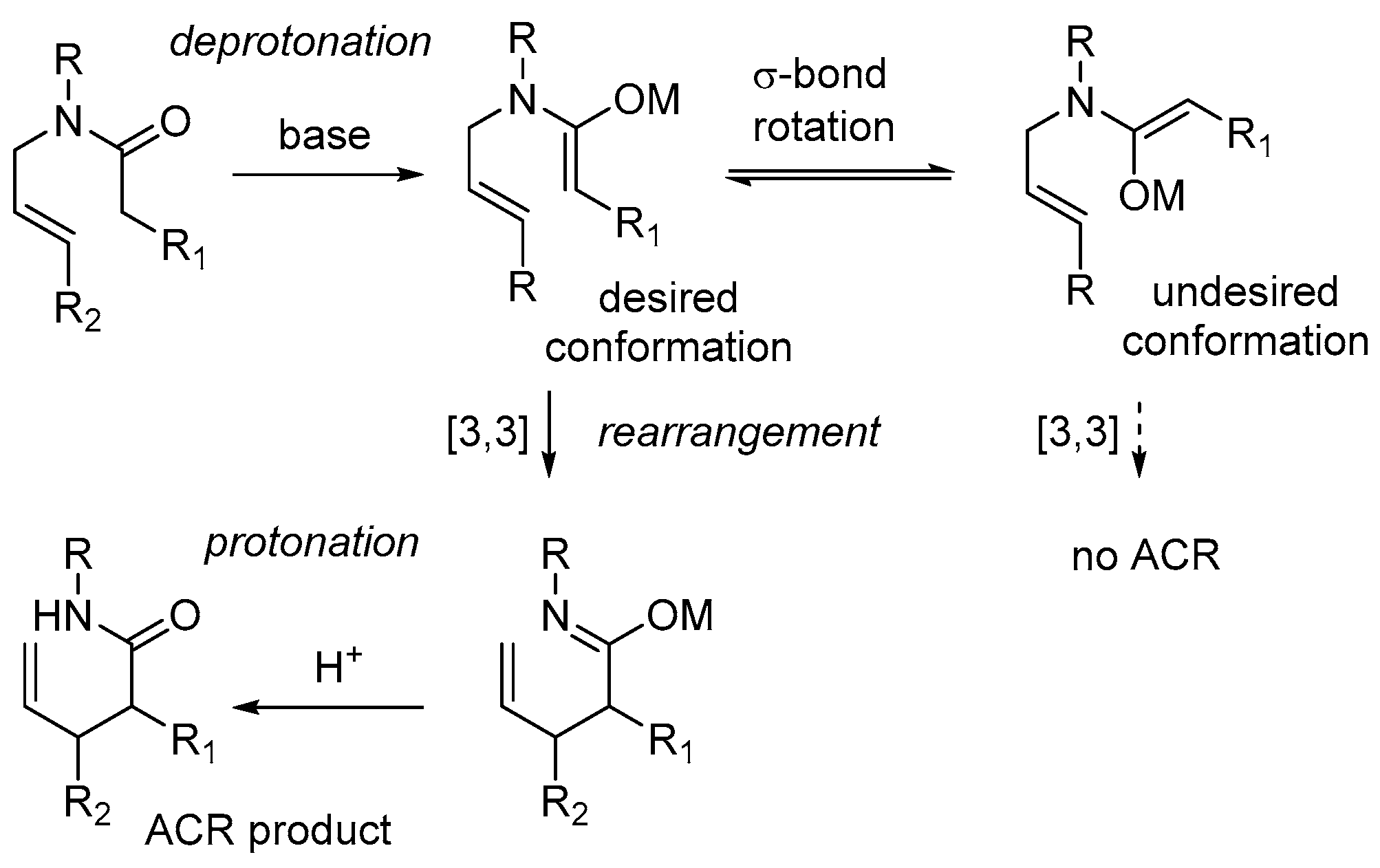
3.1. General Information
Unless noted otherwise, all starting materials and reagents were obtained from commercial suppliers and were used without further purification. Tetrahydrofuran was distilled from sodium benzophenone ketyl. Dichloromethane was freshly distilled from calcium hydride. All solvents used for routine isolation of products and chromatography were reagent grade. Air- and moisture-sensitive reactions were performed under argon atmosphere. Flash column chromatography was performed using silica gel 60 (230–400 mesh, Merck, Darmstadt, Germany) with the indicated solvents. Thin-layer chromatography was performed using 0.25 mm silica gel plates (Merck). 1H-NMR data were reported in the order of chemical shift, multiplicity (s, singlet; d, doublet; t, triplet; q, quartet; m, multiplet and/or multiple resonance), number of protons, and coupling constant in hertz (Hz).
3.1.1. Methyl 1-butyrylpiperidine-2-carboxylate (2)
To a solution of methyl pipecolinate hydrochloride 1 (3.9 g, 22 mmol) in THF (40 mL), a solution of Na2CO3 (11 g, 110 mmol) in H2O was added at 0 °C. After addition of n-butyryl chloride (2.7 mL, 26 mmol) at 0 °C, the reaction mixture was stirred for 24 h. After filtration of insoluble solids, reaction mixture was extracted EtOAc twice. Organic layers were dried over MgSO4, filtered, evaporated, and purified by column chromatography on silica gel (EtOAc:n-hexane = 1:1) to afford methyl ester 2 (4.1 g, 89%) as a colorless oil. IR (KBr) νmax 2953, 2869, 1741, 1648, 1426, 1322 cm−1; 1H-NMR (CDCl3, 400 MHz, mixture of rotamers) δ 5.25 (d, 1H, J = 5.2 Hz), 4.47–4.40, 3.65–3.61 (m, 1H), 3.57 (s, 3H), 3.09 (dt, 1H, J = 2.8, 12.8 Hz), 2.48–2.47, 2.18–2.03 (m, 2H), 2.24 (t, 2H, J = 7.6 Hz), 1.59–1.43 (m, 4H), 1.36–1.13 (m, 2H), 0.83 (t, 3H, J = 7.6 Hz); 13C-NMR (CDCl3, 100 MHz, mixture of rotamers) δ 172.7, 172.3, 171.6, 171.1, 55.7, 52.0, 51.7, 51.4, 43.1, 38.9, 35.0, 34.7, 27.0, 26.3, 25.0, 24.3, 20.7, 20.6, 18.2, 13.5; LRMS (FAB) m/z 214 (M + H+); HRMS (FAB) calcd for C11H20NO3 (M + H+): 214.1443, found 214.1448.
