Investigation of the Biological Activities and Characterization of Bioactive Constituents of Ophiorrhiza rugosa var. prostrata (D.Don) & Mondal Leaves through In Vivo, In Vitro, and In Silico Approaches
Abstract
1. Introduction
2. Results
2.1. GC-MS Analysis
2.2. Acute Toxicity Test
2.3. Qualitative Phytochemical Screening
2.4. Effects of EEOR on Castor Oil-Induced Diarrhea in Mice
2.4.1. Effects of EEOR on Castor Oil-Induced Enteropooling in Mice
2.4.2. Effects of EEOR on Charcoal-Induced Intestinal Transit in Mice
2.5. Effects of EEOR on Histamine-Induced Mouse Paw Edema
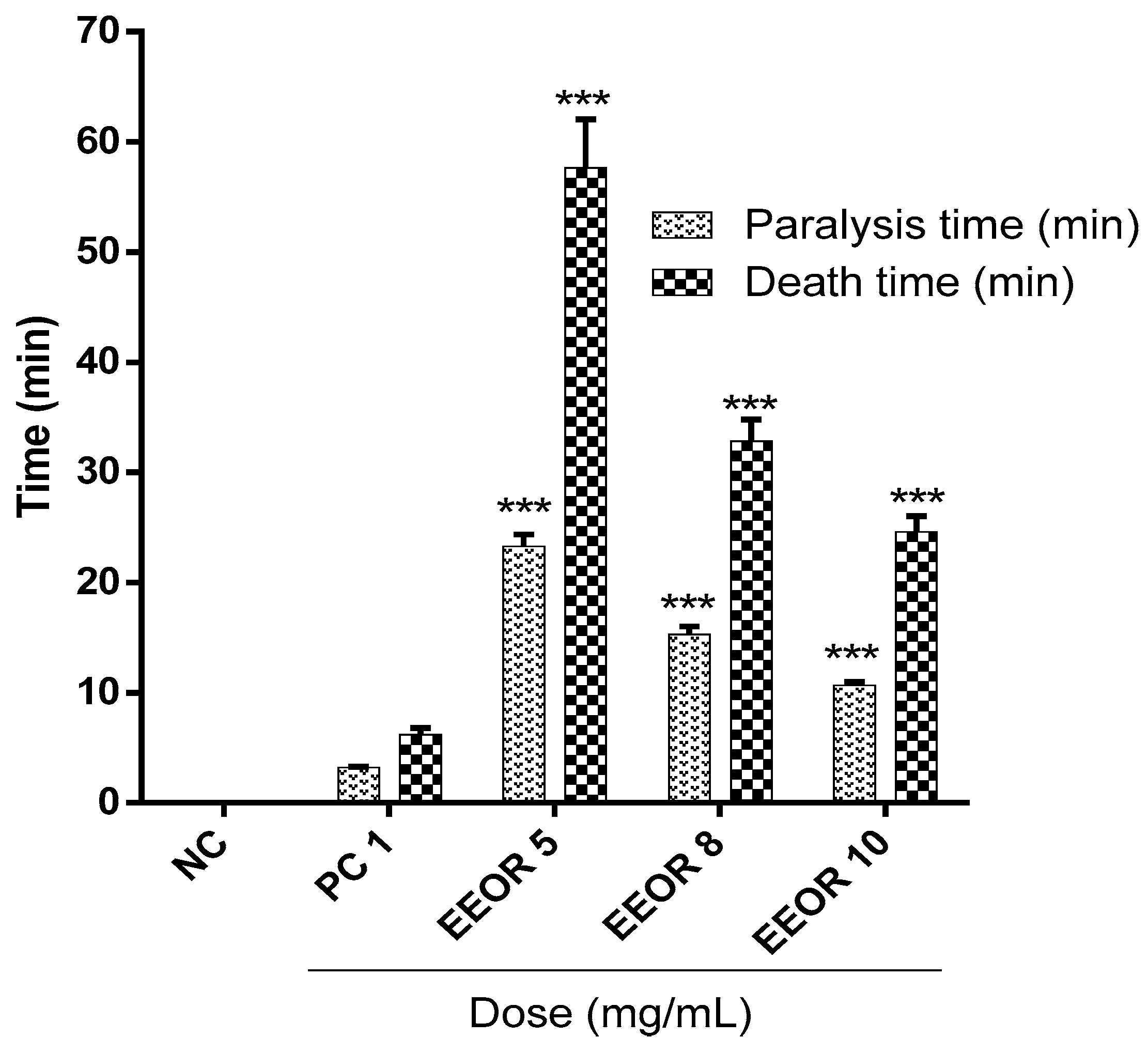
2.6. Anthelmintic Activity
2.7. Antibacterial Activity
2.8. Molecular Docking Study for Antidiarrheal Activity
2.9. Molecular Docking Study for Anthelmintic Activity
2.10. Molecular Docking Study for Antibacterial Activity
2.11. ADME Analysis
2.12. PASS Prediction
3. Discussion
4. Materials and Methods
4.1. Drugs and Chemicals
4.2. Chemical Compounds Studied in This Article
4.3. Plant Collection, Identification, and Extraction
4.4. Animals and Ethical Statements
4.5. GC-MS (Gas Chromatography-Mass Spectroscopy) Analysis of EEOR
4.6. Acute Toxicity Testing of EEOR
4.7. Qualitative Phytochemical Screening of EEOR
4.8. Antidiarrheal Activity Evaluation of EEOR (In Vivo)
4.8.1. Castor Oil-Induced Diarrhea
4.8.2. Castor Oil-Induced Enteropooling
4.8.3. Gastrointestinal Motility
4.9. Histamine-Induced Paw Edema
4.10. Anthelmintic Activity of EEOR (In Vitro)
4.11. Antibacterial Activity of EEOR (In Vitro)
4.12. In silico Molecular Docking
4.12.1. Ligand Preparation
4.12.2. Receptor Preparation
4.12.3. Grid Generation and Molecular Docking
4.13. In Silico ADME Analysis
4.14. In Silico PASS Prediction
5. Statistical Analysis
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| EEOR | Ethanol extract of Ophiorrhiza rugosa leaves |
| p.o. | per oral |
| i.p. | Intraperitoneal |
| ANOVA | Analysis of variance |
| BW | body weight |
| SEM | standard error of mean |
| SPSS | statistical package for social science |
| ADME/T | Absorption, Distribution, Metabolism, Excretion, and Toxicity |
| PASS | Prediction of Activity Spectra for Substances |
References
- Saritha, K.; Rajesh, A.; Manjulatha, K.; Setty, O.H.; Yenugu, S. Mechanism of antibacterial action of the alcoholic extracts of Hemidesmus indicus (L.) R. Br. ex Schult, Leucas aspera (Wild.), Plumbago zeylanica L., and Tridax procumbens (L.) R. Br. ex Schult. Front. Microbiol. 2015, 6, 577. [Google Scholar] [CrossRef] [PubMed]
- Clatworthy, A.E.; Pierson, E.; Hung, D.T. Targeting virulence: A new paradigm for antimicrobial therapy. Nat. Chem. Biol. 2007, 3, 541. [Google Scholar] [CrossRef] [PubMed]
- Perry, B.D.; Randolph, T.F. Improving the assessment of the economic impact of parasitic diseases and of their control in production animals. Vet. Parasitol. 1999, 84, 145–168. [Google Scholar] [CrossRef]
- Vasco, G.; Trueba, G.; Atherton, R.; Calvopina, M.; Cevallos, W.; Andrade, T.; Eguiguren, M.; Eisenberg, J.N.S. Identifying etiological agents causing diarrhea in low income Ecuadorian communities. Am. J. Trop. Med. Hyg. 2014, 91, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. Antimicrobial activity of essential oils and other plant extracts. J. Appl. Microbiol. 1999, 86, 985–990. [Google Scholar] [CrossRef]
- Matthews, H.B.; Lucier, G.W.; Fisher, K.D. Medicinal herbs in the United States: Research needs. Environ. Health Perspect. 1999, 107, 773–778. [Google Scholar] [CrossRef]
- Normile, D. The new face of traditional Chinese medicine. Science 2003, 299, 188–190. [Google Scholar] [CrossRef]
- Wagner, H.; Ulrich-Merzenich, G. Synergy research: Approaching a new generation of phytopharmaceuticals. Phytomedicine 2009, 16, 97–110. [Google Scholar] [CrossRef]
