Chiral Stationary Phases for Liquid Chromatography: Recent Developments
Abstract
:1. Introduction
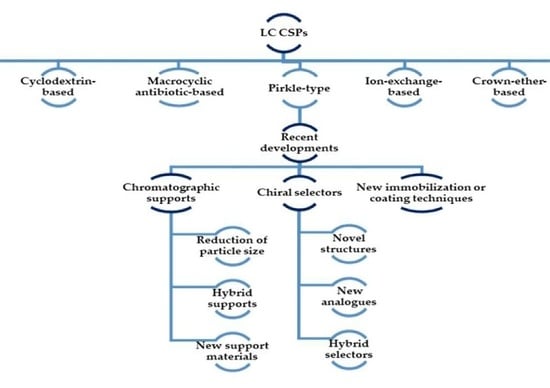
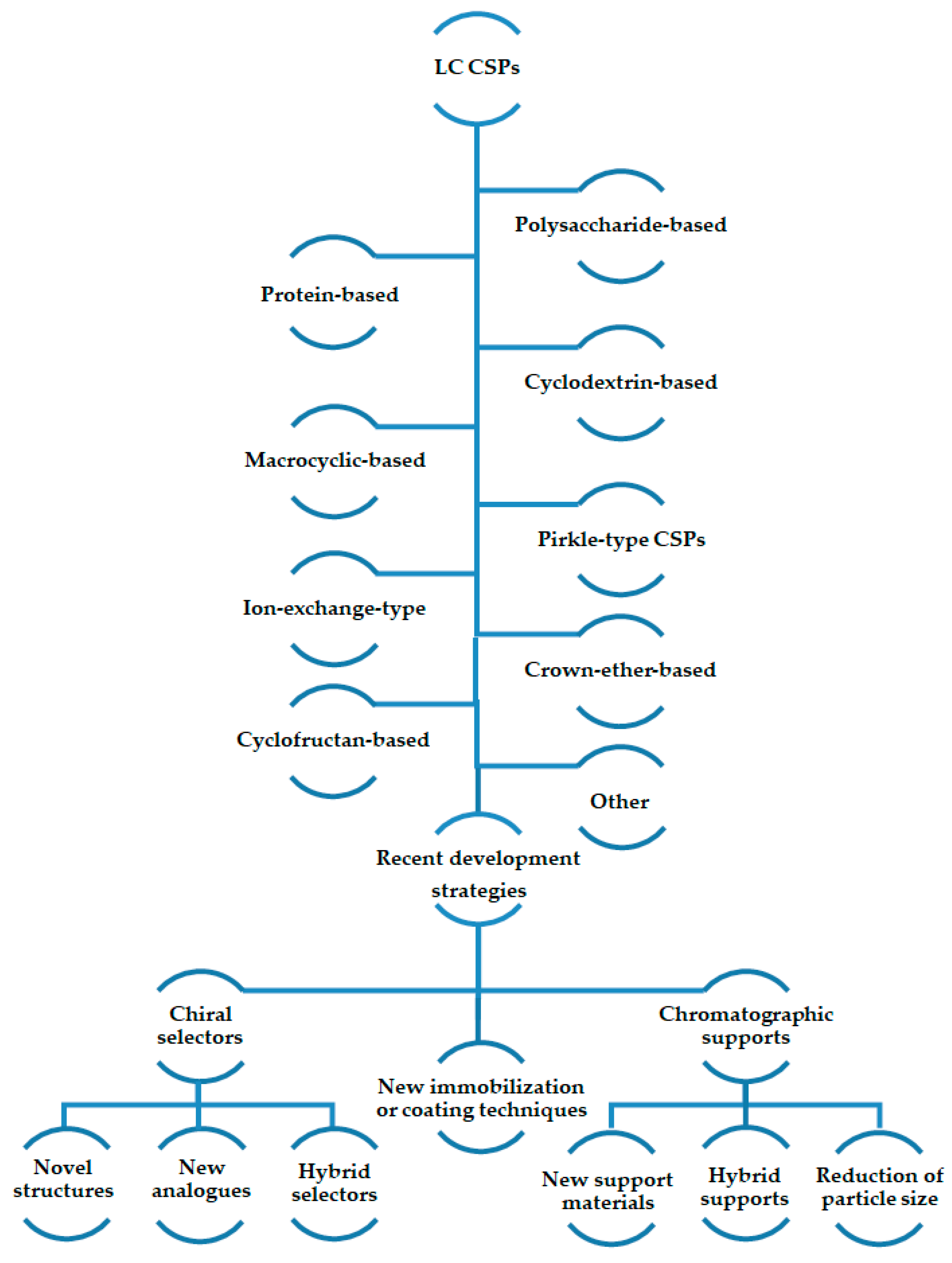
2. Chiral Stationary Phases: Recent Developments
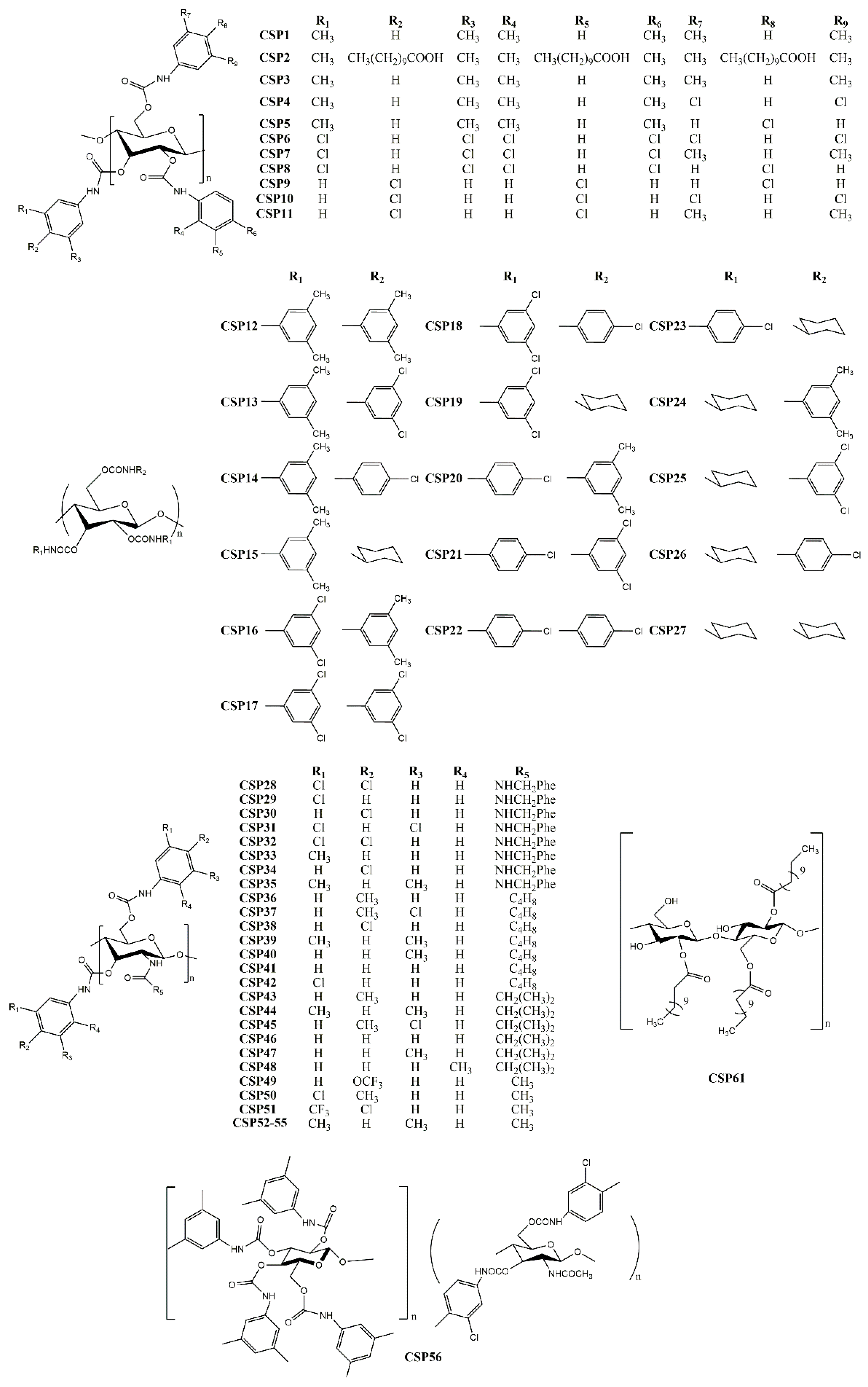
2.1. Polysaccharide-Based CSPs
2.2. Protein-Based CSPs
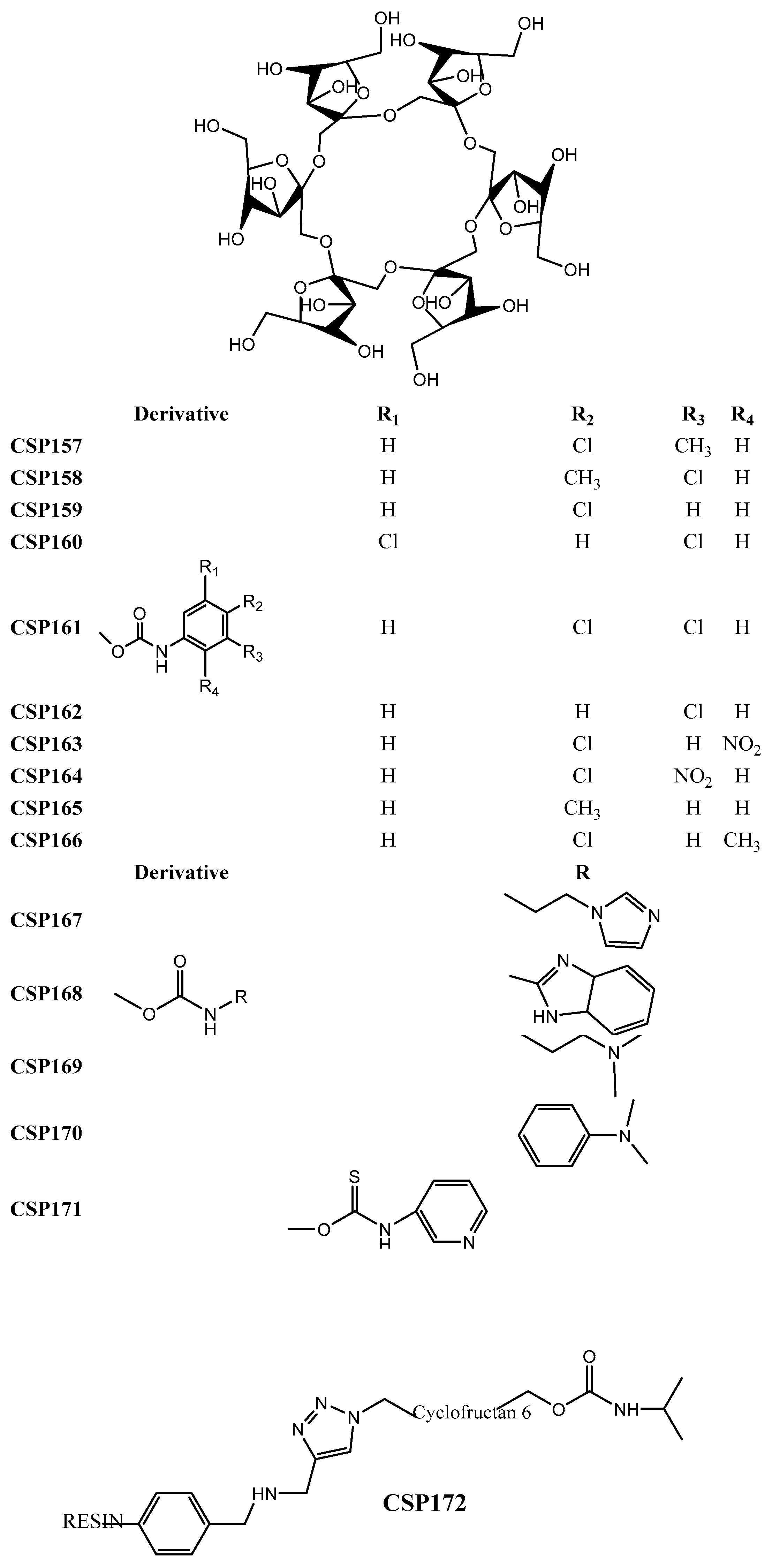
2.3. Cyclodextrin-Based CSPs
2.4. Macrocyclic Antibiotic-Based CSPs
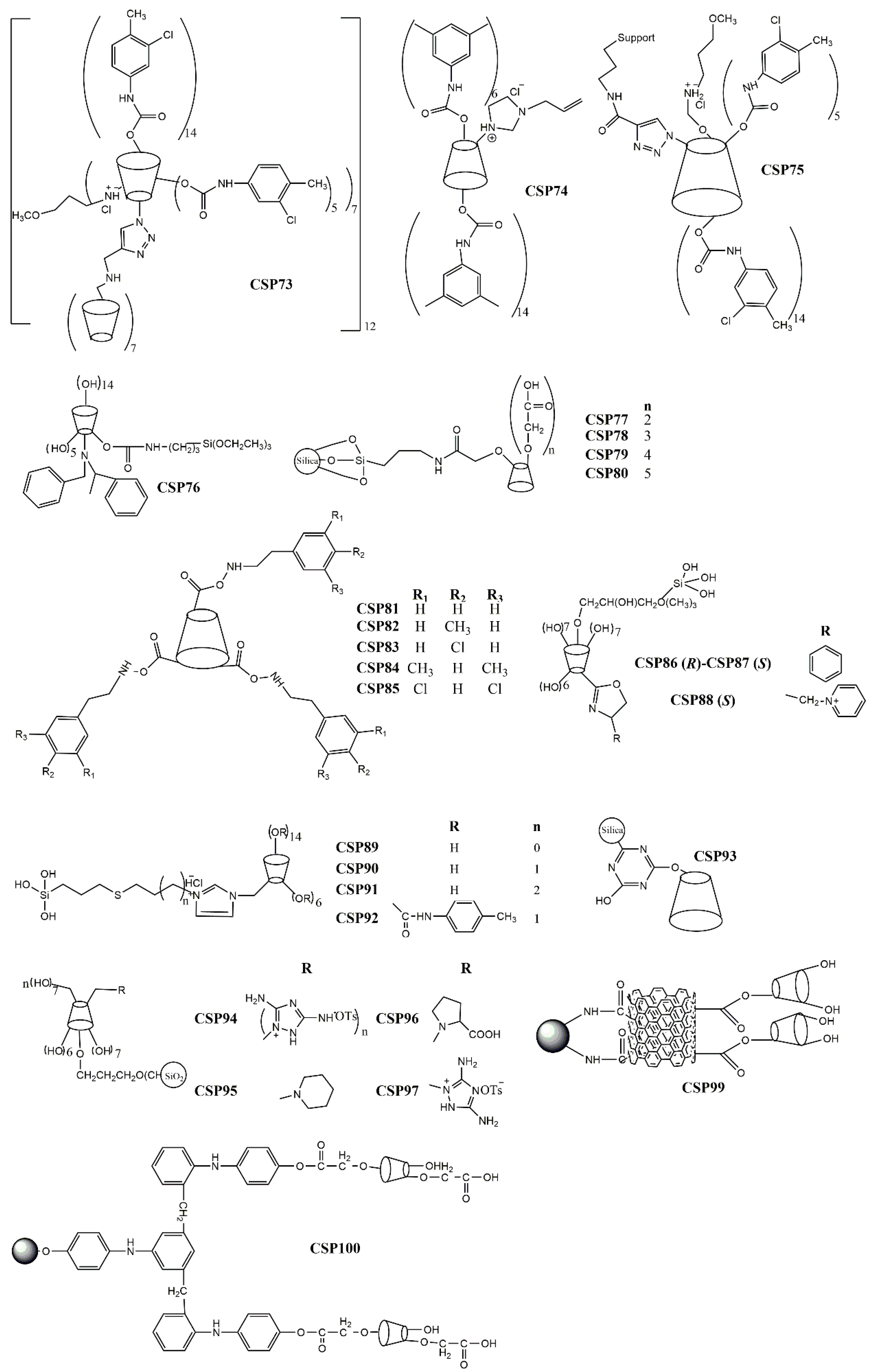
2.5. Brush-Type or Pirkle-Type CSPs
2.6. Ion-Exchange-Type CSPs
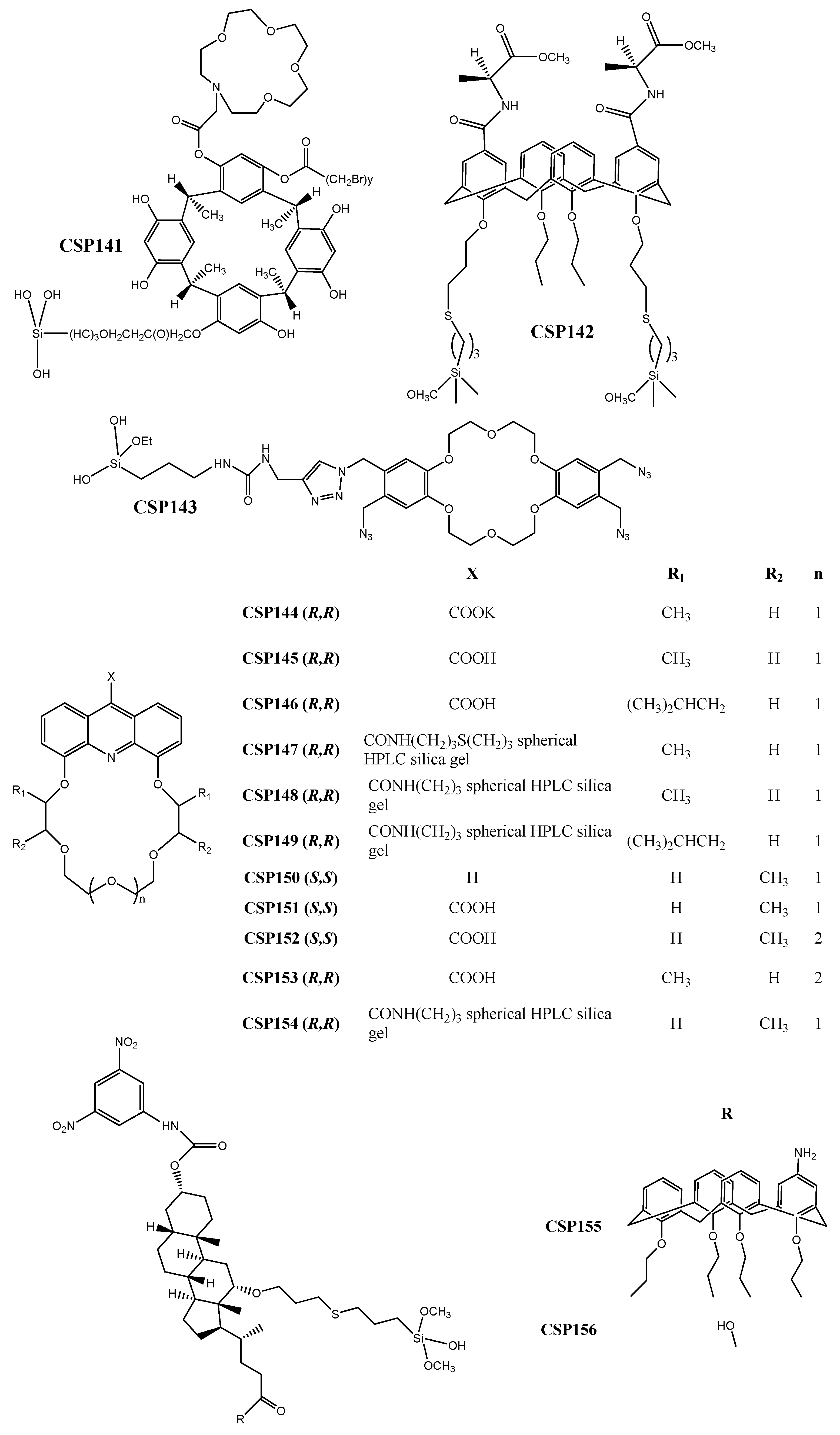
2.7. Crown-Ether-Based CSPs
2.8. Cyclofructan-Based CSPs
2.9. Molecularly-Imprinted CSPs
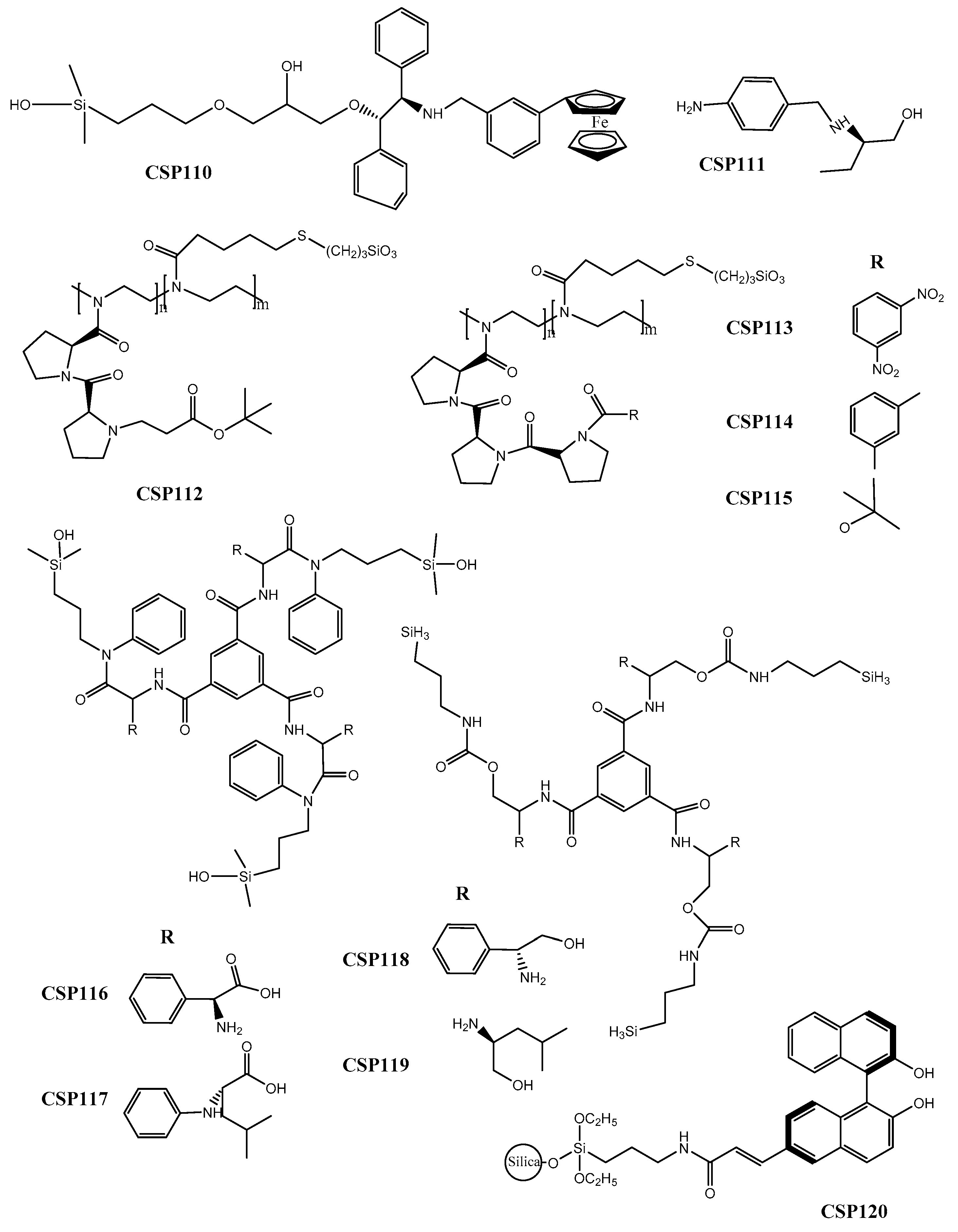
2.10. Other CSPs
3. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ward, T.J.; Ward, K.D. Chiral separations: A review of current topics and trends. Anal. Chem. 2012, 84, 626–635. [Google Scholar] [CrossRef] [PubMed]
- Phyo, Y.Z.; Cravo, S.; Palmeira, A.; Tiritan, M.E.; Kijjoa, A.; Pinto, M.M.M.; Fernandes, C. Enantiomeric resolution and docking studies of chiral xanthonic derivatives on chirobiotic columns. Molecules 2018, 23, 142. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, C.; Palmeira, A.; Santos, A.; Tiritan, M.E.; Afonso, C.; Pinto, M.M. Enantioresolution of Chiral Derivatives of Xanthones on (S,S)-Whelk-O1 and L-Phenylglycine Stationary Phases and Chiral Recognition Mechanism by Docking Approach for (S,S)-Whelk-O1. Chirality 2013, 25, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Speybrouck, D.; Lipka, E. Preparative supercritical fluid chromatography: A powerful tool for chiral separations. J. Chromatogr. A 2016, 1467, 33–55. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, L.; Naidu, H.; Asokan, K.; Shaik, K.M.; Kaspady, M.; Arunachalam, P.; Wu, D.R.; Mathur, A.; Sarabu, R. Integrating a post-column makeup pump into preparative supercritical fluid chromatography systems to address stability and recovery issues during purifications. J. Chromatogr. A 2017, 1511, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Zehani, Y.; Lemaire, L.; Millet, R.; Lipka, E. Small scale separation of isoxazole structurally related analogues by chiral supercritical fluid chromatography. J. Chromatogr. A 2017, 1505, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Bishop, R. Aspects of crystallization and chirality. In Chirality in Supramolecular Assemblies: Causes and Consequences; John Wiley & Sons, Ltd.: London, UK, 2016; pp. 65–93. [Google Scholar] [CrossRef]
- Kovalenko, V.N.; Kozyrkov, Y.Y. A simple method for resolution of endo-/exo-monoesters of trans-norborn-5-ene-2,3-dicarboxylic acids into their enantiomers. Chirality 2015, 27, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, C.; Tiritan, M.E.; Pinto, M.M.M. Chiral separation in preparative scale: A brief overview of membranes as tools for enantiomeric separation. Symmetry 2017, 9, 206. [Google Scholar] [CrossRef]
- Afonso, C.A.M.; Crespo, J.G. Recent advances in chiral resolution through membrane-based approaches. Angew. Chem. Int. Ed. 2004, 43, 5293–5295. [Google Scholar] [CrossRef] [PubMed]
- Andrushko, V.; Andrushko, N. Stereoselective Synthesis of Drugs and Natural Products; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013; Volume 1. [Google Scholar]
- Mun, S.; Wang, N.H.L. Improvement of the performances of a tandem simulated moving bed chromatography by controlling the yield level of a key product of the first simulated moving bed unit. J. Chromatogr. A 2017, 1488, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Song, J.Y.; Kim, K.M.; Lee, C.H. High-performance strategy of a simulated moving bed chromatography by simultaneous control of product and feed streams under maximum allowable pressure drop. J. Chromatogr. A 2016, 1471, 102–117. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, M.; Ohkuma, T.; Tokunaga, M.; Noyori, R. Dynamic kinetic resolution in binap-ruthenium(ii) catalyzed hydrogenation of 2-substituted 3-oxo carboxylic esters. Tetrahedron Asymmetry 1990, 1, 1–4. [Google Scholar] [CrossRef]
- Schoemaker, H.E.; Mink, D.; WubboLts, M.G. Dispelling the myths—Biocatalysis in industrial synthesis. Science 2003, 299, 1694–1697. [Google Scholar] [CrossRef] [PubMed]
- Miller, L.; Potter, M. Preparative chromatographic resolution of racemates using HPLC and SFC in a pharmaceutical discovery environment. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2008, 875, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Leek, H.; Andersson, S. Preparative scale resolution of enantiomers enables accelerated drug discovery and development. Molecules 2017, 22, 158. [Google Scholar] [CrossRef] [PubMed]
- Kirkpatrick, A.D.; Fain, M.; Yang, J.; Trehy, M. Enantiomeric impurity analysis using circular dichroism spectroscopy with United States Pharmacopeia Liquid Chromatographic Methods. J. Pharm. Biomed. Anal. 2018, 156, 366–371. [Google Scholar] [CrossRef] [PubMed]
- Batista, A.N.L.; dos Santos, F.M.; Batista, J.M.; Cass, Q.B. Enantiomeric mixtures in natural product chemistry: Separation and absolute configuration assignment. Molecules 2018, 23, 492. [Google Scholar] [CrossRef] [PubMed]
- Dawood, A.W.H.; de Souza, R.O.M.A.; Bornscheuer, U.T. Asymmetric synthesis of chiral halogenated amines using amine transaminases. ChemCatChem 2018, 10, 951–955. [Google Scholar] [CrossRef]
- Chen, J.; Xu, X.; He, Z.; Qin, H.; Sun, W.; Fan, B. Nickel/Zinc iodide co-catalytic asymmetric [2 + 2] cycloaddition reactions of azabenzonorbornadienes with terminal alkynes. Adv. Synth. Catal. 2018, 360, 427–431. [Google Scholar] [CrossRef]
- Wu, S.; Harada, S.; Morikawa, T.; Nishidda, A. Enantioselective total synthesis of a natural hydrocarbazolone alkaloid, identification of its stereochemistry, and revision of its spectroscopic data. Tetrahedron Asymmetry 2017, 28, 1083–1088. [Google Scholar] [CrossRef]
- Gholami, H.; Zhang, J.; Anyika, M.; Borhan, B. Absolute stereochemical determination of asymmetric sulfoxides via central to axial induction of chirality. Org. Lett. 2017, 19, 1722–1725. [Google Scholar] [CrossRef] [PubMed]
- Toki, H.; Ichikawa, T.; Mizuno-Yasuhira, A.; Yamaguchi, J. A rapid and sensitive chiral LC-MS/MS method for the determination of ketamine and norketamine in mouse plasma, brain and cerebrospinal fluid applicable to the stereoselective pharmacokinetic study of ketamine. J. Pharm. Biomed. Anal. 2018, 148, 288–297. [Google Scholar] [CrossRef] [PubMed]
- Jiao, H.; Li, Y.; Sun, L.; Zhang, H.; Yu, L.; Yuan, Z.; Xie, L.; Chen, J.; Wang, Y. A chiral LC-MS/MS method for the enantioselective determination of R-(+)- and S-(−)-pantoprazole in human plasma and its application to a pharmacokinetic study of S-(−)-pantoprazole sodium injection. Biomed. Chromatogr. 2017, 31, e3980. [Google Scholar] [CrossRef] [PubMed]
- Silva, B.; Pereira, J.A.; Cravo, S.; Araújo, A.M.; Fernandes, C.; Pinto, M.M.M.; Pinho, P.G.; Remião, F. Multi-milligram resolution and determination of absolute configuration of pentedrone and methylone enantiomers. J. Chromatogr. B 2018, 110, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, C.; Santos, C.; Gonçalves, V.; Ramos, A.; Afonso, C.; Tiritan, M.E. Chiral drug analysis in forensic chemistry: An overview. Molecules 2018, 23, 262. [Google Scholar] [CrossRef] [PubMed]
- Silva, B.; Fernandes, C.; Guedes-de-Pinho, P.; Remião, F. Chiral resolution and enantioselectivity of synthetic cathinones: A brief review. J. Anal. Toxicol. 2018, 42, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.R.; Maia, A.S.; Cass, Q.B.; Tiritan, M.E. Enantioseparation of chiral pharmaceuticals in biomedical and environmental analyses by liquid chromatography: An overview. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2014, 968, 8–21. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, C.; Ribeiro, A.R.; Maia, A.S.; Tiritan, M.E. Occurrence of chiral bioactive compounds in the aquatic environment: A review. Symmetry 2017, 9, 215. [Google Scholar] [CrossRef]
- Ribeiro, A.R.; Gonçalves, V.M.; Maia, A.S.; Ribeiro, C.; Castro, P.M.; Tiritan, M.E. Dispersive liquid–liquid microextraction and HPLC to analyse fluoxetine and metoprolol enantiomers in wastewaters. Environ. Chem. Lett. 2015, 13, 203–210. [Google Scholar] [CrossRef]
- Cosimelli, B.; Greco, G.; Laneri, S.; Novellino, E.; Sacchi, A.; Collina, S.; Rossi, D.; Cosconati, S.; Barresi, E.; Taliani, S.; et al. Studies on enantioselectivity of chiral 4-acetylamino-6-alkyloxy-2-alkylthiopyrimidines acting as antagonists of the human adenosine receptor. MedChemComm 2018, 9, 81–86. [Google Scholar] [CrossRef] [PubMed]