3.1.2. 1-Butyrylpiperidine-2-carbaldehyde (3)
To a solution of methyl ester 2 (850 mg, 4.0 mmol) in CH2Cl2 (10 mL), DIBAL (1.0 M in toluene, 8.0 mL, 8.0 mmol) was added at –78 °C and stirred for 3 h. Then, 15% sodium potassium tartrate solution (10 mL) was added to reaction mixture and stirred for 12 h at room temperature. Reaction mixture was extracted CH2Cl2 twice. Organic layers were dried over MgSO4, filtered, evaporated, and purified by column chromatography on silica gel (EtOAc:n-hexane = 1:1) to afford aldehyde 3 (300 mg, 41%) as a colorless oil. IR (KBr) νmax 2939, 2869, 1731, 1643, 1425 cm−1; 1H-NMR (CDCl3, 400 MHz, mixture of rotamers) δ 9.62 (s), 9.45 (s, 1H), 5.33 (d, J = 5.2 Hz), 5.07–5.05 (m, 1H), 4.62–4.58 (m), 4.35–4.33 (m), 3.72–3.64 (m, 2H), 3.08 (dt, 1H, J = 2.8, 11.6 Hz), 2.35–2.13 (m, 4H), 1.68–1.53 (m, 5H), 1.43–1.18 (m, 2H), 0.89 (t, 3H, J = 7.2 Hz); 13C-NMR (CDCl3, 100 MHz, mixture of rotamers) δ 201.0, 200.2, 173.1, 62.3, 58.9, 51.9, 51.6, 44.1, 43.3, 35.2, 35.0, 26.5, 25.3, 24.5, 23.2, 20.9, 20.8, 18.5, 13.7; LRMS (FAB) m/z 184 (M + H+); HRMS (FAB) calcd for C10H18NO2 (M + H+): 184.1338, found 184.1340.
3.1.3. 1-(2-Vinylpiperidin-1-yl)butan-1-one (4)
To a suspension of methyl triphenylphosphonium bromide (850 mg, 2.4 mmol) in THF (5 mL), KOtBu (1.0 M in THF, 2.2 mL, 2.2 mmol) was added at 0 °C and stirred for 30 min. After addition of aldehyde 2 (290 mg, 1.6 mmol) in THF (2 mL), reaction mixture was stirred for 10 min and quenched with addition of H2O. Reaction mixture was extracted EtOAc twice. Organic layers were dried over MgSO4, filtered, evaporated and purified by column chromatography on silica gel (EtOAc:n-hexane = 1:2 to 1:1) to afford allyl amide 4 (220 mg, 77%) as a colorless oil. IR (KBr) νmax 2937, 2867, 1645, 1425, 1244 cm−1; 1H-NMR (CDCl3, 400 MHz, mixture of rotamers) δ 5.71–5.68 (m, 1H), 5.17 (m, 1H), 5.34 (bs), 3.61–3.58 (m, 1H), 5.02–4.98 (m, 1H), 4.48 (bs, 1H), 3.08 (t, J = 11.6 Hz), 2.60 (t, 1H, J = 11.6 Hz), 2.32–2.20 (m, 2H), 1.79–1.76 (m, 1H), 1.66–1.40 (m, 6H), 1.34 (m, 1H), 0.93–0.89 (m, 3H); 13C-NMR (CDCl3, 100 MHz) δ 172.4, 171.7, 153.5, 136.5, 116.2, 115.8, 54.4, 51.6, 49.5, 43.3, 41.7, 37.2, 35.5, 35.3, 35.0, 30.0, 28.3, 26.3, 25.2, 19.5, 18.8, 13.9; LRMS (FAB) m/z 182 (M + H+); HRMS (FAB) calcd for C11H20NO (M + H+): 182.1545, found 182.1543.
3.1.4. (E)-3-ethyl-3,4,7,8,9,10-hexahydroazecin-2(1H)-one (5)
Procedure A; To a solution of allyl amide 4 (29 mg, 0.15 mmol) in toluene (1 mL), iPrMgCl (2.0 M in THF, 0.15 mL, 0.30 mmol) was added at reflux condition. After stirring at same temperature for 30 min, reaction mixture was cooled down to room temperature and quenched with brine and extracted with EtOAc. Organic layers were dried over MgSO4, filtered, evaporated and purified by column chromatography on silica gel (EtOAc:n-hexane = 2:1) to afford lactam 5 (22 mg, 78%) as an amorphous solid.
Procedure B; To a solution of allyl amide 4 (29 mg, 0.15 mmol) in toluene (2 mL), LHMDS (1.0 M in n-hexane, 0.30 mL, 0.30 mmol) was added at reflux condition. After stirring at same temperature for 12 h, reaction mixture was cooled down to room temperature and quenched with brine and extracted with EtOAc. Organic layers were dried over MgSO4, filtered, evaporated, and purified by column chromatography on silica gel (EtOAc:n-hexane = 2:1) to afford lactam 5 (18 mg, 62%) as an amorphous solid. IR (KBr) νmax 3315, 2925, 2441, 1637, 1550, 1451 cm−1; 1H-NMR (CD3OD, 300 MHz) δ 7.37 (bs, 1H), 5.38–5.16 (m, 2H), 3.49–3.41 (m, 1H), 2.69–2.62 (m, 1H), 2.17–2.10 (m, 1H), 2.07 -1.97 (m, 2H), 1.82–1.74 (m, 3H), 1.67–1.52 (m, 2H), 1.40–1.37 (m, 1H), 1.30–1.19 (m, 2H), 0.77 (t, 3H, J = 3.9 Hz); 13C-NMR (CDCl3, 75 MHz) δ 174.8, 134.5, 127.7, 52.7, 40.2, 37.3, 32.8, 29.7, 29.0, 24.1, 12.4; LRMS (FAB) m/z 182 (M + H+); HRMS (FAB) calcd for C11H20NO (M + H+): 182.1545, found 182.1543.
3.1.5. (E)-tert-butyl 2-(3-hydroxyprop-1-enyl)piperidine-1-carboxylate (7)
To a solution of unsaturated ester 6 (2.9 g, 10 mmol) in THF (20 mL), DIBAL (1.0 M in toluene, 22 mL, 22 mmol) was added at 0 °C and stirred for 30 min. 15% sodium potassium tartrate solution was added to reaction mixture and stirred for 5 h at room temperature. Reaction mixture was extracted EtOAc twice. Organic layers were dried over MgSO4, filtered, evaporated and purified by column chromatography on silica gel (EtOAc:n-hexane = 1:1) to afford primary alcohol 7 (2.0 g, 81%) as a colorless oil. IR (KBr) νmax 3443, 2936, 2862, 1690, 1515, 1415, 1367 cm−1; 1H-NMR (CDCl3, 500 MHz, mixture of rotamers) δ 5.64–5.56 (m, 2H), 4.74 (b, 1H) 5.58–5.54 (m, 1H), 4.10–4.09 (m, 2H), 3.88 (d, 2H, J = 13.5 Hz), 2.77 (t, 1H, J = 12.5 Hz), 2.21 (b, 1H), 1.72–1.58 (m, 2H), 1.54–1. 52 (m, 2H), 1.40 (s, 9H); 13C-NMR (CDCl3, 100 MHz, mixture of rotamers) δ 171.0, 155.2, 130.4, 129.6, 128.6, 79.3, 62.8, 60.2, 51.4, 50.3, 39.6, 29.0, 28.3, 25.3, 20.8, 19.3, 18.9, 14.0; LRMS (FAB) m/z 242 (M + H+); HRMS (FAB) calcd for C13H24NO3 (M + H+): 242.1756, found 242.1752.
