Magnetic Hyperthermia for Cancer Treatment: Main Parameters Affecting the Outcome of In Vitro and In Vivo Studies
Abstract
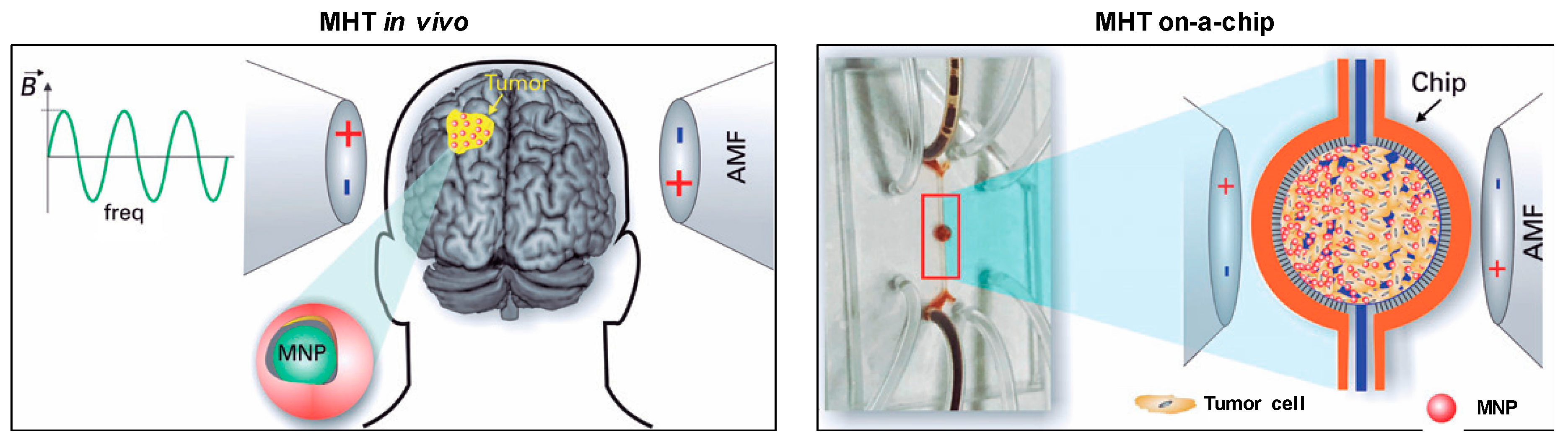
1. Introduction to Magnetic Hyperthermia: Concepts and Terminology
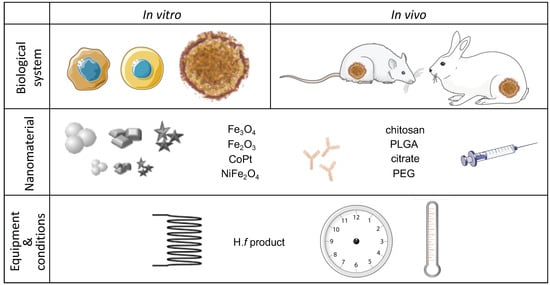
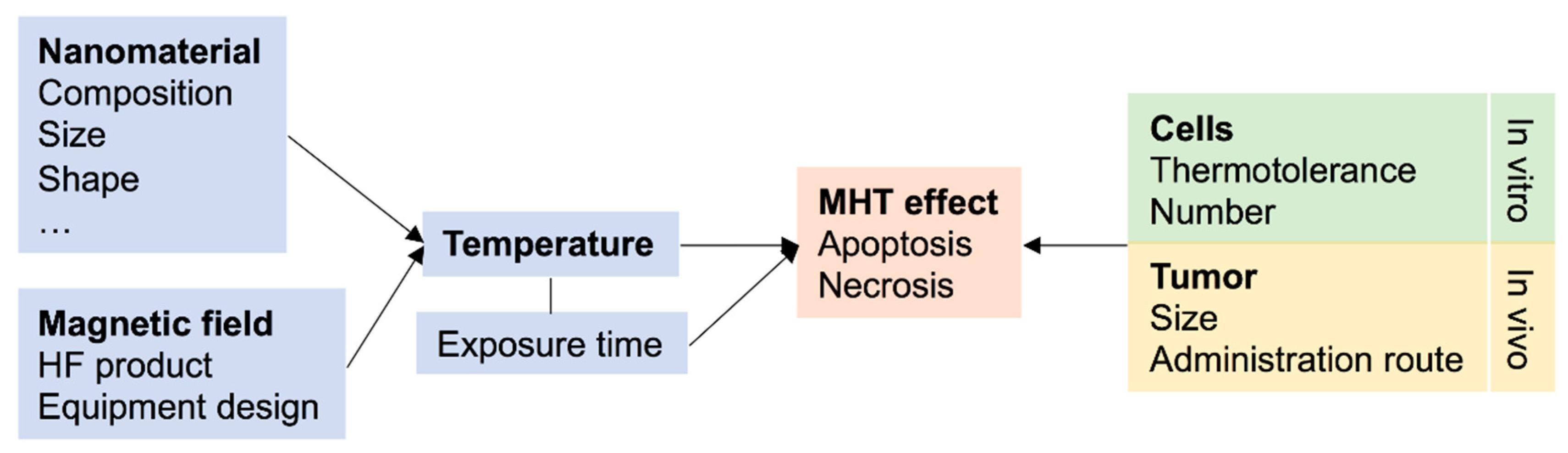
2. Main Parameters Influencing the Outcome of a Preclinical Magnetic Hyperthermia Study
2.1. The Biological Component—Cells, Cell Lines, and Animal Models
2.1.1. Relevance and Thermal Susceptibility
2.1.2. Cell Number, Configuration, and Tumor Size
2.1.3. Animal Models for In Vivo Studies
2.1.4. Parallel Tests in Normal Cells
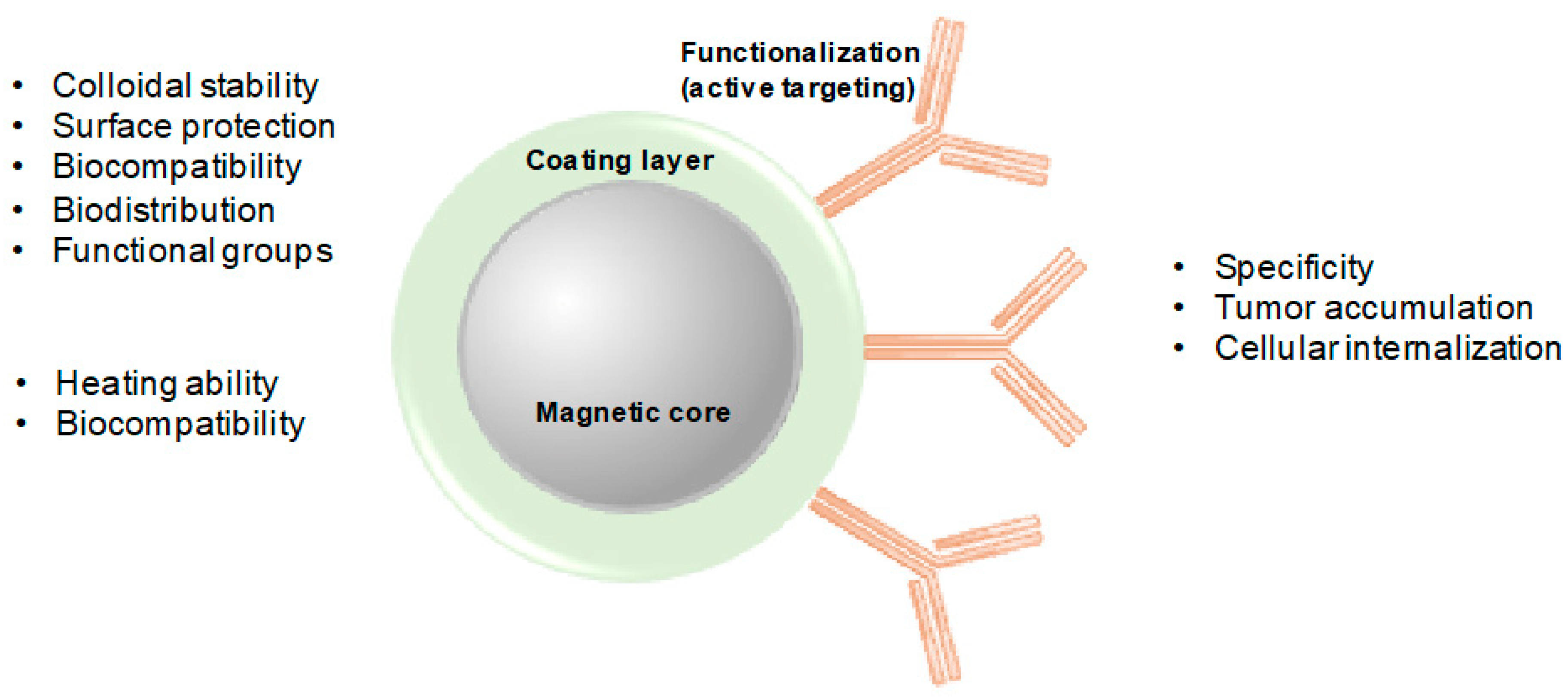
2.2. The Nanomaterial Component
2.2.1. Size, Coating, and Chemical Composition
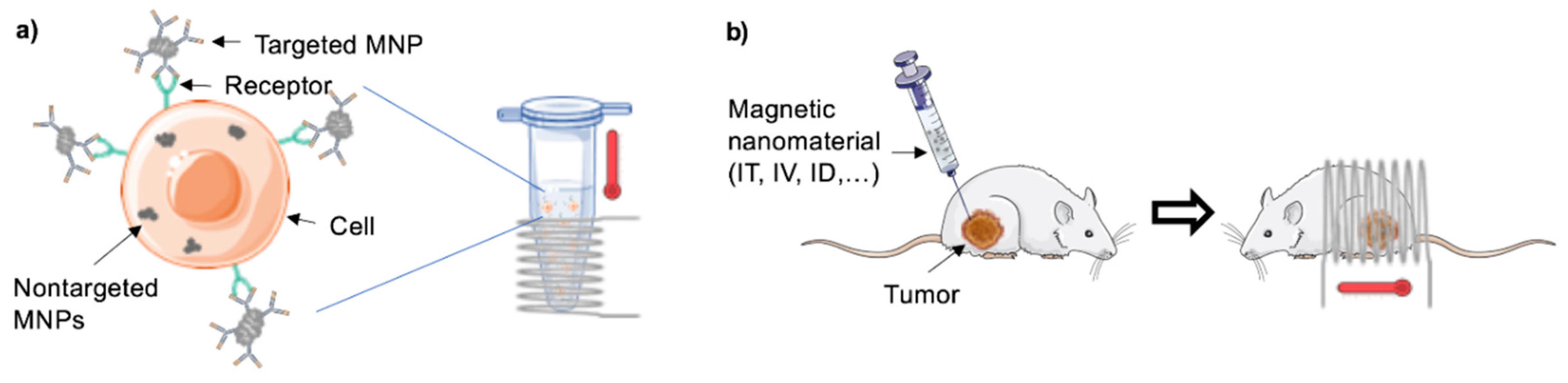
2.2.2. Targeting
2.2.3. Concentration, Time of Incubation with the Cells, and Nanomaterial Excess Removal
2.2.4. Administration Route
2.3. The Alternating Magnetic Field Component
2.3.1. AMF Power
2.3.2. Reached Temperature and Time of Exposure
2.3.3. Apoptosis or Necrosis?
2.4. Assessment of MHT Efficiency
2.4.1. Time-Point after MHT
2.4.2. Most Commonly Used Tests
3. Remarks and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Cell Line | MNPs | Inc. Time (h) | [Fe] Sample | MNPs Excess Removal | Magnetic Field | Reported Temp (°C) | % Cell Viability (h after MHT) Method | Obs. | Ref. | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Coating | Size nm (Technique) | Initial Conc. (g·L−1) | F (kHz) | H (Ka m−1) | Time (min) | ||||||||
| 4T1 + C166 + MEF | D-mannitol | ~150 (SEM) | 0.1 (LD), 1 (HD) | 24 | NR | Yes | 300 | 55 | 60 | NR | Significant increase cell death/damage (72) Sytox® Blue | Co-culture of three distinct cell types to simulate a 3D in vitro model of triple negative breast cancer lung metastasis. | [14] |
| A2780cp20; HeyA8 | Carboxy methyl dextran | ~60 and ~200 (bimodal, DLS) | 0.5 (Fe) | 0 | NA | No | 245 | 24 or 36 | 30 or 60 | 41, 43 or 45 | 41 °C = cell viability unaffected; 43 °C: <40; 45 °C: <10; (48) TB | Combined with HSP70 inhibition (HSPA6 siRNA or PES) potentiated MHT at 41 °C (HSPA6: 15–25% cell viability HeyA8, 25–50% A2780cp20; PES: <40% cell viability both cell lines). | [10] |
| A549; MDA-MB-231 | Myristic acid + pluronic F127 | 12 ± 3 (TEM); ~185 (DLS) | 1.5 (Fe3O4) | 0 | NA | No | 386 | 6 | 5, 15, 30 | 43–46 | ~60, ~30, ~10, resp. (2) LDH | Similar efficiency for both cell lines; no colonies formed two weeks after MHT; MHT for cancer stem cell elimination. | [75] |
| BV2 | PAA | 36 ± 8 (TEM) | 0.1 | 24 | NA | Yes | 560 | 23.9 | 30 | 46 | 25 (4.5), TB | Cell pellets used for treatments. Comparable efficiency between MHT and water bath hyperthermia. Suggest the use of microglial cells as MNP carriers to treat glioma with MHT. | [12] |
| C6 | Aminosilane | NR | 10 (Fe) | 0 | NA | No | 305 | 23,9 | 30 | 41–43 | 80 (10 min); 0 (0.5) Live/dead® | Tumor-on-a-chip to mimic glioblastoma | [15] |
| CT-26 | PEG | The SPIONs are ~20 (TEM) | 0.1 | 2 | NA | No | 293 | 12.57 | 15 | ~40 | 90, 85, 74, (48, 72, 96), MTT | 3 cycles of MHT days 1, 2 and 3. MTT on days 2, 3 and 4. Incorporation of DOX resulted in synergistic (45%, 30%, and 18% cell viability at 48, 72 and 96 h, resp.). Theranostic formulation (great MRI contrast). | [109] |
| DU-145 | Starch | ~108 (DLS) | 0.015, 0.075, 0.1, 0.15 (Fe) | 16–24 | 5, 70, 105, 199 pg/cell, resp. | Yes | 150 | 88 | 30 | 37–49.1 | <10 for T > 43 °C (10–14 days) clonogenic survival assay | PDL as facilitator of MNPs uptake; different reached temperature according to cell pellet size and iron/cell. | [13] |
| DX3 | Citric acid | ~17 (TEM); ~66 (DLS) | 0.5 | 12 | Susp: 210-400; Adhe: 315 pgFe3O4/cell | Yes | Susp: 911 or 950; Adhe: 950 | Susp: 6.6; 10.5; 12; 14.7 or 16.1; Adhe: 10.8 | 120 | 40 to 50 | Susp:~55 to ~5; Adhe: 3 (24) Annexin-V/Pi | No normal cells control; experiments in adhe cells suggest the occurrence of cell specific events, supporting individual cell hyperthermia. | [11] |
| ESO26, OE21, NIH-3T3 | NA | NA | NA | 24 | NR | NA | 270 | 29.4 or 34.7 | 10 sec, day 1 and 2 | 45 | CPI of ~1.3 for both cancer cell lines (12) WST-8 | Poly(NIPAAm-CO-HMAAm)/polyurethane coating a nitinol stent; NIH-3T3 only for biocompatibility studies; CPI drops to 0.1 when combined with 5-FU and PTX. | [110] |
| HeLa | Phospho lipid-PEG | ~14 (TEM) | 0.1 (Fe) | 0 | NA | No | 355 | 24 | 60 | 43–46 | ~55 (24) MTT | When using DOX-MNPs = 32% cell viability. | [111] |
| HepG2 | NA | 689 ± 155 (DLS) | 0.2 mg | 0 | NA | No | 750 | 0.8 | 30 | 43 | ~50 (24) WST1 | MHT increased intracellular ROS levels and DNA damage. | [112] |
| HT-1080 | Dextran | ~225 (DLS) | NR (drug conc 147 nM) | NR | NA | No | 250 | 27.9 | 30 | ~43.5 | 69 (48) TB | When PTX-loaded magnetic liposomes are used for MHT cell viability decreases to 28%. | [113] |
| Jurkat | PMAO-PEG | 12, 13, 16 (TEM) ~20 (DLS) | 0.6, 0.6 or 0.49, resp. | 0.25 | NA | No | 373 | 14 | 15 | ~38.5, ~40 and ~43.5, | 90, 75, and 40, resp. (0.5) ATP levels | Optimization of MNP size and polydispersity to enhance MHT response at a selected AMF frequency. | [33] |
| KB | Pullulan acetate | ~10 (TEM); 25.8 ± 6.1 (DLS) | NR | 0 | NA | No | 100 | 10.4 | 20 | 45 or 47 | 45 °C = 45, 47 °C = 20 (24) MTT | L929 (normal) cell viability without AMF application = 90%. | [22] |
| KB | PEG | 19 ± 3 (TEM) 37 ± 11 (DLS) | 1 (Fe) | 24 | 25–170 pg/cell 1.3–5.4 g/L | Yes | 110 | 20 | 60 | 43 | 0–75 (NR) TB | 2–4gFe/L to reach 40–45 °C = 50% cell viability; at 5gFe/L reached 65 °C = 0% cell viability. | [68] |
| MCF-7 | Triethylene glycol: triethanol amine | ~44 (TEM) | 0.5 (Fe) | 0 | NA | No | 240 | 89 | 60 | 45 | 25 (48) MTT | Nanoclusters also suitable for MRI in vivo for the un-clustered 10 nm MNPs = 40% cell viability. | [72] |
| MCF-7 | Chitosan | 20–30 (TEM) | 1 | 0 | NR | NR | 267 | 24 | 120 | 44–45 | 60 (1) TB | L929 (normal) = 93% cell viability. | [25] |
| MCF-7 | Terephthalic acid | 10 ± 2 (TEM) 150.9 ± 0.5 (DLS) | 1 | 0.25 | NA | No | 751.5 | 10.9 | 60 | 45 | 0 (24) TB | Highly effective MHT but without MNP removal, so not intracellular hyperthermia. | [70] |
| MCF-7 | Oleic acid + aspartic acid | 11 (TEM) | 1.5 or 2.5 | 3 | NA | No | 265 | 26.7 | 20 | NR | 17 for the 1.5 and 23 for the 2.5 (24)% of sub-G1 cells | The aspartate coated MNPs exhibited enhanced interaction with cells and superior killing effects when compared to pristine MNPs. | [114] |
| MCF-7 | Polyamidoamine dendrimer | 10 ± 4 (TEM); 120 (DLS) | 0.5 | 2 | 120 pg/cell | No | 300 | 12 | 120 | NR | 36.7 (0) MTT | Normal HDF1 cell viability = 63.5% when treated similarly. | [28] |
| MCF-7; MCF-7/ADR | mPEG−PCL | ~100 (TEM and DLS) | 0.2 (MNCs), 0.1 (MNPs) | 0 | NA | No | 114 | 115 | 15 | NR | 10 (24) MTT | MnFe2O4/MNC vs. Mn0.6Zn0.4Fe2O4/MNC, both with similar MHT efficiencies, in both cell lines. Use low AMF exposure times. | [74] |
| MDA-MB-231 | PEG bis(amine) | ~15 (TEM) | 0.2 | 5 | NA | No | 500 | 37.4 | 60 | 43 ± 1 | 75 (NR) WST-8 | When using GdTx-MNPs for MHT = 36% cell viability (GdTx as sensitizer to MHT). | [115] |
| MDA-MB-231 | Chitosan | ~18 (TEM), ~90 (DLS) | 1.5 (Mn and Fe) | 0 | NA | No | 307 | 50 (then to 20 or 35) | 30 | 42 or 52, resp. | 22.5 and 18, resp. (24) Annexin-V/Pi | 24, 48, and 72h incubation w/ MNPs yielded 100, 112 and 146 pg Fe/cell, MHT42 = apoptosis, MHT52 = necrosis. | [73] |
| MDA-MB-468, Caco-2, A2780 | Carboxy methyl dextran | 69 ± 4 (TEM) | 3.8 | 0 | NA | No | 233 | 29.4 or 34.7 | 30 | 43 or 45, resp. | MDA-MB-468 43 °C = 50, 45 °C = 30, Caco-2 43 °C = 35, 45 °C = 15, A2780 43 °C = 25, 45 °C = 5 (48) TB | Reported enhanced effects of bortezomib in combination with MHT (cell viability <20% in all cases). | [8] |