- George, R.; Haywood, A.; Good, P.; Hennig, S.; Khan, S.; Norris, R.; Hardy, J. Can saliva and plasma methadone concentrations be used for enantioselective pharmacokinetic and pharmacodynamic studies in patients with advanced cancer? Clin. Ther. 2017, 39, 1840–1848. [Google Scholar] [CrossRef] [PubMed]
- Ilisz, I.; Péter, A.; Lindner, W. State-of-the-art enantioseparations of natural and unnatural amino acids by high-performance liquid chromatography. TrAC-Trends Anal. Chem. 2016, 81, 11–22. [Google Scholar] [CrossRef]
- Tanwar, S.; Bhushan, R. Enantioresolution of Amino Acids: A Decade’s Perspective, Prospects and Challenges. Chromatographia 2015, 78, 1113–1134. [Google Scholar] [CrossRef]
- Hancu, G.; Cârcu-Dobrin, M.; Budău, M.; Rusu, A. Analytical methodologies for the stereoselective determination of fluoxetine: An overview. Biomed. Chromatogr. 2018, 32, e4040. [Google Scholar] [CrossRef] [PubMed]
- Ali, I.; Suhail, M.; Asnin, L. Chiral separation of quinolones by liquid chromatography and capillary electrophoresis. J. Sep. Sci. 2017, 40, 2863–2882. [Google Scholar] [CrossRef] [PubMed]
- Batra, S.; Bhushan, R. Resolution of enantiomers of bupropion and its metabolites by liquid chromatography. Biomed. Chromatogr. 2016, 30, 670–682. [Google Scholar] [CrossRef] [PubMed]
- Felix, G.; Berthod, A. Commercial chiral stationary phases for the separations of clinical racemic drugs. Sep. Purif. Rev. 2007, 36, 285–481. [Google Scholar] [CrossRef]
- Shaikh, S.; Muneera, M.S.; Thusleem, O.A. Chiral chromatography and its application to the pharmaceutical industry. Pharm. Rev. 2009, 7, 1371–1381. [Google Scholar]
- Peluso, P.; Mamane, V.; Aubert, E.; Cossu, S. Recent trends and applications in liquid-phase chromatography enantioseparation of atropisomers. Electrophoresis 2017, 38, 1830–1850. [Google Scholar] [CrossRef]
- Bertucci, C.; Tedesco, D. Human serum albumin as chiral selector in enantioselective high-performance liquid chromatography. Curr. Med. Chem. 2017, 24, 743–757. [Google Scholar] [CrossRef] [PubMed]
- Lämmerhofer, M. Chiral recognition by enantioselective liquid chromatography: Mechanisms and modern chiral stationary phases. J. Chromatogr. A 2010, 1217, 814–856. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, C.; Tiritan, M.E.; Cravo, S.; Phyo, Y.Z.; Kijjoa, A.; Silva, A.M.S.; Cass, Q.B.; Pinto, M.M.M. New chiral stationary phases based on xanthone derivatives for liquid chromatography. Chirality 2017, 29, 430–442. [Google Scholar] [CrossRef] [PubMed]
- Scriba, G.K.E. Chiral recognition in separation science—An update. J. Chromatogr. A 2016, 1467, 56–78. [Google Scholar] [CrossRef] [PubMed]
- Kalíková, K.; Riesová, M.; Tesařová, E. Recent chiral selectors for separation in HPLC and CE. Cent. Eur. J. Chem. 2011, 10, 450–471. [Google Scholar] [CrossRef]
- Henderson, G.M.; Rule, H.G. A new method of resolving a racemic compound. Nature 1938, 142, 917–918. [Google Scholar] [CrossRef]
- Hesse, G.; Hagel, R. Über Inclusions-Chromatographie und ein neues Retentionsprinzip für benzolderivative. Chromatographia 1976, 9, 62–68. [Google Scholar] [CrossRef]
- Chen, X.; Yamamoto, C.; Okamoto, Y. Polysaccharide derivatives as useful chiral stationary phases in high-performance liquid chromatography. Pure Appl. Chem. 2007, 79, 1561–1573. [Google Scholar] [CrossRef]
- Ali, I.; Saleem, K.; Hussain, I.; Gaitonde, V.D.; Aboul-Enein, H.Y. Polysaccharides chiral stationary phases in liquid chromatography. Sep. Purif. Rev. 2009, 38, 97–147. [Google Scholar] [CrossRef]
- Ribeiro, J.; Tiritan, M.E.; Pinto, M.M.M.; Fernandes, C. Chiral stationary phases for liquid chromatography based on chitin and chitosan-derived marine polysaccharides. Symmetry 2017, 9, 190. [Google Scholar] [CrossRef]
- Chankvetadze, B. Recent developments on polysaccharide-based chiral stationary phases for liquid-phase separation of enantiomers. J. Chromatogr. A 2012, 1269, 26–51. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, Y.; Kawashima, M.; Hatada, K. Useful chiral packing materials for high-performance liquid chromatographic resolution of enantiomers: Phenylcarbamates of polysaccharides coated on silica gel. J. Am. Chem. Soc. 1984, 106, 5357–5359. [Google Scholar] [CrossRef]
- Park, J.H.; Whang, Y.C.; Jung, Y.J.; Okamoto, Y.; Yamamoto, C.; Carr, P.W.; McNeff, C.V. Separation of racemic compounds on amylose and cellulose dimethylphenylcarbamate-coated zirconia in HPLC. J. Sep. Sci. 2003, 26, 1331–1336. [Google Scholar] [CrossRef]
- Moldoveanu, S.C.; David, V. Stationary phases and columns for chiral chromatography. In Selection of the HPLC Method in Chemical Analysis; Elsevier: Amsterdam, The Netherlands, 2017; pp. 363–376. [Google Scholar]
- Ali, I.; Aboul-Enein, H.Y. Impact of immobilized polysaccharide chiral stationary phase on enantiomeric separations. J. Sep. Sci. 2006, 29, 762–769. [Google Scholar] [CrossRef] [PubMed]
- Ali, I.; Aboul-Enein, H. Immobilized Polysaccharide CSPs: An Advancement in Enantiomeric Separations. Curr. Pharm. Anal. 2007, 3, 71–82. [Google Scholar] [CrossRef]
- Ikai, T.; Okamoto, Y. Structure control of polysaccharide derivatives for efficient separation of enantiomers by chromatography. Chem. Rev. 2009, 109, 6077–6101. [Google Scholar] [CrossRef] [PubMed]
- Al-Othman, Z.A.; Ali, I.; Asim, M.; Khan, T.A. Recent trends in chiral separations on immobilized polysaccharides CSPs. Comb. Chem. High Throughput Screen. 2012, 15, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Francotte, E.; Huynh, D. Immobilized halogenophenylcarbamate derivatives of cellulose as novel stationary phases for enantioselective drug analysis. J. Pharm. Biomed. Anal. 2002, 27, 421–429. [Google Scholar] [CrossRef]
- Khatiashvili, T.; Kakava, R.; Matarashvili, I.; Tabani, H.; Fanali, C.; Volonterio, A.; Farkas, T.; Chankvetadze, B. Separation of enantiomers of selected chiral sulfoxides with cellulose tris(4-chloro-3-methylphenylcarbamate)-based chiral columns in high-performance liquid chromatography with very high separation factor. J. Chromatogr. A 2018, 1545, 22–24. [Google Scholar] [CrossRef] [PubMed]
- Cass, Q.B.; Bassi, A.L.C.; Calafatti, S.A.; Matlin, S.A.; Tiritan, M.E.; de Campos, L.M.M. Carbohydrate carbamate coated onto microporous silica: Application to chiral analysis of commercial pharmaceutical drugs. Chirality 1996, 8, 143–146. [Google Scholar] [CrossRef]
- Matlin, S.A.; Tiritan, M.E.; Cass, Q.B.; Boyd, D.R. Enantiomeric resolution of chiral sulfoxides on polysaccharide phases by HPLC. Chirality 1996, 8, 147–152. [Google Scholar] [CrossRef]
- Cass, Q.B.; Degani, A.L.G.; Tiritan, M.E.; Matlin, S.A.; Curran, D.P.; Balog, A. Enantiomeric resolution by HPLC of axial chiral amides using amylose tris[(S)-1-phenylethylcarbamate]. Chirality 1997, 9, 109–112. [Google Scholar] [CrossRef]
- Cass, Q.B.; Watanabe, C.S.; Rabi, J.A.; Bottari, P.Q.; Costa, M.R.; Nascimento, R.M.; Cruz, J.E.; Ronald, R.C. Polysaccharide-based chiral phase under polar organic mode of elution in the determination of the enantiomeric purity of emtricitabine an anti-HIV analogue nucleoside. J. Pharm. Biomed. Anal. 2003, 33, 581–587. [Google Scholar] [CrossRef]
- Lourenço, T.C.; Armstrong, D.W.; Cass, Q.B. Enantiomeric Resolution of a Chiral Sulfoxide Series by LC on Synthetic Polymeric Columns with Multimodal Elution. Chromatographia 2010, 71, 361–372. [Google Scholar] [CrossRef]
- Jie, Z.; Qiuzheng, D.; Suzhen, Z.; Fang, S.; Xinyu, L.; Zhenzhong, Z. Enantioseparation of Three Important Intermediates of Tanikolide with Immobilized Cellulose Chiral Stationary Phase. J. Chromatogr. Sci. 2015, 53, 959–962. [Google Scholar] [CrossRef]
- Zacharis, C.K.; Vastardi, E. A Validated LC Method for the Determination of Enantiomeric Purity of Clopidogrel Intermediate Using Amylose-Based Stationary Phase. Chromatographia 2015, 78, 819–824. [Google Scholar] [CrossRef]
- Ramisetti, N.R.; Arnipalli, M.S.; Nimmu, N.V.; Bondigalla, R. UHPLC Determination of Besifloxacin Enantiomers on Immobilized Amylose tris(3,5-dichlorophenylcarbamate) Chiral Stationary Phase. Chromatographia 2017, 80, 1509–1515. [Google Scholar] [CrossRef]
- Tiritan, M.E.; Cass, Q.B.; Del Alamo, A.; Matlin, S.A.; Grieb, S.J. Preparative enantioseparation on polysaccharide phase using microporous silica as a support. Chirality 1998, 10, 573–577. [Google Scholar] [CrossRef]
- Lourenco, T.C.; Batista, J.M., Jr.; Furlan, M.; He, Y.; Nafie, L.A.; Santana, C.C.; Cass, Q.B. Albendazole sulfoxide enantiomers: Preparative chiral separation and absolute stereochemistry. J. Chromatogr. A 2012, 1230, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Belaz, K.R.; Coimbra, M.; Barreiro, J.C.; Montanari, C.A.; Cass, Q.B. Multimilligram enantioresolution of sulfoxide proton pump inhibitors by liquid chromatography on polysaccharide-based chiral stationary phase. J. Pharm. Biomed. Anal. 2008, 47, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Cass, Q.B.; Oliveira, R.V. Separation of Multi-Milligram Quantities of Gossypol Enantiomers on Polysaccharide-Based Stationary Phases. J. Liq. Chromatogr. Relat. Technol. 2007, 25, 819–829. [Google Scholar] [CrossRef]
