Neuroprotective Effects of Ginsenosides against Cerebral Ischemia
Abstract
1. Introduction
2. Results
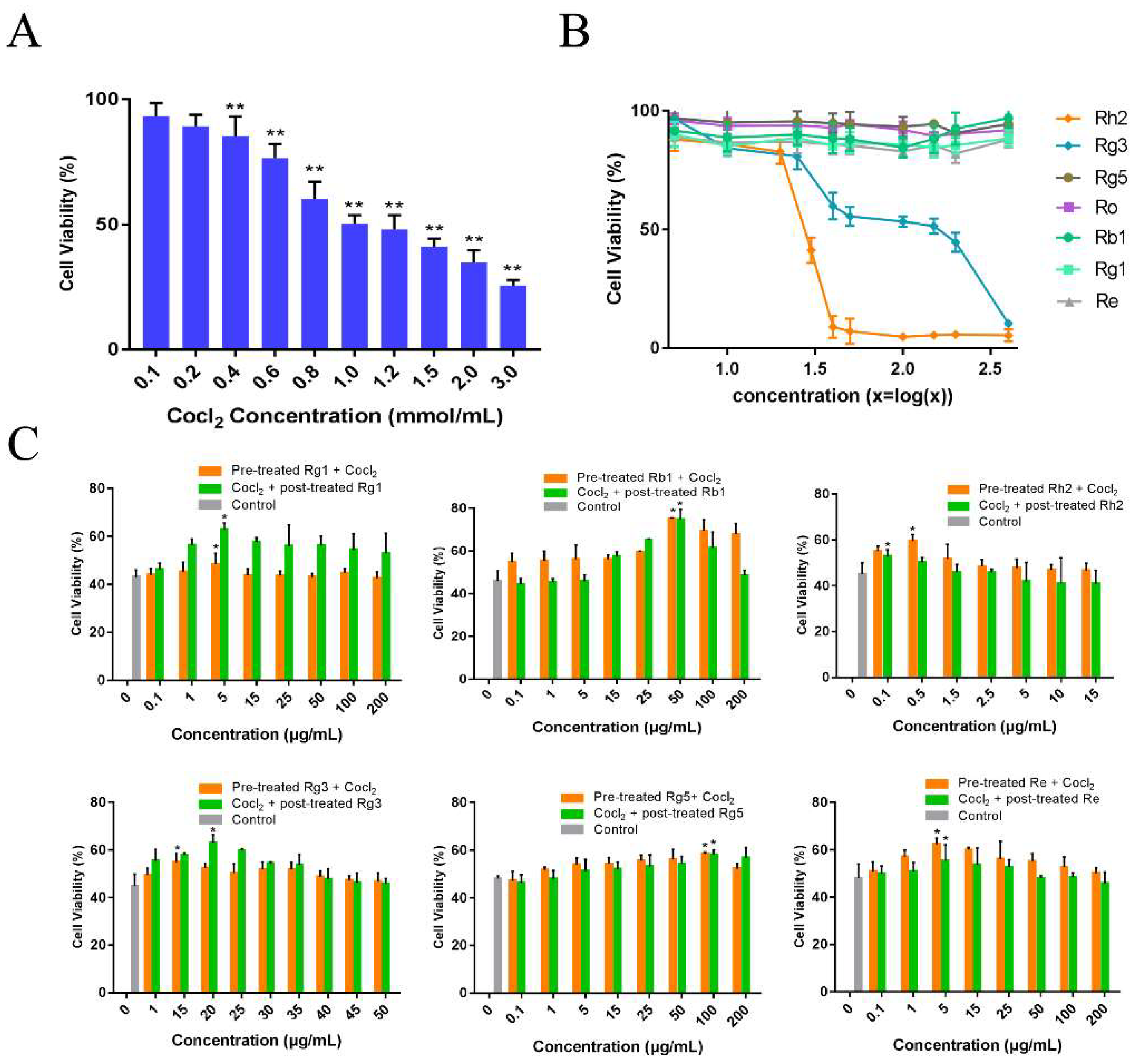
2.1. Ginsenosides Attenuated CoCl2-Induced Cytotoxicity of PC12 Cells
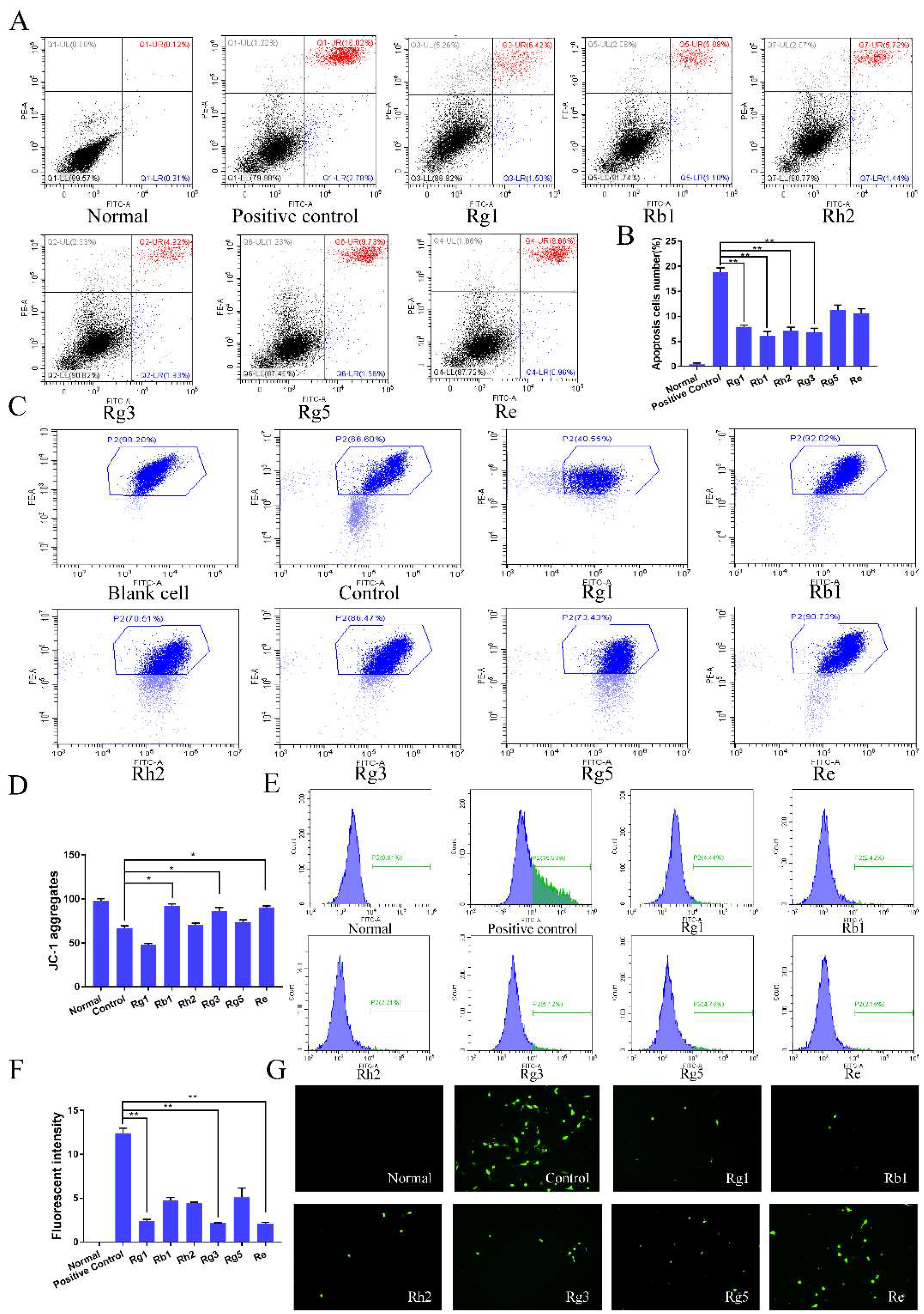
2.2. Ginsenosides Reduced Neuronal Apoptosis, ROS Level and Increased Mitochondrial Membrane Potential of PC12 Cells
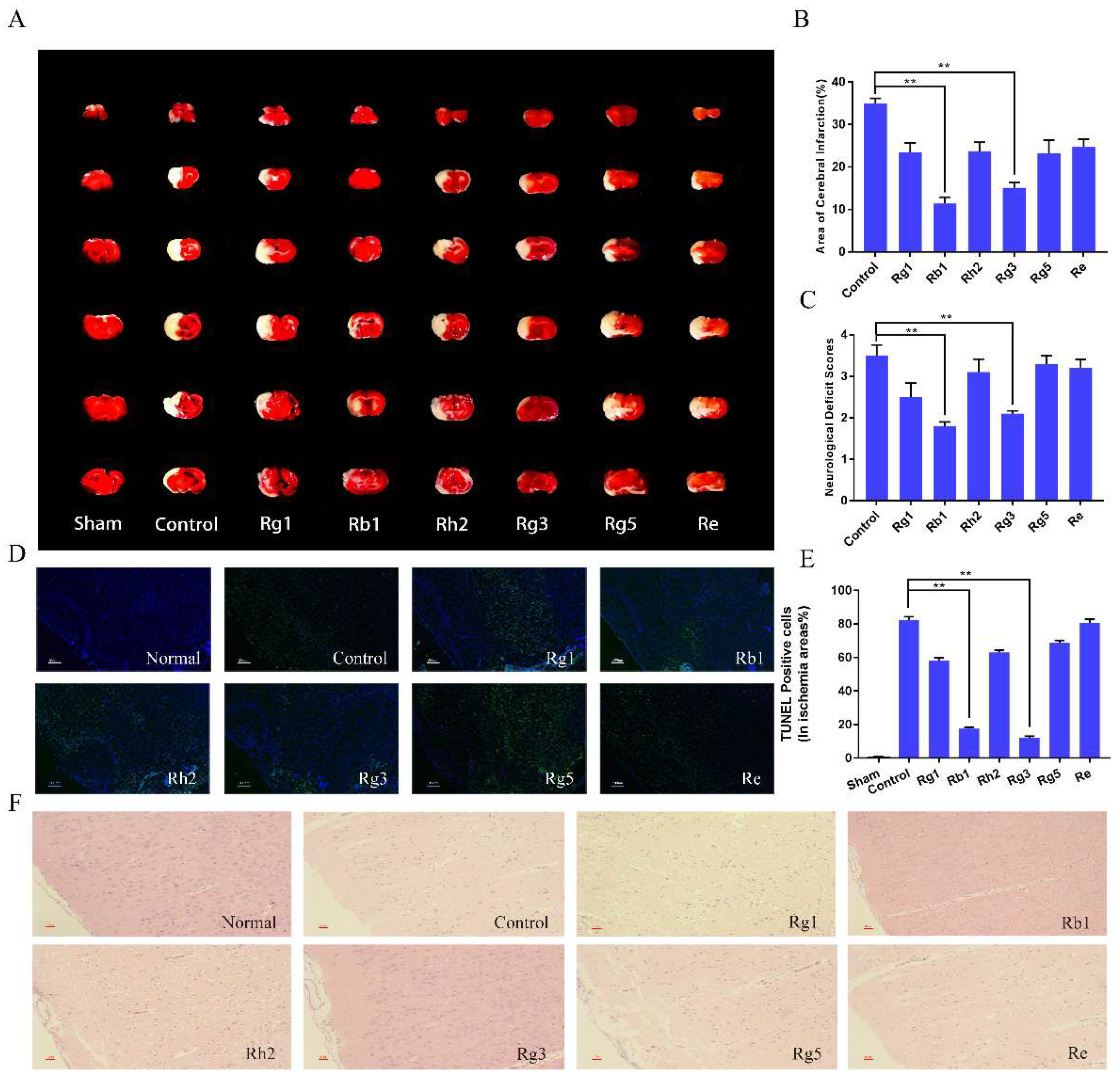
2.3. Ginsenosides Attenuated both the Cerebral Ischemic Injury and the Neurological Deficits Caused by I/S
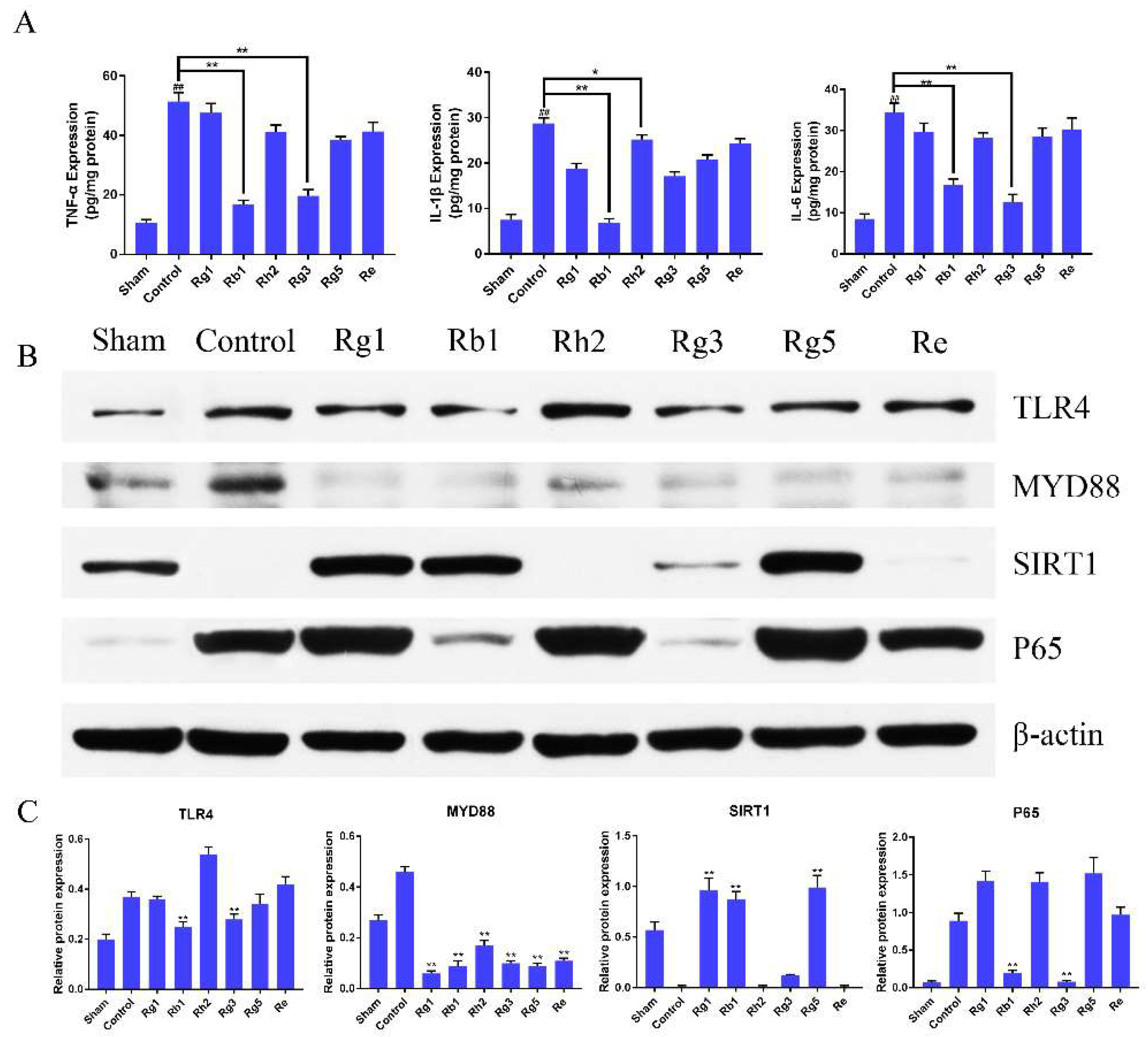
2.4. Ginsenosides Reduced the Level of TNF-a, IL-6, and IL-1β in Penumbra Tissue of I/R Rats
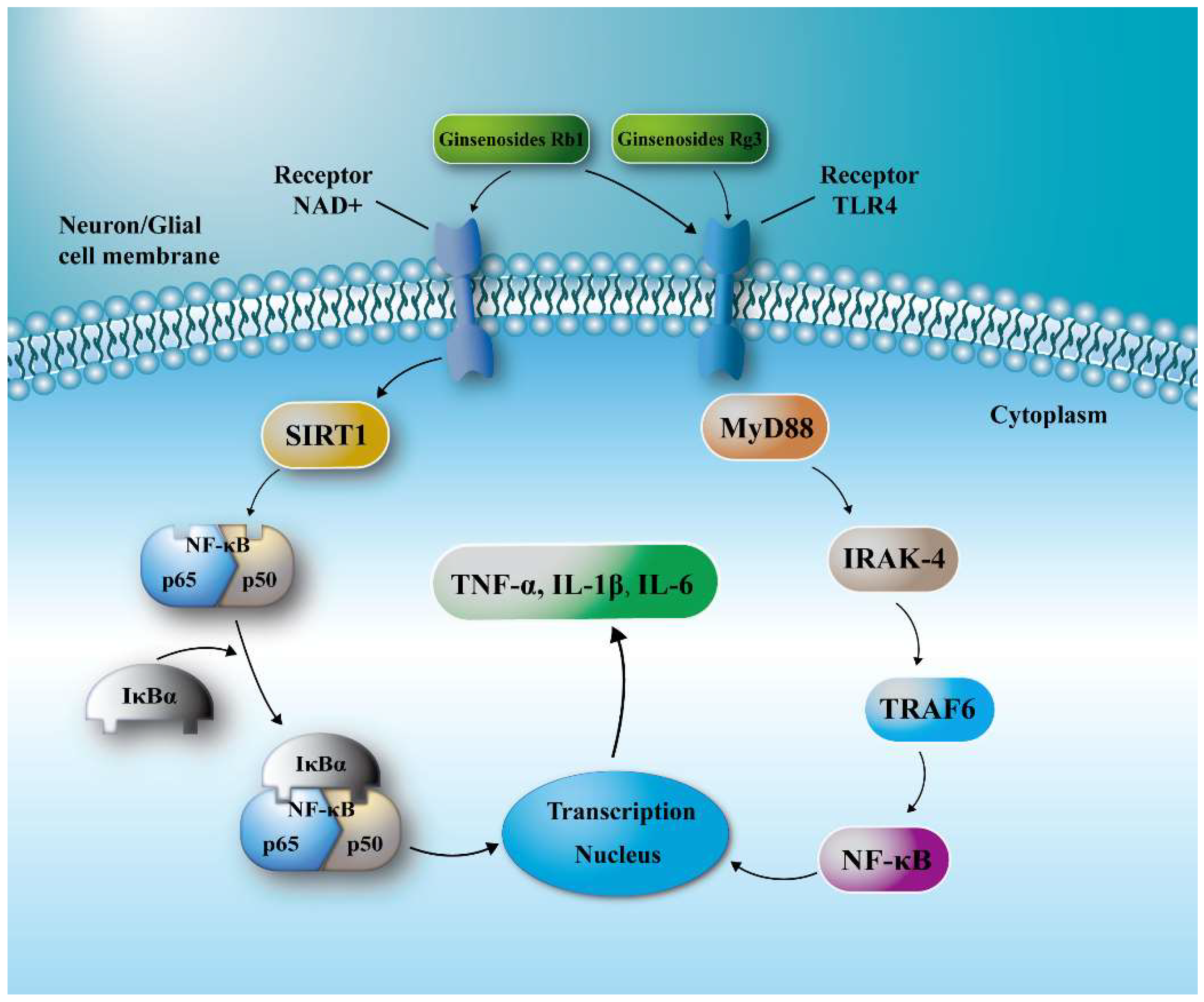
2.5. Ginsenosides Inhibited TLR4/MyD88 and SIRT1 Activation Signaling Pathway in Penumbra Tissue
3. Discussion
4. Materials and Methods
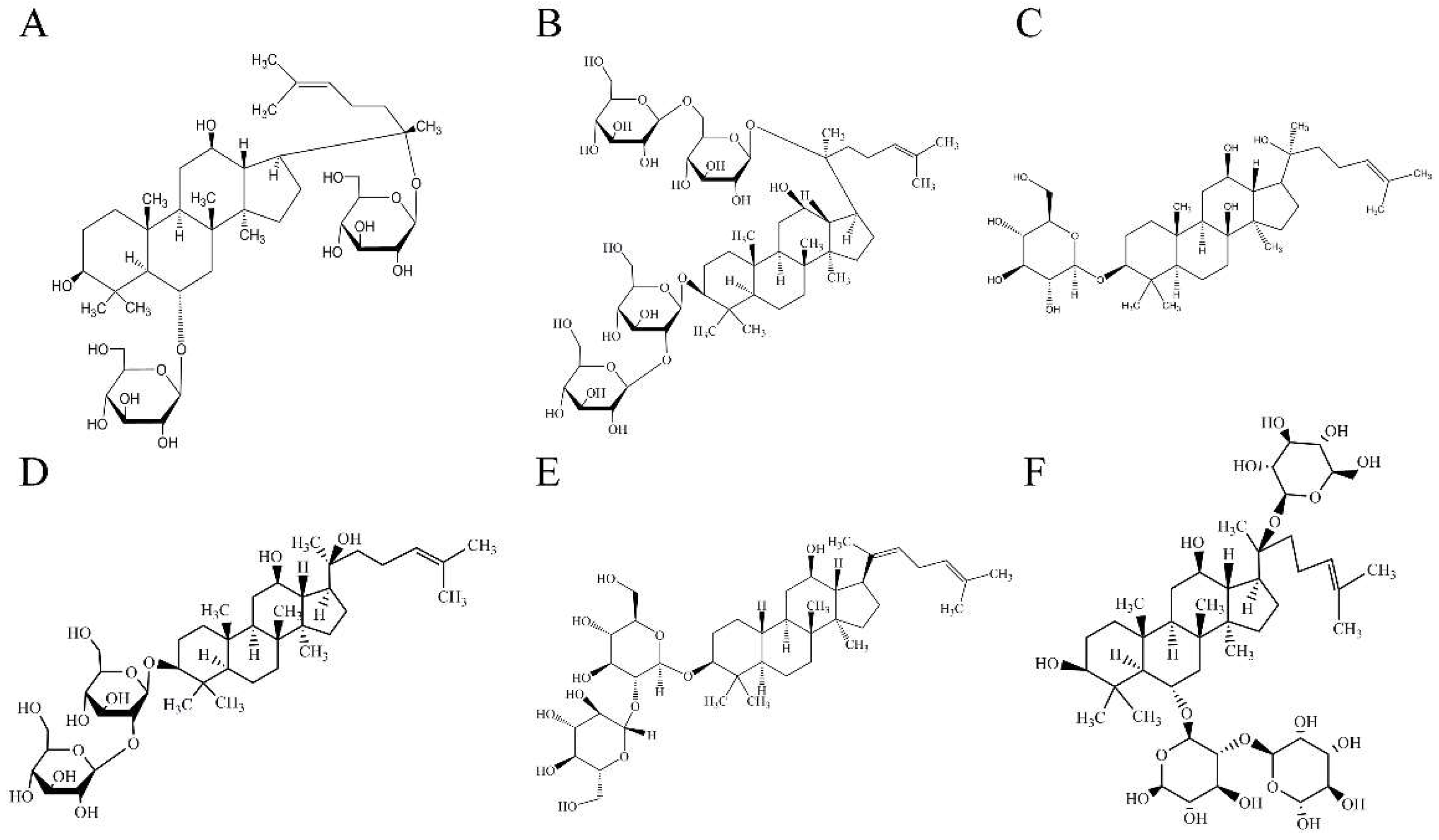
4.1. Materials
4.2. Cytotoxicity of Ginsenosides
4.3. Effect of Ginsenosides on PC12 Cells Injury Induced by CoCl2
4.4. Establishment of Middle Cerebral Artery Occlusion (MCAO) Rat Model
4.5. Effect of Ginsenosides on MCAO Rats
4.6. Measurement of Cytokines by ELISA
4.7. Western Blot Analysis
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Moskowitz, M.A.; Lo, E.H.; Iadecola, C. The Science of Stroke: Mechanisms in Search of Treatments. Neuron 2010, 67, 181–198. [Google Scholar] [CrossRef] [PubMed]
- Danton, G.H.; Dietrich, W.D. Inflammatory mechanisms after ischemia and stroke. J. Neuropath. Exp. Neur. 2003, 62, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Sahota, P.; Savitz, S.I. Investigational Therapies for Ischemic Stroke: Neuroprotection and Neurorecovery. Neurotherapeutics 2011, 8, 434–451. [Google Scholar] [CrossRef] [PubMed]
