Non-Destructive Determination of Alkylresorcinol (ARs) Content on Wheat Seed Surfaces and Prediction of ARs Content in Whole-Grain Flour
Abstract
1. Introduction
2. Results
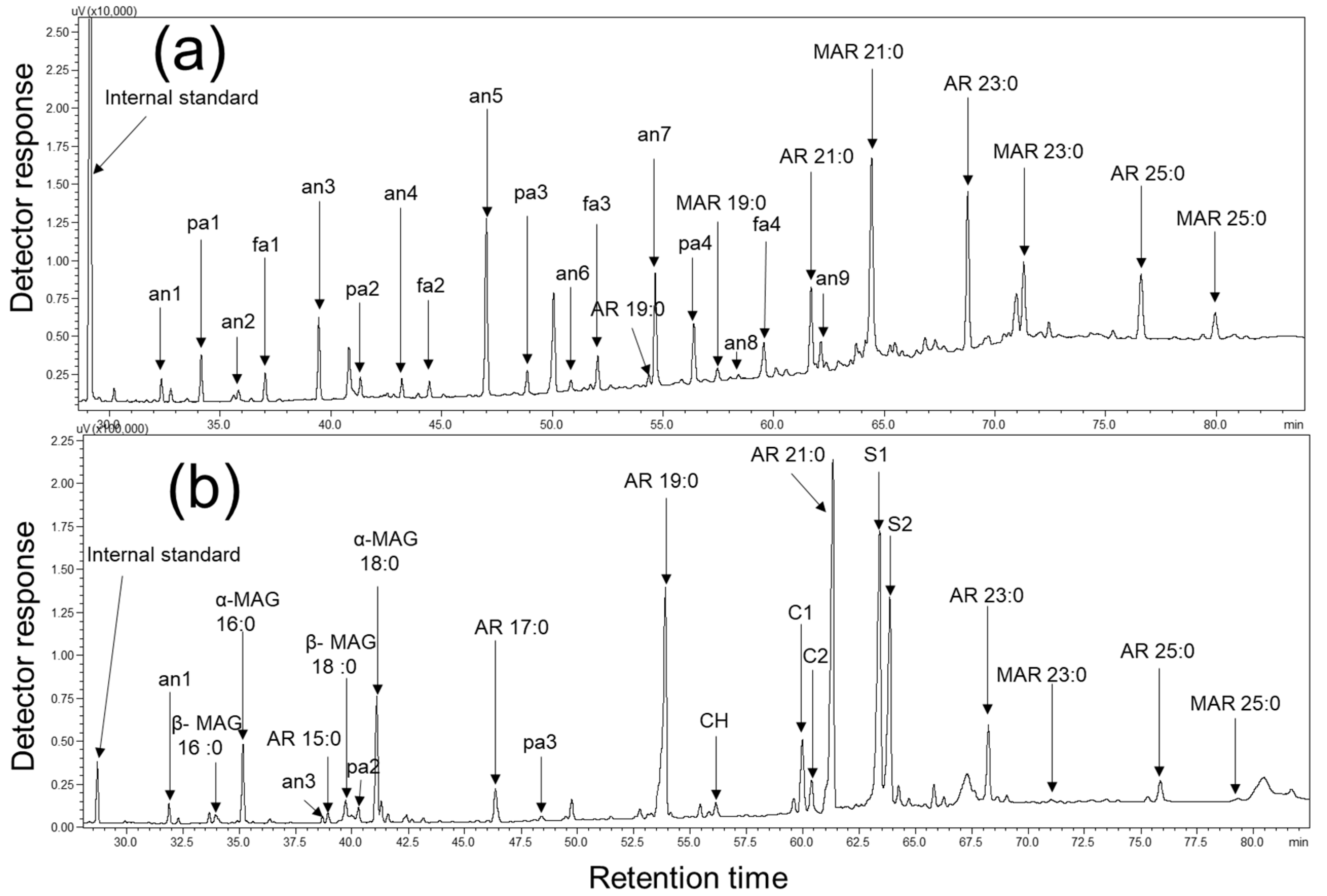
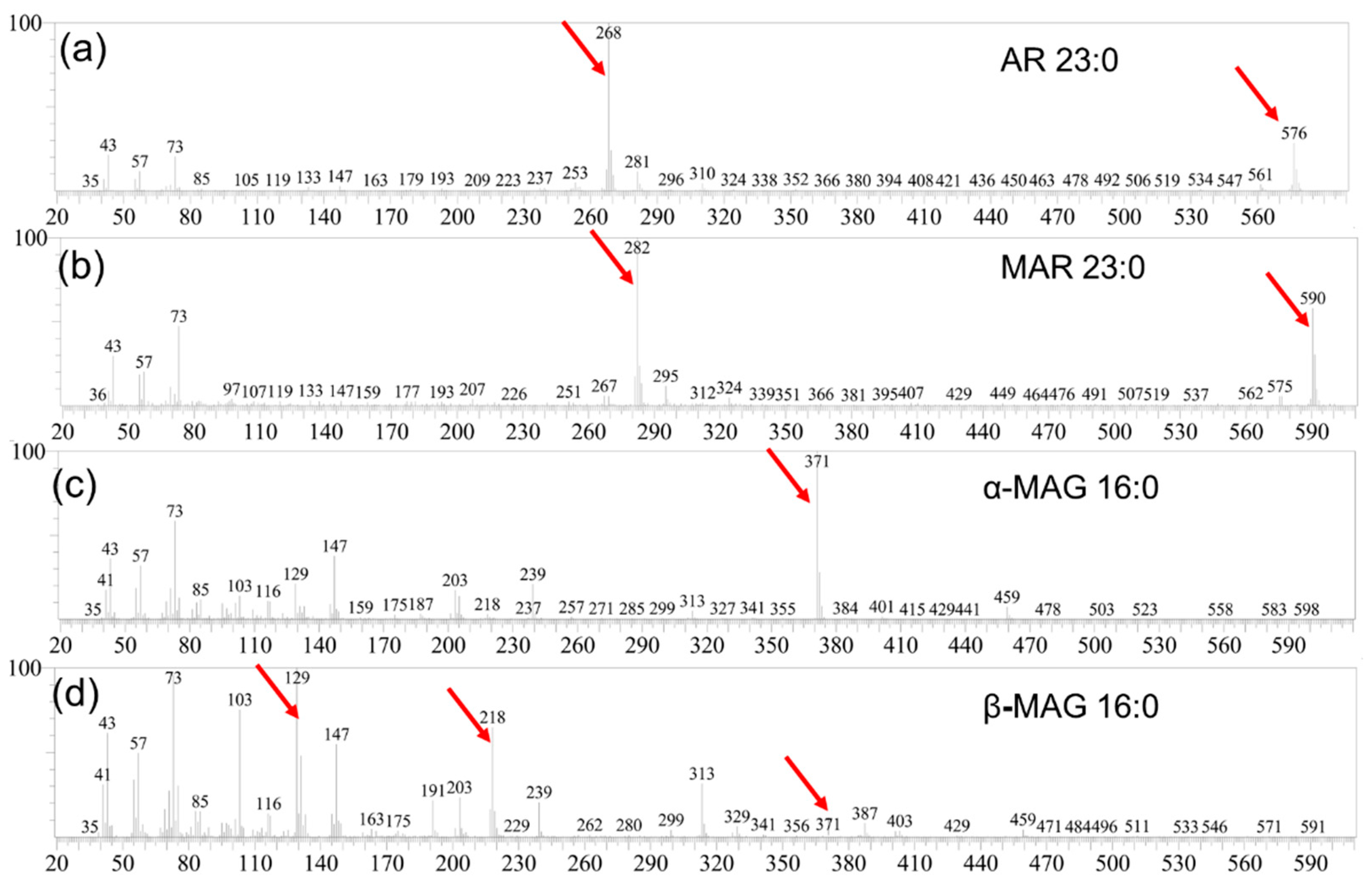
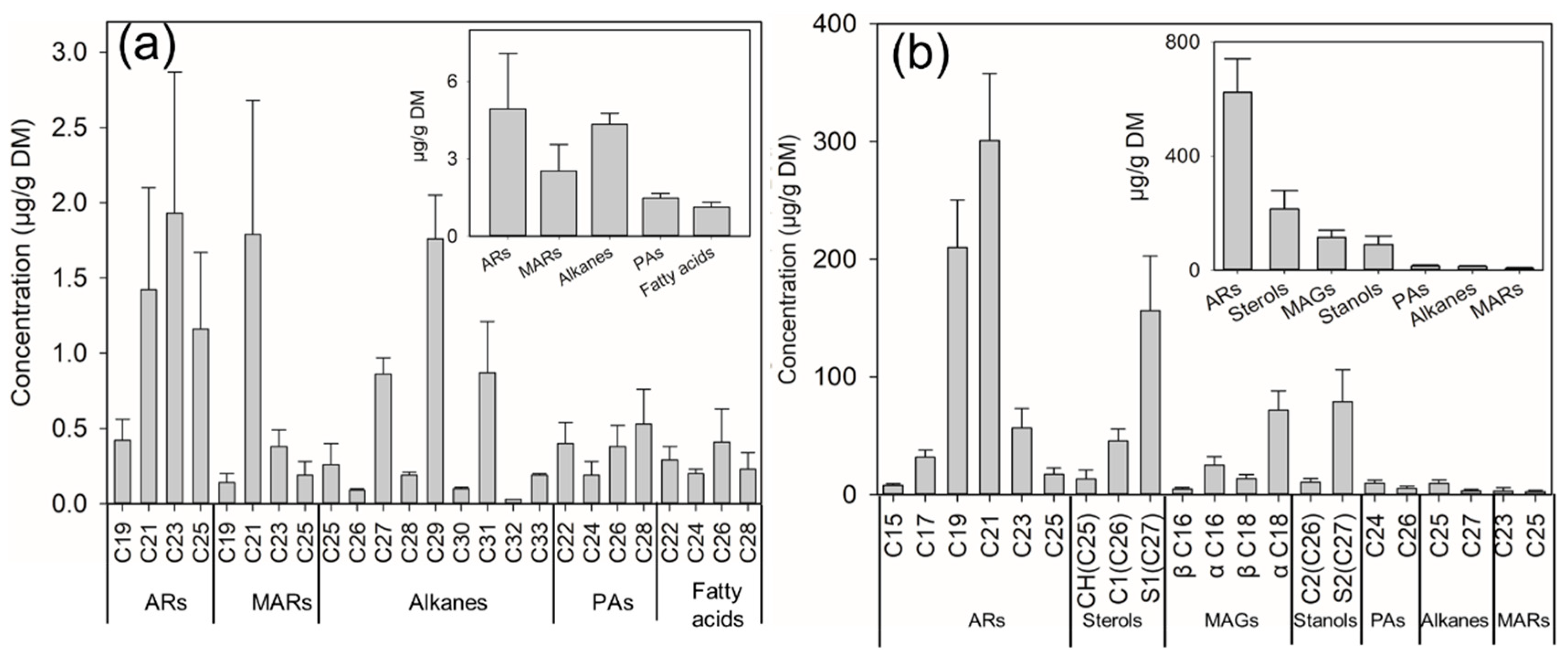
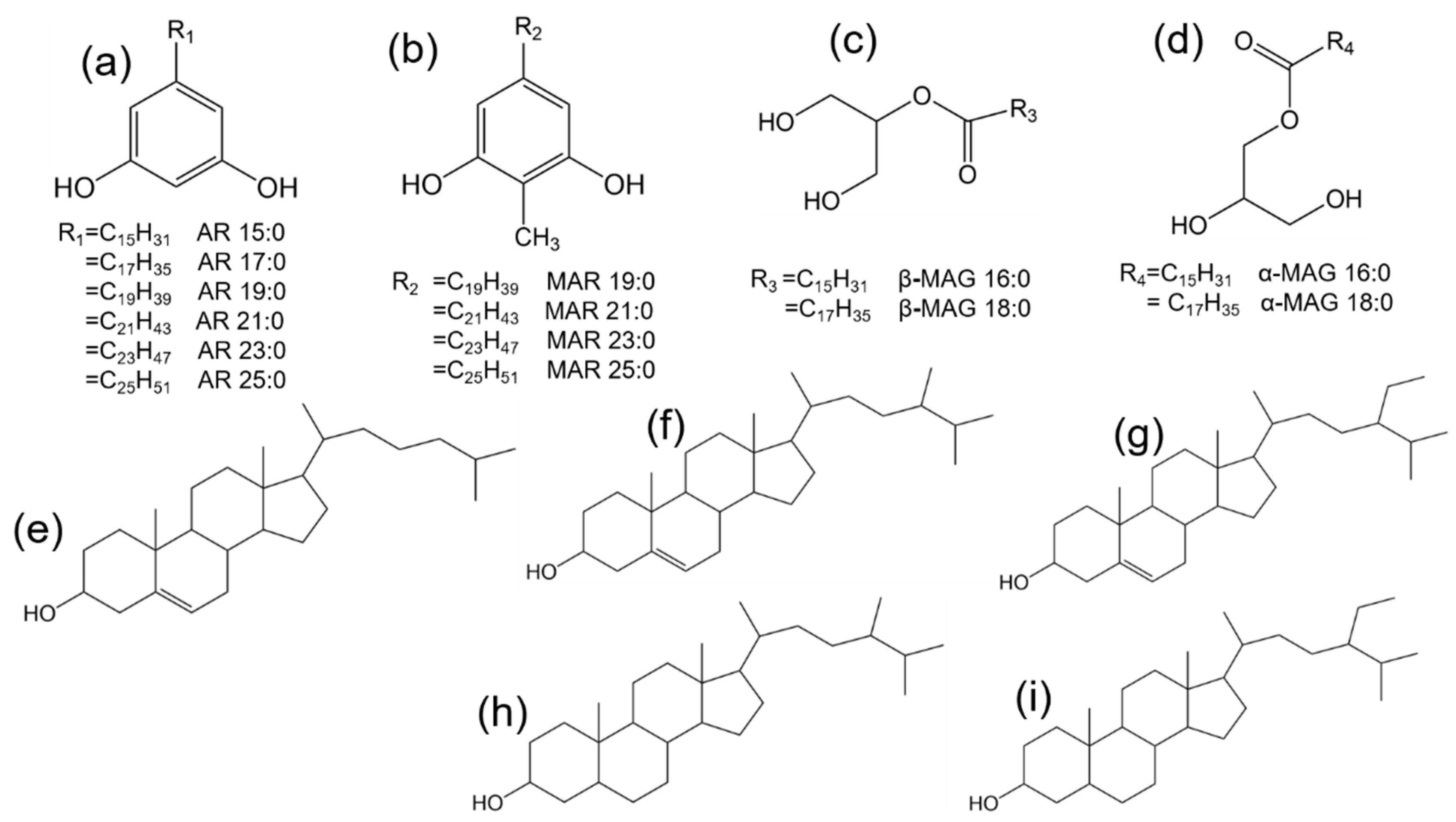
2.1. Identification and Quantification of SSL Components
2.2. Identification and Quantification of WGL Components
2.3. Differences in ARs Concents and Homologue Compositions on Seed Surfaces and in Whole-Grain Plour from 36 Wheat Cultivars
2.4. Correlation Analysis
2.4.1. Correlation Analysis between Homologues of ARs
2.4.2. Correlation Analysis between AR Homologues in Whole Grains and that on Seed Surfaces
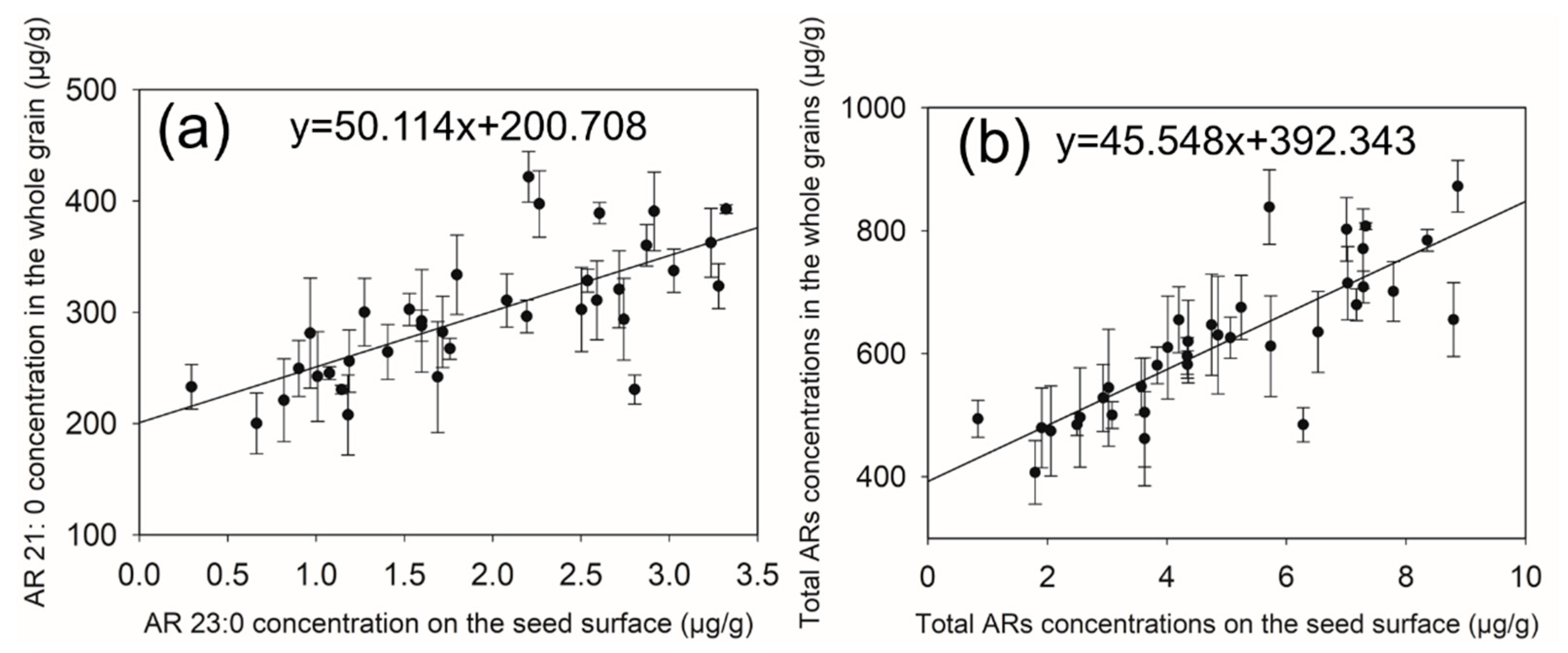
2.4.3. Predictive Model (Linear Regression Equations) Construction
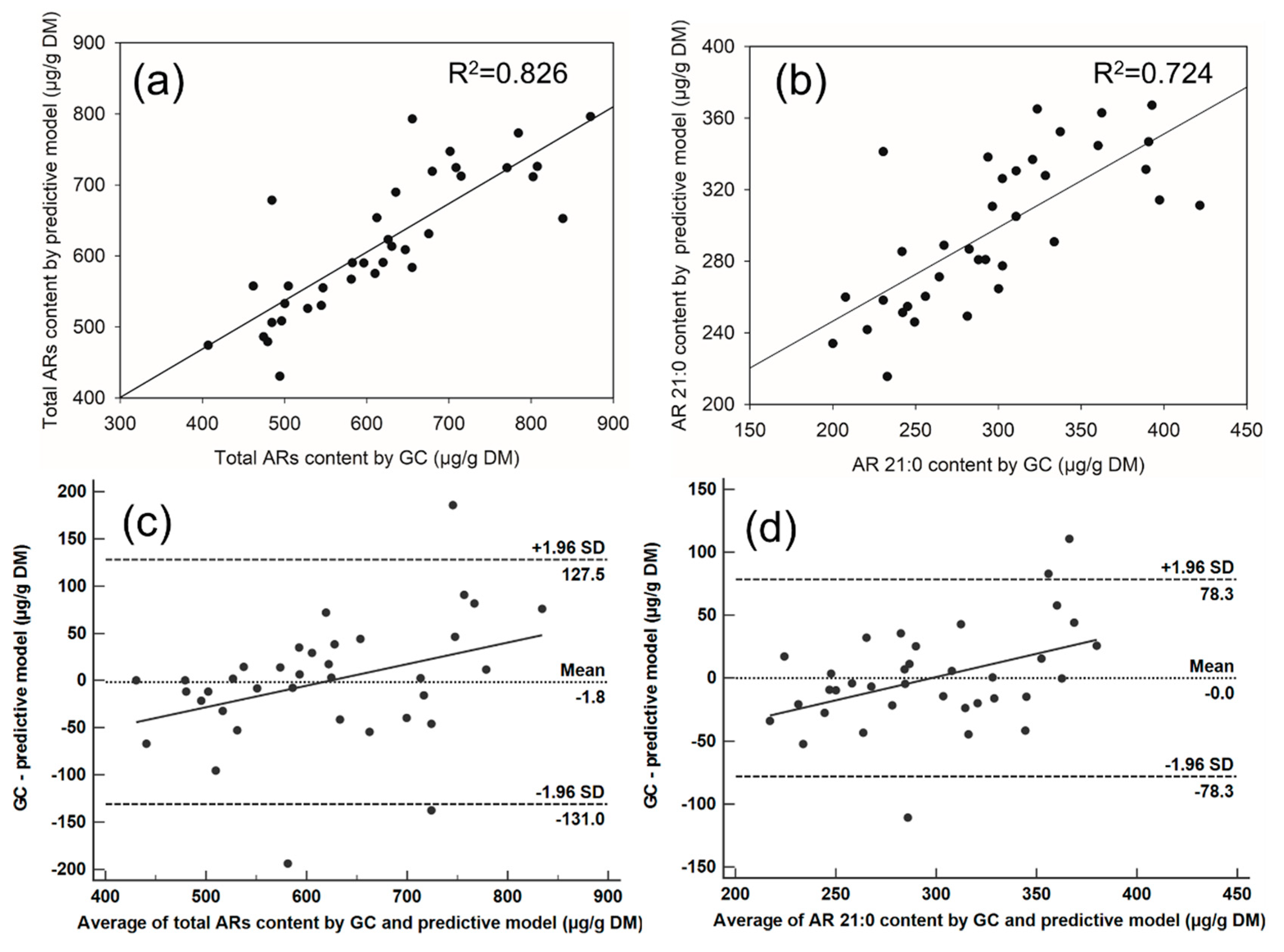
2.5. Methods Comparison
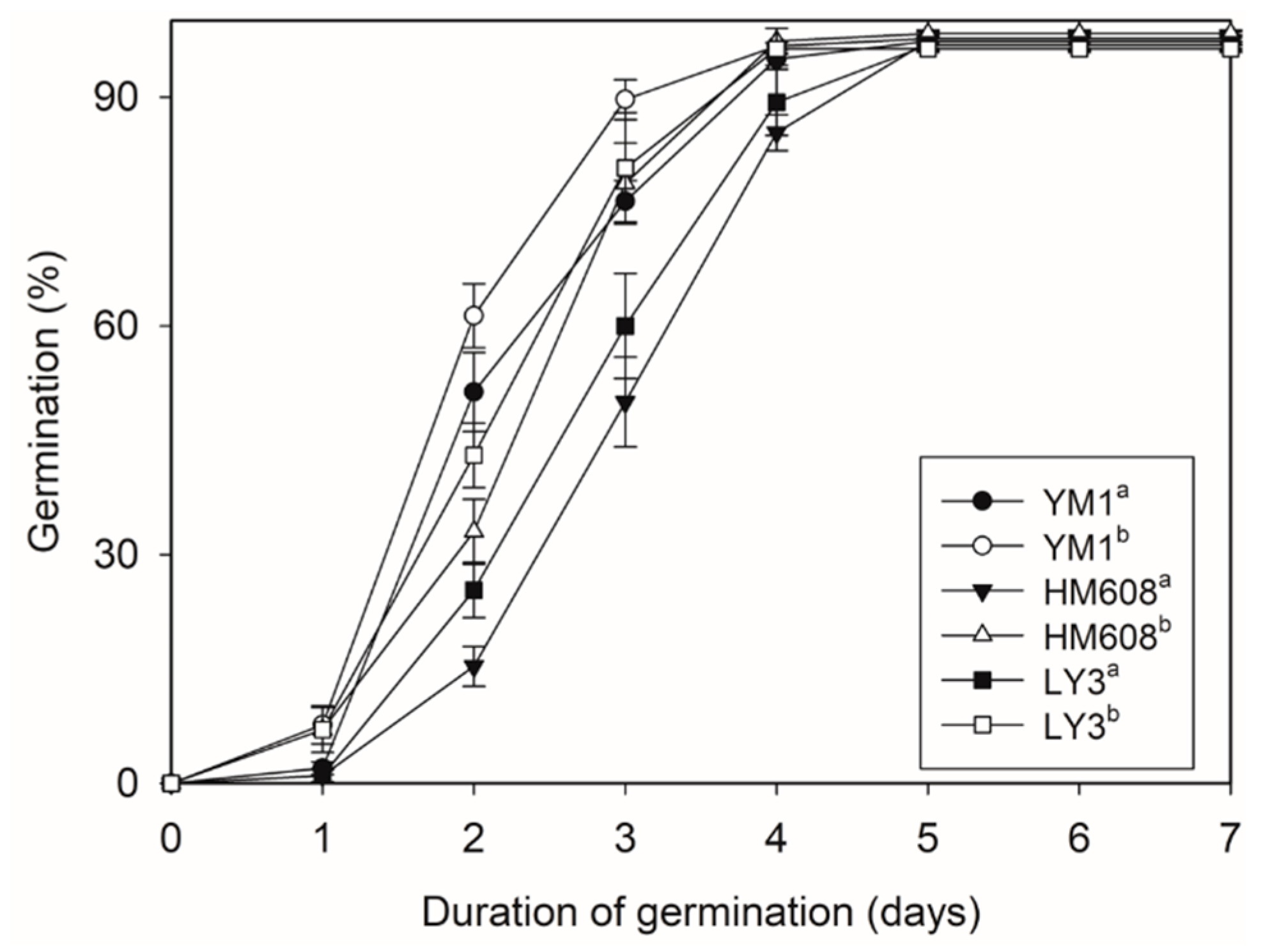
2.6. Wheat Germination Results
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Reagents
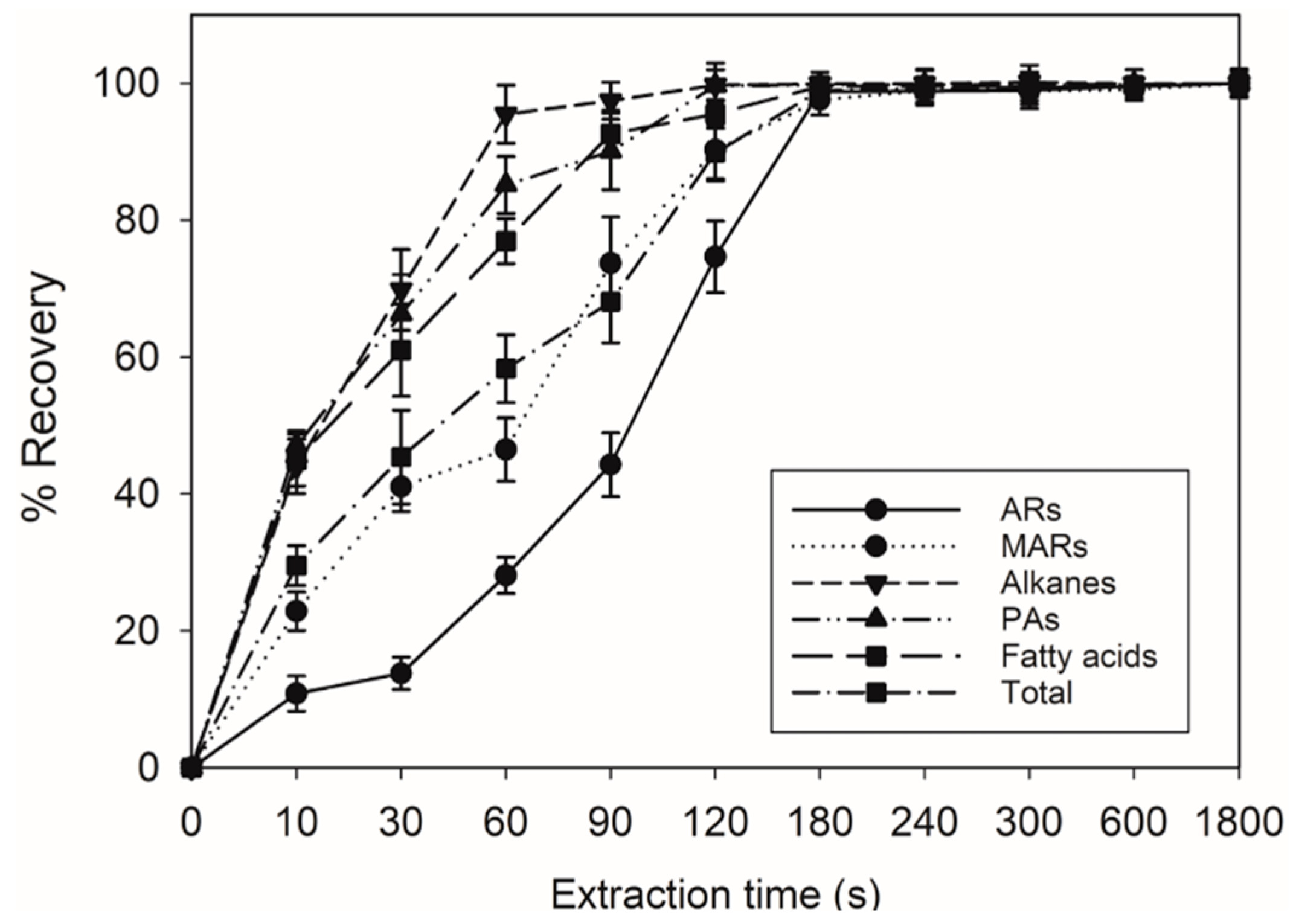
4.2. Developing a Suitable Method for Extracting Lipids from Seed Surfaces
4.3. Isolation of Lipids from Whole Grain Flour
4.4. Derivatization Reactions
4.5. Chemical Analysis of Lipids
4.6. Wheat Germination Tests
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Sample | Cultivar | Location | Latitude and Longitude |
|---|---|---|---|
| HWM6 | Huawanmai6 | Hefei, Anhui, China | 34°52′ N 117°17′ E |
| YMM1 | Yongminmai1 | Hefei, Anhui, China | 34°52′ N 117°17′ E |
| HH7 | Huaihe7 | Hefei, Anhui, China | 34°52′ N 117°17′ E |
| ZZM13 | Zhongzhimai13 | Beijing, China | 39°55′ N 116°24′ E |
| ZPM30 | Zhengpinmai30 | Zhengzhou, Henan, China | 34°46′ N 113°40′ E |
| CX29 | Chuangxing29 | Zhengzhou, Henan, China | 34°46′ N 113°40′ E |
| FDCM21 | Fengdecunmai21 | Zhengzhou, Henan, China | 34°46′ N 113°40′ E |
| FDCM23 | Fengdecunmai23 | Zhengzhou, Henan, China | 34°46′ N 113°40′ E |
| XR507 | Xiangrui507 | Zhengzhou, Henan, China | 34°46′ N 113°40′ E |
| GM8 | Guanmai8 | Zhengzhou, Henan, China | 34°46′ N 113°40′ E |
| HC878 | Huachang878 | Huaxian, Henan, China | 35°58′ N 114°52′ E |
| HZ166 | Huazhan166 | Suzhou, Anhui, China | 33°38′ N 116°58′ E |
| PA0658 | Pingan0658 | Jiaozuo, Henan, China | 35°14′ N 113°12′ E |
| HDM981 | Houdemai981 | Zhengzhou, Henan, China | 34°46′ N 113°40′ E |
| LY3 | Liangyuan3 | Luohe, Henan, China | 33°33′ N 114°02′ E |
| ZM162 | Zhengmai162 | Zhengzhou, Henan, China | 34°46′ N 113°40′ E |
| XM45 | Xinmai45 | Xinxiang, Henan, China | 35°18′ N 113°52′ E |
| YM118 | Youmai118 | Zhengzhou, Henan, China | 34°46′ N 113°40′ E |
| YM1 | Youmai1 | Zhengzhou, Henan, China | 34°46′ N 113°40′ E |
| XN10 | Xunong10 | Xuchang, Henan, China | 34°01′ N 113°49′ E |
| PX9 | Puxing9 | Puyang, Henan, China | 35°44′ N 115°01′ E |
| PM116 | Pumai116 | Puyang, Henan, China | 35°44′ N 115°01′ E |
| SM178 | Shangmai178 | Shangqiu, Henan, China | 34°26′ N 115°38′ E |
| CX216 | Chuangxing216 | Zhengzhou, Henan, China | 34°46′ N 113°40′ E |
| ZY1 | Zhongyuan1 | Zhengzhou, Henan, China | 34°46′ N 113°40′ E |
| HM608 | Huaimai608 | Huai’an, Jiangsu, China | 33°30′ N 119°09′ E |
| HH15173 | Huaihe15173 | Huai’an, Jiangsu, China | 33°30′ N 119°09′ E |
| XZ12 | Xiza12 | Hefei, Anhui, China | 34°52′ N 117°17′ E |
| SM119 | Shengmai119 | Jingning, Shandong, China | 35°23′ N 116°33′ E |
| RQM32 | Ruiquanmai32 | Zhengzhou, Henan, China | 34°46′ N 113°40′ E |
