Foliar Application of Bio-Stimulants Enhancing the Production and the Toxicity of Origanum majorana Essential Oils Against Four Rice Seed-Borne Fungi
Abstract
1. Introduction
2. Results and Discussion
2.1. Field Study
2.1.1. Effects of Ascorbic Acid, Tryptophan and Moringa Leaf Extract on Vegetative Growth
2.1.2. Effects of Ascorbic Acid, Tryptophan and Moringa Extract on the Chemical Parameters of O. majorana
Oil Percentage (%)
Total Chlorophyll and Carbohydrate Content
Chemical Composition of the Essential Oils
2.2. In Vitro Study
2.2.1. Isolation and Identification of the Fungal Isolates
2.2.2. Molecular Identification through the Internal Transcribed Spacer (ITS) Region
2.3. Antifungal Activity of Essential Oils
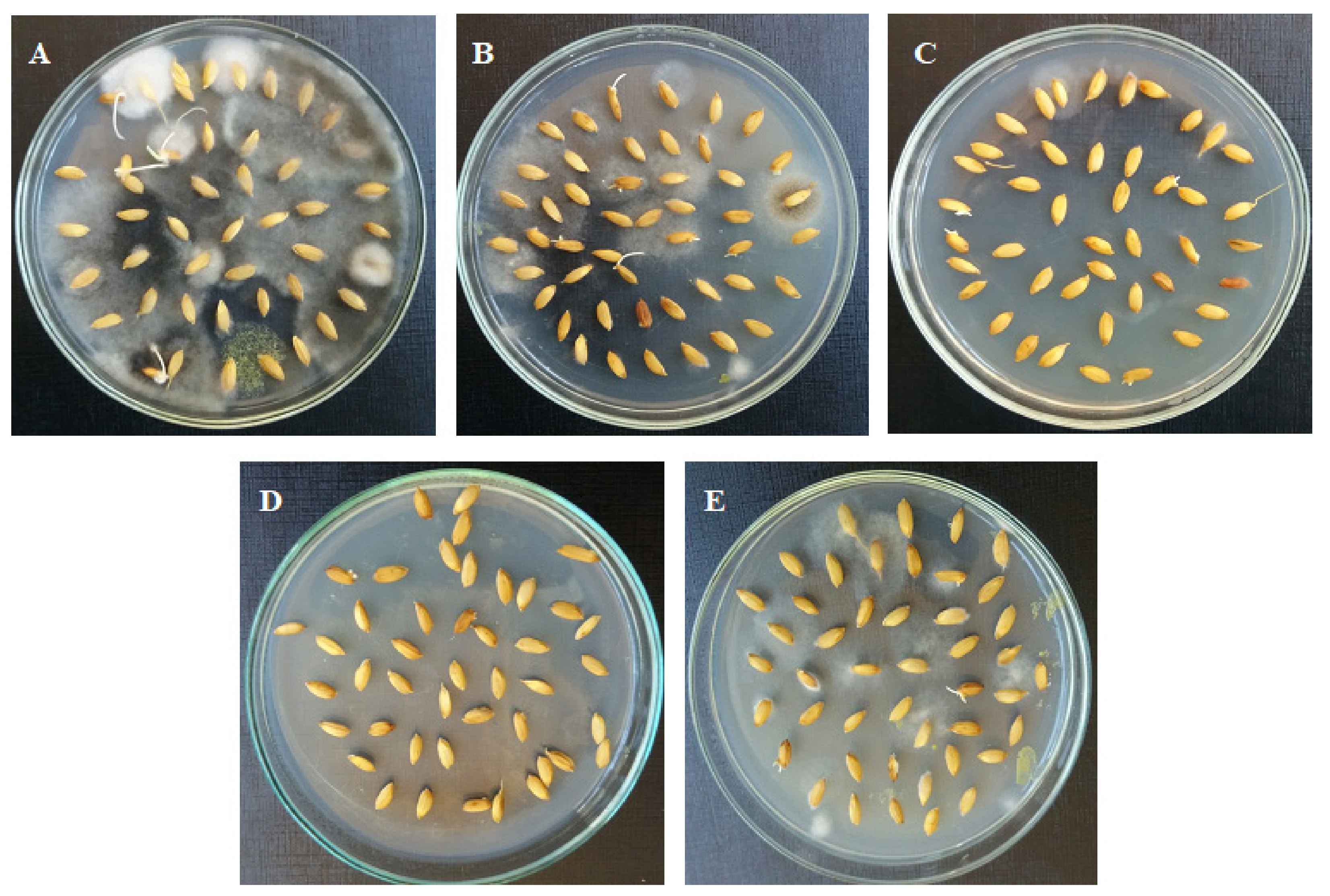
2.4. Efficacy of Seed Treatment with Essential Oils Against Seed-Borne Fungi
3. Materials and Methods
3.1. Preparation of Aqueous Extracts of Moringa Oleifera, Ascorbic Acid and Tryptophan
3.2. Field Study
3.3. Vegetative Growth and Chemical Parameters
3.4. Extraction of the Essential Oils
3.5. GC/MS Analysis of the Essential Oils
3.6. In Vitro Study for Fungal Isolation
3.7. In Vitro Study for Fungal Identification
3.7.1. Culture Characteristics and Microscopic Examination
3.7.2. Molecular Identification through the Internal Transcribed Spacer (ITS) Region
3.7.3. Sequencing of the Amplified ITS Region
3.8. Antifungal Activity and Minimum Inhibitory Concentration (MIC) Assays of Essential Oils (First Investigation)
3.9. Efficacy of Seed Treatment with Selected Essential Oils Against Seed-Borne Fungi (Second Investigation)
3.10. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- El-Khateeb, M.A.; El-Attar, A.B.; Nour, R.M. Application of plant biostimulants to improve the biological responses and essential oil production of marjoram (Majorana hortensis, Moench) plants. Middle East J. Agric. Res. 2017, 7, 928–941. [Google Scholar]
- Hashem, H.A.E.A. Response of Marjoram (Majorana Hortensis L.) Plant to foliar spraying by some antioxidants under Siwa Oasis conditions. J. Agric. Vet. Sci. 2018, 11, 51–58. [Google Scholar]
- Kovaaevia, N. Folk Medicine, 1st ed.; Faculty of Pharmacy, University of Belgrade: Belgrade, Serbia, 2001. [Google Scholar]
- Bağci, Y.; Kan, Y.; Doğu, S.; Çelik, S.A. The essential Oil compositions of Origanum majorana L. cultivated in Konya and collected from Mersin-Turkey. Indian J. Pharm. Educ. Res. 2017, 51, s463–s469. [Google Scholar]
- Heghes, S.C.; Vostinaru, O.; Rus, L.M.; Mogosan, C.; Iuga, C.A.; Filip, L. Antispasmodic Effect of Essential Oils and Their Constituents. Molecules 2019, 24, 1675. [Google Scholar] [CrossRef]
- El-Sabrout, A.M.; Salem, M.Z.M.; Bin-Jumah, M.; Allam, A.A. Toxicological activity of some plant essential oils against Tribolium castaneum and Culex pipiens larvae. Processes 2019, 7, 933. [Google Scholar] [CrossRef]
- Salem, M.Z.M.; Hamed, S.A.M.; Mansour, M.M.A. Assessment of efficacy and effectiveness of some extracted bio-chemicals as bio-fungicides on Wood. Drv. Ind. 2019, 70, 337–350. [Google Scholar] [CrossRef]
- Lambert, R.J.W.; Skandamis, P.N.; Coote, P.; Nychas, G.J.E. A study of the minimum inhibitory concentration and mode of action of oregano essential oil, thymol, and carvacrol. J. Appl. Microbiol. 2001, 91, 453–462. [Google Scholar] [CrossRef]
- Rowayshed, G.H.; Abd-Elhameed, A.A.; Abd-Elghany, M.E.A.; Shahat, A.A.; Younes, O.A.A. Effective chemical compounds and antibacterial activities of Marjoram leaves, Teucrium leaves and Fennel fruits essential oils. Middle East J. Appl. Sci. 2014, 4, 637–647. [Google Scholar]
- Tripathy, B.; Satyanarayana, S.; Abedulla Khan, K.; Raja, K.; Tripathy, S. Preliminary Phytochemical Screening and Comparison study of In Vitro Antioxidant activity of selected Medicinal Plants. Int. J. Pharm. Life Sci. 2017, 8, 5598–5604. [Google Scholar]
- Du Jardin, P. Plant Biostimulants: Definition, Concept, Main Categories and Regulation. Sci. Hortic. 2015, 196, 3–14. [Google Scholar] [CrossRef]
- Chiaiese, P.; Corrado, G.; Colla, G.; Kyriacou, M.C.; Rouphae, Y. Renewable Sources of Plant Biostimulation: Microalgae as a Sustainable Means to Improve Crop Performance. Front. Plant Sci. 2018, 9, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Drobek, M.; Frąc, M.; Cybulska, J. Plant Biostimulants: Importance of the quality and yield of horticultural crops and the improvement of plant tolerance to abiotic stress—A Review. Agronomy 2019, 9, 335. [Google Scholar] [CrossRef]