3.1.6. (E)-tert-butyl 2-(3-(tert-butyldiphenylsilyloxy)prop-1-enyl)piperidine-1-carboxylate (8)
To a solution of alcohol 7 (920 mg, 3.8 mmol) in dimethylformamide (DMF, 8 mL), imidazole (390 mg, 5.7 mmol), and TBDPSCl (0.99 mL, 3.8 mmol) were added at 0 °C and stirred for 12 h. After addition of H2O, reaction mixture was diluted with EtOAc and washed with H2O three times. Organic layers were dried over MgSO4, filtered, evaporated, and purified by column chromatography on silica gel (EtOAc:n-hexane=1:10 to 1:5) to afford TBDPS ether 8 (1.4 g, 77%) as a colorless oil. IR (KBr) νmax 3431, 2934, 2857, 1692, 1469, 1424 cm−1; 1H-NMR (CDCl3, 300 MHz, mixture of rotamers) δ 7.67–7.65 (m, 4H), 7.42–7.32 (m, 6H), 5.66 (dd, 1H, J = 4.8, 15.6 Hz), 5.58–5.54 (m, 1H), 4.76 (s, 1H), 4.20 (dd, 2H, J = 1.5, 4.4 Hz), 3.90 (d, 1H, J = 12Hz), 2.76 (t, 1H, J = 12.7 Hz), 1.67–1.40 (m, 6H), 1.42 (s, 9H), 1.03 (s, 9H); 13C-NMR (CDCl3, 100 MHz, mixture of rotamers) δ 170.9, 15.2, 135.4, 133.6, 130.0, 129.5, 128.6, 127.5, 79.0, 64.0, 60.2, 29.3, 28.3, 26.7, 25.4, 20.8, 19.4, 19.1, 14.0; LRMS (FAB) m/z 480 (M + H+).
3.1.7. (E)-1-(2-(3-(tert-butyldiphenylsilyloxy)prop-1-enyl)piperidin-1-yl)butan-1-one (9)
To a solution of TBDPS ether 8 (62 mg, 0.13 mmol) in CH2Cl2 (2 mL), trifluoroacetic acid (2 mL) was added and stirred for 30 min. After evaporation of reaction mixture, the residue was dissolved in CH2Cl2 (2 mL) and treated with iPr2NEt (0.1 mL) and n-butyryl chloride (0.05 mL) at 0 °C and stirred for 12 h. Reaction mixture was quenched with H2O and extracted with CH2Cl2 twice. Organic layers were dried over MgSO4, filtered, evaporated, and purified by column chromatography on silica gel (EtOAc:n-hexane = 1:3 to 1:2) to afford butyryl amide 9 (55 mg, 95%) as a colorless oil. IR (KBr) νmax 2935, 2858, 1732, 1644, 1427, 1238 cm−1; 1H-NMR (CDCl3, 500 MHz, mixture of rotamers) δ 7.68–7.62 (m, 4H), 7.43–7.36 (m, 6H), 5.68 (b, 1H), 5.55 (d, 1H, J = 15.5 Hz), 4.52 (b, 1H), 4.23 (s, 1H), 3.70–3.59 (m, 1H), 3.07, 2.60 (m, 1H), 2.31 (t, 2H, J = 7.5 Hz), 2.33–2.24 (m, 1H), 1.75 (m, 1H), 1.69–1.35 (m, 7H), 1.06 (s, 9H), 0.96 (t, 3H, J = 7.5 Hz); 13C-NMR (CDCl3, 125MHz) δ 177.3, 135.5, 135.4, 133.5, 130.7, 129.6, 128.2, 127.6, 64.0, 63.7, 53.7, 48.7, 37.2, 35.8, 35.6, 35.1, 30.4, 28.9, 26.7, 26.4, 25.3, 19.5, 19.1, 18.9, 18.2, 13.9, 13.6; LRMS (FAB) m/z 450 (M + H+); HRMS (FAB) calcd for C28H40NO7Si (M + H+): 450.2828, found 450.2832.
3.1.8. (E)-1-(2-(3-hydroxyprop-1-enyl)piperidin-1-yl)butan-1-one (10)
To a solution of n-butyrl amide 9 (370 mg, 0.82 mmol) in THF (10 mL), acetic acid (0.1 mL, 1.6 mmol) and TBAF (1.0 M in THF, 1.2 mL, 1.2 mmol) were added and stirred for 30 min. After addition of aq. NaHCO3, reaction mixture was extracted with EtOAc three times. Organic layers were dried over MgSO4, filtered, evaporated and purified by column chromatography on silica gel (EtOAc:MeOH = 20:1) to afford primary alcohol 10 (160 mg, 92%) as a colorless oil. IR (KBr) νmax 3398, 2936, 2865, 1620, 1436, 1253 cm−1; 1H-NMR (CDCl3, 400 MHz, mixture of rotamers) δ 5.66–5.58 (m, 2H), 5.31, 4.49 (bs, 1H), 4.43, 3.39 (d, 1H, J = 12.8 Hz), 3.09, 2.60 (t, 1H, J = 12.4 Hz), 2.26 (t, 2H, J = 7.2 Hz), 2.26–2.19 (m, 1H), 1.74 (m, 1H), 1.64–1.54 (m, 7H), 1.34 (m, 1H), 0.90 (t, 3H, J = 6.0 Hz); 13C-NMR (CDCl3, 100 MHz, mixture of rotamers) δ 175.6, 172.4, 171.9, 131.2, 130.9, 129.3, 128.8, 62.9, 62.5, 53.6, 48.8, 41.8, 37.2, 35.5, 35.1, 30.2, 28.7, 26.1, 25.2, 19.4, 18.8, 13.9; LRMS (FAB) m/z 212 (M + H+); HRMS (FAB) calcd for C12H22NO2 (M + H+): 212.1651, found 212.1644.
