In-Vitro Evaluation of 52 Commercially-Available Essential Oils Against Leishmania amazonensis
Abstract
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Essential Oils
3.2. Parasites
3.3. In-vitro Anti-promastigote Screening
3.4. Mouse Peritoneal Macrophage Cytotoxicity Screening
3.5. In-vitro Intracellular Anti-amastigote Screening
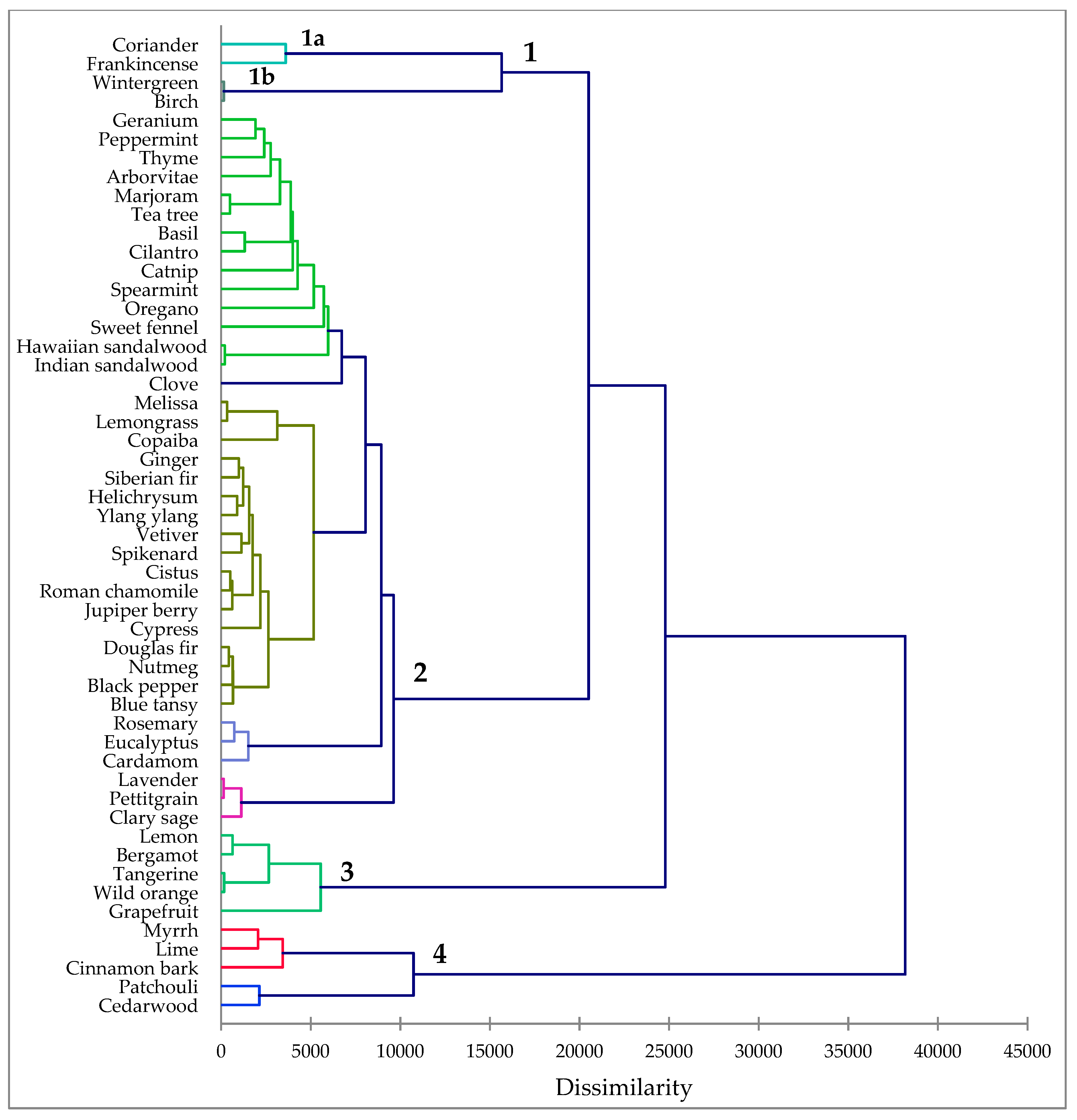
3.6. Hierarchical Cluster Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Torres-Guerrero, E.; Quintanilla-Cedillo, M.R.; Ruiz-Esmenjaud, J.; Arenas, R. Leishmaniasis: A review. F1000Research 2017, 6, 750. [Google Scholar] [CrossRef]
- Alvar, J.; Arana, B. Leishmaniasis, impact and therapeutic needs. In Drug Discovery for Leishmaniasis; Livas, L., Gil, C., Eds.; Royal Society of Chemistry: London, UK, 2018; pp. 3–23. [Google Scholar]
- World Health Organization. Neglected Tropical Diseases. Available online: https://www.who.int/neglected_diseases/diseases/en/ (accessed on 1 March 2019).