| XC998 | Xichun998 | Yangling, Shaanxi, China | 34°14′ N 107°59′ E |
| XN923 | Xinong923 | Yangling, Shaanxi, China | 34°14′ N 107°59′ E |
| TZS528 | Tezaoshu528 | Puyang, Henan, China | 35°44′ N 115°01′ E |
| THM6 | Taihemai6 | Zhengzhou, Henan, China | 34°46′ N 113°40′ E |
| JM208 | Jiamai208 | Huaibei, Anhui, China | 33°16′ N 116°23′ E |
| SC8658 | Shicha8658 | Xinxiang, Henan, China | 35°18′ N 113°52′ E |
| Sample | Alkylresorcinols Concentrations (µg/g DM) | 19:0/21:0 | ||||
|---|---|---|---|---|---|---|
| AR 19:0 | AR 21:0 | AR 23:0 | AR 25:0 | Total AR | ||
| HWM6 | 0.31 ± 0.02 | 1.21 ± 0.05 | 1.76 ± 0.17 | 1.07 ± 0.11 | 4.34 ± 0.28 | 0.26 |
| YMM1 | 0.06 ± 0.01 | 0.67 ± 0.11 | 1.80 ± 0.34 | 1.67 ± 0.25 | 4.20 ± 1.01 | 0.08 |
| HH7 | 0.06 ± 0.01 | 0.16 ± 0.04 | 0.30 ± 0.06 | 0.32 ± 0.08 | 0.84 ± 0.17 | 0.40 |
| ZZM13 | 0.30 ± 0.09 | 1.61 ± 0.20 | 2.50 ± 0.18 | 0.44 ± 0.19 | 4.85 ± 0.40 | 0.19 |
| ZPM30 | 0.38 ± 0.17 | 2.03 ± 0.22 | 3.24 ± 0.18 | 1.64 ± 0.14 | 7.28 ± 0.22 | 0.19 |
| CX29 | 0.37 ± 0.03 | 1.91 ± 0.14 | 2.80 ± 0.27 | 1.19 ± 0.11 | 6.28 ± 0.53 | 0.19 |
| FDCM21 | 0.30 ± 0.05 | 0.77 ± 0.11 | 1.08 ± 0.11 | 0.94 ± 0.10 | 3.08 ± 0.34 | 0.39 |
| FDCM23 | 1.92±0.15 | 3.51 ± 0.26 | 2.20 ± 0.16 | 1.23 ± 0.10 | 8.87 ± 0.64 | 0.55 |
| XR507 | 0.33 ± 0.03 | 0.81 ± 0.11 | 0.97 ± 0.12 | 0.92 ± 0.13 | 3.02 ± 0.51 | 0.41 |
| GM8 | 0.50 ± 0.07 | 1.47 ± 0.29 | 1.53 ± 0.32 | 0.84 ± 0.19 | 4.34 ± 0.18 | 0.34 |
| HC878 | 0.25 ± 0.04 | 1.31 ± 0.17 | 2.72 ± 0.30 | 2.26 ± 0.18 | 6.53 ± 1.18 | 0.19 |
| HZ166 | 0.09 ± 0.03 | 0.46 ± 0.09 | 0.66 ± 0.17 | 0.58 ± 0.12 | 1.79 ± 0.40 | 0.20 |
| PA0658 | 0.10 ± 0.02 | 0.98 ± 0.05 | 1.69 ± 0.13 | 0.86 ± 0.04 | 3.62 ± 0.19 | 0.10 |
| HDM981 | 0.16 ± 0.03 | 0.89 ± 0.07 | 1.19 ± 0.12 | 0.69 ± 0.02 | 2.93 ± 0.21 | 0.19 |
| LY3 | 0.47 ± 0.13 | 2.13 ± 0.24 | 3.28 ± 0.06 | 1.91 ± 0.01 | 7.79 ± 0.42 | 0.22 |
| ZM162 | 0.44 ± 0.07 | 1.68 ± 0.23 | 2.87 ± 0.43 | 2.18 ± 0.35 | 7.17 ± 1.15 | 0.26 |
| XM45 | 0.23 ± 0.03 | 1.22 ± 0.10 | 2.26 ± 0.34 | 2.00 ± 0.26 | 5.71 ± 0.74 | 0.19 |
| YM118 | 0.90 ± 0.14 | 2.57 ± 0.35 | 3.32 ± 0.10 | 1.56 ± 0.15 | 8.36 ± 0.18 | 0.35 |
| YM1 | 0.55 ± 0.06 | 2.05 ± 0.27 | 3.02 ± 0.38 | 1.40 ± 0.07 | 7.02 ± 0.73 | 0.27 |
| XN10 | 0.33 ± 0.13 | 1.36 ± 0.27 | 2.08 ± 0.23 | 1.47 ± 0.10 | 5.24 ± 0.71 | 0.24 |
| PX9 | 0.30 ± 0.14 | 1.33 ± 0.35 | 1.72 ± 0.35 | 1.01 ± 0.19 | 4.35 ± 0.95 | 0.22 |
| PM116 | 0.17 ± 0.01 | 1.09 ± 0.12 | 2.59 ± 0.66 | 0.90 ± 0.13 | 4.75 ± 0.46 | 0.16 |
| SM | 0.44 ± 0.07 | 2.04 ± 0.24 | 2.91 ± 0.03 | 1.61 ± 0.09 | 7.01 ± 0.24 | 0.22 |
| CX216 | 0.22±0.07 | 1.31 ± 0.21 | 2.19 ± 0.22 | 1.34 ± 0.10 | 5.06 ± 0.53 | 0.17 |
| ZY1 | 0.42 ± 0.08 | 1.33 ± 0.06 | 1.18 ± 0.32 | 0.69 ± 0.18 | 3.63 ± 0.40 | 0.32 |
| HM608 | 0.21 ± 0.08 | 0.97 ± 0.06 | 1.41 ± 0.32 | 0.98 ± 0.18 | 3.57 ± 0.40 | 0.22 |
| HH15173 | 0.04 ± 0.01 | 0.45 ± 0.08 | 0.90 ± 0.19 | 0.50 ± 0.08 | 1.90 ± 0.34 | 0.10 |
| XZ12 | 0.09 ± 0.02 | 0.58 ± 0.03 | 1.01 ± 0.04 | 0.86 ± 0.07 | 2.55 ± 0.07 | 0.15 |
| SM119 | 0.08 ± 0.02 | 0.67 ± 0.04 | 1.60 ± 0.03 | 1.50 ± 0.05 | 3.84 ± 0.12 | 0.11 |
| RQM32 | 0.13 ± 0.04 | 0.51 ± 0.05 | 0.82 ± 0.04 | 0.60 ± 0.02 | 2.06 ± 0.13 | 0.25 |
| XC998 | 0.16 ± 0.02 | 0.81 ± 0.13 | 1.60 ± 0.16 | 1.45 ± 0.26 | 4.01 ± 0.40 | 0.20 |
| XN923 | 0.09 ± 0.04 | 0.66 ± 0.03 | 1.14 ± 0.09 | 0.61 ± 0.16 | 2.49 ± 0.21 | 0.13 |
| TZS528 | 2.84 ± 0.50 | 4.20 ± 0.53 | 1.27 ± 0.13 | 0.48 ± 0.14 | 8.79 ± 0.78 | 0.68 |