3.1.9. (E)-1-(2-(3-(tert-butyldimethylsilyloxy)prop-1-enyl)piperidin-1-yl)butan-1-one (11)
To a solution of alcohol 10 (72 mg, 0.34 mmol) in CH2Cl2 (2 mL), iPr2NEt (0.2 mL) and TBSOTf (0.05 mL) were added at 0 °C and stirred for 12 h. After addition of H2O, reaction mixture was extracted with CH2Cl2. Organic layers were dried over MgSO4, filtered, evaporated, and purified by column chromatography on silica gel (EtOAc:n-hexane = 1:3 to 1:2) to afford TBS ether 11 (110 mg, 99%) as a colorless oil. IR (KBr) νmax 2934, 2857, 1645, 1539, 1424 cm−1; 1H-NMR (CDCl3, 400 MHz, mixture of rotamers) δ 5.59–5.49 (m, 2H), 5.35 (bs), 3.59–3.57 (m, 1H), 4.49–4.45 (m, 1H), 4.13 (s, 2H), 3.09, 259 (bs, 1H), 2.26 (t, 1H, J = 5.6 Hz), 2.20 (bs, 1H), 1.73 (m, 1H), 1.64–1.50 (m, 6H), 1.34 (b, 1H), 0.90 (t, 3H, J = 4.6 Hz), 0.85 (s, 9H), 0.00 (s, 6H); 13C-NMR (CDCl3, 100 MHz, mixture of rotamers) δ 172.2, 171.5, 131.1, 128.0, 127.8, 63.3, 62.9, 53.6, 48.6, 41.7, 37.2, 35.5, 35.1, 30.3, 28.8, 26.3, 25.8, 25.3, 19.5, 18.8, 18.2, 13.9; LRMS (FAB) m/z 326 (M + H+); HRMS (FAB) calcd for C18H36NO2Si (M + H+): 326.2515, found 326.2520.
3.1.10. (E)-1-(2-(3-(triethylsilyloxy)prop-1-enyl)piperidin-1-yl)butan-1-one (12)
To a solution of alcohol 10 (100 mg, 0.47 mmol) in CH2Cl2 (2 mL), iPr2NEt (0.17 mL, 0.95 mmol) and TESCl (1.0 M in THF, 0.62 mL, 0.62 mmol were added at 0 °C and stirred for 2 h. After addition of H2O, reaction mixture was extracted with CH2Cl2. Organic layers were dried over MgSO4, filtered, evaporated and purified by column chromatography on silica gel (EtOAc:n-hexane = 1:3 to 1:2) to afford TES ether 12 (120 mg, 78%) as a colorless oil. IR (KBr) νmax 2955, 1645, 1537, 1458, 1118 cm−1; 1H-NMR (CDCl3, 300 MHz, mixture of rotamers) δ 5.57–5.36 (m, 2H), 4.50 (b, 1H), 4.13 (s, 2H), 3.57–3.47 (m), 3.41–3.37 (m, 1H), 3.10 (t, J = 12.0 Hz), 2.60 (t, 1H, J = 12.0 Hz), 2.27 (t, 2H, J = 7.5 Hz), 1.74–1.35 (m, 7H), 0.92 (t, 9H, J = 8.1 Hz), 0.57 (q, 6H, J = 8.1 Hz); LRMS (FAB) m/z 326 (M + H+); HRMS (FAB) calcd for C18H36NO2Si (M + H+): 326.2515, found 326.2519.
3.1.11. (E)-1-(2-(3-(tert-butyldiphenylsilyloxy)prop-1-enyl)piperidin-1-yl)hex-5-yn-1-one (13)
To a solution of TBDPS ether 8 (72 mg, 0.15 mmol) in CH2Cl2 (1 mL), trifluoroacetic acid (1 mL) was added and stirred for 30 min. After evaporation of reaction mixture, the residue was dissolved in CH2Cl2 (2 mL) and treated with EDCI (57 mg, 0.3 mmol), DMAP (37 mg, 0.3 mmol) and 5-hexynoic acid (33 μL, 0.3 mmol) at 0 °C and stirred for 12 h. Reaction mixture was quenched with H2O and extracted with CH2Cl2 twice. Organic layers were dried over MgSO4, filtered, evaporated, and purified by column chromatography on silica gel (EtOAc:n-hexane = 1:3 to 1:2) to afford 5-hexynyl amide 13 (62 mg, 87%) as a colorless oil. IR (KBr) νmax 3300, 2935, 2857, 1642, 1428, 1256 cm−1; 1H-NMR (CDCl3, 400 MHz, mixture of rotamers) δ 7.66–7.60 (m, 4H), 7.42–7.24 (m, 6H), 5.71–5.63 (m, 1H), 5.57–5.50 (m, 1H), 4.53–4.45 (m, 1H), 4.11 (d, 2H, J = 7.2 Hz), 3.69–3.60 (m, 1H), 3.08–3.02 (m, 1H), 2.47–2.44 (m, 2H), 2.32 (b, 2H), 1.94–1.51 (m, 9H), 1.12 (s, 9H); 13C-NMR (CDCl3, 100 MHz, mixture of rotamers) δ 135.6, 135.4, 133.5, 130.8, 129.6, 127.6, 83.8, 68.8, 63.8, 31.9, 31.4, 30.4, 28.9, 26.7, 26.3, 23.9, 19.5, 19.2 18.0; LRMS (FAB) m/z 474 (M + H+); HRMS (FAB) calcd for C30H40NO7Si(M + H+): 474.2828, found 474.2816.
3.1.12. (E)-1-(2-(3-(tert-butyldiphenylsilyloxy)prop-1-enyl)piperidin-1-yl)hex-5-yn-1-one (14)
To a solution of TBDPS ether 8 (1.0 g, 2.1 mmol) in CH2Cl2 (10 mL), trifluoroacetic acid (5 mL) was added and stirred for 1 h. After addition of aq. Na2CO3, reaction mixture was extracted with CH2Cl2. Organic layers were dried over MgSO4, filtered, evaporated, and dissolved in CH2Cl2 (20 mL). To this solution, 3-phenylpropionic acid (0.5 g, 3.6 mmol), EDCI (0.7 g, 3.6 mmol) and DMAP (0.9 g, 7.2 mmol) were added and stirred for 12 h. Reaction mixture was quenched with aq. NH4Cl and extracted with CH2Cl2. Organic layers were dried over MgSO4, filtered, evaporated, and purified by column chromatography on silica gel (EtOAc:n-hexane = 1:5) to afford amide 14 (0.9 g, 83% for 2 steps) as a pale yellow oil. 1H-NMR (CDCl3, 300 MHz, mixture of rotamers) δ 7.77–7.63 (m, 4H), 7.49–7.35(m, 6H), 7.33–7.18 (m, 5H), 5.74–5.43 (m, 2H), 4.50 (s, 1H), 4.25 (s, 2H), 3.46 (m, 1H), 3.00 (t, 2H, J = 7.8, 15.9 Hz), 2.67 (t, 2H, J = 7.5, 15.6 Hz), 1.48 (m, 6H), 1.07 (m, 10H); 13C-NMR (CDCl3, 125 MHz, mixture of rotamers) δ 71.6, 170.9, 141.3, 135.5, 134.7, 134.6, 133.6, 130.9, 129.7(2), 129.6, 128.5, 128.4, 128.0, 127.8, 127.6, 127.4(2) 126.1, 77.3, 77.2, 77.0, 76.8, 64.1, 63.7, 53.9, 49.1, 41.8, 37.6, 34.9, 31.7, 30.3, 26.8, 26.2, 25.9, 25.8, 25.2, 19.5, 19.2, 18.6, 18.5. LRMS (EI) m/z 512 (M + H+); HRMS (EI) calcd for C33H41O2NSi (M+): 511.2907, found 511.2903.
