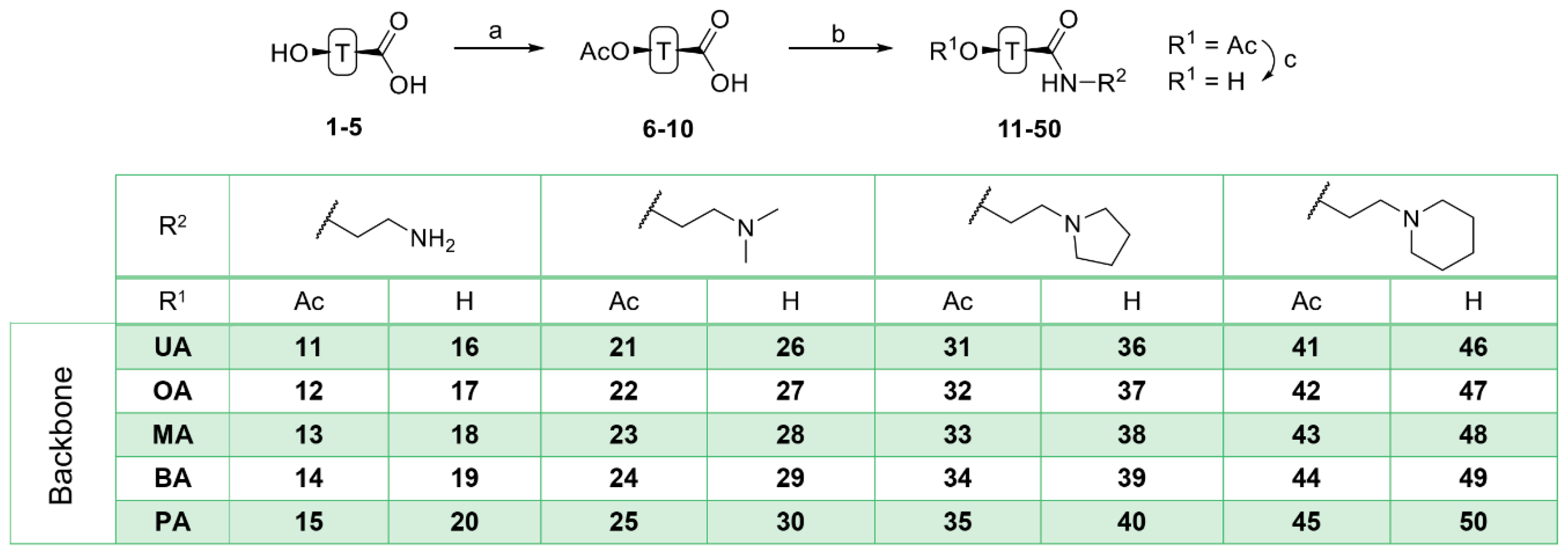
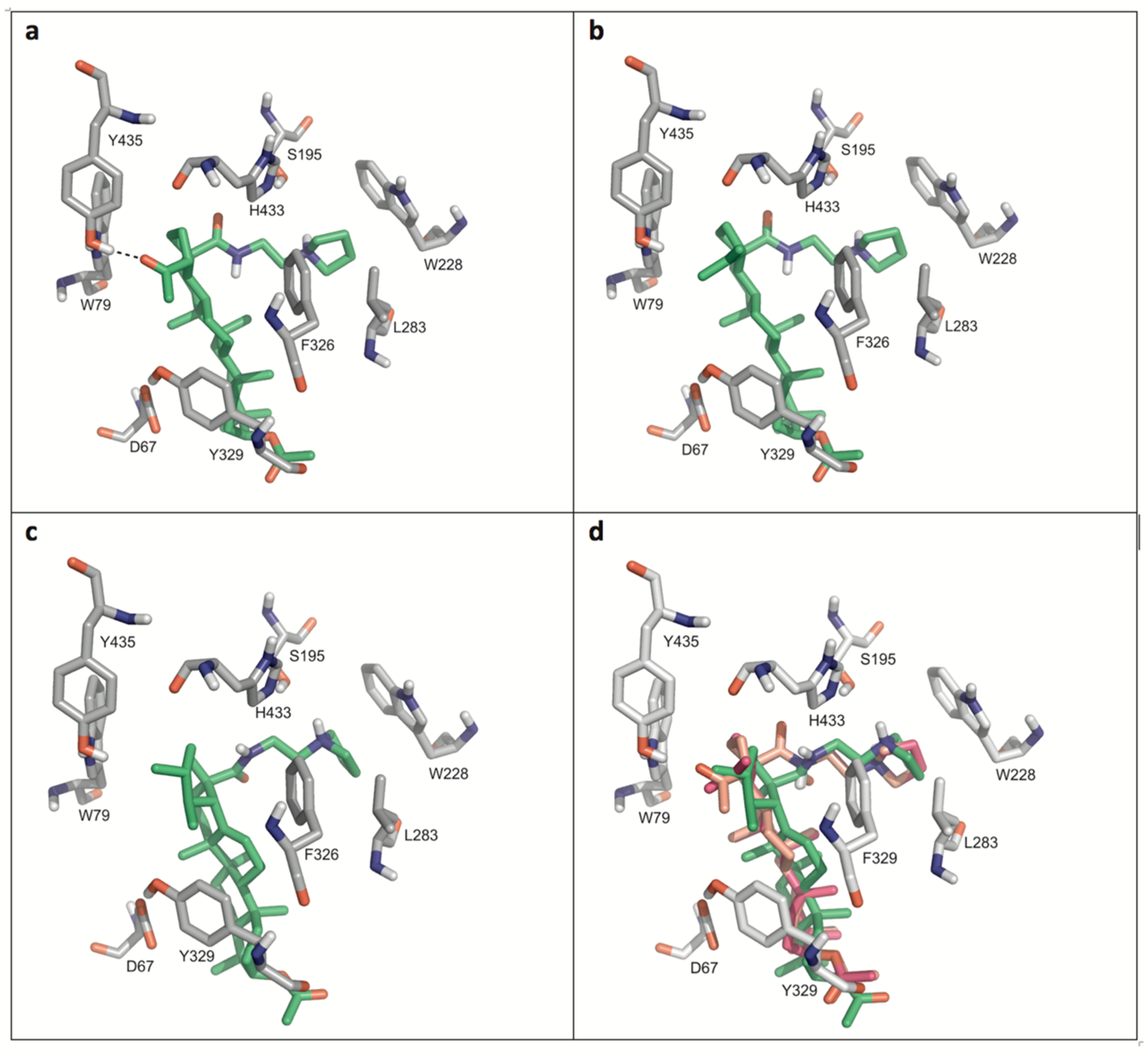
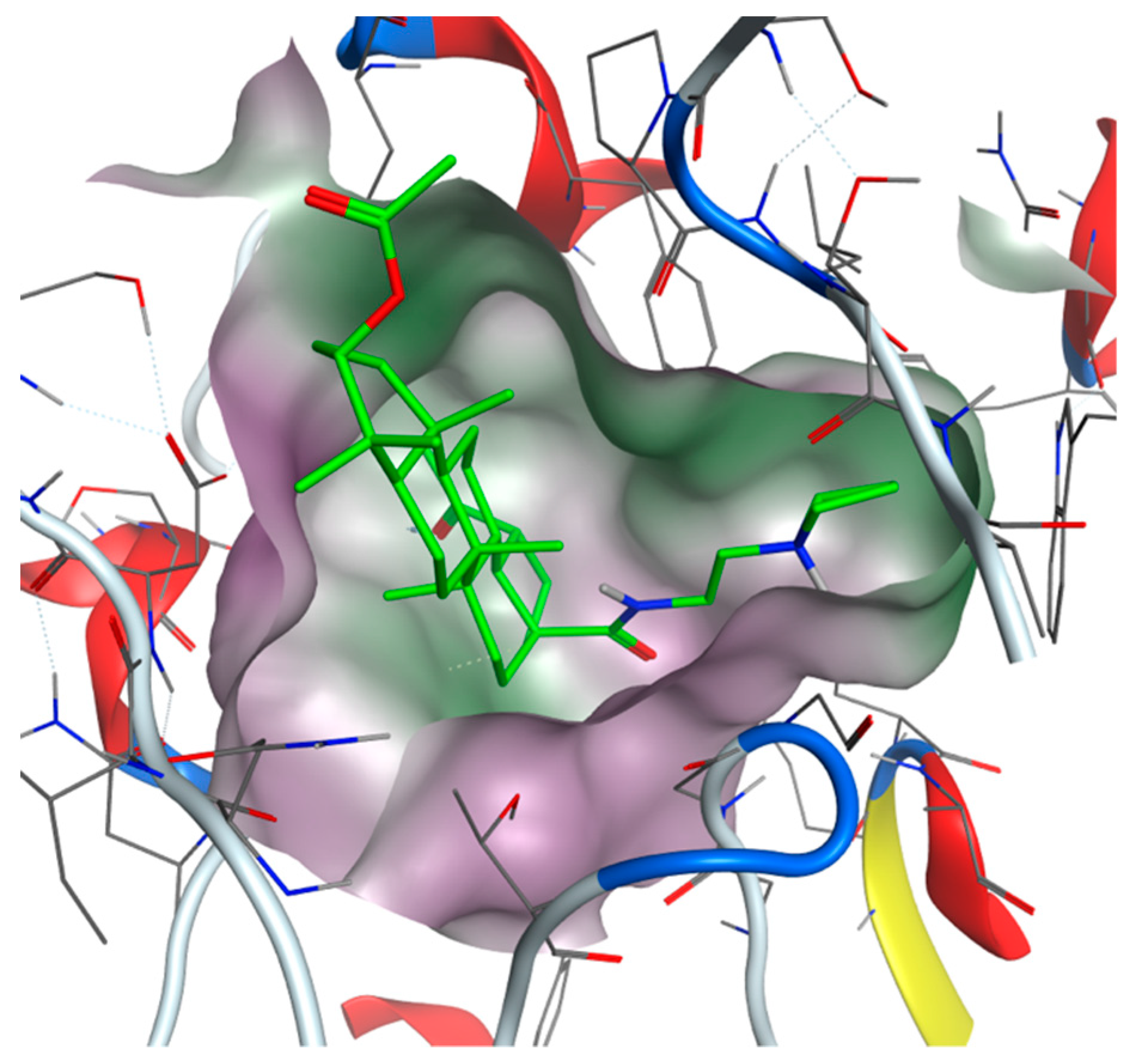
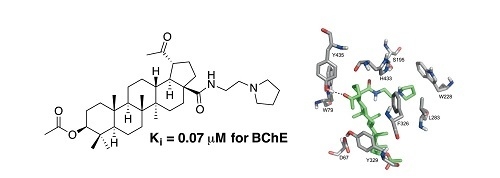
4.4. General Procedure C for the Deacetylation of Triterpenoic Amides (16–20, 26–30, 36–40, and 46–50)
To a solution of the acetylated amide (0.33 mmol) in methanol (10 mL) was added a solution of potassium hydroxide (1.65 mmol) in methanol (2 mL). The mixture was stirred at 25 °C for 2 or 3 days. After completion of the reaction (as indicated by TLC), aq. HCl was added until pH = 7. After usual work-up, the solvent was removed under reduced pressure and the residue was subjected to column chromatography (silica gel, chloroform/methanol mixtures) yielding compounds 16–20, 26–30, 36–40, and 46–50 each as colorless solids.