- Heydari, R.; Shamsipur, M. Enantiomeric Separation and Quantitation of Tenofovir Disoproxil Fumarate Using Amylose-Based Chiral Stationary Phases by High-Performance Liquid Chromatography. Acta Chromatogr. 2015, 27, 583–595. [Google Scholar] [CrossRef]
- Lal, M.; Bhushan, R. Analytical and semi-preparative enantioresolution of (RS)-ketorolac from pharmaceutical formulation and in human plasma by HPLC. Biomed. Chromatogr. 2016, 30, 1526–1534. [Google Scholar] [CrossRef] [PubMed]
- Sadutto, D.; Ferretti, R.; Zanitti, L.; Casulli, A.; Cirilli, R. Analytical and semipreparative high performance liquid chromatography enantioseparation of bicalutamide and its chiral impurities on an immobilized polysaccharide-based chiral stationary phase. J. Chromatogr. A 2016, 1445, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, Y.; Aburatani, R.; Miura, S.; Hatada, K. Chiral stationary phases for HPLC: Cellulose tris(3,5-dimethylphenylcarbamate) and tris(3,5-dichlorophenylcarbamate) chemically bonded to silica gel. J. Liq. Chromatogr. 1987, 10, 1613–1628. [Google Scholar] [CrossRef]
- Witte, D.T.; Bruggeman, F.J.; Franke, J.P.; Copinga, S.; Jansen, J.M.; De Zeeuw, R.A. Comparison between cellulose and amylose tris(3,5-dimethylphenylcarbamate) chiral stationary phases for enantiomeric separation of 17 amidotetralins. Chirality 1993, 5, 545–553. [Google Scholar] [CrossRef]
- Matlin, S.A.; Tiritan, M.E.; Crawford, A.J.; Cass, Q.B.; Boyd, D.R. HPLC with carbohydrate carbamate chiral phases: Influence of chiral phase structure on enantioselectivity. Chirality 1994, 6, 135–140. [Google Scholar] [CrossRef]
- Franco, P.; Senso, A.; Oliveros, L.; Minguillón, C. Covalently bonded polysaccharide derivatives as chiral stationary phases in high-performance liquid chromatography. J. Chromatogr. A 2001, 906, 155–170. [Google Scholar] [CrossRef]
- Fernandes, C.; Brandão, P.; Santos, A.; Tiritan, M.E.; Afonso, C.; Cass, Q.B.; Pinto, M.M.M. Resolution and determination of enantiomeric purity of new chiral derivatives of xanthones using polysaccharide-based stationary phases. J. Chromatogr. A 2012, 1269, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Sousa, E.P.; Tiritan, M.E.; Oliveira, R.V.; Afonso, C.M.; Cass, Q.B.; Pinto, M.M. Enantiomeric resolution of kielcorin derivatives by HPLC on polysaccharide stationary phases using multimodal elution. Chirality 2004, 16, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Sousa, M.E.; Tiritan, M.E.; Belaz, K.R.; Pedro, M.; Nascimento, M.S.; Cass, Q.B.; Pinto, M.M. Multimilligram enantioresolution of low-solubility xanthonolignoids on polysaccharide chiral stationary phases using a solid-phase injection system. J. Chromatogr. A 2006, 1120, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Silva, B.; Fernandes, C.; Tiritan, M.E.; Pinto, M.M.; Valente, M.J.; Carvalho, M.; de Pinho, P.G.; Remiao, F. Chiral enantioresolution of cathinone derivatives present in “legal highs”, and enantioselectivity evaluation on cytotoxicity of 3,4-methylenedioxypyrovalerone (MDPV). Forensic Toxicol. 2016, 34, 372–385. [Google Scholar] [CrossRef] [PubMed]
- Taylor, D.R.; Maher, K. Chiral separations by high-performance liquid chromatography. J. Chromatogr. Sci. 1992, 67–84. [Google Scholar] [CrossRef]
- Cavazzini, A.; Pasti, L.; Massi, A.; Marchetti, N.; Dondi, F. Recent applications in chiral high performance chiral chromatography: A review. Anal. Chim. Acta 2011, 706, 205–222. [Google Scholar] [CrossRef] [PubMed]
- Haginaka, J. Pharmaceutical and biomedical applications of enantioseparations using liquid chromatographic techniques. J. Pharm. Biomed. Anal. 2002, 27, 357–372. [Google Scholar] [CrossRef]
- Tang, M.; Zhang, J.; Zhuang, S.; Liu, W. Development of chiral stationary phases for high-performance liquid chromatographic separation. TrAC-Trends Anal. Chem. 2012, 39, 180–194. [Google Scholar] [CrossRef]
- Padró, J.M.; Keunchkarian, S. State-of-the-art and recent developments of immobilized polysaccharide-based chiral stationary phases for enantioseparations by high-performance liquid chromatography. Microchem. J. 2018, 140, 142–157. [Google Scholar] [CrossRef]
- Xie, S.; Yuan, L. Recent development trends for chiral stationary phases based on chitosan derivatives, cyclofructan derivatives and chiral porous materials in high performance liquid chromatography. J. Sep. Sci. 2019, 6–20. [Google Scholar] [CrossRef] [PubMed]
- Yashima, E. Polysaccharide-based chiral stationary phases for high-performance liquid chromatographic enantioseparation. J. Chromatogr. A 2001, 906, 105–125. [Google Scholar] [CrossRef]
- Ikai, T.; Yamamoto, C.; Kamigaito, M.; Okamoto, Y. Immobilized-type chiral packing materials for HPLC based on polysaccharide derivatives. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2008, 875, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, Y.; Ikai, T. Chiral HPLC for efficient resolution of enantiomers. Chem. Soc. Rev. 2008, 37, 2593–2608. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Jin, X.; Yang, H.; Liu, X.; Liu, Y.; Ji, S. Controlled synthesis, immobilization and chiral recognition of carboxylic acid functionalized cellulose tris(3,5-dimethylphenylcarbamate). Carbohydr. Polym. 2017, 172, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, X.-C.; Chen, W.; Bai, Z.-W. Synthesis of substituted phenylcarbamates of N-cyclobutylformylated chitosan and their application as chiral selectors in enantioseparation. Analyst 2016, 141, 4470–4480. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Bin, Q.; Chen, W.; Bai, Z.-W.; Huang, S.H. Chiral stationary phases based on chitosan bis(methylphenylcarbamate)-(isobutyrylamide) for high-performance liquid chromatography. J. Chromatogr. A 2016, 1440, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Mei, X.; Chen, W.; Huang, S.H.; Bai, Z.-W. A high-performance chiral selector derived from chitosan (p-methylbenzylurea) for efficient enantiomer separation. Talanta 2018, 185, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, Z.Q.; Chen, W.; Bai, Z.-W. Preparation and enantioseparation of biselector chiral stationary phases based on amylose and chitin derivatives. Anal. Sci. 2015, 31, 1091–1097. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhu, N.; Ma, Y.; Li, Q.; Li, P. Preparation of polysaccharide-based chiral stationary phases on SiO2@Ag core-shell particles by means of coating and intermolecular polycondensation and comparative liquid chromatography enantioseparations. Anal. Bioanal. Chem. 2018, 410, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Wang, F.; Bi, W.; Liu, B.; Liu, S.; Okamoto, Y. Synthesis of cellulose carbamates bearing regioselective substituents at 2,3- and 6-positions for efficient chromatographic. J. Chromatogr. A 2018, 1572, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Mei, X.M.; Chen, W.; Bai, Z.-W. Enantioseparation characteristics of the chiral stationary phases based on natural and regenerated chitins. J. Sep. Sci. 2017, 40, 1710–1717. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, Z.Q.; Wang, X.-C.; Zhang, J.J.; Bai, Z.-W.; Chen, W. Enantioseparation characteristics of tadalafil and its intermediate on chitin derived chiral stationary phases. Analyst 2015, 140, 5593–5600. [Google Scholar] [CrossRef] [PubMed]
- Fouad, A.; Marzouk, A.A.; Ibrahim, S.M.; El-Adl, S.M.; Ghanem, A. Functionalized polymer monoliths with carbamylated amylose for the enantioselective reversed phase nano-liquid chromatographic separation of a set of racemic pharmaceuticals. J. Chromatogr. A 2017, 1515, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.Q.; Liu, J.D.; Chen, W.; Bai, Z.-W. Enantioseparation characteristics of biselector chiral stationary phases based on derivatives of cellulose and amylose. J. Chromatogr. A 2014, 1346, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Li, X.; Jin, Z.; Tian, L.; Wang, F.; Liu, G.; Tang, S.; Pan, F. Preparation of Regioselectively Modified Amylose Derivatives and Their Applications in Chiral HPLC. Chromatographia 2012, 75, 1347–1354. [Google Scholar] [CrossRef]
- Bezhitashvili, L.; Bardavelidze, A.; Mskhiladze, A.; Gumustas, M.; Ozkan, S.A.; Volonterio, A.; Farkas, T.; Chankvetadze, B. Application of cellulose 3,5-dichlorophenylcarbamate covalently immobilized on superficially porous silica for the separation of enantiomers in high-performance liquid chromatography. J. Chromatogr. A 2018, 1571, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Yin, C.; Chen, W.; Zhang, J.; Zhang, M.; Zhang, J. A facile and efficient method to fabricate high-resolution immobilized cellulose-based chiral stationary phases via thiol-ene click chemistry. Sep. Purif. Technol. 2019, 210, 175–181. [Google Scholar] [CrossRef]
