Simvastatin Attenuates H2O2-Induced Endothelial Cell Dysfunction by Reducing Endoplasmic Reticulum Stress
Abstract
1. Introduction
2. Results
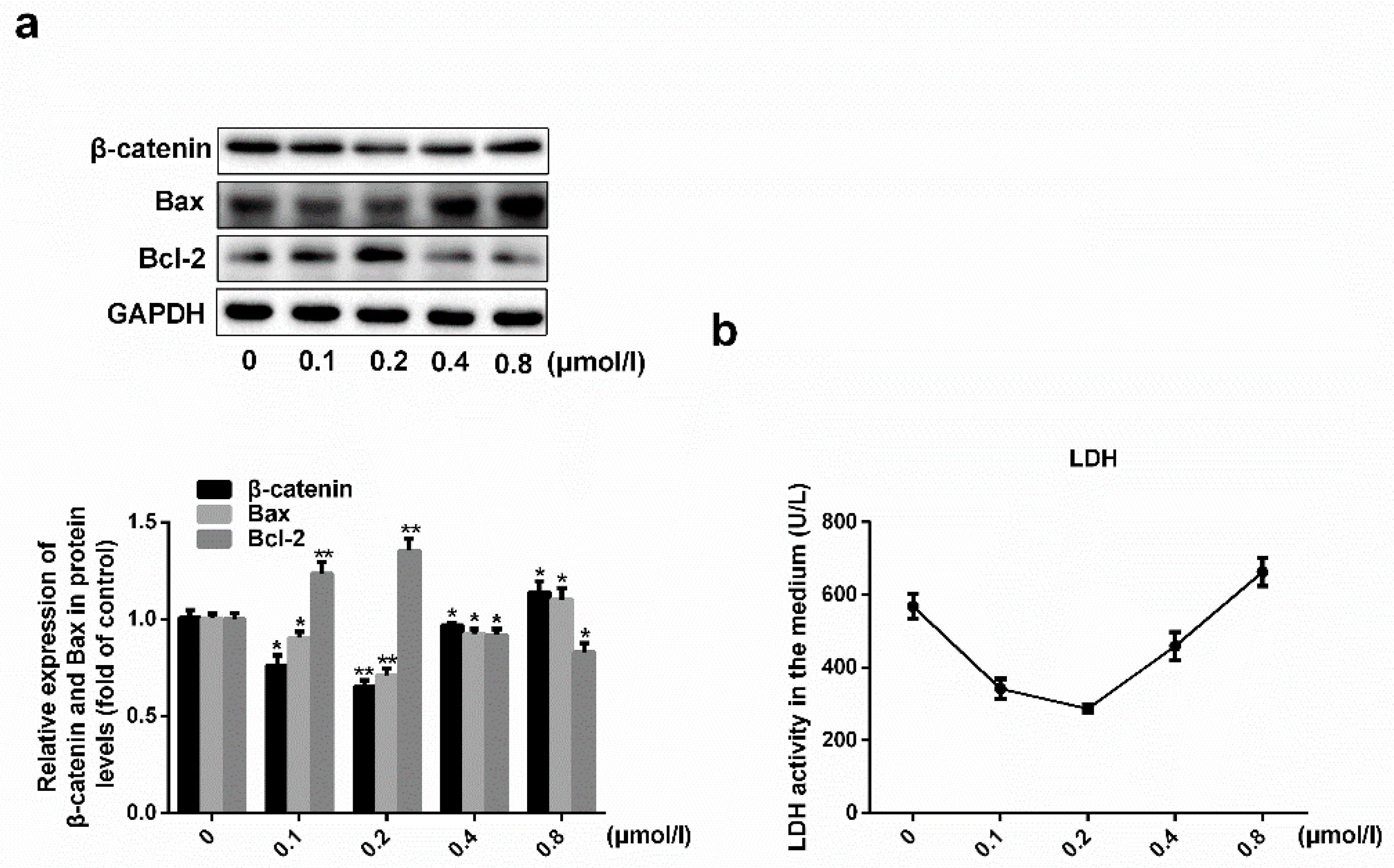
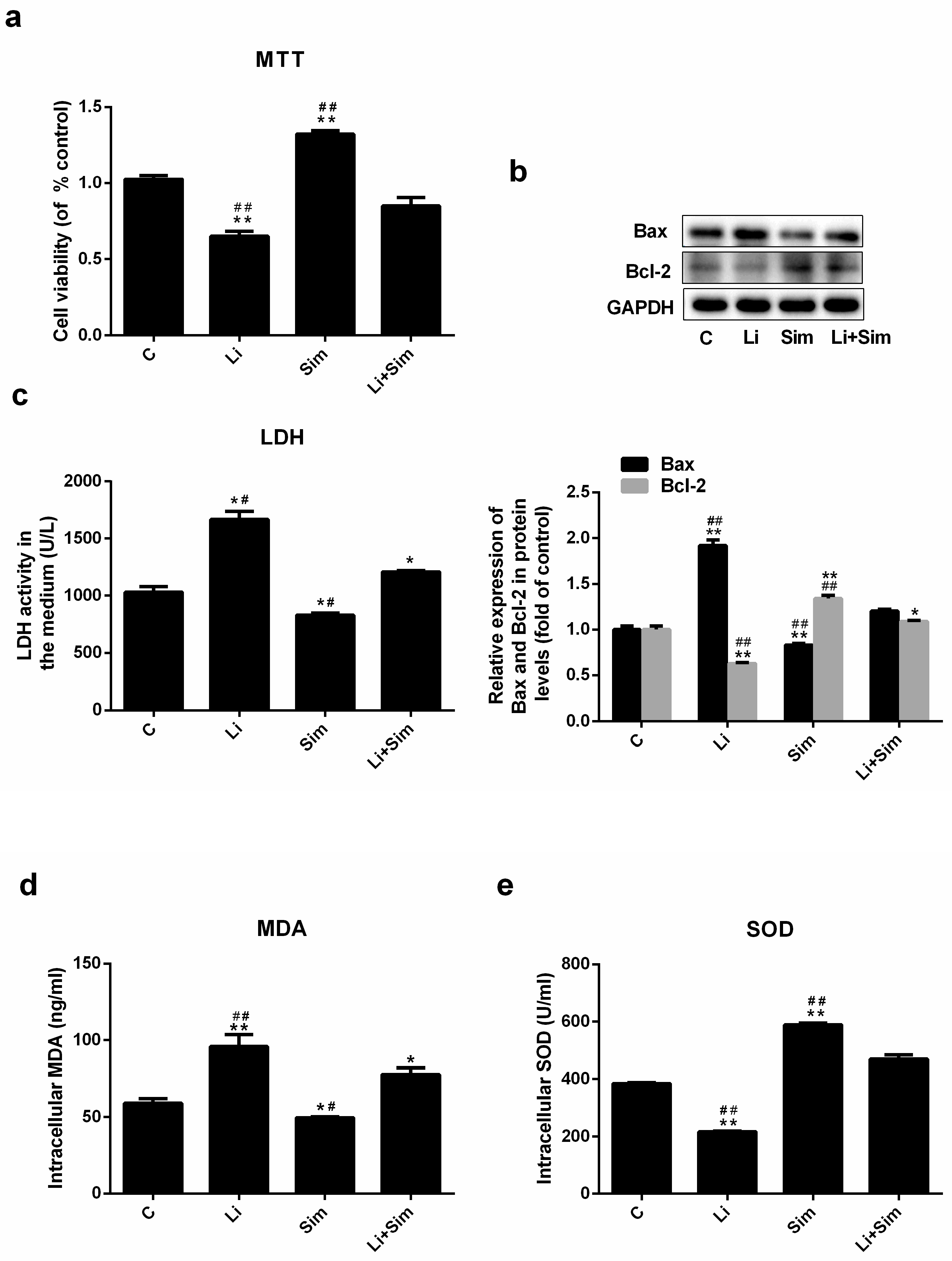
2.1. Effects of Simvastatin on the Viability of HUVECs
2.2. Simvastatin Attenuates Oxidative Stress-Induced Endothelial Cell Dysfunction by Inhibiting the Wnt/β-Catenin Pathway
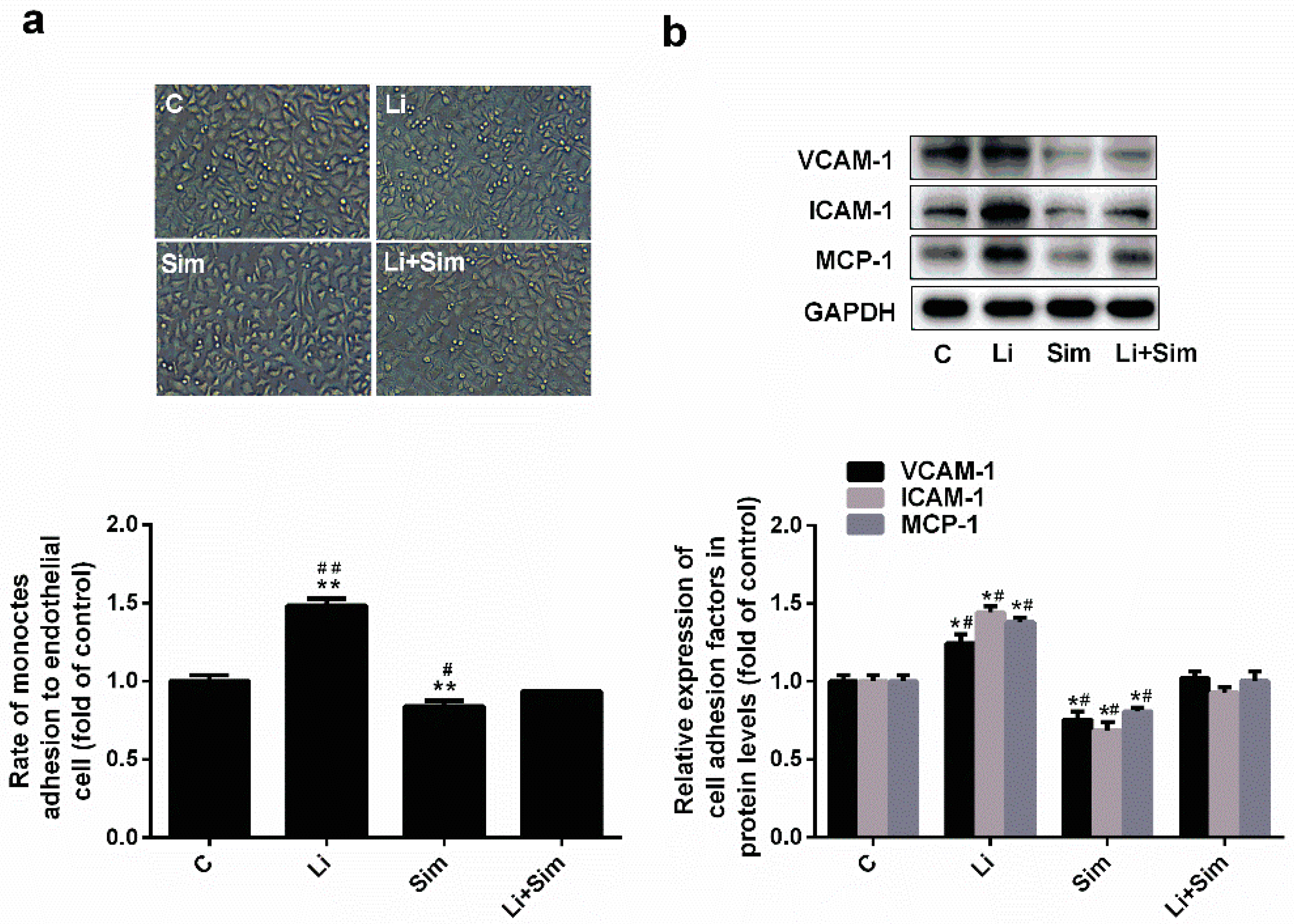
2.3. Simvastatin Reduces Endothelial Cell Adhesion by Inhibiting the Wnt/β-Catenin Pathway
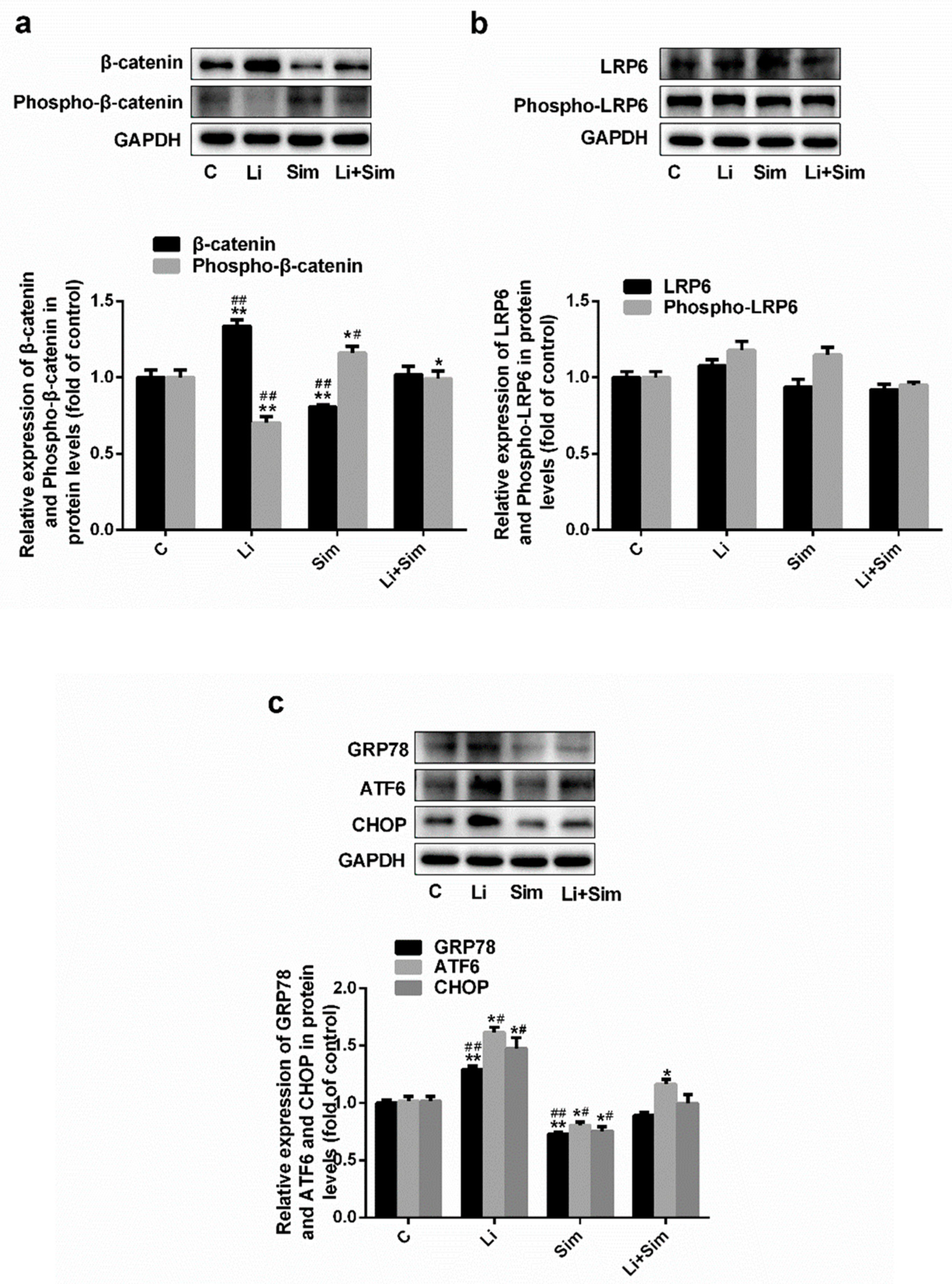
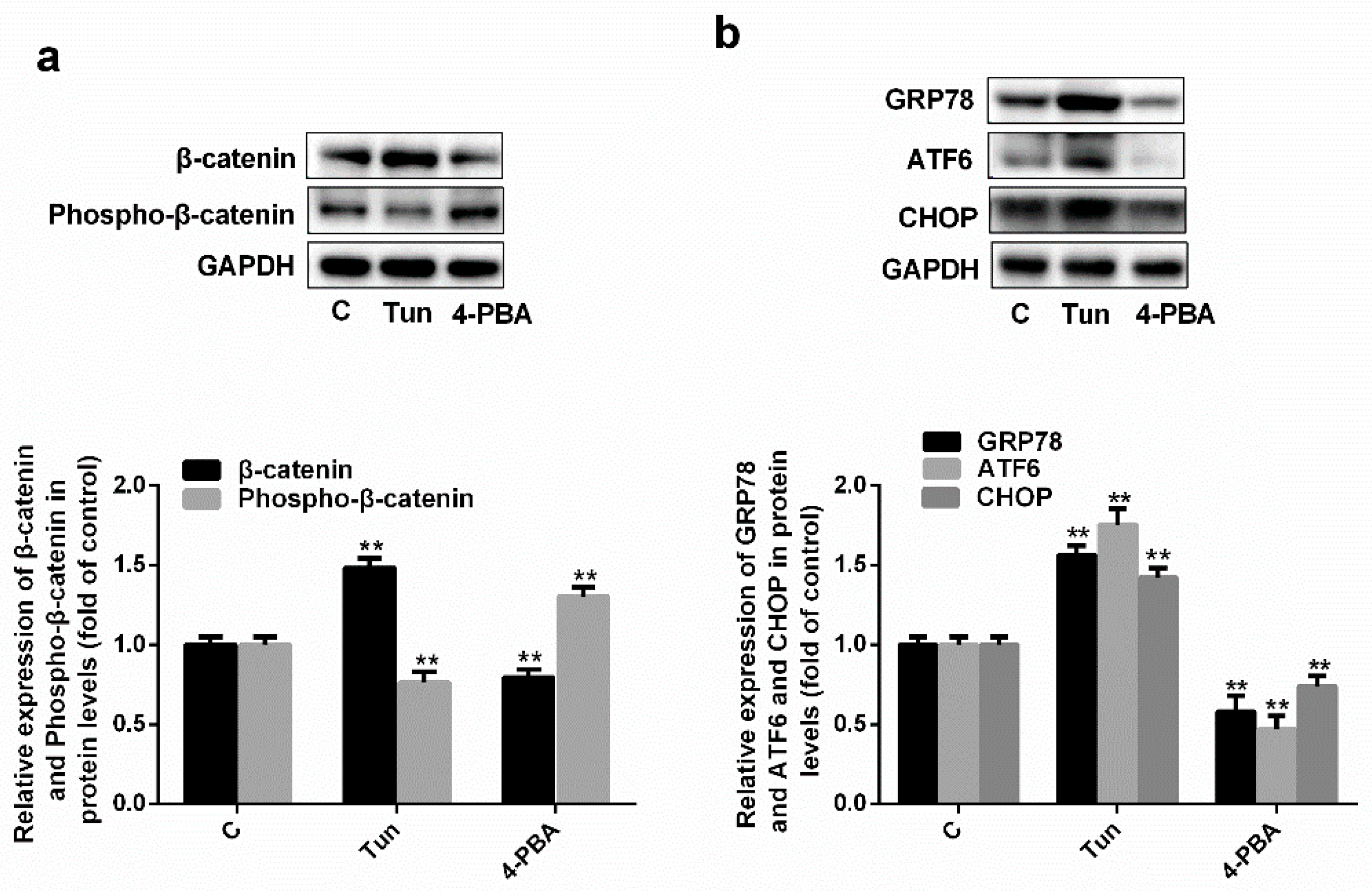
2.4. Simvastatin Inhibits ER Stress via the Wnt/β-Catenin Pathway
2.5. Endoplasmic Reticulum Stress Affects the Wnt/β-Catenin Pathway
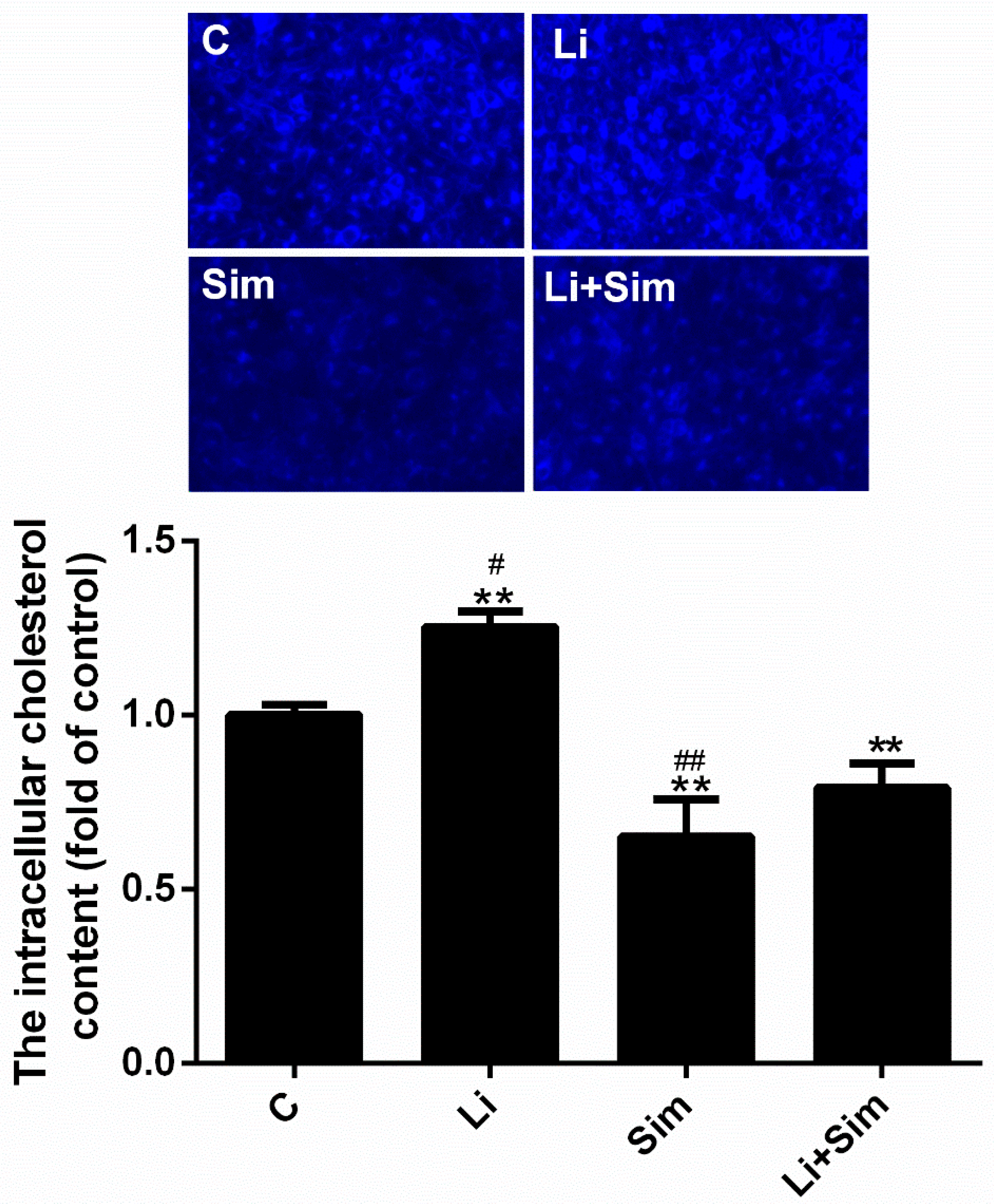
2.6. Simvastatin Reduces the Deposition of Cholesterol in the Cytoplasm
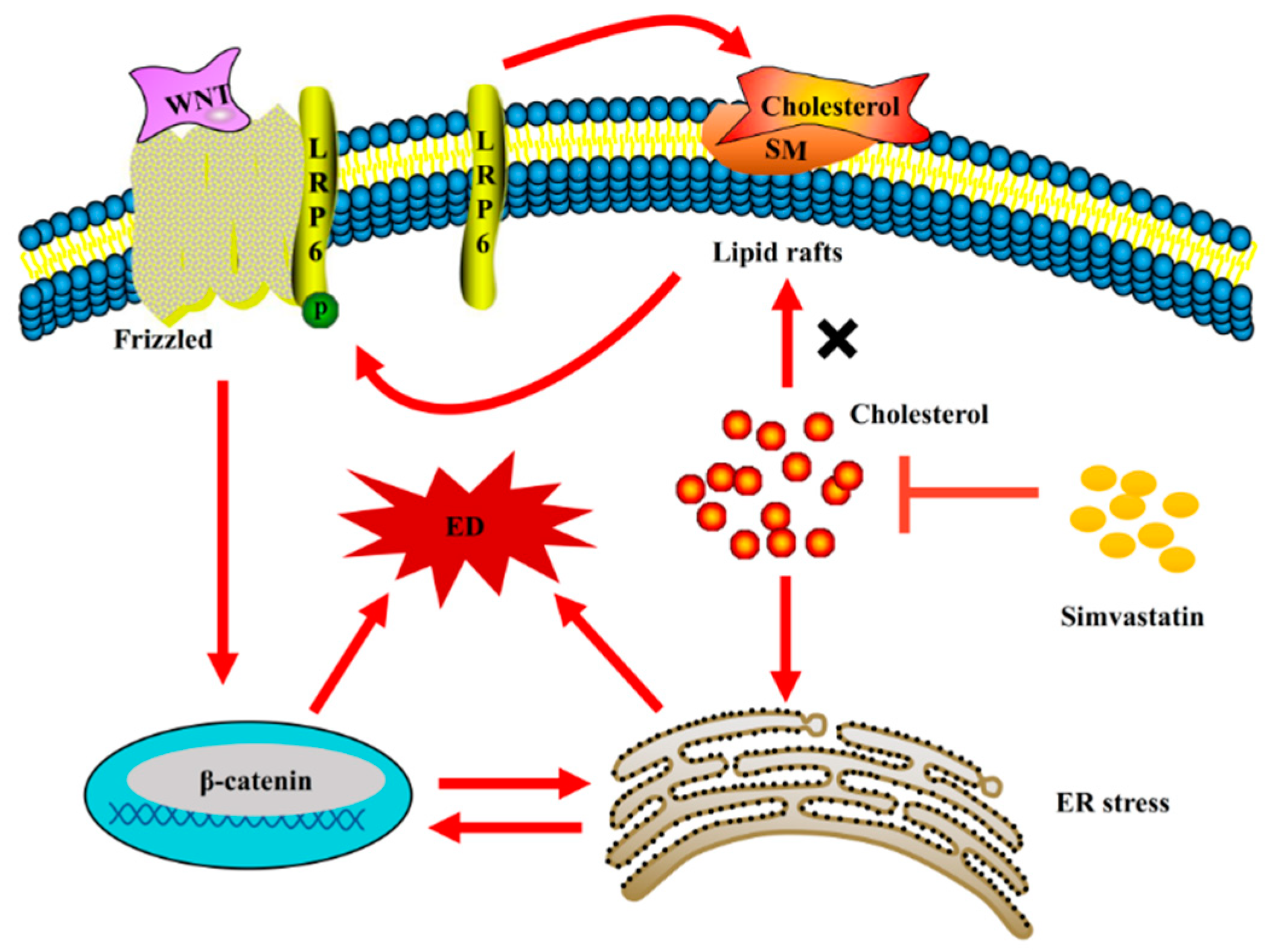
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Treatment
4.2. Grouping and Dosing
4.3. Measurement of LDH Levels in the Medium and Intracellular SOD Activity and MDA Concentration
4.4. Measurement of Adhesion Ability of HUVECs to THP-1 Cells
4.5. Western Blotting Analysis
4.6. Measurement of Cell Viability by MTT Assay
4.7. Filipin Staining
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lüscher, T.F.; Ulf, L.; Arnold, V.E.; Fogelman, A.M. High-density lipoprotein: Vascular protective effects, dysfunction, and potential as therapeutic target. Circ. Res. 2014, 114, 171–182. [Google Scholar] [CrossRef]