| MG-63 | Sodium oleate | 25–40 (TEM) | 5 | 0 | NA | No | 186 | 17 | 60 | 37–49 | >90 up to 43 °C and 54 at 47 °C (0) MTT | MHT on average 16% more efficient than water-based hyperthermia. The short time after MHT for assessing cytotoxicity does not allow to quantify apoptotic effects. | [116] |
| MIA-PaCa-2 | PLGA | 1:1:10–204 1:1:20–245 (DLS) | 1 | NR | NA | No | 0.323 | 90 | 180 | NR | 1:1:10–25, 1:1:20–50 (0) TB | MHT to potentiate chemotherapy with HSP90 inhibitor 17AAG. Elevated cytotoxicity L929 cells (normal). | [24] |
| SaOS-2 | Citric acid | NR | 1 | 48 | 0.5 g/L | Yes | 765 | 20–24 | 10 (2 cycles with 48 h distance) | MnFe: 45 CoFe:41–45 | Single pulse: MnFe: 70, CoFe: 70 Multiple pulse: MnFe: 35, CoFe 70, (0) TB | Testing 2 binary ferrites in single vs. multiple pulse for MHT. Multiple pulse resulted in enhanced cytotoxicity specifically in cancer cells. Low cytotoxicity was observed for the normal cell line (3T3-L1) treated similarly. | [91] |
| SH-SY5Y | PEI | NR | 0.1 | 24 | NR | Yes | 570 | 3.98–23.9 (to control target temp) | 30 | 37–51 | 37 °C: 90; 40 °C: 75, 42 °C: ~50, 44 °C: ~40; 46 °C: ~25; 48 °C: ~10, 50 °C: <5 (6) TP | MHT induced higher cytotoxicity than water bath for the same target temp. Cytotoxic effects increased with increased time-point after treatment. | [100] |
| SKOV-3 | Liposomes | 150 (TEM), 200 (DLS) | 0.5–5 mM (Fe) | 1–4 | ~20 pg/cell (for the 5 mM Fe) | Yes | 700 | 24 | 30 | NR | 10 (12) AlamarBlue | When combined with PDT = 0% cell viability, missing MNP concentration of reported cell viability data. | [93] |
| SKOV-3 | Gallol-PEG | ~20 (TEM) | 0.2 mM (Fe) | 2 | ~6 pg/cell | Yes | 520 | 20 | 10 | 38–40 | 75 (NR) AlamarBlue | When combined with PTT = 15% cell viability and T = 50 °C. | [94] |
| SMMC-7721 | NA | 17 ± 2 (TEM) | 1 | 0 | NA | No | 50 | 34 | 40 | 42, 44 and 44.3 | 75.4, 61.5 and 53.6 (24) MTT | Application of a static magnetic field to limit the heating to a restricted area. | [71] |
| U87 | PEG | 177±17 (DLS) 153 (TEM) | 0.3 (Fe) | 4 | NA | No | Varied to keep temp | Variable to keep temp | 30 | Multiple MHT:44 °C; single MTA: 50 °C | NR (0) Annexin-V/Pi | Increased number of apoptotic cells with increasing number of MHT cycles. Gradual progression from apoptosis to necrosis from single to multiple MHT. Extensive necrosis for MTA. | [99] |
| U87 | Methoxy-PEG-silane 500 Da | 22.8 ± 3.3 (DLS) | 0.7 | NR | 500 pg/cell (for 0.5 g·L−1) | Yes | 99 | 12.33 | 25 | 63.5 | Complete necrosis (NR) CCK-8 | Resovist as a control reached 37.5 °C and induced no change in cell viability. | [45] |
| U87MG | PEG | 50–100 (TEM) | NR | 0 | NA | No | 750 | 16 | 120/ day x 4 days | 43 | 44.5 (72) Annexin-V/Pi | No significant improvement was found when the nano-vectors were loaded with temozolomide | [46] |
| U87-EGFRvIII | PEI | 77 ± 11 (DLS) | 0.01 | 5 | NR | Yes | 225 | 5 | 45 | 44.1 | 60 (24) MTS; 80 in spheroids | Magnetofection to facilitate MNPs uptake; if combined with let-7a microRNA = 34% cell viability (45% in spheroids). | [117] |
| Functionalization | Cell Line | MNPs | Inc. Time (h) | [Fe] Sample | MNPs Excess Removal | Magnetic Field | Reported T ( °C) | % Cell Viability (h after MHT) | Observation | Ref. | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Coating | Size (nm) | Initial Conc. (g·L−1) | F (kHz) | H (kA m−1) | Time (min) | |||||||||
| Anti- αβγ3 a | MDA-MB231 | PEG | 30–35 (TEM) | 520 μM (Fe) | 6 | NR | NR | 480 | 10 | 15 | 44 | 7.3 (0) MTS | Cell viability results from targeting + DOX + MHT. Only MHT = 48%, MHT + DOX = 31%. | [56] |
| Anti-CD90 b | Huh7 | PEG | 10–20 (TEM), 130 ± 4.6 | 0.34 (Fe) | 1 | NA | Yes | 200 | NR | 60 | 44 | 30 (24) MTT | Thermosensitive magneto-liposomes. CD90+ separated from CD90− by MACS and then treated with MHT. | [87] |
| Anti-CXCR4 b | Jurkat | Targ: dextran-PA; Non-targ: PAA | Targ: 250, Non-targ 18 (TEM) | Targ: 0.362; Non-targ: 0.396 (Fe) | Targ: 1; Non-targ 2 | Targ: 122; Non-targ: 4.3; comb: 155 pg/cell | Yes | 1st: 869 2nd: 554 | 1st: 20 2nd: 24 | 30 + 30 | ~43 or ~45 | 16 (when 43 °C); 0 (when 45 °C) (72) PB | Biphasic AMF to push temp to max and then stabilize it. MHT using targeted MNPs-only cell viability = 75%. Induction of necrosis is more effective than apoptosis. | [61] |
| Anti-CXCR4 b | LN229 HK-2 | Targ: dextran-PA; Non-targ: PAA | Targ: 250, Non-targ 18 (TEM) | Targ: 0.264; Non-targ: 0.260 (Fe) | Targ: 1; Non-targ 2.5 | LN229: 108; HK-2: 38 pg/cell | Yes | 1st: 869 2nd: 554 | 1st: 20 2nd: 24 | 30 + 30 | LN229: 46.9; HK-2: 41.2 | LN229: <10 and 0 HK-2: 75 and 80 (24 and 72) PB | Optimization of the MHT approach to tumor cells expressing lower levels of target receptor; HK-2 (normal) cells practically undamaged. | [21] |
| CREKA b | A549 | Dextran | 5–13 (TEM); | 3 (Fe3O4) | NA | NR | Yes? | 292 | 58 | 30 | NR | 40 (48 & 72) Calcein-AM | Incubation suspended cells + NP. Reported additive effects of cysplatin to 20% cell viability after 72 h. | [118] |
| Dipeptide (Arg-∆Phe) b | A549, NCI-H460, HLF-1 (normal) | NA | ~146 (TEM); ~123 (DLS) | 0.08 | 3? | A549 2.5 Non-targ, 1.9 Targ; NCI-H460 2.8 Non-targ, 3.23 Targ (µg/mL) | No | 50 | 175 mA | 180 | NR | A549: Non-targ 59; Targ 72; NCI-H460: Non-targ 65; Targ 85; (24) Pi | Use pulsed electromagnetic field. No significant differences in cell viability between targeted and non-targeted MNPs. HLF-1 (normal) cells not affected by similar treatment. | [26] |
| Folate a | MCF-7, G1 | Carboxy methyl cellulose | 100–150 (TEM); 80–200 (DLS) | 2 and 4 | NA | NR | NR | 305 | 18 | 60 | NR | 20 (24) TB | If combined with 5-FU = 5% cell viability. | [119] |
| Folate b | HeLa (FR + ) | PEG | 84.9 (TEM) | 0.5 | 1 | 0.3 g/L | No | 750 | 0.8 | 10 | 43–45 | NR | Reported LDH values of 0.76 compared to 0.45 for untreated control. Normal human fibroblasts not affected. | [27] |
| Folic acid a | HeLa | Poly acrylic acid | 8–10 (TEM) | 2 | 24 | ~250 pg/cell (24h inc. w/0.3 g L−1) | Yes | 265 | 27 | 10 | NR | 65 (24) SRB | When using DOX loaded FA-MNPs = 50% cell viability, DOX-FA-MNPs + AMF = 10% cell viability. | [108] |
| Folic acid a | SKOV3 | PEG | 120–140 (TEM) | 0.5 | NR | NR | NR | 200 | NR | 30 | NR | ~14 (72) MTT | Magnetic thermosensitive liposomes are loaded with HSP90 inhibitor, 17AAG, combining chemotherapy with targeted-MHT | [57] |
| Galactose b | HepG2 | Alginate | 109.1–146.9 (HD) | 0.5 | 4 | 364.4 pg/cell | Yes | 780 | 19 | 20 | NR | 5 (18) MTT | Only applicable for hepatic tumors. | [29] |
| Herceptin a | SKBR3 | APTES-PEG | NR | 100 μg (Fe) | NR | NR | NR | 100 | ~22 | 5 | 42 | 33 (48) TB | If combined with RIT cell viability = 3.3%. First study on combined use of RIT + DOX + targeted SPIONs to kill HER overexpressing cells. | [60] |
| Herceptin b | SK-BR-3 | Dextran | 138 ± 7.6 (DLS) | 28.6 pg Fe3O4/ cell | 4 | 16.5 pg Fe3O4/ cell | Yes | 360 | 9.6 | 30 | 42.5 | 25 (24) TB | Cell viability recovered after 5 days in culture. When AMF was repeated 24 h after, cell viability <10% after 5 days. | [120] |
| Herceptin a | SK-BR-3 | PLA-TPGS/ TPGS-COOH | 155.2 ± 0.17 (DLS) | 0.86; (177 ug Fe) | 24 | NR | NR | 240 | 42 | 20 or 30 | NR | 30 (12) MTT | If combined with docetaxel = 10% cell viability. | [58] |
| Herceptin a | MIAPaCa-2 | PLGA | 524 ± 9 (DLS) | 0.1 | 48 | NR | Yes | 440 | 16.2 | 15 | NR | NR (0) | Gemcitabine released with MHT, AO-EB staining showed late cell apoptosis/ necrosis and decreased Bcl2 and cyclin-D1 expression. | [23] |
