Development of Biopolymer and Conducting Polymer-Based Optical Sensors for Heavy Metal Ion Detection
Abstract
1. Introduction
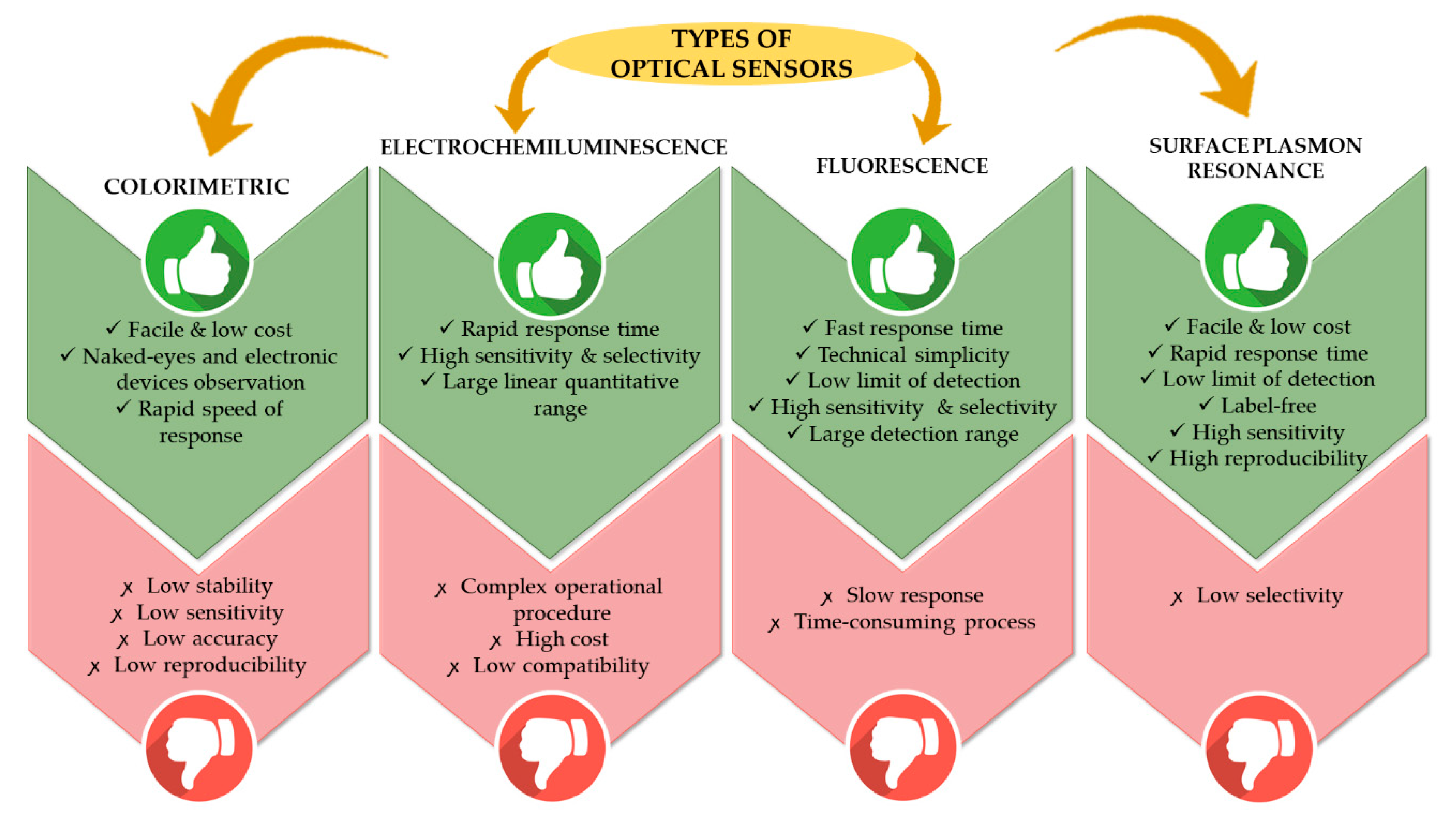
2. Application of Polymer Based Material with Optical Sensor for Heavy Metal Ion Detection
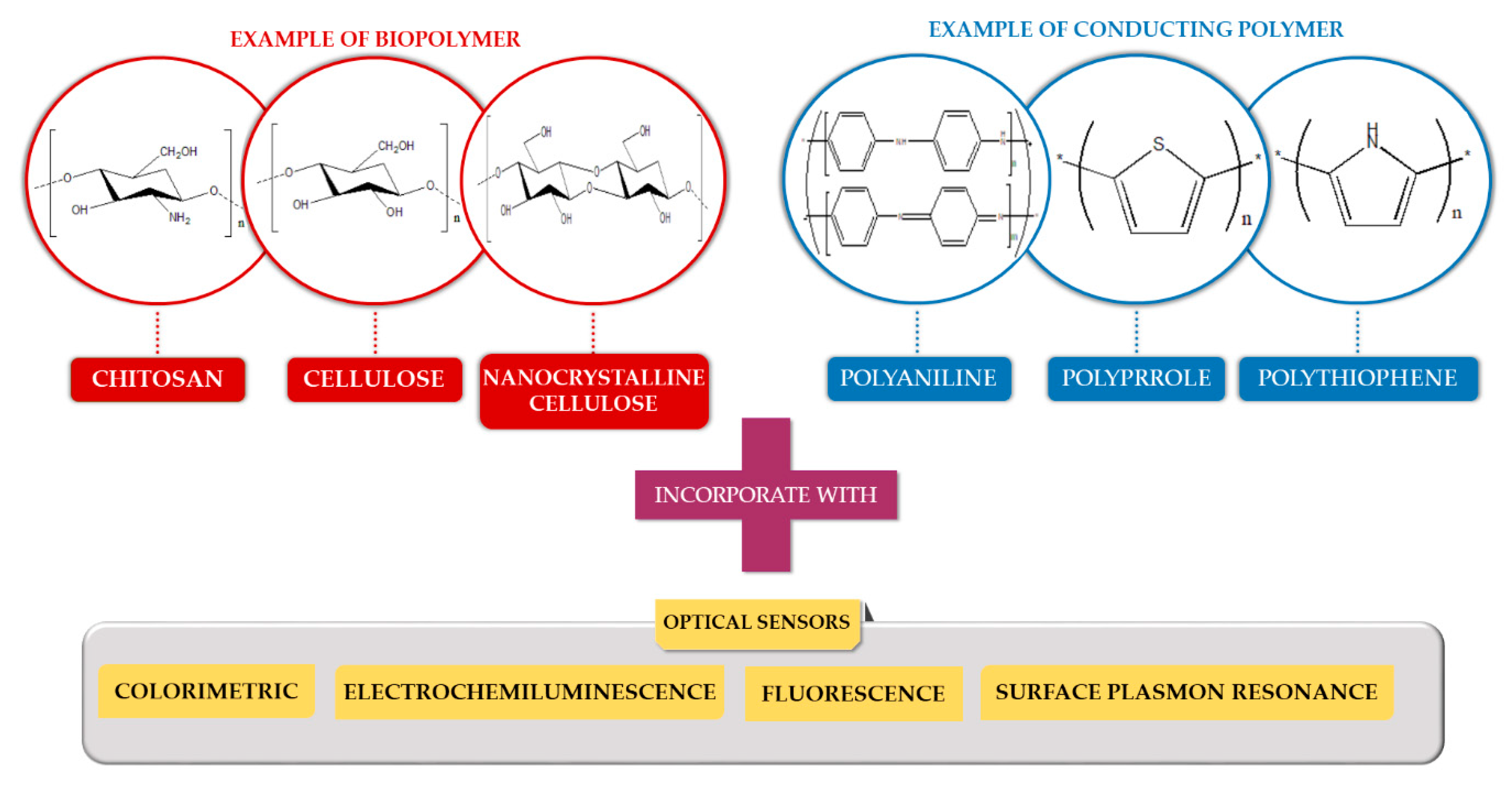
2.1. Biopolymer-Based Material
2.2. Conducting Polymer-Based Material
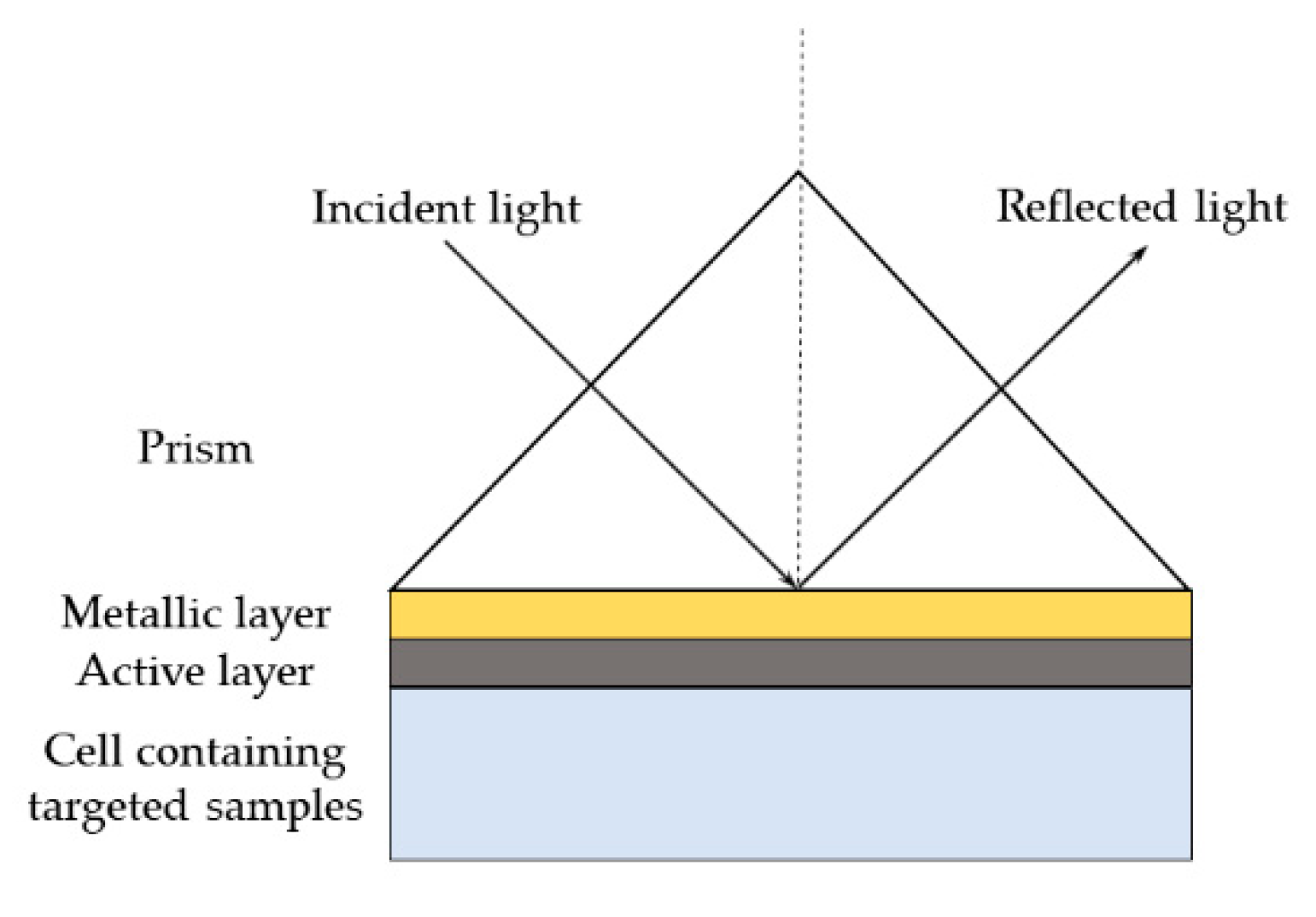
3. Surface Plasmon Resonance
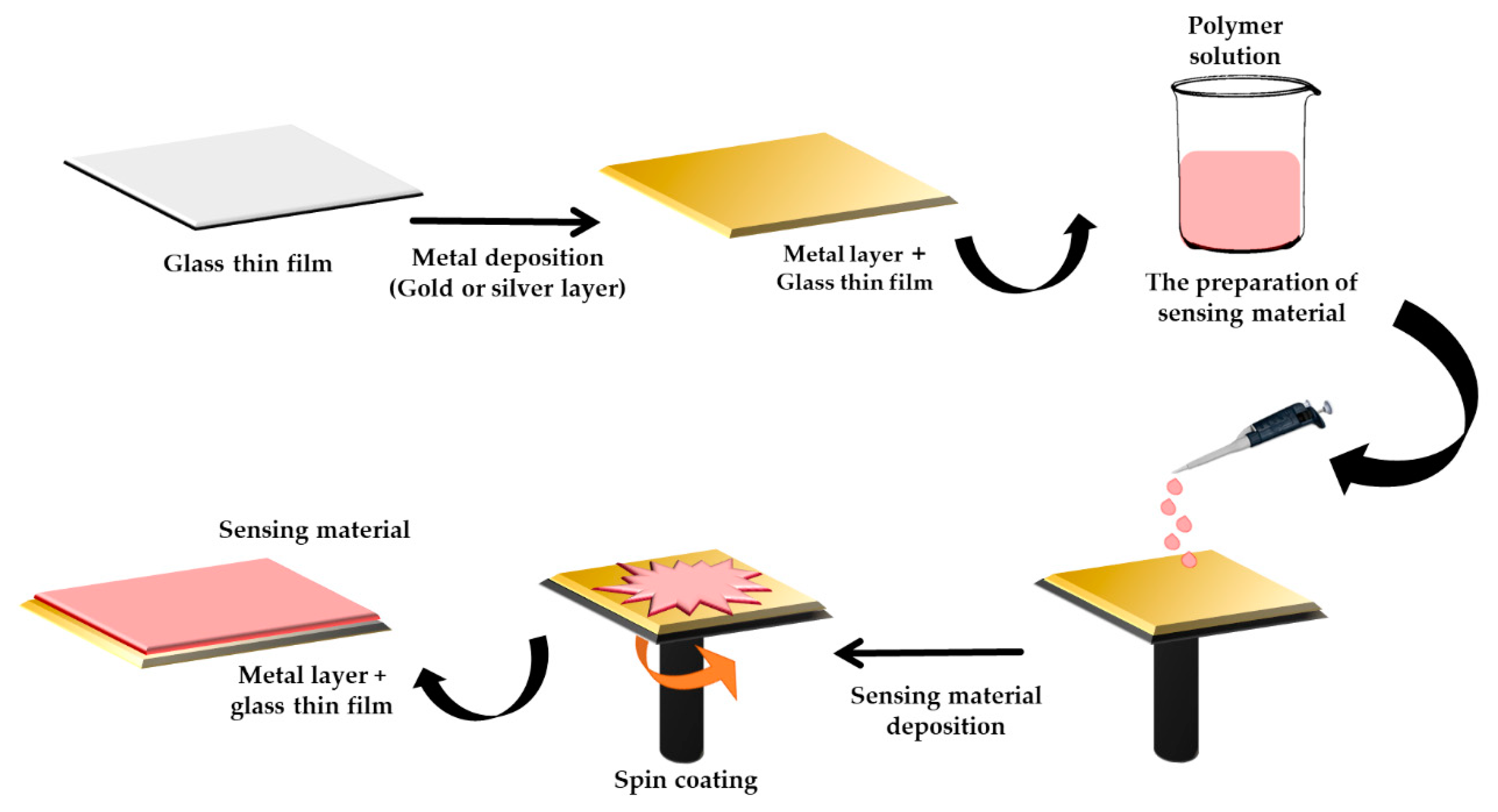
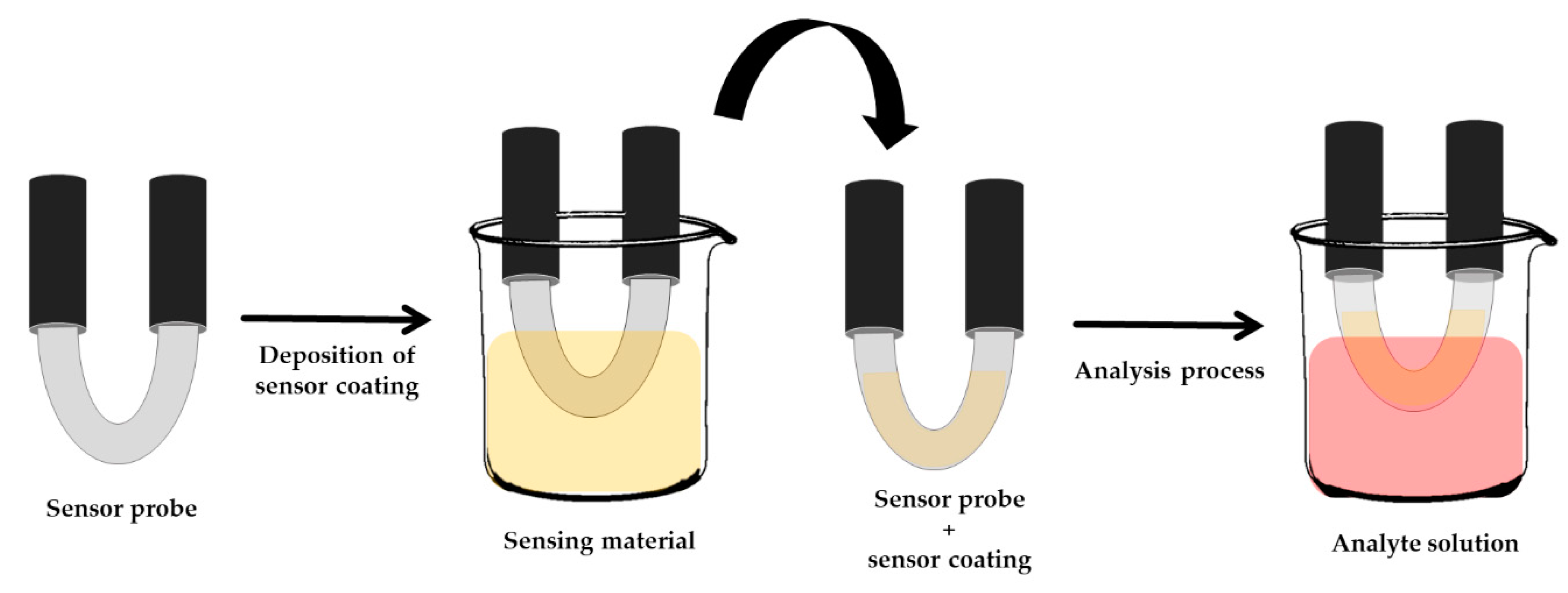
3.1. Incorporation of Biopolymer with Surface Plasmon Resonance
3.2. Incorporation of Conducting Polymer with Surface Plasmon Resonance
4. Future Perspectives in the Development of Biopolymers and Conducting Polymers with Surface Plasmon Resonance for Heavy Metal Ion Detection
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Afroz, R.; Masud, M.M.; Akhtar, R.; Duasa, J.B. Water pollution: Challenges and future direction for water resource management policies in Malaysia. Environ. Urban. ASIA 2014, 5, 63–81. [Google Scholar] [CrossRef]
- Xie, P.; Guo, F.; Yang, S.; Yao, D.; Yang, G.; Xie, L. A novel ratiometric fluorescent mercury probe based on deprotonation-ICT mechanism. J. Fluoresc. 2014, 24, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Bhatti, P.; Stewart, P.A.; Hutchinson, A.; Rothman, N.; Linet, M.S.; Inskip, P.D.; Rajaraman, P. Lead exposure, polymorphisms in genes related to oxidative stress, and risk of adult brain tumors. Cancer Epidemiol. Biomark. Prev. 2009, 18, 1841–1848. [Google Scholar] [CrossRef] [PubMed]
- Nawrot, T.S.; Staessen, J.A.; Roels, H.A.; Munters, E.; Cuypers, A.; Richart, T.; Ruttens, A.; Smeets, K.; Clijsters, H.; Vangronsveld, J. Cadmium exposure in the population: From health risks to strategies of prevention. Biometals 2010, 23, 769–782. [Google Scholar] [CrossRef] [PubMed]
- Terra, I.A.A.; Mercante, L.A.; Andre, R.S.; Correa, D.S. Fluorescent and colorimetric electrospun nanofibers for heavy-metal sensing. Biosensors 2017, 7, 61. [Google Scholar] [CrossRef] [PubMed]
- Fen, Y.W.; Yunus, W.M.M. Surface plasmon resonance spectroscopy as an alternative for sensing heavy metal ions: A review. Sens. Rev. 2013, 33, 305–314. [Google Scholar] [CrossRef]
- Kumar, P.; Kim, K.; Bansal, V.; Lazarides, T.; Kumar, N. Journal of industrial and engineering chemistry progress in the sensing techniques for heavy metal ions using nanomaterials. J. Ind. Eng. Chem. 2017, 54, 30–43. [Google Scholar] [CrossRef]
- Khan, N.I.; Song, E. Lab-on-a-chip systems for aptamer-based biosensing. Micromachines 2020, 11, 220. [Google Scholar] [CrossRef]
- Rizwan, M.; Mohd-Naim, N.F.; Ahmed, M.U. Trends and Advances in Electrochemiluminescence Nanobiosensors. Sensors 2018, 18, 166. [Google Scholar] [CrossRef]
- Abdi, M.M.; Mat Yunus, W.M.; Reayi, M.; Bahrami, A. Plasmonic Conducting Polymers for Heavy Metal Sensing. In Plasmonics—Principles and Applications; InTech: Rijeka, Croatia, 2012; Volume 395, pp. 116–124. [Google Scholar] [CrossRef][Green Version]
- Su, Y.; Lv, Y. Graphene and graphene oxides: Recent advances in chemiluminescence and electrochemiluminescence. RSC Adv. 2014, 4, 29324–29339. [Google Scholar] [CrossRef]
- Omar, N.A.S.; Fen, Y.W.; Abdullah, J.; Sadrolhosseini, A.R.; Kamil, Y.M.; Fauzi, N.I.M.; Hashim, H.S.; Mahdi, M.A. Quantitative and selective surface plasmon resonance response based on a reduced graphene oxide—Polyamidoamine nanocomposite for detection of dengue virus E-proteins. Nanomaterials 2020, 10, 569. [Google Scholar] [CrossRef] [PubMed]
- Chiu, N.-F.; Huang, T.-Y.; Lai, H.-C. Graphene oxide based surface plasmon resonance biosensors. In Advances in Graphene Science; InTech: Rijeka, Croatia, 2013; pp. 191–216. [Google Scholar] [CrossRef]
- Mohd Daniyal, W.M.E.M.; Saleviter, S.; Fen, Y.W. Development of surface plasmon resonance spectroscopy for metal ion detection. Sens. Mater. 2018, 30, 2023–2038. [Google Scholar] [CrossRef]
- Li, M.; Li, X.; Xiao, H.N.; James, T.D. Fluorescence sensing with cellulose-based materials. ChemistryOpen 2017, 6, 685–696. [Google Scholar] [CrossRef] [PubMed]
- Golmohammadi, H.; Morales-Narváez, E.; Naghdi, T.; Merkoçi, A. Nanocellulose in sensing and biosensing. Chem. Mater. 2017, 29, 5426–5446. [Google Scholar] [CrossRef]
- Chen, Y.; Zhou, S.; Li, L.; Zhu, J. Nanomaterials-based sensitive electrochemiluminescence biosensing. Nano Today 2017, 12, 98–115. [Google Scholar] [CrossRef]
- Bertoncello, P.; Forster, R.J. Nanostructured materials for electrochemiluminescence (ECL)-based detection methods: Recent advances and future perspectives. Biosens. Bioelectron. 2009, 24, 3191–3200. [Google Scholar] [CrossRef]
- Anas, N.A.A.; Fen, Y.W.; Omar, N.A.S.; Daniyal, W.M.E.M.M.; Ramdzan, N.S.M.; Saleviter, S. Development of graphene quantum dots-based optical sensor for toxic metal ion detection. Sensors 2019, 19, 3850. [Google Scholar] [CrossRef]
