Convergent Synthesis of Polysubstituted Furans via Catalytic Phosphine Mediated Multicomponent Reactions
Abstract
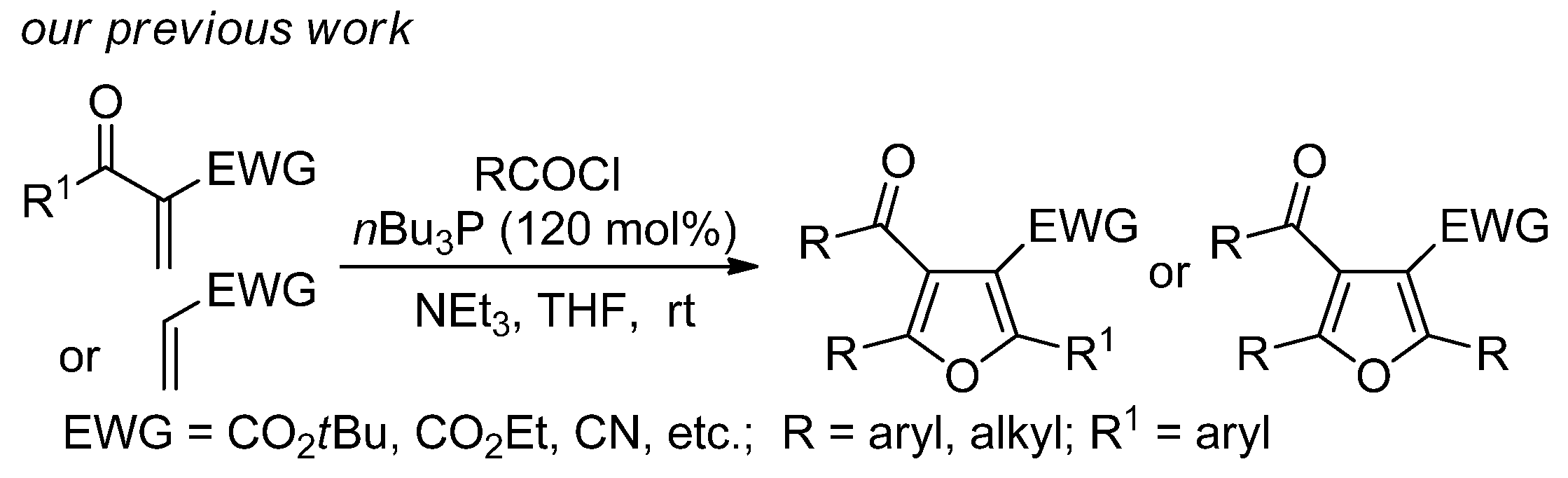
1. Introduction
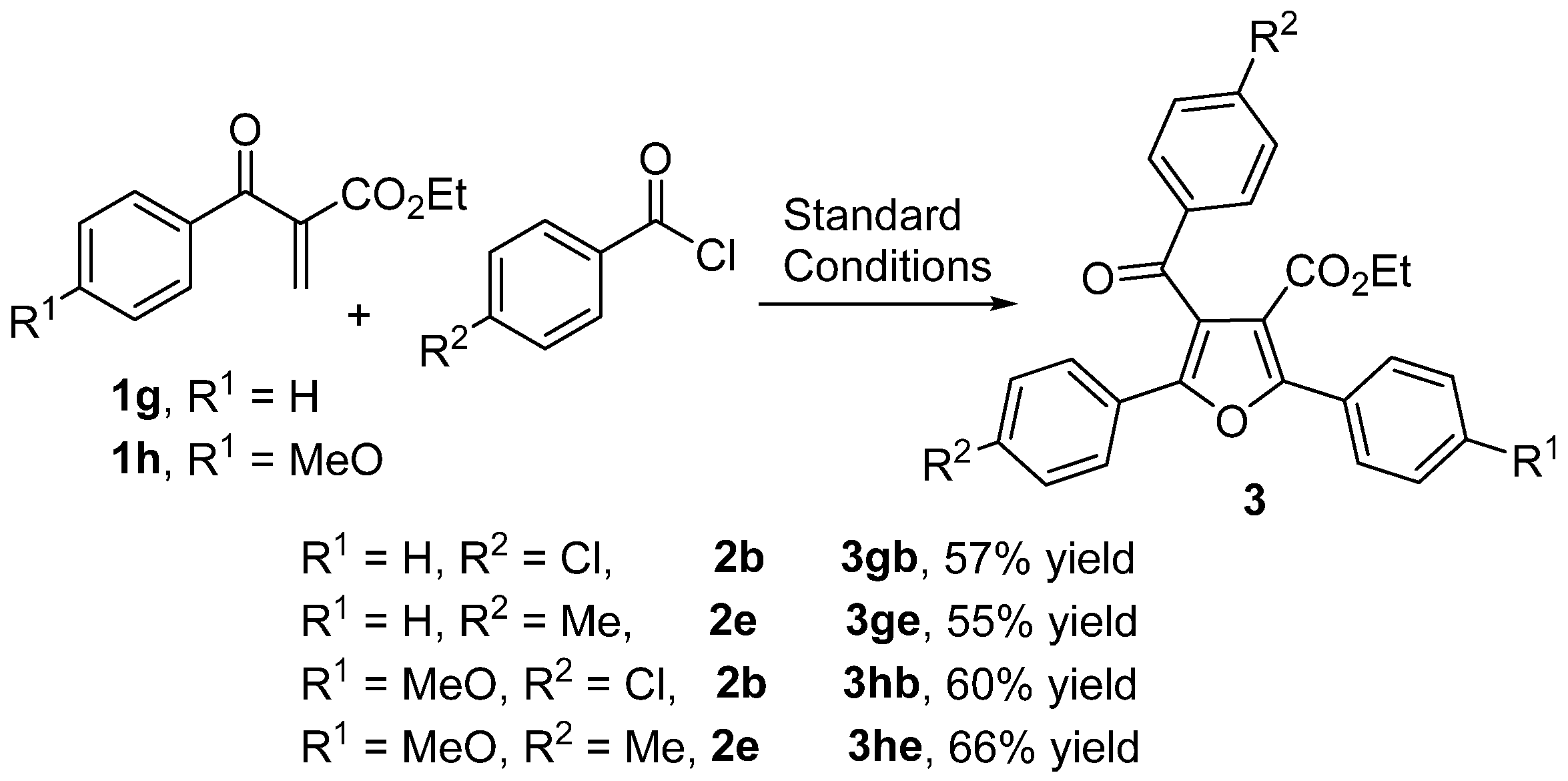
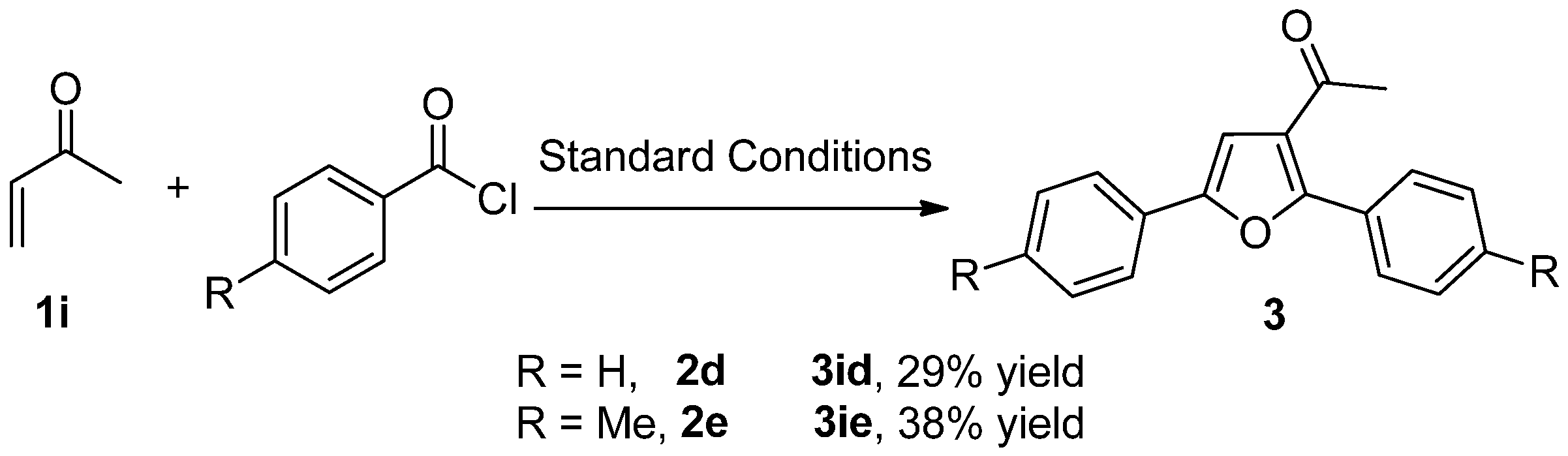
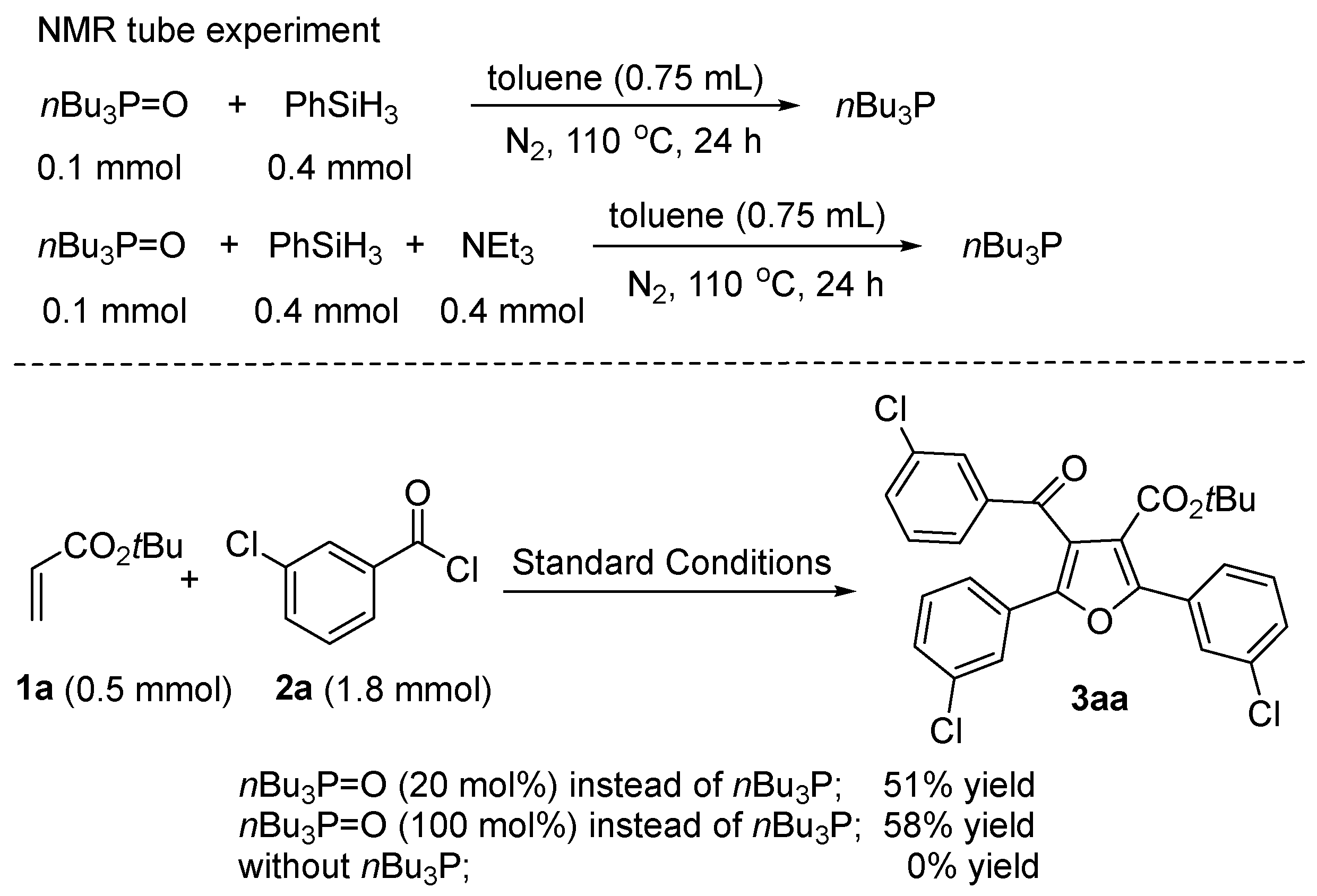
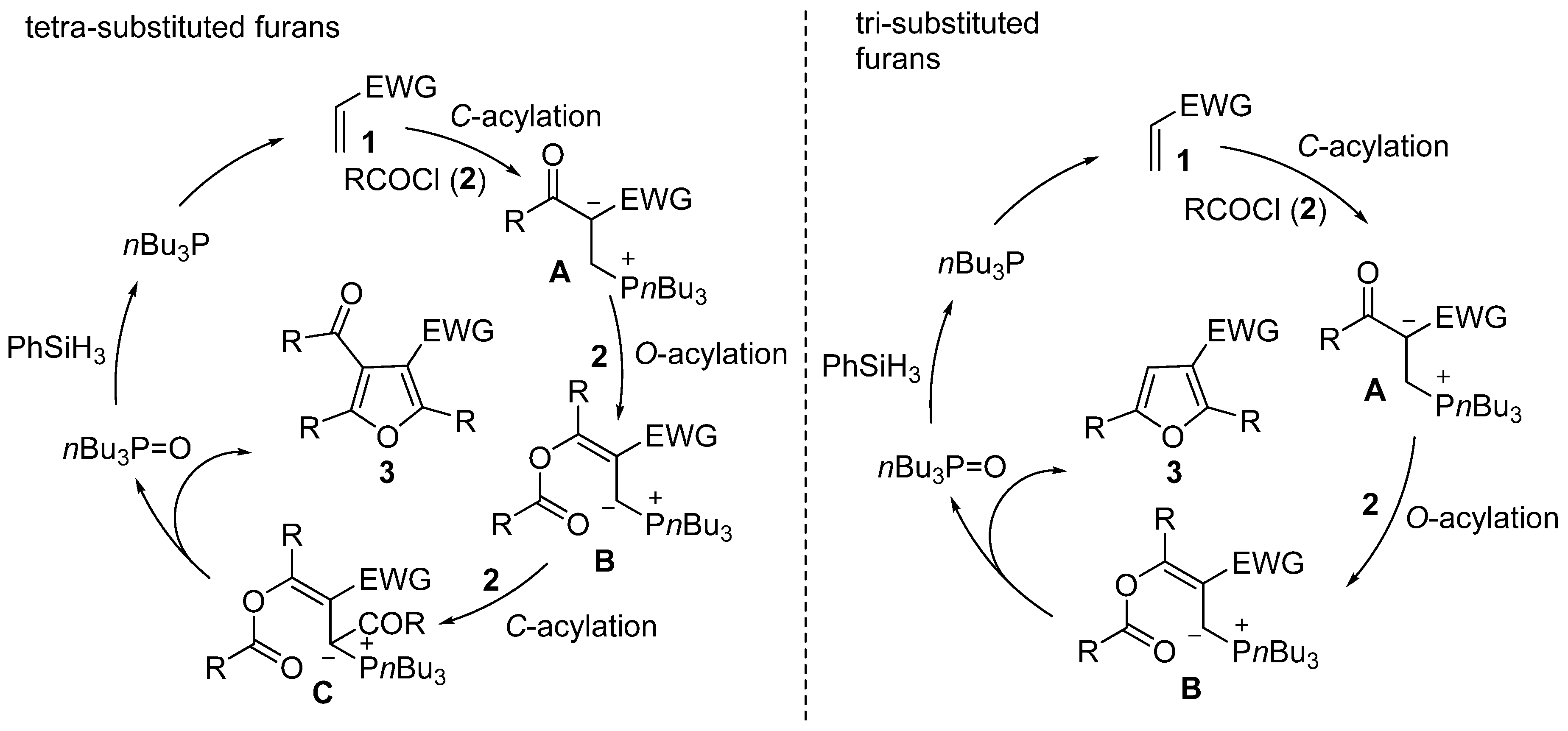
2. Results and Discussion
3. Experimental Section
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Byrne, P.A.; Gilheany, D.G. The modern interpretation of the Wittig reaction mechanism. Chem. Soc. Rev. 2013, 42, 6670–6696. [Google Scholar] [CrossRef]
- Chelucci, G. Synthesis and Metal-Catalyzed Reactions of gem-Dihalovinyl Systems. Chem. Rev. 2012, 112, 1344–1462. [Google Scholar] [CrossRef]
- Perin, G.; Lenardão, E.J.; Jacob, R.G.; Panatieri, R.B. Synthesis of Vinyl Selenides. Chem. Rev. 2009, 109, 1277–1301. [Google Scholar] [CrossRef]
- Flynn, A.B.; Ogilvie, W.W. Stereocontrolled Synthesis of Tetrasubstituted Olefins. Chem. Rev. 2007, 107, 4698–4745. [Google Scholar] [CrossRef]
- Batesky, D.C.; Goldfogel, M.J.; Weix, D.J. Removal of Triphenylphosphine Oxide by Precipitation with Zinc Chloride in Polar Solvents. J. Org. Chem. 2017, 82, 9931–9936. [Google Scholar] [CrossRef]
- Bergbreiter, D.E.; Yang, Y.-C.; Hobbs, C.E. Polyisobutylene-Supported Phosphines as Recyclable and Regenerable Catalysts and Reagents. J. Org. Chem. 2011, 76, 6912–6917. [Google Scholar] [CrossRef]
- Constable, D.J.C.; Dunn, P.J.; Hayler, J.D.; Humphrey, G.R.; Leazer, J.J.L.; Linderman, R.J.; Lorenz, K.; Manley, J.; Pearlman, B.A.; Wells, A.; et al. Key green chemistry research areas-a perspective from pharmaceutical manufacturers. Green Chem. 2007, 9, 411–420. [Google Scholar] [CrossRef]
- Dandapani, S.; Curran, D.P. Separation-Friendly Mitsunobu Reactions: A Microcosm of Recent Developments in Separation Strategies. Chem. Eur. J. 2004, 10, 3130–3138. [Google Scholar] [CrossRef]
- Dutartre, M.; Bayardon, J.; Juge, S. Applications and stereoselective syntheses of P-chirogenic phosphorus compounds. Chem. Soc. Rev. 2016, 45, 5771–5794. [Google Scholar] [CrossRef]
- Wang, Z.; Xu, X.; Kwon, O. Phosphine catalysis of allenes with electrophiles. Chem. Soc. Rev. 2014, 43, 2927–2940. [Google Scholar] [CrossRef]