- Rouphael, Y.; Spichal, L.; Panzarova, K.; Casa, R.; Colla, G. High-Throughput Plant Phenotyping for Developing Novel Biostimulants: From Lab to Field or from Field to Lab? Front. Plant Sci. 2018, 9, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Bradshaw, T.L.; Berkett, L.P.; Grith, M.C.; Darby, H.M.; Moran, R.E.; Garcia, M.E. Assessment of Kelp Extract Biostimulants on Tree Growth, Yield, and Fruit Quality in a Certified Organic Apple Orchard. In Proceedings of the II International Organic Fruit Symposium, Leavenworth, WA, USA, 18–22 June 2012; International Society for Horticultural Science: Brabant, Belgium, 2013; pp. 191–198. [Google Scholar]
- Souri, M.K.; Bakhtiarizade, M. Biostimulation effects of Rosemary essential oil on growth and nutrient uptake of Tomato seedlings. Sci. Hortic. 2019, 243, 472–476. [Google Scholar] [CrossRef]
- Tejada, M.; Rodríguez-Morgado, B.; Gómez, I.; Franco-Andreu, L.; Benítez, C.; Parrado, J. Use of Biofertilizers Obtained from Sewage Sludges on Maize Yield. Eur. J. Agron. 2016, 78, 13–19. [Google Scholar] [CrossRef]
- Foidl, N.; Makkar, H.P.S.; Becker, K. The potential of Moringa oleifera for agricultural and industrial uses. In Proceedings of the International Workshop What development potential for moringa products, Dar-es-Salaam, Tanzania, 29 October–2 November 2001; pp. 47–67. [Google Scholar]
- Abbassy, M.M.S.; Salem, M.Z.M.; Rashad, N.M.; Afify, S.M.; Salem, A.Z.M. Nutritive and biocidal properties of agroforestry trees of Moringa oleifera Lam., Cassia fistula L., and Ceratonia siliqua L. as non-conventional edible vegetable oils. Agrofor. Syst. 2018. [Google Scholar] [CrossRef]
- Nambiar, V.S.; Mehta, R.; Daniel, M. Polyphenols content of three Indian green leafy vegetables. J. Food Sci. Technol. 2005, 42, 312–315. [Google Scholar]
- El-Baha, A.M.; Abou-Gazia, H.A.; Salem, M.Z.M.; Mohamed, N.H.; Salaheldin, P.A. Initial growth response and chemical composition of Moringa oleifera seedlings to different levels of NPK and biofertilizer at two different soil types. Alex. Sci. Exch. J. 2016, 37, 186–196. [Google Scholar]
- Siddhuraju, P.; Becker, K. Antioxidant properties of various solvent extracts of total phenolic constituents from three different agroclimatic origins of drumstick tree (Moringa oleifera Lam.) leaves. J. Agric. Food Chem. 2003, 51, 2144–2155. [Google Scholar] [CrossRef]
- Nagar, P.K.; Iyer, R.I.; Sircar, P.K. Cytokinins in developing fruits of Moringa pterigosperma Gaertn. Physiol. Plant 2006, 55, 45–50. [Google Scholar] [CrossRef]
- Rady, M.M.; Varma, B.; Howladar, S.M. Common bean (Phaseolus vulgaris L.) seedlings overcome NaCl stress as a result of presoaking in Moringa oleifera leaf extract. Sci. Hortic. 2013, 162, 63–70. [Google Scholar] [CrossRef]
- Semida, W.M.; Rady, M.M. Presoaking application of propolis and maize grain extracts alleviates salinity stress in common bean (Phaseolus vulgaris L.). Sci. Hortic. 2014, 168, 210–217. [Google Scholar] [CrossRef]
- Davey, M.W.; Montagu, M.V.; Inzé, D.; Sanmartin, M.; Kanellis, A.; Smirnoff, N.; Benzie, J.; Strain, J.J.; Favell, D.; Fletcher, J. Plant L-ascorbic acid: Chemistry, function, metabolism, bioavailability and effects of processing. J. Sci. Food Agric. 2000, 80, 825–860. [Google Scholar] [CrossRef]
- Horemans, N.; Foyer, C.H.; Asard, H. Transport and action of ascorbate at the plant plasma membrane. Trends Plant Sci. 2000, 5, 263–267. [Google Scholar] [CrossRef]
- Horemans, N.; Foyer, C.H.; Potters, G.; Asard, H. Ascorbate function and associated transport systems in plants. Plant Physiol. Biochem. 2000, 38, 531–540. [Google Scholar] [CrossRef]
- Beltaji, M.S. Exogenous ascorbic acid (vitamin C) induced anabolic changes for salt tolerance in chick pea (Cicer arietinum L.) plants. Afr. J. Plant Sci. 2008, 2, 118–123. [Google Scholar]
- Taha, A.M. Effect of Concentration and Application Method of Ascorbic Acid, Thiamine and Tryptophan on the Growth of Tuberose Plant. Master’s Thesis, Faculty of Agriculture, Alexandria University, Alexandria, Egypt, 2005. [Google Scholar]