- Tang, F.A.N.; Zhang, L.; Zhang, Z.; Cheng, Z.; Zhu, X. Cellulose filter paper with antibacterial activity from cellulose filter paper with antibacterial activity from surface-initiated ATRP. J. Macromelucular Sci. 2009, 46, 989–996. [Google Scholar] [CrossRef]
- Habibi, Y.; Lucia, L.A.; Rojas, O.J. Cellulose Nanocrystals: Chemistry, Self-Assembly, and Applications. Chem. Rev. 2010, 110, 3479–3500. [Google Scholar] [CrossRef]
- Edwards, J.V.; Prevost, N.; French, A.; Concha, M.; DeLucca, A.; Wu, Q. Nanocellulose-based biosensors: Design, preparation, and activity of peptide-linked cotton cellulose nanocrystals having fluorimetric and colorimetric elastase detection sensitivity. Engineering 2013, 5, 20–28. [Google Scholar] [CrossRef]
- Badawy, M.E.I.; Rabea, E.I. A Biopolymer chitosan and its derivatives as promising antimicrobial agents against plant pathogens and their applications in crop protection. Int. J. Carbohydr. Chem. 2011, 2011. [Google Scholar] [CrossRef]
- Klemm, D.; Heublein, B.; Fink, H.; Bohn, A. Polymer science cellulose: Fascinating biopolymer and sustainable raw material angewandte. Angew. Chem. 2005, 44, 3358–3393. [Google Scholar] [CrossRef] [PubMed]
- Janmanee, R.; Chuekachang, S.; Sriwichai, S.; Baba, A.; Phanichphant, S. Functional conducting polymers in the application of SPR biosensors. J. Nanotechnol. 2012, 2012, 1–7. [Google Scholar] [CrossRef]
- Wang, H.; Xu, C.; Yuan, B. Polymer-based electrochemical sensing platform for heavy metal ions detection—A critical review. Int. J. Electrochem. Sci. 2019, 14, 8760–8771. [Google Scholar] [CrossRef]
- Kurauchi, Y.; Hayashi, R.; Egashira, N.; Ohga, K. Fluorometric determination of zinc, cadmium and gallium ions with a fiber-optic sensor having a pyridoxal isomer-modified chitosan/agarose gel as a sensing probe. Anal. Sci. 1992, 8, 837–840. [Google Scholar] [CrossRef]
- Lai, S.; Chang, X.; Fu, C. Cadmium sulfide quantum dots modified by chitosan as fluorescence probe for copper (II) ion determination. Microchim. Acta 2009, 165, 39–44. [Google Scholar] [CrossRef]
- Wang, X.; Su, Y.; Yang, H.; Dong, Z.; Ma, J. Highly sensitive fluorescence probe based on chitosan nanoparticle for selective detection of Hg2+ in water. Colloids Surf. A Physicochem. Eng. Asp. 2012, 402, 88–93. [Google Scholar] [CrossRef]
- Liu, L.; Xiao, L.; Zhu, H.; Shi, X. Preparation of magnetic and fluorescent bifunctional chitosan nanoparticles for optical determination of copper ion. Microchim. Acta 2012, 178, 413–419. [Google Scholar] [CrossRef]
- Dang, J.; Guo, Z.; Zheng, X. Label-free sensitive electrogenerated chemiluminescence aptasensing based on chitosan/Ru(Bpy)32+/silica nanoparticles modified electrode. Anal. Chem. 2014, 86, 8943–8950. [Google Scholar] [CrossRef]
- Geng, Z.; Zhang, H.; Xiong, Q.; Zhang, Y.; Zhao, H.; Wang, G. A fluorescent chitosan hydrogel detection platform for the sensitive and selective determination of trace mercury (II) in water. J. Mater. Chem. A 2015, 3, 19455–19460. [Google Scholar] [CrossRef]
- Chen, Z.; Zhang, C.; Tan, Y.; Zhou, T.; Ma, H.; Wan, C.; Lin, Y.; Li, K. Chitosan-functionalized gold nanoparticles for colorimetric detection of mercury ions based on chelation-induced aggregation. Microchim. Acta 2014, 182, 611–616. [Google Scholar] [CrossRef]
- Chauhan, K.; Singh, P.; Singhal, R.K. New chitosan-thiomer: An efficient colorimetric sensor and effective sorbent for mercury at ultralow concentration. ACS Appl. Mater. Interfaces 2015, 7, 26069–26078. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.; Yan, F.; Zhou, X.; Zheng, T.; Shi, Y.; Fu, W.; Chen, L. Preconcentration and fluorometric detection of mercury ions using magnetic core-shell chitosan microspheres modified with a rhodamine spirolactam. Microchim. Acta 2016, 183, 319–327. [Google Scholar] [CrossRef]
- Shi, D.; Yan, F.; Wang, M.; Zou, Y.; Zheng, T.; Zhou, X.; Chen, L. Rhodamine derivative functionalized chitosan as efficient sensor and adsorbent for mercury (II) detection and removal. Mater. Res. Bull. 2015, 70, 958–964. [Google Scholar] [CrossRef]
- Mehta, V.N.; Basu, H.; Singhal, R.K.; Kailasa, S.K. Simple and sensitive colorimetric sensing of Cd2+ ion using chitosan dithiocarbamate functionalized gold nanoparticles as a probe. Sens. Actuators B Chem. 2015, 220, 850–858. [Google Scholar] [CrossRef]
- Nivethaa, E.A.K.; Narayanan, V.; Stephen, A. Synthesis and spectral characterization of silver embedded chitosan matrix nanocomposite for the selective colorimetric sensing of toxic mercury. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 143, 242–250. [Google Scholar] [CrossRef]
- Kumar, A.; Chowdhuri, A.R.; Laha, D.; Chandra, S.; Karmakar, P.; Sahu, S.K. One-pot synthesis of carbon dot-entrenched chitosan-modified magnetic nanoparticles for fluorescence-based Cu2+ ion sensing and cell imaging. RSC Adv. 2016, 6, 58979–58987. [Google Scholar] [CrossRef]