- Wei, Y.; Shi, M. Applications of Chiral Phosphine-Based Organocatalysts in Catalytic Asymmetric Reactions. Chem. Asian J. 2014, 9, 2720–2734. [Google Scholar] [CrossRef] [PubMed]
- Marinetti, A.; Voituriez, A. Enantioselective Phosphine Organocatalysis. Synlett 2010, 174–194. [Google Scholar] [CrossRef]
- Glueck, D.S. Catalytic asymmetric synthesis of chiral phosphanes. Chem. Eur. J. 2008, 14, 7108–7117. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-M.; Kwong, F.-Y.; Yu, W.-Y.; Chan, A.S.C. Recent advances in developing new axially chiral phosphine ligands for asymmetric catalysis. Coord. Chem. Rev. 2007, 251, 2119–2144. [Google Scholar] [CrossRef]
- Pietrusiewicz, K.M.; Zablocka, M. Preparation of Scalemic P-Chiral Phosphines and Their Derivatives. Chem. Rev. 1994, 94, 1375–1411. [Google Scholar] [CrossRef]
- Rein, T.; Pedersen, T.M. Palladium-catalyzed reaction of olefins with PhI(OAc)2-TBAB system: An efficient and highly selective bisfunctionalization strategy. Synthesis 2002, 2002, 579–594. [Google Scholar] [CrossRef]
- Rein, T.; Reiser, O. Recent advances in asymmetric Wittig-type reactions. Acta Chem. Scand. 1996, 50, 369–379. [Google Scholar] [CrossRef]
- Zhang, K.; Lu, L.-Q.; Yao, S.; Chen, J.-R.; Shi, D.-Q.; Xiao, W.-J. Enantioconvergent Copper Catalysis: In Situ Generation of the Chiral Phosphorus Ylide and Its Wittig Reactions. J. Am. Chem. Soc. 2017, 139, 12847–12854. [Google Scholar] [CrossRef]
- Wong, G.W.; Landis, C.R. Iterative Asymmetric Hydroformylation/Wittig Olefination Sequence. Angew. Chem. Int. Ed. 2013, 52, 1564–1567. [Google Scholar] [CrossRef]
- Enders, D.; Grossmann, A.; Gieraths, B.; Duezdemir, M.; Merkens, C. Organocatalytic One-Pot Asymmetric Synthesis of 4H,5H-Pyrano[2,3-c]pyrazoles. Org. Lett. 2012, 14, 4254–4257. [Google Scholar] [CrossRef]
- Lin, A.; Wang, J.; Mao, H.; Ge, H.; Tan, R.; Zhu, C.; Cheng, Y. Organocatalytic Asymmetric Michael-Type/Wittig Reaction of Phosphorus Ylides: Synthesis of Chiral α-Methylene-δ-Ketoesters. Org. Lett. 2011, 13, 4176–4179. [Google Scholar] [CrossRef] [PubMed]
- Gramigna, L.; Duce, S.; Filippini, G.; Fochi, M.; Franchini, M.C.; Bernardi, L. Organocatalytic asymmetric Wittig reactions: Generation of enantioenriched axially chiral olefins breaking a symmetry plane. Synlett 2011, 2745–2749. [Google Scholar] [CrossRef]
