Optical Properties of Composites Based on Graphene Oxide and Polystyrene
Abstract
1. Introduction
2. Materials and Methods
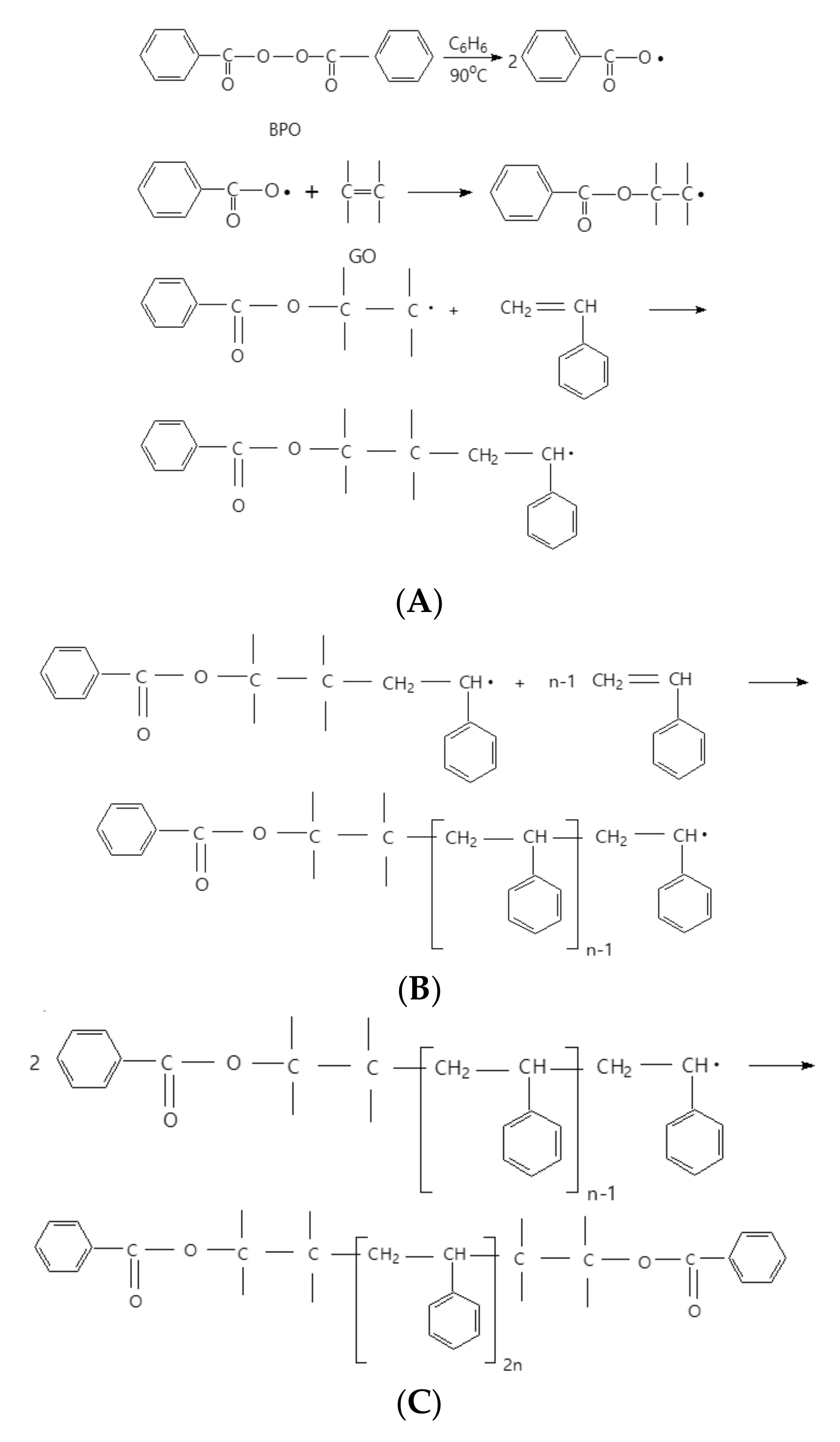
3. Results and Discussions
3.1. Optical Properties of the PS Microspheres and Their Composites with GO
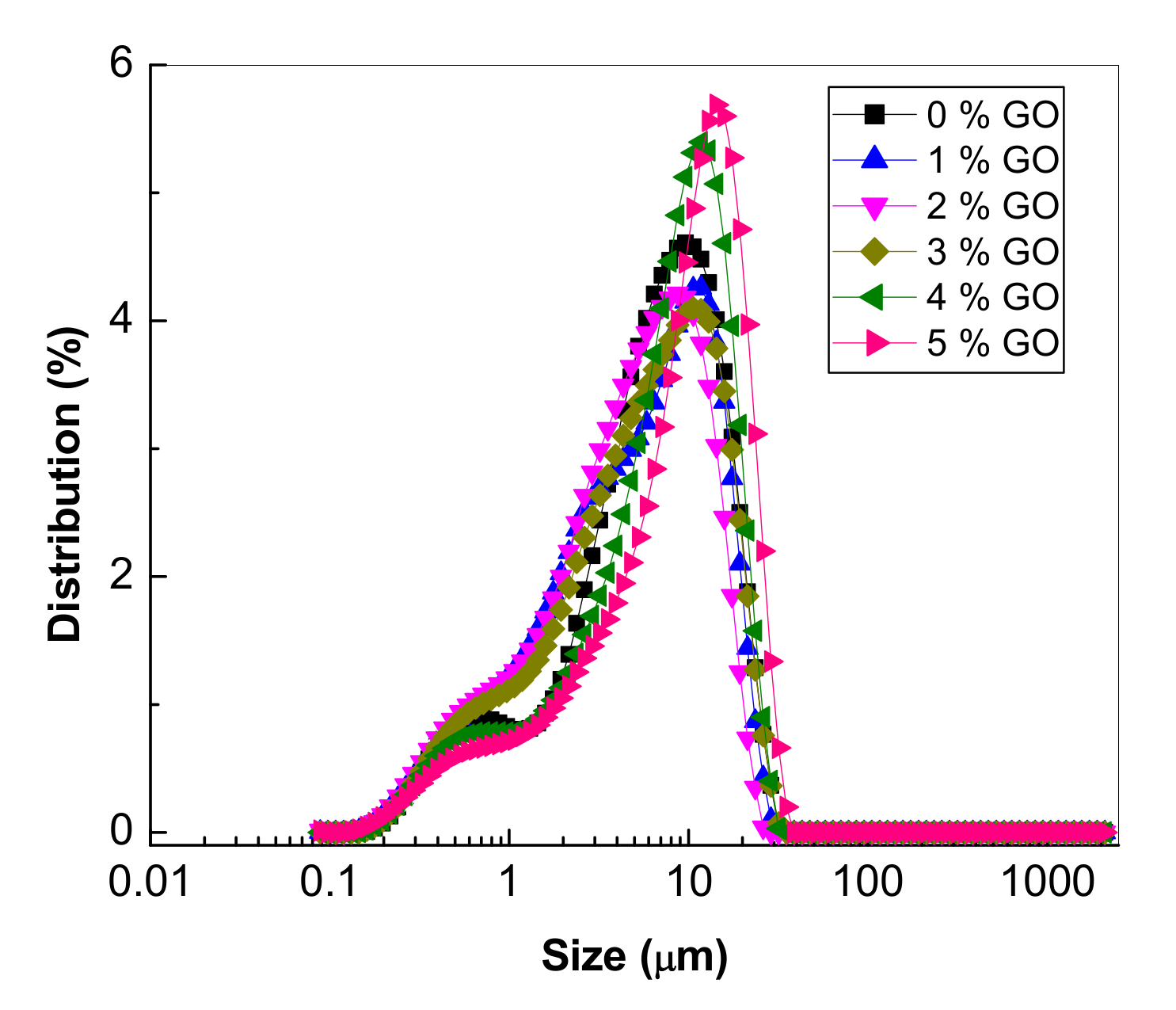
3.1.1. Morphological Properties of the PS Microspheres and Their Composites with GO
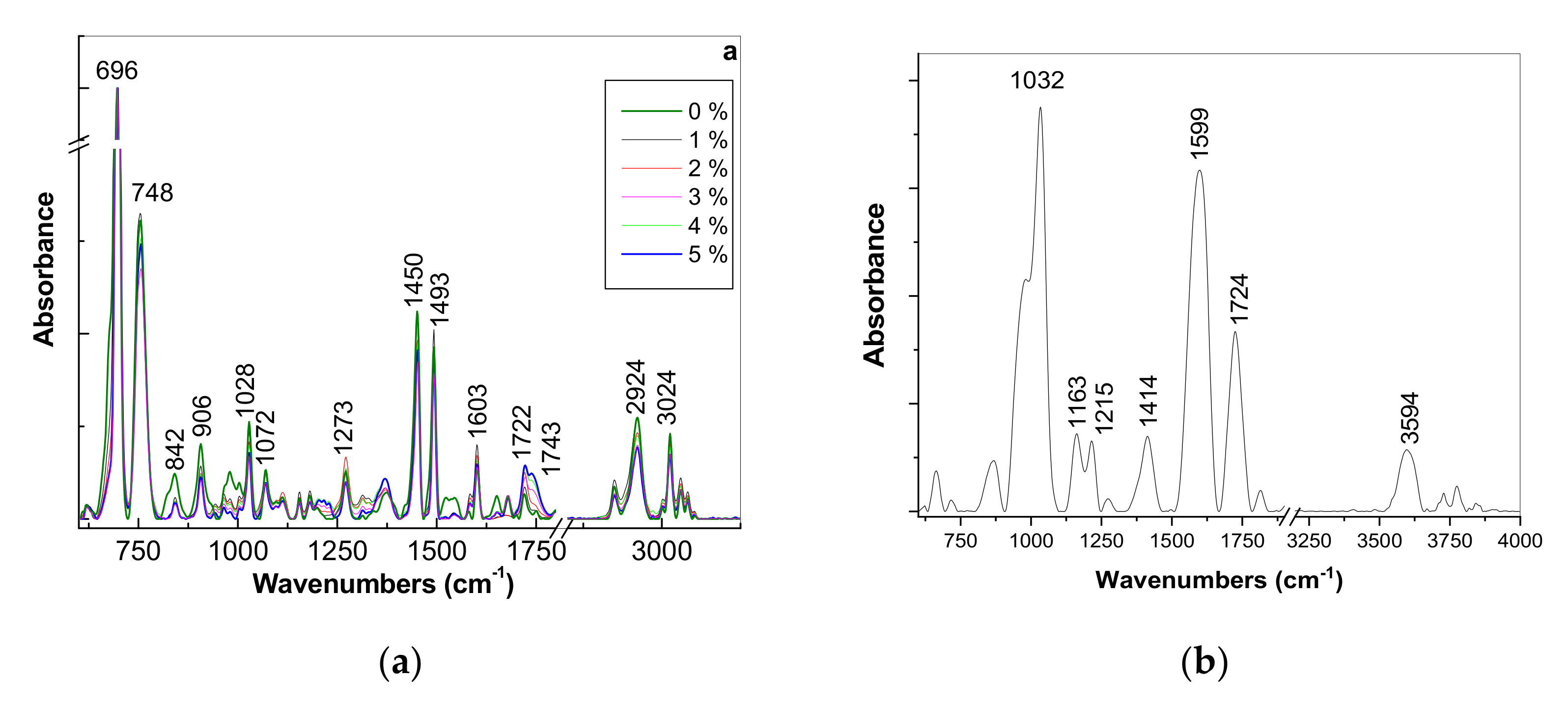
3.1.2. Vibrational Properties of the PS Microspheres and Their Composites with GO
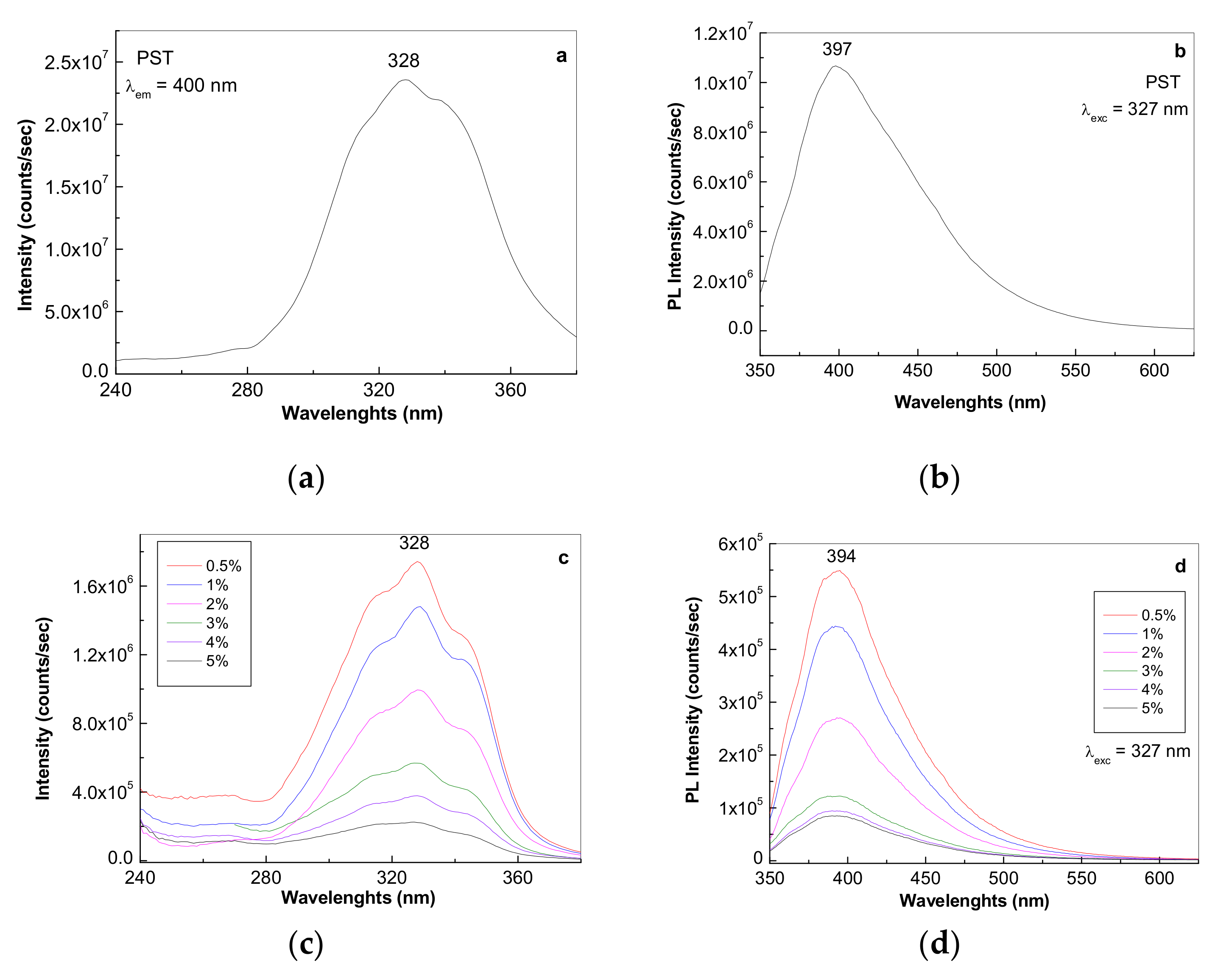
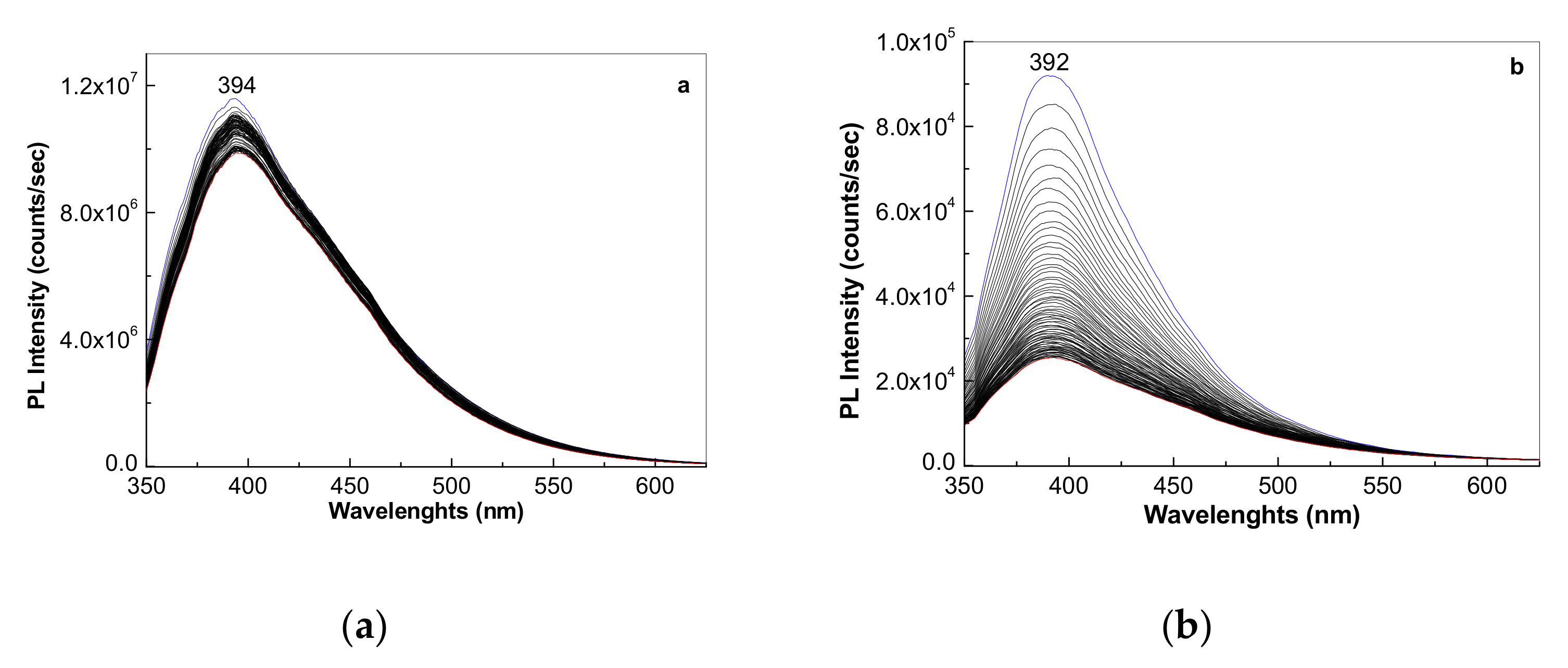
3.1.3. Photoluminescence of the PS Microspheres and Their Composites with GO
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cui, J.; Zhou, S. Facile fabrication of highly conductive polystyrene/nanocarbon composites with robust interconnected network via electrostatic attraction strategy. J. Mater. Chem. C 2018, 6, 550–557. [Google Scholar] [CrossRef]
- Wang, X.; Tan, D.; Chu, Z.; Chen, L.; Chen, X.; Zhao, J.; Chen, G. Mechanical properties of polymer composites reinforced by functionalized graphene prepared via direct exfoliation of graphite flakes in styrene. RSC Adv. 2016, 6, 112486–112492. [Google Scholar] [CrossRef]
