Enoxacin and Epigallocatechin Gallate (EGCG) Act Synergistically to Inhibit the Growth of Cervical Cancer Cells in Culture
Abstract
1. Introduction
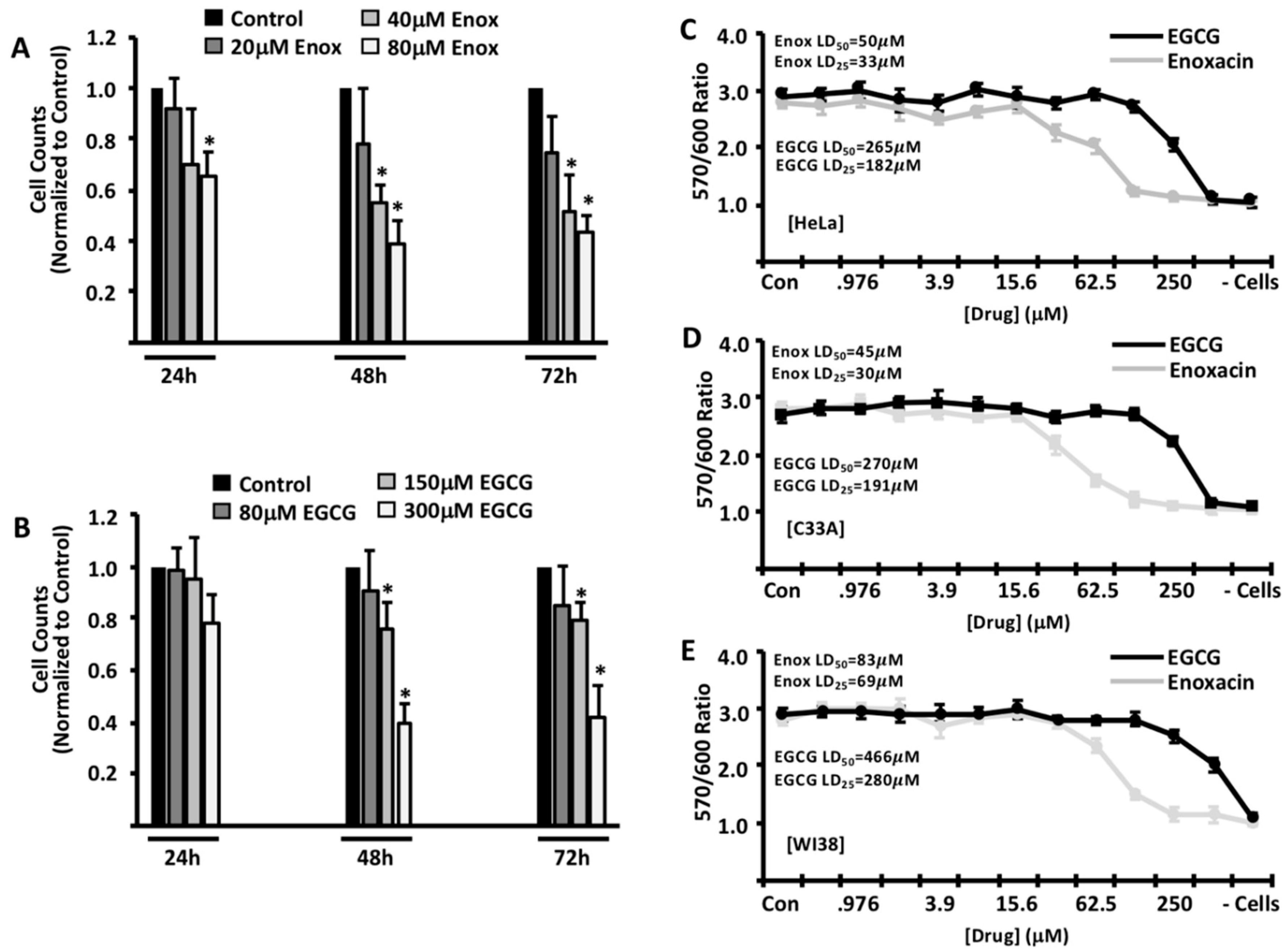
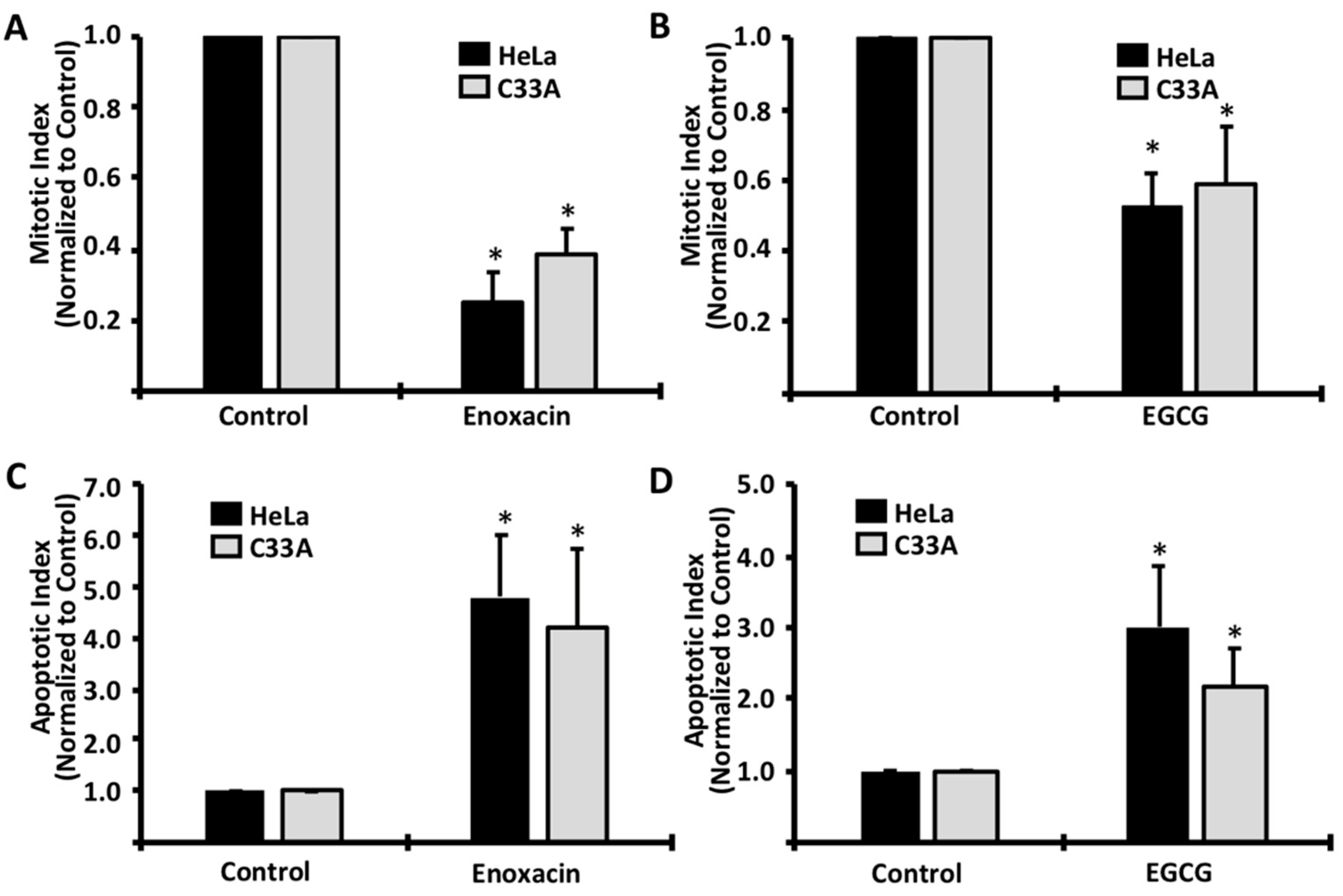
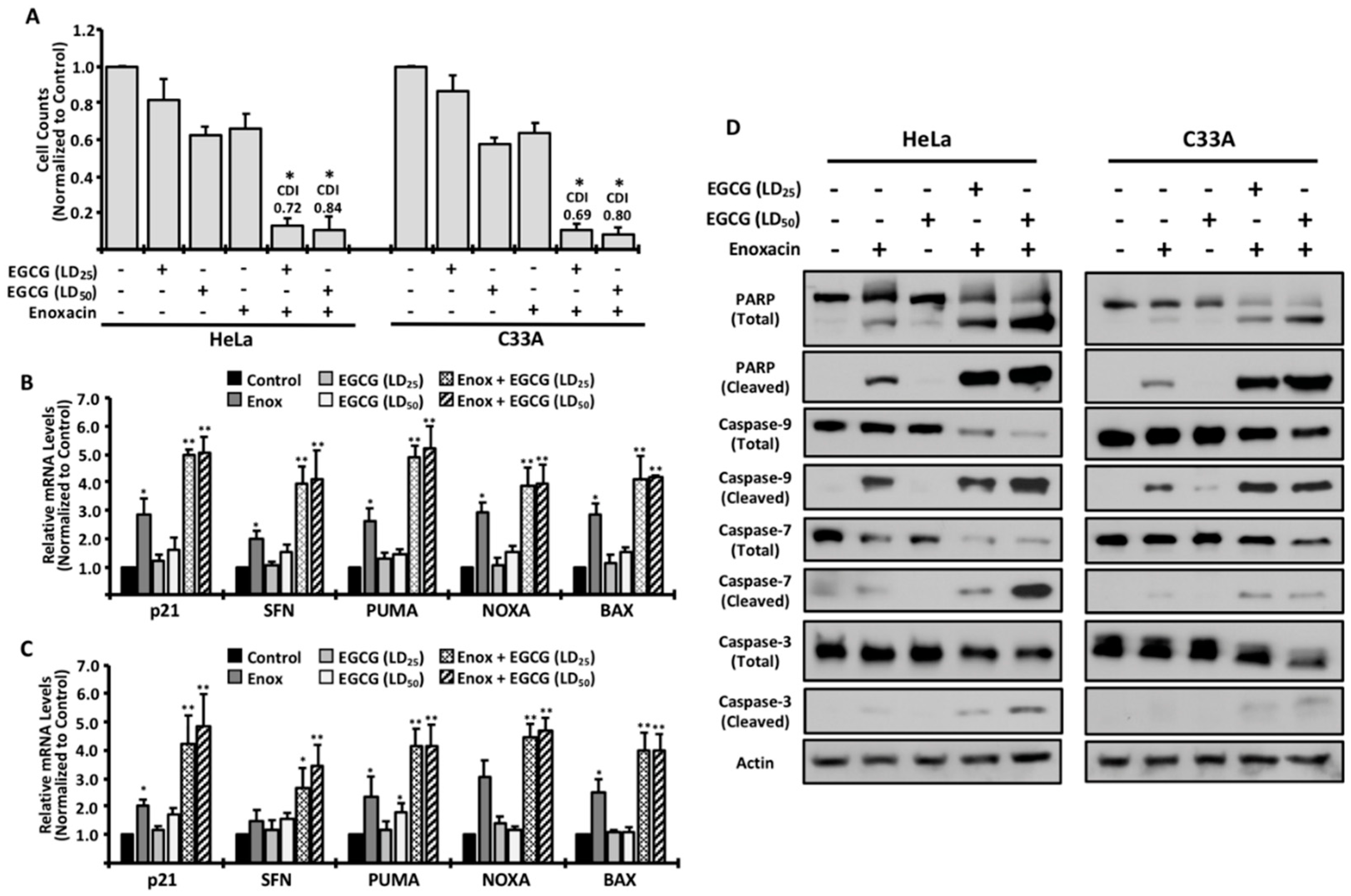
2. Results
3. Discussion
4. Materials and Methods
4.1. Cell lines and Culture Conditions
4.2. Cell Proliferation and Viability Assays
4.3. Mitotic and Apoptotic Analyses
4.4. Gene Expression Analysis
4.5. Analysis of Drug Synergism
4.6. Western Blotting Procedures
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Small, W., Jr.; Bacon, M.A.; Bajaj, A.; Chuang, L.T.; Fisher, B.J.; Harkenrider, M.M.; Jhingran, A.; Kitchener, H.C.; Mileshkin, L.R.; Viswanathan, A.N.; et al. Cervical cancer: A global health crisis. Cancer 2017. [Google Scholar] [CrossRef] [PubMed]
- Senapati, R.; Senapati, N.N.; Dwibedi, B. Molecular mechanisms of HPV mediated neoplastic progression. Infect. Agent Cancer 2016, 11, 59. [Google Scholar] [CrossRef] [PubMed]
- Mayr, C.; Wagner, A.; Neureiter, D.; Pichler, M.; Jakab, M.; Illig, R.; Berr, F.; Kiesslich, T. The green tea catechin epigallocatechin gallate induces cell cycle arrest and shows potential synergism with cisplatin in biliary tract cancer cells. BMC Complement Altern. Med. 2015, 15, 194. [Google Scholar] [CrossRef] [PubMed]
- Lang, M.; Henson, R.; Braconi, C.; Patel, T. Epigallocatechin-gallate modulates chemotherapy-induced apoptosis in human cholangiocarcinoma cells. Liver Int. 2009, 29, 670–677. [Google Scholar] [CrossRef] [PubMed]