3.1.13. (E)-tert-butyl 2-(3-(benzyloxy)prop-1-enyl)piperidine-1-carboxylate (15)
To a solution of alcohol 7 (690 mg, 3.2 mmol) in THF (15 mL), TBAI (120 mg, 0.32 mmol), NaH (60% mineral oil, 150 mg, 3.7 mmol) and benzyl bromide (0.42 mL, 3.5 mmol) were added at 0 °C and stirred for 12 h at room temperature. After addition of H2O, reaction mixture was extracted with EtOAc twice. Organic layers were dried over MgSO4, filtered, evaporated and purified by column chromatography on silica gel (EtOAc:n-hexane = 1:10 to 1:5) to afford benzyl ether 15 (920 mg, 86%) as a colorless oil. IR (KBr) νmax 2935, 2857, 1691, 1453, 1409, 1365 cm−1; 1H-NMR (CDCl3, 400 MHz, mixture of rotamers) δ 7.31–7.22 (m, 5H), 5.68–5.55 (m, 2H), 4.80 (s, 1H), 4.48 (d, 2H, J = 4.8 Hz), 4.01–4.00 (m, 2H), 3.92 (d,1H, J = 13.2 Hz), 2.80 (t, 1H, J = 11.6 Hz), 1.72–1.62 (m, 2H), 1.60–1.50 (m, 2H), 1.47–1.31 (m, 2H), 1.43 (s, 9H); 13C-NMR (CDCl3, 100 MHz, mixture of rotamers) δ 155.0, 138.1, 131.8, 128.2, 127.5, 127.4, 79.1, 72.3, 71.7, 70.2, 66.1, 39.6, 28.9, 28.3, 25.3, 19.4; LRMS (FAB) m/z 332 (M + H+); HRMS (FAB) calcd for C20H30NO3 (M + H+): 332.2226, found 332.2226.
3.1.14. (E)-1-(2-(3-(benzyloxy)prop-1-enyl)piperidin-1-yl)propan-1-one (16)
Benzyl ether
15 (120 mg, 0.36 mmol) was converted to propyl amide
16 (75 mg, 72%) using same procedure as described in
Section 3.1.7. IR (KBr) ν
max 2936, 2857, 1644, 1538, 1426, 1251 cm
−1;
1H-NMR (CDCl
3, 400 MHz, mixture of rotamers) δ 7.33–7.11 (m, 5H), 5.70–5.55 (m, 2H), 5.39 (bs), 3.60 (d, 1H,
J = 11.8 Hz), 4.48 (s, 2H), 4.48 (bs, 1H), 3.97 (d, 2H,
J = 5.8 Hz), 3.09 (t,
J = 12.3 Hz), 2.62 (t, 1H,
J = 12.3 Hz), 2.34–2.27 (m, 2H), 1.79–1.76 (m, 1H), 1.63–1.52 (m, 4H), 1.27 (m, 1H), 1.09 (t, 3H,
J = 6.0 Hz);
13C-NMR (CDCl
3, 100 MHz, mixture of rotamers) δ 173.0, 172.4, 138.1, 131.4, 130.9, 128.3, 128.0, 127.6, 126.9, 72.2, 72.0, 70.3, 69.9, 53.5, 48.8, 41.6, 37.3, 30.1, 28.5, 26.7, 26.2, 25.2, 19.6, 9.4, 9.0; LRMS (FAB)
m/
z 288 (M + H
+); HRMS (FAB) calcd for C
18H
26NO
2 (M + H
+): 288.1964, found 288.1956.
3.1.15. (E)-1-(2-(3-(benzyloxy)prop-1-enyl)piperidin-1-yl)butan-1-one (17)
Benzyl ether
15 (140 mg, 0.42 mmol) was converted to butyl amide
17 (97 mg, 77%) using same procedure as described in
Section 3.1.7. IR (KBr) ν
max 2935, 2858, 1641, 1425, 1362 cm
−1;
1H-NMR (CDCl
3, 400 MHz, mixture of rotamers) δ 7.33–7.23 (m, 5H), 5.70–5.60 (m, 2H), 5.39 (bs), 3.60 (d, 1H,
J = 9.0 Hz), 4.48 (m, 3H), 4.01 (d, 2H,
J = 3.2 Hz), 3.09 (t,
J = 12.3 Hz), 2.58 (t, 1H,
J = 12.3 Hz), 2.31–2.21 (m, 2H), 1.79–1.76 (m, 1H), 1.67–1.58 (m, 6H), 1.36 (m, 1H), 0.93 (t, 3H,
J = 7.2 Hz);
13C-NMR (CDCl
3, 100 MHz, mixture of rotamers) δ 176.6, 172.3, 171.8, 138.1, 137.9, 131.4, 130.9, 128.3, 127.7, 127.6, 72.2, 71.9, 70.3, 69.9, 53.6, 48.7, 41.8, 37.2, 35.8, 35.5, 35.1, 30.2, 28.6, 26.2, 25.2, 19.6, 19.4, 18.8, 18.6, 18.2, 13.9, 13.5; LRMS (FAB)
m/
z 302 (M + H
+); HRMS (FAB) calcd for C
19H
28NO
2 (M + H
+): 302.2120, found 302.2120.
