Applications of a Standardized Green Tea Catechin Preparation for Viral Warts and Human Papilloma Virus-Related and Unrelated Cancers
Abstract
1. Introduction
2. The Efficacy of PSV on GW and Proactive Sequential Therapy after Ablative Treatments
3. PSV Effects on Other HPV-Related Diseases
4. Case Eeports of PSV Effects on Viral Warts and Other HPV-Related Diseases
5. PSV Effects on HPV-Unrelated Cancers
6. Clinical Studies of PSV Effects on Other Diseases
7. Inconsistency among Study Results and Adverse Effects of PSV
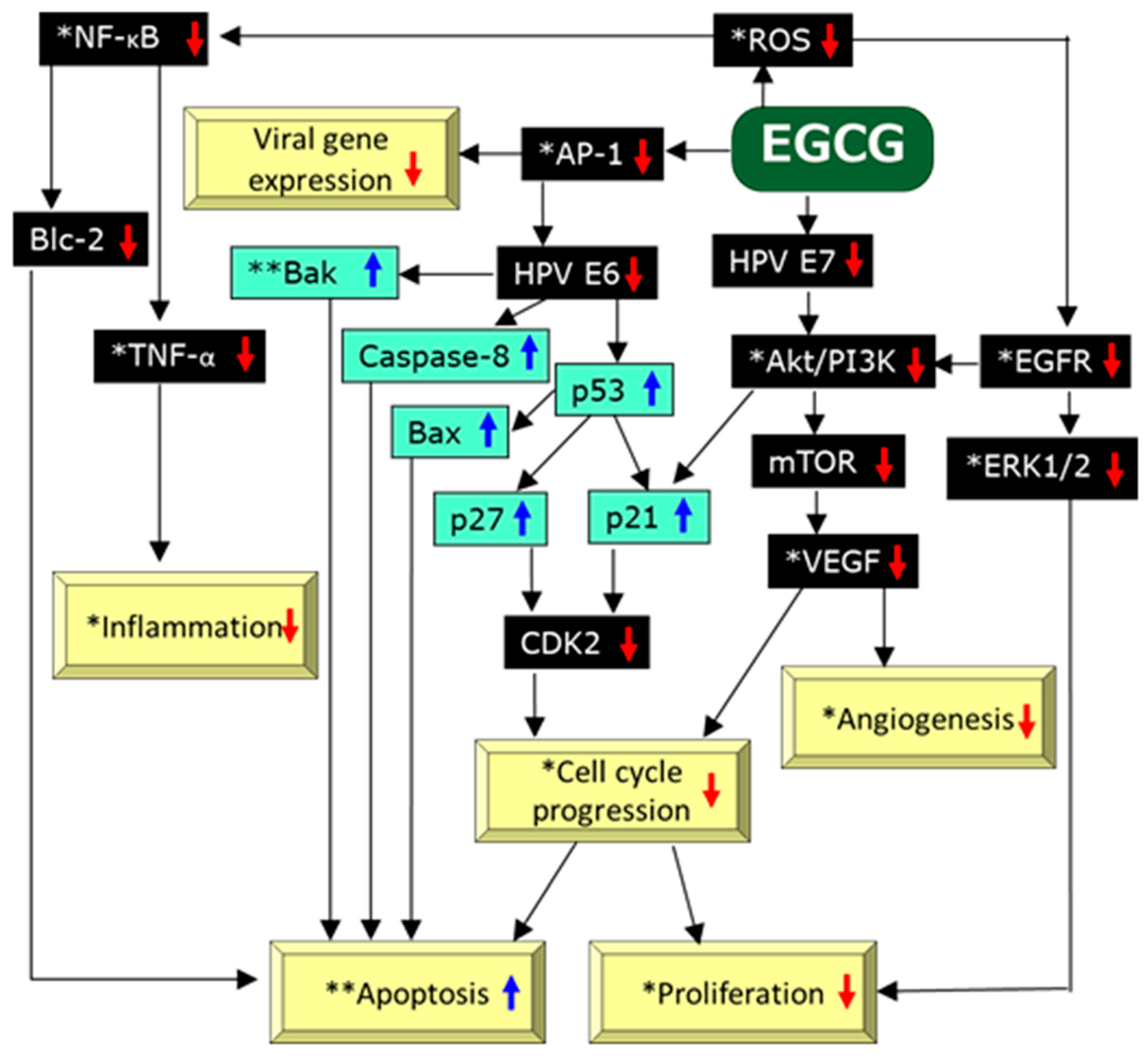
8. Mechanistic Consideration
Author Contributions
Funding
Conflicts of Interest
References
- Hara, Y. Tea catechins and their applications as supplements and pharmaceutics. Pharmacol. Res. 2011, 64, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Narotzki, B.; Reznick, A.Z.; Aizenbud, D.; Levy, Y. Green tea: A promising natural product in oral health. Arch. Oral Biol. 2012, 57, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Chacko, S.M.; Thambi, P.T.; Kuttan, R.; Nishigaki, I. Beneficial effects of green tea: A literature review. Chin. Med. 2010, 5, 13. [Google Scholar] [CrossRef] [PubMed]
- Dunne, E.F.; Friedman, A.; Datta, S.D.; Markowitz, L.E.; Workowski, K.A. Updates on human papillomavirus and genital warts and counseling messages from the 2010 Sexually Transmitted Diseases Treatment Guidelines. Clin. Infect. Dis. 2011, 53, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Lee, S.-L.; Taylor, C.; Li, J.; Chan, Y.-M.; Agarwal, R.; Temple, R.; Throckmorton, D.; Tyner, K. Scientific and regulatory approach to botanical drug development: A U.S. FDA Perspective. J. Nat. Prod. 2020, 83, 552–562. [Google Scholar] [CrossRef]
- Gross, G.; Meyer, K.-G.; Pres, H.; Thielert, C.; Tawfik, H.; Mescheder, A. A randomized, double-blind, four-arm parallel-group, placebo-controlled Phase II/III study to investigate the clinical efficacy of two galenic formulations of Polyphenon E in the treatment of external genital warts. J. Eur. Acad. Dermatol. Venereol. 2007, 21, 1404–1412. [Google Scholar] [CrossRef]
- Stockfleth, E.; Beti, H.; Orasan, R.; Grigorian, F.; Mescheder, A.; Tawfik, H.; Thielert, C. Topical Polyphenon E in the treatment of external genital and perianal warts: A randomized controlled trial. Br. J. Dermatol. 2008, 158, 1329–1338. [Google Scholar] [CrossRef]
- Tatti, S.; Swinehart, J.M.; Thielert, C.; Tawfik, H.; Mescheder, A.; Beutner, K.R. Sinecatechins, a defined green tea extract, in the treatment of external anogenital warts: A randomized controlled trial. Obstet. Gynecol. 2008, 111, 1371–1379. [Google Scholar] [CrossRef]
- Tzellos, T.G.; Sardeli, C.; Lallas, A.; Papazisis, G.; Chourdakis, M.; Kouvelas, D. Efficacy, safety and tolerability of green tea catechins in the treatment of external anogenital warts: A systematic review and meta-analysis. J. Eur. Acad. Dermatol. Venereol. 2011, 25, 345–353. [Google Scholar] [CrossRef]