- Wise, R.; Andrews, J.M.; Danks, G. In-vitro activity of enoxacin (CL-919), a new quinoline derivative, compared with that of other antimicrobial agents. J. Antimicrob. Chemother. 1984, 13, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Sun, R.; Wang, W.; Meng, X.; Zhang, Y.; Zhang, N.; Yang, S. RNA helicase DHX9 may be a therapeutic target in lung cancer and inhibited by enoxacin. Am. J. Transl. Res. 2017, 9, 674–682. [Google Scholar] [PubMed]
- Mondal, E.R.; Das, S.K.; Mukherjee, P. Comparative evaluation of antiproliferative activity and induction of apoptosis by some fluoroquinolones with a human non-small cell lung cancer cell line in culture. Asian Pac. J. Cancer Prev. 2004, 5, 196–204. [Google Scholar] [PubMed]
- Sousa, E.; Graca, I.; Baptista, T.; Vieira, F.Q.; Palmeira, C.; Henrique, R.; Jeronimo, C. Enoxacin inhibits growth of prostate cancer cells and effectively restores microRNA processing. Epigenetics 2013, 8, 548–558. [Google Scholar] [CrossRef] [PubMed]
- Melo, S.; Villanueva, A.; Moutinho, C.; Davalos, V.; Spizzo, R.; Ivan, C.; Rossi, S.; Setien, F.; Casanovas, O.; Simo-Riudalbas, L.; et al. Small molecule enoxacin is a cancer-specific growth inhibitor that acts by enhancing TAR RNA-binding protein 2-mediated microRNA processing. Proc. Natl. Acad. Sci. USA 2011, 108, 4394–4399. [Google Scholar] [CrossRef] [PubMed]