- Huang, M.; Ma, H.; Niu, M.; Hu, F.; Wang, S.; Li, L.; Lv, C. Preparation of silica microspheres with a broad pore size distribution and their use as the support for a coated cellulose derivative chiral stationary phase. J. Sep. Sci. 2018, 41, 1232–1239. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, Y.; Kawashima, M.; Hatada, K. Chromatographic resolution: XI. Controlled chiral recognition of cellulose triphenylcarbamate derivatives supported on silica gel. J. Chromatogr. 1986, 363, 173–186. [Google Scholar] [CrossRef]
- Vieira, A.T.; Assunc, R.M.N.; Faria, A.M. Stationary phase based on cellulose dodecanoate physically immobilized on silica particles for high-performance liquid chromatography. J. Chromatogr. A 2018, 1572, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Petrie, B.; Camacho Muñoz, M.D.; Martín, J. Stereoselective LC–MS/MS methodologies for environmental analysis of chiral pesticides. TrAC-Trends Anal. Chem. 2019, 110, 249–258. [Google Scholar] [CrossRef]
- D’ Orazio, G.; Asensio-Ramos, M.; Fanali, C. Enantiomers separation by capillary electrochromatography using polysaccharide-based stationary phases. J. Sep. Sci. 2019, 42, 360–384. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Q.; Wen, Q.; Xiang, Y.; Zhang, L. Chromatographic enantioseparation of chiral sulfinamide derivatives on polysaccharide-based chiral stationary phases. J. Chromatogr. A 2018, 1571, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Beridze, N.; Tsutskiridze, E.; Takaishvili, N.; Farkas, T.; Chankvetadze, B. Comparative enantiomer-resolving ability of coated and covalently immobilized versions of two polysaccharide-based chiral selectors in high-performance liquid chromatography. Chromatographia 2018, 81, 611–621. [Google Scholar] [CrossRef]
- Bouanini, M.; Belboukhari, N.; Menéndez, J.C.; Sekkoum, K.; Cheriti, A.; Aboul-Enein, H.Y. Chiral separation of novel iminonaringenin derivatives. Chirality 2018, 30, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Stewart, K.K.; Doherty, R.F. Resolution of DL-Tryptophan by affinity chromatography on bovine-serum albumin-agarose columns. Proc. Natl. Acad. Sci. USA 1973, 70, 2850–2852. [Google Scholar] [CrossRef] [PubMed]
- Bocian, S.; Skoczylas, M.; Buszewski, B. Amino acids, peptides, and proteins as chemically bonded stationary phases—A review. J. Sep. Sci. 2016, 39, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Hage, D.S.; Anguizola, J.A.; Bi, C.; Li, R.; Matsuda, R.; Papastavros, E.; Pfaunmiller, E.; Vargas, J.; Zheng, X. Pharmaceutical and biomedical applications of affinity chromatography: Recent trends and developments. J. Pharm. Biomed. Anal. 2012, 69, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Haginaka, J. Protein-based chiral stationary phases for high-performance liquid chromatography enantioseparations. J. Chromatogr. A 2001, 906, 253–273. [Google Scholar] [CrossRef]
- Wallmorth, D.M.; Lee, J.T. Chiral bioanalysis. Handb. Anal. Sep. 2003, 4, 129–184. [Google Scholar]
- Mallik, R.; Hage, D.S. Affinity monolith chromatography. J. Sep. Sci. 2006, 29, 1686–1704. [Google Scholar] [CrossRef] [PubMed]
- Moldoveanu, S.C.; David, V. Stationary phases and columns for immunoaffinity type separations. In Selection of the HPLC Method in Chemical Analysis; Elsevier: Amsterdam, The Netherlands, 2017; pp. 387–392. [Google Scholar]
- Haginaka, J. Recent progresses in protein-based chiral stationary phases for enantioseparations in liquid chromatography. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2008, 875, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Bi, C.; Jackson, A.; Vargas-Badilla, J.; Li, R.; Rada, G.; Anguizola, J.A.; Pfaunmiller, E.; Hage, D.S. Entrapment of α1-acid glycoprotein in high-performance affinity columns for drug-protein binding studies. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2016, 1021, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Pfaunmiller, E.; Hartmann, M.; Dupper, C.M.; Soman, S.; Hage, D.S. Optimization of human serum albumin monoliths for chiral separations and high-performance affinity chromatography. J. Chromatogr. A 2012, 1269, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, H.; Haginaka, J. Separation of enantiomers on chiral stationary phase based on chicken α1-acid glycoprotein: Effect of silica particle diameters on column performance. J. Chromatogr. A 2014, 1363, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Hellerstein, M.K.; Sasak, V.; Ordovas, J.; Munro, H.N. Isolation of α1-acid glycoprotein from human plasma using high-performance liquid chromatography. Anal. Biochem. 1985, 146, 366–371. [Google Scholar] [CrossRef]
- Matsunaga, H.; Haginaka, J. Separation of enantiomers on chiral stationary phase based on cellulase: Effect of preparation method and silica particle diameters on chiral recognition ability. J. Chromatogr. A 2016, 1467, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Vandenbosch, C.; Lindner, W.; Massart, D.L. Evaluation of the enantioselectivity of an ovomucoid and a cellulase chiral stationary phase towards a set of β blocking agents. Anal. Chim. Acta 1992, 270, 1–12. [Google Scholar] [CrossRef]
- Zheng, X.; Podariu, M.; Bi, C.; Hage, D.S. Development of enhanced capacity affinity microcolumns by using a hybrid of protein cross-linking/modification and immobilization. J. Chromatogr. A 2015, 1400, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Bi, C.; Matsuda, R.; Zhang, C.; Isingizwe, Z.; Clarke, W.; Hage, D.S. Studies of drug interactions with α1-acid glycoprotein by using on-line immunoextraction and high-performance affinity chromatography. J. Chromatogr. A 2017, 1519, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, R.; Donald, J.; Beyersdorf, J.; Hage, D.S. Analysis of drug–protein binding using on-line immunoextraction and high-performance affinity microcolumns: Studies with normal and glycated human serum albumin. J. Chromatogr. A 2015, 1416, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Fedorova, I.A.; Shapovalova, E.N.; Shpigun, O.A.; Staroverov, S.M. Bovine serum albumin adsorbed on eremomycin and grafted on silica as new mixed-binary chiral sorbent for improved enantioseparation of drugs. J. Food Drug Anal. 2016, 24, 848–854. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Wang, X.; Ji, Y. Monoliths with proteins as chiral selectors for enantiomer separation. Talanta 2012, 91, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Cârje, A.G.; Ion, V.; Muntean, D.; Balint, A.; Imre, S. Enantioseparation of indapamide by high-performance liquid chromatography using ovomucoid glycoprotein as chiral selector. Farmacia 2016, 64, 181–186. [Google Scholar]
- Batra, S.; Bhushan, R. Bioassay, determination and separation of enantiomers of atenolol by direct and indirect approaches using liquid chromatography: A review. Biomed. Chromatogr. 2018, 32, e4090. [Google Scholar] [CrossRef] [PubMed]
- Chrysanthakopoulos, M.; Vallianatou, T.; Giaginis, C.; Tsantili-Kakoulidou, A. Investigation of the retention behaviour of structurally diverse drugs on α1-acid glycoprotein column: Insight on the molecular factors involved and correlation with protein binding data. Eur. J. Pharm. Sci. 2014, 60, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Zi, Z.; Hage, D.S. Analysis of stereoselective drug interactions with serum proteins by high-performance affinity chromatography: A historical perspective. J. Pharm. Biomed. Anal. 2017, 144, 12–24. [Google Scholar] [CrossRef]
- Souza, M.C.O.; Marques, M.P.; Duarte, G.; Lanchote, V.L. Analysis of bupivacaine enantiomers in plasma as total and unbound concentrations using LC-MS/MS: Application in a pharmacokinetic study of a parturient with placental transfer. J. Pharm. Biomed. Anal. 2019, 164, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, D.W.; DeMond, W. Cyclodextrin bonded phases for the liquid chromatographic separation of optical, geometrical, and structural isomers. J. Chromatogr. Sci. 1984, 22, 411–415. [Google Scholar] [CrossRef]
- Chmielewska, A.; Konieczna, L.; Baczek, T. A novel two-step liquid-liquid extraction procedure combined with stationary phase immobilized human serum albumin for the chiral separation of cetirizine enantiomers along with M and P parabens. Molecules 2016, 21, 1654. [Google Scholar] [CrossRef] [PubMed]