3.1.16. (E)-1-(2-(3-(benzyloxy)prop-1-enyl)piperidin-1-yl)pentan-1-one (18)
Benzyl ether
15 (190 mg, 0.57 mmol) was converted to pentyl amide
18 (99 mg, 55%) using same procedure as described in
Section 3.1.7. IR (KBr) ν
max 2933, 2858, 1642, 1424, 1263 cm
−1;
1H-NMR (CD
3OD, 400 MHz, mixture of rotamers) δ 7.32–7.23 (m, 5H), 5.80–5.56 (m, 2H), 5.29 (bs), 4.67 (bs, 1H), 4.49 (s, 2H), 4.40 (d,
J = 12.3 Hz), 3.73 (d, 1H,
J = 12.4 Hz), 4.01 (d, 2H,
J = 3.2 Hz), 3.15 (t,
J = 12.9 Hz), 2.58 (t, 1H,
J = 12.5 Hz), 2.45–2.25 (m, 2H), 1.82 (b, 1H), 1.68–1.53 (m, 6H), 1.39–1.20 (m, 3H), 0.93–0.86 (m, 3H);
13C-NMR (CDCl
3, 100 MHz, mixture of rotamers) δ 176.4, 172.4, 1719, 138.1, 131.4, 131.0, 128.3, 128.0, 127.5, 72.0, 70.3, 69.9, 53.7, 48.7, 41.8, 37.3, 33.6, 33.3, 32.9, 30.2, 28.6, 27.5, 26.8, 26.3, 25.2, 22.5, 22.1, 19.6, 14.0, 13.6; LRMS (FAB)
m/
z 316 (M + H
+); HRMS (FAB) calcd for C
20H
30NO
2 (M + H
+): 316.2277, found 316.2278.
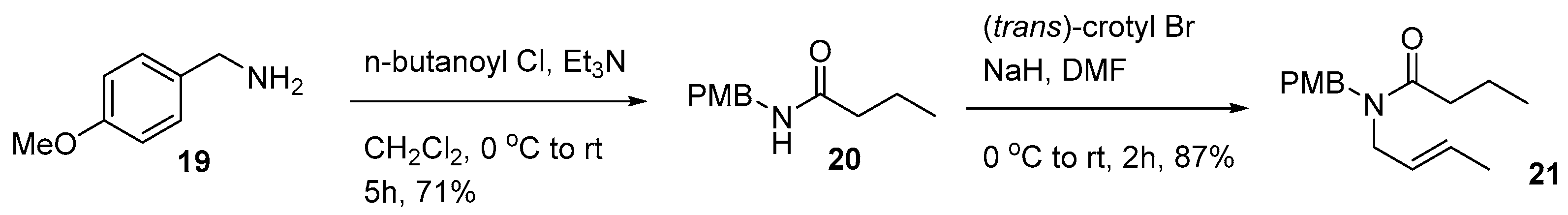
3.1.17. N-(4-methoxybenzyl)butyramide (20)
To a solution of 4-methoxybenzylamine 19 (300 mg, 2.2 mmol) in CH2Cl2 (10 mL), Et3N (0.4 mL, 2.8 mmol) and n-butyryl chloride (0.25 mL, 2.4 mmol) were at 0 °C and stirred for 5 h at room temperature. Reaction mixture was quenched with H2O and extracted with CH2Cl2 twice. Organic layers were dried over MgSO4, filtered, evaporated, and purified by column chromatography on silica gel (EtOAc:n-hexane = 1:1) to afford para-nethoxyl benzyl (PMB) amide 20 (320 mg, 71%) as a white amorphous solid. IR (KBr) νmax 3290, 2959, 1632, 1554, 1513 cm−1; 1H-NMR (CDCl3, 400 MHz) δ 7.17 (d, 2H, J = 8.6 Hz), 6.83 (d, 2H, J = 8.6 Hz), 4.85 (s, 1H), 4.26 (s, 2H), 3.72 (s, 3H), 2.17 (t, 2H, J = 7.2 Hz), 1.67–1.58 (m, 2H), 0.92 (t, 3H, J = 7.3Hz); 13C-NMR (CDCl3, 100 MHz) δ 176.4, 161.0, 132.8, 130.6, 115.6, 56.4, 44.4, 39.7, 21.1, 14.8; LRMS (FAB) m/z 208 (M + H+); HRMS (FAB) calcd for C12H18NO2 (M + H+): 208.1338, found 208.1334.
3.1.18. N-(but-2-enyl)-N-(4-methoxybenzyl)butyramide (21)
To a solution of PMB amide 20 (115 mg, 0.55 mmol) in DMF (3 mL), NaH (60% mineral oil, 26 mg, 0.66 mmol) and trans-crotyl bromide (0.10 mL, 0.83 mmol) were added at 0 °C and stirred for 2 h at room temperature. After dilution with EtOAc and reaction mixture was washed with H2O 3 times. Organic layers were dried over MgSO4, filtered, evaporated, and purified by column chromatography on silica gel (EtOAc:n-hexane = 1:3) to afford crotyl amide 21 (128 mg, 87%) as a colorless oil. IR (KBr) νmax 2962, 1645, 1513, 1459, 1248 cm−1; 1H-NMR (CDCl3, 400 MHz, mixtures of rotamers) δ 7.14–7.12 (m, 2H), 6.85–6.78 (m, 2H), 5.60–5.45 (m, 1H), 5.44–5.27 (m, 1H), 4.46 (s), 4.39 (s, 2H), 3.99 (d, J = 6.8 Hz), 3.79 (d, 1H, J = 6.8 Hz), 3.87 (d, J = 6.0 Hz), 3.68 (d, 1H, J = 6.0 Hz), 3. 76 (s), 3.74 (s, 3H), 2.32–2.27 (m, 2H), 1.71–1.50 (m, 5H), 0.96–0.84 (m, 3H); 13C-NMR (CDCl3, 100 MHz, mixtures of rotamers) δ 172.9, 172.8, 158.8, 158.7, 130.0, 129.8, 129.4, 128.8, 128.7, 128.1, 127.7, 127.5, 127.4, 127.3, 125.9, 125.8, 125.5, 114.1, 113.7, 55.1, 49.4, 49.1, 48.1, 47.3, 47.0, 46.7, 43.4, 41.2, 35.1, 34.9, 18.7, 17.6, 17.5, 13.9, 12.8; LRMS (FAB) m/z 262 (M + H+); HRMS (FAB) calcd for C16H24NO2 (M + H+): 262.1807, found 262.1800.