- Anderson, S.E.; Barton, C.E. The cardiac glycoside convallatoxin inhibits the growth of colorectal cancer cells in a p53-independent manner. Mol. Genet. Metab. Rep. 2017, 13, 42–45. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.Q.; Li, Q.; Xu, S.P.; Shen, Y.X.; Sun, G.Y. Effect of lumiracoxib on proliferation and apoptosis of human nonsmall cell lung cancer cells in vitro. Chin. Med. J. 2008, 121, 602–607. [Google Scholar] [CrossRef] [PubMed]
- Kessler, T.A. Cervical Cancer: Prevention and Early Detection. Semin Oncol Nurs 2017, 33, 172–183. [Google Scholar] [CrossRef] [PubMed]
- Markowitz, L.E.; Liu, G.; Hariri, S.; Steinau, M.; Dunne, E.F.; Unger, E.R. Prevalence of HPV After Introduction of the Vaccination Program in the United States. Pediatrics 2016, 137, e20151968. [Google Scholar] [CrossRef] [PubMed]
- Bhanot, S.K.; Singh, M.; Chatterjee, N.R. The chemical and biological aspects of fluoroquinolones: reality and dreams. Curr. Pharm. Des. 2001, 7, 311–335. [Google Scholar] [CrossRef] [PubMed]
- Scheirer, K.E.; Higgins, N.P. The DNA cleavage reaction of DNA gyrase. Comparison of stable ternary complexes formed with enoxacin and CcdB protein. J. Biol. Chem. 1997, 272, 27202–27209. [Google Scholar] [CrossRef] [PubMed]
- Sinha, B.K. Topoisomerase inhibitors. A review of their therapeutic potential in cancer. Drugs 1995, 49, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.N.; Shankar, S.; Srivastava, R.K. Green tea catechin, epigallocatechin-3-gallate (EGCG): mechanisms, perspectives and clinical applications. Biochem. Pharmacol. 2011, 82, 1807–1821. [Google Scholar] [CrossRef] [PubMed]