- Mattiasson, G.; Shamloo, M.; Gido, G.; Mathi, K.; Tomasevic, G.; Yi, S.L.; Warden, C.H.; Castilho, R.F.; Melcher, T.; Gonzalez-Zulueta, M.; et al. Uncoupling protein-2 prevents neuronal death and diminishes brain dysfunction after stroke and brain trauma. Nat. Med. 2003, 9, 1062–1068. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.Y.; Lee, J.M.; Shin, H.S.; Park, S.Y.; Yang, J.E.; Cho, S.K.; Yi, T.H. Anti-Cancer Effect of Ginsenoside F2 against Glioblastoma Multiforme in Xenograft Model in SD Rats. J. Ginseng. Res. 2012, 36, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Tuttolomondo, A.; Di Raimondo, D.; Pecoraro, R.; Casuccio, A.; Butta, C.; Della Corte, V.; Guggino, G.; Arnao, V.; Simonetta, I.; Pinto, A. Peripheral Frequency of Cd4 + Cd28-Cells in Acute Ischemic Stroke: Relationship with Stroke Subtype and Severity Markers. J. Hypertens 2015, 33, E445. [Google Scholar] [CrossRef] [PubMed]
- Tuttolomondo, A.; Di Sciacca, R.; Di Raimondo, D.; Pedone, C.; La Placa, S.; Pinto, A.; Licata, G. Effects of clinical and laboratory variables and of pretreatment with cardiovascular drugs in acute ischaemic stroke: A retrospective chart review from the GIFA study. Int. J. Cardiol. 2011, 151, 318–322. [Google Scholar] [CrossRef] [PubMed]
- Licata, G.; Tuttolomondo, A.; Corrao, S.; Di Raimondo, D.; Fernandez, P.; Caruso, C.; Avellone, G.; Pinto, A. Immunoinflammatory activation during the acute phase of lacunar and non-lacunar ischemic stroke: Association with time of onset and diabetic state. Int. J. Immunopath. Pharmacol. 2006, 19, 639–646. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.H.; Lee, Y.G.; Park, T.Y.; Kim, H.B.; Rhee, M.H.; Cho, J.Y. Ginsenoside Rp1, a ginsenoside derivative, blocks lipopolysaccharide-induced interleukin-1beta production via suppression of the NF-kappaB pathway. Planta Med. 2009, 75, 321–326. [Google Scholar] [CrossRef]