| Hyaluronic acid a | 4T1 | Polypyrrole | 83.6 (DLS) | 0.493 (Fe3O4) | 24 | NR | NR | 635 | 30 A- | 15 | NR | CTR = 25 MHT = 14 (12) ALDH + cells | When Notch inhibitor is incorporated in the formulation ALDH+ cells = 9%, mammosphere cells: [CTR = 35 MHT = 17.5 MHT + Notch inhibitor = 9] ×104 cells. Claim effective elimination of cancer stem cells. | [59] |
| Hyaluronic acid b | SCC7, NIH3T3 (normal) | None vs. PEG | 100–272 (DLS) | 0.1 | 1 | NR | NR | 368 | 1 | 10 | 42 | ~30 (24) MTS | CD44- cell viability unchanged under similar MHT protocol. No differences in MHT outcome between PEG-coated or non-coated MNP. | [121] |
| iRGD a | U87-EGFRvIII; MDA-MB-231 | PEI + PEG | 46.8 ± 2.3 (DLS) | 0.02 | 24 | NR | Yes | 300 | 5 | 45 | NR | 40 (48) MTS | Magnetofection to facilitate MNPs uptake. MHT as an enhancer of peptide therapeutics. | [122] |
| LHRH peptide a | A2780/AD | PMAO + PEI + PEG | ~40 (TEM) ~90 (DLS) | 0.015 (Fe) | 12 | 14.9 pg/cell | Yes | 393 | 33.5 | 30 | 44 | 5 (48) Calcein-AM | Similar cell viability achieved with DOX loaded LHRH-MNPs combined with MHT at 40 °C. | [69] |
| Cell Line (Number of Cells) | In Vivo Model | Initial Tumor Size | MNPs | MNP. Inj. Mode | Magnetic Field | T (°C) | Outcome | Observation | Ref. | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Coating | Size (nm) | Initial Amount | Time after Inj. | F (kHz) | H (kA m−1) | Time (min) | Cycles | ||||||||
| Human breast cancer MCF-7 (2 × 106) | BALB/c female mice | 50 mm3 | - | 97.85 ± 0.74 (DLS) | NR | IV | 0 | 423 | 10 | 30 | 5 | 42 | Delayed tumor progression | AMF to favor tumor accumulation. Triple effect: MHT, cell-penetrating peptides to increase DOX uptake. Thermo-responsive DOX release. | [16] |
| Human breast cancer MCF-7 (2 × 106) | BALB/c female mice | 100 mm3 | - | NR | NR | IV | 0 | 423 | 10 | 30 | 5 | 42–43 | Delayed tumor progression, increased tumor accumulation, augmented c-Myc silencing | AMF to favor tumor accumulation. Triple effect: MHT, cell-penetrating peptides to increase siRNA delivery, thermo-responsive siRNA-CPP release. | [123] |
| Human breast cancer MDA-MB-231 (NR) | NS (nude mice) | 100 mm3 | PEG bis (amine) | ~15 (TEM) | 75 µg | IT | 0 | 500 | 37.4 | 30 | 1 | 43 | Incomplete tumor regression day 8, tumor regrowth day 12 | When using GdTx-MNPs for MHT = tumor eliminated within eight days. | [115] |
| Human breast cancer SKBR3 (3 × 105) | BALB/c female mice | 0.2 cm3 | APTES-PEG | NR | 0.5 mgFe | IV | 48 | 100 | ~22 | 15 | 2 | NR | Tumor volume inhibitory rate day 28 = 85%, nearly complete tissue necrosis | Permanent magnet for magnetic delivery w/o detectable damage to surrounding tissues. Trastuzumab-conjugated, radiolabeled, DOX-loaded MNPs suitable detection by MRI or SPECT (targeted MHT + radio + chemo + imaging). WBC decreased by 23% day 28. | [60] |
| Human chronic myeloid leukemia K562/A02 (1 × 107) | BALB/c mice | 950 ± 150 mm3 | Oleic acid + Pluronic F-127 | 18.4 ± 1.8 (TEM) | 22 µg MCL/g body weight | IT | 0 | 219 | 10.5–310 | 40 | 1 | ~42 | 40% decrease relative tumor volume | If combined with DNR + 5-BrTet = 80% decrease relative tumor volume and decreased P-gp expression. | [124] |
| Human epidermoid carcinoma A431 (5 × 106) | SCID female mice | 200–400 mm3 (calc.) | PLGA-b-PEG | 77.8 ± 2.1 (DLS) | 400–800 µL of 4.5 g Fe3O4/L, days 16, 17, 18, 19, and 22 | IT | 0 | 173 | 25 | 30 | 5 | 5–6 inc. | 1.7 × inc. MS | hEGFR-targeted MNPs. Observed increased temperature in subsequent treatments. Higher accumulation in liver and lungs than tumor after IV injection of MNPs. | [80] |
| Human fibrosarcoma HT-1080 (1 × 106) | Swiss female mice | 8–15 mm diameter | Dextran | 225 ± 45 (DLS) | SD = 1, DD = 2 (mg MNPs) | IT | 0 | 250 | 27.9 | 30 | SD = 3, DD = 5 | 42.5 | SD: Significantly slower tumor growth both w/ and w/o PTX, DD: w/ PTX significantly different from w/o PTX | CTR mice sacrificed on day 12 due to tumor burden; SD sacrificed day 16; DD sacrificed day 22 (days after 1st dose); treatment considered harmless to the body. | [113] |
| Human glioblastoma U251 (1 × 107) | BALB/c female mice | NR | - | NR | 50 mM | SC | 0 | 280 | 335.4 Arms | 60 | 1 | 43.1 | Tumor size reduction: Fe(Salen) + MHT = 80–90%, but not significantly different from Fe(Salen) only = 50% | The tumor model was injected in the mice leg due to limited injection volume in the mice brain; sacrifice day 28. | [125] |
| Human glioblastoma U87MG (1 × 107) | BALB/c nude mice | 100–150 mm3 | PEG | 177 ± 16.9 (DLS) | MHT: 3.5 µgFe/µL, MTA: 8.7 µgFe/µL | IT multi | 0 | 389 | 19.5 | 25 | MHT: 1, 2, 3, or 4. MTA: 1 | MHT: 45 (43-44th cycle). MTA: 53.1 | MHT: Significant tumor regression after 3 or 4 cycles, but tumor recurrence if only 3 cycles are performed. MTA: complete tumor regression | Sacrifice day 25. MTA lead to serious bleeding and infection. | [99] |
| Human glioblastoma U87MG (1 × 107) | BALB/c nude mice | 100–150 mm3 | PEG | SPIONs: 18; hydrogel: 519 ± 141 (TEM) | 2.9 µgFe/µL | IT multi | 0 | 366 | 13.3 | 60 | 2 | 43 ± 1 | Significant inhibition of tumor growth specially after 2 cycles. 100% survival rate | Sacrifice day 25; TRAIL-loaded hydrogel; 2 MHT cycles enhanced TRAIL-induced cytotoxicity; neither kidney nor liver damage; long-term MRI imaging. | [105] |
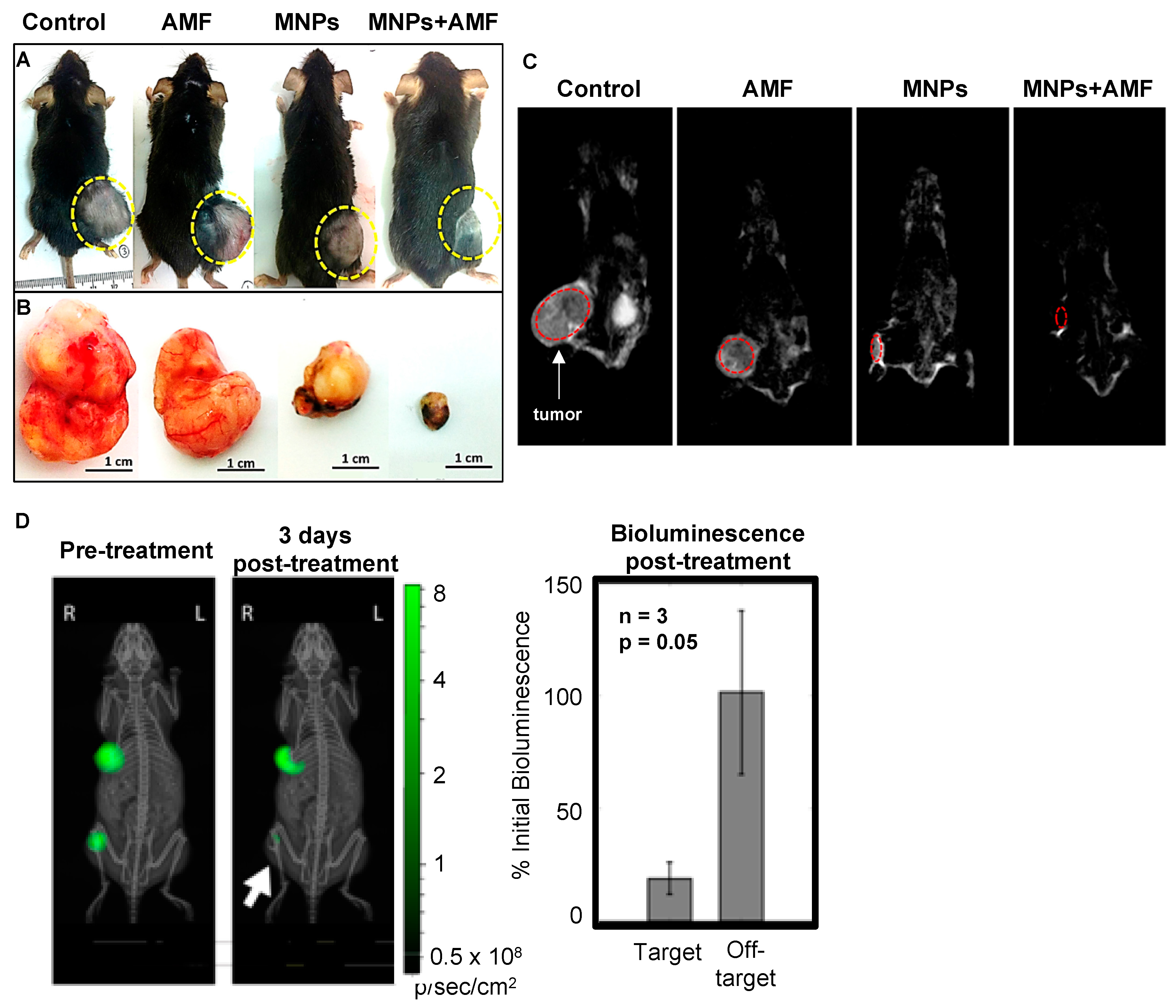
| Human hepatocellular carcinoma Hep3B (NR) | NS (nude mice) | 1000 mm3 | Methoxy-PEG-silane 500 Da | 22.8 ± 3.3 (DLS) | 100 µL of 1.15 g/L | IT | 0 | 99 | 12.33 | 15 | 1 | 50.2 | Complete tumor elimination 2 days after MHT (bioluminescence imaging) | Mice under observation for 1 month: no tumor regrowth, no severe side effects. | [45] |
| Human hepatocellular carcinoma HepG2 (1 × 106) | BALB/c female mice | 0.5 cm diameter | PEI | 20–30 (TEM) | 1 mg/cm3 tumor | IT | 0 | 230 | NR | 60 | 2 | 42–44 | 50% or 90% reduction tumor mass 28 days after MHT, resp. MHT and MHT + gene therapy | Combination of MHT with gene therapy targeting α-fetoprotein in hepatocarcinoma. | [88] |
| Human hepatocellular carcinoma HepG2 (2 × 106) | BALB/c female nude mice | 0.3–0.5 cm3 | PEI | 15–20 (TEM) | 5 mg of 10 g/L | IT multi | 24 h | 230 | NR (30 A) | 60 | 3 | 42–45 | Decrease tumor mass 6 weeks after MHT = 77%, MHT + radio + gene = 94% | MNPs functionalized w/ anti-α-fetoprotein antibody. Multimodality treatment combining MHT, radio and gene therapy. No side effects on liver, kidney and no inhibition of hematopoiesis. | [77] |
| Human hepatocellular carcinoma HepG2 (1 × 107) | BALB/c male nude mice | ~0.4 cm3 | PRO | 15–20 (TEM) | 500 mg/mL | IT multi | 24 and 48 h | 110 | 8.8 | 30 | 2 | 43 | MHT-only and gene therapy-only did not block tumor growth. The combination of both caused the tumor to shrink. | Sacrifice day 30 after injection; gene therapy = delivery of the TNFα gene. | [86] |
| Human hepatocellular carcinoma Huh7 CD90+ (2 × 104) | NOD/ SCID mice | 600 mm3 | PEG | 10–20 (TEM). 130 ± 4.6 (DLS) | NR | IT | 24 h | 200 | NR | 60 | 3 | NR | 27.3 ± 9.8% complete tumor regression 70 days after injection | When using Anti-CD90-MNPs = 78 ± 19.1% complete tumor regression 70 days after injection. Rectal T < 40 °C. | [87] |
| Human hepatocellular carcinoma Huh7 (2 × 104) | Athymic nude female mice | NR | Silica | NR | 932 μg/mL | IT | 0 | 750–800 | NR | 30 | 1 | ~43 | Attenuation of tumor growth compared to control, but still tumors grew from day 0. | Heat-induced release of ansamitocin (chemo + MHT), sacrifice day ~10,A follow-up treatment would be necessary to sustain inhibitory effect on tumor growth. | [85] |
| Human hepatocellular carcinoma SMMC-7721 (1 × 106) | NS (nude mice) | ~450 mm3 | PLGA | NA | NR (50 μL of 20% Fe) | IT | 0 | 626 | NR (28.6 A) | ~2 | 1 | 52 | Tumor ablation and elimination 5 days after MHT observed in synergy with DOX release upon heating. | MHT alone produces tumor ablation with residual growth 8 days after treatment. | [84] |