- Talaat, I.M.; Youssef, A.A. The role of the amino acids lysine and ornithine in growth and chemical constituents of basil plants. Egypt. J. Appl. Sci. 2002, 17, 83–95. [Google Scholar]
- Talaat, I.M. Physiological effect of salicylic acid and tryptophan on Pelargonium graveolens L. Egypt. J. Appl. Sci. 2005, 20, 751–760. [Google Scholar]
- Verma, J.; Dubey, N.K. Prospectives of botanical and microbial products as pesticides of tomorrow. Curr. Sci. 1999, 76, 172–179. [Google Scholar]
- EL-Hefny, M.; Mohamed, A.A.; Salem, M.Z.M.; Abd El-Kareem, M.S.M.; Ali, H.M. Chemical composition, antioxidant capacity and antibacterial activity against some potato bacterial pathogens of fruit extracts from Phytolacca dioica and Ziziphus spina-christi grown in Egypt. Sci. Hortic. 2018, 233, 225–232. [Google Scholar] [CrossRef]
- Mohamed, A.A.; Behiry, S.I.; Ali, H.M.; EL-Hefny, M.; Salem, M.Z.M.; Ashmawy, N.A. Phytochemical compounds of branches from P. halepensis oily liquid extract and S. terebinthifolius essential oil and their potential antifungal activity. Processes 2020, 8, 330. [Google Scholar] [CrossRef]
- Singh, A.K.; Meena, M.K.; Bharati, R.C.; Gade, R.M. Effect of sulphur and zinc management on yield, nutrient uptake, changes in soil fertility and economics in rice (Oryza sativa)-lentil (Lens culinaris) cropping system. Indian J. Agric. Sci. 2013, 83, 344–348. [Google Scholar]
- Ou, S.H. Rice Diseases, 2nd ed.; CMI: Kew, UK, 1985. [Google Scholar]
- Muthukumar, A.; Sangeetha, G.; Naveenkumar, R. Antimicrobial activity of essential oil against seed borne fungi of rice (Oryza sativa L.). J. Environ. Biol. 2016, 37, 1429–1436. [Google Scholar]
- Sakr, W.R.A.; El-Sayed, A.A.; Hammouda, A.M.; Saad El Deen, F.S.A. Effect of NPK, Aloe Gel and Moringa Extracts on Geranium Plants. J. Hortic. Sci. Ornam. Plants 2018, 10, 01–16. [Google Scholar]
- Culver, M.; Fanuel, T.; Chiteka, A. Effect of Moringa extract on growth and yield of tomato. Greener J. Agric. Sci. 2012, 2, 207–211. [Google Scholar]
- Gad El-Hak, S.H.; Ahmed, A.M.; Moustafa, Y.M.M. Effect of Foliar Application with Two Antioxidants and Humic Acid on Growth, Yield and Yield Components of Peas (Pisum sativum L.). J. Hortic. Sci. Ornam. Plants 2012, 4, 318–328. [Google Scholar]
- AbdelKader, M.A.I.; Hamad, E.H.A. Response of growth, yield and chemical constituents of Roselle plant to foliar application of Ascorbic Acid and Salicylic Acid. Glob. J. Agric. Food Saf. Sci. 2014, 1, 126–136. [Google Scholar]
- Ali, A.F.; Hassan, E.A.; Hamad, E.H.; Abo-Quta, W.M.H. Effect of Compost, Ascorbic Acid and Salicylic Acid Treatments on Growth, Yield and Oil Production of Fennel Plant. Assiut J. Agric. Sci. 2017, 48, 139–154. [Google Scholar]
- Khalil, S.E.; Abd El- Aziz, N.G.; Abou Leila, B.H. Effect of Water Stress, Ascorbic Acid and Spraying Time on Some Morphological and Biochemical Composition of Ocimum basilicum plant. J. Am. Sci. 2010, 6, 33–44. [Google Scholar]
- Radwanski, E.R.; Last, R.L. Tryptophan Biosynthesis and metabolism: Biochemical and Molecular Genetics. Plant Cell 1995, 7, 921–934. [Google Scholar]
- Ertani, A.; Schiavon, M.; Nardi, S. Transcriptome-wide identification of differentially expressed genes in Solanum Lycopersicon, L. in response to an alfalfa-protein hydrolysate using microarrays. Front. Plant Sci. 2017, 8, 1159. [Google Scholar] [CrossRef] [PubMed]
- Colla, G.; Rouphael, Y.; Canaguier, R.; Svecova, E.; Cardarelli, M. Biostimulant action of a plant-derived protein hydrolysate produced through enzymatic hydrolysis. Front. Plant Sci. 2014, 5, 448. [Google Scholar] [CrossRef] [PubMed]
- Zaki, S.S.; Rady, M.M. Moringa oleifera leaf extract improves growth, physiochemical attributes, antioxidant defense system and yields of salt-stressed Phaseolus vulgaris L. plants. Int. J. ChemTech Res. 2015, 8, 120–134. [Google Scholar]
- Gharib, F.A.; Moussa, L.A.; Massoud, O.N. Effect of Compost and Bio-fertilizers on Growth, Yield and Essential Oil of Sweet Marjoram (Majorana hortensis) Plant. Int. J. Agric. Biol. 2008, 10, 381–387. [Google Scholar]
- Lawrence, B.M. Progress in essential oils. Perfum. Flavorist 2011, 36, 56–59. [Google Scholar]
- Baratta, T.M.; Dorman, D.H.J.; Deans, S.G.; Figueiredo, C.A.; Barroso, J.G.; Ruberto, G. Antimicrobial and antioxidant properties of some commercial essential oils. Flavour Fragr. J. 1998, 13, 235–244. [Google Scholar] [CrossRef]
