyme activity and substrate storage in a cell culture model system for Gaucher disease. J. Inherit. Metab. Dis. 2004, 27, 649–658. [Google Scholar] [CrossRef]
- Leinekugel, P.; Michel, S.; Conzelmann, E.; Sandhoff, K. Quantitative correlation between the residual activity of -hexosaminidase A and arylsulfatase A and the severity of the resulting lysosomal storage disease. Hum. Genet. 1992, 88, 513–523. [Google Scholar] [CrossRef]
- Luzi, P.; Rafi, M.A.; Wenger, D.A. Multiple mutations in the GALC gene in a patient with adult-onset krabbe disease. Ann. Neurol. 1996, 40, 116–119. [Google Scholar] [CrossRef]
- Desnick, R.J.; Wasserstein, M.P.; Banikazemi, M. Fabry disease (alpha-galactosidase A deficiency): Renal involvement and enzyme replacement therapy. Contrib. Nephrol. 2001, 174–192. [Google Scholar] [CrossRef]
- Manganelli, F.; Ruggiero, L. Clinical features of Pompe disease. Acta Myol. 2013, 32, 82–84. [Google Scholar] [PubMed]
- Nalysnyk, L.; Rotella, P.; Simeone, J.C.; Hamed, A.; Weinreb, N. Gaucher disease epidemiology and natural history: A comprehensive review of the literature. Hematology 2017, 22, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Stirnemann, J.; Belmatoug, N.; Camou, F.; Serratrice, C.; Froissart, R.; Caillaud, C.; Levade, T.; Astudillo, L.; Serratrice, J.; Brassier, A.; et al. A Review of Gaucher Disease Pathophysiology, Clinical Presentation and Treatments. Int. J. Mol. Sci. 2017, 18, 441. [Google Scholar] [CrossRef]
- Charrow, J.; Andersson, H.C.; Kaplan, P.; Kolodny, E.H.; Mistry, P.; Pastores, G.; Rosenbloom, B.E.; Scott, C.R.; Wappner, R.S.; Weinreb, N.J.; et al. The gaucher registry: Demographics and disease characteristics of 1698 patients with gaucher disease. Arch. Intern. Med. 2000, 160, 2835–2843. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Fernández, E.M.; García Fernández, J.M.; Ortiz Mellet, C. Glycomimetic-based pharmacological chaperones for lysosomal storage disorders: Lessons from Gaucher, GM1-gangliosidosis and Fabry diseases. Chem. Commun. 2016, 52, 5497–5515. [Google Scholar] [CrossRef] [PubMed]
- Benito, J.M.; García Fernández, J.M.; Mellet, C.O. Pharmacological chaperone therapy for Gaucher disease: A patent review. Expert Opin. Ther. Pat. 2011, 21, 885–903. [Google Scholar] [CrossRef]
- Maegawa, G.H.B.; Tropak, M.B.; Buttner, J.D.; Rigat, B.A.; Fuller, M.; Pandit, D.; Tang, L.; Kornhaber, G.J.; Hamuro, Y.; Clarke, J.T.R.; et al. Identification and characterization of ambroxol as an enzyme enhancement agent for Gaucher disease. J. Biol. Chem. 2009, 284, 23502–23516. [Google Scholar] [CrossRef]
- Lukas, J.; Pockrandt, A.-M.; Seemann, S.; Sharif, M.; Runge, F.; Pohlers, S.; Zheng, C.; Gläser, A.; Beller, M.; Rolfs, A.; et al. Enzyme enhancers for the treatment of Fabry and Pompe disease. Mol. Ther. 2015, 23, 456–464. [Google Scholar] [CrossRef]
- Delamarre, A.; Meissner, W.G. Epidemiology, environmental risk factors and genetics of Parkinson’s disease. Presse Médicale 2017, 46, 175–181. [Google Scholar] [CrossRef]
- Avenali, M.; Blandini, F.; Cerri, S. Glucocerebrosidase Defects as a major risk factor for Parkinson’s disease. Front. Aging Neurosci. 2020, 12, 97. [Google Scholar] [CrossRef]
- Li, Y.; Li, P.; Liang, H.; Zhao, Z.; Hashimoto, M.; Wei, J. Gaucher-associated parkinsonism. Cell. Mol. Neurobiol. 2015, 35, 755–761. [Google Scholar] [CrossRef][Green Version]
- Butters, T.D.; Dwek, R.A.; Platt, F.M. Imino sugar inhibitors for treating the lysosomal glycosphingolipidoses. Glycobiology 2005, 15, 43R–52R. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Padia, J.; Urban, D.J.; Jadhav, A.; Goker-Alpan, O.; Simeonov, A.; Goldin, E.; Auld, D.; LaMarca, M.E.; Inglese, J.; et al. Three classes of glucocerebrosidase inhibitors identified by quantitative high-throughput screening are chaperone leads for Gaucher disease. Proc. Natl. Acad. Sci. USA 2007, 104, 13192–13197. [Google Scholar] [CrossRef]
- Urban, D.; Zheng, W.; Goker-Alpan, O.; Jadhav, A.; LaMarca, M.; Inglese, J.; Sidransky, E.; Austin, C. Optimization and validation of two miniaturized glucocerebrosidase enzyme assays for high throughput screening. Comb. Chem. High. Throughput Screen. 2008, 11, 817–824. [Google Scholar] [CrossRef]