| THM6 | 0.13 ± 0.02 | 1.31 ± 0.20 | 2.74 ± 0.25 | 1.55 ± 0.14 | 5.74 ± 0.52 | 0.10 |
| JM208 | 0.96 ± 0.12 | 2.66 ± 0.14 | 2.54 ± 0.31 | 1.13 ± 0.15 | 7.29 ± 0.43 | 0.36 |
| SC8658 | 0.80 ± 0.11 | 2.45 ± 0.27 | 2.60 ± 0.19 | 1.47 ± 0.12 | 7.32 ± 0.66 | 0.33 |
| Mean | 0.42 | 1.42 | 1.93 | 1.16 | 4.93 | 0.25 |
| C.V. (%) | 89.25 | 61.57 | 43.32 | 43.76 | 43.63 | 50.45 |
| Sample | Alkylresorcinols Concentrations (µg/g DM) | 17:0/21:0 | 19:0/21:0 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| AR 15:0 | AR 17:0 | AR 19:0 | AR 21:0 | AR 23:0 | AR 25:0 | Total AR | |||
| HWM6 | 9.2 ± 2.1 | 33.5 ± 3.3 | 212.7 ± 13.7 | 267.2 ± 9.4 | 45.1 ± 2.3 | 14.7 ± 1.4 | 582.4 ± 22.3 | 0.13 | 0.80 |
| YMM1 | 7.2 ± 0.5 | 34.1 ± 1.2 | 223.8 ± 19.4 | 333.7 ± 35.7 | 44.5 ± 2.4 | 11.8 ± 0.6 | 655.2 ± 53.7 | 0.10 | 0.67 |
| HH7 | 5.2 ± 0.1 | 26.1 ± 1.4 | 168.2 ± 9.0 | 232.9 ± 20.0 | 47.6 ± 1.6 | 14.3 ± 0.5 | 494.1 ± 30.1 | 0.11 | 0.72 |
| ZZM13 | 8.8 ± 2.5 | 34.2 ± 8.5 | 211.9 ± 33.9 | 302.5 ± 37.9 | 61.7 ± 11.4 | 17.2 ± 2.7 | 630.5 ± 95.6 | 0.11 | 0.70 |
| ZPM30 | 7.8 ± 0.9 | 36.7 ± 1.4 | 253.1 ± 25.1 | 362.5 ± 30.9 | 86.5 ± 7.1 | 24.1 ± 2.2 | 770.7 ± 64.9 | 0.10 | 0.70 |
| CX29 | 7.0 ± 0.7 | 25.8 ± 2.0 | 170.2 ± 12.0 | 230.5 ± 13.1 | 39.0 ± 3.0 | 12.0 ± 2.1 | 484.6 ± 27.8 | 0.11 | 0.74 |
| FDCM21 | 7.1 ± 1.9 | 25.9 ± 3.7 | 168.1 ± 11.4 | 245.2 ± 5.8 | 42.6 ± 4.7 | 13.0 ± 2.8 | 500.3 ± 21.8 | 0.11 | 0.69 |
| FDCM23 | 8.3 ± 0.5 | 41.8 ± 1.5 | 295.3 ± 10.4 | 421.7 ± 22.7 | 81.4 ± 7.1 | 23.9 ± 3.6 | 872.3 ± 42.0 | 0.10 | 0.70 |
| XR507 | 8.7 ± 1.7 | 25.6 ± 4.5 | 175.5 ± 30.6 | 281.3 ± 49.4 | 40.5 ± 7.2 | 13.1 ± 2.3 | 544.7 ± 95.1 | 0.09 | 0.62 |
| GM8 | 8.2 ± 1.2 | 31.4 ± 1.5 | 204.7 ± 13.8 | 302.6 ± 14.4 | 37.2 ± 1.7 | 12.1 ± 0.6 | 596.3 ± 29.8 | 0.10 | 0.68 |
| HC878 | 7.4 ± 0.4 | 29.9 ± 2.3 | 222.9 ± 24.8 | 320.7 ± 34.5 | 40.7 ± 4.3 | 13.8 ± 1.6 | 635.4 ± 66.0 | 0.09 | 0.69 |
| HZ166 | 8.1 ± 0.4 | 18.7 ± 1.5 | 132.8 ± 16.3 | 200.1 ± 27.3 | 33.7 ± 4.4 | 13.6 ± 2.6 | 407.0 ± 51.6 | 0.09 | 0.66 |
| PA0658 | 10.9 ± 1.0 | 25.3 ± 5.9 | 165.1 ± 26.1 | 241.8 ± 49.8 | 48.9 ± 8.9 | 12.5 ± 2.0 | 504.5 ± 88.7 | 0.10 | 0.68 |
| HDM981 | 6.2 ± 0.2 | 23.2 ± 0.5 | 179.5 ± 17.6 | 256.0 ± 28.0 | 47.9 ± 9.9 | 15.2 ± 2.9 | 528.0 ± 54.4 | 0.09 | 0.70 |
| LY3 | 7.3 ± 1.0 | 34.6 ± 4.3 | 234.6 ± 18.1 | 323.6 ± 20.1 | 75.4 ± 6.7 | 25.9 ± 4.7 | 701.4 ± 48.4 | 0.11 | 0.73 |
| ZM162 | 7.7 ± 0.2 | 31.8 ± 2.7 | 206.1 ± 8.7 | 360.2 ± 18.7 | 56.7 ± 1.5 | 17.2 ± 0.5 | 679.7 ± 26.0 | 0.09 | 0.57 |
| XM45 | 8.6 ± 0.7 | 38.2 ± 1.6 | 285.9 ± 21.4 | 397.4 ± 29.9 | 81.6 ± 6.2 | 27.0 ± 2.1 | 838.6 ± 60.5 | 0.10 | 0.72 |
| YM118 | 8.6 ± 1.8 | 42.4 ± 5.7 | 226.0 ± 6.1 | 392.8 ± 3.9 | 85.7 ± 6.3 | 29.0 ± 6.5 | 784.5 ± 17.7 | 0.11 | 0.58 |
| YM1 | 9.1 ± 2.4 | 32.2 ± 0.8 | 227.6 ± 38.3 | 337.4 ± 19.5 | 84.2 ± 7.7 | 24.2 ± 3.2 | 714.8 ± 59.5 | 0.10 | 0.67 |
| XN10 | 9.2 ± 1.0 | 36.8 ± 2.8 | 252.6 ± 19.5 | 310.7 ± 24.0 | 45.3 ± 3.5 | 20.8 ± 1.8 | 675.4 ± 52.1 | 0.12 | 0.81 |
| PX9 | 7.7 ± 0.1 | 31.4 ± 3.3 | 222.7 ± 20.4 | 282.4 ± 32.0 | 59.6 ± 12.9 | 16.2 ± 1.5 | 619.9 ± 67.1 | 0.11 | 0.79 |
| PM116 | 10.1 ± 2.2 | 32.5 ± 5.4 | 219.7 ± 27.8 | 310.8 ± 35.5 | 54.8 ± 9.9 | 19.1 ± 2.0 | 647.0 ± 82.2 | 0.10 | 0.71 |
| SM | 9.2 ± 0.6 | 40.3 ± 2.6 | 281.1 ± 8.4 | 390.8 ± 35.4 | 64.2 ± 5.2 | 16.8 ± 1.9 | 802.3 ± 51.5 | 0.10 | 0.72 |
| CX216 | 9.0 ± 0.7 | 29.7 ± 2.8 | 182.0 ± 10.3 | 296.4 ± 14.8 | 80.5 ± 8.9 | 28.4 ± 2.3 | 626.0 ± 33.5 | 0.10 | 0.61 |