- Elansary, H.O. Chemical Diversity and Antioxidant Capacity of Essential Oils of Marjoram in Northwest Egypt. J. Essent. Oil Bear. Plants 2015, 18, 917–924. [Google Scholar] [CrossRef]
- Gharib, F.A.; Badr, S.E.A.; Al-Ghazali, B.A.S.; Zahran, M.K. Chemical Composition, Antioxidant and Antibacterial Activities of Lavender and Marjoram Essential Oils. Egypt. J. Chem. 2013, 56, 1–24. [Google Scholar]
- Traber, M.G.; Stevens, J.F. Vitamins C and E: Beneficial effects from a mechanistic perspective. Free Radic. Biol. Med. 2011, 51, 1000–1013. [Google Scholar] [CrossRef]
- Mazid, M.; Khan, T.A.; Khan, Z.H.; Quddusi, S.; Mohammad, F. Occurrence, biosynthesis and potentialities of ascorbic acid in plants. Inter. J. Plant Anim. Environ. Sci. 2011, 1, 167–184. [Google Scholar]
- Bartel, B. Auxin biosynthesis. Ann. Rev. Plant Physiol. Plant Mol. Biol. 1997, 48, 51–66. [Google Scholar] [CrossRef] [PubMed]
- Vogt, T. Phenylpropanoid biosynthesis. Mol. Plant 2010, 3, 2–20. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.A.; Gomaa, F.H. Molecular characterization and biological control of some rice seed-borne fungal pathogens. J. Phytopathol. Pest Manag. 2019, 6, 40–53. [Google Scholar]
- Sivanesan, A. Graminicolous Species of Bipolaris, Curvularia, Drechslera, Exserohilum and Their Teleomorphs; CABI: Wallingford, UK, 1987; pp. 1–261. [Google Scholar]
- Indira, S.; Xu, X.; Iamsupasit, N.; Shetty, H.S.; Vasanthi, N.S.; Singh, S.D.; Bandyopadhyay, R. Diseases of sorghum and pearl millet in Asia. In Sorghum and Millet Diseases; Leslie, J.F., Ed.; Blackwell Publishing: Hoboken, NJ, USA, 2002; pp. 393–402. [Google Scholar]
- Perczak, A.; Gwiazdowska, D.; Marchwińska, K.; Juś, K.; Gwiazdowski, R.; Waśkiewicz, A. Antifungal activity of selected essential oils against Fusarium culmorum and F. graminearum and their secondary metabolites in wheat seeds. Arch. Microbiol. 2019, 201, 1085–1097. [Google Scholar] [CrossRef]
- Leeja, L.; Thoppil, J.E. Antimicrobial activity of methanol extract of Origanum majorana L. (Sweet marjoram). J. Environ. Biol. 2007, 28, 145. [Google Scholar]
- Aoudou, Y.; Léopold, T.N.; Michel, J.D.P.; Xavier, E.F.; Moses, M.C. Antifungal properties of essential oils and some constituents to reduce foodborne pathogen. J. Yeast Fungal Res. 2010, 1, 1–8. [Google Scholar]
- Campaniello, D.; Corbo, M.R.; Sinigaglia, M. Activity of eugenol against Penicillium, Aspergillus and Fusarium species. J. Food Prot. 2010, 73, 1124–1128. [Google Scholar] [CrossRef]
- Morcia, C.; Malanati, M.; Terzi, V. In vitro activity of terpinen-4-ol, eugenol, carvone, 1,8-cineole (eucalyptol) and thymol against mycotoxigenic plant pathogens. Food Addit. Contam. 2012, 29, 415–422. [Google Scholar]
- Sikkema, J.; De Bont, J.A.; Poolman, B. Interactions of cyclic hydrocarbons with biological membranes. J. Biol. Chem. 1994, 269, 8022–8028. [Google Scholar]
- Khan, T.Z.; Gill, M.A.; Khan, M.G. Seed borne fungi of rice from Central Punjab and their control. Pak. J. Phytopathol. 2000, 12, 12–14. [Google Scholar]
- Javaid, M.S.; Wahid, A.; Idrees, M.; Gill, M.A.; Saleem, A. Seed mycoflora studies in rice. Pak. J. Phytopathol. 2002, 14, 132–134. [Google Scholar]
- Neergaard, P. Seed pathology of rice disease problems. In Proceedings of the International Symposium on Plant Pathology, New Delhi, India, 14–18 December 1970; pp. 69–81. [Google Scholar]
- Moghaddam, M.; Mehdizadeh, L. Essential Oil and Antifungal Therapy. In Recent Trends in Antifungal Agents and Antifungal Therapy; Basak, A., Chakraborty, R., Mandal, S.M., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 29–74. [Google Scholar] [CrossRef]
- Jackson, M.L. Soil Chemical Analysis; Prentice Hall, Inc.: Englewood Cliffs, NJ, USA, 1958. [Google Scholar]
- Al_husnan, L.A.; Alkahtani, M.D.F. Impact of Moringa aqueous extract on pathogenic bacteria and fungi in vitro. Ann. Agric. Sci. 2016, 61, 247–250. [Google Scholar] [CrossRef]
- Dhawi, F. Images Are More than Documentation: They Are Research Data. Biomed. J. Sci. Tech. Res. 2018, 2, 2276–2277. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.; Hamilton, J.; Rebers, P.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Abdelsalam, N.R.; Salem, M.Z.M.; Ali, H.M.; Mackled, M.I.; EL-Hefny, M.; Elshikh, M.S.; Hatamleh, A.A. Morphological, biochemical, molecular, and oil toxicity properties of Taxodium trees from different locations. Ind. Crops Prod. 2019, 139, 111515. [Google Scholar] [CrossRef]