- Nivethaa, E.A.K.; Dhanavel, S.; Narayanan, V.; Stephen, A. Chitosan stabilized Ag-Au nanoalloy for colorimetric sensing and 5-fluorouracil delivery. Int. J. Biol. Macromol. 2017, 95, 862–872. [Google Scholar] [CrossRef]
- Tian, K.; Siegel, G.; Tiwari, A. A simple and selective colorimetric mercury (II) sensing system based on chitosan stabilized gold nanoparticles and 2,6-pyridinedicarboxylic acid. Mater. Sci. Eng. C 2017, 71, 195–199. [Google Scholar] [CrossRef]
- Wang, S.; Gao, Y. Fabrication of chitosan/silver nanocomposite films and their fluorescence sensing of aluminum ions. Mater. Technol. 2017, 32, 569–573. [Google Scholar] [CrossRef]
- Liu, Y.L.; Wang, Z.K.; Qin, W.; Hu, Q.L.; Tang, B.Z. Fluorescent detection of Cu (II) by chitosan-based AIE bioconjugate. Chin. J. Polym. Sci. 2017, 35, 365–371. [Google Scholar] [CrossRef]
- Lee, H.M.; Kim, M.H.; Yoon, Y.I.; Park, W.H. Fluorescent property of chitosan oligomer and its application as a metal ion sensor. Mar. Drugs 2017, 15, 105. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Li, Z.; Wu, Y.; Guo, W.; Wang, J.; Jiang, Q. Colorimetric detection of Hg2+ based on enhancement of peroxidase-like activity of chitosan-gold nanoparticles. Bull. Korean Chem. Soc. 2018, 39, 625–630. [Google Scholar] [CrossRef]
- Butwong, N.; Kunthadong, P.; Soisungnoen, P.; Chotichayapong, C.; Srijaranai, S.; Luong, J.H.T. Silver-doped CdS quantum dots incorporated into chitosan-coated cellulose as a colorimetric paper test stripe for mercury. Microchim. Acta 2018, 185, 1–9. [Google Scholar] [CrossRef]
- Sharma, P.; Mourya, M.; Choudhary, D.; Goswami, M.; Kundu, I.; Dobhal, M.P.; Tripathi, C.S.P.; Guin, D. Thiol terminated chitosan capped silver nanoparticles for sensitive and selective detection of mercury (II) ions in water. Sens. Actuators B Chem. 2018, 268, 310–318. [Google Scholar] [CrossRef]
- Amanulla, B.; Perumal, K.N.; Ramaraj, S.K. Chitosan functionalized gold nanoparticles assembled on sulphur doped graphitic carbon nitride as a new platform for colorimetric detection of trace Hg2+. Sens. Actuators B Chem. 2019, 281, 281–287. [Google Scholar] [CrossRef]
- Hu, L.; Zhu, B.; Zhang, L.; Yuan, H.; Zhao, Q.; Yan, Z. Chitosan-gold nanocomposite and its functionalized paper strips for reversible visual sensing and removal of trace Hg2+ in practice. Analyst 2019, 144, 474–480. [Google Scholar] [CrossRef]
- Zhao, L.; Wang, Y.; Li, Z.; Deng, Y.; Zhao, X.; Xia, Y. Facile synthesis of chitosan-gold nanocomposite and its application for exclusively sensitive detection of Ag+ ions. Carbohydr. Polym. 2019, 226, 115290. [Google Scholar] [CrossRef]
- Egorov, V.M.; Smirnova, S.V.; Formanovsky, A.A.; Pletnev, I.V.; Zolotov, Y.A. Dissolution of cellulose in ionic liquids as a way to obtain test materials for metal-ion detection. Anal. Bioanal. Chem. 2007, 387, 2263–2269. [Google Scholar] [CrossRef]
- Zhang, X.; Huang, J. Functional surface modification of natural cellulose substances for colorimetric detection and adsorption of Hg2+ in aqueous media. Chem. Commun. 2010, 46, 6042–6044. [Google Scholar] [CrossRef]
- Wang, M.; Meng, G.; Huang, Q.; Qian, Y. Electrospun 1,4-DHAQ-doped cellulose nanofiber films for reusable fluorescence detection of trace Cu2+ and further for Cr3+. Environ. Sci. Technol. 2012, 46, 367–373. [Google Scholar] [CrossRef]
- Zhang, L.; Li, Q.; Zhou, J.; Zhang, L. Synthesis and photophysical behavior of pyrene-bearing cellulose nanocrystals for Fe3+ sensing. Macromol. Chem. Phys. 2012, 213, 1612–1617. [Google Scholar] [CrossRef]
- Li, Y.; Wen, Y.; Wang, L.; He, J.; Al-Deyab, S.S.; El-Newehy, M.; Yu, J.; Ding, B. Simultaneous visual detection and removal of lead (II) ions with pyromellitic dianhydride-grafted cellulose nanofibrous membranes. J. Mater. Chem. A 2015, 3, 18180–18189. [Google Scholar] [CrossRef]
- You, J.; Hu, H.; Zhou, J.; Zhang, L.; Zhang, Y.; Kondo, T. Novel cellulose polyampholyte-gold nanoparticle-based colorimetric competition assay for the detection of cysteine and mercury(II). Langmuir 2013, 29, 5085–5092. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.Q.; Neoh, K.G.; Kang, E.T.; Fu, G.D. Rhodamine derivative-modified filter papers for colorimetric and fluorescent detection of Hg2+ in aqueous media. J. Mater. Chem. A 2013, 1, 2526–2532. [Google Scholar] [CrossRef]
- Kumari, S.; Chauhan, G.S. New cellulose-lysine schiff-base-based sensor-adsorbent for mercury ions. ACS Appl. Mater. Interfaces 2014, 6, 5908–5917. [Google Scholar] [CrossRef]
- Nawaz, H.; Tian, W.; Zhang, J.; Jia, R.; Chen, Z.; Zhang, J. Cellulose-based sensor containing phenanthroline for the highly selective and rapid detection of Fe2+ ions with naked eye and fluorescent dual modes. ACS Appl. Mater. Interfaces 2018, 10, 2114–2121. [Google Scholar] [CrossRef]