| ZY1 | 9.4 ± 0.8 | 27.5 ± 5.1 | 161.9 ± 26.7 | 207.7 ± 36.0 | 39.4 ± 7.0 | 15.9 ± 2.3 | 461.9 ± 76.4 | 0.13 | 0.78 |
| HM608 | 6.9 ± 0.3 | 22.8 ± 1.4 | 182.6 ± 15.2 | 264.3 ± 24.7 | 54.6 ± 3.5 | 15.4 ± 1.0 | 546.7 ± 45.8 | 0.09 | 0.69 |
| HH15173 | 7.4 ± 0.5 | 20.3 ± 2.4 | 144.8 ± 31.4 | 249.4 ± 25.2 | 45.7 ± 5.4 | 11.9 ± 1.3 | 479.5 ± 64.8 | 0.08 | 0.58 |
| XZ12 | 7.7 ± 0.4 | 25.3 ± 3.6 | 172.3 ± 26.2 | 242.3 ± 40.4 | 41.8 ± 9.8 | 11.0 ± 1.7 | 496.4 ± 80.7 | 0.10 | 0.71 |
| SM119 | 6.1 ± 1.7 | 29.3 ± 1.7 | 183.6 ± 11.0 | 288.0 ± 14.1 | 57.5 ± 5.2 | 16.7 ± 1.6 | 581.1 ± 29.8 | 0.10 | 0.64 |
| RQM32 | 6.8 ± 2.0 | 22.7 ± 2.9 | 169.3 ± 19.6 | 220.9 ± 37.3 | 43.4 ± 13.4 | 11.4 ± 1.7 | 474.4 ± 73.2 | 0.10 | 0.77 |
| XC998 | 5.7 ± 2.2 | 29.2 ± 4.5 | 221.8 ± 25.2 | 292.2 ± 46.1 | 48.2 ± 6.3 | 12.9 ± 1.7 | 610.1 ± 83.8 | 0.10 | 0.76 |
| XN923 | 7.0 ± 1.8 | 25.8 ± 5.7 | 170.2 ± 6.1 | 230.5 ± 3.9 | 39.0 ± 6.3 | 12.0 ± 6.5 | 484.6 ± 17.7 | 0.11 | 0.74 |
| TZS528 | 11.1 ± 0.3 | 40.3 ± 1.9 | 244.1 ± 26.1 | 300.1 ± 30.5 | 47.2 ± 2.0 | 12.7 ± 1.0 | 655.5 ± 60.0 | 0.13 | 0.81 |
| THM6 | 9.1 ± 2.0 | 33.4 ± 5.7 | 215.3 ± 31.0 | 293.7 ± 36.7 | 46.3 ± 7.9 | 14.5 ± 1.9 | 612.3 ± 82.0 | 0.11 | 0.73 |
| JM208 | 6.5 ± 0.6 | 33.2±1.7 | 233.7 ± 17.9 | 328.5 ± 10.4 | 83.9 ± 18.8 | 22.7 ± 0.9 | 708.6 ± 25.9 | 0.10 | 0.71 |
| SC8658 | 7.2 ± 0.1 | 33.2 ± 1.3 | 280.7 ± 2.3 | 389.1 ± 9.6 | 74.6 ± 10.3 | 22.8 ± 1.4 | 807.5 ± 5.4 | 0.09 | 0.72 |
| Mean | 8.01 | 31.81 | 210.01 | 300.81 | 56.61 | 17.27 | 624.22 | 0.10 | 0.70 |
| C.V. (%) | 16.82 | 18.71 | 19.28 | 18.98 | 29.03 | 30.82 | 18.75 | 11.44 | 8.57 |
References
- Ross, A.B.; Shepherd, M.J.; Schupphaus, M.; Sinclair, V.; Alfaro, B.; Kamal-Eldin, A.; Aman, P. Alkylresorcinols in cereals and cereal products. J. Agric. Food Chem. 2003, 51, 4111–4118. [Google Scholar] [CrossRef]
- Racovita, R.C.; Hen-Avivi, S.; Fernandez-Moreno, J.-P.; Granell, A.; Aharoni, A.; Jetter, R. Composition of cuticular waxes coating flag leaf blades and peduncles of Triticum aestivum cv. Bethlehem. Phytochemistry 2016, 130, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Landberg, R.; Kamal-Eldin, A.; Andersson, R.; Aman, P. Alkylresorcinol content and homologue composition in durum wheat (Triticum durum) kernels and pasta products. J. Agric. Food Chem. 2006, 54, 3012–3014. [Google Scholar] [CrossRef] [PubMed]
- Geng, P.; Harnly, J.M.; Chen, P. Differentiation of Whole Grain from Refined Wheat (T-aestivum) Flour Using Lipid Profile of Wheat Bran, Germ, and Endosperm with UHPLC-HRAM Mass Spectrometry. J. Agric. Food Chem. 2015, 63, 6189–6211. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Conklin, D.R.; Chen, H.; Wang, L.; Sang, S. 5-Alk(en)ylresorcinols as the major active components in wheat bran inhibit human colon cancer cell growth. Bioorganic Med. Chem. 2011, 19, 3973–3982. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.B.; Kamal-Eldin, A.; Aman, P. Dietary alkylresorcinols: Absorption, bioactivities, and possible use as biomarkers of whole-grain wheat- and rye-rich foods. Nutr. Rev. 2004, 62, 81–95. [Google Scholar] [CrossRef] [PubMed]
- Landberg, R.; Kamal-Eldin, A.; Andersson, A.; Vessby, B.; Aman, P. Alkylresorcinols as biomarkers of whole-grain wheat and rye intake: Plasma concentration and intake estimated from dietary records. Am. J. Clin. Nutr. 2008, 87, 832–838. [Google Scholar] [CrossRef]