- Behiry, S.I.; Nasser, R.A.; Abd El-Kareem, M.S.M.; Ali, H.M.; Salem, M.Z.M. Mass Spectroscopic Analysis, MNDO Quantum Chemical Studies and Antifungal Activity of Essential and Recovered Oil Constituents of Lemon-Scented Gum against Three Common Molds. Processes 2020, 8, 275. [Google Scholar] [CrossRef]
- Salem, M.Z.M.; Mansour, M.M.A.; Elansary, H.O. Evaluation of the effect of inner and outer bark extracts of Sugar Maple (Acer saccharum var. saccharum) in combination with citric acid against the growth of three common molds. J. Wood Chem. Technol. 2019, 39, 136–147. [Google Scholar] [CrossRef]
- Ashmawy, N.A.; Behiry, S.I.; Al-Huqail, A.A.; Ali, H.M.; Salem, M.Z.M. Bioactivity of Selected Phenolic Acids and Hexane Extracts from Bougainvilla spectabilis and Citharexylum spinosum on the Growth of Pectobacterium carotovorum and Dickeya solani Bacteria: An Opportunity to Save the Environment. Processes 2020, 8, 482. [Google Scholar] [CrossRef]
- ISTA. Handbook on Seedling Evaluation, 3rd ed.; International Seed Testing Association: Bassersdorf, Switzerland, 2003. [Google Scholar]
- Mothlagh, M.R.S.; Kavian, B. Characterization of new Bipolaris Spp.: The causal agent of rice brown spot disease in the North of Iran. Int. J. Agric. Biol. 2008, 10, 638–642. [Google Scholar]
- Edel, V.; Steinberg, C.; Gautheron, N.; Recorbet, G.; Alabouvette, C. Genetic diversity of Fusarium oxysporum populations isolated from different soils in France. FEMS Microbiol. Ecol. 2001, 36, 61–71. [Google Scholar] [CrossRef]
- Moore, D.; Robson, G.D.; Trirci, A.P.J. 21ST Century Guidebook to Fungi; Cambridge University Press: Cambridge, UK, 2011; p. 640. [Google Scholar]
- Mansour, M.M.A.; EL-Hefny, M.; Salem, M.Z.M.; Ali, H.M. The Biofungicide Activity of Some Plant Essential Oils for the Cleaner Production of Model Linen Fibers Similar to Those Used in Ancient Egyptian Mummification. Processes 2020, 8, 79. [Google Scholar] [CrossRef]
- El-Mougy, N.S. Effect of some essential oils for limiting early blight (Alternaria solani) development in potato field. J. Plant Prot. Res. 2009, 49, 57–61. [Google Scholar] [CrossRef]
- Rahman, M.; Begum, M.; Alam, M. Screening of Trichoderma isolates as a biological control agent against Ceratocystis paradoxa causing pineapple disease of sugarcane. Mycobiology 2009, 37, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute (CLSI). Reference Method for Broth Dilution Antifungal Susceptibility Testing of Filamentous Fungi; Approved Standard, 2nd ed.; CLSI document M38-A2; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2008. [Google Scholar]
- Gangopadhyay, S.; Kapoor, K.S. Control of Fusarium wilt of okra with seed treatment. Indian J. Mycol. Plant Pathol. 1977, 7, 147–149. [Google Scholar]
- Gopalakrishnan, C.; Kamalakannan, A.; Valluvaparidasan, V. Survey of seed-borne fungi associated with rice seeds in Tamil Nadu, India. Libyan Agric. Res. Cent. J. Int. 2010, 1, 307–309. [Google Scholar]
- Ahmad, L.; Pathak, N.; Zaidi, R.K. Antifungal Potential of Plant Extracts against Seed-borne Fungi Isolated from Barley Seeds (Hordeum vulgare L.). J. Plant Pathol. Microbiol. 2016, 7. [Google Scholar] [CrossRef]
- SAS. User Guide: Statistics (Release 8.02); SAS Institute: Cary, NC, USA, 2001. [Google Scholar]
- Snedecor, G.W.; Cochran, W.G. Statistical Methods Applied to Experiments in Agriculture and Biology, 5th ed.; Iowa State University Press: Ames, IA, USA, 1956. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |
| Treatments | Plant Height (cm) | Plant Diameter (cm) | Branch Number | Leaf Fresh Weight (g) | Leaf Dry Weight (g) | Leaf Area | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | |
| Control | 17.16 d* ± 1.87 | 23.16 b ± 3.95 | 21.33 b ± 0.76 | 26.00 e ± 1.39 | 95.16 b ± 8.54 | 143.50 c ± 14.23 | 29.62 e ± 1.92 | 51.86 c ± 0.23 | 11.55 b ± 4.29 | 12.42 d ± 1.01 | 0.323 f ± 0.030 | 0.453 d ± 0.063 |
| AA 100 mg/L | 20.00 ab ± 1.39 | 25.66 ab ± 2.46 | 23.58 ab ± 0.76 | 29.50 ab ± 0.25 | 150.00 a ± 15.78 | 223.00 a ± 13.61 | 35.77 abc ± 1.45 | 60.68 ab ± 6.73 | 19.11 ab ± 1.82 | 21.20 abc ± 4.83 | 0.431 becd ± 0.114 | 0.713 ab ± 0.135 |
