Multi-year Quantitative Evaluation of Stilbenoids Levels Among Selected Muscadine Grape Cultivars
Abstract
:1. Introduction
2. Results
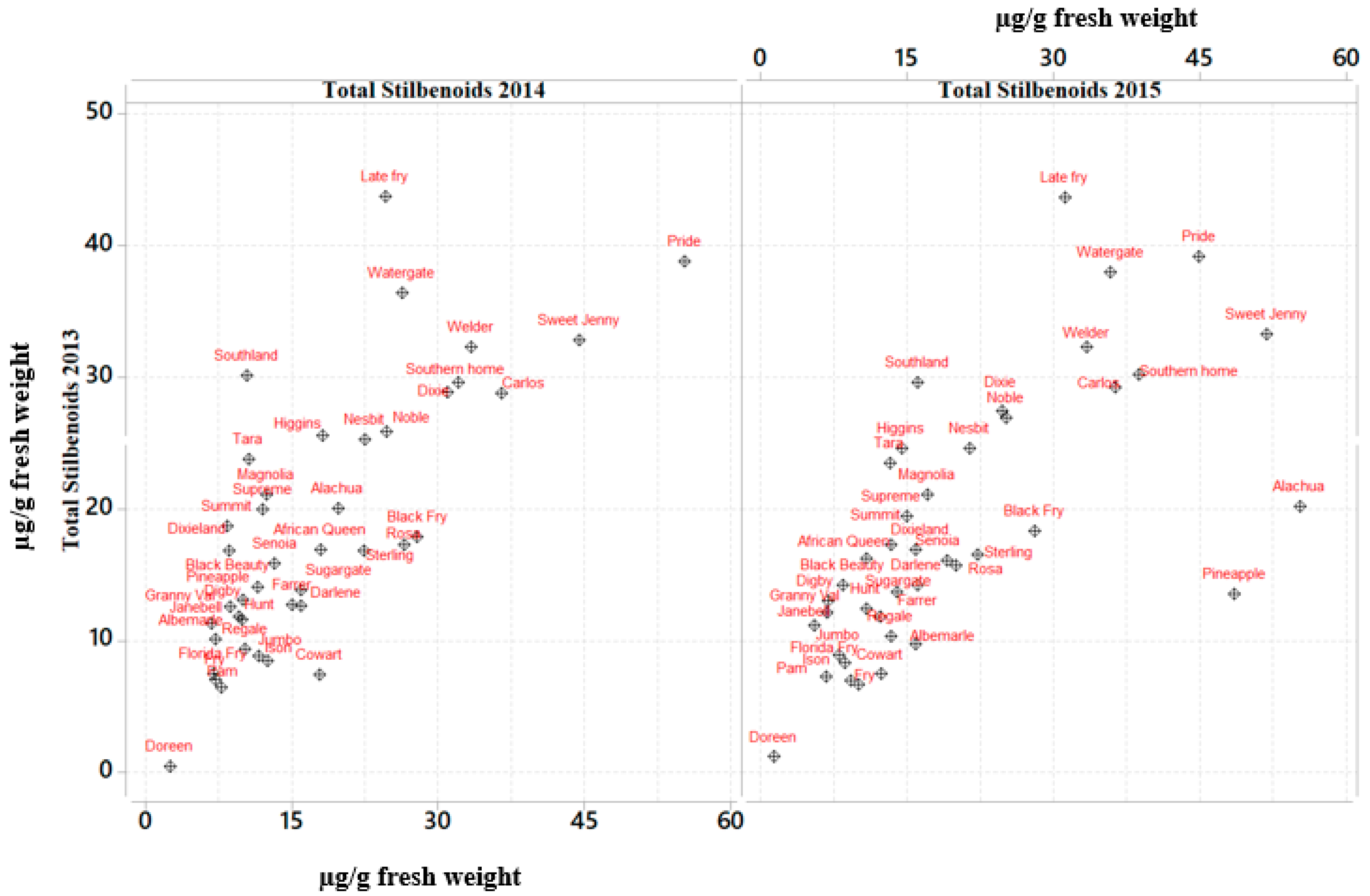
2.1. Stilbenoid Composition
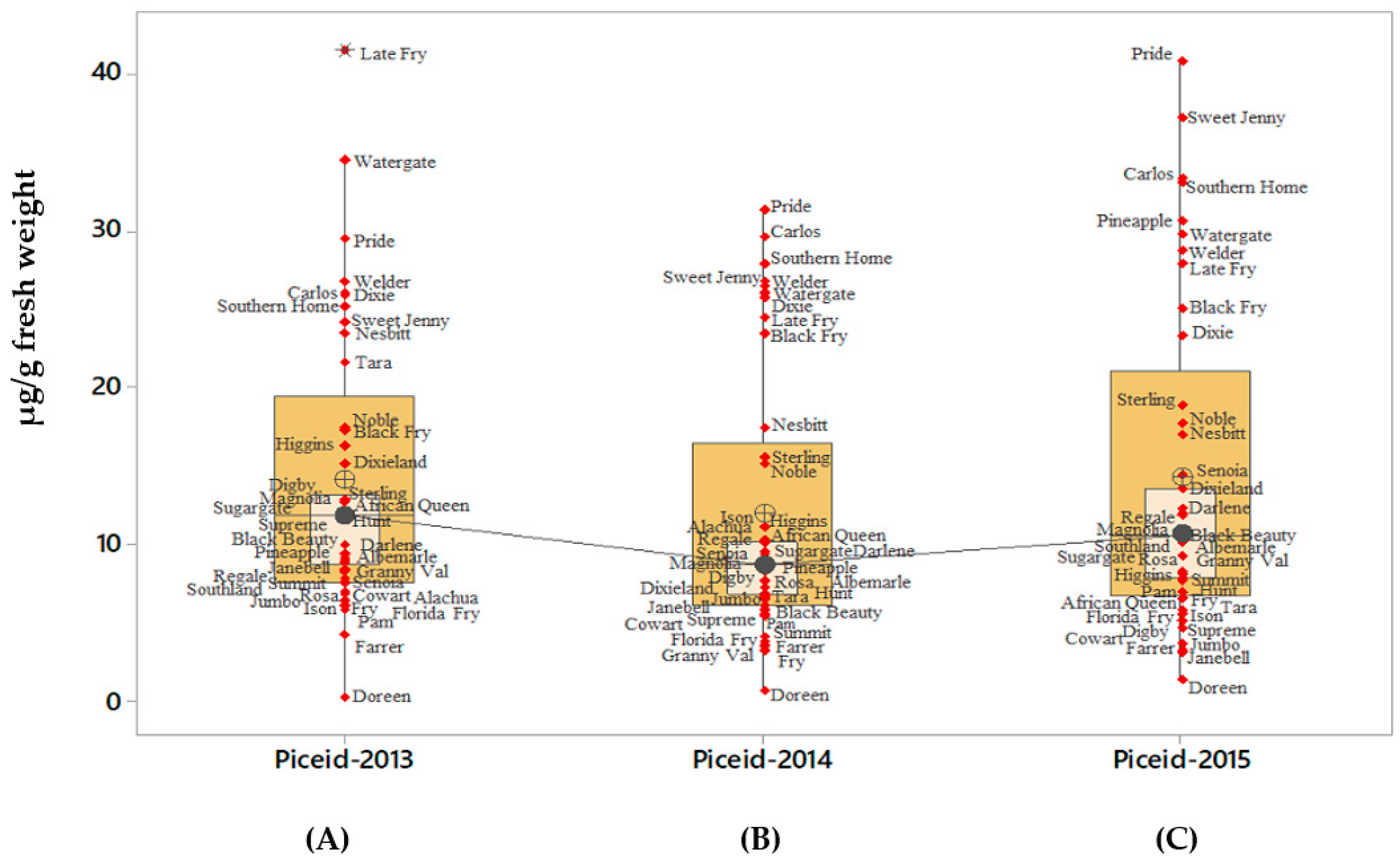
2.2. t-Piceid
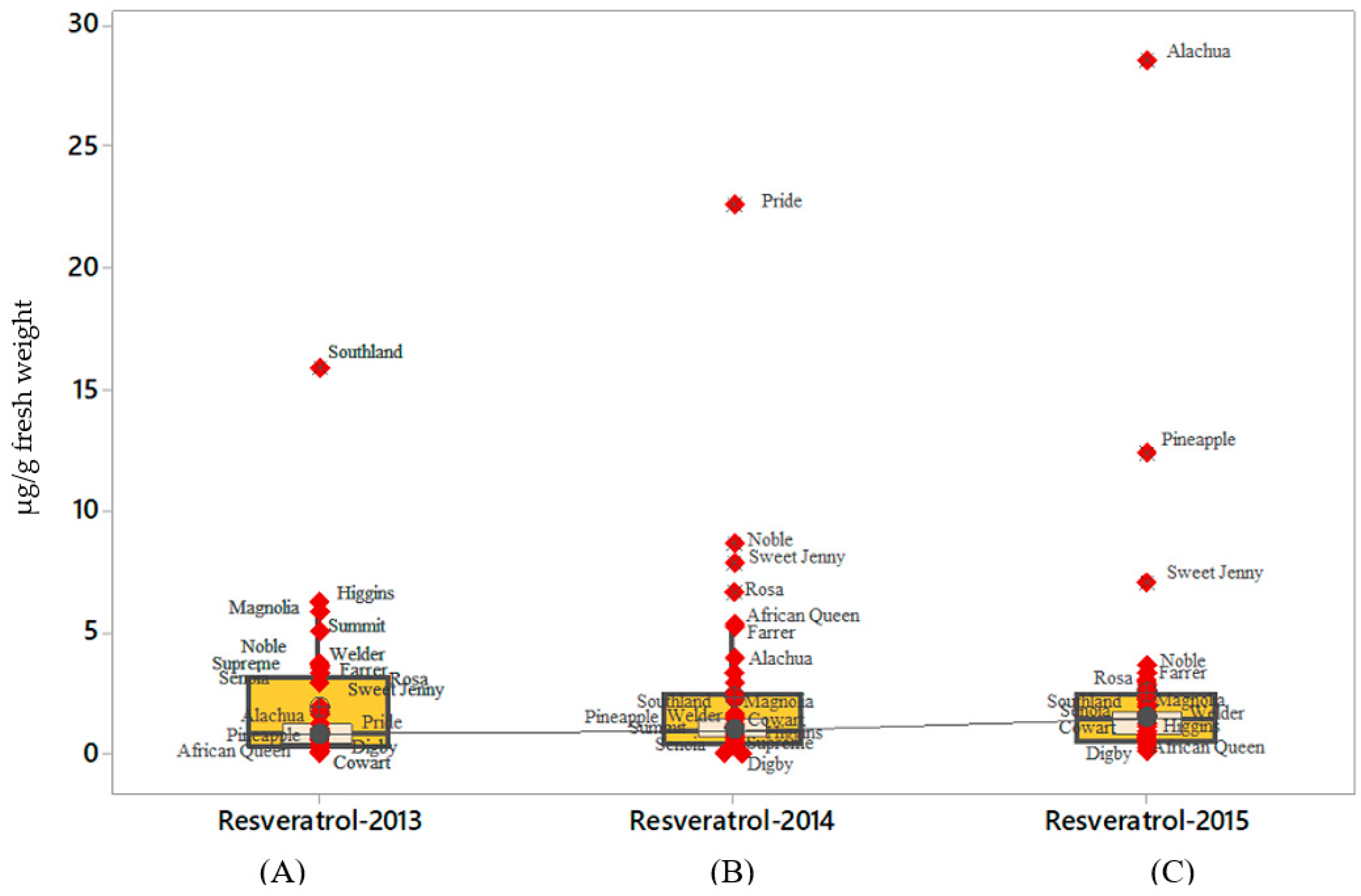
2.3. t-Resveratrol
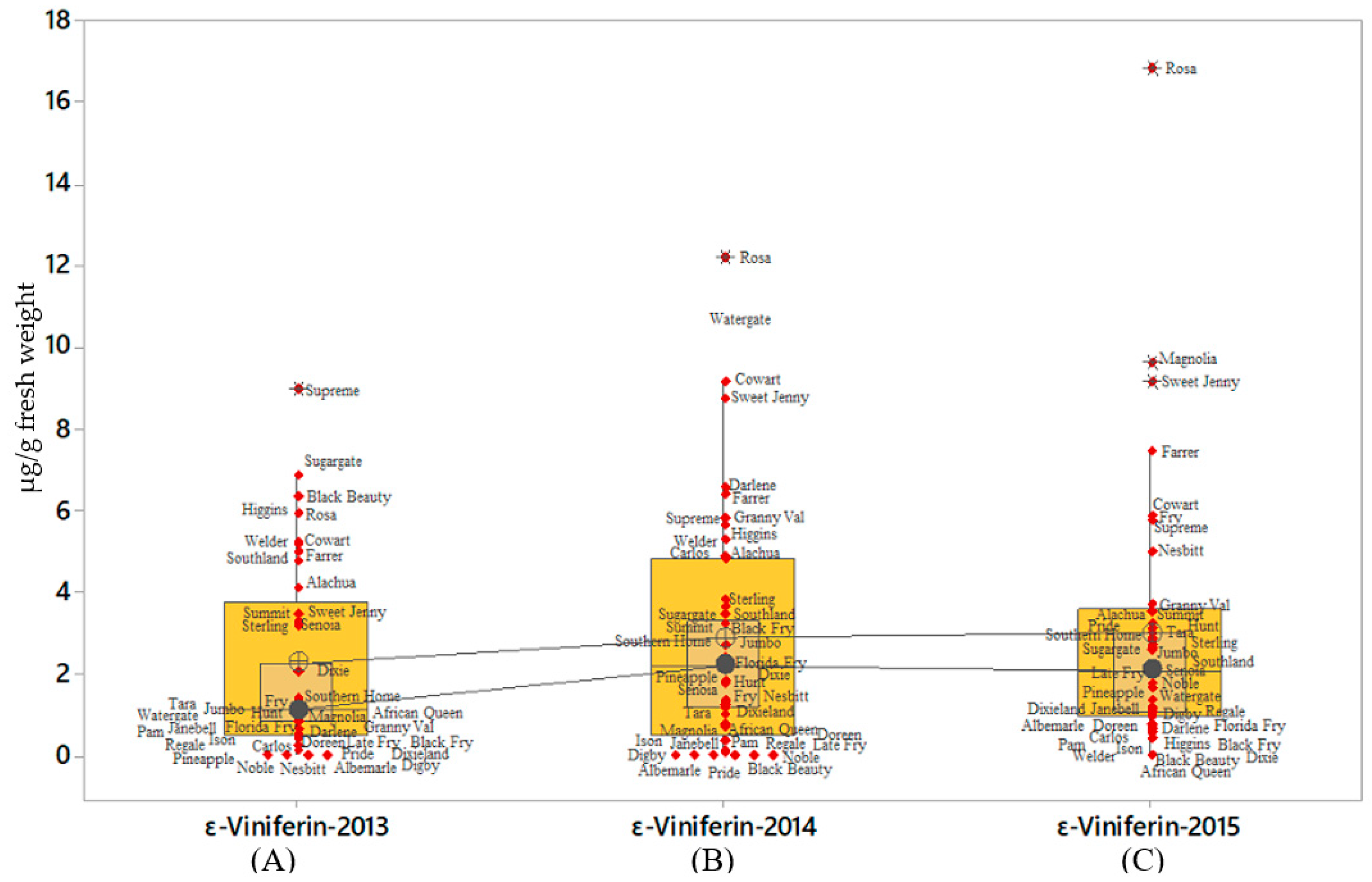
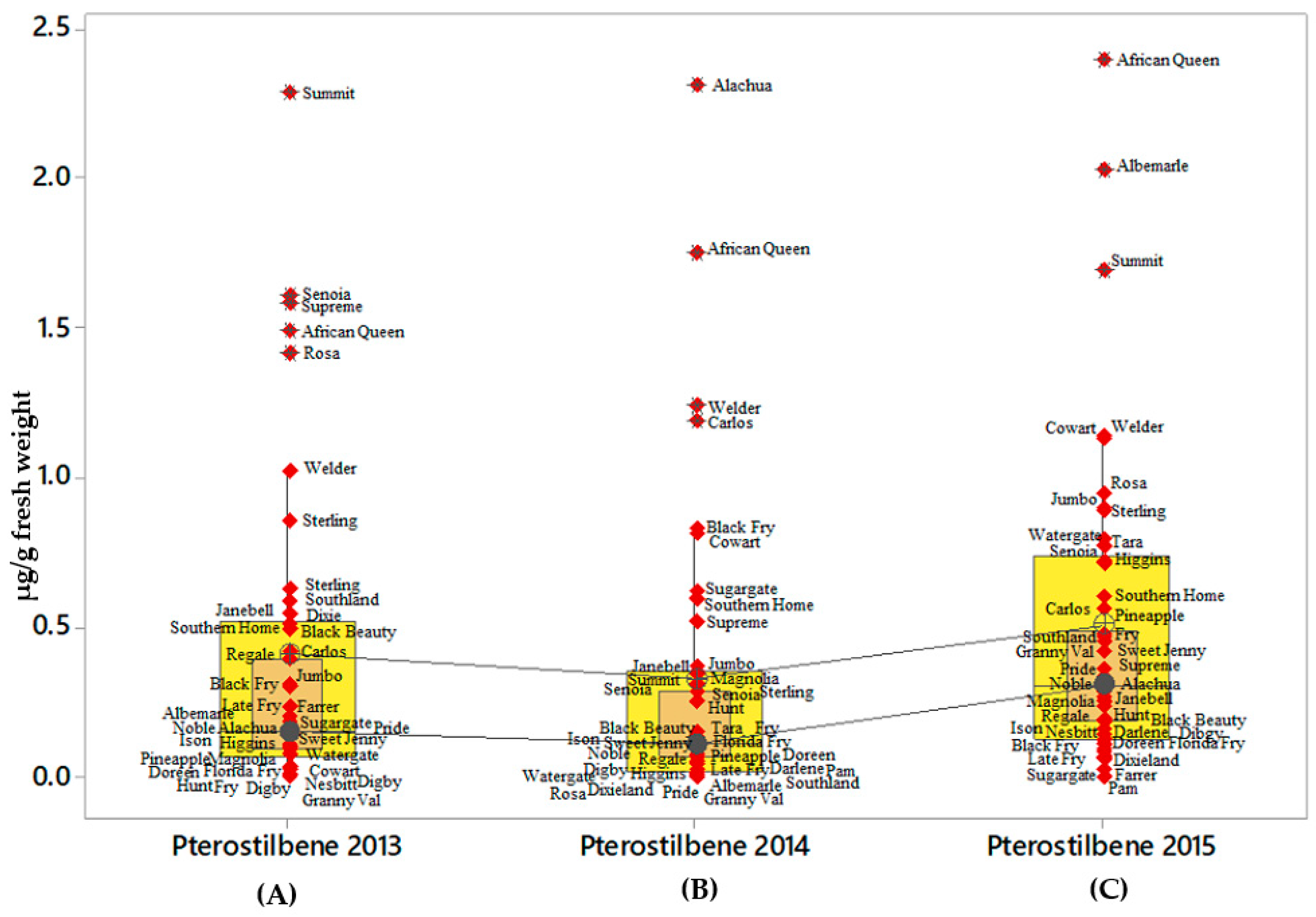
2.4. Ɛ-Viniferin and t-Pterostilbene
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Berry Samples
4.3. Extraction of Stilbenoids
4.4. Processing and Analysis of Stilbenoids
4.5. Representative Chromatogram of Stilbenoids’ Standards and Cultivars
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hartmann, T. From waste products to ecochemicals: Fifty years research of plant secondary metabolism. Phytochemistry 2007, 68, 2831–2846. [Google Scholar] [CrossRef] [PubMed]
- Soto, M.; Falqué, E.; Domínguez, H. Relevance of natural phenolics from grape and derivative Products in the Formulation of Cosmetics. Cosmetics 2015, 2, 259–276. [Google Scholar] [CrossRef]
- Yilmaz, Y.; Toledo, R.T. Major favonoids in grape seeds and skins: Antioxidant capacity of catechin, epicatechin, and gallic acid. J. Agric. Food Chem. 2004, 52, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Ector, B.J.; Magee, J.B.; Hegwood, C.P.; Coign, M.J. Resveratrol concentration in muscadine berries, juice, pomace, purees, seeds, and wines. Am. J. Enol. Vitic. 1996, 47, 57–62. [Google Scholar]