- Yun-Fei, Z.; Maimaiti, H.; Bo, Z. Preparation of cellulose-based fluorescent carbon nanoparticles and their application in trace detection of Pb (II). RSC Adv. 2017, 7, 2842–2850. [Google Scholar] [CrossRef]
- Milindanuth, P.; Pisitsak, P. A novel colorimetric sensor based on Rhodamine-B derivative and bacterial cellulose for the detection of Cu (II) ions in water. Mater. Chem. Phys. 2018, 216, 325–331. [Google Scholar] [CrossRef]
- Faham, S.; Golmohammadi, H.; Ghavami, R.; Khayatian, G. A nanocellulose-based colorimetric assay kit for smartphone sensing of iron and iron-chelating deferoxamine drug in biofluids. Anal. Chim. Acta 2019, 1087, 104–112. [Google Scholar] [CrossRef]
- Ding, B.; Si, Y.; Wang, X.; Yu, J.; Feng, L.; Sun, G. Label-free ultrasensitive colorimetric detection of copper (II) ions utilizing polyaniline/polyamide-6 nano-fiber/net sensor strips. J. Mater. Chem. 2011, 21, 13345–13353. [Google Scholar] [CrossRef]
- Si, Y.; Wang, X.; Li, Y.; Chen, K.; Wang, J.; Yu, J.; Wang, H.; Ding, B. Optimized colorimetric sensor strip for mercury (II) assay using hierarchical nanostructured conjugated polymers. J. Mater. Chem. A 2014, 2, 645–652. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, J.; Zou, W.; Wang, R. Facile synthesis of polyaniline/carbon dot nanocomposites and their application as a fluorescent probe to detect mercury. RSC Adv. 2015, 5, 41914–41919. [Google Scholar] [CrossRef]
- Wang, X.; Xuan, H.; Zhang, J.; Chen, S.; Zhang, F.; Zou, W. Facile synthesis of fluorescent polyaniline microspheres and their use for the detection of mercury ions. New J. Chem. 2015, 39, 6261–6266. [Google Scholar] [CrossRef]
- Saikia, A.; Karak, N. Polyaniline nanofiber/carbon dot nanohybrid as an efficient fluorimetric sensor for as (III) in water and effective antioxidant. Mater. Today Commun. 2018, 14, 82–89. [Google Scholar] [CrossRef]
- Tavoli, F.; Alizadeh, N. Electrically induced fluorescence Fe3+ sensing behavior of nanostructured tiron doped polypyrrole. Anal. Chim. Acta 2016, 946, 88–95. [Google Scholar] [CrossRef]
- Lo, M.; Diaw, A.K.D.; Gningue-Sall, D.; Oturan, M.A.; Chehimi, M.M.; Aaron, J.J. A novel fluorescent sensor based on electrosynthesized benzene sulfonic acid-doped polypyrrole for determination of Pb (II) and Cu (II). Luminescence 2019, 34, 489–499. [Google Scholar] [CrossRef]
- McIlwee, H.A.; Schauer, C.L.; Praig, V.G.; Boukherroub, R.; Szunerits, S. Thin chitosan films as a platform for SPR sensing of ferric ions. Analyst 2008, 133, 673–677. [Google Scholar] [CrossRef]
- Fahnestock, K.J.; Manesse, M.; McIlwee, H.A.; Schauer, C.L.; Boukherroub, R.; Szunerits, S. Selective detection of hexachromium ions by localized surface plasmon resonance measurements using gold nanoparticles/chitosan composite interfaces. Analyst 2009, 134, 881–886. [Google Scholar] [CrossRef]
- Fen, Y.W.; Yunus, W.M.M.; Yusof, N.A. Detection of mercury and copper ions using surface plasmon resonance optical sensor. Sens. Mater. 2011, 23, 325–334. [Google Scholar] [CrossRef]
- Fen, Y.W.; Yunus, W.M.M.; Yusof, N.A. Surface Plasmon Resonance Optical Sensor for Detection of Essential Heavy Metal Ions with Potential for Toxicity: Copper, Zinc and Manganese Ions. Sens. Lett. 2011, 9, 1704. [Google Scholar] [CrossRef]
- Fen, Y.W.; Yunus, W.M.M.; Yusof, N.A. Surface plasmon resonance optical sensor for detection of Pb2+ based on immobilized p-tert-butylcalix[4]arene-tetrakis in chitosan thin film as an active layer. Sens. Actuators B Chem. 2012, 171, 287–293. [Google Scholar] [CrossRef]
- Fen, Y.W.; Yunus, W.M.M.; Talib, Z.A. Analysis of Pb(II) ion sensing by crosslinked chitosan thin film using surface plasmon resonance spectroscopy. Optik (Stuttg) 2013, 124, 126–133. [Google Scholar] [CrossRef]
- Lokman, N.F.; Bakar, A.A.A.; Suja, F.; Abdullah, H.; Rahman, W.B.W.A.; Huang, N.M.; Yaacob, M.H. Highly sensitive SPR response of Au/Chitosan/Graphene oxide nanostructured thin films toward Pb (II) ions. Sens. Actuators B Chem. 2014, 195, 459–466. [Google Scholar] [CrossRef]
- Fen, Y.W.; Yunus, W.M.M.; Talib, Z.A.; Yusof, N.A. Development of surface plasmon resonance sensor for determining zinc ion using novel active nanolayers as probe. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 134, 48–52. [Google Scholar] [CrossRef]
- Kamaruddin, N.H.; Bakar, A.A.A.; Yaacob, M.H.; Mahdi, M.A.; Zan, M.S.D.; Shaari, S. Enhancement of chitosan-graphene oxide SPR sensor with a multi-metallic layers of Au-Ag-Au nanostructure for lead(II) ion detection. Appl. Surf. Sci. 2016, 361, 177–184. [Google Scholar] [CrossRef]
- Kamaruddin, N.H.; Bakar, A.A.A.; Mobarak, N.N.; Dzulkefly Zan, M.S.; Arsad, N. Binding affinity of a highly sensitive Au/Ag/Au/Chitosan-Graphene oxide sensor based on direct detection of Pb2+ and Hg2+ ions. Sensors 2017, 17, 2277. [Google Scholar] [CrossRef]