(3β
)-3-Acetyloxy-urs-12-en-28-oic acid (
6), Compound
6 was prepared according to general procedure A from ursolic acid (
1). Yield: 96%; m.p. 287–290 °C (lit.: 289–290 °C [
24]).
(3β
)-3-Acetyloxy-olean-12-en-28-oic acid (
7), Compound
7 was prepared according to general procedure A from oleanolic acid (
2). Yield: 90%; m.p. 259–261 °C (lit.: 255–257 °C [
25]).
(2α
,3β
)-2,3-Diacetyloxy-olean-12-en-28-oic acid (
8), Compound
8 was prepared according to general procedure A from maslinic acid (
3). Yield: 91%; m.p. 172–175 °C (l.: 170–173 °C [
26]).
(3β
)-3-Acetyloxy-lup-20(29)en-28-oic acid (
9), Compound
9 was prepared according to general procedure A from betulinic acid (
4). Yield: 93%; m.p. 281–284 °C (lit.: 280–282 °C [
27]).
(3β
)-3-Acetyloxy-20-oxo-30-norlupan-28-oic acid (
10), Compound
10 was prepared according to general procedure A from platanic acid (
5). Yield: 94%; m.p. 256–259 °C (lit.: 252–255 °C [
28]).
(3β)-N-(2-Aminoethyl)-3-acetyloxy-olean-12-en-28-amide (12), Compound 12 was prepared from 7 according to general procedure B using ethylenediamine as amino compound. Column chromatography (SiO2, CHCl3/MeOH 9:1) gave 12 (yield: 75%); m.p. 212–215 °C (decomp.); [α]D = +37.8° (c 0.350, CHCl3); Rf = 0.67 (CHCl3/MeOH/NH4OH 90:10:1); IR (ATR): ν = 2944 m, 1732 m, 1628 m, 1523 m, 1364 s, 1244 s, 1027 m, 985 m, 824 m, 752 m cm−1; 1H-NMR (400 MHz, CDCl3): δ = 7.04 (t, J = 5.5 Hz, 1H, NH), 5.40 (t, J = 3.4 Hz, 1H, 12-H), 4.53–4.45 (m, 1H, 3-H), 3.68–3.56 (m, 1H, 31-Ha), 3.40–3.29 (m, 1H, 31-Hb), 3.24–3.11 (m, 2H, 32-H), 2.65 (dd, J = 12.7, 4.6 Hz, 1H, 18-H), 2.04 (s, 3H, Ac), 2.01–1.82 (m, 3H, 16-Ha, 11-Ha, 11-Hb), 1.79–1.22 (m, 14H, 19-Ha, 1-Ha, 2-Ha, 2-Hb, 7-Ha, 7-Hb, 9-H, 16-Hb, 6-Ha, 15-Ha, 22-Ha, 6-Hb, 21-Ha, 22-Hb), 1.22–1.11 (m, 2H, 19-Hb, 21-Hb), 1.14 (s, 3H, 27-H), 1.10–0.95 (m, 2H, 1-Hb, 15-Hb), 0.93 (s, 3H, 25-H), 0.90 (s, 3H, 30-H), 0.89 (s, 3H, 29-H), 0.86 (s, 3H, 23-H), 0.85 (s, 3H, 24-H), 0.84–0.79 (m, 1H, 5-H), 0.73 (s, 3H, 26-H) ppm; 13C-NMR (101 MHz, CDCl3): δ = 180.8 (C-28), 171.0 (Ac), 144.0 (C-13), 123.1 (C-12), 80.7 (C-3), 55.2 (C-5), 47.5 (C-9), 46.6 (C-19), 46.3 (C-17), 41.8 (C-14), 41.4 (C-18), 40.4 (C-32), 39.4 (C-8), 38.2 (C-1), 38.1 (C-31), 37.7 (C-4), 36.9 (C-10), 34.2 (C-21), 33.1 (C-30), 32.3 (C-22), 32.2 (C-7), 30.6 (C-20), 28.0 (C-23), 27.2 (C-15), 25.8 (C-27), 23.6 (C-2), 23.5 (C-16), 23.5 (C-11), 23.5 (C-29), 21.3 (Ac), 18.2 (C-6), 16.9 (C-26), 16.7 (C-24), 15.5 (C-25) ppm; MS (ESI, MeOH): m/z = 541.3 (100%, [M + H]+); analysis calcd. for C34H56N2O3 (540.83): C 75.51, H 10.44, N 5.18; found: C 75.42, H 10.57, N 5.07.
(2α,3β)-N-(2-Aminoethyl)-2,3-diacetyloxy-olean-12-en-28-amide (13), Compound 13 was prepared from 8 according to general procedure B using ethylenediamine as amino compound. Column chromatography (SiO2, CHCl3/MeOH 9:1) gave 13 (yield: 74%); m.p. 151–154 °C; [α]D = +18.7° (c 0.330, CHCl3); Rf = 0.63 (CHCl3/MeOH/NH4OH 90:10:1); IR (KBr): ν = 3426 br s, 2946 s, 1742 s, 1636m, 1522m, 1458m, 1436w, 1368m, 1254s, 1044m cm−1; 1H-NMR (400 MHz, CDCl3): δ = 6.36 (t, J = 5.5 Hz, 1H, NH), 5.37 (t, J = 3.6 Hz, 1H, 12-H), 5.08 (ddd, J = 11.1, 10.9, 4.6 Hz, 1H, 2-H), 4.73 (d, J = 10.3 Hz, 1H, 3-H), 3.48–3.39 (m, 1H, 31-Ha), 3.12–3.02 (m, 1H, 31-Hb), 2.87–2.76 (m, 2H, 32-H), 2.56 (dd, J = 13.1, 4.3 Hz, 1H, 18-H), 2.04 (s, 3H, Ac), 2.08–1.83 (m, 4H, 1-Ha, 16-Ha, 11-Ha, 11-Hb), 1.97 (s, 3H, Ac), 1.80–1.24 (m, 11H, 19-Ha, 22-Ha, 16-Hb, 9-H, 22-Hb, 6-Ha, 15-Ha, 7-Ha, 6-Hb, 21-Ha, 7-Hb), 1.23–1.09 (m, 2H, 21-Hb, 19-Hb), 1.14 (s, 3H, 27-H), 1.11–0.99 (m, 2H, 1-Hb, 15-Hb), 1.04 (s, 3H, 25-H), 0.99–0.92 (m, 1H, 5-H), 0.90 (s, 9H, 24-H, 29-H, 30-H), 0.89 (s, 3H, 23-H), 0.76 (s, 3H, 26-H) ppm; 13C-NMR (101 MHz, CDCl3): δ = 178.8 (C-28), 170.9 (Ac), 170.7 (Ac), 144.9 (C-13), 122.5 (C-12), 80.7 (C-3), 70.1 (C-2), 54.9 (C-5), 47.6 (C-9), 46.8 (C-19), 46.5 (C-17), 44.0 (C-1), 42.3 (C-18), 42.2 (C-14), 41.8 (C-31), 41.4 (C-32), 39.6 (C-8), 39.5 (C-4), 38.2 (C-10), 34.3 (C-21), 33.1 (C-30), 32.9 (C-22), 32.3 (C-7), 30.9 (C-20), 28.5 (C-23), 27.4 (C-15), 25.9 (C-27), 23.8 (C-16), 23.8 (C-29), 23.7 (C-11), 21.3 (Ac), 21.0 (Ac), 18.3 (C-6), 17.8 (C-24), 17.1 (C-26), 16.6 (C-25) ppm; MS (ESI, MeOH): m/z = 599 (100%, [M + H]+); analysis calcd. for C36H58N2O5 (598.87): C 72.20, H 9.76, N 4.68; found: C 72.01, H 9.92, N 4.44.