- Yi, W.; Fischer, J.; Akoh, C.C. Study of anticancer activities of muscadine grape phenolics in vitro. J. Agric. Food Chem. 2005, 53, 8804–8812. [Google Scholar] [CrossRef] [PubMed]
- Banini, A.E.; Boyd, L.C.; Allen, J.C.; Allen, H.G.; Sauls, D.L. Muscadine grape products intake, diet and blood constituents of non-diabetic and type 2 diabetic subjects. Nutrition 2006, 22, 1137–1145. [Google Scholar] [CrossRef] [PubMed]
- You, Q.; Chen, F.; Wang, X.; Sharp, J.L.; You, Y. Analysis of phenolic composition of noble muscadine (Vitis rotundifolia) by HPLC-MS and the relationship to its antioxidant capacity. J. Food Sci. 2012, 77, C1115–C1123. [Google Scholar] [CrossRef] [PubMed]
- Burton, L.J.; Rivera, M.; Hawsawi, O.; Zou, J.; Hudson, T.; Wang, G.; Zhang, Q.; Cubano, L.; Boukli, N.; Odero-Marah, V. Muscadine grape skin extract induces an unfolded protein response-mediated autophagy in prostate cancer cells: A TMT-based quantitative proteomic analysis. PLoS ONE 2016, 11, e0164115. [Google Scholar] [CrossRef] [PubMed]
- Pastrana-Bonilla, E.; Akoh, C.C.; Sellappan, S.; Krewer, G. Phenolic content and antioxidant capacity of muscadine grapes. J. Agric. Food Chem. 2003, 51, 5497–5503. [Google Scholar] [CrossRef] [PubMed]
- Sandhu, A.K.; Gu, L. Antioxidant capacity, phenolic content, and profiling of phenolic compounds in the seeds, skin, and pulp of Vitis rotundifolia (muscadine grapes) as determined by HPLC-DAD-ESI-MSn. J. Agric. Food Chem. 2010, 58, 4681–4692. [Google Scholar] [CrossRef] [PubMed]
- Marshall, A.; Stringer, S.J.; Spiers, J.D. Stilbene, ellagic acid, flavonol, and phenolic content of muscadine grape (Vitis rotundifolia Michx.) cultivars. Pharm. Crop. 2012, 3, 69–77. [Google Scholar] [CrossRef]