- Zainudin, A.A.; Fen, Y.W.; Yusof, N.A.; Al-Rekabi, S.H.; Mahdi, M.A.; Omar, N.A.S. Incorporation of surface plasmon resonance with novel valinomycin doped chitosan-graphene oxide thin film for sensing potassium ion. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2018, 191, 111–115. [Google Scholar] [CrossRef]
- Saleviter, S.; Fen, Y.W.; Omar, N.A.S.; Zainudin, A.A.; Daniyal, W.M.E.M.M. Optical and structural characterization of immobilized 4-(2-pyridylazo)resorcinol in chitosan-graphene oxide composite thin film and its potential for Co2+ sensing using surface plasmon resonance technique. Results Phys. 2018, 11, 118–122. [Google Scholar] [CrossRef]
- Saleviter, S.; Fen, Y.W.; Sheh Omar, N.A.; Mustaqim Mohd Daniyal, W.M.E.; Abdullah, J.; Mat Zaid, M.H. Structural and optical studies of cadmium sulfide quantum dot-graphene oxide-chitosan nanocomposite thin film as a novel SPR spectroscopy active layer. J. Nanomater. 2018, 2018. [Google Scholar] [CrossRef]
- Boruah, B.S.; Biswas, R. An optical fiber based surface plasmon resonance technique for sensing of lead ions: A toxic water pollutant. Opt. Fiber Technol. 2018, 46, 152–156. [Google Scholar] [CrossRef]
- Abdullah, S.; Azeman, N.H.; Mobarak, N.N.; Zan, M.S.D.; Ahmad, A.A. Sensitivity enhancement of localized SPR sensor towards Pb (II) ion detection using natural bio-polymer based carrageenan. Optik (Stuttg) 2018, 168, 784–793. [Google Scholar] [CrossRef]
- Anas, N.A.A.; Fen, Y.W.; Omar, N.A.S.; Ramdzan, N.S.M.; Daniyal, W.M.E.M.M.; Saleviter, S.; Zainudin, A.A. Optical properties of chitosan/hydroxyl-functionalized graphene quantum dots thin film for potential optical detection of ferric (III) ion. Opt. Laser Technol. 2019, 120. [Google Scholar] [CrossRef]
- Ramdzan, N.S.M.; Fen, Y.W.; Omar, N.A.S.; Anas, N.A.A.; Daniyal, W.M.E.M.M.; Saleviter, S.; Zainudin, A.A. Optical and surface plasmon resonance sensing properties for chitosan/carboxyl-functionalized graphene quantum dots thin film. Optik (Stuttg) 2019, 178, 802–812. [Google Scholar] [CrossRef]
- Roshidi, M.D.A.; Fen, Y.W.; Daniyal, W.M.E.M.M.; Omar, N.A.S.; Zulholinda, M. Structural and optical properties of chitosan-poly(amidoamine) dendrimer composite thin film for potential sensing Pb2+ using an optical spectroscopy. Optik (Stuttg) 2019, 185, 351–358. [Google Scholar] [CrossRef]
- Ding, Z.W.; Ravikumar, R.; Zhao, C.L.; Chen, L.H.; Chan, C.C. Chitosan/Poly (acrylic acid) based fiber-optic surface plasmon resonance sensor for Cu2+ ions detection. J. Light. Technol. 2019, 37, 2246–2252. [Google Scholar] [CrossRef]
- Wang, W.; Zhou, X.; Wu, S.; Li, S.; Wu, W.; Xiong, Z.; Shi, W.; Tian, X.; Yu, Q. Reusable surface plasmon resonance sensor for rapid detection of Cu2+ based on modified-chitosan thin film as an active layer. Sens. Actuators A Phys. 2019, 286, 59–67. [Google Scholar] [CrossRef]
- Saleviter, S.; Wing Fen, Y.; Daniyal, W.M.E.M.M.; Abdullah, J.; Sadrolhosseini, A.R.; Omar, N.A.S. Design and analysis of surface plasmon resonance optical sensor for determining cobalt ion based on chitosan-graphene oxide decorated quantum dots-modified gold active layer. Opt. Express 2019, 27, 32294. [Google Scholar] [CrossRef]
- Omar, N.A.S.; Fen, Y.W.; Saleviter, S.; Daniyal, W.M.E.M.M.; Anas, N.A.A.; Ramdzan, N.S.M.; Roshidi, M.D.A. Development of a graphene-based surface plasmon resonance optical sensor chip for potential biomedical application. Materials 2019, 12, 1928. [Google Scholar] [CrossRef]
- Daniyal, W.M.E.M.M.; Fen, Y.W.; Abdullah, J.; Sadrolhosseini, A.R.; Saleviter, S.; Omar, N.A.S. Exploration of surface plasmon resonance for sensing copper ion based on nanocrystalline cellulose-modified thin film. Opt. Express 2018, 26, 34880. [Google Scholar] [CrossRef]
- Daniyal, W.M.E.M.M.; Fen, Y.W.; Abdullah, J.; Sadrolhosseini, A.R.; Saleviter, S.; Omar, N.A.S. Label-free optical spectroscopy for characterizing binding properties of highly sensitive nanocrystalline cellulose-graphene oxide based nanocomposite towards nickel ion. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 212, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Daniyal, W.M.E.M.M.; Fen, Y.W.; Anas, N.A.A.; Omar, N.A.S.; Ramdzan, N.S.M.; Nakajima, H.; Mahdi, M.A. Enhancing the sensitivity of a surface plasmon resonance-based optical sensor for zinc ion detection by the modification of a gold thin film. RSC Adv. 2019, 9, 41729–41736. [Google Scholar] [CrossRef]
- Yu, J.C.C.; Lai, E.P.C.; Sadeghi, S. Surface plasmon resonance sensor for Hg(II) detection by binding interactions with polypyrrole and 2-mercaptobenzothiazole. Sens. Actuators B Chem. 2004, 101, 236–241. [Google Scholar] [CrossRef]
- Sadrolhosseini, A.R.; Noor, A.S.M.; Moksin, M.M.; Abdi, M.M.; Soleimani, H.; Abas, A.F.; Talib, Z.A. Polypyrrole thin film sensor base surface plasmon resonance for detection of Cu (II) and Fe (III) in aqueous solution. AIP Conf. Proc. 2012, 1482, 200–204. [Google Scholar] [CrossRef]