- Marugan, J.J.; Zheng, W.; Motabar, O.; Southall, N.; Goldin, E.; Westbroek, W.; Stubblefield, B.K.; Sidransky, E.; Aungst, R.A.; Lea, W.A.; et al. Evaluation of quinazoline analogues as glucocerebrosidase inhibitors with chaperone activity. J. Med. Chem. 2011, 54, 1033–1058. [Google Scholar] [CrossRef]
- Marugan, J.J.; Huang, W.; Motabar, O.; Zheng, W.; Xiao, J.; Patnaik, S.; Southall, N.; Westbroek, W.; Lea, W.A.; Simeonov, A.; et al. Non-iminosugar glucocerebrosidase small molecule chaperones. Med. Chem Commun 2012, 3, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Goldin, E.; Zheng, W.; Motabar, O.; Southall, N.; Choi, J.H.; Marugan, J.; Austin, C.P.; Sidransky, E. High throughput screening for small molecule therapy for gaucher disease using patient tissue as the source of mutant glucocerebrosidase. PLoS ONE 2012, 7, e29861. [Google Scholar] [CrossRef][Green Version]
- Rogers, S.; Patnaik, S.; Schoenen, F.; Zheng, W.; Choi, J.; Motabar, O.; Southall, N.; Westbroek, W.; Goldin, E.; Sidransky, E.; et al. Discovery, SAR, and biological evaluation of non-inhibitory chaperones of glucocerebrosidase. In Probe Reports from the NIH Molecular Libraries Program; National Center for Biotechnology Information (US): Bethesda, MD, USA, 2010; Available online: http://www.ncbi.nlm.nih.gov/books/NBK143537/ (accessed on 9 July 2020).
- Aflaki, E.; Goldin, E.; Marugan, J.J.; Patnaik, S.; Rogers, S.A.; Schoenen, F.J.; Sidransky, E.; Southall, N.; Westbroek, W.; Zheng, W. Salicylic acid derivatives useful as glucocerebrosidase activators. U.S. Patent US2015065469A1, 11 October 2015. [Google Scholar]
- Aflaki, E.; Borger, D.K.; Moaven, N.; Stubblefield, B.K.; Rogers, S.A.; Patnaik, S.; Schoenen, F.J.; Westbroek, W.; Zheng, W.; Sullivan, P.; et al. A new glucocerebrosidase chaperone reduces -synuclein and glycolipid levels in ipsc-derived dopaminergic neurons from patients with gaucher disease and parkinsonism. J. Neurosci. 2016, 36, 7441–7452. [Google Scholar] [CrossRef]
- Patnaik, S.; Zheng, W.; Choi, J.H.; Motabar, O.; Southall, N.; Westbroek, W.; Lea, W.A.; Velayati, A.; Goldin, E.; Sidransky, E.; et al. Discovery, structure–activity relationship, and biological evaluation of noninhibitory small molecule chaperones of glucocerebrosidase. J. Med. Chem. 2012, 55, 5734–5748. [Google Scholar] [CrossRef] [PubMed]
- Aflaki, E.; Stubblefield, B.K.; Maniwang, E.; Lopez, G.; Moaven, N.; Goldin, E.; Marugan, J.; Patnaik, S.; Dutra, A.; Southall, N.; et al. Macrophage models of gaucher disease for evaluating disease pathogenesis and candidate drugs. Sci. Transl. Med. 2014, 6, 240ra73. [Google Scholar] [CrossRef]
- Mazzulli, J.R.; Zunke, F.; Tsunemi, T.; Toker, N.J.; Jeon, S.; Burbulla, L.F.; Patnaik, S.; Sidransky, E.; Marugan, J.J.; Sue, C.M.; et al. Activation of β-glucocerebrosidase reduces pathological α-synuclein and restores lysosomal function in Parkinson’s patient midbrain neurons. J. Neurosci. 2016, 36, 7693–7706. [Google Scholar] [CrossRef] [PubMed]
- Skerlj, R.T.; Lansbury, P.T.; Bourque, E.M.J.; Good, A.C. Methods of treatment and combination therapies using Gcase activator heterobicyclic and related compounds. World Patent WO2017192841A1, 2017. [Google Scholar]
- Bourque, E.M.J.; Good, A.C.; Krainc, D.; Lansbury, P.T.; Silverman, R.B.; Skerlj, R.T.; Zheng, J. Substituted pyrrolo[1,2-a]pyrimidines and their use in the treatment of medical disorders. U.S. Patent US2020030331A1, 5 July 2020. [Google Scholar]
- Hilt, D.C.; Heijer, J.; Cullen, V.; Dudgeon, L.; Lansbury, P.; Kruithof, A.; Berendse, H.; de Bie, R.; Bonifati, V.; Boon, A.; et al. Late-breaking abstract LBA8 A Dose Ranging, Placebo-Controlled, 28-Day, Safety and Biomarker Phase 2a Study in GBA-PD Patients with the Selective GCase Activator, LTI-291. In Proceedings of the International Congress of Parkinson’s Disease and Movement Disorders, Nice, France, 22–26 September 2019. [Google Scholar]
- Parkinson’s Therapies Seek to Stem Progression | ALZFORUM. Available online: https://www.alzforum.org/news/conference-coverage/parkinsons-therapies-seek-stem-progression (accessed on 29 May 2020).