- Wang, Y.J.; Chen, G.; Yu, X.D.; Li, Y.C.; Zhang, L.; He, Z.Z.; Zhang, N.N.; Yang, X.P.; Zhao, Y.S.; Li, N.; et al. Salvianolic Acid B Ameliorates Cerebral Ischemia/Reperfusion Injury Through Inhibiting TLR4/MyD88 Signaling Pathway. Inflammation 2016, 39, 1503–1513. [Google Scholar] [CrossRef]
- Xu, S.X.; Zhong, A.Q.; Ma, H.N.; Li, D.; Hu, Y.; Xu, Y.Z.; Zhang, J.P. Neuroprotective effect of salvianolic acid B against cerebral ischemic injury in rats via the CD40/NF-kappa B pathway associated with suppression of platelets activation and neuroinflammation. Brain Res. 2017, 1661, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.M.; Yao, Q.Z.; Chen, C.Y. Ginseng Compounds: An Update on their Molecular Mechanisms and Medical Applications. Curr. Vasc. Pharmacol. 2009, 7, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Attele, A.S.; Wu, J.A.; Yuan, C.S. Ginseng pharmacology-Multiple constituents and multiple actions. Biochem. Pharmacol. 1999, 58, 1685–1693. [Google Scholar] [CrossRef]
- Xie, W.J.; Zhou, P.; Sun, Y.F.; Meng, X.B.; Dai, Z.R.; Sun, G.B.; Sun, X.B. Protective Effects and Target Network Analysis of Ginsenoside Rg1 in Cerebral Ischemia and Reperfusion Injury: A Comprehensive Overview of Experimental Studies. Cells-Basel 2018, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xu, Q.Q.; Shan, C.S.; Shi, Y.H.; Wang, Y.; Zheng, G.Q. Combined Use of Emodin and Ginsenoside Rb1 Exerts Synergistic Neuroprotection in Cerebral Ischemia/Reperfusion Rats. Front Pharmacol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Han, J.Y.; Cui, Y.C.; Pan, C.S.; Yan, L.; Hu, B.H.; Chang, X.; Liu, Y.Y.; Sun, K.; Li, Q.; Fan, J.Y. Ginsenoside Rb1 protects against ischemia/reperfusion-induced myocardial injury via energy metabolism regulation mediated by RhoA signaling pathway. Faseb J. 2017, 31. [Google Scholar]