- Xu, C.; Yagiz, Y.; Zhao, L.; Simonne, A.; Lu, J.; Marshall, M.R. Fruit quality, nutraceutical and antimicrobial properties of 58 muscadine grape varieties (Vitis rotundifolia Michx.) grown in United States. Food Chem. 2017, 15, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Zhang, Y.; Lu, J. Phenolic contents and compositions in skins of red wine grape cultivars among various genetic backgrounds and originations. Int. J. Mol. Sci. 2012, 13, 3492–3510. [Google Scholar] [CrossRef] [PubMed]
- Pezet, R.; Pont, V. Mise en évidence de ptérostilbène dans les grappes de Vitis vinifera. Plant Physiol. Biochem. 1988, 26, 603–607. [Google Scholar]
- Mattivi, F.; Reniero, F.; Korhammer, S. Isolation, characterization, and evolution in red wine vinification of resveratrol monomers. J. Agric. Food Chem. 1995, 43, 1820–1823. [Google Scholar] [CrossRef]
- Langcake, P.; Pryce, R.J. A new class of phytoalexins from grapevines. Experientia 1977, 33, 151–152. [Google Scholar] [CrossRef] [PubMed]
- Paller, C.; Rudek, M.; Zhou, X.; Wagner, W.D.; Hudson, T.S.; Anders, N.; Hammers, H.J.; Dowling, D.; King, S.; Antonarakis, E.S.; et al. A phase I study of muscadine grape skin extract in men with biochemically recurrent prostate cancer: Safety, tolerability, and dose determination. Prostate 2015, 75, 1518–1525. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Yagiz, Y.; Xu, C.; Lu, J.; Chung, S.; Marshall, M.R. Muscadine grape seed oil as a novel source of tocotrienols to reduce adipogenesis and adipocyte inflammation. Food Funct. 