| Human lung squamous carcinoma A549-Luc (1 × 106) | Fox Chase SCID female mice | 0.5 × 106 photons/s | Myristic acid + pluronic F127 | 369 ± 34 (DLS) | NR | IH | 7 days | 386 | 6 | 30 | 1 | NR | Reduction in tumor growth rate over 1 month after MHT. Reduced tumor weight. | No significant side effects. Inhalation led to higher tumor MNPs accumulation than other organs. | [126] |
| Human ovarian cancer HeyA8 or A2780cp20 (1 × 106) | Athymic nude mice | 35–113 mm3 | Carboxy methyl dextran | ~60 and ~200 (bimodal, DLS) | 5 mgFe/cm3 | IT or IP | 4 h | 245 | 23 | 30 | 3 | 43 | Reduction in tumor growth (volume and weight) is enhanced by the number of MHT treatments. | Combination with HSP70 silencing tumor growth is inhibited. | [10] |
| Human ovarian carcinoma OVCAR-3 (1 × 106) | Balb/c nu/nu female mice | 100 mm3 | Carboxy dextran | 77 (DLS) | 300 µgFe/mL (5 × 105 SPION-labeled MSC) | SC | NA | 1050 | 8 (10 mT) | 20 | 4 | 41.5; 40.8; 39.7; 38.2, resp. | No difference in tumor growth (tumor volume similar to control). | SPION-labeled MSC injected simultaneously with tumor cells. Magnetic heating effect decreased with cycles: heat-induced MSC death and clearance? | [18] |
| Human ovarian carcinoma SKOV3 (5 × 106) | BALB/c female nude mice | NR | PEG | ~130 (TEM) | 10 mgFe/Kg | IV | NR | 200 | NR | 30 | 1 (or 4?) | NR | MHT = 63%. MHT + 17-AAG = 68%; MHT + 17-AAG + FA = 85% tumor weight inhibition rate. | 4 IV injections of MLS. Not clear whether AMF is applied only one time or one time after each injection (total 4 times). | [57] |
| Human pancreatic cancer MIAPaCa-2 (1.5 × 107) | B6.CB17-Prkdscid/szJ mice | >50 mm3 | PLGA | 524 ± 9 (DLS) | 2 mg/Kg GCT equivalent | IT | 0 | 440 | 16.2 | 15 | 10 | 6 inc. | 86% reduction tumor volume after 30 days. Reduced expression of Bcl-2 and cyclin-D1 | Herceptin-targeted nanospheres containing fluorescent IONP and GCT, tested MHT and MRI, | [23] |
| Mouse breast cancer 4T1 (5 × 106) | BALB/c female mice | 50–80 mm3(calc.) | PEG | 54.6 (DLS) | 6 × 30 µg Fe/g body weight (every other day) | IV | 30 min | 390 | 2.6 | 30 | 15 | Passive: 38.7–42.5; Active: 39.6–44.1 | 59% apoptotic cells and delayed tumor growth | Passive vs. active targeting (ανβ3 integrin—targeted MNCs); studied bioaccumulation in the main organs, combined MRI and MHT, no clear toxicity | [81] |
| Mouse breast cancer 4T1 (5 × 106) | BALB/c nude mice | ~80 mm3 | Polypyrrole | 83.6 (DLS) | 18.64 mgFe/Kg | IT | 0 | 635 | 30 A | 15 | 4 | 45 (calc.) | Slower tumor growth, significantly lower tumor weight, decreased number of CD44 + cells: targeted-MHT = 57%, targeted-MHT + chemo = 33% | Theranostic tool: chemotherapy mediated by Notch inhibitor + targeted-MHT + dual-mode MRI and photoacoustic imaging | [59] |
| Mouse breast cancer 4T1 (5 × 106) | BALB/c female mice | ~80 mm3 (calc.) | PEG | 30–45 (TEM) | 0.25 mgFe/ 100 mm3 | IT | 60 min | 480 | 10 | 15 | 1 | NR | Tumor volume inhibition day 16 —MHT = 18%, MHT + DOX = 88%, targ-MHT + DOX = 89% w/ absence of metastasis | Targeting of ανβ3 integrin contributes for the absence of metastasis. Enhanced MRI-T2 contrast. | [56] |
| Mouse colon carcinoma CT-26 (1 × 105) or mouse melanoma B16F10 (1.25 × 105) | BALB/c or C57BL/6 mice | 5 × 6 mm (2 tumors/ mouse) | BNF-Starch | 100 (TEM) | 140 µg Fe | ID | 0 | 167.5 | 36–44 | 20 or 30 | 1 | 42.5–43 for 30 min; 44.5–45 for 20 min | 43 °C = Complete elimination of treated CT26 tumor in BALB/c in 5 days + untreated tumor grew slower, incomplete elimination of treated B16 tumor in C57BL/6 + untreated tumor grew slower, 45 °C = complete elimination of treated B16 tumor in C57BL/6 + no effect in the untreated tumor | Hyperthermia-induced immunologic response at 43 but not at 45 °C. Immunologic response was not observed for Lewis lung carcinoma tumors, even at 43 °C. rectum T = 35.5–37 °C | [19] |
| Mouse Lewis lung cancer (2 × 106) | C57/BL6 male mice | 0.8 ± 0.1 cm diameter | NR | NR | 15 mg magnetic fluid | IT | 24 h | 150 | Variable to keep temp | 30 | 1 | ~43 | Tumor volume decreased ~38% in MHT treated group and ~71% in MHT + IL-2 group | Improved treatment for Lewis lung cancer in mice when MHT is combined w/ regular IL-2 injections. | [64] |
| Mouse squamous cell carcinoma SCC7 (2 × 105) | NCr nude mice | ~150 mm3 | PEG | 11.3 ± 2.3 (TEM) 23.8 ± 0.1 (DLS) | 1.7 g Fe/Kg body weight | IV (tail vein) | 24 h | 980 | 38 | 2 | 1 | 60 | Complete tumor ablation in 78–90% cases | Muscle w/ MNPs T = 42 °C; Muscle w/o MNPs T = 36 °C; Liver uptake > tumors. 16:1 MNP ration tumor to non-tumor. | [83] |
| Rabbit carcinoma VX-2 (1 × 105) | New Zealand white rabbits | 1.4 cm3 | PLGA | NR | 100 µL of 30% Fe3O4 | IT | 0 | 626 | NR (28.6 A) | 3 | 1 | 72.3 ± 2.2 | MHT-only = Incomplete tumor ablation, if combined w/ cisplatin release = residual tumor elimination by day 21 | Larger tumor and animal model. possible to track by ultrasound or computer tomography. | [17] |
References
- Gilchrist, R.K.; Medal, R.; Shorey, W.D.; Hanselman, R.C.; Parrott, J.C.; Taylor, C.B. Selective inductive heating of lymph nodes. Ann. Surg. 1957, 146, 596–606. [Google Scholar] [CrossRef] [PubMed]
- Gordon, R.T.; Hines, J.R.; Gordon, D. Intracellular hyperthermia. A biophysical approach to cancer treatment via intracellular temperature and biophysical alterations. Med. Hypotheses 1979, 5, 83–102. [Google Scholar] [CrossRef]
- Frank, D.; Tyagi, C.; Tomar, L.; Choonara, Y.E.; Du Toit, L.C.; Kumar, P.; Penny, C.; Pillay, V. Overview of the role of nanotechnological innovations in the detection and treatment of solid tumors. Int. J. Nanomed. 2014, 9, 589–613. [Google Scholar] [CrossRef]
- Gobbo, O.L.; Sjaastad, K.; Radomski, M.W.; Volkov, Y.; Prina-Mello, A. Magnetic nanoparticles in cancer theranostics. Theranostics 2015, 5, 1249–1263. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.P.; Patel, P.B.; Parekh, B.B. Application of nanotechnology in cancers prevention, early detection and treatment. J. Cancer Res. Ther. 2014, 10, 479–486. [Google Scholar] [CrossRef]
- Sohail, A.; Ahmad, Z.; Beg, O.A.; Arshad, S.; Sherin, L. A review on hyperthermia via nanoparticle-mediated therapy. B Cancer 2017, 104, 452–461. [Google Scholar] [CrossRef]
- Armour, E.P.; McEachern, D.; Wang, Z.; Corry, P.M.; Martinez, A. Sensitivity of human cells to mild hyperthermia. Cancer Res. 1993, 53, 2740–2744. [Google Scholar]
- Alvarez-Berrios, M.P.; Castillo, A.; Rinaldi, C.; Torres-Lugo, M. Magnetic fluid hyperthermia enhances cytotoxicity of bortezomib in sensitive and resistant cancer cell lines. Int. J. Nanomedicine 2014, 9, 145–153. [Google Scholar] [CrossRef][Green Version]
- Hildebrandt, B.; Wust, P.; Ahlers, O.; Dieing, A.; Sreenivasa, G.; Kerner, T.; Felix, R.; Riess, H. The cellular and molecular basis of hyperthermia. Crit. Rev. Oncol. Hemat. 2002, 43, 33–56. [Google Scholar] [CrossRef]
- Court, K.A.; Hatakeyama, H.; Wu, S.Y.; Lingegowda, M.S.; Rodriguez-Aguayo, C.; Lopez-Berestein, G.; Ju-Seog, L.; Rinaldi, C.; Juan, E.J.; Sood, A.K.; et al. HSP70 inhibition synergistically enhances the effects of magnetic fluid hyperthermia in ovarian cancer. Mol. Cancer Ther. 2017, 16, 966–976. [Google Scholar] [CrossRef]
- Blanco-Andujar, C.; Ortega, D.; Southern, P.; Nesbitt, S.A.; Thanh, N.T.; Pankhurst, Q.A. Real-time tracking of delayed-onset cellular apoptosis induced by intracellular magnetic hyperthermia. Nanomedicine 2016, 11, 121–136. [Google Scholar] [CrossRef] [PubMed]