- Coan, K.E.D.; Ottl, J.; Klumpp, M. Non-stoichiometric inhibition in biochemical high-throughput screening. Expert Opin. Drug Discov. 2011, 6, 405–417. [Google Scholar] [CrossRef] [PubMed]
- Sneader, W. Drug Discovery: A History; John Wiley & Sons: Hoboken, NJ, USA, 2005; ISBN 0471899798. [Google Scholar]
- Bangladesh Ethnobotany Online Database. Ophiorrhiza Horrisiana Heyne. Available online: http://www.ebbd.info/ophiorrhiza-horrisiana.html (accessed on 4 December 2018).
- Quattrocchi, U. CRC World Dictionary of Medicinal and Poisonous Plants. Common Names, Scientific Names, Eponyms, Synonyms, and Etymology; CRC Press: Boca Raton, FL, USA, 2012; ISBN 9788578110796. [Google Scholar]
- Gherman, C.; Culea, M.; Cozar, O. Comparative analysis of some active principles of herb plants by GC/MS. Talanta 2000, 53, 253–262. [Google Scholar] [CrossRef]
- Yi, T.; Li, S.-M.; Fan, J.-Y.; Fan, L.-L.; Zhang, Z.-F.; Luo, P.; Zhang, X.-J.; Wang, J.-G.; Zhu, L.; Zhao, Z.-Z. Comparative analysis of EPA and DHA in fish oil nutritional capsules by GC-MS. Lipids Health Dis. 2014, 13, 190. [Google Scholar] [CrossRef]
- Huertas-Pérez, J.F.; Ernest, M.; Badoud, F. Quantification of folpet and phthalimide in tea and herbal infusions by LC-high-resolution MS and GC–MS/MS. Food Addit. Contam. Part A 2019, 36, 109–119. [Google Scholar] [CrossRef]
- Hodges, K.; Gill, R. Infectious diarrhea: Cellular and molecular mechanisms. Gut Microb. 2010, 1, 4–21. [Google Scholar] [CrossRef]
- Petri, W.A.; Miller, M.; Binder, H.J.; Levine, M.M.; Dillingham, R.; Guerrant, R.L. Enteric infections, diarrhea, and their impact on function and development. J. Clin. Investig. 2008, 118, 1277–1290. [Google Scholar] [CrossRef]
- Dubreuil, J. Antibacterial and antidiarrheal activities of plant products against enterotoxinogenic Escherichia coli. Toxins 2013, 5, 2009–2041. [Google Scholar] [CrossRef]
- Tadesse, E.; Engidawork, E.; Nedi, T.; Mengistu, G. Evaluation of the anti-diarrheal activity of the aqueous stem extract of Lantana camara Linn (Verbenaceae) in mice. BMC Complement. Altern. Med. 2017, 17, 190. [Google Scholar] [CrossRef]
- Taylor, P.W. Alternative natural sources for a new generation of antibacterial agents. Int. J. Antimicrob. Agents 2013, 42, 195–201. [Google Scholar] [CrossRef]
- Tangpu, V.; Yadav, A.K. Antidiarrhoeal activity of Rhus javanica ripen fruit extract in albino mice. Fitoterapia 2004, 75, 39–44. [Google Scholar] [CrossRef]
- Bose, S.K.; Dewanjee, S.; Gupta, A.S.; Samanta, K.C.; Kundu, M.; Mandal, S.C. In vivo evaluation of antidiarrhoeal activity of Rhus semialata fruit extract in rats. African J. Tradit. Complement. Altern. Med. 2008, 5, 97–102. [Google Scholar] [CrossRef]
- Boeing, J.S.; Barizão, É.O.; e Silva, B.C.; Montanher, P.F.; de Cinque Almeida, V.; Visentainer, J.V. Evaluation of solvent effect on the extraction of phenolic compounds and antioxidant capacities from the berries: Application of principal component analysis. Chem. Cent. J. 2014, 8, 48. [Google Scholar] [CrossRef]
- Adnan, M.; Chy, M.N.U.; Kamal, A.T.M.M.; Barlow, J.W.; Faruque, M.O.; Yang, X.; Uddin, S.B. Evaluation of anti-nociceptive and anti-inflammatory activities of the methanol extract of Holigarna caustica (Dennst.) Oken leaves. J. Ethnopharmacol. 2019, 236, 401–411. [Google Scholar] [CrossRef]
- Kulkarni, S.R.; Pandit, A.B. Enzymatic hydrolysis of castor oil: An approach for rate enhancement and enzyme economy. Indian J. Biotechnol. 2005, 4, 241–245. [Google Scholar]
- Mathias, J.R.; Martin, J.L.; Burns, T.W.; Carlson, G.M.; Shields, R.P. Ricinoleic acid effect on the electrical activity of the small intestine in rabbits. J. Clin. Investig. 1978, 61, 640–644. [Google Scholar] [CrossRef]
- Tunaru, S.; Althoff, T.F.; Nüsing, R.M.; Diener, M.; Offermanns, S. Castor oil induces laxation and uterus contraction via ricinoleic acid activating prostaglandin EP3 receptors. Proc. Natl. Acad. Sci. USA 2012, 109, 9179–9184. [Google Scholar] [CrossRef]