- Ross, A.B. Present status and perspectives on the use of alkylresorcinols as biomarkers of wholegrain wheat and rye intake. J. Nutr. Metab. 2012, 2012, 462967. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.B.; Bourgeois, A.; Macharia, H.N.u.; Kochhar, S.; Jebb, S.A.; Brownlee, I.A.; Seal, C.J. Plasma alkylresorcinols as a biomarker of whole-grain food consumption in a large population: Results from the WHOLEheart Intervention Study. Am. J. Clin. Nutr. 2012, 95, 204–211. [Google Scholar] [CrossRef]
- Kyro, C.; Tjonneland, A.; Overvad, K.; Olsen, A.; Landberg, R. Higher Whole-Grain Intake Is Associated with Lower Risk of Type 2 Diabetes among Middle-Aged Men and Women: The Danish Diet, Cancer, and Health Cohort. J. Nutr. 2018, 148, 1434–1444. [Google Scholar] [CrossRef]
- Della Pepa, G.; Vetrani, C.; Vitale, M.; Riccardi, G. Wholegrain Intake and Risk of Type 2 Diabetes: Evidence from Epidemiological and Intervention Studies. Nutrients 2018, 10, 1288. [Google Scholar] [CrossRef] [PubMed]
- Biskup, I.; Kyro, C.; Marklund, M.; Olsen, A.; van Dam, R.M.; Tjonneland, A.; Overvad, K.; Lindahl, B.; Johansson, I.; Landberg, R. Plasma alkylresorcinols, biomarkers of whole-grain wheat and rye intake, and risk of type 2 diabetes in Scandinavian men and women. Am. J. Clin. Nutr. 2016, 104, 88–96. [Google Scholar] [CrossRef]
- Ross, A.B.; Chen, Y.; Frank, J.; Swanson, J.E.; Parker, R.S.; Kozubek, A.; Lundh, T.; Vessby, B.; Aman, P.; Kamal-Eldin, A. Cereal alkylresorcinols elevate gamma-tocopherol levels in rats and inhibit gamma-tocopherol metabolism in vitro. J. Nutr. 2004, 134, 506–510. [Google Scholar] [CrossRef]
- Ma, J.; Ross, A.B.; Shea, M.K.; Bruce, S.J.; Jacques, P.F.; Saltzman, E.; Lichtenstein, A.H.; Booth, S.L.; McKeown, N.M. Plasma Alkylresorcinols, Biomarkers of Whole-Grain Intake, Are Related to Lower BMI in Older Adults. J. Nutr. 2012, 142, 1859–1864. [Google Scholar] [CrossRef]
- Horikawa, K.; Hashimoto, C.; Kikuchi, Y.; Makita, M.; Fukudome, S.-i.; Okita, K.; Wada, N.; Oishi, K. Wheat alkylresorcinols reduce micellar solubility of cholesterol in vitro and increase cholesterol excretion in mice. Nat. Prod. Res. 2017, 31, 578–582. [Google Scholar] [CrossRef] [PubMed]
- Oishi, K.; Yamamoto, S.; Itoh, N.; Nakao, R.; Yasumoto, Y.; Tanaka, K.; Kikuchi, Y.; Fukudome, S.-i.; Okita, K.; Takano-Ishikawa, Y. Wheat Alkylresorcinols Suppress High-Fat, High-Sucrose Diet-Induced Obesity and Glucose Intolerance by Increasing Insulin Sensitivity and Cholesterol Excretion in Male Mice. J. Nutr. 2015, 145, 199–206. [Google Scholar] [CrossRef]
- Landberg, R.; Kamal-Eldin, A.; Andersson, S.-O.; Johansson, J.-E.; Zhang, J.-X.; Hallmans, G.; Aman, P. Reproducibility of Plasma Alkylresorcinols during a 6-Week Rye Intervention Study in Men with Prostate Cancer. J. Nutr. 2009, 139, 975–980. [Google Scholar] [CrossRef]
- Newby, P.K.; Maras, J.; Bakun, P.; Muller, D.; Ferrucci, L.; Tucker, K.L. Intake of whole grains, refined grains, and cereal fiber measured with 7-d diet records and associations with risk factors for chronic disease. Am. J. Clin. Nutr. 2007, 86, 1745–1753. [Google Scholar] [CrossRef] [PubMed]
- Poutanen, K.; Shepherd, R.; Shewry, P.R.; Delcour, J.A.; Bjorck, I.; van der Kamp, J.-W. Beyond whole grain: The European HEALTHGRAIN project aims at healthier cereal foods. Cereal Foods World 2008, 53, 32–35. [Google Scholar] [CrossRef]