- Kollipara, R.; Ekhlassi, E.; Downing, C.; Guidry, J.; Lee, M.; Tyring, S.K. Advancements in pharmacotherapy for noncancerous manifestations of HPV. J. Clin. Med. 2015, 4, 832–846. [Google Scholar] [CrossRef]
- Werner, R.N.; Westfechtel, L.; Dressler, C.; Nast, A. Self-administered interventions for anogenital warts in immunocompetent patients: A systematic review and meta-analysis. Sex. Transm. Infect. 2017, 93, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Barton, S.; Wakefield, V.; O’Mahony, C.; Edwards, S. Effectiveness of topical and ablative therapies in treatment of anogenital warts: A systematic review and network meta-analysis. BMJ Open 2019, 9, e027765. [Google Scholar] [CrossRef] [PubMed]
- Deeb, M.; Levy, R.; Pope, E.; Lara-Corrales, I. Sinecatechins ointment for the treatment of warts in children. Pediatr. Dermatol. 2019, 36, 121–124. [Google Scholar] [CrossRef] [PubMed]
- On, S.C.J.; Linkner, R.V.; Haddican, M.; Yaroshinsky, A.; Gagliotti, M.; Singer, G.; Goldenberg, G. A single-blinded randomized controlled study to assess the efficacy of twice daily application of sinecatechins 15% ointment when used sequentially with cryotherapy in the treatment of external genital warts. J. Drugs Dermatol. 2014, 13, 1400–1405. [Google Scholar]
- Juhl, M.E.; Seferovic, V.; Antonijevic, S.; Krunic, A. Combined treatment of anogenital HPV infection with cryodestruction, podophyllin 25% and post-ablation immunomodulation with sinecatechins 15% ointment—A retrospective analysis. Int. J. Std Aids 2016, 27, 1071–1078. [Google Scholar] [CrossRef]
- Stockfleth, E.; Meyer, T. The use of sinecatechins (polyphenon E) ointment for treatment of external genital warts. Expert Opin. Biol. Ther. 2012, 12, 783–793. [Google Scholar] [CrossRef]
- Puviani, M.; Galluzzo, M.; Talamonti, M.; Mazzilli, S.; Campione, E.; Bianchi, L.; Milani, M.; Luppino, I.; Micali, G. Efficacy of sinecatechins 10% as proactive sequential therapy of external genital warts after laser CO2 ablative therapy: The PACT study (post-ablation immunomodulator treatment of condylomata with sinecatechins): A randomized, masked outcome assessment. Int. J. Std Aids 2019, 30, 131–136. [Google Scholar] [CrossRef]
- Manyere, N.R.; Dube Mandishora, R.S.; Magwali, T.; Mtisi, F.; Mataruka, K.; Mtede, B.; Palefsky, J.M.; Chirenje, Z.M. Human papillomavirus genotype distribution in genital warts among women in Harare-Zimbabwe. J. Obstet. Gynaecol. 2019, 1–7. [Google Scholar] [CrossRef]
- Ahn, W.-S.; Yoo, J.; Huh, S.-W.; Kim, C.-K.; Lee, J.-M.; Namkoong, S.-E.; Bae, S.-M.; Lee, I.P. Protective effects of green tea extracts (polyphenon E and EGCG) on human cervical lesions. Eur. J. Cancer Prev. 2003, 12, 383–390. [Google Scholar] [CrossRef]