- Ye, L.-W.; Wang, S.-B.; Wang, Q.-G.; Sun, X.-L.; Tang, Y.; Zhou, Y.-G. Asymmetric tandem Michael addition-ylide olefination reaction for the synthesis of optically active cyclohexa-1,3-diene derivatives. Chem. Commun. 2009, 3092–3094. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-k.; Ma, C.; Jiang, K.; Liu, T.-Y.; Chen, Y.-C. Asymmetric Tandem Michael Addition-Wittig Reaction to Cyclohexenone Annulation. Org. Lett. 2009, 11, 2848–2851. [Google Scholar] [CrossRef] [PubMed]
- Hong, B.-C.; Jan, R.-H.; Tsai, C.-W.; Nimje, R.Y.; Liao, J.-H.; Lee, G.-H. Organocatalytic Enantioselective Cascade Michael-Michael-Wittig Reactions of Phosphorus Ylides: One-Pot Synthesis of the all-cis Trisubstituted Cyclohexenecarboxylates via the [1 + 2 + 3] Annulation. Org. Lett. 2009, 11, 5246–5249. [Google Scholar] [CrossRef] [PubMed]
- Voituriez, A.; Saleh, N. From phosphine-promoted to phosphine-catalyzed reactions by in situ phosphine oxide reduction. Tetrahedron Lett. 2016, 57, 4443–4451. [Google Scholar] [CrossRef]
- Xu, S.; Tang, Y. Catalytic approaches to stoichiometric phosphine-mediated organic reactions. Lett. Org. Chem. 2014, 11, 524–533. [Google Scholar] [CrossRef]
- Van Kalkeren, H.A.; van Delft, F.L.; Rutjes, F.P.J.T. Organophosphorus Catalysis to Bypass Phosphine Oxide Waste. ChemSusChem 2013, 6, 1615–1624. [Google Scholar] [CrossRef]
- Zhao, Q.; Curran, D.P.; Malacria, M.; Fensterbank, L.; Goddard, J.-P.; Lacôte, E. N-Heterocyclic Carbene-Catalyzed Hydrosilylation of Styryl and Propargylic Alcohols with Dihydrosilanes. Chem. Eur. J. 2011, 17, 9911–9914. [Google Scholar] [CrossRef]
- Quin, L.D.; Caster, K.C.; Kisalus, J.C.; Mesch, K.A. Bridged ring systems containing phosphorus: Structural influences on the stereochemistry of silane reductions of P-oxides and on carbon-13 and phosphorus-31 NMR properties of phosphines. J. Am. Chem. Soc. 1984, 106, 7021–7032. [Google Scholar] [CrossRef]
- Busacca, C.A.; Raju, R.; Grinberg, N.; Haddad, N.; James-Jones, P.; Lee, H.; Lorenz, J.C.; Saha, A.; Senanayake, C.H. Reduction of Tertiary Phosphine Oxides with DIBAL-H. J. Org. Chem. 2008, 73, 1524–1531. [Google Scholar] [CrossRef] [PubMed]
- Busacca, C.A.; Lorenz, J.C.; Grinberg, N.; Haddad, N.; Hrapchak, M.; Latli, B.; Lee, H.; Sabila, P.; Saha, A.; Sarvestani, M.; et al. A Superior Method for the Reduction of Secondary Phosphine Oxides. Org. Lett. 2005, 7, 4277–4280. [Google Scholar] [CrossRef] [PubMed]