- Racusen, L.C.; Binder, H.J. Ricinoleic acid stimulation of active anion secretion in colonic mucosa of the rat. J. Clin. Investig. 1979, 63, 743–749. [Google Scholar] [CrossRef]
- Pierce, N.F.; Carpenter, C.C.J.; Elliott, H.L.; Greenough, W.B. Effects of prostaglandins, theophylline, and cholera exotoxin upon transmucosal water and electrolyte movement in the canine jejunum. Gastroenterology 1971, 60, 22–32. [Google Scholar]
- Schiller, L.R.; Santa Ana, C.A.; Morawski, S.G.; Fordtran, J.S. Mechanism of the antidiarrheal effect of loperamide. Gastroenterology 1984, 86, 1475–1480. [Google Scholar]
- Trease, G.E.; Evans, W.C. Pharmacognosy, 13th ed.; Bailliere Tindall Ltd.: London, UK, 1989. [Google Scholar]
- Agbon, A.N.; Kwaneshie, H.O.; Hamman, W.O. Antidiarrheal activity of aqueous fruit extract of Phoenix dactylifera (DATE PALM) in Wistar rats. Br. J. Pharmacol. Toxicol. 2013, 4, 121–127. [Google Scholar] [CrossRef]
- Islam, M.M.; Pia, R.S.; Sifath-E-Jahan, K.; Chowdhury, J.; Akter, F.; Parvin, N.; Akter, S. Antidiarrheal activity of Dillenia indica bark extract. Int. J. Pharm. Sci. Res. 2013, 4, 682. [Google Scholar]
- Awouters, F.; Niemegeers, C.J.E.; Lenaerts, F.M.; Janssen, P.A.J. Delay of castor oil diarrhoea in rats: A new way to evaluate inhibitors of prostaglandin biosynthesis. J. Pharm. Pharmacol. 1978, 30, 41–45. [Google Scholar] [CrossRef]
- Abbas, A.K.; Lichtman, A.H.; Pober, J.S. Cellular and Molecular Immunology; WB Saunders Company: Philadelphia, PA, USA, 1994. [Google Scholar]
- Tamaddonfard, E.; Farshid, A.A.; Hosseini, L. Crocin alleviates the local paw edema induced by histamine in rats. Avicenna J. Phytomed. 2012, 2, 97. [Google Scholar]
- Gilbert, M.A.; Granath, W.O., Jr. Whirling disease of salmonid fish: Life cycle, biology, and disease. J. Parasitol. 2003, 89, 658–667. [Google Scholar] [CrossRef]
- Jamkhande, P.G.; Barde, S.R. Evaluation of anthelmintic activity and in silico PASS assisted prediction of Cordia dichotoma (Forst.) root extract. Anc. Sci. Life 2014, 34, 39–43. [Google Scholar] [CrossRef]
- Patra, J.K.; Baek, K.-H. Antibacterial activity and action mechanism of the essential oil from Enteromorpha linza L. against foodborne pathogenic bacteria. Molecules 2016, 21, 388. [Google Scholar] [CrossRef]
- Khan, R.; Islam, B.; Akram, M.; Shakil, S.; Ahmad, A.A.; Ali, S.M.; Siddiqui, M.; Khan, A.U. Antimicrobial activity of five herbal extracts against multi drug resistant (MDR) strains of bacteria and fungus of clinical origin. Molecules 2009, 14, 586–597. [Google Scholar] [CrossRef]
- Okeke, M.I.; Iroegbu, C.U.; Eze, E.N.; Okoli, A.S.; Esimone, C.O. Evaluation of extracts of the root of Landolphia owerrience for antibacterial activity. J. Ethnopharmacol. 2001, 78, 119–127. [Google Scholar] [CrossRef]
- Akter, K.N.; Karmakar, P.; Das, A.; Anonna, S.N. Evaluation of antibacterial and anthelmintic activities with total phenolic contents of Piper betel leaves. Avicenna J. Phytomed. 2014, 4, 320–329. [Google Scholar]
- Macauder, P.J. Flavonoids affect acetylcholine, prostaglandin E and antigen mediated muscle contration. Prog. Clin. Biol. Res. 1986, 231, 489–492. [Google Scholar]
- Carlo, G.D.; Mascolo, N.; Izzo, A.A.; Capasso, F. Effects of quercetin on the gastrointestinal tract in rats and mice. Phyther. Res. 1994, 8, 42–45. [Google Scholar] [CrossRef]
- Daswani, P.G.; Brijesh, S.; Tatali, P.; Antia, N.H.; Birdi, T.J. Antidiarrhoeal activity of Zingiber officinale (Rosc.). Curr. Sci. 2010, 98, 222–229. [Google Scholar]
- Bhaskar, V.H.; Balakrishnan, N. Analgesic, anti-inflammatory and antipyretic activities of Pergularia daemia and Carissa carandas. Daru 2009, 17, 168–174. [Google Scholar]
- Duke, D. Dr. Duke’s Phytochemical and Ethnobotanical Databases. Available online: https://phytochem.nal.usda.gov/phytochem/search/list (accessed on 17 January 2019).