- Krainc, D.; Silverman, R.B.; Zheng, J. Substituted 4-methyl-pyrrolo[1,2-a]pyrimidine-8-carboxamide compounds and uses thereof for modulating glucocerebrosidase activity. U.S. Patent US2017002013A1, 2017. [Google Scholar]
- Krainc, D.; Silverman, R.B.; Zheng, J. Pyrrolopyrimidine compounds and uses thereof for modulating Glucocerebrosidase activity. US. Patent 2020. [Google Scholar]
- Tropak, M.B.; Kornhaber, G.J.; Rigat, B.A.; Maegawa, G.H.; Buttner, J.D.; Blanchard, J.E.; Murphy, C.; Tuske, S.J.; Coales, S.J.; Hamuro, Y.; et al. Identification of pharmacological chaperones for gaucher disease and characterization of their effects on β-glucocerebrosidase by hydrogen/deuterium exchange mass spectrometry. ChemBioChem 2008, 9, 2650–2662. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Chen, L.; Schwake, M.; Silverman, R.B.; Krainc, D. Design and synthesis of potent quinazolines as selective β-glucocerebrosidase modulators. J. Med. Chem. 2016, 59, 8508–8520. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Chen, L.; Skinner, O.S.; Ysselstein, D.; Remis, J.; Lansbury, P.; Skerlj, R.; Mrosek, M.; Heunisch, U.; Krapp, S.; et al. β-Glucocerebrosidase modulators promote dimerization of β-glucocerebrosidase and reveal an allosteric binding site. J. Am. Chem. Soc. 2018, 140, 5914–5924. [Google Scholar] [CrossRef]
- Zheng, J.; Jeon, S.; Jiang, W.; Burbulla, L.F.; Ysselstein, D.; Oevel, K.; Krainc, D.; Silverman, R.B. Conversion of quinazoline modulators from inhibitors to activators of β-glucocerebrosidase. J. Med. Chem. 2019, 62, 1218–1230. [Google Scholar] [CrossRef]
- Burbulla, L.F.; Jeon, S.; Zheng, J.; Song, P.; Silverman, R.B.; Krainc, D. A modulator of wild-type glucocerebrosidase improves pathogenic phenotypes in dopaminergic neuronal models of Parkinson’s disease. Sci. Transl. Med. 2019, 11, eaau6870. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.J.; Kanack, A.J.; Dahms, N.M. Progress in the understanding and treatment of Fabry disease. Biochim. Biophys. Acta BBA Gen. Subj. 2020, 1864, 129437. [Google Scholar] [CrossRef]
- Vedder, A.C.; Linthorst, G.E.; Breemen, M.J.v.; Groener, J.E.M.; Bemelman, F.J.; Strijland, A.; Mannens, M.M.a.M.; Aerts, J.M.F.G.; Hollak, C.E.M. The Dutch Fabry cohort: Diversity of clinical manifestations and Gb3 levels. J. Inherit. Metab. Dis. 2007, 30, 68–78. [Google Scholar] [CrossRef]
- Burton, B.K.; Charrow, J.; Hoganson, G.E.; Waggoner, D.; Tinkle, B.; Braddock, S.R.; Schneider, M.; Grange, D.K.; Nash, C.; Shryock, H.; et al. Newborn screening for lysosomal storage disorders in illinois: the initial 15-month experience. J. Pediatr. 2017, 190, 130–135. [Google Scholar] [CrossRef]
- Hwu, W.-L.; Chien, Y.-H.; Lee, N.-C.; Chiang, S.-C.; Dobrovolny, R.; Huang, A.-C.; Yeh, H.-Y.; Chao, M.-C.; Lin, S.-J.; Kitagawa, T.; et al. Newborn screening for Fabry disease in Taiwan reveals a high incidence of the later-onset GLA mutation c.936+919G>A (IVS4+919G>A). Hum. Mutat. 2009, 30, 1397–1405. [Google Scholar] [CrossRef] [PubMed]