- Jia, Y.; Hu, T.; Hang, C.-Y.; Yang, R.; Li, X.; Chen, Z.-L.; Mei, Y.-D.; Zhang, Q.-H.; Huang, K.-C.; Xiang, Q.-Y.; et al. Case-control study of diet in patients with cervical cancer or precancerosis in Wufeng, a high incidence region in China. Asian Pac. J. Cancer Prev. 2012, 13, 5299–5302. [Google Scholar] [CrossRef]
- Garcia, F.A.R.; Cornelison, T.; Nuño, T.; Greenspan, D.L.; Byron, J.W.; Hsu, C.-H.; Alberts, D.S.; Chow, H.-H.S. Results of a phase II randomized, double-blind, placebo-controlled trial of Polyphenon E in women with persistent high-risk HPV infection and low-grade cervical intraepithelial neoplasia. Gynecol. Oncol. 2014, 132, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Chamseddin, B.H.; Agim, N.G.; Jarin, J.; Wilson, E.E.; Mir, A. Therapy for anogenital verrucae in preadolescent children with topical and systemic treatment. Pediatr. Dermatol. 2019, 36, 623–627. [Google Scholar] [CrossRef] [PubMed]
- Clouth, A.; Schöfer, H. Treatment of recalcitrant facial verrucae vulgares with sinecatechins (greentea catechins) ointment. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 178–179. [Google Scholar] [CrossRef] [PubMed]
- Grandolfo, M.; Milani, M. Efficacy and tolerability of Polyphenon E in “difficult-to-treat” multiple genital warts in an HIV-positive male subject. Case Rep. Dermatol. 2006, 9, 55–59. [Google Scholar] [CrossRef]
- Bilenchi, R.; Campoli, M.; Trovato, E.; Cinotti, E.; Rubegni, P.; Fimiani, M. Sinecatechins 10% ointment for genital warts: Case report of a beneficial reaction in an HIV-positive woman. Int. J. Std Aids 2018, 29, 1033–1035. [Google Scholar] [CrossRef]
- Rob, F.; Jůzlová, K.; Sečníková, Z.; Jiráková, A.; Hercogová, J. Successful treatment with 10% sinecatechins ointment for recurrent anogenital warts in an eleven-year-old child. Pediatr. Infect. Dis. J. 2017, 36, 235–236. [Google Scholar] [CrossRef]
- Meloni, G.; Milani, M. Efficacy and tolerability of topical green tea extract (Polyphenon E) application in a “therapy-resistant” plantar wart. Case Rep. Dermatol. 2018, 10, 127–132. [Google Scholar] [CrossRef]
- Gupta, N.; Rodriguez, E.; Andikyan, V.; Salob, S.P.; Chi, D. A case report of vulvar carcinoma in situ treated with sinecatechins with complete response. Gynecol. Oncol. Case Rep. 2013, 6, 10–12. [Google Scholar] [CrossRef]
- Pizzini, L.; De Luca, G.; Milani, M. Efficacy and tolerability of topical Polyphenon E in multiple “seborrheic keratosis-like” lesions of the groin in an immunocompetent 26-year-old man. Case Rep. Dermatol. 2019, 11, 310–316. [Google Scholar] [CrossRef]
- Henrickson, S.E.; Treat, J.R. Topical cidofovir for recalcitrant verrucae in individuals with severe combined immunodeficiency after hematopoietic stem cell transplantation. Pediatr. Dermatol. 2017, 34, 24–25. [Google Scholar] [CrossRef]