- Norbert, W.; Christiane, K.; Ulrich, H.; Joseph, L. Endothelial dysfunction and atherothrombosis in mild hyperhomocysteinemia. Vas. Med. 2012, 7, 227–239. [Google Scholar]
- Kattoor, A.J.; Pothineni, N.V.K.; Palagiri, D.; Mehta, J.L. Oxidative Stress in Atherosclerosis. Curr. Atheroscler. Rep. 2017, 19, 42. [Google Scholar] [CrossRef]
- Assies, J.; Mocking, R.J.T.; Lok, A.; Ruhé, H.G.; Pouwer, F.; Schene, A.H. Effects of oxidative stress on fatty acid- and one-carbon-metabolism in psychiatric and cardiovascular disease comorbidity. Acta Psychiatr. Scand. 2015, 130, 163–180. [Google Scholar] [CrossRef]
- Daverey, A.; Agrawal, S.K. Curcumin alleviates oxidative stress and mitochondrial dysfunction in astrocytes. Neuroscience 2016, 333, 92–103. [Google Scholar] [CrossRef]
- Zhang, G.Q.; Tao, Y.K.; Bai, Y.P.; Yan, S.T.; Zhao, S.P. Inhibitory Effects of Simvastatin on Oxidized Low-Density Lipoprotein-Induced Endoplasmic Reticulum Stress and Apoptosis in Vascular Endothelial Cells. Chin. Med. J. 2018, 131, 950. [Google Scholar] [CrossRef]
- Yin, M.; Liu, Q.; Yu, L.; Yang, Y.; Lu, M.; Wang, H.; Luo, D.; Rong, X.; Tang, F.; Guo, J.V.R. Downregulations of CD36 and Calpain-1, Inflammation, and Atherosclerosis by Simvastatin in Apolipoprotein E Knockout Mice. J. Vasc. Res. 2017, 54, 123–130. [Google Scholar] [CrossRef]
- Kim, K.; Theusch, E.; Kuang, Y.L.; Dose, A.; Mitchel, K.; Cubitt, C.; Chen, Y.I.; Krauss, R.M.; Medina, M.W. ZNF542P is a pseudogene associated with LDL response to simvastatin treatment. Sci. Rep. 2018, 8, 12443. [Google Scholar] [CrossRef]