- Calatayud, M.P.; Soler, E.; Torres, T.E.; Campos-Gonzalez, E.; Junquera, C.; Ibarra, M.R.; Goya, G.F. Cell damage produced by magnetic fluid hyperthermia on microglial BV2 cells. Sci. Rep. 2017, 7, 8627. [Google Scholar] [CrossRef] [PubMed]
- Hedayati, M.; Thomas, O.; Abubaker-Sharif, B.; Zhou, H.M.; Cornejo, C.; Zhang, Y.G.; Wabler, M.; Mihalic, J.; Gruettner, C.; Westphal, F.; et al. The effect of cell cluster size on intracellular nanoparticle-mediated hyperthermia: Is it possible to treat microscopic tumors? Nanomedicine 2013, 8, 29–41. [Google Scholar] [CrossRef]
- Stocke, N.A.; Sethi, P.; Jyoti, A.; Chan, R.; Arnold, S.M.; Hilt, J.Z.; Upreti, M. Toxicity evaluation of magnetic hyperthermia induced by remote actuation of magnetic nanoparticles in 3D micrometastasic tumor tissue analogs for triple negative breast cancer. Biomaterials 2017, 120, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Mamani, J.B.; Marinho, B.S.; Rego, G.N.A.; Nucci, M.P.; Alvieri, F.; Santos, R.S.D.; Ferreira, J.V.M.; Oliveira, F.A.; Gamarra, L.F. Magnetic hyperthermia therapy in glioblastoma tumor on-a-Chip model. Einstein 2020, 18, eAO4954. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Xie, X.; Yang, Y.; Fu, X.; Liu, H.; Yang, Y.; Deng, J. Thermosensitive magnetic liposomes with doxorubicin cell-penetrating peptides conjugate for enhanced and targeted cancer therapy. Drug Deliv. 2016, 23, 3436–3443. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, F.; Zheng, K.; Deng, L.; Yang, L.; Zhang, N.; Xu, C.; Ran, H.; Wang, Z.; Wang, Z.; et al. Injectable PLGA/Fe3O4 implants carrying cisplatin for synergistic magnetic hyperthermal ablation of rabbit VX2 tumor. PloS ONE 2017, 12, e0177049. [Google Scholar] [CrossRef]
- Kalber, T.L.; Ordidge, K.L.; Southern, P.; Loebinger, M.R.; Kyrtatos, P.G.; Pankhurst, Q.A.; Lythgoe, M.F.; Janes, S.M. Hyperthermia treatment of tumors by mesenchymal stem cell-delivered superparamagnetic iron oxide nanoparticles. Int. J. Nanomed. 2016, 11, 1973–1983. [Google Scholar] [CrossRef]
- Toraya-Brown, S.; Sheen, M.R.; Zhang, P.; Chen, L.; Baird, J.R.; Demidenko, E.; Turk, M.J.; Hoopes, P.J.; Conejo-Garcia, J.R.; Fiering, S. Local hyperthermia treatment of tumors induces CD8(+) T cell-mediated resistance against distal and secondary tumors. Nanomedicine 2014, 10, 1273–1285. [Google Scholar] [CrossRef]
- Hegyi, G.; Szigeti, G.P.; Szasz, A. Hyperthermia versus oncothermia: Cellular effects in complementary cancer therapy. Evid Based Complement. Alternat. Med. 2013, 2013, 672873. [Google Scholar] [CrossRef]
- Vilas-Boas, V.; Espina, B.; Kolen’ko, Y.V.; Banobre-Lopez, M.; Brito, M.; Martins, V.; Duarte, J.A.; Petrovykh, D.Y.; Freitas, P.; Carvalho, F. Effectiveness and Safety of a Nontargeted Boost for a CXCR4-Targeted Magnetic Hyperthermia Treatment of Cancer Cells. ACS Omega 2019, 4, 1931–1940. [Google Scholar] [CrossRef]
- Gao, F.P.; Cai, Y.Y.; Zhou, J.; Xie, X.X.; Ouyang, W.W.; Zhang, Y.H.; Wang, X.F.; Zhang, X.D.; Wang, X.W.; Zhao, L.Y.; et al. Pullulan acetate coated magnetite nanoparticles for hyper-thermia: Preparation, characterization and in vitro experiments. Nano Res. 2010, 3, 23–31. [Google Scholar] [CrossRef]
- Jaidev, L.R.; Chellappan, D.R.; Bhavsar, D.V.; Ranganathan, R.; Sivanantham, B.; Subramanian, A.; Sharma, U.; Jagannathan, N.R.; Krishnan, U.M.; Sethuraman, S. Multi-functional nanoparticles as theranostic agents for the treatment & imaging of pancreatic cancer. Acta Biomater. 2017, 49, 422–433. [Google Scholar] [CrossRef] [PubMed]
- Rochani, A.K.; Balasubramanian, S.; Ravindran Girija, A.; Raveendran, S.; Borah, A.; Nagaoka, Y.; Nakajima, Y.; Maekawa, T.; Kumar, D.S. Dual mode of cancer cell destruction for pancreatic cancer therapy using Hsp90 inhibitor loaded polymeric nano magnetic formulation. Int. J. Pharm. 2016, 511, 648–658. [Google Scholar] [CrossRef]
- Thorat, N.D.; Otari, S.V.; Patil, R.M.; Bohara, R.A.; Yadav, H.M.; Koli, V.B.; Chaurasia, A.K.; Ningthoujam, R.S. Synthesis, characterization and biocompatibility of chitosan functionalized superparamagnetic nanoparticles for heat activated curing of cancer cells. Dalton Trans. 2014, 43, 17343–17351. [Google Scholar] [CrossRef] [PubMed]
- Baskar, G.; Ravi, M.; Panda, J.J.; Khatri, A.; Dev, B.; Santosham, R.; Sathiya, S.; Babu, C.S.; Chauhan, V.S.; Rayala, S.K.; et al. Efficacy of dipeptide-coated magnetic nanoparticles in lung cancer models under pulsed electromagnetic field. Cancer Invest. 2017, 35, 431–442. [Google Scholar] [CrossRef]
- Sadhasivam, S.; Savitha, S.; Wu, C.J.; Lin, F.H.; Stobinski, L. Carbon encapsulated iron oxide nanoparticles surface engineered with polyethylene glycol-folic acid to induce selective hyperthermia in folate over expressed cancer cells. Int. J. Pharm. 2015, 480, 8–14. [Google Scholar] [CrossRef]
- Salimi, M.; Sarkar, S.; Saber, R.; Delavari, H.; Alizadeh, A.M.; Mulder, H.T. Magnetic hyperthermia of breast cancer cells and MRI relaxometry with dendrimer-coated iron-oxide nanoparticles. Cancer Nanotechnol. 2018, 9, 7. [Google Scholar] [CrossRef]
- Liao, S.H.; Liu, C.H.; Bastakoti, B.P.; Suzuki, N.; Chang, Y.; Yamauchi, Y.; Lin, F.H.; Wu, K.C. Functionalized magnetic iron oxide/alginate core-shell nanoparticles for targeting hyperthermia. Int. J. Nanomed. 2015, 10, 3315–3328. [Google Scholar] [CrossRef]
- Shi, B.; Abrams, M.; Sepp-Lorenzino, L. Expression of asialoglycoprotein receptor 1 in human hepatocellular carcinoma. J. Histochem. Cytochem. 2013, 61, 901–909. [Google Scholar] [CrossRef]
- Wu, K.; Su, D.; Liu, J.; Saha, R.; Wang, J.P. Magnetic nanoparticles in nanomedicine: A review of recent advances. Nanotechnology 2019, 30, 502003. [Google Scholar] [CrossRef] [PubMed]
- Lanier, O.L.; Korotych, O.I.; Monsalve, A.G.; Wable, D.; Savliwala, S.; Grooms, N.W.F.; Nacea, C.; Tuitt, O.R.; Dobson, J. Evaluation of magnetic nanoparticles for magnetic fluid hyperthermia. Int. J. Hyperth. 2019, 36, 687–701. [Google Scholar] [CrossRef]
- Khandhar, A.P.; Ferguson, R.M.; Simon, J.A.; Krishnan, K.M. Enhancing cancer therapeutics using size-optimized magnetic fluid hyperthermia. J. Appl. Phys. 2012, 111, 07B306. [Google Scholar] [CrossRef] [PubMed]
- Barrera, G.; Allia, P.; Tiberto, P. Temperature-dependent heating efficiency of magnetic nanoparticles for applications in precision nanomedicine. Nanoscale 2020, 12, 6360–6377. [Google Scholar] [CrossRef]
- Myrovali, E.; Maniotis, N.; Samaras, T.; Angelakeris, M. Spatial focusing of magnetic particle hyperthermia. Nanoscale Adv. 2020, 2, 408. [Google Scholar] [CrossRef]
- Lu, A.H.; Salabas, E.L.; Schuth, F. Magnetic nanoparticles: Synthesis, protection, functionalization, and application. Angew. Chem. Int. Ed. 2007, 46, 1222–1244. [Google Scholar] [CrossRef]
- Pamme, N. Magnetism and microfluidics. Lab Chip 2006, 6, 24–38. [Google Scholar] [CrossRef] [PubMed]
- Bychkova, A.V.; Sorokina, O.N.; Rosenfeld, M.A.; Kovarski, A.L. Multifunctional biocompatible coatings on magnetic nanoparticles. Russ. Chem. Rev. 2012, 81, 1026–1050. [Google Scholar] [CrossRef]
- Iyer, A.K.; Khaled, G.; Fang, J.; Maeda, H. Exploiting the enhanced permeability and retention effect for tumor targeting. Drug Discov. Today 2006, 11, 812–818. [Google Scholar] [CrossRef]
- Stylianopoulos, T. EPR-effect: Utilizing size-dependent nanoparticle delivery to solid tumors. Ther. Deliv. 2013, 4, 421–423. [Google Scholar] [CrossRef]
- Danhier, F. To exploit the tumor microenvironment: Since the EPR effect fails in the clinic, what is the future of nanomedicine? J. Control. Release 2016, 244, 108–121. [Google Scholar] [CrossRef] [PubMed]