- Liu, A.; Zhu, W.; Sun, L.; Han, G.; Liu, H.; Chen, Z.; Zhuang, L.; Jiang, W.; Xue, X. Ginsenoside Rb1 administration attenuates focal cerebral ischemic reperfusion injury through inhibition of HMGB1 and inflammation signals. Experim. Ther. Med. 2018, 16, 3020–3026. [Google Scholar] [CrossRef]
- Ling, C.L.; Liang, J.M.; Zhang, C.; Li, R.X.; Mou, Q.Q.; Qin, J.; Li, X.F.; Wang, J.X. Synergistic effects of Salvianolic Acid B and Puerarin on cerebral ischemia reperfusion injury. Molecules 2018, 23, 3. [Google Scholar] [CrossRef]
- Zhang, C.; Ling, C.L.; Pang, L.; Wang, Q.; Liu, J.X.; Wang, B.S.; Liang, J.M.; Guo, Y.Z.; Qin, J.; Wang, J.X. Direct Macromolecular Drug Delivery to Cerebral Ischemia Area using Neutrophil-Mediated Nanoparticles. Theranostics 2017, 7, 3260–3275. [Google Scholar] [CrossRef]
- Zou, W.G.; Yan, M.D.; Xu, W.J.; Huo, H.R.; Sun, L.Y.; Zheng, Z.C.; Liu, X.Y. Cobalt chloride induces PC12 cells apoptosis through reactive oxygen species and accompanied by AP-1 activation. J. Neurosci. Res. 2001, 64, 646–653. [Google Scholar] [CrossRef]
- Morimoto, Y.; Morimoto, Y.; Nishihira, J.; Kemmotsu, O.; Shibano, T.; Gando, S.; Shikama, H. Pentobarbital inhibits apoptosis in neuronal cells. Crit. Care. Med. 2000, 28, 1899–1904. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Wang, Y.H.; Zhang, X.Y.; Peng, T.; Li, Y.Q.; Zhang, Y. Protective effect of ginsenoside R0 on anoxic and oxidative damage In vitro. Biomol. Ther. 2012, 20, 544–549. [Google Scholar] [CrossRef] [PubMed]