- Liu, N.; Yang, G.; Hu, M.; Cai, Y.; Hu, Z.; Jia, C.; Zhang, M. Association of ABCC2 polymorphism and gender with high-density lipoprotein cholesterol response to simvastatin. Pharmacogenomics 2018, 19, 1125–1132. [Google Scholar] [CrossRef]
- Santos, L.; Davel, A.P.; Almeida, T.I.R.; Almeida, M.R.; Soares, E.A.; Fernandes, G.J.M.; Magalhães, S.F.; Barauna, V.G.; Garcia, J.A.D.; Santos, L. Soy milk versus simvastatin for preventing atherosclerosis and left ventricle remodeling in LDL receptor knockout mice. Braz. J. Med. Biol. Res. 2017, 50, e5854. [Google Scholar] [CrossRef]
- Shang, S.; Hua, F.; Hu, Z.W. The regulation of β-catenin activity and function in cancer: Therapeutic opportunities. Oncotarget 2017, 8, 33972–33989. [Google Scholar] [CrossRef]
- Foulquier, S.; Daskalopoulos, E.P.; Lluri, G.; Kcm, H.; Deb, A.; Blankesteijn, W.M. WNT Signaling in Cardiac and Vascular Disease. Pharmacol. Rev. 2018, 70, 68–141. [Google Scholar] [CrossRef]
- Zheng, J.; Chen, K.; Wang, H.; Chen, Z.; Xi, Y.; Yin, H.; Lai, K.; Liu, Y. SIRT7 Regulates the Vascular Smooth Muscle Cells Proliferation and Migration via Wnt/β-Catenin Signaling Pathway. Biomed. Res. Int. 2018, 2018, 4769596. [Google Scholar] [CrossRef]
- Popovic, D.S.; Mitrovic, M.; Tomic-Naglic, D.; Icin, T.; Bajkin, I.; Vukovic, B.; Benc, D.; Zivanovic, Z.; Kovacev-Zavisic, B.; Stokic, E. The Wnt/β-catenin Signalling Pathway Inhibitor Sclerostin is a Biomarker for Early Atherosclerosis in Obesity. Curr. Neurovasc. Res. 2017, 14, 200–206. [Google Scholar] [CrossRef]
- Bhatt, P.M.; Ramiro, M. Wnt5a: A player in the pathogenesis of atherosclerosis and other inflammatory disorders. Atherosclerosis 2014, 237, 155–162. [Google Scholar] [CrossRef]