| AA 200 mg/L | 20.66 ab ± 3.12 | 27.08 a ± 3.95 | 23.66 ab ± 2.75 | 30.41 ab ± 0.52 | 150.33 a ± 8.69 | 230.16 a ± 59.25 | 37.56 ab ± 6.29 | 64.12 a ± 2.02 | 19.41 ab ± 6.74 | 21.57 ab ± 7.14 | 0.365 ef ± 0.012 | 0.412 d ± 0.034 |
| AA 300 mg/L | 21.33 a ± 2.24 | 27.08 a ± 3.16 | 24.75 a ± 0.66 | 31.08 a ± 0.62 | 152.33 a ± 9.50 | 232.16 a ± 10.39 | 39.08 a ± 6.29 | 64.37 a ± 2.68 | 20.72 a ± 5.62 | 25.03 a ± 4.79 | 0.733 a ± 0.053 | 0.852 a ± 0.07 |
| Trp 100 mg/L | 19.58 abc ± 4.15 | 25.33 ab ± 2.50 | 22.50 ab ± 1.56 | 27.33 cde ± 0.38 | 139.83 a ± 6.00 | 214.50 ab ± 36.87 | 30.24 de ± 1.34 | 54.20 bc ± 3.25 | 11.97 b ± 4.88 | 13.42 cd ± 1.31 | 0.392 cedf ± 0.025 | 0.458 cd ± 0.051 |
| Trp 200 mg/L | 19.75 abc ± 0.66 | 25.50 ab ± 3.27 | 22.58 ab ± 0.52 | 28.66bcd ± 0.2 | 143.00 a ± 7.54 | 219.00 a ± 38.50 | 30.61 cde ± 1.88 | 54.96 bc ± 8.56 | 12.48 b ± 0.86 | 14.90 bcd ± 5.47 | 0.517 b ± 0.120 | 0.611 bc ± 0.084 |
| Trp 300 mg/L | 19.75 abc ± 2.17 | 25.50 ab ± 1.80 | 23.25 ab ± 4.02 | 28.83 bc ± 1.84 | 145.16 a ± 4.53 | 221.50 a ± 21.21 | 31.12 cde ± 3.00 | 57.09 abc ± 3.18 | 13.41 ab ± 2.55 | 15.56 bcd ± 1.06 | 0.474 cbd ± 0.036 | 0.61 bc ± 0.045 |
| MLE 2.5% | 17.91 cd ± 1.75 | 24.33 b ± 2.02 | 21.91 b ± 1.28 | 26.66 de ± 3.35 | 107.16 b ± 26.27 | 168.16 bc ± 13.32 | 32.54 bcde ± 0.85 | 58.08 abc ± 2.26 | 13.47 ab ± 7.26 | 16.93 bcd ± 5.50 | 0.385 def ± 0.006 | 0.477 cd ± 0.077 |
| MLE 5% | 18.75 bcd ± 2.61 | 24.50 ab ± 2.00 | 22.41 ab ± 1.18 | 26.75 de ± 1.14 | 136.83 a ± 17.32 | 209.16 ab ± 20.93 | 33.77 abcde ± 1.98 | 58.66 abc ± 5.03 | 15.46 ab ± 1.28 | 17.22 abcd ± 7.69 | 0.5 bc ± 0.080 | 0.741 ab ± 0.035 |
| MLE 10% | 19.66 abc ± 1.25 | 24.91 ab ± 3.18 | 22.41 ab ± 0.80 | 27.08 cde ± 0.52 | 138.16 a ± 31.60 | 213.66 ab ± 14.15 | 35.37 abcd ± 0.27 | 59.06 abc ± 3.37 | 16.39 ab ± 5.97 | 20.09 abcd ± 4.27 | 0.497 bc ± 0.0155 | 0.669 b ± 0.190 |
| Treatment | Total Chlorophyll Content (SPAD) | Total Carbohydrate Content (%) | Oil Percentage (%) | |||
|---|---|---|---|---|---|---|
| 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | |
| Control | 44.68 c* ± 10.10 | 47.66 c ± 10.80 | 7.50 f ± 0.45 | 7.66 g ± 0.47 | 0.664 b ± 0.050 | 0.658 abc ± 0.219 |
| AA 100 mg/L | 53.13 abc ± 3.07 | 54.05 bc ± 4.29 | 8.71 c ± 0.46 | 8.86 d ± 0.47 | 0.892 ab ± 0.168 | 0.535 c ± 0.128 |
| AA 200 mg/L | 57.01 abc ± 11.93 | 58.10 abc ± 5.96 | 8.81 c ± 0.45 | 9.04 c ± 0.44 | 0.864 ab ± 0.094 | 0.546 bc ± 0.178 |
| AA 300 mg/L | 57.71 ab ± 2.86 | 58.08 abc ± 3.28 | 8.86 c ± 0.47 | 9.08 c ± 0.45 | 0.982 a ± 0.462 | 0.737 abc ± 0.240 |
| Trp 100 mg/L | 45.33 c ± 14.07 | 51.31 bc ± 8.58 | 8.01 e ± 0.45 | 8.42 e ± 0.45 | 0.726 ab ± 0.199 | 0.683 abc ± 0.148 |
| Trp 200 mg/L | 47.06 bc ± 3.79 | 53.53 bc ± 2.91 | 8.06 e ± 0.45 | 7.98 f ± 0.45 | 0.8 ab ± 0.156 | 0.826 a ± 0.245 |
| Trp 300 mg/L | 52.55 abc ± 1.16 | 53.90 bc ± 3.51 | 8.37 d ± 0.44 | 8.44 e ± 0.40 | 0.748 ab ± 0.054 | 0.822 ab ± 0.148 |
| MLE 2.5% | 56.03 abc ± 5.04 | 60.11 ab ± 4.23 | 9.10 b ± 0.45 | 9.10 c ± 0.45 | 0.977 ab ± 0.218 | 0.799 abc ± 0.017 |
| MLE 5% | 56.28 abc ± 8.14 | 60.68 ab ± 11.48 | 9.73 a ± 0.15 | 9.29 b ± 0.45 | 0.917 ab ± 0.127 | 0.81 abc ± 0.146 |
| MLE 10% | 59.90 a ± 7.20 | 66.36 a ± 8.31 | 9.62 a ± 0.30 | 9.79 a ± 0.15 | 1.065 a ± 0.313 | 0.867 a ± 0.058 |
| Percentage in the Oil (%) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Compound Name | Control (Untreated Plants) | AA (mg/L) | MLE (%) | Trp (mg/L) | ||||||
| 100 | 200 | 300 | 2.5 | 5 | 10 | 100 | 200 | 300 | ||
| α-Pinene | 0.53 (913,910) * | 0.41 (937,921) | 0.61 (916,910) | ND | 0.68 (914,911) | ND | ND | ND | ND | 0.45 (920,914) |
| Sabinene | 6.83 (863,861) | 8.66 (878,876) | 7.82 (877,876) | 5.49 (918,909) | 6.89 (917,911) | 2.76 (937,930) | 3.13 (932,924) | 5.79 (918,917) | 3.53 (929,928) | 6.67 (935,929) |
| α-Terpinene | 0.42 (927,923) | ND | 0.84 (908,907) | 0.33 (923,919) | 3.55 (909,907) | ND | 0.8 (923,919) | 0.7 (921,905) | ND | ND |
| 2-Carene | ND | 2.9 (881,881) | 1.42 (886,885) | 0.73 (869,864) | ND | ND | 0.61 (868,867) | ND | ND | ND |
| Cymene | 4.5 (865,850) | 3.83 (846,809) | 4.55 (864,846) | 3.29 (829,813) | 3.9 (906,888) | 3.53 (914,903) | 3.04 (929,909) | 4.41 (824,815) | 7.2 (893,875) | 17.75 (896,884) |