- Tyagia, T.; Argawak, M. Phytochemical screening and GC-MS analysis of bioactive constituents in the ethanolic extract of Pistia stratiotes L. and Eichhornia crassipes (Mart.) solms. J. Pharmacogn. Phytochem. 2017, 6, 195–206. [Google Scholar]
- Rukshana, M.S.; Doss, A.; Kumari, P.R. Phytochemical screening and GC-MS ANALYSIS of leaf extract of Pergularia daemia (Forssk) Chiov. Asian J. Plant Sci. Res. 2017, 7, 9–15. [Google Scholar]
- Mustapa, A.N.; Martin, Á.; Mato, R.B.; Cocero, M.J. Extraction of phytocompounds from the medicinal plant Clinacanthus nutans Lindau by microwave-assisted extraction and supercritical carbon dioxide extraction. Ind. Crops Prod. 2015, 74, 83–94. [Google Scholar] [CrossRef]
- Khan, S.; Nazir, M.; Raiz, N.; Saleem, M.; Zengin, G.; Fazal, G.; Saleem, H.; Mukhtar, M.; Tousif, M.I.; Tareen, R.B. Phytochemical profiling, in vitro biological properties and in silico studies on Caragana ambigua stocks (Fabaceae): A comprehensive approach. Ind. Crops Prod. 2019, 131, 117–124. [Google Scholar] [CrossRef]
- Duffy, F.J.; Devocelle, M.; Shields, D.C. Computational approaches to developing short cyclic peptide modulators of protein–protein interactions. In Computational Peptidology; Springer: Berlin, Germany, 2015; pp. 241–271. [Google Scholar]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 1997, 23, 3–25. [Google Scholar] [CrossRef]
- Daina, A.; Michielin, O.; Zoete, V. iLOGP: A simple, robust, and efficient description of n-octanol/water partition coefficient for drug design using the GB/SA approach. J. Chem. Inf. Model. 2014, 54, 3284–3301. [Google Scholar] [CrossRef]
- Organisation for Economic Co-operation and Development. Test No. 420: Acute Oral Toxicity—Fixed Dose Procedure; OECD Publishing: Paris, France, 2002; ISBN 9789264070943. [Google Scholar]
- Tiwari, P.; Kumar, B.; Kaur, M.; Kaur, G.; Kaur, H. Phytochemical screening and extraction: A review. Int. Pharm. Sci. 2011, 1, 98–106. [Google Scholar]
- Robert, A.; Nezamis, J.E.; Lancaster, C.; Hanchar, A.J.; Klepper, M.S. Enteropooling assay: A test for diarrhea produced by prostaglandins. Prostaglandins 1976, 11, 809–828. [Google Scholar] [CrossRef]
- Mascolo, N.; Izzo, A.A.; Autore, G.; Barbato, F.; Capasso, F. Nitric oxide and castor oil-induced diarrhea. J. Pharmacol. Exp. Ther. 1994, 268, 291–295. [Google Scholar]
- Singh, S.; Majumdar, D.K.; Rehan, H.M.S. Evaluation of anti-inflammatory potential of fixed oil of Ocimum sanctum (Holybasil) and its possible mechanism of action. J. Ethnopharmacol. 1996, 54, 19–26. [Google Scholar] [CrossRef]
- Ajaiyeoba, E.O.; Onocha, P.A.; Olarenwaju, O.T. In vitro anthelmintic properties of Buchholzia coriaceae and Gynandropsis gynandra extracts. Pharm. Biol. 2001, 39, 217–220. [Google Scholar] [CrossRef]
- Adnan, M.; Chy, M.N.U.; Rudra, S.; Tahamina, A.; Das, R.; Tanim, M.A.H.; Siddique, T.I.; Hoque, A.; Tasnim, S.M.; Paul, A. Evaluation of Bonamia semidigyna (Roxb.) for antioxidant, antibacterial, anthelmintic and cytotoxic properties with the involvement of polyphenols. Orient. Pharm. Exp. Med. 2018, 1–13. [Google Scholar] [CrossRef]
- Bauer, A.W.; Kirby, W.M.M.; Sherris, J.C.; Turck, M. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol. 1966, 45, 493–496. [Google Scholar] [CrossRef]
- Berman, H.M.; Battistuz, T.; Bhat, T.N.; Bluhm, W.F.; Bourne, P.E.; Burkhardt, K.; Feng, Z.; Gilliland, G.L.; Iype, L.; Jain, S. The protein data bank Acta Crystallogr. D Biol. Crystallogr. 2002, 58, 899–907. [Google Scholar] [CrossRef]
- Isupov, M.N.; Obmolova, G.; Butterworth, S.; Badet-Denisot, M.-A.; Badet, B.; Polikarpov, I.; Littlechild, J.A.; Teplyakov, A. Substrate binding is required for assembly of the active conformation of the catalytic site in Ntn amidotransferases: Evidence from the 1.8 Å crystal structure of the glutaminase domain of glucosamine 6-phosphate synthase. Structure 1996, 4, 801–810. [Google Scholar] [CrossRef]
- Ravelli, R.B.G.; Gigant, B.; Curmi, P.A.; Jourdain, I.; Lachkar, S.; Sobel, A.; Knossow, M. Insight into tubulin regulation from a complex with colchicine and a stathmin-like domain. Nature 2004, 428, 198. [Google Scholar] [CrossRef]
- Price, K.L.; Lillestol, R.K.; Ulens, C.; Lummis, S.C.R. Varenicline interactions at the 5-HT3 receptor ligand binding site are revealed by 5-HTBP. ACS Chem. Neurosci. 2015, 6, 1151–1157. [Google Scholar] [CrossRef]
- Thorsen, T.S.; Matt, R.; Weis, W.I.; Kobilka, B.K. Modified T4 lysozyme fusion proteins facilitate G protein-coupled receptor crystallogenesis. Structure 2014, 22, 1657–1664. [Google Scholar] [CrossRef]
- Friesner, R.A.; Murphy, R.B.; Repasky, M.P.; Frye, L.L.; Greenwood, J.R.; Halgren, T.A.; Sanschagrin, P.C.; Mainz, D.T. Extra precision glide: Docking and scoring incorporating a model of hydrophobic enclosure for protein-ligand complexes. J. Med. Chem. 2006, 49, 6177–6196. [Google Scholar] [CrossRef]
- Friesner, R.A.; Banks, J.L.; Murphy, R.B.; Halgren, T.A.; Klicic, J.J.; Mainz, D.T.; Repasky, M.P.; Knoll, E.H.; Shelley, M.; Perry, J.K. Glide: A new approach for rapid, accurate docking and scoring. 1. Method and assessment of docking accuracy. J. Med. Chem. 2004, 47, 1739–1749. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2012, 64, 4–17. [Google Scholar] [CrossRef]
- Swiss ADME. Available online: http://www.swissadme.ch/index.php (accessed on 17 January 2019).
- Way2Drug—Main. Available online: http://www.pharmaexpert.ru/PASSonline/index.php (accessed on 17 January 2019).