- Bhatt, P.M.; Lewis, C.J.; House, D.L.; Keller, C.M.; Kohn, L.D.; Silver, M.J.; Mccall, K.D.; Goetz, D.J.; Malgor, R. Increased Wnt5a mRNA Expression in Advanced Atherosclerotic Lesions, and Oxidized LDL Treated Human Monocyte-Derived Macrophages. Open Circ. Vasc. J. 2012, 5, 1–7. [Google Scholar] [CrossRef]
- Ma, S.; Yao, S.; Tian, H.; Jiao, P.; Yang, N.; Zhu, P.; Qin, S. Pigment epithelium-derived factor alleviates endothelial injury by inhibiting Wnt/β-catenin pathway. Lipids Health Dis. 2017, 16, 31. [Google Scholar] [CrossRef]
- Zhang, P.; Hua, L.; Hou, H.; Du, X.; He, Z.; Liu, M.; Hu, X.; Yan, N. Sphingomyelin synthase 2 promotes H2O2-induced endothelial dysfunction by activating the Wnt/beta-catenin signaling pathway. International journal of molecular medicine. Int. J. Mol. Med. 2018, 42, 3344–3354. [Google Scholar]
- Ira, T.; David, R. Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress. Nat. Cell Biol. 2011, 13, 184. [Google Scholar]
- Ira, T. The role of endoplasmic reticulum stress in the progression of atherosclerosis. Circ. Res. 2010, 107, 839–850. [Google Scholar]
- Sozen, E.; Karademir, B.; Ozer, N.K. Basic mechanisms in endoplasmic reticulum stress and relation to cardiovascular diseases. Free Radic. Biol. Med. 2015, 78, 30–41. [Google Scholar] [CrossRef] [PubMed]
- Huang, A.; Patel, S.; Mcalpine, C.S.; Werstuck, G.H. The Role of Endoplasmic Reticulum Stress-Glycogen Synthase Kinase-3 Signaling in Atherogenesis. Int. J. Mol. Sci. 2018, 19, 1607. [Google Scholar] [CrossRef] [PubMed]