- Shanafelt, T.D.; Call, T.G.; Zent, C.S.; Leis, J.F.; LaPlant, B.; Bowen, D.A.; Roos, M.; Laumann, K.; Ghosh, A.K.; Lesnick, C.; et al. Phase 2 trial of daily, oral Polyphenon E in patients with asymptomatic, Rai stage 0 to II chronic lymphocytic leukemia. Cancer 2013, 119, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Gee, J.R.; Saltzstein, D.R.; Kim, K.; Kolesar, J.; Huang, W.; Havighurst, T.C.; Wollmer, B.W.; Stublaski, J.; Downs, T.; Mukhtar, H.; et al. A phase II randomized, double-blind, presurgical trial of Polyphenon E in bladder cancer patients to evaluate pharmacodynamics and bladder tissue biomarkers. Cancer Prev. Res. (Phila.) 2017, 10, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Crew, K.D.; Ho, K.A.; Brown, P.; Greenlee, H.; Bevers, T.B.; Arun, B.; Sneige, N.; Hudis, C.; McArthur, H.L.; Chang, J.; et al. Effects of a green tea extract, Polyphenon E, on systemic biomarkers of growth factor signalling in women with hormone receptor-negative breast cancer. J. Hum. Nutr. Diet. 2015, 28, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.M.; Ahmann, F.R.; Nagle, R.B.; Hsu, C.-H.; Tangrea, J.A.; Parnes, H.L.; Sokoloff, M.H.; Gretzer, M.B.; Chow, H.-H.S. Randomized, double-blind, placebo-controlled trial of polyphenon E in prostate cancer patients before prostatectomy: Evaluation of potential chemopreventive activities. Cancer Prev. Res. (Phila). 2012, 5, 290–298. [Google Scholar] [CrossRef]
- Kumar, N.B.; Patel, R.; Pow-Sang, J.; Spiess, P.E.; Salup, R.; Williams, C.R.; Schell, M.J. Long-term supplementation of decaffeinated green tea extract does not modify body weight or abdominal obesity in a randomized trial of men at high risk for prostate cancer. Oncotarget 2017, 8, 99093–99103. [Google Scholar] [CrossRef]
- Dryden, G.W.; Lam, A.; Beatty, K.; Qazzaz, H.H.; McClain, C.J. A pilot study to evaluate the safety and efficacy of an oral dose of (-)-epigallocatechin-3-gallate-rich polyphenon E in patients with mild to moderate ulcerative colitis. Inflamm. Bowel Dis. 2013, 19, 1904–1912. [Google Scholar] [CrossRef]
- Chiaverini, C.; Roger, C.; Fontas, E.; Bourrat, E.; Bourdon-Lanoy, E.; Labrèze, C.; Mazereeuw, J.; Vabres, P.; Bodemer, C.; Lacour, J.-P. Oral epigallocatechin-3-gallate for treatment of dystrophic epidermolysis bullosa: A multicentre, randomized, crossover, double-blind, placebo-controlled clinical trial. Orphanet J. Rare Dis. 2016, 11, 31. [Google Scholar] [CrossRef][Green Version]
- Lovera, J.; Ramos, A.; Devier, D.; Garrison, V.; Kovner, B.; Reza, T.; Koop, D.; Rooney, W.; Foundas, A.; Bourdette, D. Polyphenon E, non-futile at neuroprotection in multiple sclerosis but unpredictably hepatotoxic: Phase I single group and phase II randomized placebo-controlled studies. J. Neurol. Sci. 2015, 358, 46–52. [Google Scholar] [CrossRef]