- Bressolle, F.; Audran, M.; Pham, T.-N.; Vallon, J.-J. Cyclodextrins and enantiomeric separations of drugs by liquid chromatography and capillary electrophoresis: Basic principles and new developments. J. Chromatogr. B Biomed. Appl. 1996, 687, 303–336. [Google Scholar] [CrossRef]
- Berthood, A. Chiral recognition mechanisms in enantiomers separations: A general view. In Chiral Recognition in Separation Methods; Springer: Berlin/Heidelberg, Germany, 2010; pp. 1–32. [Google Scholar]
- Xiao, Y.; Ng, S.C.; Tan, T.T.Y.; Wang, Y. Recent development of cyclodextrin chiral stationary phases and their applications in chromatography. J. Chromatogr. A 2012, 1269, 52–68. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Lin, Y.; Xiao, Y.; Crommen, J.; Jiang, Z. Recent developments on cyclodextrin functionalized monolithic columns for the enantioseparation of chiral drugs. J. Pharm. Biomed. Anal. 2016, 130, 110–125. [Google Scholar] [CrossRef] [PubMed]
- Szabó, Z.-I.; Szőcs, L.; Horváth, P.; Komjáti, B.; Nagy, J.; Jánoska, Á.; Muntean, D.-L.; Noszál, B.; Tóth, G. Liquid chromatography with mass spectrometry enantioseparation of pomalidomide on cyclodextrin-bonded chiral stationary phases and the elucidation of the chiral recognition mechanisms by NMR spectroscopy and molecular modeling(Article). J. Sep. Sci. 2016, 39, 2941–2949. [Google Scholar] [CrossRef] [PubMed]
- Arsad, S.R.; Maarof, H.; Ibrahim, W.A.W.; Aboul-Enein, H.Y. Theoretical and Molecular Docking Study of Ketoconazole on Heptakis(2,3,6-tri-O-methyl)-β-cyclodextrin as Chiral Selector. Chirality 2016, 28, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.J.; Yang, G.L.; Xu, X.D.; Sheng, J.J.; Shen, J. Preparation and chromatographic evaluation of β-cyclodextrin derivative CSPs bearing substituted phenylcarbamate groups for HPLC. J. Liq. Chromatogr. Relat. Technol. 2016, 39, 647–657. [Google Scholar] [CrossRef]
- Li, L.; Cheng, B.; Zhou, R.; Cao, Z.; Zeng, C.; Li, L. Preparation and evaluation of a novel N-benzyl-phenethylamino-β-cyclodextrin-bonded chiral stationary phase for HPLC. Talanta 2017, 174, 179–191. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhang, M.; Wang, Y.; Zhou, W.; Zhou, Z. Preparation of chiral oxazolinyl-functionalized β-cyclodextrin-bonded stationary phases and their enantioseparation performance in high-performance liquid chromatography. J. Sep. Sci. 2016, 39, 4136–4146. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Li, X.; Sun, Y.; Xiao, Y.; Wang, Y. Thiol-ene click derived structurally well-defined per(3,5-dimethyl)phenylcarbamoylated cationic cyclodextrin separation material for achiral and chiral chromatography. J. Sep. Sci. 2018, 41, 2710–2718. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Yang, B.; Tang, J.; Tang, W. A cationic cyclodextrin clicked bilayer chiral stationary phase for versatile chiral separation in HPLC. New J. Chem. 2018, 42, 3526–3533. [Google Scholar] [CrossRef]
- Chu, C.; Liu, R. Application of click chemistry on preparation of separation materials for liquid chromatography. Chem. Soc. Rev. 2011, 40, 2177–2188. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Dong, S.; Han, F.; Zhao, Y.; Zhang, X.; Zhang, X.; Qiu, H.; Zhao, L. Spherical β-cyclodextrin-silica hybrid materials for multifunctional chiral stationary phases. J. Chromatogr. A 2015, 1383, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Li, L.; Wang, Y.; Zhou, Z. Preparation of polar group derivative β-cyclodextrin bonded hydride silica chiral stationary phases and their chromatography separation performances. Chin. Chem. Lett. 2018. [Google Scholar] [CrossRef]
- Ghanem, A.; Adly, F.G.; Sokerik, Y.; Antwi, N.Y.; Shenashen, M.A.; El-Safty, S.A. Trimethyl-β-cyclodextrin-encapsulated monolithic capillary columns: Preparation, characterization and chiral nano-LC application. Talanta 2017, 169, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Qiang, L.I.; Yuan-yuan, L.I.; Nan, Z.H.U.; Zhu-xian, G.A.O.; Tian-jun, L.I.; Tong, Z.; Yu-long, M.A. Preparation of cyclodextrin type stationary phase based on graphene oxide and its application in enantioseparation and hydrophilic interaction chromatography. Chin. J. Anal. Chem. 2018, 46, 1455–1463. [Google Scholar] [CrossRef]
- Tang, Q.; Yu, B.; Gao, L.; Cong, H.; Zhang, S. Light-assisted preparation of cyclodextrin-based chiral stationary phase and its separation performance in liquid chromatography. New J. Chem. 2018, 42, 1115–1120. [Google Scholar] [CrossRef]
- Zhou, J.; Yang, B.; Tang, J.; Tang, W. Cationic cyclodextrin clicked chiral stationary phase for versatile enantioseparations in high-performance liquid chromatography. J. Chromatogr. A 2016, 1467, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Long, Y.; Zhi, Y.; Xu, X. Preparation and chromatographic evaluation of a chiral stationary phase based on carboxymethyl-β-cyclodextrin for high-performance liquid chromatography. Chin. Chem. Lett. 2018, 29, 1399–1403. [Google Scholar] [CrossRef]
- Li, X.; Jin, X.; Yao, X.; Ma, X.; Wang, Y. Thioether bridged cationic cyclodextrin stationary phases: Effect of spacer length, selector concentration and rim functionalities on the enantioseparation. J. Chromatogr. A 2016, 1467, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Pesek, J.J.; Matyska, M.T. Silica Hydride-Based Packing Materials: HPLC Stationary Phases for a Global Approach to Complex Sample Analysis. Curr. Chrom. 2018, 5, 33–42. [Google Scholar] [CrossRef]
- Armstrong, D.W.; Tang, Y.; Chen, S.; Zhou, Y.; Bagwlll, C.; Chen, J. Macrocyclic antibiotics as a new class of chiral selectors for liquid chromatography. Anal. Chem. 1994, 1473–1484. [Google Scholar] [CrossRef]
- Ilisz, I.; Pataj, Z.; Aranyi, A.; Péter, A. Macrocyclic antibiotic selectors in direct HPLC enantioseparations. Sep. Purif. Rev. 2012, 41, 207–249. [Google Scholar] [CrossRef]
- Ward, T.J.; Farris, A.B. Chiral separations using the macrocyclic antibiotics: A review. J. Chromatogr. A 2001, 906, 73–89. [Google Scholar] [CrossRef]
- Ilisz, I.; Berkecz, R.; Péter, A. Retention mechanism of high-performance liquid chromatographic enantioseparation on macrocyclic glycopeptide-based chiral stationary phases. J. Chromatogr. A 2009, 1216, 1845–1860. [Google Scholar] [CrossRef] [PubMed]
- Aburachid Cardoso, P.; Costa César, I. Chiral method development strategies for HPLC using macrocyclic glycopeptide-based stationary phases. Chromatographia 2018, 81, 841–850. [Google Scholar] [CrossRef]
- Beesley, T.E.; Lee, J.T. Method development strategy and applications update for CHIROBIOTIC chiral stationary phases. J. Liq. Chromatogr. Relat. Technol. 2009, 32, 1733–1767. [Google Scholar] [CrossRef]
- Fernandes, C.; Tiritan, M.E.; Cass, Q.; Kairys, V.; Fernandes, M.X.; Pinto, M.M. Enantioseparation and chiral recognition mechanism of new chiral derivatives of xanthones on macrocyclic antibiotic stationary phases. J. Chromatogr. A 2012, 1241, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Hrobonová, K.; Lehotay, J.; Cizmárik, J. HPLC enantioseparation of phenylcarbamic acid derivatives by using macrocyclic chiral stationary phases. Nov. Biotechnol. Chim. 2016, 15, 12–22. [Google Scholar] [CrossRef]
- Min, Y.; Sui, Z.; Liang, Z.; Zhang, L.; Zhang, Y. Teicoplanin bonded sub-2 micrometer superficially porous particles for enantioseparation of native amino acids. J. Pharm. Biomed. Anal. 2015, 114, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Ismail, O.H.; Ciogli, A.; Villani, C.; De Martino, M.; Pierini, M.; Cavazzini, A.; Bell, D.S.; Gasparrini, F. Ultra-fast high-efficiency enantioseparations by means of a teicoplanin-based chiral stationary phase made on sub-2 µm totally porous silica particles of narrow size distribution. J. Chromatogr. A 2016, 1427, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Rocchi, S.; Rocco, A.; Pesek, J.J.; Matyska, M.T.; Capitani, D.; Fanali, S. Enantiomers separation by nano-liquid chromatography: Use of a novel sub-2 micrometer vancomycin silica hydride stationary phase. J. Chromatogr. A 2015, 1381, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Shao, H.; Luo, R.; Wang, Q.; Sánchez-López, E.; Fanali, S.; Marina, M.L.; Jiang, Z. A facile and efficient single-step approach for the fabrication of vancomycin functionalized polymer-based monolith as chiral stationary phase for nano-liquid chromatography. J. Chromatogr. A 2018, 1557, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Hellinghausen, G.; Lopez, D.A.; Lee, J.T.; Wang, Y.; Weatherly, C.A.; Portillo, A.E.; Berthod, A.; Armstrong, D.W. Evaluation of the Edman degradation product of vancomycin bonded to core-shell particles as a new HPLC chiral stationary phase. Chirality 2018, 30, 1067–1078. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.; Zhang, S.; Li, G.; Cong, H. Light-assisted preparation of vancomycin chiral stationary phase based on diazotized silica and its enantioseparation evaluation by high-performance liquid chromatography. Talanta 2018, 182, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Dolzan, M.D.; Shu, Y.; Smuts, J.P.; Petersen, H.; Ellegaard, P.; Micke, G.A.; Armstrong, D.W.; Breitbach, Z.S. Enantiomeric separation of citalopram analogues by HPLC using macrocyclic glycopeptide and cyclodextrin based chiral stationary phase. J. Liq. Chromatogr. Relat. Technol. 2016, 39, 154–160. [Google Scholar] [CrossRef]