(3β)-N-(2-Aminoethyl)-3-hydroxy-olean-12-en-28-amide (17), Compound 17 was prepared from 12 according to general procedure C. Column chromatography (SiO2, CHCl3/MeOH/NH4OH 90:10:0.1) gave 17 (yield: 80%); m.p. 221–224 °C (decomp.); [α]D = +55.3° (c 0.315, CHCl3); Rf = 0.61 (CHCl3/MeOH/NH4OH 90:10:1); IR (ATR): ν = 3374w, 2941m, 1622m, 1530m, 1456m, 1387s, 1361s, 1344s, 1322s, 1187m, 1137m, 1097m, 1023m, 996m, 825m cm−1; 1H-NMR (500 MHz, CD3OD): δ = 5.36 (t, J = 3.7 Hz, 1H, 12-H), 3.50–3.43 (m, 1H, 31-Ha), 3.37–3.32 (m, 1H, 31-Hb), 3.15 (dd, J = 11.3, 4.8 Hz, 1H, 3-H), 3.07–2.95 (m, 2H, 32-H), 2.79 (dd, J = 13.2, 4.4 Hz, 1H, 18-H), 2.10 (ddd, J = 13.8, 13.0, 4.0 Hz, 1H, 16-Ha), 1.99–1.86 (m, 2H, 11-Ha, 11-Hb), 1.83–1.74 (m, 1H, 19-Ha), 1.70–1.35 (m, 12H, 22-Ha, 1-Ha, 9-H, 15-Ha, 2-Ha, 2-Hb, 16-Hb, 6-Ha, 22-Hb, 7-Ha, 6-Hb, 21-Ha), 1.33–1.27 (m, 1H, 7-Hb), 1.25–1.14 (m, 2H, 21-Hb, 19-Hb), 1.18 (s, 3H, 27-H), 1.11–0.99 (m, 2H, 15-Hb, 1-Hb), 0.97 (s, 3H, 23-H), 0.96 (s, 3H, 29-H), 0.94 (s, 3H, 25-H), 0.92 (s, 3H, 30-H), 0.79 (s, 3H, 26-H), 0.78 (s, 3H, 24-H), 0.77–0.74 (m, 1H, 5-H) ppm; 13C-NMR (126 MHz, CD3OD): δ = 182.0 (C-28), 145.0 (C-13), 124.2 (C-12), 79.6 (C-3), 56.7 (C-5), 49.0 (C-9), 47.6 (C-17), 47.6 (C-19), 42.9 (C-14), 42.4 (C-18), 40.8 (C-32), 40.7 (C-8), 39.8 (C-4), 39.8 (C-1), 38.8 (C-31), 38.1 (C-10), 35.0 (C-21), 34.2 (C-22), 33.8 (C-7), 33.5 (C-30), 31.6 (C-20), 28.7 (C-23), 28.5 (C-15), 27.8 (C-2), 26.5 (C-27), 24.5 (C-11), 24.0 (C-29), 24.0 (C-16), 19.5 (C-6), 17.9 (C-26), 16.3 (C-24), 15.9 (C-25) ppm; MS (ESI, MeOH): m/z = 499.3 (100%, [M + H]+); analysis calcd. for C32H54N2O2 (498.80): C 77.06, H 10.91, N 5.62; found: C 76.90, H 11.05, N 5.41.
(2α,3β)-N-(2-Aminoethyl)-2,3-dihydroxy-olean-12-en-28-amide (18), Compound 18 was prepared from 13 according to general procedure C. Column chromatography (SiO2, CHCl3/MeOH 9:1) gave 18 (yield: 60%); m.p. 260–266 °C (decompn.); [α]D = +47.6° (c 0.335, CHCl3); Rf = 0.36 (CHCl3/MeOH/NH4OH 90:10:1); IR (KBr): ν = 3424 br s, 944m, 1636m, 1528m, 1460w, 1384w, 1166w, 1050m cm−1; 1H-NMR (400 MHz, CD3OD): δ = 5.36 (t, J = 3.7 Hz, 1H, 12-H), 3.61 (ddd, J = 11.3, 9.5, 4.5 Hz, 1H, 2-H), 3.43–3.35 (m, 1H, 31-Ha), 3.29–3.22 (m, 1H, 31-Hb), 2.90 (d, J = 9.6 Hz, 1H, 3-H), 2.89 (t, J = 6.5 Hz, 2H, 32-H), 2.81 (dd, J = 13.5, 4.5 Hz, 1H, 18-H), 2.14–2.03 (m, 1H, 16-Ha), 2.03–1.88 (m, 3H, 11-Ha, 11-Hb, 1-Ha), 1.78 (t, J = 13.5 Hz, 1H, 19-Ha), 1.70–1.26 (m, 10H, 9-H, 22-Ha, 15-Ha, 16-Hb, 6-Ha, 22-Hb, 7-Ha, 6-Hb, 21-Ha, 7-Hb), 1.23–1.13 (m, 2H, 19-Hb, 21-Hb), 1.18 (s, 3H, 27-H), 1.10–1.02 (m, 1H, 15-Hb), 1.01 (s, 3H, 23-H), 1.00 (s, 3H, 25-H), 0.95 (s, 3H, 30-H), 0.91 (s, 3H, 29-H), 0.93–0.89 (m, 1H, 1-Hb), 0.88–0.82 (m, 1H, 5-H), 0.80 (s, 3H, 24-H), 0.78 (s, 3H, 26-H) ppm; 13C-NMR (101 MHz, CD3OD): δ = 181.5 (C-28), 145.2 (C-13), 123.9 (C-12), 84.4 (C-3), 69.4 (C-2), 56.6 (C-5), 48.9 (C-9), 48.1 (C-1), 47.6 (C-17), 47.5 (C-19), 42.9 (C-14), 42.5 (C-18), 41.2 (C-32), 40.7 (C-8), 40.5 (C-4), 40.4 (C-31), 39.2 (C-10), 35.1 (C-21), 34.3 (C-22), 33.7 (C-7), 33.5 (C-30), 31.6 (C-20), 29.3 (C-23), 28.5 (C-15), 26.5 (C-27), 24.6 (C-11), 24.0 (C-29), 23.9 (C-16), 19.5 (C-6), 17.9 (C-26), 17.4 (C-24), 17.1 (C-25) ppm; MS (ESI, MeOH): m/z = 515 (100%, [M + H]+); analysis calcd. for C32H54N2O3 (514.80): C 74.66, H 10.57, N 5.44; found: C 74.49, H 10.74, N 5.28.
(3β
)-N-[2-(Dimethylamino)-ethyl]-3-acetyloxy-olean-12-en-28-amide (
22), Compound
22 was prepared from
7 according to general procedure B using N,N-dimethylethylene diamine as amino compound. Column chromatography (SiO
2, CHCl
3/MeOH 95:5) gave
22 (yield: 95%); m.p. 227–230 °C (decomp.), Lit.: 255 °C (decomp.) [
29]; [α]
D = +53.0° (c 0.320, CHCl
3), Lit.: +51.8° (c 0.34, CHCl
3)[
29]; R
f = 0.38 (CHCl
3/MeOH 9:1); IR (ATR): ν = 2944m, 1732m, 1640w, 1464m, 1365m, 1244s, 1027m, 986m, 753m cm
−1;
1H-NMR (400 MHz, CDCl
3): δ = 6.77 (t, J = 5.3 Hz, 1H, NH), 5.38 (t, J = 3.6 Hz, 1H, 12-H), 4.51–4.45 (m, 1H, 3-H), 3.61–3.51 (m, 1H, 31-H
a), 3.37–3.27 (m, 1H, 31-H
b), 2.89–2.83 (m, 2H, 32-H), 2.65–2.56 (m, 1H, 18-H), 2.61 (s, 6H, 33-H, 33′-H), 2.03 (s, 3H, Ac), 2.02–1.93 (m, 1H, 16-H
a), 1.93–1.85 (m, 2H, 11-H
a, 11-H
b), 1.72 (t, J = 13.4 Hz, 1H, 19-H
a), 1.66–1.22 (m, 13H, 22-H
a, 1-H
a, 2-H
a, 2-H
b, 16-H
b, 9-H, 22-H
b, 6-H
a, 15-H
a, 7-H
a, 6-H
b, 21-H
a, 7-H
b), 1.21–1.14 (m, 2H, 19-H
b, 21-H
b), 1.14 (s, 3H, 27-H), 1.10–0.97 (m, 2H, 1-H
b, 15-H
b), 0.92 (s, 3H, 25-H), 0.90 (s, 3H, 29-H), 0.89 (s, 3H, 30-H), 0.85 (s, 3H, 23-H), 0.84 (s, 3H, 24-H), 0.83–0.79 (m, 1H, 5-H), 0.74 (s, 3H, 26-H) ppm;
13C-NMR (101 MHz, CDCl
3): δ = 179.3 (C-28), 171.1 (Ac), 144.2 (C-13), 123.1 (C-12), 81.0 (C-3), 57.8 (C-32), 55.4 (C-5), 47.6 (C-9), 46.6 (C-19), 46.5 (C-17), 44.7 (C-33, C-33′), 42.0 (C-14), 41.9 (C-18), 39.6 (C-8), 38.3 (C-1), 37.8 (C-4), 37.0 (C-10), 36.1 (C-31), 34.3 (C-21), 33.2 (C-30), 32.9 (C-22), 32.5 (C-7), 30.8 (C-20), 28.2 (C-23), 27.5 (C-15), 25.9 (C-27), 23.7 (C-29), 23.6 (C-16, C-2, C-11), 21.4 (Ac), 18.3 (C-6), 17.1 (C-24), 16.8 (C-26), 15.6 (C-25) ppm; MS (ESI, MeOH): m/z = 569.5 (100%, [M + H]
+); analysis calcd. for C
36H
60N
2O
3 (568.89): C 76.01, H 10.63, N 4.92; found: C 75.86, H 10.83, N 4.77.