- Iqbal, H.; Ishfaq, M.; Wahab, A.; Abbas, M.N.; Ahmad, I.; Rehman, A.; Zakir, M. Therapeutic modalities to combat leishmaniasis, a review. Asian Pacific J. Trop. Dis. 2016, 6, 1–5. [Google Scholar] [CrossRef]
- Hailu, T.; Yimer, M.; Mulu, W.; Abera, B. Challenges in visceral leishmaniasis control and elimination in the developing countries: A review. J. Vector Borne Dis. 2016, 53, 193–198. [Google Scholar] [PubMed]
- Sunyoto, T.; Potet, J.; Boelaert, M. Why miltefosine—a life-saving drug for leishmaniasis—is unavailable to people who need it the most. BMJ Glob. Heal. 2018, 3, e000709. [Google Scholar] [CrossRef]
- Firenzuoli, F.; Jaitak, V.; Horvath, G.; Henri, I.; Bassolé, N.; Setzer, W.N.; Gori, L. Essential Oils: New Perspectives in Human Health and Wellness; Hindawi Publishing Corp.: London, UK, 2014. [Google Scholar]
- Başer, K.H.C.; Buchbauer, G. Handbook of Essential Oils: Science, Technology, and Applications; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar]
- Monzote, L.; Alarcón, O.; Setzer, W.N. Antiprotozoal activity of essential oils. Agric. Conspec. Sci. 2012, 77, 167–175. [Google Scholar]
- Bero, J.; Kpoviessi, S.; Quetin-Leclercq, J. Anti-parasitic activity of essential oils and their active constituents against Plasmodium, Trypanosoma and Leishmania. In Novel Plant Bioresources: Applications in Food, Medicine and Cosmetics; Gurub-Fakim, A., Ed.; John Wiley & Sons Ltd.: Oxford, UK, 2014; pp. 455–470. [Google Scholar]
- Mikus, J.; Harkenthal, M.; Steverding, D.; Reichling, J. In vitro effect of essential oils and isolated mono- and sesquiterpenes on Leishmania major and Trypanosoma brucei. Planta Med. 2000, 66, 366–368. [Google Scholar] [CrossRef]
- Powers, C.N.; Osier, J.L.; McFeeters, R.L.; Brazell, C.B.; Olsen, E.L.; Moriarity, D.M.; Satyal, P.; Setzer, W.N. Antifungal and cytotoxic activities of sixty commercially-available essential oils. Molecules 2018, 23, 1549. [Google Scholar] [CrossRef] [PubMed]
- Orchard, A.; Viljoen, A.; van Vuuren, S. Wound pathogens: Investigating antimicrobial activity of commercial essential oil combinations against reference strains. Chem. Biodivers. 2018, 15, e1800405. [Google Scholar] [CrossRef]
- Orchard, A.; van Vuuren, S.F.; Viljoen, A.M. Commercial essential oil combinations against topical fungal pathogens. Nat. Prod. Commun. 2019, 14, 151–158. [Google Scholar] [CrossRef]
- Serra, E.; Hidalgo-Bastida, L.A.; Verran, J.; Williams, D.; Malic, S. Antifungal activity of commercial essential oils and biocides against Candida albicans. Pathogens 2018, 7, 15. [Google Scholar] [CrossRef]
- Cannas, S.; Usai, D.; Tardugno, R.; Benvenuti, S.; Pellati, F.; Zanetti, S.; Molicotti, P.; Cannas, S.; Usai, D.; Tardugno, R.; et al. Chemical composition, cytotoxicity, antimicrobial and antifungal activity of several essential oils. Nat. Prod. Res. 2016, 30, 332–339. [Google Scholar] [CrossRef]
- Andrade, M.A.; Azevedo, S.; Motta, F.N.; Lucília, M.; Silva, C.L.; De Santana, J.M.; Bastos, I.M.D. Essential oils: In vitro activity against Leishmania amazonensis, cytotoxicity and chemical composition. BMC Complement. Altern. Med. 2016, 16, 444. [Google Scholar] [CrossRef]