- Baibarac, M.; Baltog, I.; Lefrant, S.; Mevellec, J.Y.; Bucur, C. Vibrational and photoluminescence properties of the polystyrene functionalized single-walled carbon nanotubes. Diam. Relat. Mater. 2008, 17, 1380–1388. [Google Scholar] [CrossRef]
- Wu, N.; She, X.; Yang, D.; Wu, X.; Su, F.; Chen, Y. Synthesis of network reduced graphene oxide in polystyrene matrix by a two-step reduction method for superior conductivity of the composite. J. Mater. Chem. 2012, 22, 17254–17261. [Google Scholar] [CrossRef]
- Wan, Y.J.; Yang, W.H.; Yu, S.H.; Sun, R.; Wong, C.P.; Liao, W.H. Covalent polymer functionalization of graphene for improved dielectric properties and thermal stability of epoxy composites. Compos. Sci. Technol. 2016, 122, 27–35. [Google Scholar] [CrossRef]
- Han, Y.; Wu, Y.; Shen, M.; Huang, X.; Zhu, J.; Zhang, X. Preparation and properties of polystyrene nanocomposites with graphite oxide and graphene as flame retardants. J. Mater. Sci. 2013, 48, 4214–4222. [Google Scholar] [CrossRef]
- Hu, W.; Yu, B.; Jiang, S.D.; Song, L.; Hu, Y.; Wang, B. Hyper-branched polymer grafting graphene oxide as an effective flame retardant and smoke suppressant for polystyrene. J. Hazard. Mater. 2015, 300, 58–66. [Google Scholar] [CrossRef]