- Sirrs, S.; Hollak, C.; Merkel, M.; Sechi, A.; Glamuzina, E.; Janssen, M.C.; Rachmann, R.; Langendonk, J.; Scarpelli, M.; Ben Omran, T.; et al. The frequencies of different inborn errors of metabolism in adult metabolic centres: Report from the SSIEM adult metabolic physicians group. JIMD Rep. 2016, 27, 85–91. [Google Scholar] [CrossRef]
- Garman, S.C.; Garboczi, D.N. The molecular defect leading to fabry disease: structure of human α-galactosidase. J. Mol. Biol. 2004, 337, 319–335. [Google Scholar] [CrossRef] [PubMed]
- Citro, V.; Peña-García, J.; den-Haan, H.; Pérez-Sánchez, H.; Del Prete, R.; Liguori, L.; Cimmaruta, C.; Lukas, J.; Cubellis, M.V.; Andreotti, G. Identification of an allosteric binding site on human lysosomal alpha-galactosidase opens the way to new pharmacological chaperones for fabry disease. PLoS ONE 2016, 11, e0165463. [Google Scholar] [CrossRef]
- Bellotti, A.S.; Andreoli, L.; Ronchi, D.; Bresolin, N.; Comi, G.P.; Corti, S. Molecular approaches for the treatment of pompe disease. Mol. Neurobiol. 2020, 57, 1259–1280. [Google Scholar] [CrossRef]
- Parenti, G.; Andria, G. Pompe disease: From new views on pathophysiology to innovative therapeutic strategies. Curr. Pharm. Biotechnol. 2011, 12, 902–915. [Google Scholar] [CrossRef]
- Parenti, G.; Zuppaldi, A.; Pittis, M.G.; Tuzzi, M.R.; Annunziata, I.; Meroni, G.; Porto, C.; Donaudy, F.; Rossi, B.; Rossi, M.; et al. Pharmacological enhancement of mutated α-glucosidase activity in fibroblasts from patients with pompe disease. Mol. Ther. 2007, 15, 508–514. [Google Scholar] [CrossRef]
- Parenti, G.; Fecarotta, S.; la Marca, G.; Rossi, B.; Ascione, S.; Donati, M.A.; Morandi, L.O.; Ravaglia, S.; Pichiecchio, A.; Ombrone, D.; et al. A chaperone enhances blood α-glucosidase activity in pompe disease patients treated with enzyme replacement therapy. Mol. Ther. 2014, 22, 2004–2012. [Google Scholar] [CrossRef]
- Porto, C.; Cardone, M.; Fontana, F.; Rossi, B.; Tuzzi, M.R.; Tarallo, A.; Barone, M.V.; Andria, G.; Parenti, G. The pharmacological chaperone N-butyldeoxynojirimycin enhances enzyme replacement therapy in pompe disease fibroblasts. Mol. Ther. 2009, 17, 964–971. [Google Scholar] [CrossRef] [PubMed]
- Motabar, O.; Shi, Z.-D.; Goldin, E.; Liu, K.; Southall, N.; Sidransky, E.; Austin, C.P.; Griffiths, G.L.; Zheng, W. A new resorufin-based α-glucosidase assay for high-throughput screening. Anal. Biochem. 2009, 390, 79–84. [Google Scholar] [CrossRef][Green Version]
- Marugan, J.J.; Zheng, W.; Ferrer, M.; Motabar, O.; Southall, N.; Goldin, E.; Westbroek, W.; Sidransky, E. Discovery, SAR, and Biological Evaluation of a Non-Inhibitory Chaperone for Acid Alpha Glucosidase. In Probe Reports from the NIH Molecular Libraries Program; National Center for Biotechnology Information: Bethesda, MD, USA, 2013; Available online: https://www.ncbi.nlm.nih.gov/book01s/NBK153221/ (accessed on 1 June 2020).