- Fujisaki, R.; Kamei, K.; Yamamura, M.; Nishiya, H.; Inouye, S.; Takahashi, M.; Abe, S. In vitro and in vivo anti-plasmodial activity of essential oils, including himokitiol. Southeast Asian J. Trop. Med. Public Health 2012, 43, 270–279. [Google Scholar] [PubMed]
- Rodrigues, K.A.D.F.; Amorim, L.V.; Dias, C.N.; Moraes, D.F.C.; Carneiro, S.M.P.; Carvalho, F.A.D.A. Syzygium cumini (L.) Skeels essential oil and its major constituent α-pinene exhibit anti-Leishmania activity through immunomodulation in vitro. J. Ethnopharmacol. 2015, 160, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Rondon, F.C.M.; Bevilaqua, C.M.L.; Accioly, M.P.; de Morais, S.M.; de Andrade-Júnior, H.F.; de Carvalho, C.A.; Lima, J.C.; Magalhães, H.C.R. In vitro efficacy of Coriandrum sativum, Lippia sidoides and Copaifera reticulata against Leishmania chagasi. Rev. Bras. Parasitol. Veterinária 2012, 21, 185–191. [Google Scholar] [CrossRef]
- Rosa, M.D.S.S.; Mendonça-Filho, R.R.; Bizzo, H.R.; Rodrigues, I.D.A.; Soares, R.M.A.; Souto-Padrón, T.; Alviano, C.S.; Lopes, A.H.C.S. Antileishmanial activity of a linalool-rich essential oil from Croton cajucara. Antimicrob. Agents Chemother. 2003, 47, 1895–1901. [Google Scholar] [CrossRef]
- Santos, A.O.; Ueda-Nakamura, T.; Dias Filho, B.P.; Veiga Junior, V.F.; Pinto, A.C.; Nakamura, C.V. Effect of Brazilian copaiba oils on Leishmania amazonensis. J. Ethnopharmacol. 2008, 120, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Soares, D.C.; Portella, N.A.; Ramos, M.F.D.S.; Siani, A.C.; Saraiva, E.M. trans-β-Caryophyllene: An effective antileishmanial compound found in commercial copaiba oil (Copaifera spp.). Evidence-Based Complement. Altern. Med. 2013, 2013, 761323. [Google Scholar] [CrossRef] [PubMed]
- do Carmo, D.F.M.; Amaral, A.C.F.; Machado, G.M.C.; Leon, L.L.; Silva, J.R.D.A. Chemical and biological analyses of the essential oils and main constituents of Piper species. Molecules 2012, 17, 1819–1829. [Google Scholar] [CrossRef]
- Essid, R.; Zahra, F.; Msaada, K.; Sghair, I.; Hammami, M.; Bouratbine, A.; Aoun, K.; Limam, F. Antileishmanial and cytotoxic potential of essential oils from medicinal plants in Northern Tunisia. Ind. Crop. Prod. 2015, 77, 795–802. [Google Scholar] [CrossRef]
- Arévalo, Y.; Robledo, S.; Muñoz, D.L.; Granados-Falla, D.; Cuca, L.E.; Delgado, G. Evaluación in vitro de la actividad de aceites esenciales de plantas colombianas sobre Leishmania braziliensis. Rev. Colomb. Ciencias Químico-Farm. 2009, 38, 131–141. [Google Scholar]
- Satyal, P.; Jones, T.H.; Lopez, E.M.; McFeeters, R.L.; Ali, N.A.A.; Mansi, I.; Al-Kaf, A.G.; Setzer, W.N. Chemotypic characterization and biological activity of Rosmarinus officinalis. Foods (Basel, Switzerland) 2017, 6, 20. [Google Scholar] [CrossRef]