- Kou, D.Q.; Jiang, Y.L.; Qin, J.H.; Huang, Y.H. Magnolol attenuates the inflammation and apoptosis through the activation of SIRT1 in experimental stroke rats. Pharmacol. Rep. 2017, 69, 642–647. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhang, X.J.; Zhang, C.; Bai, X.; Zhang, J.; Zhao, X.M.; Chen, L.Y.; Wang, L.N.; Zhu, C.H.; Cui, L.L.; et al. Nobiletin promotes antioxidant and anti-inflammatory responses and elicits protection against ischemic stroke in vivo. Brain Res. 2016, 1636, 130–141. [Google Scholar] [CrossRef] [PubMed]
- Swanson, R.A.; Morton, M.T.; Tsaowu, G.; Savalos, R.A.; Davidson, C.; Sharp, F.R. A semiautomated method for measuring brain infarct volume. J. Cerebr Blood F Met. 1990, 10, 290–293. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Jiang, Y.; Wu, L.; Lu, T.; Xu, G.; Liu, X. Suppression of local inflammation contributes to the neuroprotective effect of ginsenoside Rb1 in rats with cerebral ischemia. Neuroscience 2012, 202, 342–351. [Google Scholar] [CrossRef]
- Li, Z.Y.; Qi, H.; Xuan, Z.; Yan, Y.; Li, L.Y.; Hou, Y.H.; He, F.N.; Yong, Z.; Jing, Z. Foxo1-mediated inflammatory response after cerebral hemorrhage in rats. Neurosci. Lett. 2016, 629, 131–136. [Google Scholar] [CrossRef]
- Gao, M.; Chen, L.; Yu, H.; Sun, Q.; Kou, J.; Yu, B. Diosgenin down-regulates NF-kappaB p65/p50 and p38MAPK pathways and attenuates acute lung injury induced by lipopolysaccharide in mice. Int. Immunol. 2013, 15, 240–245. [Google Scholar]
- Jin, A.; Li, X.; Zhu, Y.Y.; Yu, H.Y.; Pi, H.F.; Zhang, P.; Ruan, H.L. Four new compounds from the bulbs of Lycoris aurea with neuroprotective effects against CoCl2 and H2O2-induced SH-SY5Y cell injuries. Arch Pharm. Res. 2014, 37, 315–323. [Google Scholar] [CrossRef]
- De Nardo, D.; Latz, E. NLRP3 inflammasomes link inflammation and metabolic disease. Trends Immunol. 2011, 32, 373–379. [Google Scholar] [CrossRef]