- Marugan, J.J.; Zheng, W.; Motabar, O.; Southall, N.; Goldin, E.; Sidransky, E.; Aungst, R.A.; Liu, K.; Sadhukhan, S.K.; Austin, C.P. Evaluation of 2-thioxo-2,3,5,6,7,8-hexahydropyrimido[4,5-d]pyrimidin-4(1H)-one analogues as GAA activators. Eur. J. Med. Chem. 2010, 45, 1880–1897. [Google Scholar] [CrossRef][Green Version]
- Porto, C.; Ferrara, M.C.; Meli, M.; Acampora, E.; Avolio, V.; Rosa, M.; Cobucci-Ponzano, B.; Colombo, G.; Moracci, M.; Andria, G.; et al. Pharmacological enhancement of α-glucosidase by the allosteric chaperone n-acetylcysteine. Mol. Ther. 2012, 20, 2201–2211. [Google Scholar] [CrossRef] [PubMed]
- Roig-Zamboni, V.; Cobucci-Ponzano, B.; Iacono, R.; Ferrara, M.C.; Germany, S.; Bourne, Y.; Parenti, G.; Moracci, M.; Sulzenbacher, G. Structure of human lysosomal acid α-glucosidase–a guide for the treatment of Pompe disease. Nat. Commun. 2017, 8, 1–10. [Google Scholar] [CrossRef]
- D’Alonzo, D.; De Fenza, M.; Porto, C.; Iacono, R.; Huebecker, M.; Cobucci-Ponzano, B.; Priestman, D.A.; Platt, F.; Parenti, G.; Moracci, M.; et al. N-Butyl-l-deoxynojirimycin (l-NBDNJ): Synthesis of an allosteric enhancer of α-glucosidase activity for the treatment of pompe disease. J. Med. Chem. 2017, 60, 9462–9469. [Google Scholar] [CrossRef] [PubMed]
- Won, J.-S.; Singh, A.K.; Singh, I. Biochemical, cell biological, pathological, and therapeutic aspects of Krabbe’s disease. J. Neurosci. Res. 2016, 94, 990–1006. [Google Scholar] [CrossRef] [PubMed]
- Graziano, A.C.E.; Cardile, V. History, genetic, and recent advances on Krabbe disease. Gene 2015, 555, 2–13. [Google Scholar] [CrossRef]
- Orsini, J.J.; Escolar, M.L.; Wasserstein, M.P.; Caggana, M. Krabbe Disease. In GeneReviews®®; Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J., Stephens, K., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993–2020; Available online: https://www.ncbi.nlm.nih.gov/books/NBK1238/ (accessed on 1 June 2020).
- Tappino, B.; Biancheri, R.; Mort, M.; Regis, S.; Corsolini, F.; Rossi, A.; Stroppiano, M.; Lualdi, S.; Fiumara, A.; Bembi, B.; et al. Identification and characterization of 15 novel galc gene mutations causing krabbe disease. Hum. Mutat. 2010, 31, E1894–E1915. [Google Scholar] [CrossRef]
- Escolar, M.L.; West, T.; Dallavecchia, A.; Poe, M.D.; LaPoint, K. Clinical management of Krabbe disease. J. Neurosci. Res. 2016, 94, 1118–1125. [Google Scholar] [CrossRef]
- Wright, M.D.; Poe, M.D.; DeRenzo, A.; Haldal, S.; Escolar, M.L. Developmental outcomes of cord blood transplantation for Krabbe disease. Neurology 2017, 89, 1365–1372. [Google Scholar] [CrossRef]
- Hossain, M.A.; Higaki, K.; Saito, S.; Ohno, K.; Sakuraba, H.; Nanba, E.; Suzuki, Y.; Ozono, K.; Sakai, N. Chaperone therapy for Krabbe disease: Potential for late-onset GALC mutations. J. Hum. Genet. 2015, 60, 539–545. [Google Scholar] [CrossRef]
- Lee, W.C.; Kang, D.; Causevic, E.; Herdt, A.R.; Eckman, E.A.; Eckman, C.B. Molecular characterization of mutations that cause globoid cell leukodystrophy and pharmacological rescue using small molecule chemical chaperones. J. Neurosci. 2010, 30, 5489–5497. [Google Scholar] [CrossRef] [PubMed]
- Berardi, A.S.; Pannuzzo, G.; Graziano, A.; Costantino-Ceccarini, E.; Piomboni, P.; Luddi, A. Pharmacological chaperones increase residual β-galactocerebrosidase activity in fibroblasts from Krabbe patients. Mol. Genet. Metab. 2014, 112, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Spratley, S.J.; Deane, J.E. New therapeutic approaches for Krabbe disease: The potential of pharmacological chaperones. J. Neurosci. Res. 2016, 94, 1203–1219. [Google Scholar] [CrossRef] [PubMed]
- Underhaug, J.; Aubi, O.; Martinez, A. Phenylalanine hydroxylase misfolding and pharmacological chaperones. Curr. Top. Med. Chem. 2012, 12, 2534–2545. [Google Scholar] [CrossRef]
- Muntau, A.C.; Röschinger, W.; Habich, M.; Demmelmair, H.; Hoffmann, B.; Sommerhoff, C.P.; Roscher, A.A. Tetrahydrobiopterin as an alternative treatment for mild phenylketonuria. N. Engl. J. Med. 2002, 347, 2122–2132. [Google Scholar] [CrossRef]