- Goel, R.K.; Singh, D.; Lagunin, A.; Poroikov, V. PASS-assisted exploration of new therapeutic potential of natural products. Med. Chem. Res. 2011, 20, 1509–1514. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
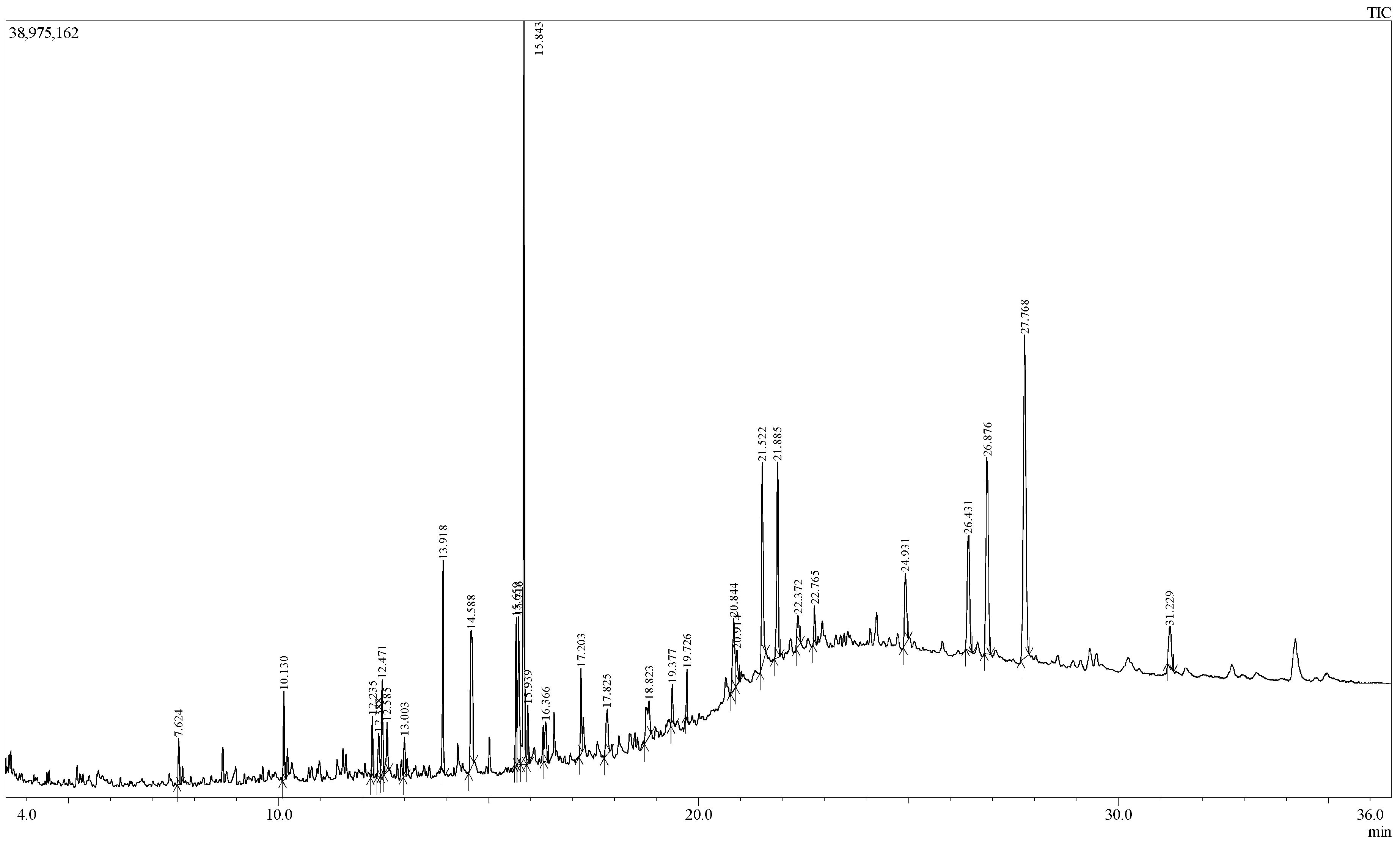
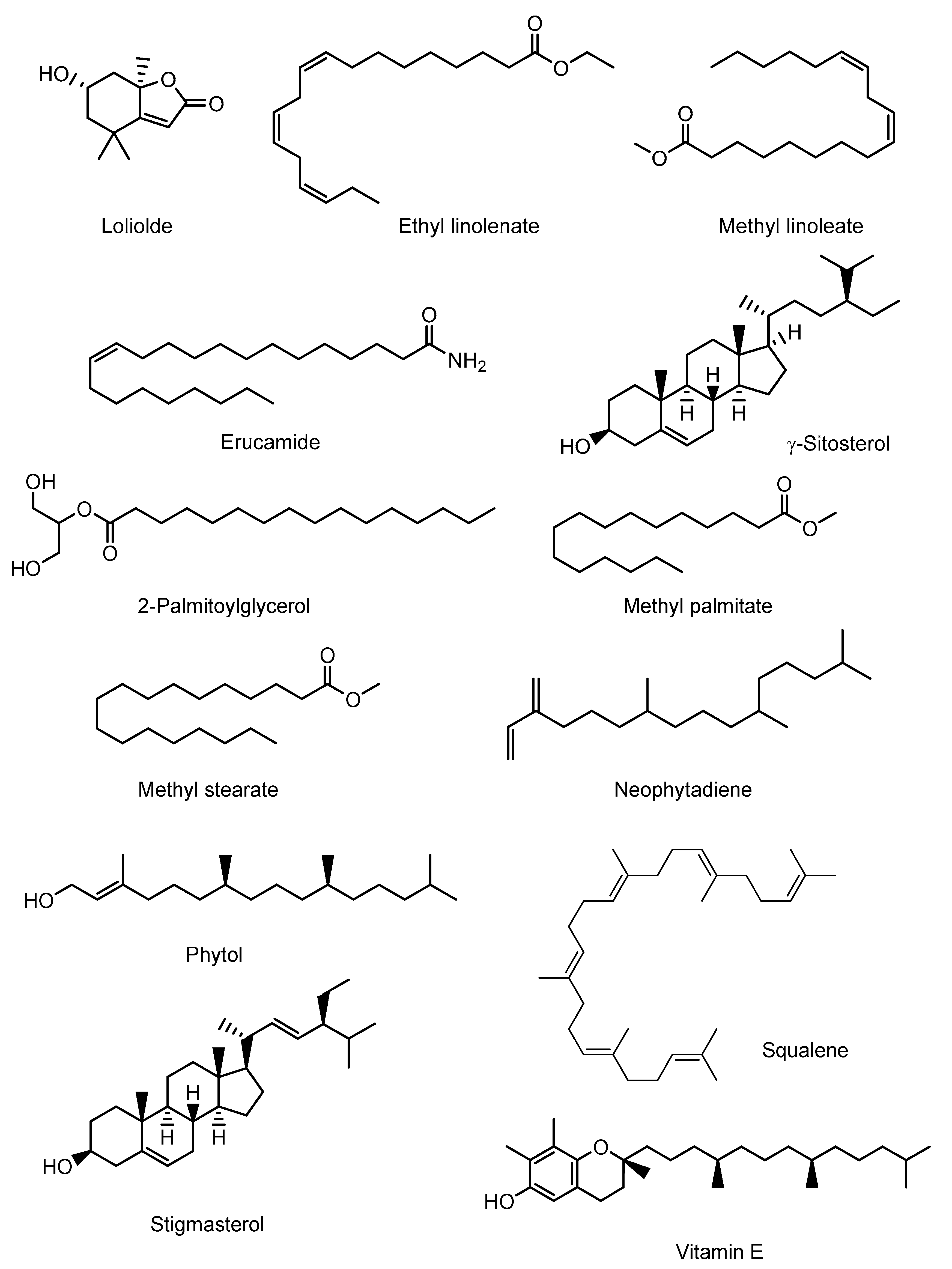
| S.N. | RT (min) | PA (%) | Name of Compound | Molecular Formula |
|---|---|---|---|---|
| 1 | 7.624 | 0.92 | Carbonic acid, hexadecyl methyl ester | C18H36O3 |
| 2 | 10.130 | 1.77 | 1-Nonadecene | C19H38 |
| 3 | 12.235 | 1.29 | Succinic acid, tridec-2-yn-1-yl trans-4-methylcyclohexyl ester | C24H40O4 |
| 4 | 12.388 | 1.10 | 6-Hydroxy-4,4,7α-trimethyl-5,6,7,7α-tetrahydrobenzofuran-2(4H)-one, or Loliolide | C11H16O3 |
| 5 | 12.471 | 1.97 | 1-Nonadecene | C19H38 |
| 6 | 12.585 | 1.38 | 2-Cyclohexen-1-one, 4-hydroxy-3,5,6-trimethyl-4-(3-oxo-1-butenyl)- | C13H18O3 |
| 7 | 13.003 | 0.86 | Neophytadiene | C20H38 |
| 8 | 13.918 | 3.95 | Hexadecanoic acid, methyl ester or Methyl Palmitate | C17H34O2 |
| 9 | 14.588 | 5.47 | 9H-Pyrido[3,4-b]indole, 1-methyl- | C12H10N2 |
| 10 | 15.659 | 2.96 | 9,12-Octadecadienoic acid (Z,Z)-, methyl ester or Methyl linoleate | C19H34O2 |
| 11 | 15.716 | 4.33 | 8,11,14-Docosatrienoic acid, methyl ester | C23H40O2 |
| 12 | 15.843 | 15.50 | Phytol | C20H40O |
| 13 | 15.939 | 1.21 | Methyl stearate | C19H38O2 |
| 14 | 16.366 | 1.17 | 9,12,15-Octadecatrienoic acid, ethyl ester, (Z,Z,Z)- or Ethyl linolenate | C20H34O2 |
| 15 | 17.203 | 1.92 | Octadecanoic acid, 3-hydroxypropyl ester | C21H42O3 |
| 16 | 17.825 | 1.61 | 1-Heptatriacotanol | C37H76O |
| 17 | 18.823 | 2.21 | 6,9-Octadecadienoic acid, methyl ester | C19H34O2 |
| 18 | 19.377 | 0.91 | Hexadecanoic acid, 2-hydroxy-1-(hydroxy- methyl)ethyl ester, or 2-Palmitoylglycerol | C19H38O4 |
| 19 | 19.726 | 0.95 | Diisooctyl phthalate | C24H38O4 |
| 20 | 20.844 | 2.87 | E,E,Z-1,3,12-Nonadecatriene-5,14-diol | C19H34O2 |
| 21 | 20.914 | 1.10 | Ethyl 9,12,15-octadecatrienoate | C20H34O2 |
| 22 | 21.522 | 5.39 | 13-Docosenamide, (Z)- or Erucamide | C22H43NO |
| 23 | 21.885 | 4.83 | Squalene | C30H50 |
| 24 | 22.372 | 1.14 | α-Tocospiro B | C29H50O4 |
| 25 | 22.765 | 0.92 | 1,6,10,14,18,22-Tetracosahexaen-3-ol, 2,6,10,15,19,23-hexamethyl-, (all-E)- | C30H50O |
| 26 | 24.931 | 2.51 | Vitamin E | C29H50O2 |
| 27 | 26.431 | 5.00 | Campesterol | C28H48O |
| 28 | 26.876 | 7.92 | Stigmasterol | C29H48O |
| 29 | 27.768 | 14.94 | γ-Sitosterol | C29H50O |
| 30 | 31.229 | 1.91 | Lup-20(29)-en-3-ol, acetate, (3β)- | C32H52O2 |
| Treatment (mg/kg) | Total Number of Feces | % Inhibition of Defecation | Total Number of Diarrheal Feces | % Inhibition of Diarrhea |
|---|---|---|---|---|
| Control (0.1 mL/mouse) | 14.60 ± 0.74 | 6.40 ± 0.81 | ||
| Loperamide (5) | 5.40 ± 0.24 *** | 63.01 | 2.20 ± 0.20 *** | 65.62 |
| EEOR (100) | 8.00 ± 0.44 ** | 45.20 | 5.00 ± 0.31 *** | 21.87 |
| EEOR (200) | 7.00 ± 0.83 *** | 52.05 | 3.80 ± 0.48 ** | 40.62 |
| EEOR (400) | 5.80 ± 0.20 *** | 60.27 | 2.40 ± 0.24 *** | 62.50 |
| Treatment (mg/kg) | Volume of Intestinal Content (mL) | % Inhibition | Weight of Intestinal Content (gm) | % Inhibition |