2015, 6, 2293–2302. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Tani, T.; Kanbe, T.; Watanabe, K. Effect of cilostazol on platelet aggregation and experimental thrombosis. Arzneim. Forsch. 1985, 35, 1144–1149. [Google Scholar]
- Frankel, E.N.; German, J.B.; Kinsella, J.E.; Parks, E.; Kanner, J. Inhibition of oxidation of human low-density lipoprotein by phenolic substances in red wine. Lancet 1993, 341, 454–457. [Google Scholar] [CrossRef]
- Delaunois, B.; Cordelier, S.; Conreux, A.; Clement, C.; Jeandet, P. Molecular engineering of resveratrol in plants. Plant Biotechnol. J. 2009, 7, 2–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, S.Y.; Xie, X.Q.; Xu, Y.; Zhang, C.H.; Wang, X.P.; Zhang, J.X.; Wang, Y. Genetic transformation of a fruit-specific, highly expressed stilbene synthase gene from Chinese wild Vitis quinquangularis. Planta 2016, 243, 1041–1053. [Google Scholar] [CrossRef] [PubMed]
- Vergara, C.; von Baer, D.; Mardones, C.; Wilkens, A.; Wernekinck, K.; Damm, A.; Macke, S.; Gorena, T.; Winterhalter, P. Stilbene levels in grape cane of different cultivars in southern Chile: Determination by HPLC-DAD-MS/MS method. J. Agric. Food Chem. 2012, 60, 929–933. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Wang, L.; Wang, J.; Wu, B.; Liu, W.; Fan, P.; Liang, Z.; Li, S. Resveratrols in Vitis berry skins and leaves: Their extraction and analysis by HPLC. Food Chem. 2013, 136, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Floris, T.; Filippino, G.; Scrugli, S.; Pinna, M.B.; Argiolas, F.; Argiolas, A.; Murru, M.; Reverchon, E. Antioxidant