- Stockfleth, E.; Meyer, T. Sinecatechins (Polyphenon E) ointment for treatment of external genital warts and possible future indications. Expert Opin. Biol. Ther. 2014, 14, 1033–1043. [Google Scholar] [CrossRef]
- Wang, Y.-Q.; Lu, J.-L.; Liang, Y.-R.; Li, Q.-S. Suppressive effects of EGCG on cervical cancer. Molecules 2018, 23, 2334. [Google Scholar] [CrossRef]
- Tyring, S.K. Effect of sinecatechins on HPV-activated cell growth and induction of apoptosis. J. Clin. Aesthet. Dermatol. 2012, 5, 34–41. [Google Scholar] [PubMed]
- Rosen, T. Green tea catechins: Biologic properties, proposed mechanisms of action, and clinical implications. J. Drugs Dermatol. 2012, 11, 55–60. [Google Scholar]
- Doan, H.Q.; Nguyen, H.P.; Rady, P.; Tyring, S.K. Expression patterns of immune-associated genes in external genital and perianal warts treated with sinecatechins. Viral Immunol. 2015, 28, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Thappa, D.M.; Chiramel, M.J. Evolving role of immunotherapy in the treatment of refractory warts. Indian Dermatol. Online J. 2016, 7, 364–370. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.P.; Doan, H.Q.; Brunell, D.J.; Rady, P.; Tyring, S.K. Apoptotic gene expression in sinecatechins-treated external genital and perianal warts. Viral Immunol. 2014, 27, 556–558. [Google Scholar] [CrossRef]
- Thomas, M.; Banks, L. Human papillomavirus (HPV) E6 interactions with Bak are conserved amongst E6 proteins from high and low risk HPV types. J. Gen. Virol. 1999, 80, 1513–1517. [Google Scholar] [CrossRef]
- Hayakawa, S.; Saito, K.; Miyoshi, N.; Ohishi, T.; Oishi, Y.; Miyoshi, M.; Nakamura, Y. Anti-cancer effects of green tea by either anti- or pro- oxidative mechanisms. Asian Pac. J. Cancer Prev. 2016, 17, 1649–1654. [Google Scholar] [CrossRef]
- Nguyen, H.P.; Doan, H.Q.; Rady, P.; Tyring, S.K. Cellular signaling in sinecatechins-treated external genital and perianal warts: Unraveling the mechanism of action of a botanical therapy. Virol. Sin. 2015, 30, 214–217. [Google Scholar] [CrossRef]
- Leong, H.; Mathur, P.S.; Greene, G.L. Inhibition of mammary tumorigenesis in the C3(1)/SV40 mouse model by green tea. Breast Cancer Res. Treat. 2008, 107, 359–369. [Google Scholar] [CrossRef]
- Shimizu, M.; Deguchi, A.; Lim, J.T.E.; Moriwaki, H.; Kopelovich, L.; Weinstein, I.B. (-)-Epigallocatechin gallate and polyphenon E inhibit growth and activation of the epidermal growth factor receptor and human epidermal growth factor receptor-2 signaling pathways in human colon cancer cells. Clin. Cancer Res. 2005, 11, 2735–2746. [Google Scholar] [CrossRef]