- Pirkle, W.H.; House, D.W. Chiral high-performance liquid chromatographic stationary phases. 1. Separation of the enantiomers of sulfoxides, amines, amino acids, alcohols, hydroxy acids, lactones and mercaptans. J. Org. Chem. 1979, 44, 1957–1960. [Google Scholar] [CrossRef]
- Davankov, V.A. The nature of chiral recognition: Is it a three-point interaction? Chirality 1997, 9, 99–102. [Google Scholar] [CrossRef]
- Welch, C.J. Evolution of chiral stationary phase design in the Pirkle laboratories. J. Chromatogr. A 1994, 666, 3–26. [Google Scholar] [CrossRef]
- Pirkle, W.H.; Däppen, R. Reciprocity in Chiral Recognition. Comparison of several chiral stationary phases. J. Chromatogr. A 1987, 404, 107–115. [Google Scholar] [CrossRef]
- Pirkle, W.H.; Hyun, M.H.; Tsipouras, A.; Hamper, B.C.; Banks, B. A rational approach to the design of highly effective chiral stationary phases for the liquid chromatographic separation of enantiomers. J. Pharm. Biomed. Anal. 1984, 2, 173–181. [Google Scholar] [CrossRef]
- Pirkle, W.H.; Hyun, M.H. Preparation and use of hydantion-based chiral stationary phases. J. Chromatogr. A 1985, 322, 309–320. [Google Scholar] [CrossRef]
- Pirkle, W.H.; Murray, P.G.; Wilson, S.R. X-ray crystallographic evidence in support of a proposed chiral recognition mechanism. J. Org. Chem. 1996, 61, 4775–4777. [Google Scholar] [CrossRef] [PubMed]
- Pirkle, W.H.; Selness, S.R. Chiral recognition studies: Intra- and intermolecular 1H{1H}-nuclear overhauser effects as effective tools in the study of bimolecular complexes. J. Org. Chem. 1995, 60, 3252–3256. [Google Scholar] [CrossRef]
- Fernandes, C.; Tiritan, M.E.; Pinto, M.M.M. Small molecules as chromatographic tools for HPLC enantiomeric resolution: Pirkle-type chiral stationary phases evolution. Chromatographia 2013, 76, 871–897. [Google Scholar] [CrossRef]
- Dungelova, J.; Lehotay, J.; Cizmarik, J.; Armstrong, D.W. Study of the mechanism of enantioseparation. IV. Study of enantioseparation of some derivatives of phenylcarbamic acid using p-complex stationary phase in HPLC. J. Liq. Chromatogr. Relat. Technol. 2003, 26, 2331–2350. [Google Scholar] [CrossRef]
- Asnin, L.D.; Guiochon, G. The adsorption of naproxen enantiomers on the chiral stationary phase (R, R)-Whelk-O1 under reversed-phase conditions: The effect of mobile phase composition. J. Chromatogr. A 2010, 1217, 2871–2878. [Google Scholar] [CrossRef] [PubMed]
- Welch, C.J.; Szczerba, T.; Perrin, S.R. Some recent high-performance liquid chromatography separations of the enantiomers of pharmaceuticals and other compounds using the Whelk-O 1 chiral stationary phase. J. Chromatogr. A 1997, 758, 93–98. [Google Scholar] [CrossRef]
- Fernandes, C.; Phyo, Y.Z.; Silva, A.S.; Tiritan, M.E.; Kijjoa, A.; Pinto, M.M.M. Chiral stationary phases based on small molecules: An update of the last 17 years. Sep. Purif. Rev. 2018, 47, 89–123. [Google Scholar] [CrossRef]
- Gasparrini, F.; Misiti, D.; Villani, C. High-performance liquid chromatography chiral stationary phases based on low-molecular mass selectors. J. Chromatogr. A 2001, 906, 35–50. [Google Scholar] [CrossRef]
- Villani, C.; D’Acquarica, I.; Gasparrini, F.; Ritchi, H.; Kotoni, D.; Pierini, M.; Ciogli, A.; Kocergin, J. The evolution of chiral stationary phases from HPLC to UHPLC. LC-GC Eur. 2014, 27, 128–137. [Google Scholar]
- Waterlot, C.; Ghinet, A.; Lipka, E. Core-shell Particles: A Way to Greening Liquid Chromatography in Environmental Applications. Curr. Chromatogr. 2018, 5. [Google Scholar] [CrossRef]
- Spudeit, D.A.; Dolzan, M.D.; Breitbach, Z.S.; Barber, W.E.; Micke, G.A.; Armstrong, D.W. Superficially porous particles vs fully porous particles for bonded high-performance liquid chromatographic chiral stationary phases: Isopropyl cyclofructan 6. J. Chromatogr. A 2014, 1363, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Catani, M.; Ismail, O.; Gasparrini, F.; Antonelli, M.; Pasti, L.; Marchetti, N.; Fellettia, S.; Cavazzini, A. Recent advancements and future directions of superficially porous chiral stationary phases for ultrafast high-performance enantioseparations. Analyst 2017, 142, 555–566. [Google Scholar] [CrossRef] [PubMed]
- Qiao, L.; Zhou, X.; Li, X.; Du, W.; Yu, A.; Zhang, S.; Wu, Y. Synthesis and performance of chiral ferrocene modified silica gel for mixed-mode chromatography. Talanta 2017, 163, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Çakmak, R.; Ercan, S.; Sünkür, M.; Yilmaz, H.; Topal, G. Design, preparation and application of a Pirkle-type chiral stationary phase for enantioseparation of some racemic organic acids and molecular dynamic studies. Org. Commun. 2017, 10, 216–227. [Google Scholar] [CrossRef]
- Shen, H.; Du, G.; Liu, K.; Ye, L.; Xie, S.; Jiang, L. Synthesis and evaluation of pseudopeptide chiral stationary phases for enantioselective resolution. J. Chromatogr. A 2017, 1521, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Armstrong, D.W.; Ryoo, J.J. Synthesis of new C3 symmetric amino acid- and aminoalcohol-containing chiral stationary phases and application to HPLC enantioseparations. Chirality 2018, 30, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, D.; Zhang, Y.; Tang, Y.; Zhao, J.; Shen, B. Synthesis of a Novel Chiral Stationary Phase by (R)-1,1′-Binaphthol and the Study on Mechanism of Chiral Recognition. Symmetry 2018, 10, 704. [Google Scholar] [CrossRef]
- Yang, J.; Carmella, S.G.; Hecht, S.S. Analysis of N′-nitrosonornicotine enantiomers in human urine by chiral stationary phase liquid chromatography–nanoelectrospray ionization–high resolution tandem mass spectrometry. J. Chromatogr. B 2017, 1044, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Yip, S.H.; Wu, D.-R.; Li, P.; Sun, D.; Watterson, S.H.; Zhao, R.; Tino, J.; Mathur, A. Separation of Bruton’s tyrosine kinase inhibitor atropisomers by supercritical fluid chromatography. J. Chromatogr. A 2019, 1586, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Felletti, S.; De Luca, C.; Ismail, O.H.; Pasti, L.; Costa, V.; Gasparrini, F.; Cavazzini, A.; Catani, M. On the effect of chiral selector loading and mobile phase composition on adsorption properties of latest generation fully- and superficially-porous Whelk-O1 particles for high-efficient ultrafast enantioseparations. J. Chromatogr. A 2018, 1579, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Tumashov, A.A.; Gruzdev, D.A.; Vigorov, A.Y.; Musiyak, V.V.; Chulakov, E.N.; Levit, G.L.; Krasnov, V.P.; Charushin, V.N. Analysis of racemic conjugates of purine with heterocyclic amines by chiral high-performance liquid chromatography. Russ. Chem. Bull. 2018, 67, 1704–1709. [Google Scholar] [CrossRef]
- Knežević, A.; Novak, J.; Pescitelli, G.; Vinković, V. Determination of the Absolute Configuration of (S)-N-(1-Aryl-allyl)-3,5-dinitrobenzamides and Their Elution Order on Brush-Type Chiral Stationary Phases. Eur. J. Org. Chem. 2018, 2018, 3982–3991. [Google Scholar] [CrossRef]
- Rosini, C.; Bertucci, C.; Pini, D.; Altemura, P.; Salvadori, P. Cinchona alkaloids for preparing new, easily accessible chiral stationary phases. I. 11-(10,11-dihydro-6′-methoxy-cinchonan-9-OL)-tiopropylsilanized silica. Tetrahedron Lett. 1985, 26, 3361–3364. [Google Scholar] [CrossRef]
- Lämmerhofer, M. Liquid chromatographic enantiomer separation with special focus on zwitterionic chiral ion-exchangers. Anal. Bioanal. Chem. 2014, 406, 6095–6103. [Google Scholar] [CrossRef] [PubMed]
- Vander Heyden, Y.; Mangelings, D.; Matthijs, N.; Perrin, C. Chiral separations. In Handbook of Pharmaceuticals Analysis by HPLC; Elsevier: Amsterdam, The Netherlands, 2005; Volume 6, pp. 447–498. [Google Scholar]
- Natalini, B.; Sardella, R. Chromatographic separations and analysis: Chiral ion and ligand exchange stationary phases. Compr. Chir. 2012, 8, 115–152. [Google Scholar]
- Hoffmann, C.V.; Pell, R.; Lämmerhofer, M.; Lindner, W. Synergistic effects on enantioselectivity of zwitterionic chiral stationary phases for separations of chiral acids, bases and amino acids by HPLC. Anal. Chem. 2008, 80, 8780–8789. [Google Scholar] [CrossRef] [PubMed]
- Nesterenko, P.N.; Haddad, P.R. Zwitterionic ion-exchangers in liquid chromatography. Anal. Sci. 2000, 16, 565–574. [Google Scholar] [CrossRef]