compounds recovery from grape residues by a supercritical antisolvent assisted process. J. Supercrit. Fluids 2010, 54, 165–170. [Google Scholar] [CrossRef]
- Wang, W.; Tang, K.; Yang, H.R.; Wen, P.F.; Zhang, P.; Wang, H.L.; Huang, W.D. Distribution of resveratrol and stilbene synthase in young grape plants (Vitis vinifera L. cv. Cabernet Sauvignon) and the effect of UV-C on its accumulation. Plant Physiol. Biochem. 2010, 48, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Sobolev, V.S.; Cole, R.J.; Dorner, J.W.; Yagen, R. Isolation, purification, and liquid chromatographic determination of stilbene phytoalexins in peanuts. J. Assoc. Anal. Chem. Int. 1995, 78, 1177–1182. [Google Scholar]
- Jeandet, P.; Douillet-Breuil, A.C.; Bessis, R.; Debord, S.; Sbaghi, M.; Adrian, M. Phytoalexins from the vitaceae: Biosynthesis, phytoalexin gene expression in transgenic plants, antifungal activity, and metabolism. J. Agric. Food Chem. 2002, 50, 2731–2741. [Google Scholar] [CrossRef] [PubMed]
- Gatto, P.; Vrhovsek, U.; Muth, J.; Segala, C.; Romualdi, C.; Fontana, P.; Pruefer, D.; Stefanini, M.; Moser, C.; Mattivi, F.; et al. Ripening and genotype control stilbene accumulation in healthy grapes. J. Agric. Food Chem. 2008, 56, 11773–11785. [Google Scholar] [CrossRef] [PubMed]