|---|---|---|---|---|
| Control (0.1 mL/mouse) | 0.51 ± 0.025 | 0.71±0.022 | ||
| Loperamide (5) | 0.26 ± 0.013 *** | 49.42 | 0.29±0.012 *** | 58.87 |
| EEOR (100) | 0.44 ± 0.014 ** | 13.22 | 0.55±0.030 ** | 22.25 |
| EEOR (200) | 0.40 ± 0.017 *** | 21.78 | 0.44±0.090 ** | 38.02 |
| EEOR (400) | 0.34 ± 0.063 * | 32.29 | 0.35±0.047 *** | 49.57 |
| Treatment (mg/kg) | Total Length of Intestine (cm) | Distance Travelled by Marker (cm) | Peristalsis Index (%) | % Inhibition Relative to Control |
|---|---|---|---|---|
| Control (0.1 mL/mouse) | 48.60 ± 0.51 | 41.40 ± 0.93 | 85.19 ± 1.74 | |
| Loperamide (5) | 49.20 ± 0.58 | 20.80 ± 0.73 | 42.26 ± 1.32 *** | 57.73 |
| EEOR (100) | 44.20 ± 0.37 ** | 29.20 ± 0.58 ** | 66.07 ± 1.32 *** | 33.92 |
| EEOR (200) | 43.00 ± 0.44 ** | 24.80 ± 0.86 *** | 57.69 ± 2.08 *** | 42.30 |
| EEOR (400) | 48.30 ± 0.25 *** | 20.10 ± 1.36 *** | 41.66 ± 3.02 *** | 58.33 |
| Treatment (mg/kg) | Paw Volume (mm) (% Inhibition) | |||
|---|---|---|---|---|
| 1 h | 2 h | 3 h | 4 h | |
| Control (0.1mL/mouse) | 0.454 ± 0.010 | 0.392 ± 0.012 | 0.340 ± 0.007 | 0.312 ± 0.008 |
| Diclofenac-Na (10) | 0.350 ± 0.004 *** (42.42) | 0.290 ± 0.007 *** (60.29) | 0.264 ± 0.010 *** (66.66) | 0.248 ± 0.012 *** (78.57) |
| EEOR (100) | 0.422 ± 0.005 ** (11.11) | 0.358 ± 0.015 *** (17.64) | 0.310 ± 0.010 *** (23.8) | 0.290 ± 0.004 ** (21.42) |
| EEOR (200) | 0.398 ± 0.007 *** (20.20) | 0.334 ± 0.009 *** (30.88) | 0.294 ± 0.006 *** (35.71) | 0.278 ± 0.006 *** (32.14) |
| EEOR (400) | 0.344 ± 0.012 *** (38.38) | 0.300 ± 0.006 *** (42.64) | 0.260 ± 0.005 *** (54.76) | 0.246 ± 0.005 *** (57.14) |
| Bacterial Strain | Name of the Bacteria | Zone of Inhibition (mm) | |||
|---|---|---|---|---|---|
| Concentration (μg/disc) | Kanamycin (30 µg/disc) | ||||
| EEOR 500 | EEOR 800 | EEOR 1000 | |||
| Gram-positive | Staphylococcus aureus (ATCC 6538) | - | - | - | 29.30 ± 0.60 |
| Bacillus subtilis (ATCC 6633) | 7.33 ± 0.57 | 11.70 ± 0.75 | 14.80 ± 0.72 | 32.81 ± 0.67 | |
| Bacillus cereus (ATCC 14579) | - | - | - | 27.50 ± 0.58 | |
| Gram-negative | Salmonella typhi (ATCC 29629) | - | 7.33 ± 0.57 | 12.80 ± 0.34 | 28.218±0.81 |
| Salmonella paratyphi (ATCC 9150) | - | - | - | 30.51 ± 0.50 | |
| Escherichia coli (ATCC 8739) | 8.20 ± 0.72 | 11.26 ± 1.16 | 16.23 ± 0.68 | 31.20 ± 0.82 | |
| Pseudomonas aeruginosa (ATCC 9027) | - | - | - | 26.28 ± 0.36 | |
| Compound Name | Docking Score 1 | |||
|---|---|---|---|---|
| 4U14 | 5AIN | 1SA0 | 1XFF | |
| Loliolide | −6.63 | −5.47 | −4.49 | −4.88 |
| Ethyl linolenate | −6.76 | −3.47 | −5.36 | −3.10 |
| Methyl linoleate | −3.26 | −1.65 | −1.87 | 0.25 |
| Erucamide | − | − | −2.35 | −1.21 |
| γ-Sitosterol | − | − | −7.00 | − |
| 2-Palmitoylglycerol | −3.55 | − | − | −1.16 |
| Methyl palmitate | −2.00 | −0.25 | −1.10 | +1.81 |
| Methyl stearate | − | +1.62 | − | +2.76 |
| Neophytadiene | −2.55 | −0.69 | −0.59 | +1.18 |
| Phytol | −3.62 | −2.08 | −2.30 | −0.12 |
| Squalene | − | − | − | − |
| Stigmasterol | − | − | −7.13 | − |
| Vitamin E | −8.80 | − | −6.65 | − |
| Reference drugs (Loperamide/Levamisole/Kanamycin) | −7.32 | − | −6.26 | −2.73 |