3.1.19. (Rac-3S,4R,E)-4-((tert-butyldiphenylsilyloxy)methyl)-3-ethyl-3,4,7,8,9,10-hexahydroazecin-2(1H)-one (9’)
TBDPS ether 9 (27 mg, 0.060 mmol) was converted to macrolactam 9’ as an amorphous solid using Procedure A (13 mg, 48%) or Procedure B (2 mg, 7.5%). IR (KBr) νmax 3300, 2929, 1645, 1541, 1458 cm−1; 1H-NMR (CDCl3, 300 MHz) δ 7.58–7.56 (m, 4H), 7.36–7.29 (m, 6H), 5.49–5.36 (m, 2H), 4.89 (d, 1H, J = 9.0 Hz), 3.71–3.56 (m, 3H), 2.77 (dd, 1H, J = 6.9, 12.9 Hz), 2.13 (m, 2H), 1.97 (dt, 2H, J = 7.2, 11.6 Hz), 1.89–1.13 (m, 7H), 1.00 (s, 3H), 0.76 (t, 3H, J = 7.2 Hz); 13C-NMR (CDCl3, 75MHz) δ 174.8, 135.6, 133.6, 133.2, 130.4, 129.7, 127.6, 63.7, 53.5, 49.0, 40.3, 33.1, 29.8, 26.9, 26.8, 21.4, 19.4, 12.8; LRMS (FAB) m/z 450 (M + H+); HRMS (FAB) calcd for C28H40NO7Si (M + H+): 450.2828, found 450.2832.
3.1.20. (Rac-3S,4R,E)-4-((tert-buyldimethylsilyloxy)methyl)-3-ethyl-3,4,7,8,9,10-hexahydroazecin-2(1H)-one (11’)
TBS ether 11 (48 mg, 0.15 mmol) was converted to macrolactam 11’ as an amorphous solid using Procedure A (33 mg, 69%) or Procedure B (12 mg, 25%). IR (KBr) νmax 3294, 2928, 2857, 1644, 1550 cm−1; 1H-NMR (CDCl3, 400 MHz) δ 5.43–5.36 (m, 2H), 4.97 (d, 1H, J = 5.2 Hz), 3.71–3.62 (m, 3H), 2.80 (dd, 1H, J = 6.0, 10.8 Hz), 2.21–2.13 (m, 2H), 1.97 (dt, 1H, J = 3.1, 6.5 Hz), 1.92–1.86 (m, 1H), 1.81–1.76 (m, 1H), 1.71–1.62 (m, 2H), 1.49–1.42 (m, 2H), 1.24–1.17 (m, 1H), 0.87 (s, 9H), 0.84 (t, 3H, J = 5.8 Hz), 0.01 (s, 3H), 0.01 (s, 3H); 13C-NMR (CD3OD, 100 MHz) δ 178.4, 134.3, 133.3, 65.4, 54.8, 51.6, 42.0, 34.4, 31.4, 27.2, 23.1, 20.0, 13.9, −4.3, −4.5; LRMS (FAB) m/z 326 (M + H+); HRMS (FAB) calcd for C18H36NO2Si (M + H+): 326.2515, found 326.2514.
3.1.21. (Rac-3S,4R,E)-4-((triethylsilyloxy)methyl)-3-ethyl-3,4,7,8,9,10-hexahydroazecin-2(1H)-one (12’)
TES ether 12 (40 mg, 0.12 mmol) was converted to macrolactam 12’ as an amorphous solid using Procedure A (31 mg, 69%). IR (KBr) νmax 3299, 2926, 1646, 1553, 1453, 137 cm−1; 1H-NMR (CDCl3, 400 MHz) δ 5.45–5.35 (m, 2H), 5.00 (d, 1H, J = 8.3 Hz), 3.71–3.60 (m, 3H), 2.80 (dd, 1H, J = 7.6, 13.7 Hz), 2.21–2.00 (m, 2H), 1.96 (dt, 1H, J = 4.0, 10.2 Hz), 1.92–1.87 (m, 1H), 1.81–1.67 (m, 3H), 1.50–1.42 (m, 2H), 1.25–1.16 (m, 1H), 0.92 (t, 9H, J = 8.0 Hz), 0.84 (t, 3H, J = 7.2 Hz), 0.56 (q, 6H, J = 8.0 Hz); 13C-NMR (CDCl3, 100 MHz) δ 174.9, 133.0, 130.5, 77.2, 62.5, 53.1, 49.0, 40.3, 33.1, 29.7, 21.4, 12.8, 6.7, 4.3; LRMS (FAB) m/z 326 (M + H+); HRMS (FAB) calcd for C18H36NO2Si (M + H+): 326.2515, found 326.2519.
3.1.22. (Rac-3S,4R,E)- 3-(but-3-ynyl)-4-((tert-butyldiphenylsilyloxy)methyl)-3,4,7,8,9,10-hexahydroazecin-2(1H)-one (13’)
Hexynyl amide 13 (22 mg, 46 μmol) was converted to macrolactam 13’ as an amorphous solid using Procedure A (12 mg, 55%). IR (KBr) νmax 3303, 2928, 2857, 1645, 1541, 1430 cm−1; 1H-NMR (CDCl3, 300 MHz,) δ 7.67–7.63 (m, 4H), 7.44–7.33 (m, 6H), 5.51 (dd, 1H, J = 8.7, 15.6 Hz), 5.46–5.36 (m, 1H), 5.01 (d, 1H, J = 9.6 Hz), 3.71 (d, 2H, J =2.7 Hz), 3.67 (m, 1H), 2.81 (dd, 1H, J = 7.5, 13.2 Hz), 2.44 (dd, 1H, J = 3.0, 10,8 Hz), 2.29–2.15 (m, 3H), 2.10–1.47 (m, 8H), 1.23 (m, 1H), 1.05 (s, 9H); 13C-NMR (CDCl3, 75 MHz) δ 174.1, 135.7, 135.6, 133.5, 133.4, 130.0, 129.7, 129.6, 127.7, 84.0, 68.6, 63.4, 50.0, 48.9, 40.3, 33.1, 27.0, 26.9, 19.3, 17.0; LRMS (FAB) m/z 474 (M + H+); HRMS (FAB) calcd for C30H40NO7 Si (M + H+): 474.2828, found 474.2832.
3.1.23. (Rac-3S,4R,E)- 3-benzyl-4-((tert-butyldiphenylsilyloxy)methyl)-3,4,7,8,9,10-hexahydroazecin-2(1H)-one (14’)
Phenylethyl amide 14 (1.0 g, 1.9 mmol) was converted to macrolactam 14’ as an amorphous solid using Procedure A (0.94 g, 94%). 1H-NMR (CDCl3, 500 MHz) δ 7.62–7.59 (m, 4H), 7.37–7.25 (m, 6H), 7.14–7.02 (m, 5H), 5.46–5.36 (m, 2H), 4.82 (d, 1H, J = 8.0 Hz), 3.84–3.75 (m, 2H), 3.51 (ddd, 1H, J = 9.0, 12.5, 17.0 Hz), 2.93 (dd, 1H, J = 12.0, 14.0 Hz), 2.61 (dd, 2H, J = 2.5, 13.5Hz), 2.57 (dd, 1H, J = 11.5, 19Hz), 2.39 (ddd, 1H, J = 2.5, 10.5, 13Hz), 2.24 (dd, 1H, J = 8.5, 18.5, Hz), 2.14 (dd, 1H, J = 3.5, 12.5, Hz) 1.83–1.71 (m, 2H), 1.63 (d, 1H, J = 7.0 Hz), 1.40 (dd, 1H, J = 11.5, 23.0 Hz), 1.06–0.99 (m, 9H); 13C-NMR (CDCl3, 125MHz) δ173.9, 141.0, 140.7, 135.7, 133.5, 129.9, 128.9, 128.5, 127.7, 127.0, 125.9, 65.2, 63.6, 53.8, 49.0, 40.3, 34.3, 33.1, 29.8, 27.0, 19.4; LRMS (EI) m/z 512 (M + H+); HR-MS (EI) calcd for C33H41O2NSi (M+): 511.2907, found 511.2908.