- Damsgaard, C.T.; Biltoft-Jensen, A.; Tetens, I.; Michaelsen, K.F.; Lind, M.V.; Astrup, A.; Landberg, R. Whole-Grain Intake, Reflected by Dietary Records and Biomarkers, Is Inversely Associated with Circulating Insulin and Other Cardiometabolic Markers in 8-to 11-Year-Old Children. J. Nutr. 2017, 147, 816–824. [Google Scholar] [CrossRef] [PubMed]
- Landberg, R.; Kamal-Eldin, A.; Salmenkallio-Marttila, M.; Rouau, X.; Aman, P. Localization of alkylresorcinols in wheat, rye and barley kernels. J. Cereal Sci. 2008, 48, 401–406. [Google Scholar] [CrossRef]
- Ross, A.B.; Kochhar, S. Rapid and Sensitive Analysis of Alkylresorcinols from Cereal Grains and Products Using HPLC-Coularray-Based Electrochemical Detection. J. Agric. Food Chem. 2009, 57, 5187–5193. [Google Scholar] [CrossRef]
- Menzel, C.; Kamal-Eldin, A.; Marklund, M.; Andersson, A.; Aman, P.; Landberg, R. Alkylresorcinols in Swedish cereal food products. J. Food Compos. Anal. 2012, 28, 119–125. [Google Scholar] [CrossRef]
- Kulawinek, M.; Jaromin, A.; Kozubek, A.; Zarnowski, R. Alkylresorcinols in selected polish rye and wheat cereals and whole-grain cereal products. J. Agric. Food Chem. 2008, 56, 7236–7242. [Google Scholar] [CrossRef]
- Zarnowski, R.; Suzuki, Y. Expedient Soxhlet extraction of resorcinolic lipids from wheat grains. J. Food Compos. Anal. 2004, 17, 649–663. [Google Scholar] [CrossRef]
- Knoedler, M.; Most, M.; Schieber, A.; Carle, R. A novel approach to authenticity control of whole grain durum wheat (Triticum durum Desf.) flour and pasta, based on analysis of alkylresorcinol composition. Food Chem. 2010, 118, 177–181. [Google Scholar] [CrossRef]
- Ross, A.B.; Kamal-Eldin, A.; Jung, C.; Shepherd, M.J.; Aman, P. Gas chromatographic analysis of alkylresorcinols in rye (Secale cereale L.) grains. J. Sci. Food Agric. 2001, 81, 1405–1411. [Google Scholar] [CrossRef]
- Geerkens, C.H.; Matejka, A.E.; Carle, R.; Schweiggert, R.M. Development and validation of an HPLC method for the determination of alk(en)ylresorcinols using rapid ultrasound-assisted extraction of mango peels and rye grains. Food Chem. 2015, 169, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Shewry, P.R.; Charmet, G.; Branlard, G.; Lafiandra, D.; Gergely, S.; Salgo, A.; Saulnier, L.; Bedo, Z.; Mills, E.N.C.; Ward, J.L. Developing new types of wheat with enhanced health benefits. Trends Food Sci. Technol. 2012, 25, 70–77. [Google Scholar] [CrossRef]
- Jenks, M.A.; Joly, R.J.; Peters, P.J.; Rich, P.J.; Axtell, J.D.; Ashworth, E.N. Chemically induced cuticle mutation affecting epidermal conductance to water vapor and disease susceptibility in Sorghum bicolor (L). Moench. Plant Physiol. 1994, 105, 1239–1245. [Google Scholar] [CrossRef] [PubMed]
- Holmes, M.G.; Keiller, D.R. Effects of pubescence and waxes on the reflectance of leaves in the ultraviolet and photosynthetic wavebands: A comparison of a range of species. Plant Cell Environ. 2002, 25, 85–93. [Google Scholar] [CrossRef]
- Eigenbrode, S.D.; Jetter, R. Attachment to plant surface waxes by an insect predator. Integr. Comp. Biol. 2002, 42, 1091–1099. [Google Scholar] [CrossRef]