- Hufton, S.E.; Jennings, I.G.; Cotton, R.G.H. Structure and function of the aromatic amino acid hydroxylases. Biochem. J. 1995, 311, 353–366. [Google Scholar] [CrossRef]
- Jaffe, E.K. New protein structures provide an updated understanding of phenylketonuria. Mol. Genet. Metab. 2017, 121, 289–296. [Google Scholar] [CrossRef]
- Flydal, M.I.; Alcorlo-Pagés, M.; Johannessen, F.G.; Martínez-Caballero, S.; Skjærven, L.; Fernandez-Leiro, R.; Martinez, A.; Hermoso, J.A. Structure of full-length human phenylalanine hydroxylase in complex with tetrahydrobiopterin. Proc. Natl. Acad. Sci. USA 2019, 116, 11229–11234. [Google Scholar] [CrossRef]
- Erlandsen, H.; Fusetti, F.; Martinez, A.; Hough, E.; Flatmark, T.; Stevens, R.C. Crystal structure of the catalytic domain of human phenylalanine hydroxylase reveals the structural basis for phenylketonuria. Nat. Struct. Biol. 1997, 4, 995–1000. [Google Scholar] [CrossRef]
- Andreas Andersen, O.; Flatmark, T.; Hough, E. Crystal structure of the ternary complex of the catalytic domain of human phenylalanine hydroxylase with tetrahydrobiopterin and 3-(2-thienyl)-l-alanine, and its implications for the mechanism of catalysis and substrate activation. J. Mol. Biol. 2002, 320, 1095–1108. [Google Scholar] [CrossRef]
- Arturo, E.C.; Gupta, K.; Héroux, A.; Stith, L.; Cross, P.J.; Parker, E.J.; Loll, P.J.; Jaffe, E.K. First structure of full-length mammalian phenylalanine hydroxylase reveals the architecture of an autoinhibited tetramer. Proc. Natl. Acad. Sci. USA 2016, 113, 2394–2399. [Google Scholar] [CrossRef] [PubMed]
- Patel, D.; Kopec, J.; Fitzpatrick, F.; McCorvie, T.J.; Yue, W.W. Structural basis for ligand-dependent dimerization of phenylalanine hydroxylase regulatory domain. Sci. Rep. 2016, 6, 1–10. [Google Scholar] [CrossRef]
- Zhang, S.; Fitzpatrick, P.F. Identification of the allosteric site for phenylalanine in rat phenylalanine hydroxylase. J. Biol. Chem. 2016, 291, 7418–7425. [Google Scholar] [CrossRef] [PubMed]
- Levy, H.L.; Milanowski, A.; Chakrapani, A.; Cleary, M.; Lee, P.; Trefz, F.K.; Whitley, C.B.; Feillet, F.; Feigenbaum, A.S.; Bebchuk, J.D.; et al. Efficacy of sapropterin dihydrochloride (tetrahydrobiopterin, 6R-BH4) for reduction of phenylalanine concentration in patients with phenylketonuria: A phase III randomised placebo-controlled study. Lancet 2007, 370, 504–510. [Google Scholar] [CrossRef]
- Martinez, A.; Calvo, A.C.; Teigen, K.; Pey, A.L. Rescuing proteins of low kinetic stability by chaperones and natural ligands: phenylketonuria, a case study. J. Mol. Cell Biol. 2008, 83, 89–134. [Google Scholar] [CrossRef]
- Blau, N.; Erlandsen, H. The metabolic and molecular bases of tetrahydrobiopterin-responsive phenylalanine hydroxylase deficiency. Mol. Genet. Metab. 2004, 82, 101–111. [Google Scholar] [CrossRef]
- Erlandsen, H.; Pey, A.L.; Gámez, A.; Pérez, B.; Desviat, L.R.; Aguado, C.; Koch, R.; Surendran, S.; Tyring, S.; Matalon, R.; et al. Correction of kinetic and stability defects by tetrahydrobiopterin in phenylketonuria patients with certain phenylalanine hydroxylase mutations. Proc. Natl. Acad. Sci. USA 2004, 101, 16903–16908. [Google Scholar] [CrossRef]
- Pey, A.L.; Ying, M.; Cremades, N.; Velazquez-Campoy, A.; Scherer, T.; Thöny, B.; Sancho, J.; Martinez, A. Identification of pharmacological chaperones as potential therapeutic agents to treat phenylketonuria. J. Clin. Investig. 2008, 118, 2858–2867. [Google Scholar] [CrossRef]
- Santos-Sierra, S.; Kirchmair, J.; Perna, A.M.; Reiß, D.; Kemter, K.; Röschinger, W.; Glossmann, H.; Gersting, S.W.; Muntau, A.C.; Wolber, G.; et al. Novel pharmacological chaperones that correct phenylketonuria in mice. Hum. Mol. Genet. 2012, 21, 1877–1887. [Google Scholar] [CrossRef]
- Erwin, A.L.; Desnick, R.J. Congenital erythropoietic porphyria: Recent advances. Mol. Genet. Metab. 2019, 128, 288–297. [Google Scholar] [CrossRef]