- Gübitz, G.; Schmid, M.G. Chiral separation by chromatographic and electromigration techniques. A review. Biopharm. Drug Dispos. 2001, 22, 291–336. [Google Scholar] [CrossRef] [PubMed]
- Ilisz, I.; Bajtai, A.; Lindner, W.; Péter, A. Liquid chromatographic enantiomer separations applying chiral ion-exchangers based on Cinchona alkaloids. J. Pharm. Biomed. Anal. 2018, 159, 127–152. [Google Scholar] [CrossRef] [PubMed]
- Todoroki, K.; Ishii, Y.; Ide, T.; Min, J.Z.; Inoue, K.; Huang, X.; Zhang, W.; Hamashima, Y.; Toyo’oka, T. Advanced dress-up chiral columns: New removable chiral stationary phases for enantioseparation of chiral carboxylic acids. Anal. Chim. Acta 2015, 882, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Woiwode, U.; Sievers-Engler, A.; Zimmermann, A.; Lindner, W.; Sánchez-Muñoz, O.L.; Lämmerhofer, M. Surface-anchored counterions on weak chiral anion-exchangers accelerate separations and improve their compability for mass-spectrometry-hyphenation. J. Chromatogr. A 2017, 1503, 21–31. [Google Scholar] [CrossRef] [PubMed]
- De Martino, M.; Bencivenni, G.; Mazzanti, A.; Menta, S.; Ismail, O.H.; Sabia, R.; Ciogli, A. 3,5-dinitrobenzoyl-9-amino-9-deoxy-9-epiquinine as Pirkle-anion exchange hybrid-type chiral selector in high-performance liquid chromatography. Chromatographia 2017, 80, 751–762. [Google Scholar] [CrossRef]
- Woiwode, U.; Neubauer, S.; Lindner, W.; Buckenmaier, S.; Lämmerhofer, M. Enantioselective multiple heartcut two-dimensional ultra-high-performance liquid chromatography method with a coreshell chiral stationary phase in the second dimension for analysis of all proteinogenic amino acids in a single run. J. Chromatogr. A 2018, 1562, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Woiwode, U.; Ferri, M.; Maier, N.M.; Lindner, W.; Lämmerhofer, M. Complementary enantioselectivity profiles of chiral cinchonan carbamate selectors with distinct carbamate residues and their implementation in enantioselective two-dimensional high-performance liquid chromatography of amino acids. J. Chromatogr. A 2018, 1558, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, K.; Lämmerhofer, M. Optimization of the surface modification process of cross-linked polythiol-coated chiral stationary phases synthesized by a two-step thiol-ene click reaction. J. Sep. Sci. 2018, 41, 1338–1345. [Google Scholar] [CrossRef] [PubMed]
- Patel, D.C.; Breitbach, Z.S.; Yu, J.; Nguyen, K.A.; Armstrong, D.W. Quinine bonded to superficially porous particles for high-efficiency and ultrafast liquid and supercritical fluid chromatography. Anal. Chim. Acta 2017, 963, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Orosz, T.; Forró, E.; Fülöp, F.; Lindner, W.; Ilisz, I.; Péter, A. Effects on N-methylation and amidination of cyclic β-amino acids on enantioselectivity and retention characteristics using Cinchona alkaloid- and sulfonic acid-based chiral zwitterionic stationary phases. J. Chromatogr. A 2018, 1535, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Bajtai, A.; Fekete, B.; Palkó, M.; Fülöp, F.; Lindner, W.; Kohout, M.; Ilisz, I.; Péter, A. Comparative study on the liquid chromatographic enantioseparation of cyclic β-amino acids and the related cyclic β-aminohydroxamic acids on Cinchona alkaloid-based zwitterionic chiral stationary phases. J. Sep. Sci. 2018, 41, 1216–1223. [Google Scholar] [CrossRef] [PubMed]
- Calderón, C.; Santi, C.; Lämmerhofer, M. Chiral separation of disease biomarkers with 2-hydroxycarboxylic acid structure. J. Sep. Sci. 2018, 41, 1224–1231. [Google Scholar] [CrossRef] [PubMed]
- Nakano, Y.; Taniguchi, M.; Fukusaki, E. High-sensitive liquid chromatography-tandem mass spectrometry-based chiral metabolic profiling focusing on amino acids and related metabolites. J. Biosci. Bioeng. 2018. [Google Scholar] [CrossRef] [PubMed]
- Iannia, F.; Pucciarinia, L.; Carottia, A.; Gioielloa, A.; Galarinib, R.; Natalinic, S.; Sardellaa, R.; Lindnerd, W.; Natalini, B. Improved chromatographic diastereoresolution of cyclopropyl dafachronic acid derivatives using chiral anion exchangers. J. Chromatogr. A 2018, 1557, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Sousa, L.R.; Sogah, G.D.Y.; Hoffmann, D.H.; Cram, D.J. Host-Guest Complexation. 12. Total optical resolution of amine and amino ester salts by chromatography. J. Am. Chem. Soc. 1978, 100, 4569–4576. [Google Scholar] [CrossRef]
- León-González, M.E.; Rosales-Conrado, N.; Pérez-Arribas, L.V.; Guillén-Casla, V. Two-dimensional liquid chromatography for direct chiral separations: A review. Biomed. Chromatogr. 2014, 28, 59–83. [Google Scholar] [CrossRef] [PubMed]
- Hyun, M.H. Liquid chromatographic enantioseparations on crown ether-based chiral stationary phases. J. Chromatogr. A 2016, 1467, 19–32. [Google Scholar] [CrossRef] [PubMed]
- Hyun, M.H. Development of HPLC chiral stationary phases based on (+)-(18-Crown-6)-2,3,11,12-tetracarboxylic acid and their applications. Chirality 2015, 27, 576–588. [Google Scholar] [CrossRef] [PubMed]
- Bakó, P.; Keglevich, G.; Rapi, Z.; Töke, L. The enantiomeric differentiation ability of chiral crown ethers based on carbohydrates. Curr. Org. Chem. 2012, 16, 297–304. [Google Scholar] [CrossRef]
- Mohammadzadeh Kakhki, R. Application of crown ethers as stationary phase in the chromatographic methods. J. Incl. Phenom. Macrocycl. Chem. 2013, 75, 11–22. [Google Scholar] [CrossRef]
- Paik, M.J.; Kang, J.S.; Huang, B.S.; Carey, J.R.; Lee, W. Development and application of chiral crown ethers as selectors for chiral separation in high-performance liquid chromatography and nuclear magnetic resonance spectroscopy. J. Chromatogr. A 2013, 1274, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.; Wei, Q.; Meng, M.; Yin, J.; Shan, Y.; Du, L.; Zhu, X. Preparation and application of aza-15-crown-5-capped methylcalix [4] resorcinarene-bonded silica particles for use as chiral stationary phase in HPLC. Chromatographia 2017, 80, 1007–1014. [Google Scholar] [CrossRef]
- Yaghoubnejad, S.; Ahmadi, S.H. Preparation and evaluation of a chiral HPLC stationary phase based on cone calix[4]arene functionalized at the upper rim with l-alanine units. Biomed. Chromatogr. 2018, 32, e4122. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Sheng, Z.; Zhu, C.; Yin, W.; Chu, C. Silica based click-dibenzo-18-crown-6-ether high-performance liquid chromatography stationary phase and its application in separation of fullerenes. Talanta 2018, 178, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Németh, T.; Dargó, G.; Petró, J.L.; Petrik, Z.; Lévai, S.; Krámos, B.; Béni, Z.; Nagy, J.; Balogh, G.T.; Huszthy, P.; et al. Synthesis and pKa determination of new enantiopure dimethyl-substituted acridino-crown ethers containing a carboxyl group: Useful candidates for enantiomeric recognition studies. Chirality 2017, 29, 522–535. [Google Scholar] [CrossRef] [PubMed]
- Yaghoubnejad, S.; Ahmadi, S.H. Preparation and evaluation of a deoxycholic-calix[4]arene hybrid-type receptor as a chiral stationary phase for HPLC. J. Sep. Sci. 2018, 41, 1903–1912. [Google Scholar] [CrossRef] [PubMed]
- Sun, P.; Wang, C.; Breitbach, Z.S.; Zhang, Y.; Armstrong, D.W. Development of new HPLC chiral stationary phases based on native and derivatized cyclofructans. Anal. Chem. 2009, 81, 10215–10226. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.M.; Breitbach, Z.S.; Berthood, A.; Armstrong, D.W. Chlorinated aromatic derivatives of cyclofructan 6 as HPLC chiral stationary phases. J. Liq. Chromatogr. Relat. Technol. 2016, 39, 497–503. [Google Scholar] [CrossRef]
- Janecková, L.; Kalíková, K.; Vozka, J.; Armstrong, D.W.; Bosáková, Z.; Tesarová, E. Characterization of cyclofructan-based chiral stationary phase by linear free energy relationship. J. Sep. Sci. 2011, 34, 2639–2644. [Google Scholar] [CrossRef] [PubMed]
- Padivitage, N.L.; Smuts, J.P.; Breitbach, Z.S.; Armstrong, D.W.; Berthood, A. Preparation and evaluation of HPLC chiral stationary phases based on cationic/basic derivatives of cyclofructan 6. J. Liq. Chromatogr. Relat. Technol. 2015, 38, 550–560. [Google Scholar] [CrossRef] [PubMed]
- Qiu, H.; Kiyono-Shimobe, M.; Armstrong, D.W. Native/derivatized cyclofructan 6 bound to resins via “click” chemistry as stationary phases for achiral/chiral separations. J. Liq. Chromatogr. Relat. Technol. 2014, 37, 2302–2326. [Google Scholar] [CrossRef]
- Moskalová, M.; Kozlov, O.; Gondová, T.; Budovská, M.; Armstrong, D.W. HPLC enantioseparation of novel spirobrassinin analogs on the cyclofructan chiral stationary phases. Chromatographia 2017, 80, 53–62. [Google Scholar] [CrossRef]