(2α,3β)-N-[2-(Dimethylamino)ethyl]-2,3-diacetyloxy-olean-12-en-28-amide (23), Compound 23 was prepared from 8 according to general procedure B using N,N-dimethylethylene diamine as amino compound. Column chromatography (SiO2, CHCl3/MeOH 9:1) gave 23 (yield: 94%); m.p. 131–136 °C; [α]D = +17.2° (c 0.340, CHCl3); Rf = 0.31 (CHCl3/MeOH 95:5); IR (KBr): ν = 3426br s, 2946s, 2772w, 1744s, 1654m, 1512m, 1462m, 1368m, 1252s, 1156w, 1044m cm−1; 1H-NMR (400 MHz, CDCl3): δ = 6.51 (t, J = 4.4 Hz, 1H, NH), 5.33 (t, J = 3.6 Hz, 1H, 12-H), 5.08 (ddd, J = 11.2, 10.3, 4.6 Hz, 1H, 2-H), 4.75 (d, J = 10.3 Hz, 1H, 3-H), 3.39–3.29 (m, 1H, 31-Ha), 3.18–3.09 (m, 1H, 31-Hb), 2.56–2.48 (m, 1H, 18-H), 2.36 (t, J = 6.0 Hz, 2H, 32-H), 2.21 (s, 6H, 33-H, 33′-H), 2.05 (s, 3H. Ac), 2.04–1.99 (m, 1H, 1-Ha), 1.97 (s, 3H, Ac), 1.96–1.87 (m, 3H, 16-Ha, 11-Ha, 11-Hb), 1.81–1.25 (m, 11H, 19-Ha, 22-Ha, 16-Hb, 9-H, 22-Hb, 15-Ha, 6-Ha, 7-Ha, 6-Hb, 21-Ha, 7-Hb), 1.22–1.12 (m, 2H, 21-Hb, 19-Hb), 1.14 (s, 3H, 27-H), 1.05 (s, 3H, 25-H), 1.10–0.99 (m, 2H, 1-Hb, 15-Hb), 0.99–0.94 (m, 1H, 5-H), 0.90 (s, 9H, 24-H, 29-H, 30-H), 0.89 (s, 3H, 23-H), 0.79 (s, 3H, 26-H) ppm; 13C-NMR (101 MHz, CDCl3): δ = 178.1 (C-28), 170.9 (Ac), 170.7 (Ac), 144.7 (C-13), 122.5 (C-12), 80.7 (C-3), 70.2 (C-2), 57.7 (C-32), 55.0 (C-5), 47.6 (C-9), 46.8 (C-19), 46.5 (C-17), 45.4 (C-33, C-33′), 44.1 (C-1), 42.5 (C-18), 42.2 (C-14), 39.6 (C-8), 39.5 (C-4), 38.2 (C-10), 37.0 (C-31), 34.3 (C-21), 33.2 (C-30), 32.8 (C-22), 32.5 (C-7), 30.9 (C-20), 28.6 (C-23), 27.5 (C-15), 25.8 (C-27), 23.8 (C-16), 23.7 (C-11), 23.7 (C-29), 21.3 (Ac), 21.0 (Ac), 18.4 (C-6), 17.8 (C-24), 17.0 (C-26), 16.7 (C-25) ppm; MS (ESI, MeOH): m/z = 627 (100%, [M + H]+); analysis calcd. for C38H62N2O5 (626.92): C 72.80, H 9.97, N 4.47; found: C 72.63, H 10.14, N4.29.
(3β)-N-[2-(Dimethylamino)-ethyl]-3-hydroxy-olean-12-en-28-amide (27), Compound 27 was prepared from 22 according to general procedure C. Column chromatography (SiO2, CHCl3/MeOH 95:5) gave 27 (yield: 85%); m.p. 187–190 °C; [α]D = +42.6° (c 0.320, MeOH); Rf = 0.29 (CHCl3/MeOH 9:1); IR (ATR): ν = 3383br w, 2943m; 1624m, 1532m, 1431s, 1387s, 1319s, 1186m, 1032m, 996m, 823m, 752s cm−1; 1H-NMR (500 MHz, CDCl3): δ = 6.73 (t, J = 5.2 Hz, 1H, NH), 5.40 (t, J = 3.6 Hz, 1H, 12-H), 3.63–3.53 (m, 1H, 31-Ha), 3.35–3.27 (m, 1H, 31-Hb), 3.21 (dd, J = 11.4, 4.3 Hz, 1H, 3-H), 2.87–2.81 (m, 2H, 32-H), 2.64–2.57 (m, 1H, 18-H), 2.60 (s, 6H, 33-H, 33′-H), 1.99 (ddd, J = 13.7, 13.6, 3.9 Hz, 1H, 16-Ha), 1.93–1.89 (m, 2H, 11-Ha, 11-Hb), 1.73 (t, J = 13.4 Hz, 1H, 19-Ha), 1.68–1.30 (m, 12H, 22-Ha, 16-Hb, 1-Ha, 2-Ha, 2-Hb, 9-H, 22-Hb, 6-Ha, 15-Ha, 7-Ha, 6-Hb, 21-Ha), 1.26 (ddd, J = 12.3, 3.0, 2.9 Hz, 1H, 7-Hb), 1.22–1.14 (m, 2H, 21-Hb, 19-Hb), 1.15 (s, 3H, 27-H), 1.05 (ddd, J = 14.1, 3.6, 3.5 Hz, 1H, 15-Hb), 0.98 (s, 3H, 23-H), 0.98–0.93 (m, 1H, 1-Hb), 0.92 (s, 3H, 29-H), 0.90 (s, 3H, 25-H), 0.90 (s, 3H, 30-H), 0.78 (s, 3H, 24-H), 0.75 (s, 3H, 26-H), 0.74–0.71 (m, 1H, 5-H) ppm; 13C-NMR (126 MHz, CDCl3): δ = 179.2 (C-28), 144.2 (C-13), 123.2 (C-12), 79.1 (C-3), 57.8 (C-32), 55.3 (C-5), 47.7 (C-9), 46.7 (C-19), 46.6 (C-17), 44.7 (C-33, C-33′), 42.1 (C-14), 42.0 (C-18), 39.6 (C-8), 38.9 (C-4), 38.6 (C-1), 37.1 (C-10), 36.0 (C-31), 34.3 (C-21), 33.2 (C-30), 32.9 (C-22), 32.6 (C-7), 30.8 (C-20), 28.2 (C-23), 27.5 (C-15), 27.3 (C-2), 25.9 (C-27), 23.7 (C-16), 23.7 (C-29), 23.6 (C-11), 18.5 (C-6), 17.2 (C-26), 15.7 (C-24), 15.5 (C-25) ppm; MS (ESI, MeOH): m/z = 527.4 (100%, [M + H]+); analysis calcd. for C34H58N2O2 (526.85): C 77.51, H 11.10, N 5.32; found: C 77.37, H 11.31, N 5.16.