- Machado, M.; Pires, P.; Dinis, A.M.; Santos-Rosa, M.; Alves, V.; Salgueiro, L.; Cavaleiro, C. Monoterpenic aldehydes as potential anti-Leishmania agents: Activity of Cymbopogon citratus and citral on L. infantum, L. tropica and L. major. Exp. Parasitol. 2012, 130, 223–231. [Google Scholar] [CrossRef]
- Setzer, W.N.; Schmidt, J.M.; Eiter, L.C.; Haber, W.A. The leaf oil composition of Zanthoxylum fagara (L.) Sarg. from Monteverde, Costa Rica, and its biological activities. J. Essent. Oil Res. 2005, 17, 333–335. [Google Scholar] [CrossRef]
- Wright, B.S.; Bansal, A.; Moriarity, D.M.; Takaku, S.; Setzer, W.N. Cytotoxic leaf essential oils from Neotropical Lauraceae: Synergistic effects of essential oil components. Nat. Prod. Commun. 2007, 2, 1241–1244. [Google Scholar] [CrossRef]
- Bayala, B.; Bassole, I.H.N.; Maqdasy, S.; Baron, S.; Simpore, J.; Lobaccaro, J.-M.A. Cymbopogon citratus and Cymbopogon giganteus essential oils have cytotoxic effects on tumor cell cultures. Identification of citral as a new putative anti-proliferative molecule. Biochimie 2018, 153, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Sharopov, F.S.; Wink, M.; Khalifaev, D.R.; Zhang, H.; Dosoky, N.S.; Setzer, W.N. Composition and bioactivity of the essential oil of Melissa officinalis L. growing wild in Tajikistan. Int. J. Tradit. Nat. Med. 2013, 2, 86–96. [Google Scholar]
- Santin, M.R.; dos Santos, A.O.; Nakamura, C.V.; Filho, B.P.D.; Ferreira, I.C.P.; Ferreira, P.; Ueda-Nakamura, T. In vitro activity of the essential oil of Cymbopogon citratus and its major component (citral) on Leishmania amazonensis. Parasitol. Res. 2009, 105, 1489–1496. [Google Scholar] [CrossRef] [PubMed]
- Monzote, L.; Piñón, A.; Setzer, W. Antileishmanial potential of tropical rainforest plant extracts. Medicines 2014, 1, 32–55. [Google Scholar] [CrossRef]
- Pastor, J.; García, M.; Steinbauer, S.; Setzer, W.N.; Scull, R.; Gille, L.; Monzote, L. Combinations of ascaridole, carvacrol, and caryophyllene oxide against Leishmania. Acta Trop. 2015, 145. [Google Scholar] [CrossRef] [PubMed]
- Monzote, L.; Jiménez, J.; Cuesta-Rubio, O.; Márquez, I.; Gutiérrez, Y.; da Rocha, C.Q.; Marchi, M.; Setzer, W.N.; Vilegas, W. In vitro assessment of plants growing in Cuba belonging to Solanaceae family against Leishmania amazonensis. Phyther. Res. 2016, 30. [Google Scholar]
- Sladowski, D.; Steer, S.J.; Clothier, R.H.; Balls, M. An improved MTT assay. J. Immunol. Methods 1993, 157, 203–207. [Google Scholar] [CrossRef]
- Dutta, A.; Bandyopadhyay, S.; Mandal, C.; Chatterjee, M. Development of a modified MTT assay for screening antimonial resistant field isolates of Indian visceral leishmaniasis. Parasitol. Int. 2005, 54, 119–122. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the essential oils reported in this work are available from the authors. |
| Essential Oil | Commercial Source | IC50 ± SD (μg/mL) Promastigotes L. amazonensis | CC50 ± SD (μg/mL) Macrophage from BALB/c mice | Selectivity Index (SI) | Comments |
|---|---|---|---|---|---|
| Abies sibirica Ledeb. (Siberian fir) | dōTERRA | 58.2 ± 8.5 | 42.5 ± 2.4 | 1 | Unspecific |
| Anthemis nobilis L. (Roman chamomile) | dōTERRA | 27.9 ± 4.5 | 85.7 ± 6.0 | 3 | Unspecific |