| Compound Name | MW 1 (g/mol) | HB Acceptor 2 | HB Donor 3 | Log Po/w 4 | Molar Refractivity 5 | Rule of Five 6 |
|---|---|---|---|---|---|---|
| Loliolide | 196.24 | 3 | 1 | 1.53 | 52.51 | 0 |
| Ethyl linolenate | 306.48 | 2 | 0 | 5.82 | 98.12 | 1 |
| Methyl linoleate | 297.47 | 2 | 0 | 5.69 | 98.78 | 1 |
| Erucamide | 337.58 | 1 | 1 | 6.77 | 110.30 | 1 |
| γ-Sitosterol | 414.71 | 1 | 1 | 7.19 | 133.1 | 2 |
| 2-Palmitoylglycerol | 330.50 | 4 | 2 | 4.72 | 97.06 | 0 |
| Methyl palmitate | 270.45 | 2 | 0 | 5.54 | 85.12 | 1 |
| Methyl stearate | 298.50 | 2 | 0 | 6.24 | 94.73 | 1 |
| Neophytadiene | 278.52 | 0 | 0 | 7.07 | 97.31 | 1 |
| Phytol | 296.53 | 1 | 1 | 6.22 | 98.94 | 1 |
| Squalene | 410.72 | 0 | 0 | 9.38 | 143.48 | 2 |
| Stigmasterol | 412.69 | 1 | 1 | 6.96 | 132.75 | 2 |
| Vitamin E | 430.71 | 2 | 1 | 8.27 | 139.27 | 2 |
| Compound Name | Biological Properties Predicted by Pass Online | Pa | Pi |
|---|---|---|---|
| Loliolide | Sugar-phosphatase inhibitor | 0.727 | 0.028 |
| Antibacterial | 0.418 | 0.026 | |
| Spasmolytic, urinary | 0.454 | 0.062 | |
| Anti-inflammatory | 0.416 | 0.088 | |
| Antiperistaltic | 0.345 | 0.018 | |
| Antihelmintic | 0.345 | 0.071 | |
| Ethyl linolenate | Lipid metabolism regulator | 0.951 | 0.003 |
| Anti-inflammatory | 0.826 | 0.005 | |
| Histamine release inhibitor | 0.523 | 0.028 | |
| Antiparasitic | 0.489 | 0.017 | |
| Antihelmintic | 0.488 | 0.019 | |
| Anti-inflammatory, intestinal | 0.438 | 0.015 | |
| Methyl linoleate | Lipid metabolism regulator | 0.881 | 0.004 |
| Antisecretoric | 0.781 | 0.005 | |
| Anti-inflammatory | 0.727 | 0.013 | |
| Reductant | 0.637 | 0.009 | |
| Antihelmintic (Nematodes) | 0.500 | 0.017 | |
| Anti-infective | 0.424 | 0.038 | |
| Erucamide | Sugar-phosphatase inhibitor | 0.828 | 0.012 |
| Anti-infective | 0.501 | 0.022 | |
| Prostaglandin E1 antagonist | 0.470 | 0.005 | |
| Anti-inflammatory, intestinal | 0.444 | 0.014 | |
| Albendazole monooxygenase inhibitor | 0.450 | 0.026 | |
| Antitoxic | 0.387 | 0.025 | |
| γ-Sitosterol | Antihypercholesterolemic | 0.977 | 0.001 |
| Antiviral (Influenza) | 0.686 | 0.006 | |
| Antiinflammatory | 0.572 | 0.038 | |
| Antiacne | 0.529 | 0.005 | |
| Antiprotozoal (Leishmania) | 0.316 | 0.091 | |
| Antibacterial | 0.282 | 0.067 | |
| 2-Palmitoylglycerol | Sugar-phosphatase inhibitor | 0.927 | 0.003 |
| Lipid metabolism regulator | 0.889 | 0.004 | |
| Antiinfective | 0.757 | 0.005 | |
| Anti-inflammatory, intestinal | 0.578 | 0.004 | |
| Histamine release inhibitor | 0.573 | 0.015 | |
| Antiprotozoal (Leishmania) | 0.560 | 0.018 | |
| Methyl palmitate | Anti-inflammatory, intestinal | 0.758 | 0.002 |
| Calcium channel (voltage-sensitive) activator | 0.637 | 0.014 | |
| Antihelmintic (Nematodes) | 0.619 | 0.005 | |
| Reductant | 0.523 | 0.020 | |
| Antimutagenic | 0.513 | 0.014 | |
| Antiprotozoal (Leishmania) | 0.442 | 0.035 | |
| Methyl stearate | GABA aminotransferase inhibitor | 0.820 | 0.003 |
| Anti-inflammatory, intestinal | 0.758 | 0.002 | |
| Lipid metabolism regulator | 0.740 | 0.009 | |
| Gastrin inhibitor | 0.716 | 0.004 | |
| Antihelmintic (Nematodes) | 0.619 | 0.005 | |