- Romero-Pérez, A.I.; Lamuela-Raventós, R.M.; Andrés-Lacueva, C.; De La Carmen Torre-Boronat, M. Method for the quantitative extraction of resveratrol and piceid isomers in grape berry skins. Effect of powdery mildew on the stilbene content. J. Agric. Food Chem. 2001, 49, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Ribes, A.M.; Leandro, M.C.; Belchior, A.P.; Spranger, M.I. Stilbenes: Quantitative extraction from grape skins, contribution of grape solids to wine and variation during wine maturation. Anal. Chim. Acta 2006, 563, 382–390. [Google Scholar] [CrossRef]
- Vincenzi, S.; Tomasi, D.; Gaiotti, F.; Lovat, L.; Giacosa, S.; Torchio, F.; Segade, S.R.; Rolle, L. Comparative study of the resveratrol content of twenty-one Italian red grape varieties. S. Afr. J. Enol. Vitic. 2013, 34, 30–35. [Google Scholar] [CrossRef]
- Vitrac, X.; Bornet, A.; Vanderlinde, R.; Valls, J.; Richard, T.; Delaunay, J.C.; Mérillon, J.M.; Teissédre, P.L. Determination of stilbenes (δ-viniferin, trans-astringin, trans-piceid, cis-and trans-resveratrol, ε-viniferin) in Brazilian wines. J. Agric. Food Chem. 2005, 53, 5664–5669. [Google Scholar] [CrossRef] [PubMed]
- Barchenger, D.W.; Clark, J.R.; Threlfall, R.T.; Howard, L.R.; Brownmiller, C.R. Evaluation of physicochemical and storability attributes of muscadine grapes (Vitis rotundifolia Michx.). HortScience 2015, 50, 104–111. [Google Scholar] [CrossRef]
- Coombe, B.G. Growth stages of the grapevine: Adoption of a system for identifying grapevine growth stages. Aust. J. Grape Wine Res. 1995, 1, 104–110. [Google Scholar] [CrossRef]
- R Core Team 2016. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing 2010, Vienna Austria. Available online: https://www.R-project.org (accessed on 28 January 2018).