- Phillips, J.D. Heme biosynthesis and the porphyrias. Mol. Genet. Metab. 2019, 128, 164–177. [Google Scholar] [CrossRef]
- Urquiza, P.; Laín, A.; Sanz-Parra, A.; Moreno, J.; Bernardo-Seisdedos, G.; Dubus, P.; González, E.; Gutiérrez-de-Juan, V.; García, S.; Eraña, H.; et al. Repurposing ciclopirox as a pharmacological chaperone in a model of congenital erythropoietic porphyria. Sci. Transl. Med. 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Almási, T.; Guey, L.T.; Lukacs, C.; Csetneki, K.; Vokó, Z.; Zelei, T. Systematic literature review and meta-analysis on the epidemiology of methylmalonic acidemia (MMA) with a focus on MMA caused by methylmalonyl-CoA mutase (mut) deficiency. Orphanet J. Rare Dis. 2019, 14, 84. [Google Scholar] [CrossRef] [PubMed]
- Fowler, B.; Leonard, J.V.; Baumgartner, M.R. Causes of and diagnostic approach to methylmalonic acidurias. J. Inherit. Metab. Dis. 2008, 31, 350–360. [Google Scholar] [CrossRef]
- Zhang, J.; Dobson, C.M.; Wu, X.; Lerner-Ellis, J.; Rosenblatt, D.S.; Gravel, R.A. Impact of cblB mutations on the function of ATP:cob(I)alamin adenosyltransferase in disorders of vitamin B12 metabolism. Mol. Genet. Metab. 2006, 87, 315–322. [Google Scholar] [CrossRef]
- Jorge-Finnigan, A.; Aguado, C.; Sánchez-Alcudia, R.; Abia, D.; Richard, E.; Merinero, B.; Gámez, A.; Banerjee, R.; Desviat, L.R.; Ugarte, M.; et al. Functional and structural analysis of five mutations identified in methylmalonic aciduria cbIB type. Hum. Mutat. 2010, 31, 1033–1042. [Google Scholar] [CrossRef] [PubMed]
- Brasil, S.; Richard, E.; Jorge-Finnigan, A.; Leal, F.; Merinero, B.; Banerjee, R.; Desviat, L.R.; Ugarte, M.; Pérez, B. Methylmalonic aciduria cblB type: Characterization of two novel mutations and mitochondrial dysfunction studies. Clin. Genet. 2015, 87, 576–581. [Google Scholar] [CrossRef][Green Version]
- Brasil, S.; Briso-Montiano, A.; Gámez, A.; Underhaug, J.; Flydal, M.I.; Desviat, L.; Merinero, B.; Ugarte, M.; Martinez, A.; Pérez, B. New perspectives for pharmacological chaperoning treatment in methylmalonic aciduria cblB type. Biochim. Biophys. Acta BBA - Mol. Basis Dis. 2018, 1864, 640–648. [Google Scholar] [CrossRef] [PubMed]
- Jorge-Finnigan, A.; Brasil, S.; Underhaug, J.; Ruiz-Sala, P.; Merinero, B.; Banerjee, R.; Desviat, L.R.; Ugarte, M.; Martinez, A.; Perez, B. Pharmacological chaperones as a potential therapeutic option in methylmalonic aciduria cblB type. Hum. Mol. Genet. 2013, 22, 3680–3689. [Google Scholar] [CrossRef]
- Sparks, S.E.; Krasnewich, D.M. PMM2-CDG (CDG-Ia). In GeneReviews®®; Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J., Stephens, K., Amemiya, A., Eds.; University of Washington, Seattle: Seattle, WA, USA, 1993–2020; Available online: https://www.ncbi.nlm.nih.gov/books/NBK1110/ (accessed on 9 July 2020).
- Jaeken, J.; Matthijs, G. Congenital disorders of glycosylation. Annu. Rev. Genom. Hum. Genet. 2001, 2, 129–151. [Google Scholar] [CrossRef]
- Andreotti, G.; Monti, M.C.; Citro, V.; Cubellis, M.V. Heterodimerization of Two Pathological Mutants Enhances the Activity of Human Phosphomannomutase2. PLoS ONE 2015, 10, e0139882. [Google Scholar] [CrossRef] [PubMed]
- Yuste-Checa, P.; Gámez, A.; Brasil, S.; Desviat, L.R.; Ugarte, M.; Pérez-Cerdá, C.; Pérez, B. The effects of pmm2-cdg-causing mutations on the folding, activity, and stability of the PMM2 protein. Hum. Mutat. 2015, 36, 851–860. [Google Scholar] [CrossRef] [PubMed]
- Andreotti, G.; Pedone, E.; Giordano, A.; Cubellis, M.V. Biochemical phenotype of a common disease-causing mutation and a possible therapeutic approach for the phosphomannomutase 2-associated disorder of glycosylation. Mol. Genet. Genom. Med. 2013, 1, 32–44. [Google Scholar] [CrossRef] [PubMed]
- Andreotti, G.; Vaca, I.C.d.; Poziello, A.; Monti, M.C.; Guallar, V.; Cubellis, M.V. Conformational response to ligand binding in phosphomannomutase2. insights into inborn glycosilation disorder. J. Biol. Chem. 2014, 289, 34900–34910. [Google Scholar] [CrossRef]