- Shinozaki, S.; Chiba, T.; Kokame, K.; Miyata, T.; Kaneko, E.; Shimokado, K. A deficiency of Herp, an endoplasmic reticulum stress protein, suppresses atherosclerosis in ApoE knockout mice by attenuating inflammatory responses. PLoS ONE 2013, 8, e75249. [Google Scholar] [CrossRef]
- Carina, H.; Jan, M.; Jenny, D.M.; Ken, M.; Tibor, H.; Heikki, T.; Lakshman, K.P.; Katrin, F.R.; Tibor, H.; Gunnar, S. WNT signaling in activated microglia is proinflammatory. Glia 2011, 59, 119–131. [Google Scholar]
- Amodio, G.; Moltedo, O.; Faraonio, R.; Remondelli, P. Targeting the Endoplasmic Reticulum Unfolded Protein Response to Counteract the Oxidative Stress-Induced Endothelial Dysfunction. Oxid. Med. Cell Longev. 2018, 2018, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Battson, M.L.; Lee, D.M.; Gentile, C.L. Endoplasmic reticulum stress and the development of endothelial dysfunction. Am. J. Physiol. Heart Circ. Physiol. 2017, 312, H355–H367. [Google Scholar] [CrossRef]
- Duvetorp, A.; Olsen, R.S.; Nyström, H.; Skarstedt, M.; Seifert, O. Expression of low-density lipoprotein related receptors 5 and 6 (LRP5/6) in psoriasis skin. Exp. Dermatol. 2017, 26, 1033–1038. [Google Scholar] [CrossRef]
- Simons, K.; Ikonen, E. Functional rafts in cell membranes. Nature 1997, 387, 569–572. [Google Scholar] [CrossRef]
- Li, Y.; Guan, J.; Wang, W.; Hou, C.; Zhou, L.; Ma, J.; Cheng, Y.; Jiao, S.; Zhou, Z. TRAF3-interacting JNK-activating modulator promotes inflammation by stimulating translocation of Toll-like receptor 4 to lipid rafts. J. Biol. Chem. 2019, 294, 2744–2756. [Google Scholar] [CrossRef]