- Adamski, N.M.; Bush, M.S.; Simmonds, J.; Turner, A.S.; Mugford, S.G.; Jones, A.; Findlay, K.; Pedentchouk, N.; von Wettstein-Knowles, P.; Uauy, C. The Inhibitor of wax 1 locus (Iw1) prevents formation of β- and OH-β-diketones in wheat cuticular waxes and maps to a sub-cM interval on chromosome arm 2BS. Plant J. 2013, 74, 989–1002. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, J.; Chai, G.; Li, C.; Hu, Y.; Chen, X.; Wang, Z. Developmental Changes in Composition and Morphology of Cuticular Waxes on Leaves and Spikes of Glossy and Glaucous Wheat (Triticum aestivum L.). Plos ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Wu, H.; Xu, J.; Li, C.; Wang, Y.; Wang, Z. Five Fatty Acyl-Coenzyme A Reductases Are Involved in the Biosynthesis of Primary Alcohols in Aegilops tauschii Leaves. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Wen, M.; Jetter, R. Composition of secondary alcohols, ketones, alkanediols, and ketols in Arabidopsis thaliana cuticular waxes. J. Exp. Bot. 2009, 60, 1811–1821. [Google Scholar] [CrossRef]
- Ji, X.; Jetter, R. Very long chain alkylresorcinols accumulate in the intracuticular wax of rye (Secale cereale L.) leaves near the tissue surface. Phytochemistry 2008, 69, 1197–1207. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.B.; Svelander, C.; Karlsson, G.; Savolainen, O.I. Identification and quantification of even and odd chained 5-n alkylresorcinols, branched chain-alkylresorcinols and methylalkylresorcinols in Quinoa (Chenopodium quinoa). Food Chem. 2017, 220, 344–351. [Google Scholar] [CrossRef]
- Li, Y.; Beisson, F.; Ohlrogge, J.; Pollard, M. Monoacylglycerols are components of root waxes and can be produced in the aerial cuticle by ectopic expression of a suberin-associated acyltransferase. Plant Physiol. 2007, 144, 1267–1277. [Google Scholar] [CrossRef]
- Prinsen, P.; Gutierrez, A.; Faulds, C.B.; del Rio, J.C. Comprehensive Study of Valuable Lipophilic Phytochemicals in Wheat Bran. J. Agric. Food Chem. 2014, 62, 1664–1673. [Google Scholar] [CrossRef]
- Andersson, A.A.M.; Kamal-Eldin, A.; Fras, A.; Boros, D.; Aman, P. Alkylresorcinols in Wheat Varieties in the HEALTHGRAIN Diversity Screen. J. Agric. Food Chem. 2008, 56, 9722–9725. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.B.; Aman, P.; Andersson, R.; Kamal-Eldin, A. Chromatographic analysis of alkylresorcinols and their metabolites. J. Chromatogr. A 2004, 1054, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Gunenc, A.; HadiNezhad, M.; Tamburic-Ilincic, L.; Mayer, P.M.; Hosseinian, F. Effects of region and cultivar on alkylresorcinols content and composition in wheat bran and their antioxidant activity. J. Cereal Sci. 2013, 57, 405–410. [Google Scholar] [CrossRef]
- Shewry, P.R.; Piironen, V.; Lampi, A.-M.; Edelmann, M.; Kariluoto, S.; Nurmi, T.; Fernandez-Orozco, R.; Ravel, C.; Charmet, G.; Andersson, A.A.M.; et al. The HEALTHGRAIN Wheat Diversity Screen: Effects of Genotype and Environment on Phytochemicals and Dietary Fiber Components. J. Agric. Food Chem. 2010, 58, 9291–9298. [Google Scholar] [CrossRef]
- Andersson, A.A.M.; Kamal-Eldin, A.; Aman, P. Effects of Environment and Variety on Alkylresorcinols in Wheat in the HEALTHGRAIN Diversity Screen. J. Agric. Food Chem. 2010, 58, 9299–9305. [Google Scholar] [CrossRef]
- Liu, J.; Hao, Y.; Wang, Z.; Ni, F.; Wang, Y.; Gong, L.; Sun, B.; Wang, J. Identification, Quantification, and Anti-inflammatory Activity of 5-n-Alkylresorcinols from 21 Different Wheat Varieties. J. Agric. Food Chem. 2018, 66, 9241–9247. [Google Scholar] [CrossRef] [PubMed]
- Landberg, R.; Andersson, A.A.M.; Aman, P.; Kamal-Eldin, A. Comparison of GC and colorimetry for the determination of alkylresorcinol homologues in cereal grains and products. Food Chem. 2009, 113, 1363–1369. [Google Scholar] [CrossRef]
- Andersson, A.A.M.; Aman, P.; Wandel, M.; Frolich, W. Alkylresorcinols in wheat and rye flour and bread. J. Food Compos. Anal. 2010, 23, 794–801. [Google Scholar] [CrossRef]