- Kattimmuttathu, S.I.; Krishnappan, C.; Vellorathekkaepadil, V.; Nutenki, R.; Mandapati, V.R.; Cernik, M. Synthesis, characterization and optical properties of graphene oxide-polystyrene nanocomposites. Polym. Adv. Technol. 2015, 26, 214–222. [Google Scholar]
- Han, Y.; Wang, T.; Gao, X.; Li, T.; Zhang, Q. Preparation of thermally reduced graphene oxide and the influence of its reduction temperature on the thermal, mechanical, flame retardant performances of PS nanocomposites. Compos. Part A 2016, 84, 336–343. [Google Scholar] [CrossRef]
- Kim, Y.N.; Lee, N.H.; Yun, D.Y.; Kim, T.W. Multilevel characteristics and operating mechanisms of nonvolatile memory devices based on a floating gate of graphene oxide sheets sandwiched between two polystyrene layers. Org. Electron. 2015, 25, 165–169. [Google Scholar] [CrossRef]
- Wang, Z.; Li, S.; Wu, Z. The fabrication and properties of a graphite nanosheet/polystyrene composite based on graphite nanosheets treated with supercritical water. Comp. Sci. Technol. 2015, 112, 50–57. [Google Scholar] [CrossRef]
- Khobragade, P.S.; Hansora, D.P.; Nick, J.B.; Njuguna, J.; Mishra, S. Physico-mechanical properties of nano-polystyrene-decorated graphene oxide-epoxy composites. Polym. Int. 2017, 66, 1402–1409. [Google Scholar] [CrossRef]