- Suganuma, M.; Saha, A.; Fujiki, H. New cancer treatment strategy using combination of green tea catechins and anticancer drugs. Cancer Sci. 2011, 102, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative C(T) method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: EGCG (4143) and Enoxacin (94426) are available for purchase through Sigma-Aldrich. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McDonnell, A.M.; Pyles, H.M.; Diaz-Cruz, E.S.; Barton, C.E. Enoxacin and Epigallocatechin Gallate (EGCG) Act Synergistically to Inhibit the Growth of Cervical Cancer Cells in Culture. Molecules 2019, 24, 1580. https://doi.org/10.3390/molecules24081580
McDonnell AM, Pyles HM, Diaz-Cruz ES, Barton CE. Enoxacin and Epigallocatechin Gallate (EGCG) Act Synergistically to Inhibit the Growth of Cervical Cancer Cells in Culture. Molecules. 2019; 24(8):1580. https://doi.org/10.3390/molecules24081580
Chicago/Turabian StyleMcDonnell, Anna Margaret, Holly M. Pyles, Edgar S. Diaz-Cruz, and Christopher E. Barton. 2019. "Enoxacin and Epigallocatechin Gallate (EGCG) Act Synergistically to Inhibit the Growth of Cervical Cancer Cells in Culture" Molecules 24, no. 8: 1580. https://doi.org/10.3390/molecules24081580
APA StyleMcDonnell, A. M., Pyles, H. M., Diaz-Cruz, E. S., & Barton, C. E. (2019). Enoxacin and Epigallocatechin Gallate (EGCG) Act Synergistically to Inhibit the Growth of Cervical Cancer Cells in Culture. Molecules, 24(8), 1580. https://doi.org/10.3390/molecules24081580