- Rejman, J.; Oberle, V.; Zuhorn, I.S.; Hoekstra, D. Size-dependent internalization of particles via the pathways of clathrin- and caveolae-mediated endocytosis. Biochem. J. 2004, 377, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Saptarshi, S.R.; Duschl, A.; Lopata, A.L. Interaction of nanoparticles with proteins: Relation to bio-reactivity of the nanoparticle. J. Nanobiotechnology 2013, 11, 26. [Google Scholar] [CrossRef] [PubMed]
- Das, P.; Colombo, M.; Prosperi, D. Recent advances in magnetic fluid hyperthermia for cancer therapy. Colloids Surf. B: Biointerfaces 2019, 174, 42–55. [Google Scholar] [CrossRef]
- Jang, J.T.; Lee, J.; Seon, J.; Ju, E.; Kim, M.; Kim, Y.I.; Kim, M.G.; Takemura, Y.; Arbab, A.S.; Kang, K.W.; et al. Giant magnetic heat induction of magnesium-doped gamma-Fe2O3 superparamagnetic nanoparticles for completely killing tumors. Adv. Mater. 2018, 30. [Google Scholar] [CrossRef]
- Tapeinos, C.; Marino, A.; Battaglini, M.; Migliorin, S.; Brescia, R.; Scarpellini, A.; De Julian Fernandez, C.; Prato, M.; Drago, F.; Ciofani, G. Stimuli-responsive lipid-based magnetic nanovectors increase apoptosis in glioblastoma cells through synergic intracellular hyperthermia and chemotherapy. Nanoscale 2018, 11, 72–88. [Google Scholar] [CrossRef]
- Hervault, A.; Thanh, N.T.K. Magnetic nanoparticle-based therapeutic agents for thermo-chemotherapy treatment of cancer. Nanoscale 2014, 6, 11553–11573. [Google Scholar] [CrossRef]
- Hanini, A.; Lartigue, L.; Gavard, J.; Kacem, K.; Wilhelm, C.; Gazeau, F.; Chau, F.; Ammar, S. Zinc substituted ferrite nanoparticles with Zn0.9Fe2.1O4 formula used as heating agents for in vitro hyperthermia assay on glioma cells. J. Magn. Magn. Mater. 2016, 416, 315–320. [Google Scholar] [CrossRef]
- Hedayatnasab, Z.; Abnisa, F.; Daud, W.M.A.W. Review on magnetic nanoparticles for magnetic nanofluid hyperthermia application. Mater. Design 2017, 123, 174–196. [Google Scholar] [CrossRef]
- Osaci, M.; Cacciola, M. The influence of the magnetic nanoparticles coating from colloidal system on the magnetic relaxation time. Beilstein Arch. 2019. [Google Scholar] [CrossRef]
- Salunkhe, A.B.; Khot, V.M.; Pawar, S.H. Magnetic hyperthermia with magnetic nanoparticles: A status review. Curr. Top. Med. Chem. 2014, 14, 572–594. [Google Scholar] [CrossRef] [PubMed]
- Sperling, R.A.; Parak, W.J. Surface modification, functionalization and bioconjugation of colloidal inorganic nanoparticles. Philos. Trans. A Math. Phys. Eng. Sci. 2010, 368, 1333–1383. [Google Scholar] [CrossRef] [PubMed]
- Pirollo, K.F.; Chang, E.H. Does a targeting ligand influence nanoparticle tumor localization or uptake? Trends Biotechnol 2008, 26, 552–558. [Google Scholar] [CrossRef]
- NDong, C.; Tate, J.A.; Kett, W.C.; Batra, J.; Demidenko, E.; Lewis, L.D.; Hoopes, P.J.; Gerngross, T.U.; Griswold, K.E. Tumor cell targeting by iron oxide nanoparticles is dominated by different factors in vitro versus in vivo. PloS ONE 2015, 10, e0115636. [Google Scholar] [CrossRef] [PubMed]
- Lesniak, A.; Salvati, A.; Santos-Martinez, M.J.; Radomski, M.W.; Dawson, K.A.; Aberg, C. Nanoparticle adhesion to the cell membrane and its effect on nanoparticle uptake efficiency. J. Am. Chem. Soc. 2013, 135, 1438–1444. [Google Scholar] [CrossRef] [PubMed]
- Manigandan, A.; Handi, V.; Sundaramoorthy, N.S.; Dhandapani, R.; Radhakrishnan, J.; Sethuraman, S.; Subramanian, A. Responsive nanomicellar theranostic cages for metastatic breast cancer. Bioconjug. Chem. 2018, 29, 275–286. [Google Scholar] [CrossRef]
- Wang, X.; Yang, R.; Yuan, C.; An, Y.; Tang, Q.; Chen, D. Preparation of folic acid-targeted temperature-sensitive magnetoliposomes and their antitumor effects in vitro and in vivo. Target. Oncol. 2018, 13, 481–494. [Google Scholar] [CrossRef]
- Mi, Y.; Liu, X.; Zhao, J.; Ding, J.; Feng, S.S. Multimodality treatment of cancer with herceptin conjugated, thermomagnetic iron oxides and docetaxel loaded nanoparticles of biodegradable polymers. Biomaterials 2012, 33, 7519–7529. [Google Scholar] [CrossRef]
- Tang, J.; Zhou, H.; Liu, J.; Liu, J.; Li, W.; Wang, Y.; Hu, F.; Huo, Q.; Li, J.; Liu, Y.; et al. Dual-mode imaging-guided synergistic chemo- and magnetohyperthermia therapy in a versatile nanoplatform to eliminate cancer stem cells. ACS Appl. Mater. Interfaces 2017, 9, 23497–23507. [Google Scholar] [CrossRef]
- Zolata, H.; Afarideh, H.; Davani, F.A. Triple therapy of HER2(+) cancer using radiolabeled multifunctional iron oxide nanoparticles and alternating magnetic field. Cancer Biother. Radiopharm. 2016, 31, 324–329. [Google Scholar] [CrossRef]
- Vilas-Boas, V.; Espina, B.; Kolen’ko, Y.V.; Banobre-Lopez, M.; Duarte, J.A.; Martins, V.C.; Petrovykh, D.Y.; Freitas, P.P.; Carvalho, F.D. Combining CXCR4-targeted and nontargeted nanoparticles for effective unassisted in vitro magnetic hyperthermia. Biointerphases 2018, 13, 011005. [Google Scholar] [CrossRef] [PubMed]
- Zwicke, G.L.; Mansoori, G.A.; Jeffery, C.J. Utilizing the folate receptor for active targeting of cancer nanotherapeutics. Nano Rev. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Mitri, Z.; Constantine, T.; O’Regan, R. The HER2 receptor in breast cancer: Pathophysiology, clinical use, and new advances in therapy. Chemother. Res. Pract. 2012, 2012, 743193. [Google Scholar] [CrossRef] [PubMed]
- Hu, R.; Ma, S.; Ke, X.; Jiang, H.; Wei, D.; Wang, W. Effect of interleukin-2 treatment combined with magnetic fluid hyperthermia on Lewis lung cancer-bearing mice. Biomed. Rep. 2016, 4, 59–62. [Google Scholar] [CrossRef]
- Prijic, S.; Prosen, L.; Cemazar, M.; Scancar, J.; Romih, R.; Lavrencak, J.; Bregar, V.B.; Coer, A.; Krzan, M.; Znidarsic, A.; et al. Surface modified magnetic nanoparticles for immuno-gene therapy of murine mammary adenocarcinoma. Biomaterials 2012, 33, 4379–4391. [Google Scholar] [CrossRef]
- Rago, G.; Bauer, B.; Svedberg, F.; Gunnarsson, L.; Ericson, M.B.; Bonn, M.; Enejder, A. Uptake of gold nanoparticles in healthy and tumor cells visualized by nonlinear optical microscopy. J. Phys. Chem. B 2011, 115, 5008–5016. [Google Scholar] [CrossRef]
- Sims, L.B.; Curtis, L.T.; Frieboes, H.B.; Steinbach-Rankins, J.M. Enhanced uptake and transport of PLGA-modified nanoparticles in cervical cancer. J. Nanobiotechnology 2016, 14, 33. [Google Scholar] [CrossRef]
- Guardia, P.; Di Corato, R.; Lartigue, L.; Wilhelm, C.; Espinosa, A.; Garcia-Hernandez, M.; Gazeau, F.; Manna, L.; Pellegrino, T. Water-soluble iron oxide nanocubes with high values of specific absorption rate for cancer cell hyperthermia treatment. ACS Nano 2012, 6, 3080–3091. [Google Scholar] [CrossRef]
- Taratula, O.; Dani, R.K.; Schumann, C.; Xu, H.; Wang, A.; Song, H.; Dhagat, P.; Taratula, O. Multifunctional nanomedicine platform for concurrent delivery of chemotherapeutic drugs and mild hyperthermia to ovarian cancer cells. Int. J. Pharm. 2013, 458, 169–180. [Google Scholar] [CrossRef]
- Kandasamy, G.; Sudame, A.; Bhati, P.; Chakrabarty, A.; Kale, S.N.; Maity, D. Systematic magnetic fluid hyperthermia studies of carboxyl functionalized hydrophilic superparamagnetic iron oxide nanoparticles based ferrofluids. J. Colloid Interface Sci. 2018, 514, 534–543. [Google Scholar] [CrossRef]
- Ma, M.; Zhang, Y.; Shen, X.L.; Xie, J.; Li, Y.; Gu, N. Targeted inductive heating of nanomagnets by a combination of alternating current (AC) and static magnetic fields. Nano Res. 2015, 8, 600–610. [Google Scholar] [CrossRef]
- Maity, D.; Chandrasekharan, P.; Pradhan, P.; Chuang, K.H.; Xue, J.M.; Feng, S.S.; Ding, J. Novel synthesis of superparamagnetic magnetite nanoclusters for biomedical applications. J. Mater. Chem 2011, 21, 14717–14724. [Google Scholar] [CrossRef]
- Oh, Y.; Lee, N.; Kang, H.W.; Oh, J. In vitro study on apoptotic cell death by effective magnetic hyperthermia with chitosan-coated MnFe2O4. Nanotechnology 2016, 27, 115101. [Google Scholar] [CrossRef]