- Li, Y.; Xu, Y.; Zhaou, T.; Zhang, A.; Bao, J. A method to construct perfect 3D polymer/graphene oxide core-shell microspheres via electrostatic self-assembly. RSC Adv. 2015, 5, 32469–32478. [Google Scholar] [CrossRef]
- Yeole, N.; Kutcherlapati, S.N.R.; Jana, T. Polystyrene-graphene oxide (GO) nanocomposite synthesized by interfacial interactions between RAFT modified GO and core-shell polymeric nanoparticles. J. Colloid. Interface Sci. 2015, 43, 137–142. [Google Scholar] [CrossRef]
- Zhang, L.; Tu, S.; Wang, H.; Du, Q. Preparation of polymer/graphene oxide nanocomposites by a two-step strategy composed of in situ polymerization and melt processing. Compos. Sci. Technol. 2018, 154, 1–7. [Google Scholar] [CrossRef]
- Cotet, L.C.; Magyari, K.; Todea, M.; Dudescu, M.C.; Danciu, V.; Baia, L. Versatile self-assembled graphene oxide membranes obtained under ambient conditions by using a water-ethanol suspension. J. Mat. Chem. A 2017, 5, 2132–2142. [Google Scholar] [CrossRef]
- Pérez Del Pino, A.; György, E.; Cotet, C.; Baia, L.; Logofatu, C. Laser-induced chemical transformation of free-standing graphene oxide membranes in liquid and gas ammonia environments. RSC Adv. 2016, 6, 50034–50042. [Google Scholar] [CrossRef]