| Betula lenta L. (birch) | dōTERRA | 32.2 ± 6.6 | 136.8 ± 9.2 | 4 | Unspecific |
| BoswelliaRoxb. ex Colebr. spp. (frankincense) | dōTERRA | <12.5 | 59.3 ± 1.8 | 5 | Active/Follow up |
| Cananga odorata (Lam.) Hook. f. & Thomson (ylang ylang) | dōTERRA | 36.6 ± 3.9 | 55.6 ± 0.5 | 2 | Unspecific |
| Cinnamomum zeylanicum Blume (cinnamon bark) | dōTERRA | <12.5 | <12.5 | - | Too Toxic |
| Cistus ladanifer L. (cistus) | Albert Vielle | 19.2 ± 4.6 | 56.9 ± 8.1 | 3 | Unspecific |
| Citrus aurantium L. (pettitgrain) | dōTERRA | 56.9 ± 1.8 | 77.4 ± 0.9 | 1 | Unspecific |
| Citrus aurantium L. (wild orange) | dōTERRA | 34.8 ± 2.2 | 35.2 ± 1.7 | 1 | Unspecific |
| Citrus aurantifolia Swingle (lime) | dōTERRA | <12.5 | <12.5 | - | Too toxic |
| Citrus × bergamia Risso & Poit. (bergamot) | dōTERRA | 59.2 ± 6.3 | 44.9 ± 1.6 | 1 | Unspecific |
| Citrus limon (L.) Osbeck (lemon) | dōTERRA | 71.2 ± 1.9 | 71.1 ± 1.6 | 1 | Unspecific |
| Citrus × paradisi Macfad. (grapefruit) | dōTERRA | 35.5 ± 4.4 | <12.5 | - | Too Toxic |
| Citrus reticulata Blanco (tangerine) | dōTERRA | 70.7 ± 7.8 | 54.3 ± 1.0 | 1 | Unspecific |
| Commiphora myrrha (T. Nees) Engl. (myrrh) | dōTERRA | <12.5 | <12.5 | - | Too Toxic |
| Coriandrum sativum L. (coriander) | dōTERRA | <12.5 | 141.7 ± 4.1 | >8 | Active/Follow up |
| Coriandrum sativum L. (cilantro) | dōTERRA | 34.4 ± 4.8 | 45.9 ± 1.3 | 1 | Unspecific |
| Copaifera L. spp. (copaiba) | dōTERRA | 17.2 ± 0.1 | 23.3 ± 7.1 | 1 | Unspecific |
| Cupressus sempervirens L. (cypress) | Améo | 40.0 ± 2.3 | 27.2 ± 0.7 | 1 | Unspecific |
| Cymbopogon flexuosus (Nees) Will. Watson (lemongrass) | dōTERRA | 27.8 ± 0.4 | 30.0 ± 2.0 | 1 | Unspecific |
| Elettaria cardamomum (L.) Maton (cardamom) | dōTERRA | 109.5 ± 1.3 | 60.5 ± 6.0 | 1 | Unspecific |
| Eugenia caryophyllata Thunb. (syn. Syzygium aromaticum (L.) Merr. & L.M. Perry) (clove) | dōTERRA | >200 | 143.1 ± 11.9 | - | Inactive |
| Eucalyptus radiata Sieber ex DC. (eucalyptus) | dōTERRA | 164.7 ± 8.3 | 100.2 ± 8.4 | 1 | Unspecific |
| Foeniculum vulgare Mill. (sweet fennel) | dōTERRA | >200 | 66.9 ± 7.9 | - | Inactive |
| Gualtheria fragrantissima Wall.(wintergreen) | dōTERRA | 20.7 ± 1.6 | 135.4 ± 5.7 | 7 | Active/Follow up |
| Helichrysum italicum G. Don f. (helichrysum) | Améo | 42.8 ± 2.2 | 55.2 ± 2.4 | 1 | Unspecific |
| Juniperus communis L. (juniper berry) | Améo | 27.4 ± 0.5 | 103.0 ± 9.3 | 4 | Unspecific |
| Juniperus virginiana L. (cedarwood) | dōTERRA | 53.2 ± 8.4 | <12.5 | - | Too Toxic |
| Lavandula angustifolia Mill. (lavender) | Améo | 70.7 ± 5.0 | 82.3 ± 2.4 | 1 | Unspecific |
| Melaleuca alternifolia Cheel (tea tree) | dōTERRA | 70.7 ± 6.2 | 42.5 ± 1.4 | 1 | Unspecific |
| Melissa officinalis L. (melissa) | dōTERRA | 24.6 ± 0.7 | 37.3 ± 1.4 | 2 | Unspecific |
| Mentha piperita L. (peppermint) | dōTERRA | 172.8 ± 4.1 | 67.3 ± 4.4 | 0 | Unspecific |
| Mentha spicata L. (spearmint) | dōTERRA | 79.8 ± 3.0 | 90.4 ± 5.6 | 1 | Unspecific |