- Qu, Y.; Li, J.; Ren, J.; Leng, J.; Lin, C.; Shi, D. Enhanced magnetic fluid hyperthermia by micellar magnetic nanoclusters composed of Mn(x)Zn(1-x)Fe2O4 nanoparticles for induced tumor cell apoptosis. ACS Appl. Mater. Inter. 2014, 6, 16867–16879. [Google Scholar] [CrossRef] [PubMed]
- Sadhukha, T.; Niu, L.; Wiedmann, T.S.; Panyam, J. Effective elimination of cancer stem cells by magnetic hyperthermia. Mol. Pharm. 2013, 10, 1432–1441. [Google Scholar] [CrossRef] [PubMed]
- Hergt, R.; Dutz, S. Magnetic particle hyperthermia-biophysical limitations of a visionary tumour therapy. J. Magn. Magn. Mater. 2007, 311, 187–192. [Google Scholar] [CrossRef]
- Lin, M.; Huang, J.; Jiang, X.; Zhang, J.; Yu, H.; Ye, J.; Zhang, D. A combination hepatoma-targeted therapy based on nanotechnology: pHRE-Egr1-HSV-TK/(131)I-antiAFPMcAb-GCV/MFH. Sci. Rep. 2016, 6, 33524. [Google Scholar] [CrossRef]
- Engelmann, U.M.; Roeth, A.A.; Eberbeck, D.; Buhl, E.M.; Neumann, U.P.; Schmitz-Rode, T.; Slabu, I. Combining bulk temperature and nanoheating enables advanced magnetic fluid hyperthermia efficacy on pancreatic tumor cells. Sci. Rep. 2018, 8, 13210. [Google Scholar] [CrossRef]
- Gaharwar, U.S.; Meena, R.; Rajamani, P. Biodistribution, clearance and morphological alterations of intravenously administered iron oxide nanoparticles in male wistar rats. Int. J. Nanomed. 2019, 14, 9677–9692. [Google Scholar] [CrossRef]
- Baldi, G.; Ravagli, C.; Mazzantini, F.; Loudos, G.; Adan, J.; Masa, M.; Psimadas, D.; Fragogeorgi, E.A.; Locatelli, E.; Innocenti, C.; et al. In vivo anticancer evaluation of the hyperthermic efficacy of anti-human epidermal growth factor receptor-targeted PEG-based nanocarrier containing magnetic nanoparticles. Int. J Nanomed. 2014, 9, 3037–3056. [Google Scholar] [CrossRef][Green Version]
- Xie, J.; Yan, C.; Yan, Y.; Chen, L.; Song, L.; Zang, F.; An, Y.; Teng, G.; Gu, N.; Zhang, Y. Multi-modal Mn-Zn ferrite nanocrystals for magnetically-induced cancer targeted hyperthermia: A comparison of passive and active targeting effects. Nanoscale 2016. [Google Scholar] [CrossRef] [PubMed]
- Kirpotin, D.B.; Drummond, D.C.; Shao, Y.; Shalaby, M.R.; Hong, K.; Nielsen, U.B.; Marks, J.D.; Benz, C.C.; Park, J.W. Antibody targeting of long-circulating lipidic nanoparticles does not increase tumor localization but does increase internalization in animal models. Cancer Res. 2006, 66, 6732–6740. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.S.; Hainfeld, J.F. Intravenous magnetic nanoparticle cancer hyperthermia. Int. J. Nanomed. 2013, 8, 2521–2532. [Google Scholar] [CrossRef]
- Gao, W.; Zheng, Y.; Wang, R.; Chen, H.; Cai, X.; Lu, G.; Chu, L.; Xu, C.; Zhang, N.; Wang, Z.; et al. A smart, phase transitional and injectable DOX/PLGA-Fe implant for magnetic-hyperthermia-induced synergistic tumor eradication. Acta Biomater. 2016, 29, 298–306. [Google Scholar] [CrossRef]
- Seidel, K.; Balakrishnan, A.; Alexiou, C.; Janko, C.; Komoll, R.M.; Wang, L.L.; Kirschning, A.; Ott, M. Synthesis of magnetic-nanoparticle/ansamitocin conjugates-inductive heating leads to decreased cell proliferation in vitro and attenuation of tumour growth in vivo. Chemistry 2017, 23, 12326–12337. [Google Scholar] [CrossRef]
- Teo, P.; Wang, X.; Chen, B.; Zhang, H.; Yang, X.; Huang, Y.; Tang, J. Complex of TNF-alpha and modified Fe3O4 nanoparticles suppresses tumor growth by magnetic induction hyperthermia. Cancer Biother. Radiopharm. 2017, 32, 379–386. [Google Scholar] [CrossRef]
- Yang, R.; An, L.Y.; Miao, Q.F.; Li, F.M.; Han, Y.; Wang, H.X.; Liu, D.P.; Chen, R.; Tang, S.Q. Effective elimination of liver cancer stem-like cells by CD90 antibody targeted thermosensitive magnetoliposomes. Oncotarget 2016, 7, 35894–35916. [Google Scholar] [CrossRef]
- Yuan, C.; An, Y.; Zhang, J.; Li, H.; Zhang, H.; Wang, L.; Zhang, D. Magnetic nanoparticles for targeted therapeutic gene delivery and magnetic-inducing heating on hepatoma. Nanotechnology 2014, 25, 345101. [Google Scholar] [CrossRef][Green Version]
- Hervault, A.; Dunn, A.E.; Lim, M.; Boyer, C.; Mott, D.; Maenosono, S.; Thanh, N.T. Doxorubicin loaded dual pH- and thermo-responsive magnetic nanocarrier for combined magnetic hyperthermia and targeted controlled drug delivery applications. Nanoscale 2016, 8, 12152–12161. [Google Scholar] [CrossRef]
- Soetaert, F.; Kandala, S.K.; Bakuzis, A.; Ivkov, R. Experimental estimation and analysis of variance of the measured loss power of magnetic nanoparticles. Sci. Rep. 2017, 7, 6661. [Google Scholar] [CrossRef]
- Makridis, A.; Tziomaki, M.; Topouridou, K.; Yavropoulou, M.P.; Yovos, J.G.; Kalogirou, O.; Samaras, T.; Angelakeris, M. A novel strategy combining magnetic particle hyperthermia pulses with enhanced performance binary ferrite carriers for effective in vitro manipulation of primary human osteogenic sarcoma cells. Int. J. Hyperth. 2016, 32, 778–785. [Google Scholar] [CrossRef] [PubMed]
- Kumar, C.S.; Mohammad, F. Magnetic nanomaterials for hyperthermia-based therapy and controlled drug delivery. Adv. Drug Deliv. Rev. 2011, 63, 789–808. [Google Scholar] [CrossRef] [PubMed]
- Di Corato, R.; Bealle, G.; Kolosnjaj-Tabi, J.; Espinosa, A.; Clement, O.; Silva, A.K.; Menager, C.; Wilhelm, C. Combining magnetic hyperthermia and photodynamic therapy for tumor ablation with photoresponsive magnetic liposomes. ACS Nano 2015, 9, 2904–2916. [Google Scholar] [CrossRef] [PubMed]
- Espinosa, A.; Di Corato, R.; Kolosnjaj-Tabi, J.; Flaud, P.; Pellegrino, T.; Wilhelm, C. Duality of iron oxide nanoparticles in cancer therapy: Amplification of heating efficiency by magnetic hyperthermia and photothermal bimodal treatment. ACS Nano 2016, 10, 2436–2446. [Google Scholar] [CrossRef]
- Thompson, S.A.; Martínez, I.A.; Haro-González, P.; Adam, A.P.; Jaque, D.; Nieder, J.B.; De la Rica, R. Plug and play anisotropy-based nanothermometers. ACS Photonics 2018, 5, 2676–2681. [Google Scholar] [CrossRef]
- Wust, P.; Hildebrandt, B.; Sreenivasa, G.; Rau, B.; Gellermann, J.; Riess, H.; Felix, R.; Schlag, P.M. Hyperthermia in combined treatment of cancer. Lancet Oncol. 2002, 3, 487–497. [Google Scholar] [CrossRef]
- Maier-Hauff, K.; Ulrich, F.; Nestler, D.; Niehoff, H.; Wust, P.; Thiesen, B.; Orawa, H.; Budach, V.; Jordan, A. Efficacy and safety of intratumoral thermotherapy using magnetic iron-oxide nanoparticles combined with external beam radiotherapy on patients with recurrent glioblastoma multiforme. J. Neurooncol 2011, 103, 317–324. [Google Scholar] [CrossRef]
- Song, C.W. Effect of local hyperthermia on blood-flow and microenvironment—a review. Cancer Res. 1984, 44, 4721–4730. [Google Scholar]
- Zhang, Z.Q.; Song, S.C. Thermosensitive/superparamagnetic iron oxide nanoparticle-loaded nanocapsule hydrogels for multiple cancer hyperthermia. Biomaterials 2016, 106, 13–23. [Google Scholar] [CrossRef]
- Sanz, B.; Calatayud, M.P.; Torres, T.E.; Fanarraga, M.L.; Ibarra, M.R.; Goya, G.F. Magnetic hyperthermia enhances cell toxicity with respect to exogenous heating. Biomaterials 2017, 114, 62–70. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Vistica, D.T.; Skehan, P.; Scudiero, D.; Monks, A.; Pittman, A.; Boyd, M.R. Tetrazolium-based assays for cellular viability: A critical examination of selected parameters affecting formazan production. Cancer Res. 1991, 51, 2515–2520. [Google Scholar] [PubMed]
- Kroll, A.; Pillukat, M.H.; Hahn, D.; Schnekenburger, J. Interference of engineered nanoparticles with in vitro toxicity assays. Arch. Toxicol. 2012, 86, 1123–1136. [Google Scholar] [CrossRef] [PubMed]