- Reynolds, N.M.; Hsu, S.L. A normal vibrational analysis of syndiotactic polystyrene. Macromolecules 1990, 23, 3463–3471. [Google Scholar] [CrossRef]
- Jasse, B.; Chao, R.S.; Koening, J.L. Laser Raman scattering in uniaxially oriented atactic polystyrene. J. Polym. Sci. Polym. Phys. 1978, 16, 2157–2169. [Google Scholar] [CrossRef]
- Kellar, E.J.C.; Galiotis, C.; Andrews, E.H. Raman vibrational studies of syndiotactic polystyrene. 1. Assignments in a conformational/crystallinity sensitive spectral region. Macromolecules 1996, 29, 3515–3520. [Google Scholar] [CrossRef]
- Eigler, S.; Dotzer, C.; Hirsch, A. Visualization of defect densities in reduced graphene oxide. Carbon 2012, 50, 3666–3673. [Google Scholar] [CrossRef]
- Eggl, P.; Pink, D.; Quinn, B.; Ringsdorf, H.; Sackmann, E. Diffusion in quasi two-dimensional macromolecular solutions. Macromolecules 1990, 23, 3472–3480. [Google Scholar] [CrossRef]
- Najo, H.K.; Oda, A.M.; Abdulaljeleel, W.; Abdilkadhim, H.; Hefdhi, R. ZnO-Ag/PS and ZnO/PS films for photocatalytic degradation of methylene blue. Indones.J. Chem. 2020, 20, 314–323. [Google Scholar] [CrossRef]
- Wu, H.D.; Wu, S.C.; Wu, I.D.; Chang, F.C. Novel determination of the crystallinity of syndiotactic polystyrene using FTIR spectrum. Polymer 2001, 42, 4719–4725. [Google Scholar] [CrossRef]