- Imamoto, T.; Kikuchi, S.-i.; Miura, T.; Wada, Y. Stereospecific Reduction of Phosphine Oxides to Phosphines by the Use of a Methylation Reagent and Lithium Aluminum Hydride. Org. Lett. 2001, 3, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Henson, P.D.; Naumann, K.; Mislow, K. Stereomutation of phosphine oxide by lithium aluminum hydride. J. Amer. Chem. Soc. 1969, 91, 5645–5646. [Google Scholar] [CrossRef]
- Sowa, S.; Stankevic, M.; Flis, A.; Pietrusiewicz, K.M. Reduction of Tertiary Phosphine Oxides by BH3 Assisted by Neighboring Activating Groups. Synthesis 2018, 50, 2106–2118. [Google Scholar]
- Provis-Evans, C.B.; Emanuelsson, E.A.C.; Webster, R.L. Rapid Metal-Free Formation of Free Phosphines from Phosphine Oxides. Adv. Synth. Catal. 2018, 360, 3999–4004. [Google Scholar] [CrossRef]
- Elias, J.S.; Costentin, C.; Nocera, D.G. Direct Electrochemical P(V) to P(III) Reduction of Phosphine Oxide Facilitated by Triaryl Borates. J. Am. Chem. Soc. 2018, 140, 13711–13718. [Google Scholar] [CrossRef]
- Mehta, M.; Garcia de la Arada, I.; Perez, M.; Porwal, D.; Oestreich, M.; Stephan, D.W. Metal-Free Phosphine Oxide Reductions Catalyzed by B(C6F5)3 and Electrophilic Fluorophosphonium Cations. Organometallics 2016, 35, 1030–1035. [Google Scholar] [CrossRef]
- Stepen, A.J.; Bursch, M.; Grimme, S.; Stephan, D.W.; Paradies, J. Electrophilic Phosphonium Cation-Mediated Phosphane Oxide Reduction Using Oxalyl Chloride and Hydrogen. Angew. Chem. Int. Ed. 2018, 57, 15253–15256. [Google Scholar] [CrossRef]
- Li, P.; Wischert, R.; Metivier, P. Mild Reduction of Phosphine Oxides with Phosphites To Access Phosphines. Angew. Chem. Int. Ed. 2017, 56, 15989–15992. [Google Scholar] [CrossRef]
- Kuroboshi, M.; Kita, T.; Aono, A.; Katagiri, T.; Kikuchi, S.; Yamane, S.; Kawakubo, H.; Tanaka, H. Reduction of phosphine oxides to the corresponding phosphine derivatives in Mg/Me3SiCl/DMI system. Tetrahedron Lett. 2015, 56, 918–920. [Google Scholar] [CrossRef]
- Pehlivan, L.; Metay, E.; Delbrayelle, D.; Mignani, G.; Lemaire, M. Reduction of phosphine oxides to phosphines with the InBr3/TMDS system. Tetrahedron 2012, 68, 3151–3155. [Google Scholar] [CrossRef]