- Dayan, F.E.; Kagan, I.A.; Rimando, A.M. Elucidation of the biosynthetic pathway of the allelochemical sorgoleone using retrobiosynthetic NMR analysis. J. Biol. Chem. 2003, 278, 28607–28611. [Google Scholar] [CrossRef] [PubMed]
- Raharjo, T.J.; Chang, W.T.; Choi, Y.H.; Peltenburg-Looman, A.M.G.; Verpoorte, R. Olivetol as product of a polyketide synthase in Cannabis saliva L. Plant Sci. 2004, 166, 381–385. [Google Scholar] [CrossRef]
- Kozubek, A.; Tyman, J.H.P. Resorcinolic lipids, the natural non-isoprenoid phenolic amphiphiles and their biological activity. Chem. Rev. 1999, 99, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet (Lond. Engl.) 1986, 1, 307–310. [Google Scholar] [CrossRef]
Sample Availability: Samples of wheat seeds are available from the authors for limit time. |
| Samples | AR Homologue (%) | |||||||
|---|---|---|---|---|---|---|---|---|
| AR15:0 | AR 17:0 | AR 19:0 | AR 21:0 | AR 23:0 | AR 25:0 | 17:0/21:0 | 19:0/21:0 | |
| Seed surface | nd | nd | 7.35 | 27.47 | 39.83 | 25.35 | nd | 0.25 |
| C.V. a (%) | nd | nd | 78.20 | 23.07 | 17.50 | 31.20 | nd | 50.45 |
| Whole grain | 1.28 | 5.10 | 33.64 | 48.19 | 9.07 | 2.77 | 0.10 | 0.70 |
| C.V. b (%) | 24.19 | 9.09 | 6.01 | 3.88 | 17.11 | 20.29 | 11.44 | 8.57 |
| AR Homologue | AR Homologue | |||||
|---|---|---|---|---|---|---|
| AR17:0 | AR19:0 | AR21:0 | AR23:0 | AR25:0 | Total ARs | |
| Whole grain | ||||||
| AR 15:0 | 0.423 * | 0.208 | 0.219 | 0.084 | 0.249 | 0.260 |
| AR 17:0 | 0.873 ** | 0.830 ** | 0.571 ** | 0.610 ** | 0.867 ** | |
| AR 19:0 | 0.897 ** | 0.646 ** | 0.611 ** | 0.945 ** | ||
| AR 21:0 | 0.711 ** | 0.679 ** | 0.974 ** | |||
| AR 23:0 | 0.815 ** | 0.765 ** | ||||
| AR 25:0 | 0.730 ** | |||||
| Seed surface | ||||||
| AR 19:0 | 0.917 ** | 0.555 ** | 0.279 | 0.799 ** | ||
| AR 21:0 | 0.754 ** | 0.400 * | 0.932 ** | |||
| AR 23:0 | 0.753 ** | 0.855 ** | ||||
| AR 25:0 | 0.620 ** | |||||
| AR Homologue | AR Homologue | ||||
|---|---|---|---|---|---|
| AR19:0 a | AR 21:0 a | AR 23:0 a | AR 25:0 a | Total Ars a | |
| AR19:0 b | 0.591 ** | 0.713 ** | 0.698 ** | 0.642 ** | 0.825 ** |
| AR 21:0 b | 0.573 ** | 0.682 ** | 0.760 ** | 0.703 ** | 0.813 ** |
| AR 23:0 b | 0.390 * | 0.551 ** | 0.629 ** | 0.440 ** | 0.622 ** |
| AR 25:0 b | 0.534 ** | 0.612 ** | 0.658 ** | 0.498 ** | 0.644 ** |
| Total Ars b | 0.627 ** | 0.750 ** | 0.775 ** | 0.676 ** | 0.863 ** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Gao, X.; Wang, Z. Non-Destructive Determination of Alkylresorcinol (ARs) Content on Wheat Seed Surfaces and Prediction of ARs Content in Whole-Grain Flour. Molecules 2019, 24, 1329. https://doi.org/10.3390/molecules24071329
Wang J, Gao X, Wang Z. Non-Destructive Determination of Alkylresorcinol (ARs) Content on Wheat Seed Surfaces and Prediction of ARs Content in Whole-Grain Flour. Molecules. 2019; 24(7):1329. https://doi.org/10.3390/molecules24071329
Chicago/Turabian StyleWang, Jiahuan, Xin Gao, and Zhonghua Wang. 2019. "Non-Destructive Determination of Alkylresorcinol (ARs) Content on Wheat Seed Surfaces and Prediction of ARs Content in Whole-Grain Flour" Molecules 24, no. 7: 1329. https://doi.org/10.3390/molecules24071329
APA StyleWang, J., Gao, X., & Wang, Z. (2019). Non-Destructive Determination of Alkylresorcinol (ARs) Content on Wheat Seed Surfaces and Prediction of ARs Content in Whole-Grain Flour. Molecules, 24(7), 1329. https://doi.org/10.3390/molecules24071329