- Bhutto, A.A.; Vesely, D.; Gabrys, B.J. Miscibility and interactions in polystyrene and sodium sulfonated polystyrene with poly (vinyl methyl ether) PVME blends. Part II. FTIR. Polymer 2003, 44, 6627–6631. [Google Scholar] [CrossRef]
- Wild, S.; Kesmodel, L.L.; Apai, G. High-resolution electron energy loss spectroscopy of polystyrene surfaces. J. Phys. Chem. B 2000, 104, 3179–3182. [Google Scholar] [CrossRef]
- Coates, J. Interpretation of Infrared Spectra, A Practical Approach. In Encyclopedia of Analytical Chemistry; Meyers, R.A., Ed.; John Wiley& Sons Ltd.: Hoboken, NJ, USA, 1999; Volume 12, pp. 1–22. [Google Scholar]
- Herman, V.; Takacs, H.; Duclairoir, F.; Renault, O.; Tortai, J.H.; Viala, B. Core double-shell cobalt/graphene/polystyrene magnetic nanocomposites synthesized by in situ sonochemical polymerization. RSC Adv. 2015, 5, 51371–51381. [Google Scholar] [CrossRef]
- Fei, H.; Wu, G.; Cheng, W.Y.; Yan, W.; Xu, H.; Zhnag, D.; Zhao, Y.; Lv, Y.; Chen, Y.; Zhang, L.; et al. Enhanced NO2 sensing at room temperature with graphene via monodisperse polystyrene bead decoration. ACS Omega 2019, 4, 3812–3819. [Google Scholar] [CrossRef]