- Strober, W. Trypan blue exclusion test of cell viability. Curr. Protoc. Immunol. 2015, 111, A3B1–A3B3. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.Q.; Song, S.C. Multiple hyperthermia-mediated release of TRAIL/SPION nanocomplex from thermosensitive polymeric hydrogels for combination cancer therapy. Biomaterials 2017, 132, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Tay, Z.W.; Chandrasekharan, P.; Chiu-Lam, A.; Hensley, D.W.; Dhavalikar, R.; Zhou, X.Y.; Yu, E.Y.; Goodwill, P.W.; Zheng, B.; Rinaldi, C.; et al. Magnetic particle imaging-guided heating in vivo using gradient fields for arbitrary localization of magnetic hyperthermia therapy. ACS Nano 2018, 12, 3699–3713. [Google Scholar] [CrossRef]
- Khandhar, A.P.; Ferguson, R.M.; Simon, J.A.; Krishnan, K.M. Tailored magnetic nanoparticles for optimizing magnetic fluid hyperthermia. J. Biomed. Mater. Res. A 2012, 100A, 728–737. [Google Scholar] [CrossRef]
- Gupta, J.; Mohapatra, J.; Bhargava, P.; Bahadur, D. A pH-responsive folate conjugated magnetic nanoparticle for targeted chemo-thermal therapy and MRI diagnosis. Dalton Trans. 2016, 45, 2454–2461. [Google Scholar] [CrossRef]
- Ramachandra Kurup Sasikala, A.; Thomas, R.G.; Unnithan, A.R.; Saravanakumar, B.; Jeong, Y.Y.; Park, C.H.; Kim, C.S. Multifunctional nanocarpets for cancer theranostics: Remotely controlled graphene nanoheaters for thermo-chemosensitisation and magnetic resonance imaging. Sci. Rep. 2016, 6, 20543. [Google Scholar] [CrossRef]
- Aguilar, L.E.; GhavamiNejad, A.; Park, C.H.; Kim, C.S. On-demand drug release and hyperthermia therapy applications of thermoresponsive poly-(NIPAAm-co-HMAAm)/polyurethane core-shell nanofiber mat on non-vascular nitinol stents. Nanomedicine 2017, 13, 527–538. [Google Scholar] [CrossRef]
- Quinto, C.A.; Mohindra, P.; Tong, S.; Bao, G. Multifunctional superparamagnetic iron oxide nanoparticles for combined chemotherapy and hyperthermia cancer treatment. Nanoscale 2015. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.T.; Li, K.Y.; Meng, F.Q.; Lin, J.F.; Young, I.C.; Ivkov, R.; Lin, F.H. ROS-induced HepG2 cell death from hyperthermia using magnetic hydroxyapatite nanoparticles. Nanotechnology 2018, 29, 375101. [Google Scholar] [CrossRef] [PubMed]
- Gogoi, M.; Jaiswal, M.K.; Sarma, H.D.; Bahadur, D.; Banerjee, R. Biocompatibility and therapeutic evaluation of magnetic liposomes designed for self-controlled cancer hyperthermia and chemotherapy. Integr. Biol. 2017, 9, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Sharma, K.S.; Ningthoujam, R.S.; Dubey, A.K.; Chattopadhyay, A.; Phapale, S.; Juluri, R.R.; Mukherjee, S.; Tewari, R.; Shetake, N.G.; Pandey, B.N.; et al. Synthesis and characterization of monodispersed water dispersible Fe3O4 nanoparticles and in vitro studies on human breast carcinoma cell line under hyperthermia condition. Sci. Rep. 2018, 8, 14766. [Google Scholar] [CrossRef]
- Yoo, D.; Jeong, H.; Preihs, C.; Choi, J.S.; Shin, T.H.; Sessler, J.L.; Cheon, J. Double-effector nanoparticles: A synergistic approach to apoptotic hyperthermia. Angew. Chem. Int. Ed. 2012, 51, 12482–12485. [Google Scholar] [CrossRef] [PubMed]
- Herea, D.D.; Danceanu, C.; Radu, E.; Labusca, L.; Lupu, N.; Chiriac, H. Comparative effects of magnetic and water-based hyperthermia treatments on human osteosarcoma cells. Int. J. Nanomed. 2018, 13, 5743–5751. [Google Scholar] [CrossRef]
- Yin, P.T.; Shah, B.P.; Lee, K.B. Combined magnetic nanoparticle-based microRNA and hyperthermia therapy to enhance apoptosis in brain cancer cells. Small 2014, 10, 4106–4112. [Google Scholar] [CrossRef]
- Kruse, A.M.; Meenach, S.A.; Anderson, K.W.; Hilt, J.Z. Synthesis and characterization of CREKA-conjugated iron oxide nanoparticles for hyperthermia applications. Acta Biomater. 2014, 10, 2622–2629. [Google Scholar] [CrossRef]
- Sivakumar, B.; Aswathy, R.G.; Nagaoka, Y.; Suzuki, M.; Fukuda, T.; Yoshida, Y.; Maekawa, T.; Sakthikumar, D.N. Multifunctional carboxymethyl cellulose-based magnetic nanovector as a theragnostic system for folate receptor targeted chemotherapy, imaging, and hyperthermia against cancer. Langmuir 2013, 29, 3453–3466. [Google Scholar] [CrossRef]
- Ito, A.; Kuga, Y.; Honda, H.; Kikkawa, H.; Horiuchi, A.; Watanabe, Y.; Kobayashi, T. Magnetite nanoparticle-loaded anti-HER2 immunoliposomes for combination of antibody therapy with hyperthermia. Cancer Lett. 2004, 212, 167–175. [Google Scholar] [CrossRef]
- Thomas, R.G.; Moon, M.J.; Lee, H.; Sasikala, A.R.; Kim, C.S.; Park, I.K.; Jeong, Y.Y. Hyaluronic acid conjugated superparamagnetic iron oxide nanoparticle for cancer diagnosis and hyperthermia therapy. Carbohydr Polym. 2015, 131, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Shah, B.P.; Pasquale, N.; De, G.; Tan, T.; Ma, J.; Lee, K.B. Core-shell nanoparticle-based peptide therapeutics and combined hyperthermia for enhanced cancer cell apoptosis. ACS Nano 2014, 8, 9379–9387. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Xie, X.; Xu, X.; Xia, X.; Wang, H.; Li, L.; Dong, W.; Ma, P.; Yang, Y.; Liu, Y.; et al. Thermal and magnetic dual-responsive liposomes with a cell-penetrating peptide-siRNA conjugate for enhanced and targeted cancer therapy. Colloids surf B: Biointerfaces 2016, 146, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Zhang, H.; Chen, B.; Cheng, J.; Cai, X.; Liu, R.; Xia, G.; Wu, W.; Wang, S.; Ding, J.; et al. Multifunctional magnetic Fe3O4 nanoparticles combined with chemotherapy and hyperthermia to overcome multidrug resistance. Int. J. Nanomed. 2012, 7, 2261–2269. [Google Scholar] [CrossRef]
- Ohtake, M.; Umemura, M.; Sato, I.; Akimoto, T.; Oda, K.; Nagasako, A.; Kim, J.H.; Fujita, T.; Yokoyama, U.; Nakayama, T.; et al. Hyperthermia and chemotherapy using Fe(Salen) nanoparticles might impact glioblastoma treatment. Sci. Rep. 2017, 7, 42783. [Google Scholar] [CrossRef]
- Sadhukha, T.; Wiedmann, T.S.; Panyam, J. Inhalable magnetic nanoparticles for targeted hyperthermia in lung cancer therapy. Biomaterials 2013, 34, 5163–5171. [Google Scholar] [CrossRef]
| Number of Studies | Hf > 5 × 109 A∙m−1∙s−1 | Hf < 5 × 109 A∙m−1∙s−1 | No Reported T |
|---|---|---|---|
| 72 | 37 | 24 | 17 |
| % | 51.4% | 33.3% | 23.6% |
| Mild Hyperthermia | Thermoablation | ||
|---|---|---|---|
| T (°C) | 41–43 | >45 | |
| Tumor tissue | Increased blood supply Increased oxygenation | Decreased blood supply Lower dissipation rate | |
| Tumor cells | Protein denaturation ER stress response activation Inhibition of DNA repair APOPTOSIS | Induction of HSP THERMOTOLERANCE | Protein denaturation Cytoskeleton rearrangement Membrane disruption Altered DNA conformation NECROSIS |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vilas-Boas, V.; Carvalho, F.; Espiña, B. Magnetic Hyperthermia for Cancer Treatment: Main Parameters Affecting the Outcome of In Vitro and In Vivo Studies. Molecules 2020, 25, 2874. https://doi.org/10.3390/molecules25122874
Vilas-Boas V, Carvalho F, Espiña B. Magnetic Hyperthermia for Cancer Treatment: Main Parameters Affecting the Outcome of In Vitro and In Vivo Studies. Molecules. 2020; 25(12):2874. https://doi.org/10.3390/molecules25122874
Chicago/Turabian StyleVilas-Boas, Vânia, Félix Carvalho, and Begoña Espiña. 2020. "Magnetic Hyperthermia for Cancer Treatment: Main Parameters Affecting the Outcome of In Vitro and In Vivo Studies" Molecules 25, no. 12: 2874. https://doi.org/10.3390/molecules25122874
APA StyleVilas-Boas, V., Carvalho, F., & Espiña, B. (2020). Magnetic Hyperthermia for Cancer Treatment: Main Parameters Affecting the Outcome of In Vitro and In Vivo Studies. Molecules, 25(12), 2874. https://doi.org/10.3390/molecules25122874