- Rajendran, K.V.; Gilheany, D.G. Simple unprecedented conversion of phosphine oxides and sulfides to phosphine boranes using sodium borohydride. Chem. Commun. 2012, 48, 817–819. [Google Scholar] [CrossRef] [PubMed]
- Kawakubo, H.; Kuroboshi, M.; Yano, T.; Kobayashi, K.; Kamenoue, S.; Akagi, T.; Tanaka, H. Electroreduction of triphenylphosphine oxide to triphenylphosphine in the presence of chlorotrimethylsilane. Synthesis 2011, 4091–4098. [Google Scholar]
- Yano, T.; Kuroboshi, M.; Tanaka, H. Electroreduction of triphenylphosphine dichloride and the efficient one-pot reductive conversion of phosphine oxide to triphenylphosphine. Tetrahedron Lett. 2010, 51, 698–701. [Google Scholar] [CrossRef]
- Longwitz, L.; Werner, T. Recent advances in catalytic Wittig-type reactions based on P(III)/P(V) redox cycling. Pure Appl. Chem. 2019, 91, 95–102. [Google Scholar] [CrossRef]
- Podyacheva, E.; Kuchuk, E.; Chusov, D. Reduction of phosphine oxides to phosphines. Tetrahedron Lett. 2019, 60, 575–582. [Google Scholar] [CrossRef]
- Karanam, P.; Reddy, G.M.; Lin, W. Strategic Exploitation of the Wittig Reaction: Facile Synthesis of Heteroaromatics and Multifunctional Olefins. Synlett 2018, 29, 2608–2622. [Google Scholar]
- Kovacs, T.; Keglevich, G. The Reduction of Tertiary Phosphine Oxides by Silanes. Curr. Org. Chem. 2017, 21, 569–585. [Google Scholar] [CrossRef]
- Kovacs, T.; Keglevich, G. The deoxygenation of phosphine oxides under green chemical conditions. Phosphorus Sulfur Silicon Relat. Elem. 2016, 191, 359–366. [Google Scholar] [CrossRef]
- Lao, Z.; Toy, P.H. Catalytic Wittig and aza-Wittig reactions. Beilstein J. Org. Chem. 2016, 12, 2577–2587. [Google Scholar] [CrossRef] [PubMed]
- Herault, D.; Nguyen, D.H.; Nuel, D.; Buono, G. Reduction of secondary and tertiary phosphine oxides to phosphines. Chem. Soc. Rev. 2015, 44, 2508–2528. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, C.J.; Tellez, J.L.; Nixon, Z.S.; Kang, L.J.; Carter, A.L.; Kunkel, S.R.; Przeworski, K.C.; Chass, G.A. Recycling the Waste: The Development of a Catalytic Wittig Reaction. Angew. Chem. Int. Ed. 2009, 48, 6836–6839. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, C.J.; Lavigne, F.; Coyle, E.E.; Holohan, A.J.; Doonan, B.J. Breaking the Ring through a Room Temperature Catalytic Wittig Reaction. Chem. Eur. J. 2013, 19, 5854–5858. [Google Scholar]