- Wang, Y.Z.; Qing, Y.B.; Sun, Y.; Zhu, M.; Dong, S.B. A study on preparation of modified graphene oxide and flame retardancy of polystyrene composite microspheres. Design. Monomers Polym. 2020, 23, 1–5. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds PS, GO and their composites are available from the authors. |

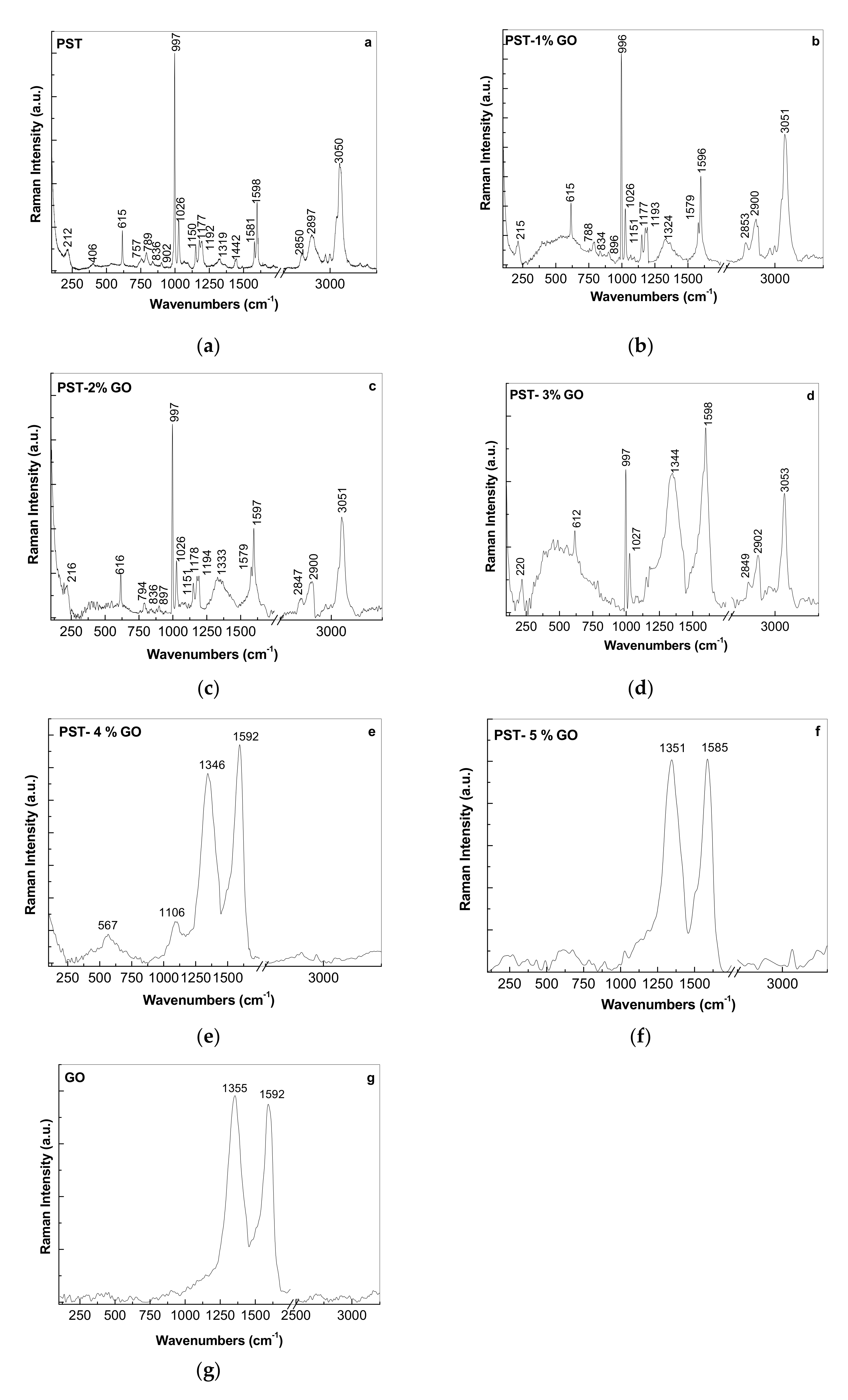


| ν, cm−1 | Assignment of Vibrational Modes [18,19,20] |
|---|---|
| 615 | out-of-plane benzene (B) ring deformation |
| 758 | CC stretching + ring CC stretching + ring trigonal + asymmetric deformation |
| 795 | out-of-plane C-H bending in mono-substituted B ring |
| 996 | in-plane bending C-H rocking |
| 1028 | C-H bending tangential E1u |
| 1151-1177-1195 | CC stretching + tangential C-H bending B2u |
| 1326 | C-H bending mode A2g + C-O stretching |
| 1448 | C-H bending mode in ethylene (B3u) |
| 1579–1598 | C=C stretching |
| 2850–2906 | C-H stretching mode on the polymer chain |
| 3053 | radial C-H stretching mode E1u |
| ν, cm−1 | Assignment of Vibrational Modes [21] |
|---|---|
| 1032 | C=C stretching mode |
| 1163 | C-O vibrational mode in ether group |
| 1212 | OH group at the GO ends |
| 1414 | the deformation mode of OH in the COOH group |
| 1599 | H2O adsorbed on the GO surface |
| 1724 | C=O stretching mode in COOH/CHO |