- Chung, C.L.; Wang, S.W.; Sun, W.C.; Shu, C.W.; Kao, Y.C.; Shiao, M.S.; Chen, C.L. Sorafenib suppresses TGF-β responses by inducing caveolae/lipid raft–mediated internalization/degradation of cell-surface type II TGF-β receptors: Implications in development of effective adjunctive therapy for hepatocellular carcinoma. Biochem. Pharmacol. 2018, 154, 39–53. [Google Scholar] [CrossRef] [PubMed]
- Özhan, G.; Sezgin, E.; Wehner, D.; Pfister, A.; Kühl, S.; Kagermeier-Schenk, B.; Kühl, M.; Schwille, P.; Weidinger, G. Lypd6 Enhances Wnt/β-Catenin Signaling by Promoting Lrp6 Phosphorylation in Raft Plasma Membrane Domains. Dev. Cell. 2013, 26, 331–345. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Yang, Y.; Gao, Y.; Wu, X.; Yang, X.; Zhu, Y.; Yang, H.; Wu, L.; Yang, C.; Song, L. Elevated expression of flotillin-1 is associated with lymph node metastasis and poor prognosis in early-stage cervical cancer. Am. J. Cancer Res. 2015, 6, 38. [Google Scholar]
- Haack, F.; Lemcke, H.; Ewald, R.; Rharass, T.; Uhrmacher, A.M. Spatio-temporal model of endogenous ROS and raft-dependent WNT/beta-catenin signaling driving cell fate commitment in human neural progenitor cells. PLoS Comput. Biol. 2015, 11, e1004106. [Google Scholar] [CrossRef] [PubMed]
- Badana, A.K.; Chintala, M.; Gavara, M.M.; Naik, S.; Kumari, S.; Kappala, V.R.; Iska, B.R.; Malla, R.R. Lipid rafts disruption induces apoptosis by attenuating expression of LRP6 and survivin in triple negative breast cancer. Biomed. Pharmacother. 2018, 97, 359–368. [Google Scholar] [CrossRef]