- Monticelli, M.; Liguori, L.; Allocca, M.; Andreotti, G.; Cubellis, M.V. β-Glucose-1,6-Bisphosphate Stabilizes pathological phophomannomutase2 mutants in vitro and represents a lead compound to develop pharmacological chaperones for the most common disorder of glycosylation, PMM2-CDG. Int. J. Mol. Sci. 2019, 20, 41164. [Google Scholar] [CrossRef]
- Yuste-Checa, P.; Brasil, S.; Gámez, A.; Underhaug, J.; Desviat, L.R.; Ugarte, M.; Pérez-Cerdá, C.; Martinez, A.; Pérez, B. Pharmacological chaperoning: a potential treatment for PMM2-CDG. Hum. Mutat. 2017, 38, 160–168. [Google Scholar] [CrossRef]
| Protein | Second Generation Pharmacological Chaperone | Reference | |||
|---|---|---|---|---|---|
| Name | Structure | Known Binding Site (Identification Mode) | |||
| Gaucher disease (OMIM# 231000) | Gcase | 2-(2-((4-bromophenyl)amino)-2-oxoethoxy)-N-(2-(methyl(phenyl)amino)-2-oxoethyl)benzamide (ML266 or NCGC00182186) |  | No | [49] |
| 2-[2-[(4-iodophenyl)amino]-2-oxoethoxy]-N-[2- (methylphenylamino)-2-oxoethyl]-benzamide (NCGC607) | 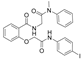 | No | [50,51] | ||
| N-(4-ethynylphenyl)-5,7-dimethylpyrazolo[1,5-a]pyrimidine-3-carboxamide (ML198 or NCGC00188758) | 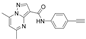 | No | [49,52,53] | ||
| 5,7-dimethyl-N-((1r,4r)-4-(pentyloxy)cyclohexyl)pyrazolo[1,5-a]pyrimidine-3-carboxamide (LTI-291) | 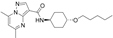 | No | [55,56] | ||
| ((S)-N-((2,3-dihydrobenzo[b][1,4]dioxin-2-yl)methyl)-N-methyl-2-(pyridin-3-yl)quinazolin-4-amine (S-181) | 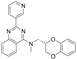 | Yes (co-crystallization with a derivatives) | [63,64,65] | ||
| Fabry disease (OMIM# 301500) | α-Gal A | 2,6-dithiopurine (DTP) | 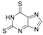 | Yes (virtual screening) | [72] |
| Pompe disease (OMIM# 232300) | GAA | 1-(3,4-dimethoxybenzyl)-6-propyl-2-thioxo-2,3,5,6,7,8-hexahydropyrimido[4,5-d]pyrimidin-4(1H)-one (ML247 or NCGC00183885) | 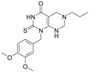 | No | [79,80] |
| N-acetylcysteine | 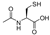 | Yes (co-crystallization) | [81,82] | ||
| N-butyl-l-deoxynojirimycin (l-NBDNJ) |  | No | [83] | ||
| Krabbe disease (OMIM# 245200) | GALC | α-Lobeline | 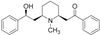 | No | [91,92] |
| 3′,4′,7-trihydroxyisoflavone |  | No | [92] | ||
| Phenylketonuria (OMIM# 261600) | PAH | (6R)-l-erythro-5,6,7,8-tetrahydrobiopterin (BH4) | 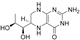 | Yes (co-crystallization) | [107,108] |
| Congenital erythropoietic porphyria (OMIM# 263700) | UORIIIS | Ciclopirox | 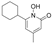 | Yes (virtual screening, NMR-based experiments) | [112] |
| Methylmalonic aciduria cblB type (OMIM# 251110) | ATR | N-(((4-chlorophenyl)carbamothioyl)amino)- 2-phenylacetamide | 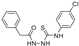 | Yes (molecular docking) | [118,119] |
| Phosphomannomutase 2 deficiency (OMIM# 212065) | PMM2 | 1-(3-chlorophenyl)-3,3-bis(pyridin-2-yl)urea | 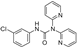 | No | [127] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tran, M.L.; Génisson, Y.; Ballereau, S.; Dehoux, C. Second-Generation Pharmacological Chaperones: Beyond Inhibitors. Molecules 2020, 25, 3145. https://doi.org/10.3390/molecules25143145
Tran ML, Génisson Y, Ballereau S, Dehoux C. Second-Generation Pharmacological Chaperones: Beyond Inhibitors. Molecules. 2020; 25(14):3145. https://doi.org/10.3390/molecules25143145
Chicago/Turabian StyleTran, My Lan, Yves Génisson, Stéphanie Ballereau, and Cécile Dehoux. 2020. "Second-Generation Pharmacological Chaperones: Beyond Inhibitors" Molecules 25, no. 14: 3145. https://doi.org/10.3390/molecules25143145
APA StyleTran, M. L., Génisson, Y., Ballereau, S., & Dehoux, C. (2020). Second-Generation Pharmacological Chaperones: Beyond Inhibitors. Molecules, 25(14), 3145. https://doi.org/10.3390/molecules25143145