| 3594 | OH stretch vibration in the free H2O molecules |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stroe, M.; Cristea, M.; Matei, E.; Galatanu, A.; Cotet, L.C.; Pop, L.C.; Baia, M.; Danciu, V.; Anghel, I.; Baia, L.; et al. Optical Properties of Composites Based on Graphene Oxide and Polystyrene. Molecules 2020, 25, 2419. https://doi.org/10.3390/molecules25102419
Stroe M, Cristea M, Matei E, Galatanu A, Cotet LC, Pop LC, Baia M, Danciu V, Anghel I, Baia L, et al. Optical Properties of Composites Based on Graphene Oxide and Polystyrene. Molecules. 2020; 25(10):2419. https://doi.org/10.3390/molecules25102419
Chicago/Turabian StyleStroe, Malvina, Mirela Cristea, Elena Matei, Andrei Galatanu, Liviu C. Cotet, Lucian C. Pop, Monica Baia, Virginia Danciu, Ion Anghel, Lucian Baia, and et al. 2020. "Optical Properties of Composites Based on Graphene Oxide and Polystyrene" Molecules 25, no. 10: 2419. https://doi.org/10.3390/molecules25102419
APA StyleStroe, M., Cristea, M., Matei, E., Galatanu, A., Cotet, L. C., Pop, L. C., Baia, M., Danciu, V., Anghel, I., Baia, L., & Baibarac, M. A. (2020). Optical Properties of Composites Based on Graphene Oxide and Polystyrene. Molecules, 25(10), 2419. https://doi.org/10.3390/molecules25102419