(2α,3β)-N-[2-(Dimethylamino)-ethyl]-2,3-dihydroxy-olean-12-en-28-amide (28), Compound 28 was prepared from 23 according to general procedure C. Column chromatography (SiO2, CHCl3/MeOH 9:1) gave 28 (yield: 66%); m.p. 146–149 °C; [α]D = +43.1° (c 0.355, CHCl3); Rf = 0.10 (CHCl3/MeOH 95:5); IR (KBr): ν = 3418br s, 2946s, 2862s, 2826m, 2778m, 1636s, 1522m, 1462s, 1386m, 1364m, 1266w, 1188w, 1158w, 1098w, 1050s cm−1; 1H-NMR (400 MHz, CDCl3): δ = 6.56 (t, J = 4.9 Hz, 1H, NH), 5.35 (t, J = 3.6 Hz, 1H, 12-H), 3.68 (ddd, J = 11.3, 9.5, 4.5 Hz, 1H, 2-H), 3.42–3.32 (m, 1H, 31-Ha), 3.20–3.11 (m, 1H, 31-Hb), 2.99 (d, J = 9.5 Hz, 1H, 3-H), 2.57–2.50 (m, 1H, 18-H), 2.40 (t, J = 6.1 Hz, 2H, 32-H), 2.25 (s, 6H, 33-H, 33‘-H), 2.01–1.90 (m, 4H, 1-Ha, 16-Ha, 11-Ha, 11-Hb), 1.80–1.24 (m, 11H, 19-Ha, 22-Ha, 16-Hb, 9-H, 22-Hb, 15-Ha, 6-Ha, 7-Ha, 6-Hb, 21-Ha, 7-Hb), 1.22–1.16 (m, 2H, 19-Hb, 21-Hb), 1.15 (s, 3H, 27-H), 1.06–0.99 (m, 1H, 15-Hb), 1.02 (s, 3H, 23-H), 0.97 (s, 3H, 25-H), 0.95–0.87 (m, 1H, 1-Hb), 0.90 (s, 6H, 29-H, 30-H), 0.86–0.82 (m, 1H, 5-H), 0.82 (s, 3H, 24-H), 0.78 (s, 3H, 26-H) ppm; 13C-NMR (101 MHz, CDCl3): δ = 178.3 (C-28), 144.6 (C-13), 122.8 (C-12), 84.0 (C-3), 67.0 (C-2), 57.8 (C-32), 55.4 (C-5), 47.7 (C-9), 46.8 (C-19), 46.6 (C-1), 46.5 (C-17), 45.3 (C-33, C-33‘), 42.4 (C-18), 42.2 (C-14), 39.6 (C-8), 39.3 (C-4), 38.3 (C-10), 36.9 (C-31), 34.3 (C-21), 33.2 (C-30), 32.8 (C-22), 32.6 (C-7), 30.9 (C-20), 28.8 (C-23), 27.5 (C-15), 25.9 (C-27), 23.8 (C-16), 23.7 (C-11), 23.7 (C-29), 18.5 (C-6), 17.1 (C-24), 16.9 (C-26), 16.8 (C-25) ppm; MS (ESI, MeOH): m/z = 543 (100%, [M + H]+); analysis calcd. for C34H58N2O3 (542.85): C 75.23, H 10.77, N 5.16; found: C 74.99, H 10.97, N 5.08.
(3β
)-N-(2-Pyrrolidin-1-ylethyl)-3-acetyloxy-olean-12-en-28-amide (
32), Compound
32 was prepared from
7 according to general procedure B using 1-(2-aminoethyl)-pyrrolidine as amino compound. Column chromatography (SiO
2, CHCl
3/MeOH 95:5) gave
32 (yield: 90%); m.p. 175–178 °C (decomp.), lit.: 258 °C (decomp.)[
29]; [α]
D = +47.9° (c 0.315, CHCl
3), Lit.: +49.8° (c 0.350, CHCl
3)[
29]; R
f = 0.39 (CHCl
3/MeOH 9:1); IR (ATR): ν = 2944m, 1731m, 1644w, 1522w, 1365m, 1244s, 1027m, 985m, 752m cm
−1;
1H-NMR (400 MHz, CDCl
3): δ = 6.89 (t, J = 5.6 Hz, 1H, NH), 5.41 (t, J = 3.6 Hz, 1H, 12-H), 4.48 (dd, J = 10.3, 5.7 Hz, 1H, 3-H), 3.90–3.76 (m, 2H, 33-H
a, 33′-H
a), 3.73–3.64 (m, 1H, 31-H
a), 3.51–3.41 (m, 1H, 31-H
b), 3.35–3.18 (m, 2H, 32-H), 2.99–2.86 (m, 2H, 33-H
b, 33′-H
b), 2.64 (dd, J = 13.5, 4.6 Hz, 1H, 18-H), 2.22–1.95 (m, 5H, 34-H, 34′-H, 16-H
a), 2.03 (s, 3H, Ac), 1.94–1.76 (m, 2H, 11-H
a, 11-H
b), 1.70 (t, J = 13.5 Hz, 1H, 19-H
a), 1.65–1.28 (m, 12H, 1-H
a, 2-H
a, 2-H
b, 7-H
a, 7-H
b, 16-H
b, 9-H, 6-H
a, 15-H
a, 22-H
a, 6-H
b, 21-H
a), 1.28–1.15 (m, 3H, 22-H
b, 19-H
b, 21-H
b), 1.13 (s, 3H, 27-H), 1.09–0.97 (m, 2H, 1-H
b, 15-H
b), 0.92 (s, 3H, 25-H), 0.91 (s, 3H, 29-H), 0.89 (s, 3H, 30-H), 0.85 (s, 3H, 23-H), 0.84 (s, 3H, 24-H), 0.84–0.79 (m, 1H, 5-H), 0.71 (s, 3H, 26-H) ppm;
13C-NMR (101 MHz, CDCl
3): δ = 179.9 (C-28), 171.1 (Ac), 143.9 (C-13), 123.3 (C-12), 81.0 (C-3), 55.3 (C-5), 55.1 (C-32), 54.8 (C-33, C-33′), 47.6 (C-9), 46.5 (C-19), 46.5 (C-17), 41.9 (C-14), 41.5 (C-18), 39.5 (C-8), 38.2 (C-1), 37.8 (C-4), 37.0 (C-10), 36.4 (C-31), 34.2 (C-21), 33.2 (C-30), 32.9 (C-7), 32.5 (C-22), 30.8 (C-20), 28.2 (C-23), 27.5 (C-15), 25.9 (C-27), 23.6 (C-2), 23.6 (C-29), 23.6 (C-11), 23.6 (C-16), 23.4 (C-34, C-34′), 21.4 (Ac), 18.3 (C-6), 17.2 (C-26), 16.8 (C-24), 15.5 (C-25) ppm; MS (ESI, MeOH): m/z = 595.5 (100%, [M + H]
+); analysis calcd. for C
38H
62N
2O
3 (594.93): C 76.72, H 10.50, N 4.71; found: C 76.51, H 10.73, N 4.69.
(2α,3β)-N-(2-Pyrrolidin-1-ylethyl)-2,3-diacetyloxy-olean-12-en-28-amide (33), Compound 33 was prepared from 8 according to general procedure B using 1-(2-aminoethyl)-pyrrolidine as amino compound. Column chromatography (SiO2, CHCl3/MeOH 9:1) gave 33 (yield: 96%); m.p. 143–146 °C (decomp.); [α]D = +20.0° (c 0.345, CHCl3); Rf = 0.30 (CHCl3/MeOH 95:5); IR (KBr): ν = 3426br s, 2948s, 2802w, 1744s, 1654m, 1508m, 1460m, 1368m, 1250s, 1152w, 1044m cm−1; 1H-NMR (400 MHz, CDCl3): δ = 6.58 (t, J = 4.3 Hz, 1H, NH), 5.30 (t, J = 3.6 Hz, 1H, 12-H), 5.08 (ddd, J = 11.5, 10.3, 4.7 Hz, 1H, 2-H), 4.74 (d, J = 10.3 Hz, 1H, 3-H), 3.46–3.37 (m, 1H, 31-Ha), 3.24–3.13 (m, 1H, 31-Hb), 2.67–2.44 (m, 7H, 32-H, 33-H, 33′-H, 18-H), 2.04 (s, 3H, Ac), 2.03–1.98 (m, 1H, 1-Ha), 1.97 (s, 3H, Ac), 1.96–1.83 (m, 3H, 11-Ha, 11-Hb, 16-Ha), 1.82–1.76 (m, 4H, 34-H, 34′-H), 1.75–1.23 (m, 11H, 19-Ha, 22-Ha, 16-Hb, 9-H, 22-Hb, 15-Ha, 6-Ha, 7-Ha, 6-Hb, 21-Ha, 7-Hb), 1.24–1.10 (m, 2H, 21-Hb, 19-Hb), 1.14 (s, 3H, 27-H), 1.05 (s, 3H, 25-H), 1.09–0.99 (m, 2H, 1-Hb, 15-Hb), 0.98–0.93 (m, 1H, 5-H), 0.90 (s, 9H, 24-H, 29-H, 30-H), 0.89 (s, 3H, 23-H), 0.77 (s, 3H, 26-H) ppm; 13C-NMR (101 MHz, CDCl3): δ = 177.9 (C-28), 170.8 (Ac), 170.5 (Ac), 144.6 (C-13), 122.3 (C-12), 80.5 (C-3), 70.0 (C-2), 54.8 (C-5), 54.2 (C-32), 53.9 (C-33, C-33′), 47.5 (C-9), 46.7 (C-19), 46.3 (C-17), 43.9 (C-1), 42.3 (C-18), 42.0 (C-14), 39.4 (C-8), 39.3 (C-4), 38.1 (C-10), 37.8 (C-31), 34.2 (C-21), 33.0 (C-30), 32.6 (C-22), 32.3 (C-7), 30.7 (C-20), 28.4 (C-23), 27.3 (C-15), 25.6 (C-27), 23.7 (C-11), 23.7 (C-34, C-34′), 23.6 (C-16), 23.5 (C-29), 21.1 (Ac), 20.9 (Ac), 18.2 (C-6), 17.6 (C-24), 16.8 (C-26), 16.5 (C-25) ppm; MS (ESI, MeOH): m/z = 653 (100%, [M + H]+); analysis calcd. for C40H64N2O5 (652.96): C 73.58, H 9.88, N 4.29; found: C 73.27, H 10.02, N 4.02.