| γ-Terpinene | 1.56 (930,925) | 4.88 (898,869) | 3.17 (901,875) | 2.53 (934,928) | 7.65 (887,885) | 0.96 (938,930) | 2.84 (931,926) | 2.53 (931,926) | 0.67 (938,930) | 0.32 (881,877) |
| 4-Thujanol (Sabinene hydrate) | 10.51 (885,884) | 11.3 (865,865) | 10.38 (875,874) | 12.18 (866,862) | 24.47 (867,865) | 46.47 (847,845) | 33.28 (873,871) | 37.17 (911,908) | 39.88 (865,862) | 9.13 (925,915) |
| Terpineol (cis-β-(1-Terpinenol)) | 33.17 (814,812) | 26.76 (856,839) | 28.44 (803,787) | 38.06 (820,806) | ND | ND | ND | ND | ND | ND |
| cis-p-2-Menthen-1-ol | 1.29 (874,871) | 1.2 (874,870) | 1.4 (877,873) | 1.09 (876,875) | 1.29 (892,887) | 1.39 (887,835) | 2 (902,896) | 1.68 (882,878) | 1.68 (878,872) | ND |
| Terpinen-4-ol | 22.25 (813,813) | 21.58 (815,815) | 21.73 (824,824) | 17.42 (837,837) | 33.34 (815,815) | 27.65 (870,864) | 32.04 (829,829) | 26.09 (822,822) | 25.9 (819,818) | 38.35 (902,896) |
| cis-Piperitol | 0.23 (847,800) | 0.19 (845,806) | 0.22 (856,814) | 0.12 (839,790) | 0.18 (884,841) | 0.18 (920,893) | 0.27 (897,877) | 0.21 (870,829) | 0.27 (871,811) | 1.82 (858,831) |
| α-Terpineol | 7.78 (824,765) | 6.24 (861,827) | 7.37 (860,814) | 7.18 (847,815) | 8.88 (857,857) | 9.48 (884,883) | 12.58 (842,832) | 10.67 (821,772) | 9.22 (873,873) | 7.91 (933,921) |
| Linalyl acetate | 2.33 (846,845) | ND | 2.41 (819,817) | 2.45 (822,821) | 0.76 (907,903) | 1.86 (933,930) | 1.01 (926,923) | 0.92 (897,895) | 3.72 (862,858) | ND |
| cis-Sabinene hydrate acetate | 2.24 (852,811) | 5.06 (867,780) | 2.09 (870,825) | 1.91 (885,786) | 3.64 (888,789) | 0.33 (925,818) | 1.7 (913,811) | 2.88 (922,877) | 2.03 (888,790) | 0.86 (922,859) |
| 1-Terpinen-4-yl acetate | 0.35 (838,830) | 0.48 (846,838) | 0.54 (822,814) | 0.32 (906,862) | 0.34 (842,833) | 0.27 (867,855) | 0.67 (836,828) | 0.41 (850,841) | 0.55 (838,829) | ND |
| 2,5-Dimethyl-3-hexyne-2,5-diol | 0.62 (796,757) | 0.32 (810,766) | 0.53 (794,758) | 0.19 (807,757) | 0.21 (822,780) | 0.36 (823,786) | 0.15 (819,771) | 0.53 (822,782) | 0.36 (817,779) | ND |
| Caryophyllene | 2.26 (894,850) | 2.71 (812,810) | 3.19 (865,806) | 2.71 (812,809) | 2.1 (899,870) | 1.84 (918,918) | 2.02 (913,903) | 2.01 (891,862) | 1.96 (890,866) | 1.46 (915,913) |
| γ-Elemene | 0.61 (863,860) | 1.16 (840,839) | 0.96 (837,835) | 1.36 (840,839) | 0.82 (857,855) | 0.53 (869,865) | 0.91 (846,844) | 0.83 (860,858) | 0.42 (878,874) | ND |
| (-)-Spathulenol | 0.72 (863,862) | 0.7 (888,873) | 0.75 (859,859) | 0.67 (900,882) | 0.23 (885,883) | 0.75 (921,906) | 0.61 (915,902) | 0.88 (865,864) | 0.84 (906,889) | 4.52 (901,900) |
| Caryophyllene oxide | 0.22 (886,884) | 0.19 (884,882) | 0.21 (904,903) | 0.29 (884,881) | 0.09 (916,912) | 0.43 (935,933) | 0.33 (922,921) | 0.16 (926,924) | 0.41 (916,915) | 0.29 (776,762) |
| Monoterpenes (%) | 94.61 | 93.81 | 93.52 | 93.29 | 95.78 | 95.24 | 94.12 | 93.99 | 95.01 | 83.26 |
| Sesquiterpenes (%) | 3.81 | 4.76 | 5.11 | 5.03 | 3.24 | 3.55 | 3.87 | 3.88 | 3.63 | 6.27 |
| Non- oxygenated constituents (%) | 16.71 | 24.55 | 22.56 | 16.44 | 25.59 | 9.62 | 13.35 | 16.27 | 13.78 | 26.65 |
| Oxygenated constituents (%) | 81.71 | 74.02 | 76.07 | 81.88 | 73.43 | 89.17 | 84.64 | 81.60 | 84.86 | 62.88 |
| Control | AA 100 mg/L | AA 200 mg/L | AA 300 mg/L | MLE 2.5% | MLE 5% | MLE 10% | Trp 100 mg/L | Trp 200 mg/L | Trp 300 mg/L | |
|---|---|---|---|---|---|---|---|---|---|---|
| Control | 1 | |||||||||
| AA 100 mg/L | 0.99 | 1 | ||||||||
| AA 200 mg/L | 0.997 | 0.999 | 1 | |||||||
| AA 300 mg/L | 1.000 | 0.991 | 0.995 | 1 | ||||||
| MLE 2.5% | 0.989 | 0.999 | 0.997 | 0.985 | 1 | |||||
| MLE 5% | 0.996 | 0.980 | 0.987 | 0.998 | 0.972 | 1 | ||||
| MLE 10% | 0.999 | 0.987 | 0.993 | 1.000 | 0.980 | 0.999 | 1 | |||
| Trp 100 mg/L | 1.000 | 0.993 | 0.997 | 1.000 | 0.988 | 0.997 | 0.999 | 1 | ||
| Trp 200 mg/L | 0.999 | 0.989 | 0.994 | 1.000 | 0.983 | 0.999 | 1.000 | 1.000 | 1 | |
| Trp 300 mg/L | 0.963 | 0.985 | 0.977 | 0.955 | 0.990 | 0.937 | 0.949 | 0.962 | 0.954 | 1 |
| Isolate Code | Cultivar | Isolated Fungi | Accession Number | Reference |
|---|---|---|---|---|
| F.g.101 | Sakha 101 | Fusarium graminearum | MK450469 | [58] |
| F.v.101 | Fusarium verticilliodies | MK450470 | [58] | |
| B.o.177 | Giza 177 | Bipolaris oryzae | MK450473 | [58] |
| C.l.177 | Curvularia lunata | MK450466 | (This study) |