- Sadrolhosseini, A.R.; Noor, A.S.M.; Bahrami, A.; Lim, H.N.; Talib, Z.A.; Mahdi, M.A. Application of Polypyrrole Multi-Walled Carbon Nanotube Composite Layer for Detection of Mercury, Lead and Iron Ions Using Surface Plasmon Resonance Technique. PLoS ONE 2014, 9, e93962. [Google Scholar] [CrossRef] [PubMed]
- Nawi, N.M.; Abdullah, S.; Bakar, A.A.A. Gold nanoparticles/graphene oxide/polyaniline nanocomposites film as sensitive LSPR-based sensor for Pb (II) ions detection. In Proceedings of the 2014 IEEE 5th International Conference on Photonics, Kuala Lumpur, Malaysia, 2–4 September 2014; pp. 188–190. [Google Scholar] [CrossRef]
- Bahrami, A.; Sadrolhosseini, A.R.; Mamdoohi, G.; Bahzad, K.; Abdi, M.M. Surface plasmon sensor based on polypyrrole multiwalled carbon nanotube composite layer to detect Al (III) in aqueous solution. Dig. J. Nanomater. Biostruct. 2015, 10, 535–541. [Google Scholar]
- Tabassum, R.; Gupta, B.D. Fiber optic manganese ions sensor using SPR and nanocomposite of ZnO-polypyrrole. Sens. Actuators B Chem. 2015, 220, 903–909. [Google Scholar] [CrossRef]
- Sadrolhosseini, A.R.; Moksin, M.M.; Yunus, W.M.M.; Talib, Z.A.; Abdi, M.M. Surface plasmon resonance detection of copper corrosion in biodiesel using polypyrrole-chitosan layer sensor. Opt. Rev. 2011, 18, 331–337. [Google Scholar] [CrossRef]
- Abdi, M.M.; Abdullah, L.C.; Sadrolhosseini, A.R.; Yunus, W.M.; Moksin, M.M.; Tahir, P.M. Surface plasmon resonance sensing detection of mercury and lead ions based on conducting polymer composite. PLoS ONE 2011, 6, e24578. [Google Scholar] [CrossRef]
- Sadrolhosseini, A.; Moksin, M.M.; Yunus, W.M.M.; Talib, Z.A. Surface plasmon resonance characterization of virgin coconut oil biodiesel: Detection of iron corrosion using polypyrrole chitosan sensing layer. Sens. Mater. 2012, 24, 221–232. [Google Scholar] [CrossRef]
- Sadrolhosseini, A.R.; Noor, A.S.M.; Moksin, M.M.; M.abdi, M.; Mohammadi, A. Application of polypyrrole-chitosan layer for detection of Zn (II) and Ni (II) in aqueous solutions using surface plasmon resonance. Int. J. Polym. Mater. Polym. Biomater. 2013, 62, 284–287. [Google Scholar] [CrossRef]
- Verma, R.; Gupta, B.D. Detection of heavy metal ions in contaminated water by surface plasmon resonance based optical fibre sensor using conducting polymer and chitosan. Food Chem. 2015, 166, 568–575. [Google Scholar] [CrossRef] [PubMed]
- Sadrolhosseini, A.R.; Naseri, M.; Rashid, S.A. Polypyrrole-chitosan/nickel-ferrite nanoparticle composite layer for detecting heavy metal ions using surface plasmon resonance technique. Opt. Laser Technol. 2017, 93, 216–223. [Google Scholar] [CrossRef]
- Sadrolhosseini, A.R.; Naseri, M.; Kamari, H.M. Surface plasmon resonance sensor for detecting of arsenic in aqueous solution using polypyrrole-chitosan-cobalt ferrite nanoparticles composite layer. Opt. Commun. 2017, 383, 132–137. [Google Scholar] [CrossRef]
- Witkowski, A.; Brajter-Toth, A. Overoxidized polypyrrole films—A model for the design of permselective electrodes. Anal. Chem. 1992, 64, 635–641. [Google Scholar] [CrossRef]
- Cui, X.Y.; Martin, D.C. Electrochemical deposition and characterization of poly(3,4-ethylenedioxythiophene) on neural microelectrode arrays. Sens. Actuators B Chem. 2003, 89, 92–102. [Google Scholar] [CrossRef]
- Yang, Y.J.; Jiang, Y.D.; Xu, J.H.; Yu, J.S. Conducting PEDOT-PSS composite films assembled by LB technique. Colloids Surf. A Physicochem. Eng. Asp. 2007, 302, 157–161. [Google Scholar] [CrossRef]
- Chen, S.; Tao, H.; Wang, Y.; Ma, Z. Process optimization of soy protein isolate-based edible films containing nanocrystalline cellulose from sunflower seed hull and chitosan. Trans. Chin. Soc. Agric. Eng. 2016, 32, 306–314. [Google Scholar] [CrossRef]
| Metal Ion | Type of Polymers | Optical Sensors | Linear Range | Limit of Detection | References |
|---|---|---|---|---|---|
| Zn2+ | Chitosan modified with 5-formyl-3-hydroxy-4-hydroxymethyl-2-methylpyridine | Fluorescence | 0–25 µM | Zn2+: 1 µM | [27] |
| Cd2+ | Cd2+: 1 mM | ||||
| Ga3+ | Ga3+: 1 mM | ||||
| Cu2+ | Cadmium sulfide quantum dots modified by chitosan | Fluorescence | 8 nM–3 μM | 1.2 nM | [28] |
| Hg2+ | Chitosan nanoparticles | Fluorescence | 0–1 mM | 1 µM | [29] |
| Cu2+ | Magnetic and fluorescent bifunctional chitosan nanoparticles | Fluorescence | 1.967–393.416 µM | 0.724 µM | [30] |
| K+ | Chitosan/Ru(bpy)32+/silica nanoparticle-modified glass carbon electrode | Electrochemiluminescence | 0–9 nM | 0.3 nM | [31] |
| Hg2+ | Three-dimensional fluorescent chitosan hydrogel | Fluorescence | 5.0–50 nM | 0.9 nM | [32] |
| Hg2+ | Chitosan-functionalized gold nanoparticles | Colorimetric | 0.05–9 µM | 1.35 µM | [33] |
| Hg2+ | Chitosan thiomer | Colorimetric | 0–498.5 µM | 2.318 µM | [34] |
| Hg2+ | Chitosan microspheres | Fluorescence | 0.5–7 µM | 0.015 µM | [35] |
| Hg2+ | Chitosan–rhodamine | Fluorescence | 0–6 µM | 3.42 µM | [36] |