3.1.24. (Rac-3S,4R,E)- 4-(benzyloxymethyl)-3-methyl-3,4,7,8,9,10-hexahydroazecin-2(1H)-one (16’)
Propyl amide 16 (20 mg, 70 μmol) was converted to macrolactam 16’ as an amorphous solid using Procedure A (16 mg, 80%) or Procedure B (7 mg, 35%). IR (KBr) νmax 3312, 2924, 1644, 1550, 1452, 1367 cm−1; 1H-NMR (CD3OD, 300 MHz) δ 7.33–7.24 (m, 5H), 5.52–5.31 (m, 2H), 4.48 (q, 2H, J = 12.0 Hz), 3.54 (d, 2H, J = 4.5 Hz), 3.57–3.47 (m, 1H), 2.73 (dd, 1H, J = 6.9, 13.5 Hz), 2.37–2.12 (m, 3H), 1.93–1.72 (m, 3H), 1.51–1.27 (m, 2H), 1.05 (d, 3H, J = 6.3 Hz); 13C-NMR (CDCl3, 100 MHz) δ 175.6, 138.2, 133.8, 130.3, 128.3, 127.6, 127.5, 126.9, 73.2, 69.7, 48.5, 45.0, 40.1, 33.0, 29.6, 13.6; LRMS (FAB) m/z 288 (M + H+); HRMS (FAB) calcd for C18H26NO2 (M + H+): 288.1964, found 288.1956.
3.1.25. (Rac-3S,4R,E)-4-(benzyloxymethyl)-3-ethyl-3,4,7,8,9,10-hexahydroazecin-2(1H)-one (17’)
Butyryl amide 17 (20 mg, 66 μmol) was converted to macrolactam 17’ as an amorphous solid using Procedure A (14 mg, 70%) or Procedure B (3 mg, 15%). IR (KBr) νmax 2926, 2856, 2421, 1637, 1457 cm–1; 1H-NMR (CD3OD, 300 MHz) δ 7.24–7.15 (m, 5H), 5.41–5.22 (m, 2H), 4.38 (q, 2H, J = 12.0 Hz), 3.44 (d, 2H, J = 4.0 Hz), 3.45–3.44 (m, 1H), 2.66 (m, 1H), 2.16–2.02 (m, 3H), 1.78–1.18 (m, 7H), 0.73 (t, 3H, J = 7.2 Hz); 13C-NMR (CD3OD, 75 MHz) δ 178.2, 140.5, 134.5, 133.3, 130.1, 129.7, 129.4, 74.9, 72.3, 55.0, 42.0, 33.9, 31.4, 23.1, 21.4, 14.9, 13.8.; LRMS (FAB) m/z 302 (M + H+); HRMS (FAB) calcd for C19H28NO2 (M + H+): 302.2120, found 302.2121.
3.1.26. (Rac-3S,4R,E)- 4-(benzyloxymethyl)-3-propyl-3,4,7,8,9,10-hexahydroazecin-2(1H)-one (18’)
Pentyryl amide 18 (44 mg, 0.14 mmol) was converted to macrolactam 18’ as an amorphous solid using Procedure A (29 mg, 66%). IR (KBr) νmax 3304, 2927, 2858, 1645, 1543, 1453 cm−1; 1H-NMR (CD3OD, 300 MHz) δ 7.23–7.15 (m, 5H), 5.42–5.22 (m, 2H), 4.43 (d, 1H, J = 12.0 Hz), 4.33 (d, 2H, J = 12.0 Hz), 3.47–3.43 (m, 2H), 2.63 (m, 1H), 2.30–2.04 (m, 2H), 1.78–0.99 (m, 10H), 0.77 (t, 3H, J = 6.9 Hz); 13C-NMR (CDCl3, 100 MHz) δ 174.8, 138.3, 133.4, 130.3, 128.3, 127.6 (2C), 73.1, 69.9, 67.6, 51.5, 47.2, 40.2, 33.0, 30.4, 29.7, 21.4, 14.1; LRMS (FAB) m/z 316 (M + H+); HRMS (FAB) calcd for C20H30NO2 (M + H+): 316.2277, found 316.2272.
3.1.27. 2-Ethyl-N-(4-methoxybenzyl)-3-methylpent-4-enamide (21’)
To a solution of crotyl amide 21 (44 mg, 0.18 mmol) in toluene (2 mL), iPrMgCl (2.0 M in THF, 0.18 mL, 0.36 mmol) was added at reflux condition. After stirring at same temperature for 12 h, additional iPrMgCl (2.0 M in THF, 0.18 mL, 0.36 mmol) was added at same temperature. Reaction mixture was refluxed 5 h and cooled down to room temperature and quenched with brine and extracted with EtOAc. Organic layers were dried over MgSO4, filtered, evaporated, and purified by column chromatography on silica gel (EtOAc:n-hexane = 1:3) to afford lactam 21’ (18 mg, 36%) as an amorphous solid. IR (KBr) νmax 3305, 2965, 1646, 1514, 1459 cm−1; 1H-NMR (CDCl3, 500 MHz) δ 7.18 (d, 2H, J = 7.6 Hz), 6.83 (d, 2H, J = 7.6 Hz), 5.79–5.72 (m, 1H), 5.59 (bs, 1H), 5.00–4.92 (m, 2H), 4.32 (d, 2H, J = 14 Hz), 3.77 (s, 3H), 2.40–2.34 (m, 1H), 1.83–1.77 (m, 1H), 1.71–1.48 (m, 3H), 1.11 (d, 3H, J = 5.5 Hz), 0.87 (t, 3H, J = 5.9 Hz); 13C-NMR (CDCl3, 125 MHz) δ 174.1, 158.9, 141.6, 130.5, 129.2, 114.2, 114.0, 55.3, 55.2, 42.8, 40.1, 23.0, 17.5, 12.2; LRMS (FAB) m/z 262 (M + H+); HRMS (FAB) calcd for C16H24NO2 (M + H+): 262.1807, found 262.1804.