- Di Raimondo, D.; Tuttolomondo, A.; Butta, C.; Miceli, S.; Licata, G.; Pinto, A. Effects of ACE-inhibitors and angiotensin receptor blockers on inflammation. Curr. Pharm. Design 2012, 18, 4385–4413. [Google Scholar] [CrossRef]
- Rathinam, V.A.K.; Vanaja, S.K.; Fitzgerald, K.A. Regulation of inflammasome signaling. Nat. Immunol. 2012, 13, 333–342. [Google Scholar] [CrossRef]
- Kroemer, G.; Galluzzi, L.; Vandenabeele, P.; Abrams, J.; Alnemri, E.S.; Baehrecke, E.H.; Blagosklonny, M.V.; El-Deiry, W.S.; Golstein, P.; Green, D.R.; et al. Classification of cell death: recommendations of the Nomenclature Committee on Cell Death 2009. Cell Death Differ 2009, 16, 3–11. [Google Scholar] [CrossRef]
- Lamkanfi, M.; Dixit, V.M. Inflammasomes and their roles in health and disease. Annu. Rev. Cell. Dev. Bi 2012, 28, 137–161. [Google Scholar] [CrossRef]
- Fann, D.Y.W.; Lee, S.Y.; Manzanero, S.; Chunduri, P.; Sobey, C.G.; Arumugam, T.V. Pathogenesis of acute stroke and the role of inflammasomes. Age. Res. Rev. 2013, 12, 941–966. [Google Scholar] [CrossRef]
- Abais, J.M.; Xia, M.; Li, G.B.; Chen, Y.; Conley, S.M.; Gehr, T.W.B.; Boini, K.M.; Li, P.L. Nod-like Receptor Protein 3 (NLRP3) inflammasome activation and podocyte injury via thioredoxin-interacting protein (TXNIP) during hyperhomocysteinemia. J. Biol. Chem. 2014, 289, 27159–27168. [Google Scholar] [CrossRef]
- Sun, X.; Song, X.; Zhang, L.; Sun, J.; Wei, X.B.; Meng, L.Y.; An, J. NLRP2 is highly expressed in a mouse model of ischemic stroke. Biochem. Bioph. Res. Co. 2016, 479, 656–662. [Google Scholar] [CrossRef]