(3β)-N-(2-Pyrrolidin-1-ylethyl)-3-hydroxy-olean-12-en-28-amide (37), Compound 37 was prepared from 32 according to general procedure C. Column chromatography (SiO2, CHCl3/MeOH 95:5) gave 37 (yield: 76%); m.p. 189–192 °C; [α]D = +40.8° (c 0.325, MeOH); Rf = 0.28 (CHCl3/MeOH 9:1); IR (ATR): ν = 3386br w, 2941m, 1631m, 1527m, 1320s, 1031m, 996m, 823m, 752m cm−1; 1H-NMR (500 MHz, CDCl3): δ = 6.94 (t, J = 5.6 Hz, 1H, NH), 5.42 (t, J = 3.7 Hz, 1H, 12-H), 3.88–3.78 (m, 2H, 33-Ha, 33′-Ha), 3.73–3.62 (m, 1H, 31-Ha), 3.51–3.41 (m, 1H, 31-Hb), 3.35–3.22 (m, 2H, 33-Hb, 33′-Hb), 3.20 (dd, J = 11.5, 4.4 Hz, 1H, 3-H), 2.99–2.88 (m, 2H, 32-H), 2.66 (dd, J = 12.9, 3.7 Hz, 1H, 18-H), 2.19–2.05 (m, 4H, 34-H, 34′-H), 2.01 (ddd, J = 13.9, 13.8, 3.9 Hz, 1H, 16-Ha), 1.97–1.84 (m, 2H, 11-Ha, 11-Hb), 1.70 (t, J = 13.4 Hz, 1H, 19-Ha), 1.65–1.29 (m, 12H, 1-Ha, 16-Hb, 2-Ha, 2-Hb, 22-Ha, 22-Hb, 9-H, 6-Ha, 15-Ha, 7-Ha, 6-Hb, 21-Ha), 1.28–1.23 (m, 1H, 7-Hb), 1.21–1.15 (m, 2H, 19-Hb, 21-Hb), 1.14 (s, 3H, 27-H), 1.04 (ddd, J = 14.4, 3.5, 3.4 Hz, 1H, 15-Hb), 0.98 (s, 3H, 23-H), 0.97–0.92 (m, 1H, 1-Hb), 0.91 (s, 3H, 29-H), 0.90 (s, 3H, 25-H), 0.89 (s, 3H, 30-H), 0.77 (s, 3H, 24-H), 0.74–0.70 (m, 1H, 5-H), 0.71 (s, 3H, 26-H) ppm; 13C-NMR (126 MHz, CDCl3): δ = 180.1 (C-28), 143.8 (C-13), 123.4 (C-12), 79.1 (C-3), 55.3 (C-5), 55.1 (C-33, C-33′), 54.9 (C-32), 47.7 (C-9), 46.5 (C-17, C-19), 41.9 (C-14), 41.5 (C-18), 39.6 (C-8), 38.9 (C-4), 38.6 (C-1), 37.1 (C-10), 36.4 (C-31), 34.3 (C-21), 33.2 (C-30), 32.9 (C-22), 32.6 (C-7), 30.8 (C-20), 28.2 (C-23), 27.5 (C-15), 27.3 (C-2), 26.0 (C-27), 23.7 (C-29), 23.6 (C-16), 23.5 (C-11), 23.4 (C-34, C-34′), 18.5 (C-6), 17.2 (C-26), 15.7 (C-24), 15.5 (C-25) ppm; MS (ESI, MeOH): m/z = 553.5 (100%, [M + H]+); analysis calcd. for C36H60N2O2 (552.89): C 78.21, H 10.94, N 5.07; found: C 78.01, H 11.13, N 4.83.
(2α,3β)-N-(2-Pyrrolidin-1-ylethyl)-2,3-dihydroxy-olean-12-en-28-amide (38), Compound 38 was prepared from 33 according to general procedure C. Column chromatography (SiO2, CHCl3/MeOH 9:1) gave 38 (yield: 67%); m.p. 153–156 °C (decomp.); [α]D = +44.3° (c 0.330, CHCl3); Rf = 0.10 (CHCl3/MeOH 95:5); IR (KBr): ν = 3406br s, 2946s, 2878s, 2808m, 1636s, 1522m, 1462m, 1386m, 1364m, 1268w, 1194w, 1150w, 1050m cm−1; 1H-NMR (400 MHz, CDCl3): δ = 6.72 (t, J = 5.1 Hz, 1H, NH), 5.34 (t, J = 3.6 Hz, 1H, 12-H), 3.67 (ddd, J = 11.3, 9.4, 4.4 Hz, 1H, 2-H), 3.53–3.43 (m, 1H, 31-Ha), 3.31–3.22 (m, 1H, 31-Hb), 2.99 (d, J = 9.5 Hz, 1H, 3-H), 2.80–2.63 (m, 6H, 32-H, 33-H, 33′-H), 2.56 (dd, J = 13.4, 4.3 Hz, 1H, 18-H), 2.02–1.89 (m, 4H, 1-Ha, 11-Ha, 11-Hb, 16-Ha), 1.89–1.83 (m, 4H, 34-H, 34′-H), 1.79–1.24 (m, 11H, 19-Ha, 22-Ha, 16-Hb, 9-H, 22-Hb, 6-Ha, 15-Ha, 7-Ha, 6-Hb, 21-Ha, 7-Hb), 1.23–1.11 (m, 2H, 21-Hb, 19-Hb), 1.14 (s, 3H, 27-H), 1.08–0.99 (m, 1H, 15-Hb), 1.02 (s, 3H, 23-H), 0.97 (s, 3H, 25-H), 0.94–0.87 (m, 1H, 1-Hb), 0.90 (s, 3H, 29-H), 0.89 (s, 3H, 30-H), 0.85–0.79 (m, 1H, 5-H), 0.81 (s, 3H, 24-H), 0.75 (s, 3H, 26-H) ppm; 13C-NMR (101 MHz, CDCl3): δ = 178.6 (C-28), 144.5 (C-13), 122.8 (C-12), 84.0 (C-3), 68.9 (C-2), 55.3 (C-5), 54.6 (C-32), 54.2 (C-33, C-33′), 47.7 (C-9), 46.8 (C-19), 46.5 (C-1), 46.5 (C-17), 42.2 (C-18), 42.1 (C-14), 39.6 (C-8), 39.3 (C-4), 38.3 (C-10), 37.6 (C-31), 34.3 (C-21), 33.2 (C-30), 32.8 (C-22), 32.5 (C-7), 30.8 (C-20), 28.8 (C-23), 27.5 (C-15), 25.9 (C-27), 23.8 (C-11), 23.7 (C-29), 23.7 (C-34, C-34′), 23.6 (C-16), 18.5 (C-6), 17.1 (C-26), 16.9 (C-24), 16.8 (C-25) ppm; MS (ESI, MeOH): m/z = 569 (100%, [M + H]+); analysis calcd. for C36H60N2O3 (568.89): C 76.01, H 10.63, N 4.92; found: C 75.86, H 10.90, N 4.77.
(3β)-N-(2-Piperidin-1-ylethyl)-3-acetyloxy-olean-12-en-28-amide (42), Compound 42 was prepared from 7 according to general procedure B using 1-(2-aminoethyl)-piperidine as amino compound. Column chromatography (SiO2, CHCl3/MeOH 9:1) gave 42 (yield: 76%); m.p. = 163–166 °C (decomp.); [α]D = +47.5° (c 0.305, CHCl3); Rf = 0.55 (silica gel, CHCl3/MeOH 9:1); IR (ATR): ν = 2943m, 2864w, 1732m, 1642w, 1523w, 1432m, 1388m, 1365s, 1330m, 1244vs, 1214m, 1149w, 1097w, 1027m, 1007m, 986m, 751m; 1H-NMR (500 MHz, CDCl3): δ = 6.93 (t, J = 5.3 Hz, 1H, NH), 5.42 (dd, J = 3.3 Hz, 3.3 Hz, 1H, 12-H), 4.51–4.44 (m, 1H, 3-H), 3.71 (dq, J = 11.9, 5.9 Hz, 1H, 31-Ha), 3.46 (dq, J = 12.2, 6.0 Hz, 1H, 31-Hb), 3.13 (s, 2H, 32-H), 2.64 (dd, J = 12.9, 3.3 Hz, 1H, 18-H), 2.03 (s, 3H, Ac), 2.02–1.84 (m, 8H, 2-Ha, 2-Hb, 16-Ha, 16-Hb. 33-H, 33′-H), 1.70 (m, 1H, 19-Ha), 1.66–1.40 (m, 15H, 1-Ha, 11-Ha, 22-Ha, 11-Hb, 9-H, 22-Hb, 6-Ha, 15-Ha, 7-Ha, 34-H, 34′-H, 35-H), 1.40–1.30 (m, 2H, 6-Hb, 21-Ha), 1.24 (m, 1H, 7-Hb), 1.22–1.14 (m, 2H, 19-Hb, 21-Hb), 1.13 (s, 3H, 27-H), 1.04 (m, 2H, 1-Hb, 15-Hb), 0.92 (s, 3H, 29-H) 0.92 (s, 3H, 26-H), 0.89 (s, 3H, 30-H), 0.85 (s, 3H, 23-H), 0.84 (s, 3H, 25-H), 0.81 (s, 1H, 5-H), 0.71 (s, 3H, 24-H); 13C-NMR (126 MHz, CDCl3): δ = 179.9 (C-28), 171.1 (Ac), 143.9 (C-13), 123.3 (C-12), 81.0 (C-3), 56.9 (C-32), 55.3 (C-5), 54.5 (C-33 + C-33′), 47.6 (C-9), 46.5 (C-19), 46.5 (C-17), 41.9 (C-14), 41.5 (C-18), 39.5 (C-8), 38.2 (C-1), 37.8 (C-4), 37.0 (C-10), 34.9 (C-31), 34.3 (C-21), 33.2 (C-30), 32.9 (C-22), 32.5 (C-7), 30.8 (C-20), 28.2 (C-23), 27.5 (C-15), 26.0 (C-27), 23.6 (C-2), 23.6 (C-29), 23.6 (C-11), 23.6 (C-16), 23.1 (C-34 + C-34‘), 22.1 (C-35), 21.4 (Ac), 18.3 (C-6), 17.2 (C-24), 16.8 (C-25), 15.5 (C-26); MS (ESI, MeOH): m/z = 609.5 (100%, [M + H]+); analysis calcd. for C39H64N2O3 (608.95): C 76.92, H 10.59, N 4.60; found: C 76.78, H 10.79, N 4.44.