| Antinociceptive | 0.538 | 0.019 | |
| Neophytadiene | Carminative | 0.691 | 0.007 |
| Gastrin inhibitor | 0.641 | 0.012 | |
| Antiulcerative | 0.585 | 0.012 | |
| Histamine release inhibitor | 0.506 | 0.034 | |
| Antiprotozoal (Leishmania) | 0.460 | 0.031 | |
| Antiparasitic | 0.395 | 0.032 | |
| Phytol | Lipid metabolism regulator | 0.828 | 0.005 |
| Antiparasitic | 0.615 | 0.008 | |
| Antihelmintic | 0.605 | 0.004 | |
| Antiprotozoal (Leishmania) | 0.601 | 0.014 | |
| Histamine release inhibitor | 0.526 | 0.027 | |
| Spasmolytic | 0.506 | 0.027 | |
| Squalene | Sugar-phosphatase inhibitor | 0.854 | 0.009 |
| Gastrin inhibitor | 0.743 | 0.003 | |
| Anti-inflammatory | 0.699 | 0.016 | |
| Antiparasitic | 0.555 | 0.011 | |
| Histamine release inhibitor | 0.558 | 0.018 | |
| Antihelmintic | 0.538 | 0.005 | |
| Stigmasterol | Dermatologic | 0.809 | 0.004 |
| Antiacne | 0.552 | 0.004 | |
| Antiinflammatory | 0.541 | 0.045 | |
| Antiprotozoal (Leishmania) | 0.403 | 0.047 | |
| Antisecretoric | 0.367 | 0.068 | |
| Bone formation stimulant | 0.306 | 0.020 | |
| Vitamin E | Lipid peroxidase inhibitor | 0.978 | 0.002 |
| Anti-inflammatory | 0.830 | 0.005 | |
| Free radical scavenger | 0.783 | 0.003 | |
| Spasmolytic | 0.525 | 0.024 | |
| Histamine release inhibitor | 0.396 | 0.093 | |
| Anti-infective | 0.277 | 0.122 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adnan, M.; Nazim Uddin Chy, M.; Mostafa Kamal, A.T.M.; Azad, M.O.K.; Paul, A.; Uddin, S.B.; Barlow, J.W.; Faruque, M.O.; Park, C.H.; Cho, D.H. Investigation of the Biological Activities and Characterization of Bioactive Constituents of Ophiorrhiza rugosa var. prostrata (D.Don) & Mondal Leaves through In Vivo, In Vitro, and In Silico Approaches. Molecules 2019, 24, 1367. https://doi.org/10.3390/molecules24071367
Adnan M, Nazim Uddin Chy M, Mostafa Kamal ATM, Azad MOK, Paul A, Uddin SB, Barlow JW, Faruque MO, Park CH, Cho DH. Investigation of the Biological Activities and Characterization of Bioactive Constituents of Ophiorrhiza rugosa var. prostrata (D.Don) & Mondal Leaves through In Vivo, In Vitro, and In Silico Approaches. Molecules. 2019; 24(7):1367. https://doi.org/10.3390/molecules24071367
Chicago/Turabian StyleAdnan, Md., Md. Nazim Uddin Chy, A.T.M. Mostafa Kamal, Md Obyedul Kalam Azad, Arkajyoti Paul, Shaikh Bokhtear Uddin, James W. Barlow, Mohammad Omar Faruque, Cheol Ho Park, and Dong Ha Cho. 2019. "Investigation of the Biological Activities and Characterization of Bioactive Constituents of Ophiorrhiza rugosa var. prostrata (D.Don) & Mondal Leaves through In Vivo, In Vitro, and In Silico Approaches" Molecules 24, no. 7: 1367. https://doi.org/10.3390/molecules24071367
APA StyleAdnan, M., Nazim Uddin Chy, M., Mostafa Kamal, A. T. M., Azad, M. O. K., Paul, A., Uddin, S. B., Barlow, J. W., Faruque, M. O., Park, C. H., & Cho, D. H. (2019). Investigation of the Biological Activities and Characterization of Bioactive Constituents of Ophiorrhiza rugosa var. prostrata (D.Don) & Mondal Leaves through In Vivo, In Vitro, and In Silico Approaches. Molecules, 24(7), 1367. https://doi.org/10.3390/molecules24071367