| Myristica fragrans Houtt. (nutmeg) | Améo | 133.5 ± 2.6 | 40.0 ± 1.1 | 0 | Unspecific |
| Nardostachys jatamansi (D. Don) DC. (spikenard) | dōTERRA | 49.8 ± 1.4 | 22.1 ± 4.0 | 0 | Unspecific |
| Nepeta cataria L. (catnip) | Mountain Rose | 54.0 ± 4.4 | 81.6 ± 0.8 | 2 | Unspecific |
| Ocimum basilicum L. (basil) | dōTERRA | >200 | 69.1 ± 9.0 | - | Inactive |
| Origanum majorana L. (marjoram) | dōTERRA | >200 | 25.7 ± 1.3 | - | Inactive |
| Origanum vulgare L. (oregano) | dōTERRA | >200 | 66.5 ± 0.9 | - | Inactive |
| Pelargonium graveolens L'Hér. ex Aiton (geranium) | Améo | >200 | 57.4 ± 3.6 | - | Inactive |
| Piper nigrum L. (black pepper) | dōTERRA | 57.7 ± 3.7 | 35.6 ± 5.7 | 1 | Unspecific |
| Pogostemon cablin (Blanco) Benth. (patchouli) | Améo | 68.7 ± 7.8 | <12.5 | - | Too Toxic |
| Pseudotsuga menziesii (Mirb.) Franco (Douglas fir) | dōTERRA | 82.5 ± 4.5 | 37.7 ± 3.2 | 0 | Unspecific |
| Rosmarinus officinalis L. (rosemary) | dōTERRA | 89.7 ± 2.0 | 83.4 ± 7.3 | 1 | Unspecific |
| Santalum album L. (Indian sandalwood) | dōTERRA | 105.5 ± 6.0 | 29.9 ± 6.3 | 0 | Unspecific |
| Santalum paniculatum Hook. & Arn. (Hawaiian sandalwood) | dōTERRA | 43.1 ± 2.2 | 25.9 ± 5.3 | 1 | Unspecific |
| Salvia sclarea L. (clary sage) | Améo | >200 | 58.6 ± 9.0 | - | Inactive |
| Tanacetum annuum L. (blue tansy) | dōTERRA | 52.2 ± 2.8 | 36.6 ± 5.8 | 1 | Unspecific |
| Thuja plicata Donn ex D. Don (arborvitae) | dōTERRA | 67.1 ± 3.1 | 61.9 ± 6.1 | 1 | Unspecific |
| Thymus vulgaris L. (thyme) | dōTERRA | >200 | 30.5 ± 5.5 | - | Inactive |
| Vetiveria zizanioides (L.) Nash (syn. Chrysopogon zizanioides (L.) Roberty) (vetiver) | dōTERRA | 19.0 ± 3.3 | 31.7 ± 2.8 | 2 | Unspecific |
| Zingiber officinale Roscoe (ginger) | dōTERRA | 39.9 ± 3.4 | 58.3 ± 4.7 | 1 | Unspecific |
| Pentamidine | 0.37 ± 0.01 | 11.7 ± 1.7 | 31 | Active |
| Essential Oil | IC50 ± SD (µg/mL) | SI |
|---|---|---|
| Boswellia spp. (frankincense) | 22.1 ± 4.2 | 2 |
| Coriandrum sativum (coriander) | 19.1 ± 0.7 | 7 |
| Gualtheria fragrantissima (wintergreen) | 22.2 ± 3.5 | 6 |
| Pentamidine | 1.3 ± 0.1 | 9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monzote, L.; Herrera, I.; Satyal, P.; Setzer, W.N. In-Vitro Evaluation of 52 Commercially-Available Essential Oils Against Leishmania amazonensis. Molecules 2019, 24, 1248. https://doi.org/10.3390/molecules24071248
Monzote L, Herrera I, Satyal P, Setzer WN. In-Vitro Evaluation of 52 Commercially-Available Essential Oils Against Leishmania amazonensis. Molecules. 2019; 24(7):1248. https://doi.org/10.3390/molecules24071248
Chicago/Turabian StyleMonzote, Lianet, Isabel Herrera, Prabodh Satyal, and William N. Setzer. 2019. "In-Vitro Evaluation of 52 Commercially-Available Essential Oils Against Leishmania amazonensis" Molecules 24, no. 7: 1248. https://doi.org/10.3390/molecules24071248
APA StyleMonzote, L., Herrera, I., Satyal, P., & Setzer, W. N. (2019). In-Vitro Evaluation of 52 Commercially-Available Essential Oils Against Leishmania amazonensis. Molecules, 24(7), 1248. https://doi.org/10.3390/molecules24071248