- O’Brien, C.J.; Nixon, Z.S.; Holohan, A.J.; Kunkel, S.R.; Tellez, J.L.; Doonan, B.J.; Coyle, E.E.; Lavigne, F.; Kang, L.J.; Przeworski, K.C. Part I: The Development of the Catalytic Wittig Reaction. Chem. Eur. J. 2013, 19, 15281–15289. [Google Scholar] [CrossRef]
- Coyle, E.E.; Doonan, B.J.; Holohan, A.J.; Walsh, K.A.; Lavigne, F.; Krenske, E.H.; O’Brien, C.J. Catalytic Wittig Reactions of Semi- and Nonstabilized Ylides Enabled by Ylide Tuning. Angew. Chem. Int. Ed. 2014, 53, 12907–12911. [Google Scholar] [CrossRef]
- Li, Y.; Das, S.; Zhou, S.; Junge, K.; Beller, M. General and Selective Copper-Catalyzed Reduction of Tertiary and Secondary Phosphine Oxides: Convenient Synthesis of Phosphines. J. Am. Chem. Soc. 2012, 134, 9727–9732. [Google Scholar] [CrossRef]
- Li, Y.; Lu, L.-Q.; Das, S.; Pisiewicz, S.; Junge, K.; Beller, M. Highly Chemoselective Metal-Free Reduction of Phosphine Oxides to Phosphines. J. Am. Chem. Soc. 2012, 134, 18325–18329. [Google Scholar] [CrossRef]
- Cai, L.; Zhang, K.; Chen, S.; Lepage, R.J.; Houk, K.N.; Krenske, E.H.; Kwon, O. Catalytic Asymmetric Staudinger-aza-Wittig Reaction for the Synthesis of Heterocyclic Amines. J. Am. Chem. Soc. 2019, 141, 9537–9542. [Google Scholar] [CrossRef]
- Longwitz, L.; Spannenberg, A.; Werner, T. Phosphetane Oxides as Redox Cycling Catalysts in the Catalytic Wittig Reaction at Room Temperature. ACS Catal. 2019, 9, 9237–9244. [Google Scholar] [CrossRef]
- Lorton, C.; Castanheiro, T.; Voituriez, A. Catalytic and Asymmetric Process via PIII/PV=O Redox Cycling: Access to (Trifluoromethyl)cyclobutenes via a Michael Addition/Wittig Olefination Reaction. J. Am. Chem. Soc. 2019, 141, 10142–10147. [Google Scholar] [CrossRef]
- Fianchini, M.; Maseras, F. DFT characterization of the mechanism for Staudinger/aza-Wittig tandem organocatalysis. Tetrahedron 2019, 75, 1852–1859. [Google Scholar] [CrossRef]
- Han, X.; Saleh, N.; Retailleau, P.; Voituriez, A. Phosphine-Catalyzed Reaction between 2-Aminobenzaldehydes and Dialkyl Acetylenedicarboxylates: Synthesis of 1,2-Dihydroquinoline Derivatives and Toward the Development of an Olefination Reaction. Org. Lett. 2018, 20, 4584–4588. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Sun, M.; Ding, M.-W. Catalytic Intramolecular Wittig Reaction Based on a Phosphine/Phosphine Oxide Catalytic Cycle for the Synthesis of Heterocycles. Eur. J. Org. Chem. 2017, 2568–2578. [Google Scholar] [CrossRef]