| Oil from Plant Treated with | Inhibition Percentage % | |||
|---|---|---|---|---|
| F. verticilliodies | F. graminearum | B. oryzae | C. lunata | |
| Control (without oil) | 0.00 f ± 0.00 * | 0.00 e ± 0.00 | 0.00 f ± 0.00 | 0.00 e ± 0.00 |
| Control (oil from untreated plants) | 63.56 e ± 0.77 | 62.87 d ± 0.37 | 45.83 e ± 1.65 | 50.74 d ± 0.74 |
| AA (100 mg/L) | 70.93 cd ± 0.67 | 70.07 b ± 1.00 | 77.65 bcd ± 1.65 | 74.07 b ± 0.97 |
| AA (200 mg/L) | 69.76 b ± 0.67 | 70.07 b ± 1.00 | 80.68 abc ± 1.73 | 81.85 a ± 0.97 |
| AA (300 mg/L) | 69.76 bc ± 0.67 | 68.18 bc ± 0.65 | 82.19 ab ± 3.23 | 81.11 a ± 0.64 |
| Trp (100 mg/L) | 65.50 de ± 1.39 | 67.80 cd ± 4.60 | 75.37 d ± 1.00 | 70.74 c ± 0.97 |
| Trp (200 mg/L) | 67.82 cd ± 1.02 | 82.19 a ± 2.10 | 77.65 bcd ± 1.36 | 72.59 bc ± 1.33 |
| Trp (300 mg/L) | 71.31 b ± 0.38 | 85.60 a ± 0.75 | 79.54 abcd ± 0.65 | 74.81 b ± 1.95 |
| MLE (2.5%) | 65.11 de ± 1.34 | 63.25 cd ± 2.00 | 76.13 cd ± 0.65 | 73.70 bc ± 0.74 |
| MLE (5%) | 94.57 a ± 1.39 | 70.07 b ± 0.37 | 79.54 abcd ± 0.65 | 73.70 bc ± 0.97 |
| MLE (10%) | 92.63 a ± 0.38 | 71.96 b ± 0.37 | 84.46 a ± 3.10 | 75.18 b ± 1.33 |
| Mancozeb (200 mg/L) ** | 53.5 ± 2.45 | 65.15 ± 2.62 | 77.35 ± 2.54 | 63.33 ± 3.33 |
| Oil from Plant Treated with | Minimum Inhibitory Concentration (MIC mg/L) | |||
|---|---|---|---|---|
| F. verticilliodies | F. graminearum | B. oryzae | C. lunata | |
| Control (Oil from untreated plants) | 300 | 300 | 400 | 400 |
| AA (100 mg/L) | 350 | 350 | 250 | 350 |
| AA (200 mg/L) | 350 | 350 | 250 | 250 |
| AA (300 mg/L) | 350 | 350 | 250 | 250 |
| Trp (100 mg/L) | 300 | 350 | 350 | 350 |
| Trp (200 mg/L) | 350 | 250 | 250 | 350 |
| Trp (300 mg/L) | 350 | 250 | 250 | 350 |
| MLE (2.5%) | 300 | 300 | 250 | 350 |
| MLE (5%) | 200 | 350 | 250 | 350 |
| MLE (10%) | 200 | 350 | 250 | 350 |
| Mancozeb(reference fungicide) | 40 | 30 | 20 | 25 |
| Fungi | Seeds without Oil | Oils from Plants Treated with | |||
|---|---|---|---|---|---|
| Untreated Plants | Ascorbic Acid (300 mg/L) | Moringa Extract (10%) | Tryptophan (300 mg/L) | ||
| F. verticilliodies | 28 | 24 | 0.0 | 0.0 | 0.0 |
| F. graminearum | 21 | 18 | 42 | 0.0 | 0.0 |
| B. oryzae | 232 | 32 | 0.0 | 70 | 7.0 |
| C. lunata | 49 | 0.0 | 0.0 | 0.0 | 0.0 |
| Total grains | |||||
| Healthy grains | 70 | 326 | 358 | 330 | 393 |
| Infected grains | 330 | 74 | 42 | 70 | 7 |
| Rice grain infection (%) | 82.5 | 18.5 | 10.5 | 17.5 | 1.75 |
| Season | pH | EC (dSm−1) | Soluble Cations (meq/L) | Soluble Anions (meq/L) | |||||
|---|---|---|---|---|---|---|---|---|---|
| Ca++ | Mg++ | Na+ | K+ | HCO3− | Cl− | SO2−− | |||
| 2018 | 8.08 | 2.53 | 18.20 | 14.20 | 23.91 | 4.49 | 7.20 | 21.00 | 27.10 |
| 2019 | 8.13 | 2.03 | 13.22 | 3.0 | 16.21 | 3.79 | 6.52 | 16.93 | 25.21 |
| Treatment | Concentration | Treatment | Concentration |
|---|---|---|---|
| Control plants | Tap water | Trp | 200 mg/L |
| AA (ascorbic acid) | 100 mg/L | Trp | 300 mg/L |
| AA | 200 mg/L | (moringa leaf extract) MLE | 2.5% |
| AA | 300 mg/L | MLE | 5% |
| Trp (tryptophan) | 100 mg/L | MLE | 10% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohamed, A.A.; El-Hefny, M.; El-Shanhorey, N.A.; Ali, H.M. Foliar Application of Bio-Stimulants Enhancing the Production and the Toxicity of Origanum majorana Essential Oils Against Four Rice Seed-Borne Fungi. Molecules 2020, 25, 2363. https://doi.org/10.3390/molecules25102363
Mohamed AA, El-Hefny M, El-Shanhorey NA, Ali HM. Foliar Application of Bio-Stimulants Enhancing the Production and the Toxicity of Origanum majorana Essential Oils Against Four Rice Seed-Borne Fungi. Molecules. 2020; 25(10):2363. https://doi.org/10.3390/molecules25102363
Chicago/Turabian StyleMohamed, Abeer A., Mervat El-Hefny, Nader A. El-Shanhorey, and Hayssam M. Ali. 2020. "Foliar Application of Bio-Stimulants Enhancing the Production and the Toxicity of Origanum majorana Essential Oils Against Four Rice Seed-Borne Fungi" Molecules 25, no. 10: 2363. https://doi.org/10.3390/molecules25102363
APA StyleMohamed, A. A., El-Hefny, M., El-Shanhorey, N. A., & Ali, H. M. (2020). Foliar Application of Bio-Stimulants Enhancing the Production and the Toxicity of Origanum majorana Essential Oils Against Four Rice Seed-Borne Fungi. Molecules, 25(10), 2363. https://doi.org/10.3390/molecules25102363