| Cd2+ | Chitosan dithiocarbamate functionalized gold nanoparticles | Colorimetric | 0.1–500 µM | 0.063 µM | [37] |
| Hg2+ | Chitosan–silver nanocomposite | Colorimetric | 0–500.870 µM | 7.2 nM | [38] |
| Cu2+ | Carbon dot-embedded fluorescent magnetic nanoparticles O-carboxymethyl chitosan | Fluorescence | 0.01–200 µM | 0.56 µM | [39] |
| Hg2+ | Chitosan/silver–gold nanocomposite | Colorimetric | - | 0.5 nM | [40] |
| Hg2+ | Chitosan stabilized gold nanoparticles and 2,6-pyridinedicarboxylic acid | Colorimetric | 0.3–5 µM | - | [41] |
| Al3+ | Chitosan/silver | Fluorescence | 2–180 µM | - | [42] |
| Cu2+ | Tetraphenylethylene–chitosan | Fluorescence | 5–100 µM | - | [43] |
| Cu2+ | Chitosan–Fluorescein isothiocyanate oligomer | Colorimetric | 0.1 µM–7 mM | 60 µM | [44] |
| Hg2+ | Chitosan–gold nanoparticles | Colorimetric | 0–13.6 µM | 0.02 µM | [45] |
| Hg2+ | Silver-doped cadmium sulfide quantum dots/chitosan | Colorimetric | 124–800 µM | 124 µM | [46] |
| Hg2+ | Thiol terminated chitosan capped silver nanoparticles | Colorimetric | 0–0.4 µM | 0.017 µM | [47] |
| Hg2+ | Chitosan functionalized gold nanoparticles assembled on Sulphur doped graphitic carbon nitride | Colorimetric | 0.1–0.5 µM | 0.275 nM | [48] |
| Hg2+ | Chitosan–gold nanocomposite | Colorimetric | 0–2 µM | 3.2 nM 0.05 µM | [49] |
| Ag+ | Chitosan functionalized gold nanoparticles | Colorimeter | 1–100 µM | 0.13 µM | [50] |
| Zn2+ | Cellulose film modified with 1-(2-pyridylazo)-2-naphthol (PAN) | Colorimetric | 10–100 µM | - | [51] |
| Mn2+ | |||||
| Ni2+ | |||||
| Hg2+ | Ruthenium dye or mercaptosilane monolayer onto metal oxide ultrathin film pre-coated cellulose nanofibers | Colorimetric | 0.050–100 µM | 0.049 µM | [52] |
| Cr3+ | 1,4-DHAQ-doped cellulose nanofiber Films | Fluorescence | Cu2+: 2.5–37.5 nM | - | [53] |
| Cr3+ | Cr3+: 2.5–25 nM | ||||
| Fe3+ | Pyrene–cellulose nanocrystals | Fluorescence | 0–5 mM | 1 µM | [54] |
| Pb2+ | Pyromellitic dianhydride-grafted cellulose nanofibrous membranes | Colorimetric | 0.048–5 µM | 0.048 µM | [55] |
| Hg2+ | Au nanoparticles/carboxylethyl quaternized cellulose | Colorimetric | 0–0.3 µM | 0.04 µM | [56] |
| Hg2+ | Cellulose–rhodamine | Fluorescence | 0–10 mM | 50 µM | [57] |
| Hg2+ | Cellulose–Lysine Schiff-Base | Fluorescence | 49.853–498 µM | 49.853 µM | [58] |
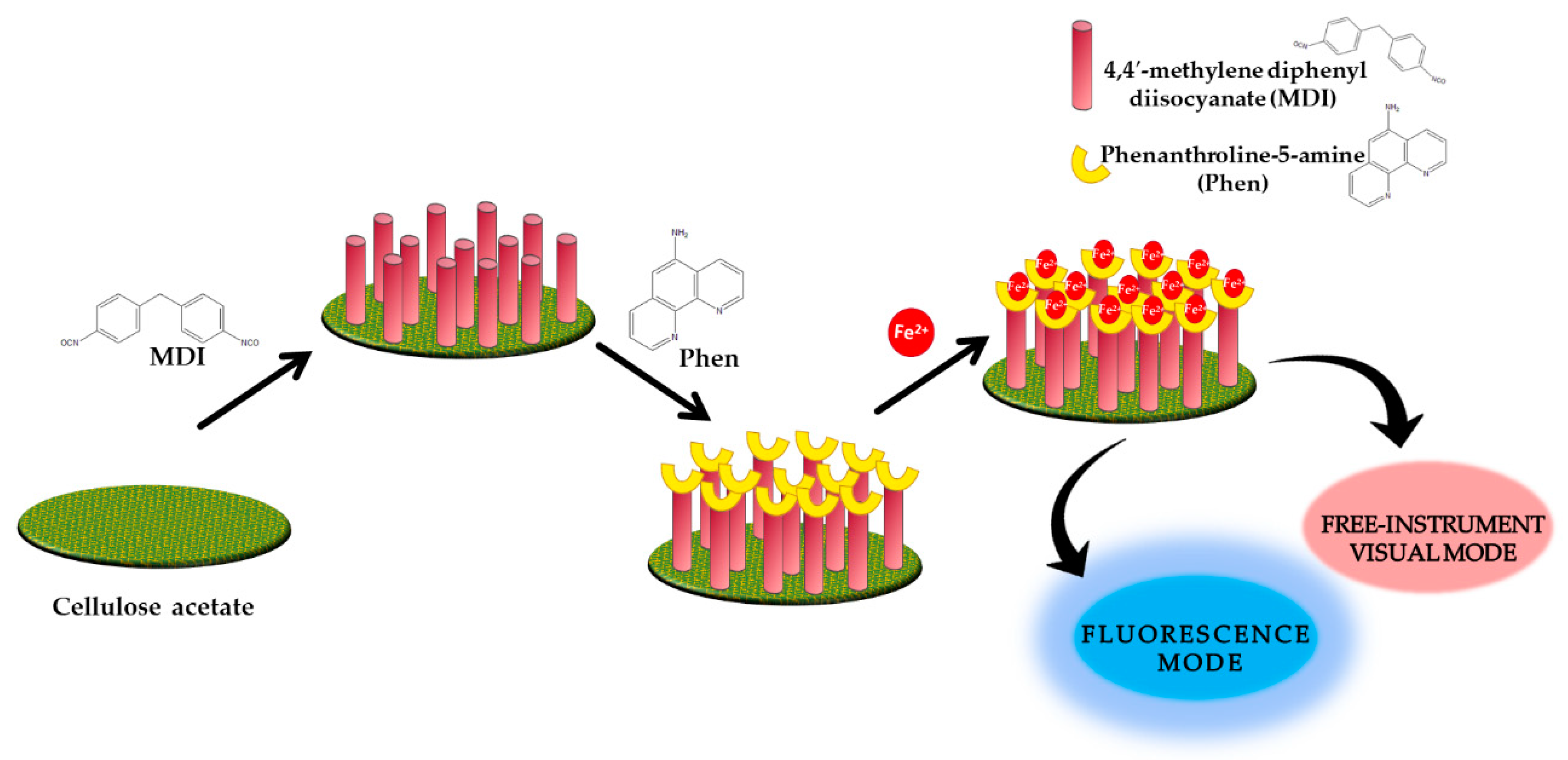
| Fe2+ | 1,10-phenanthroline-5-amine-4-4′-methylene diphenyl diisocyanate-cellulose acetate | Fluorescence | 0–17.8 µM | 46.6 nM 0.895 µM | [59] |
| Pb2+ | Nanocrystalline cellulose-ethylenediamine | Fluorescence | 18.7 nM–0.5 µM | 0.024 µM | [60] |
| Cu2+ | Rhodamine-B derivative and bacterial cellulose | Colorimeter | 4–128 µM | - | [61] |
| Fe3+ | Curcumin embedded bacterial cellulose | Colorimetric | 0.01–100 µM | 7.8 µM 8.2 µM | [62] |
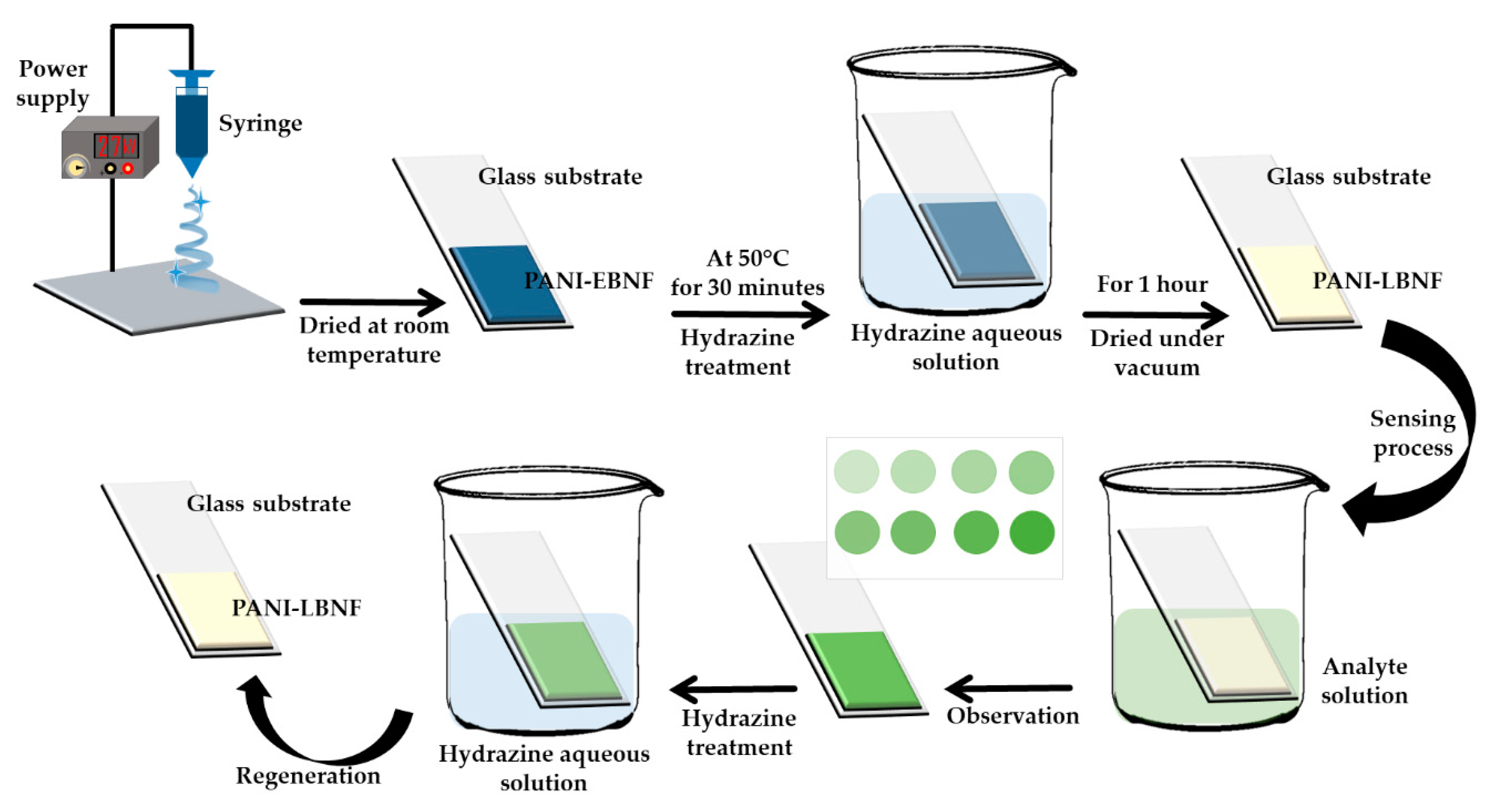
| Cu2+ | Polyaniline/polyamide-6 nano-fiber/net membranes | Colorimetric | 0–1573 µM | 15.737 µM | [63] |
| Hg2+ | Polyaniline leucoemeraldine base nanofibrous | Colorimetric | 0–150 µM | 0.005 µM | [64] |
| Hg2+ | Polyaniline/carbon dot nanocomposites | Fluorescence | 0.05–1 µM | 0.8 nM | [65] |
| Hg2+ | Polyaniline microspheres | Fluorescence | 0–1.5 µM | 0.86 nM | [66] |
| As3+ | Polyaniline nanofiber/carbon dot nanohybrid | Fluorescence | 0–0.026 µM | 1.3 pM | [67] |
| Fe3+ | Nanostructured polypyrrole film doped Tiron | Fluorescence | 0.05–1 µM | 0.01 µM | [68] |
| Pb2+ | Benzene sulfonic acid doped polypyrrole | Fluorescence | 0–9 µM | Pb2+: 0.018 µM | [69] |
| Cu2+ | Cu2+: 3.1 nM |
| Metal Ion | Type of Polymers | Linear Range | Limit of Detection | References |
|---|---|---|---|---|
| Fe3+ | Chitosan thin film | 4.477 µM–1.79 nM | 4.477 µM | [58] |
| Cr6+ | Gold nanoparticles/chitosan composite | 192.322 µM–4.808 nM | 192.322 µM | [71] |
| Hg2+ | Chitosan cross-linked glutaraldehyde solution | Hg2+: 2.493–498.529 µM | Hg2+: 2.493 µM | [72] |
| Cu2+ | Cu2+: 7.868 µM–1.573 nM | Cu2+: 7.868 µM | ||
| Cu2+ | Chitosan cross-linked glutaraldehyde solution | Cu2+: 7.868 µM–1.573 mM | Cu2+: 7.868 µM | [73] |
| Zn2+ | Zn2+: 7.648 µM–1.529 mM | Zn2+: 7.648 µM | ||
| Mn2+ | Mn2+: 9.101 µM–1.820 mM | Mn2+: 9.101 µM | ||
| Pb2+ | Immobilized p-tert-butylcalix[4]arene-tetrakis in chitosan thin film | 0.144–24.131 µM | 0.144 µM | [74] |
| Pb2+ | Crosslinked chitosan | 2.413–482.625 µM | 2.413 µM | [75] |
| Pb2+ | Gold–chitosan–graphene oxide nanostructured thin films | 0.145–24.131 µM | 0.145 µM | [76] |
| Zn2+ | Chitosan–tetrabutyl thiuram disulfide | 0.153–76.476 µM | 0.153 µM | [77] |
| Pb2+ | Chitosan–graphene oxide | 0.483–24.131 µM | 0.483 µM | [78] |
| Pb2+ | Gold/silver/gold/chitosan–graphene oxide | Pb2+: 0.483–24.131 µM | Pb2+: 0.483 µM | [79] |
| Hg2+ | Hg2+: 0.499–24.926 µM | Hg2+: 0.499 µM | ||
| K+ | Valinomycin doped chitosan–graphene oxide thin film | 0.0256 µM–2.557 mM | 0.0256 µM | [80] |