- Pan, J.; Zhang, Q.; Xiong, D.; Vedell, P.; Yan, Y.; Jiang, H.; Cui, P.; Ding, F.; Tichelaar, J.W.; Wang, Y.; et al. Transcriptomic analysis by RNA-seq reveals AP-1 pathway as key regulator that green tea may rely on to inhibit lung tumorigenesis. Mol. Carcinog. 2014, 53, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Jung, M.K.; Ha, S.; Son, J.-A.; Song, J.H.; Houh, Y.; Cho, E.; Chun, J.H.; Yoon, S.R.; Yang, Y.; Bang, S.I.; et al. Polyphenon-60 displays a therapeutic effect on acne by suppression of TLR2 and IL-8 expression via down-regulating the ERK1/2 pathway. Arch. Dermatol. Res. 2012, 304, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.; Zhou, F.-M.; Su, C.-J.; Liu, T.T.-T.; Zhou, Y.; Fan, L.; Wang, Z.-H.; Liu, X.; Huang, Y.; Liu, T.T.-T.; et al. Epigallocatechin-3-gallate attenuates acute and chronic psoriatic itch in mice: Involvement of antioxidant, anti-inflammatory effects and suppression of ERK and Akt signaling pathways. Biochem. Biophys. Res. Commun. 2018, 496, 1062–1068. [Google Scholar] [CrossRef] [PubMed]
- Sivakumar, A.S.; Hwang, I. Effects of Sunphenon and Polyphenon 60 on proteolytic pathways, inflammatory cytokines and myogenic markers in H2O2-treated C2C12 cells. J. Biosci. 2015, 40, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Neyestani, T.R.; Gharavi, A.; Kalayi, A. Selective effects of tea extract and its phenolic compounds on human peripheral blood mononuclear cell cytokine secretions. Int. J. Food Sci. Nutr. 2009, 60, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Tyring, S.K. Sinecatechins: Effects on HPV-induced enzymes involved in inflammatory mediator generation. J. Clin. Aesthet. Dermatol. 2012, 5, 19–26. [Google Scholar]
- Song, S.; Krishnan, K.; Liu, K.; Bresalier, R.S. Polyphenon E inhibits the growth of human Barrett’s and aerodigestive adenocarcinoma cells by suppressing cyclin D1 expression. Clin. Cancer Res. 2009, 15, 622–631. [Google Scholar] [CrossRef]
- Leong, H.; Mathur, P.S.; Greene, G.L. Green tea catechins inhibit angiogenesis through suppression of STAT3 activation. Breast Cancer Res. Treat. 2009, 117, 505–515. [Google Scholar] [CrossRef]
- Philips, B.J.; Coyle, C.H.; Morrisroe, S.N.; Chancellor, M.B.; Yoshimura, N. Induction of apoptosis in human bladder cancer cells by green tea catechins. Biomed. Res. 2009, 30, 207–215. [Google Scholar] [CrossRef]
- Nakano, S.; Megro, S.-I.; Hase, T.; Suzuki, T.; Isemura, M.; Nakamura, Y.; Ito, S. Computational molecular docking and X-ray crystallographic studies of catechins in new drug design strategies. Molecules 2018, 23, 2020. [Google Scholar] [CrossRef]
- Santilli, G.; Piotrowska, I.; Cantilena, S.; Chayka, O.; D’Alicarnasso, M.; Morgenstern, D.A.; Himoudi, N.; Pearson, K.; Anderson, J.; Thrasher, A.J.; et al. Polyphenon [corrected] E enhances the antitumor immune response in neuroblastoma by inactivating myeloid suppressor cells. Clin. Cancer Res. 2013, 19, 1116–1125. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Liu, X.; Mei, L.; Wang, H.; Fang, F. Epigallocatechin-3-gallate (EGCG) inhibits imiquimod-induced psoriasis-like inflammation of BALB/c mice. Bmc Complement. Altern. Med. 2016, 16, 334. [Google Scholar] [CrossRef] [PubMed]