- Cao, L.; Lei, H.; Chang, M.Z.; Liu, Z.Q.; Bie, X.H. Down-regulation of 14-3-3β exerts anti-cancer effects through inducing ER stress in human glioma U87 cells: Involvement of CHOP–Wnt pathway. Biochem. Biophys. Res. Commun. 2015, 462, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Chen, Y.; Zhao, X.; Lv, C.; Yan, J. Oncolytic vaccinia virus inhibits human hepatocellular carcinoma MHCC97-H cell proliferation via endoplasmic reticulum stress, autophagy and Wnt pathways. J. Gene Med. 2016, 18, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wu, S.; Muhammad, S.; Ren, Q.; Sun, C. miR-103/107 promote ER stress mediated apoptosis via targeting the wnt3a/β-catenin /ATF6 pathway in preadipocytes. J. Lipid Res. 2018, 59, 843–853. [Google Scholar] [CrossRef]
- Chaube, R.; Kallakunta, V.M.; Espey, M.G.; Mclarty, R.; Faccenda, A.; Ananvoranich, S.; Mutus, B. Endoplasmic reticulum stress-mediated inhibition of NSMase2 elevates plasma membrane cholesterol and attenuates NO production in endothelial cells. BBA Mol. Cell Biol. Lipids 2012, 1821, 313–323. [Google Scholar] [CrossRef]
- Liu, T.; Duan, W.; Nizigiyimana, P.; Gao, L.; Liao, Z.; Xu, B.; Liu, L.; Lei, M.J.B. Alpha-mangostin attenuates diabetic nephropathy in association with suppression of acid sphingomyelianse and endoplasmic reticulum stress. Biochem. Biophys. Res. 2018, 496, 394–400. [Google Scholar] [CrossRef]
- Röhrl, C.; Stangl, H. Cholesterol metabolism—physiological regulation and pathophysiological deregulation by the endoplasmic reticulum. Wien. Med. Wochenschr. 2018, 168, 280–285. [Google Scholar] [CrossRef]