- Yang, Z.; Zhong, L.N.; Xian, R.H.; Yuan, B.Q. MicroRNA-223 regulates inflammation and brain injury via feedback to NLRP3 inflammasome after intracerebral hemorrhage. Mol. Immunol. 2015, 65, 267–276. [Google Scholar] [CrossRef]
- Zeng, X.S.; Zhou, X.S.; Luo, F.C.; Jia, J.J.; Qi, L.; Yang, Z.X.; Zhang, W.; Bai, J. Comparative analysis of the neuroprotective effects of ginsenosides Rg1 and Rb1 extracted from Panax notoginseng against cerebral ischemia. Can J. Physiol. Pharm. 2014, 92, 102–108. [Google Scholar] [CrossRef]
- Strowig, T.; Henao-Mejia, J.; Elinav, E.; Flavell, R. Inflammasomes in health and disease. Nature 2012, 481, 278–286. [Google Scholar] [CrossRef]
- Wang, X.X.; Li, R.P.; Wang, X.; Fu, Q.; Ma, S.P. Umbelliferone ameliorates cerebral ischemia-reperfusion injury via upregulating the PPAR gamma expression and suppressing TXNIP/NLRP3 inflammasome. Neurosci. Lett. 2015, 600, 182–187. [Google Scholar] [CrossRef]
- Sun, X.T.; Liu, J.; Crary, J.F.; Malagelada, C.; Sulzer, D.; Greene, L.A.; Levy, O.A. ATF4 protects against neuronal death in cellular parkinson’s disease models by maintaining levels of parkin. J. Neurosci. 2013, 33, 2398–2407. [Google Scholar] [CrossRef]
- Jiang, Y.F.; Liu, Z.Q.; Cui, W.; Zhang, W.T.; Gong, J.P.; Wang, X.M.; Zhang, Y.; Yang, M.J. Antioxidant effect of Salvianolic Acid B on hippocampal CA1 neurons in mice with cerebral ischemia and reperfusion injury. Chin. J. Integr. Med. 2015, 21, 516–522. [Google Scholar] [CrossRef]
- Sun, Q.; Meng, Q.T.; Jiang, Y.; Liu, H.M.; Lei, S.Q.; Su, W.T.; Duan, W.N.; Wu, Y.; Xia, Z.Y.; Xia, Z.Y. Protective effect of ginsenoside Rb1 against intestinal ischemia-reperfusion Induced acute renal injury in mice. PLoS ONE 2013, 8, 12. [Google Scholar] [CrossRef]