- Frink, L.A.; Berthood, A.; Xu, Q.L.; Gao, H.; Kurti, L.; Armstrong, D.W. Separation of 2-naphtol atropisomers on cyclofructan-based chiral stationary phases. J. Liq. Chromatogr. Relat. Technol. 2016, 39, 710–717. [Google Scholar] [CrossRef]
- Gutierrez-Climente, R.; Gomez-Caballero, A.; Guerreiro, A.; Garcia-Mutio, D.; Unceta, N.; Goicolea, M.A.; Barrio, R. Molecularly imprinted nanoparticles grafted to porous silica as chiral selectors in liquid chromatography. J. Chromatogr. A 2017, 1508, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Sellergren, B. Imprinted chiral stationary phases in high-performance liquid chromatography. J. Chromatogr. A 2001, 906, 227–252. [Google Scholar] [CrossRef]
- Wei, Z.H.; Mu, L.N.; Huang, Y.P.; Liu, Z.S. Imprinted monoliths: Recent significant progress in analysis field. TrAC-Trends Anal. Chem. 2017, 86, 84–92. [Google Scholar] [CrossRef]
- Rutkowska, M.; Plotka-Wasylka, J.; Morrison, C.; Wieczorek, P.P.; Namiesnik, J.; Marc, M. Application of molecularly imprinted polymers in analytical chiral separations and analysis. TrAC-Trends Anal. Chem. 2018, 102, 91–102. [Google Scholar] [CrossRef]
- Yang, S.; Wang, Y.; Jiang, Y.; Li, S.; Liu, W. Molecularly imprinted polymers for the identification and separation of chiral drugs and biomolecules. Polymers 2016, 8, 216. [Google Scholar] [CrossRef]
- Bitas, D.; Samanidou, V. Molecularly imprinted polymers as extracting media for the chromatographic determination of antibiotics in milk. Molecules 2018, 23, 316. [Google Scholar] [CrossRef] [PubMed]
- Sierra, I.; Marina, M.L.; Pérez-Quintanilla, D.; Morante-Zarcero, S.; Silva, M. Approaches for enantioselective resolution of pharmaceuticals by miniaturised separation techniques with new chiral phases based on nanoparticles and monoliths. Electrophoresis 2016, 37, 2538–2553. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.S.; Zheng, C.; Yan, C.; Gao, R.Y. Molecularly imprinted polymers as a tool for separation in CEC. Electrophoresis 2007, 28, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, P.; Dhawan, V.; Fernandes, C.; Suares, D. Recent progress on nanofabrication of molecularly imprinted polymers. In Emerging Applications of Nanoparticles and Architecture Nanostructures; Elsevier: Amsterdam, The Netherlands, 2018; pp. 385–409. [Google Scholar]
- Moniera, M.; Shafikb, A.L.; Abdel-Latifa, D.A. Surface molecularly imprinted amino-functionalized alginate microspheres for enantio-selective extraction of l-ascorbic acid. Carbohydr. Polym. 2018, 195, 652–661. [Google Scholar] [CrossRef] [PubMed]
- Khataee, A.; Hassanzadeh, J.; Elmira Kohan, E. Specific quantification of atropine using molecularly imprinted polymer on graphene quantum dots. Spectrochim. Acta A 2018, 205, 614–621. [Google Scholar] [CrossRef] [PubMed]
- Alatawi, R.A.S.; Monier, M.; Elsayed, N.H. Chiral separation of (±)-methamphetamine racemate using molecularly imprinted sulfonic acid functionalized resin. J. Colloid Interface Sci. 2018, 541, 654–663. [Google Scholar] [CrossRef] [PubMed]
- Pisarev, O.A.; Polyakova, I.V. Molecularly imprinted polymers based on methacrylic acid and ethyleneglycol dimethacrylate for l-lysine recognition. React. Funct. Polym. 2018, 130, 98–110. [Google Scholar] [CrossRef]
- Moniera, M.; El-Mekabaty, A.; Abdel-Latifa, D.A. Synthesis and evaluation of enantio-selective l-histidine imprinted salicylic acid functionalized resin. React. Funct. Polym. 2018, 128, 104–113. [Google Scholar] [CrossRef]
- Monier, M.; Youssef, I.; Abdel-Latifa, D.A. Synthesis of imprinted styrene-maleic acid functionalized resin for enantioselective extraction of R-amphetamine. Chem. Eng. J. 2019, 356, 693–701. [Google Scholar] [CrossRef]
- Yang, Y.-J.; Liu, X.-W.; Kong, X.-J.; Qin, Z.; Jiao, Z.-H.; Li, S.-H.; Li, J.-Y. Preparation and evaluation of oseltamivir molecularly imprinted polymer silica gel as liquid chromatography stationary phase. Molecules 2018, 23, 1881. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.H.; Dong, Q.; Ying, L.L.; Chi, S.S.; Lan, Y.H.; Huang, Y.P.; Liu, Z.S. Enhancement of selective separation on molecularly imprinted monolith by molecular crowding agent. Anal. Bioanal. Chem. 2017, 409, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Okamoto, Y. Efficient separation of enantiomers using stereoregular chiral polymers. Chem. Rev. 2016, 116, 1094–1138. [Google Scholar] [CrossRef] [PubMed]
- Nakano, T. Optically active synthetic polymers as chiral stationary phases in HPLC. J. Chromatogr. A 2001, 906, 205–225. [Google Scholar] [CrossRef]
- Ding, P.; Chang, B.; Qing, G.; Sun, T. New approach for chiral separation: From polysaccharide-based materials to chirality-responsive polymers. Sci. China Chem. 2014, 57, 1492–1506. [Google Scholar] [CrossRef]
- Maeda, K.; Yashima, E. Helical polyacetylenes induced via noncovalent chiral interactions and their applications as chiral materials. Top. Curr. Chem. 2017, 375, 72. [Google Scholar] [CrossRef] [PubMed]
- Mallakpour, S.; Khadem, E. Hybrid optically active polymer/metal oxide composites: Recent advances and challenges. In Hybrid Polymer Composite Materials; Woodhead Publishing: Sawston, UK, 2017; pp. 379–406. [Google Scholar] [CrossRef]
- Ding, X.; Yang, J.; Dong, Y. Advancements in the preparation of high-performance liquid chromatographic organic polymer monoliths for the separation of small-molecule drugs. J. Pharm. Anal. 2018, 8, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K.; Maruta, M.; Sakai, Y.; Ikai, T.; Kanoh, S. Synthesis of optically active poly(diphenylacetylene)s using polymer reactions and an evaluation of their chiral recognition abilities as chiral stationary phases for HPLC. Molecules 2016, 21, 1487. [Google Scholar] [CrossRef] [PubMed]
- Ikai, T.; Awata, S.; Kudo, T.; Ishidate, R.; Maeda, K.; Kanoh, S. Chiral stationary phases consisting of π-conjugated polymers bearing glucose-linked biphenyl units: Reversible switching of resolution abilities based on a coil-to-helix transition. Polym. Chem. 2017, 29, 4190–4198. [Google Scholar] [CrossRef]
- Zhang, K.; Cai, S.L.; Yan, Y.L.; He, Z.H.; Lin, H.M.; Huang, X.L.; Zheng, S.R.; Fan, J.; Zhan, W.G. Construction of a hydrazone-linked chiral covalent organic framework-silica composite as the stationary phase for high-performance liquid chromatography. J. Chromatogr. A 2017, 1519, 100–109. [Google Scholar] [CrossRef] [PubMed]
- De Mello, A. FOCUS. Lab Chip 2002, 3, 48N–54N. [Google Scholar] [CrossRef] [PubMed]
- Grinias, J.P.; Kennedy, R.T. Advances in and prospects of microchip liquid chromatography. TrAC-Trends Anal. Chem. 2016, 81, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Kecskemeti, A.; Gaspar, A. Particle-based liquid chromatographic separations in microfluidic devices—A review. Anal. Chim. Acta 2018, 1021, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Manz, A.; Graber, N.; Widmer, H.M. Miniaturized total chemical analysis systems: A novel concept for chemical sensing. Sens. Actuator B Chem. 1990, 1, 244–248. [Google Scholar] [CrossRef]
- Baharfar, M.; Yamini, Y.; Seidi, S.; Karami, M. Quantitative analysis of clonidine and ephedrine by a microfluidic system: On-chip electromembrane extraction followed by high performance liquid chromatography. J. Chromatogr. B 2017, 1068–1069, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Payán, M.R.; Murillo, E.S.; Coello, J.; López, M.Á.B. A comprehensive study of a new versatile microchip device based liquid phase microextraction for stopped-flow and double-flow conditions. J. Chromatogr. A 2018, 1556, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Zhai, H.; Huang, L.; Chen, Z.; Su, Z.; Yuan, K.; Liang, G.; Pan, Y. Chip-based molecularly imprinted monolithic capillary array columns coated GO/SiO2 for selective extraction and sensitive determination of rhodamine B in chili powder. Food Chem. 2017, 214, 664–669. [Google Scholar] [CrossRef] [PubMed]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teixeira, J.; Tiritan, M.E.; Pinto, M.M.M.; Fernandes, C. Chiral Stationary Phases for Liquid Chromatography: Recent Developments. Molecules 2019, 24, 865. https://doi.org/10.3390/molecules24050865
Teixeira J, Tiritan ME, Pinto MMM, Fernandes C. Chiral Stationary Phases for Liquid Chromatography: Recent Developments. Molecules. 2019; 24(5):865. https://doi.org/10.3390/molecules24050865
Chicago/Turabian StyleTeixeira, Joana, Maria Elizabeth Tiritan, Madalena M. M. Pinto, and Carla Fernandes. 2019. "Chiral Stationary Phases for Liquid Chromatography: Recent Developments" Molecules 24, no. 5: 865. https://doi.org/10.3390/molecules24050865
APA StyleTeixeira, J., Tiritan, M. E., Pinto, M. M. M., & Fernandes, C. (2019). Chiral Stationary Phases for Liquid Chromatography: Recent Developments. Molecules, 24(5), 865. https://doi.org/10.3390/molecules24050865