| Co2+ | Immobilized 4-(2-pyridylazo) resorcinol in chitosan–graphene oxide composite thin film | 0.169 µM–1.696 mM | 0.169 µM | [80] |
| Co2+ | Cadmium sulfide quantum dots–graphene oxide–chitosan nanocomposite thin film | 0.169–169.684 µM | 0.169 µM | [82] |
| Pb2+ | Chitosan–glutathione coated sensor probe | 4.826–33.784 µM | 6.274 µM | [83] |
| Pb2+ | Gold nanoparticle kappa-carrageenan and chitosan | 0.048–24.131 µM | 0.048 µM | [84] |
| Fe2+ | Chitosan/hydroxyl-functionalized graphene quantum dots thin film | 8.953 µM–1.79 mM | 8.953 µM | [85] |
| Hg2+ | Chitosan/carboxyl-functionalized graphene quantum dots thin film | 2.493–498.529 µM | 2.493 µM | [86] |
| Pb2+ | Chitosan–poly(amidoamine) dendrimer composite thin film | 0.483–2.413 µM | 0.483 µM | [87] |
| Cu2+ | Chitosan/poly(acrylic acid) bilayers | 3.147 µM–7.868 mM | 0.1054 µM | [88] |
| Cu2+ | Silver/gold composite film modified by modified-chitosan (MCS) thin film | 15.737–78.683 µM | 0.283 µM | [89] |
| Co2+ | Chitosan–graphene oxide/cadmium sulphide quantum dots active layer | 0.169 µM–1.696 mM | 0.169 µM | [90] |
| Co2+ | Chitosan–graphene oxide–cadmium sulfide quantum dots composite thin film | 1.697 µM–1.696 mM | 1.697 µM | [91] |
| Cu2+ | Nanocrystalline cellulose modified by hexadecyltrimethylammonium bromide and graphene oxide composite thin film | 157.366–944.198 µM | 157.366 µM | [92] |
| Ni2+ | Nanocrystalline cellulose–graphene oxide-based nanocomposite | 0.170–170.378 µM | 0.170 µM | [93] |
| Zn2+ | Modified-nanocrystalline cellulose/graphene oxide | 0.153–152.952 µM | 0.153 µM | [94] |
| Hg2+ | Polypyrrole and 2-mercaptobenzothiazole | 0.498–49.853 µM | 0.0498 µM | [95] |
| Cu2+ | Polypyrrole thin film | Cu2+: 1.574–314.733 µM | Cu2+: 1.574 µM | [96] |
| Fe3+ | Fe3+: 1.791–358.134 µM | Fe3+: 1.791 µM | ||
| Hg2+ | Polypyrrole multi-walled carbon nanotube composite layer | Hg2+: 0.498–498.529 µM | Hg2+: 0.498 µM | [97] |
| Pb2+ | Pb2+: 0.483–482.625 µM | Pb2+: 0.483 µM | ||
| Fe2+ | Fe2+: 1.791 µM–1.79 mM | Fe2+: 1.791 µM | ||
| Pb2+ | Gold nanoparticles/graphene oxide/polyaniline nanocomposites film | 0.145–14.479 µM | 0.145 µM | [98] |
| Al3+ | Polypyrrole multiwalled carbon nanotube composite layer | 3.706 µM–3.706 mM | 3.706 µM | [99] |
| Mn2+ | Nanocomposite of polypyrrole and zinc oxide over silver | 0–0.2 M | 0.673 µM | [100] |
| Cu2+ | Polypyrrole–chitosan layer | 1.574 µM–1.573 mM | 1.574 µM | [101] |
| Hg2+ | Polypyrrole–chitosan conducting polymer composite | 2.493 µM–59.824 µM | 2.493 µM | [102] |
| Pb2+ | ||||
| Fe3+ | Polypyrrole–chitosan layer | 1.791 µM–1.343 mM | 1.791 µM | [103] |
| Zn2+ | Polypyrrole–chitosan | Zn2+: 0.153–59.824 µM | Zn2+: 0.153 µM | [104] |
| Ni2+ | Ni2+: 0.170 µM–1.277 mM | Ni2+: 0.170 µM | ||
| Cd2+ | Polypyrrole and chitosan/ITO/silver | - | Cd2+: 1.29 nM | [105] |
| Pb2+ | Pb2+: 1.58 nM | |||
| Hg2+ | Hg2+: 2.93 nM | |||
| Ni2+ Fe2+ Co2+ Al2+ Mg2+ Hg2+ Pb2+ | Polypyrrole–chitosan/nickel–ferrite nanoparticle composite layer | - | Ni2+, Fe2+, Co2+: 17.3 nM Al2+, Mg2+: 37.1 nM Hg2+, Pb2+: 4.82 nM | [106] |
| V | Polypyrrole–chitosan–cobalt ferrite nanoparticles composite layer | 0.0133–1334.721 µM | 13.3 nM | [107] |
| Optical Sensor | Biopolymers/Conducting Polymers | Lowest Limit of Detection | Heavy Metal Ion | Reference |
|---|---|---|---|---|
| Electrochemiluminescence | Chitosan/Ru(bpy)32+/silica nanoparticle-modified glass carbon electrode | 0.3 nM | K+ | [31] |
| Colorimetric | Chitosan functionalized gold nanoparticles assembled on sulphur doped graphitic carbon nitride | 0.275 nM | Hg2+ | [48] |
| Fluorescence | Polyaniline nanofiber/carbon dot nanohybrid | 1.3 pM | As3+ | [67] |
| Surface plasmon resonance | Polypyrrole–chitosan/ITO/silver | 1.29 nM | Cd2+ | [104] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramdzan, N.S.M.; Fen, Y.W.; Anas, N.A.A.; Omar, N.A.S.; Saleviter, S. Development of Biopolymer and Conducting Polymer-Based Optical Sensors for Heavy Metal Ion Detection. Molecules 2020, 25, 2548. https://doi.org/10.3390/molecules25112548
Ramdzan NSM, Fen YW, Anas NAA, Omar NAS, Saleviter S. Development of Biopolymer and Conducting Polymer-Based Optical Sensors for Heavy Metal Ion Detection. Molecules. 2020; 25(11):2548. https://doi.org/10.3390/molecules25112548
Chicago/Turabian StyleRamdzan, Nur Syahira Md, Yap Wing Fen, Nur Ain Asyiqin Anas, Nur Alia Sheh Omar, and Silvan Saleviter. 2020. "Development of Biopolymer and Conducting Polymer-Based Optical Sensors for Heavy Metal Ion Detection" Molecules 25, no. 11: 2548. https://doi.org/10.3390/molecules25112548
APA StyleRamdzan, N. S. M., Fen, Y. W., Anas, N. A. A., Omar, N. A. S., & Saleviter, S. (2020). Development of Biopolymer and Conducting Polymer-Based Optical Sensors for Heavy Metal Ion Detection. Molecules, 25(11), 2548. https://doi.org/10.3390/molecules25112548