- Song, Z.M.; Liu, Y.; Hao, B.S.; Yu, S.J.; Zhang, H.; Liu, D.H.; Zhou, B.; Wu, L.; Wang, M.; Xiong, Z.J.; et al. Ginsenoside Rb1 prevents H2O2-induced HUVEC senescence by stimulating Sirtuin-1 pathway. PLoS ONE 2014, 9, 11. [Google Scholar] [CrossRef]
- Zeng, X.S.; Ji, J.J.; Kwon, Y.; Wang, S.D.; Bai, J. The role of thioredoxin-1 in suppression of endoplasmic reticulum stress in Parkinson disease. Free Radical. Bio. Med. 2014, 67, 10–18. [Google Scholar] [CrossRef]
- Hur, J.; Kim, S.; Lee, P.; Lee, Y.M.; Choi, S.Y. The protective effects of oxyresveratrol imine derivative against hydrogen peroxide-induced cell death in PC12 cells. Free Radical. Res. 2013, 47, 212–218. [Google Scholar] [CrossRef]
- Im, J.Y.; Lee, K.W.; Woo, J.M.; Junn, E.; Mouradian, M.M. DJ-1 induces thioredoxin 1 expression through the Nrf2 pathway. Hum. Mol. Genet. 2012, 21, 3013–3024. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, Z.; Zhang, M.; Ling, C.; Zhu, Y.; Ren, H.; Hong, C.; Qin, J.; Liu, T.; Wang, J. Neuroprotective Effects of Ginsenosides against Cerebral Ischemia. Molecules 2019, 24, 1102. https://doi.org/10.3390/molecules24061102
Cheng Z, Zhang M, Ling C, Zhu Y, Ren H, Hong C, Qin J, Liu T, Wang J. Neuroprotective Effects of Ginsenosides against Cerebral Ischemia. Molecules. 2019; 24(6):1102. https://doi.org/10.3390/molecules24061102
Chicago/Turabian StyleCheng, Zhekang, Meng Zhang, Chengli Ling, Ying Zhu, Hongwei Ren, Chao Hong, Jing Qin, Tongxiang Liu, and Jianxin Wang. 2019. "Neuroprotective Effects of Ginsenosides against Cerebral Ischemia" Molecules 24, no. 6: 1102. https://doi.org/10.3390/molecules24061102
APA StyleCheng, Z., Zhang, M., Ling, C., Zhu, Y., Ren, H., Hong, C., Qin, J., Liu, T., & Wang, J. (2019). Neuroprotective Effects of Ginsenosides against Cerebral Ischemia. Molecules, 24(6), 1102. https://doi.org/10.3390/molecules24061102