- Ohishi, T.; Goto, S.; Monira, P.; Isemura, M.; Nakamura, Y. Anti-inflammatory action of green tea. Antiinflamm. Antiallergy. Agents Med. Chem. 2016, 15, 74–90. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-Y.; Kao, C.-L.; Liu, C.-M. The cancer prevention, anti-inflammatory and anti-oxidation of bioactive phytochemicals targeting the TLR4 signaling pathway. Int. J. Mol. Sci. 2018, 19, 2729. [Google Scholar] [CrossRef] [PubMed]

| Case | PSV Application | Subject | Major Outcome | Reference |
|---|---|---|---|---|
| 1 | SC 10% | 34-year old male with recalcitrant facial warts and atopic dermatitis | Complete remission within 20 days with light skin irritation | [23] |
| 2 | PE 10%3 times/day | An HIV-positive 55-year-old male with 5 GW lesions | Reduction to 3 lesions after one month and complete regression after 8 weeks | [24] |
| 3 | SC 10% | HIV-positive female with previous unsuccessful treatments of cryotherapy plus imiquimod | Complete regression of inflammation and reduced number of GW lesion after 3 weeks No recurrence during 8 months | [25] |
| 4 | SC 10% | 11-year-old child with AGW | Complete regression of the lesions after 10 weeks No notable side effects during a 12 week-follow-up | [26] |
| 5 | * PE 10%, twice/day | 67-year-old male with plantar warts with unsuccessful prior treatments including cryotherapy and 5-fluorouracil regimen | Complete regression after 3 months | [27] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miyoshi, N.; Tanabe, H.; Suzuki, T.; Saeki, K.; Hara, Y. Applications of a Standardized Green Tea Catechin Preparation for Viral Warts and Human Papilloma Virus-Related and Unrelated Cancers. Molecules 2020, 25, 2588. https://doi.org/10.3390/molecules25112588
Miyoshi N, Tanabe H, Suzuki T, Saeki K, Hara Y. Applications of a Standardized Green Tea Catechin Preparation for Viral Warts and Human Papilloma Virus-Related and Unrelated Cancers. Molecules. 2020; 25(11):2588. https://doi.org/10.3390/molecules25112588
Chicago/Turabian StyleMiyoshi, Noriyuki, Hiroki Tanabe, Takuji Suzuki, Koichi Saeki, and Yukihiko Hara. 2020. "Applications of a Standardized Green Tea Catechin Preparation for Viral Warts and Human Papilloma Virus-Related and Unrelated Cancers" Molecules 25, no. 11: 2588. https://doi.org/10.3390/molecules25112588
APA StyleMiyoshi, N., Tanabe, H., Suzuki, T., Saeki, K., & Hara, Y. (2020). Applications of a Standardized Green Tea Catechin Preparation for Viral Warts and Human Papilloma Virus-Related and Unrelated Cancers. Molecules, 25(11), 2588. https://doi.org/10.3390/molecules25112588