- Yu, C.; Jiang, F.; Zhang, M.; Luo, D.; Shao, S.; Zhao, J.; Gao, L.; Zuo, C.; Guan, Q. HC diet inhibited testosterone synthesis by activating endoplasmic reticulum stress in testicular Leydig cells. J. Cell Mol. Med. 2019, 23, 3140–3150. [Google Scholar] [CrossRef] [PubMed]
- Sághy, É.; Payrits, M.; Bíró-Sütő, T.; Skoda-Földes, R.; Szánti-Pintér, E.; Erostyák, J.; Makkai, G.; Sétáló, G.J.; Kollár, L.; Kőszegi, T.; Csepregi, R.; et al. Carboxamido steroids inhibit the opening properties of transient receptor potential ion channelsby lipid raft modulation. J. Lipid Res. 2018, 59, 1851–1863. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, Z.; He, X.; Liu, M.; Hua, L.; Wang, T.; Liu, Q.; Chen, L.; Yan, N. Simvastatin Attenuates H2O2-Induced Endothelial Cell Dysfunction by Reducing Endoplasmic Reticulum Stress. Molecules 2019, 24, 1782. https://doi.org/10.3390/molecules24091782
He Z, He X, Liu M, Hua L, Wang T, Liu Q, Chen L, Yan N. Simvastatin Attenuates H2O2-Induced Endothelial Cell Dysfunction by Reducing Endoplasmic Reticulum Stress. Molecules. 2019; 24(9):1782. https://doi.org/10.3390/molecules24091782
Chicago/Turabian StyleHe, Zhiqiang, Xuanhong He, Menghan Liu, Lingyue Hua, Tian Wang, Qian Liu, Lai Chen, and Nianlong Yan. 2019. "Simvastatin Attenuates H2O2-Induced Endothelial Cell Dysfunction by Reducing Endoplasmic Reticulum Stress" Molecules 24, no. 9: 1782. https://doi.org/10.3390/molecules24091782
APA StyleHe, Z., He, X., Liu, M., Hua, L., Wang, T., Liu, Q., Chen, L., & Yan, N. (2019). Simvastatin Attenuates H2O2-Induced Endothelial Cell Dysfunction by Reducing Endoplasmic Reticulum Stress. Molecules, 24(9), 1782. https://doi.org/10.3390/molecules24091782