(2α,3β)-N-(2-Piperidin-1-ylethyl)-2,3-diacetyloxy-olean-12-en-28-amide (43), Compound 43 was prepared from 8 according to general procedure B using 1-(2-aminoethyl)-piperidine as amino compound. Column chromatography (SiO2, CHCl3/MeOH 9:1) gave 43 (yield: 71%); m.p. = 130–133 °C (decomp.); [α]D = +15.7° (c 0.35, CHCl3); Rf = 0.47 (silica gel, CHCl3/MeOH 9:1); IR (ATR): ν = 2938m, 2863w, 1741s, 1653m, 1508m, 1503m, 1466m, 1455m, 1443w, 1434w, 1367m, 1303w, 1247vs, 1229vs, 1155w, 1127w, 1041s, 1032s; 1H-NMR (500 MHz, CDCl3) δ = 6.69 (s, 1H, NH), 5.36 (dd, J = 3.4 Hz, 1H, 12-H), 5.08 (td, J = 11.4, 4.6 Hz, 1H, 2-H), 4.74 (d, J = 10.4 Hz, 1H, 3-H), 3.40 (m, 1H, 31-Ha), 3.20 (m, 1H, 31-Hb), 2.55 (d, J = 11.7 Hz, 1H, 18-H), 2.43 (s, 6 H, 32-Ha, 32-Hb, 33-H, 33‘-H), 2.05 (s, 3H, Ac), 1.97 (s, 3H, Ac), 2.07–1.82 (m, 3H, 1-Ha, 16-Ha, 16-Hb), 1.78–1.42 (m, 15H, 19-Ha, 11-Ha, 22-Ha, 9-H, 11-Hb, 34-H, 34‘-H, 6-Ha, 15-Ha, 22-Hb, 35-H, 7-Ha), 1.42–1.13 (m, 5H, 6-Hb, 21-Ha, 7-Hb, 21-Hb, 19-Hb), 1.14 (s, 3H, 27-H), 1.04 (s, 3H, 25-H), 1.09–0.92 (m, 3H, 1-Hb, 15-Hb, 5-H), 0.90 (s, 3H, 29-H), 0.90 (s, 3H, 24-H), 0.90 (s, 3H, 30-H), 0.90 (s, 3H, 23-H), 0.75 (s, 3H, 26-H); 13C-NMR (126 MHz, CDCl3) δ = 178.0, (C-28),170.9 (Ac), 170.7 (Ac), 144.8 (C-13), 122.4 (C-12), 80.7 (C-3), 70.2 (C-2), 57.0 (C-32), 55.0 (C-33 + C-33′), 54.4 (C-5), 47.6 (C-9), 46.9 (C-19), 46.4 (C-17), 44.0 (C-1), 42.3 (C-18), 42.1 (C-14), 39.6 (C-8), 39.5 (C-4), 38.3 (C-10), 35.8 (C-31), 34.3 (C-21), 33.2 (C-30), 32.8 (C-22), 32.3 (C-7), 30.9 (C-20), 28.6 (C-23), 27.4 (C-15), 25.8 (C-38 + C-38′), 25.9 (C-27), 24.2 (C-35), 23.7 (C-16 + C-11), 23.7 (C-29), 21.3 (Ac), 21.0 (Ac), 18.3 (C-6), 17.8 (C-24), 17.0 (C-26), 16.6 (C-25); MS (ESI, MeOH): m/z = 667.5 ([M + H]+, 100%); analysis calcd. for C41H66N2O5 (666.99): C 73.83, H 9.97, N 4.20; found: C 73.69, H 10.18, N 4.01.
(3β)-N-(2-Piperidin-1-ylethyl)-3-acetyloxy-20-oxo-30-norlupan-28-amide (45), Compound 45 was prepared from 10 according to general procedure B using 1-(2-aminoethyl)-piperidine as amino compound. Column chromatography (SiO2, CHCl3/MeOH 9:1) gave 45 (yield: 63%); m.p. = 141–144 °C; [α]D = –9.5° (c 0.34, CHCl3); Rf = 0.43 (silica gel, CHCl3/MeOH); IR (ATR): ν = 2939m, 2867w, 1733m, 1711m, 1657m, 1654m, 1517m, 1468m, 1451m, 1391w, 1367m, 1352m, 1316w, 1302w, 1243vs, 1196m, 1157m, 1130w, 1110w, 1090w, 1073w, 1026m, 979m; 1H-NMR (500 MHz, CDCl3) δ = 6.76 (s, 1H, NH), 4.45 (dd, J = 11.0, 5.1 Hz, 1H, 3-H), 3.40 (m, 4H, 18-H, 31-Ha, 31-Hb), 2.63–2.52 (m, 6H, 32-Ha, 32-Hb, 33-H, 33′-H), 2.21–2.14 (m, 1H, 13-H), 2.15 (s, 3H, 29-H), 2.09–2.00 (m, 3H, 9-H, 16-Ha, 21-Ha), 2.02 (s, 3H, Ac), 1.84–1.78 (dd, 1H, 1-Ha), 1.71–1.53 (m, 8H, 34-H, 34′-H, 19-Ha, 1-Hb, 16-Hb, 2-Ha), 1.53–1.20 (m, 14H, 13-Ha, 2-Hb, 21-Hb, 6-Ha, 35-H, 11-Ha, 12-Ha, 7-Ha, 7-Hb, 6-Hb, 11-Hb, 22-Ha, 22-Hb), 1.20–1.13 (m, 1H, 12-Hb), 1.10–1.00 (m, 2H, 15-Ha, 15-Hb), 0.98 (s, 3H, 27-H), 0.96–0.91 (m, 1H, 19-Hb), 0.88 (s, 3H, 26-H), 0.82 (s, 3H, 23-H), 0.82 (s, 3H, 25-H), 0.81 (s, 3H, 26-H), 0.79–0.75 (m, 1H, 5-H); 13C-NMR (126 MHz, CDCl3) δ = 212.9 (C-20), 176.5 (Ac), 171.0 (C-28), 81.0 (C-3), 57.3 (C-32), 55.8 (C-5), 55.5 (C-17), 54.3 (C-33), 51.4 (C-18), 50.5 (C-9), 50.1 (C-19), 42.4 (C-14), 40.8 (C-8), 38.5 (C-1), 38.0 (C-4), 37.9 (C-10), 37.2 (C-13), 37.0 (C-22), 35.3 (C-31), 34.4 (C-7), 32.9 (C-16), 30.3 (C-29), 29.6 (C-12), 28.8 (C-21), 28.1 (C-23), 27.3 (C-15), 25.3 (C-34), 23.8 (C-35), 23.8 (C-2), 21.4 (Ac), 21.1 (C-11), 18.3 (C-6), 16.6 (C-24), 16.3 (C-25), 16.3 (C-26), 14.8 (C-27); MS (ESI, MeOH): m/z = 611.5 (100%, [M + H]+); analysis calcd. for C38H62N2O4 (610.92): C 74.71, H 10.23, N 4.59; found: C 74.56, H 10.51, N 4.39.