- Wang, L.; Xie, Y.-B.; Huang, N.-Y.; Yan, J.-Y.; Hu, W.-M.; Liu, M.-G.; Ding, M.-W. Catalytic aza-Wittig Reaction of Acid Anhydride for the Synthesis of 4H-Benzo[d][1,3]oxazin-4-ones and 4-Benzylidene-2-aryloxazol-5(4H)-ones. ACS Catal. 2016, 6, 4010–4016. [Google Scholar] [CrossRef]
- Saleh, N.; Voituriez, A. Synthesis of 9H-Pyrrolo[1,2-a]indole and 3H-Pyrrolizine Derivatives via a Phosphine-Catalyzed Umpolung Addition/Intramolecular Wittig Reaction. J. Org. Chem. 2016, 81, 4371–4377. [Google Scholar] [CrossRef]
- Schirmer, M.-L.; Adomeit, S.; Spannenberg, A.; Werner, T. Novel Base-Free Catalytic Wittig Reaction for the Synthesis of Highly Functionalized Alkenes. Chem. Eur. J. 2016, 22, 2458–2465. [Google Scholar] [CrossRef]
- Lee, C.-J.; Chang, T.-H.; Yu, J.-K.; Madhusudhan Reddy, G.; Hsiao, M.-Y.; Lin, W. Synthesis of Functionalized Furans via Chemoselective Reduction/Wittig Reaction Using Catalytic Triethylamine and Phosphine. Org. Lett. 2016, 18, 3758–3761. [Google Scholar] [CrossRef]
- Schirmer, M.-L.; Adomeit, S.; Werner, T. First Base-Free Catalytic Wittig Reaction. Org. Lett. 2015, 17, 3078–3081. [Google Scholar] [CrossRef]
- Tsai, Y.-L.; Lin, W. Synthesis of Multifunctional Alkenes from Substituted Acrylates and Aldehydes via Phosphine-Catalyzed Wittig Reaction. Asian J. Org. Chem. 2015, 4, 1040–1043. [Google Scholar] [CrossRef]
- Rommel, S.; Belger, C.; Begouin, J.-M.; Plietker, B. Dual [Fe+Phosphine] Catalysis: Application in Catalytic Wittig Olefination. Chemcatchem 2015, 7, 1292–1301. [Google Scholar] [CrossRef]
- Werner, T.; Hoffmann, M.; Deshmukh, S. First Enantioselective Catalytic Wittig Reaction. Eur. J. Org. Chem. 2014, 6630–6633. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Y.; Chen, M.; Ding, M.-W. Reversible P(III)/P(V) Redox: Catalytic Aza-Wittig Reaction for the Synthesis of 4(3H)-Quinazolinones and the Natural Product Vasicinone. Adv. Synth. Catal. 2014, 356, 1098–1104. [Google Scholar] [CrossRef]
- Werner, T.; Hoffmann, M.; Deshmukh, S. First Microwave-Assisted Catalytic Wittig Reaction. Eur. J. Org. Chem. 2014, 6873–6876. [Google Scholar] [CrossRef]
- Van Kalkeren, H.A.; te Grotenhuis, C.; Haasjes, F.S.; Hommersom, C.A.; Rutjes, F.P.J.T.; van Delft, F.L. Catalytic Staudinger/aza-Wittig sequence by in situ phosphane oxide reduction. Eur. J. Org. Chem. 2013, 7059–7066. [Google Scholar] [CrossRef]