Sample Availability: Samples of the compounds are not available from the authors. |
| Source | DF | SS | Mean Sq. | F-Value | Pr (>F) |
|---|---|---|---|---|---|
| variety | 40 | 11803 | 295.08 | 11.293 | 1.45 × 10−12 |
| year | 1 | 58 | 58.21 | 2.228 | 0.14319 |
| cultivar:year | 40 | 2250 | 56.25 | 2.153 | 0.00817 |
| Residuals | 41 | 1071 | 26.13 |
| Muscadine Cultivars | Brix° | 2013 (µg/g Fresh Weight) | 2014 (µg/g Fresh Weight) | 2015 (µg/g Fresh Weight) | Mean (µg/g Fresh Weight) | SD |
|---|---|---|---|---|---|---|
| African Queen | 17.5 | 15.95 | 17.6 | 10.87 | 14.81 | 3.51 |
| Alachua | 15.0 | 19.84 | 21 | 54.31 | 31.72 | 19.87 |
| Albemarle | 16.5 | 10.46 | 8.42 | 16.87 | 11.92 | 4.41 |
| Black Beauty | 19.0 | 13.3 | 10.95 | 9.23 | 11.16 | 2.04 |
| Black Fry | 17.0 | 18.61 | 28.83 | 26.86 | 24.77 | 5.42 |
| Carlos | 13.5 | 28.2 | 36.77 | 37.37 | 34.11 | 5.13 |
| Cowart | 17 | 7.91 | 16.73 | 11.32 | 11.99 | 4.45 |
| Darlene | 15.0 | 13.22 | 16.48 | 15.28 | 14.99 | 1.65 |
| Digby | 14.0 | 12.39 | 8.33 | 6.28 | 9 | 3.11 |
| Dixie | 14.5 | 28.41 | 29.8 | 24.59 | 27.6 | 2.70 |
| Dixieland | 14.5 | 15.94 | 8.45 | 15.6 | 13.33 | 4.23 |
| Doreen | 13.5 | 1.39 | 1.45 | 2.36 | 1.73 | 0.54 |
| Farrer | 15.0 | 11.97 | 14.81 | 13.05 | 13.28 | 1.43 |
| Florida Fry | 17.0 | 7.47 | 6.92 | 7.28 | 7.22 | 0.28 |
| Fry | 17.5 | 7.55 | 6.26 | 10.58 | 8.13 | 2.22 |
| Fry Seedless | 16.0 | 257.47 | 274.12 | 279.01 | 270.2 | 11.29 |
| Granny Val | 16.0 | 11.98 | 9.12 | 7.89 | 9.66 | 2.10 |
| Higgins | 16.5 | 25.48 | 17.94 | 13.19 | 18.87 | 6.20 |
| Hunt | 16.0 | 12.18 | 9.11 | 9.89 | 10.4 | 1.60 |
| Ison | 15.0 | 7.72 | 11.87 | 6.53 | 8.71 | 2.80 |
| Janebell | 13.0 | 10.45 | 7.81 | 4.21 | 7.49 | 3.13 |
| Jumbo | 14.5 | 8.38 | 10.65 | 8.83 | 9.29 | 1.20 |
| Late Fry | 17.5 | 43.7 | 25.32 | 30.43 | 33.15 | 9.49 |
| Magnolia | 13.0 | 21.9 | 11.28 | 15.75 | 16.31 | 5.33 |
| Nesbitt | 13.5 | 24.82 | 21.65 | 20.06 | 22.18 | 2.42 |
| Noble | 16.5 | 26.41 | 23.69 | 24.41 | 24.84 | 1.41 |
| Pam | 18.0 | 6.23 | 6.44 | 8.49 | 7.05 | 1.25 |
| Pineapple | 15.0 | 13.25 | 11.08 | 49.73 | 24.69 | 21.72 |
| Pride | 16.0 | 38.65 | 54 | 45.9 | 46.18 | 7.68 |
| Regale | 15.0 | 9.69 | 11 | 12.94 | 11.21 | 1.64 |
| Rosa | 13.0 | 16.3 | 26.1 | 19.67 | 20.69 | 4.98 |
| Senoia | 14.0 | 15.24 | 12.05 | 18.99 | 15.43 | 3.47 |
| Southern home | 16.0 | 29.48 | 33.17 | 39.54 | 34.07 | 5.09 |
| Southland | 17.0 | 30.3 | 11.12 | 15.97 | 19.13 | 9.97 |
| Sterling | 14.5 | 16.9 | 22.36 | 22.72 | 20.66 | 3.26 |
| Sugargate | 17.0 | 13.88 | 14.9 | 14.48 | 14.42 | 0.51 |
| Summit | 18.0 | 18.28 | 7.8 | 13.6 | 13.23 | 5.25 |
| Supreme | 17.0 | 19.33 | 13.05 | 14.88 | 15.75 | 3.23 |