(3β)-N-(2-Piperidin-1-ylethyl)-3-hydroxy-olean-12-en-28-amide (47), Compound 47 was prepared from 42 according to general procedure C. Column chromatography (SiO2, CHCl3/MeOH 95:5) gave 47 (yield: 93%); m.p. = 184–186 °C (decomp.); [α]D = +49.5° (c 0.365, CHCl3); Rf = 0.26 (silica gel, CHCl3/MeOH 9:1); IR (ATR): ν = 3372w, 2941m, 2864m, 1633m, 1527m, 1431s, 1387s, 1378s, 1357s, 1324vs, 1245m, 1212m, 1200w, 1188w, 1138w, 1093w, 1032m, 1006m, 997m, 752m; 1H-NMR (500 MHz, CDCl3) δ = 6.97 (t, J = 5.3 Hz, 1H, NH), 5.43 (dd, J = 3.4, 3.4 Hz, 1H, 12-H), 4.58 (s, 1H, OH), 3.71 (dq, J = 11.7, 5.8 Hz, 1H, 31-Ha), 3.46 (dq, J = 12.2, 5.9 Hz, 1H, 31-Hb), 3.22–3.17 (m, 1H, 3-H), 3.17–3.11 (m, 2H, 32-Ha, 32-Hb), 2.65 (dd, J = 13.4, 3.6 Hz, 2H, 18-H), 2.05–1.95 (m, 2H, 16-Ha, 16-Hb), 1.95–1.84 (m, 8H, 11-Ha, 11-Hb, 34-H, 34‘-H, 35-H), 1.74–1.67 (m, 1H, 19-Ha), 1.65–1.45 (m, 10H, 1-Ha, 16-Ha, 22-Ha, 16-Hb, 22-Hb, 2-Ha, 2-Hb, 9-H, 6-Ha, 15-Ha), 1.45–1.39 (m, 1H, 7-Ha), 1.40–1.29 (m, 2H, 6-Hb, 21-Ha), 1.29–1.22 (m, 1H, 7-Hb), 1.21–1.15 (m, 2H, 19-Hb, 21-Hb), 1.14 (s, 3H, 27-H), 1.07–1.00 (m, 1H, 15-Hb), 0.97 (s, 3H, 23-H), 0.97–0.93 (m, 1H, 1-Hb), 0.92 (s, 3H, 29-H), 0.89 (s, 3H, 25-H), 0.89 (s, 3H, 30-H), 0.77 (s, 3H, 24-H), 0.73 (s, 1H, 5-H), 0.71 (s, 3H, 26-H); 13C-NMR (126 MHz, CDCl3) δ = 179.9 (C-28), 143.8 (C-13), 123.4 (C-12), 79.1 (C-3), 55.3 (C-32), 56.9 (C-5), 54.5 (C-33 + C-33′), 47.7 (C-9), 46.5 (C-19), 46.5 (C-17), 41.9 (C-14), 41.5 (C-18), 39.5 (C-8), 38.9 (C-4), 38.6 (C-1), 37.1 (C-10), 34.9 (C-31), 34.3 (C-21), 33.2 (C-30), 32.9 (C-22), 32.6 (C-7), 30.8 (C-20), 28.2 (C-23), 27.5 (C-15), 27.3 (C-2), 26.0 (C-27), 23.6 (C-29), 23.6 (C-16), 23.6 (C-11), 23.0 (C-34 + C-34′), 22.1 (C-35), 18.4 (C-6), 17.2 (C-26), 15.7 (C-24), 15.5 (C-25); MS (ESI, MeOH): m/z = 567.5 (100%, [M + H]+); analysis calcd. for C37H62N2O2 (566.92): C 78.39, H 11.02, N 4.94; found: C 78.21,H 11.18, N 4.78.
(2α,3β)-N-(2-Piperidin-1-ylethyl)-2,3-dihydroxy-olean-12-en-28-amide (48), Compound 48 was prepared from 43 according to general procedure C. Column chromatography (SiO2, CHCl3/MeOH 9:1) gave 48 (yield: 87%); m.p. = 149–152 °C; [α]D = +45.2° (c 0.31, CHCl3); Rf = 0.21 (silica gel, CHCl3/MeOH 9:1); IR (ATR): ν = 3378m, 2934vs, 2861m, 2853m, 1635s, 1513s, 1459s, 1454s, 1386s, 1378s, 1364m, 1348m, 1328w, 1303m, 1281m, 1266m, 1259m, 1232w, 1209w, 1194m, 1154m, 1129m, 1111w, 1096m, 1084m, 1050vs, 1016m, 992m, 958m, 660m; 1H-NMR (500 MHz, CDCl3) δ = 5.41 (s, 1H, 12-H), 3.74–3.64 (m, 1H, 2-H), 3.59–3.45 (m, 1H, 31-Ha), 3.43–3.25 (m, 1H, 31-Hb), 3.00 (d, J = 9.5 Hz, 1H, 3-H), 2.72–2.60 (m, 1H, 18-H), 2.64–2.36 (m, 2H, 32-Ha, 32-Hb), 2.02–1.92 (m, 4H, 1-Ha, 16-Ha, 16-Hb, 11-Ha), 1.77–1.49 (m, 17H, 1-Hb, 33-H, 33‘-H, 9-H, 11-Hb, 34-H, 34‘-H, 6-Ha, 22-Ha, 35-H, 7-Ha, 15-Ha), 1.50–1.42 (m, 1H, 7-Hb), 1.41–1.31 (m, 2H, 6-Hb, 21-Ha), 1.31–1.28 (m, 1H, 21-Hb), 1.19 (m, 3H, 22-Hb, 19-Ha), 1.15 (s, 3H, 27-H), 1.04 (m, 1H, 15-Hb), 1.03 (s, 3H, 23-H), 0.97 (s, 3H, 25-H), 0.93 (s, 3H, 29-H), 0.91 (s, 3H, 30-H), 0.92-0.88 (m, 1H, 19-Hb), 0.86-0.82 (m, 1H, 5-H), 0.82 (s, 3H, 26-H), 0.74 (s, 3H, 24-H); 13C-NMR (126 MHz, CDCl3) δ = 179.0 (C-28), 144.0 (C-13), 122.8 (C-12), 84.1 (C-3), 69.1 (C-2), 57.2 (C-32), 55.37 (C-33 + C-33‘), 54.5 (C-5), 48.2 (C-17), 47.7 (C-9), 46.5 (C-19), 46.5 (C-1), 43.2 (C-18), 42.1 (C-14), 39.6 (C-8), 39.3 (C-4), 39.1 (C-10), 38.4 (C-31), 34.3 (C-21), 33.2 (C-30), 33.0 (C-22), 32.5 (C-7), 30.9 (C-20), 28.8 (C-23), 27.5 (C-15), 25.8 (C-34 + C-34‘), 26.0 (C-27), 24.1 (C-35), 23.7 (C-16), 23.7 (C-11), 23.7 (C-29), 18.5 (C-6), 17.2 (C-24), 16.9 (C-26), 16.8 (C-25); MS (ESI, MeOH): m/z = 569.5 ([M + H]+, 100%); analysis calcd. for C37H62N2O3 (582.91): C 76.24, H 10.72, N 4.81; found: C 75.97, H 10.93, N 4.57.
(3β)-N-(2-Piperidin-1-ylethyl)-3-hydroxy-20-oxo-30-norlupan-28-amide (50), Compound 50 was prepared from 45 according to general procedure C. Column chromatography (SiO2, CHCl3/MeOH 9:1) gave 50 (yield: 86%); m.p. = 153–156 °C (decomp.); [α]D = –21.2° (c 0.115, CHCl3); Rf = 0.16 (silica gel, CHCl3/MeOH); IR (ATR): ν = 3371w, 2934vs, 2865m, 1706m, 1643s, 1524s, 1519s, 1466s, 1450s, 1387m, 1376s, 1356s, 1328m, 1319m, 1301m, 1277m, 1245s, 1197s, 1161m, 1131m, 1109m, 1085m, 1046s, 1035s, 1003m, 987m, 973m; 1H-NMR (500 MHz, CDCl3) δ = 3.67–3.55 (m, 2H, 31-Ha, 31-Hb), 3.27 (td, J = 11.2, 3.9 Hz, 2H, 1-Ha, 1-Hb), 3.17–3.07 (m, 3H, 32-Ha, 32-Hb, 3-H), 2.20–2.12 (m, 1H, 13-H), 2.09 (s, 3H, 28-H), 2.10–2.02 (m, 1H, 16-Ha), 2.00–1.94 (m, 1H, 18-H), 1.91–1.73 (m, 6H, 12-Ha, 34-H, 34′-H, 22-Ha), 1.62–1.06 (m, 22H, 19-Ha, 33-H, 33′-H, 15-Ha, 15-Hb, 2-Ha, 16-Hb, 6-Ha, 22-Hb, 12-Hb, 11-Ha, 6-Hb, 7-Ha, 35-H, 7-Hb, 21-Ha, 9-H, 11-Hb, 21-Hb), 1.05–0.92 (m, 1H, 2-Hb), 0.90 (s, 3H, 27-H), 0.89 (s, 3H, 23-H), 0.82 (s, 3H, 25-H), 0.85–0.78 (m, 1H, 19-Hb), 0.74 (s, 3H, 24-H), 0.68 (s, 3H, 26-H), 0.63–0.58 (m, 1H, 5-H); 13C-NMR (126 MHz, CDCl3) δ = 212.7 (C-20), 177.8 (C-30), 78.7 (C-3), 56.9 (C-32), 55.6 (C-17), 55.3 (C-5), 54.0 (C-33), 51.1 (C-19), 50.4 (C-9), 50.0 (C-18), 42.1 (C-14), 40.6 (C-8), 38.8 (C-4), 38.6 (C-1), 37.6 (C-22), 37.1 (C-10), 36.7 (C-13), 34.3 (C-7), 34.2 (C-31), 32.1 (C-16), 30.0 (C-28), 29.4 (C-21), 28.4 (C-12), 28.0 (C-23), 27.3 (C-15), 27.2 (C-2), 22.8 (C-34), 21.8 (C-35), 20.90 (C-11), 18.2 (C-6), 16.1 (C-24), 16.1 (C-25), 15.4 (C-26), 14.6 (C-27); MS (ESI, MeOH): m/z = 568.5 (100%, [M + H]+); analysis calcd. for C36H60N2O3 (568.89): C 76.01, H 10.63, N 4.92; found: C 75.93, H 10.46, N 4.71.