| Sweet Jenny | 18.0 | 33.03 | 43.25 | 51.77 | 42.68 | 9.38 |
| Tara | 15.0 | 23.79 | 9.43 | 12.12 | 15.11 | 7.63 |
| Watergate | 14.5 | 37.17 | 27.31 | 35.02 | 33.17 | 5.18 |
| Welder | 14.0 | 33.12 | 32.9 | 33.19 | 33.07 | 0.15 |
| Source | DF | SS | Mean Sq. | F-Value | Pr(>F) |
|---|---|---|---|---|---|
| cultivar | 40 | 9630 | 240.74 | 26.356 | <2 × 10−16 |
| year | 1 | 1 | 1.23 | 0.134 | 0.71589 |
| cultivar:year | 40 | 898 | 22.46 | 2.459 | 0.00251 |
| Residuals | 41 | 375 | 9.13 |
| Source | DF | SS | Mean Sq. | F-Value | Pr (>F) |
|---|---|---|---|---|---|
| variety | 40 | 642.8 | 16.07 | 1.216 | 0.268 |
| year | 1 | 7.9 | 7.936 | 0.601 | 0.443 |
| cultivar:year | 40 | 595.5 | 14.887 | 1.127 | 0.352 |
| Residuals | 41 | 541.7 | 13.212 |
| Source | DF | SS | Mean Sq. | F-Value | Pr (>F) |
|---|---|---|---|---|---|
| cultivar | 40 | 664.3 | 16.607 | 6.506 | 1.2 × 10−8 |
| year | 1 | 10.7 | 10.75 | 4.211 | 0.0466 |
| cultivar:year | 40 | 208.9 | 2.046 | 2.459 | 0.0124 |
| Residuals | 41 | 375 | 9.13 |
| Source | DF | SS | Mean Sq. | F-Value | Pr (>F) |
|---|---|---|---|---|---|
| variety | 40 | 19.442 | 0.4861 | 2.145 | 0.00844 |
| year | 1 | 0.184 | 0.1836 | 0.81 | 0.37337 |
| cultivar:year | 40 | 5.31 | 0.1328 | 0.586 | 0.95337 |
| Residuals | 41 | 9.293 | 0.2266 |
| Aug. 2013 | Sept. 2013 | Aug. 2014 | Sept. 2014 | Aug. 2015 | Sept. 2015 | |
|---|---|---|---|---|---|---|
| Avg. high temp. | 34.4 °C | 31.7 °C | 35.4 °C | 31.3 °C | 33.0 °C | 32.2 °C |
| Avg. low temp. | 23.7 °C | 21.7 °C | 23.1 °C | 21.7 °C | 23.1 °C | 21.5 °C |
| Precipitation | 15.42 cm | 7.77 cm | 5.74 cm | 17.32 cm | 21.89 cm | 8.09 cm |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kambiranda, D.M.; Basha, S.M.; Stringer, S.J.; Obuya, J.O.; Snowden, J.J. Multi-year Quantitative Evaluation of Stilbenoids Levels Among Selected Muscadine Grape Cultivars. Molecules 2019, 24, 981. https://doi.org/10.3390/molecules24050981
Kambiranda DM, Basha SM, Stringer SJ, Obuya JO, Snowden JJ. Multi-year Quantitative Evaluation of Stilbenoids Levels Among Selected Muscadine Grape Cultivars. Molecules. 2019; 24(5):981. https://doi.org/10.3390/molecules24050981
Chicago/Turabian StyleKambiranda, Devaiah M., Sheikh M. Basha, Stephen J. Stringer, James O. Obuya, and Janana J. Snowden. 2019. "Multi-year Quantitative Evaluation of Stilbenoids Levels Among Selected Muscadine Grape Cultivars" Molecules 24, no. 5: 981. https://doi.org/10.3390/molecules24050981
APA StyleKambiranda, D. M., Basha, S. M., Stringer, S. J., Obuya, J. O., & Snowden, J. J. (2019). Multi-year Quantitative Evaluation of Stilbenoids Levels Among Selected Muscadine Grape Cultivars. Molecules, 24(5), 981. https://doi.org/10.3390/molecules24050981