- Beddoe, R.H.; Sneddon, H.F.; Denton, R.M. The catalytic Mitsunobu reaction: A critical analysis of the current state-of-the-art. Org. Biomol. Chem. 2018, 16, 7774–7781. [Google Scholar] [CrossRef]
- Hirose, D.; Gazvoda, M.; Košmrlj, J.; Taniguchi, T. The “Fully Catalytic System” in Mitsunobu Reaction Has Not Been Realized Yet. Org. Lett. 2016, 18, 4036–4039. [Google Scholar] [CrossRef]
- Buonomo, J.A.; Aldrich, C.C. Mitsunobu Reactions Catalytic in Phosphine and a Fully Catalytic System. Angew. Chem. Int. Ed. 2015, 54, 13041–13044. [Google Scholar] [CrossRef]
- Lenstra, D.C.; Wolf, J.J.; Mecinovic, J. Catalytic Staudinger Reduction at Room Temperature. J. Org. Chem. 2019, 84, 6536–6545. [Google Scholar] [CrossRef]
- Zhang, K.; Cai, L.; Yang, Z.; Houk, K.N.; Kwon, O. Bridged [2.2.1] bicyclic phosphine oxide facilitates catalytic γ-umpolung addition-Wittig olefination. Chem. Sci. 2018, 9, 1867–1872. [Google Scholar] [CrossRef]
- Lenstra, D.C.; Lenting, P.E.; Mecinovic, J. Sustainable organophosphorus-catalysed Staudinger reduction. Greem Chem. 2018, 20, 4418–4422. [Google Scholar] [CrossRef]
- Andrews, K.G.; Denton, R.M. A more critical role for silicon in the catalytic Staudinger amidation: Silanes as non-innocent reductants. Chem. Commun. 2017, 53, 7982–7985. [Google Scholar] [CrossRef] [PubMed]
- Saleh, N.; Blanchard, F.; Voituriez, A. Synthesis of Nitrogen-Containing Heterocycles and Cyclopentenone Derivatives via Phosphine-Catalyzed Michael Addition/Intramolecular Wittig Reaction. Adv. Synth. Catal. 2017, 359, 2304–2315. [Google Scholar] [CrossRef]
- Van Kalkeren, H.A.; Bruins, J.J.; Rutjes, F.P.J.T.; van Delft, F.L. Organophosphorus-catalyzed Staudinger reduction. Adv. Synth. Catal. 2012, 354, 1417–1421. [Google Scholar] [CrossRef]
- Longwitz, L.; Jopp, S.; Werner, T. Organocatalytic Chlorination of Alcohols by P(III)/P(V) Redox Cycling. J. Org. Chem. 2019, 84, 7863–7870. [Google Scholar] [CrossRef] [PubMed]
- Van Kalkeren, H.A.; Leenders, S.; Hommersom, C.R.A.; Rutjes, F.; van Delft, F.L. In situ phosphine oxide reduction: A catalytic Appel reaction. Chem. Eur. J. 2011, 17, 11290–11295. [Google Scholar] [CrossRef]
- Nykaza, T.V.; Ramirez, A.; Harrison, T.S.; Luzung, M.R.; Radosevich, A.T. Biphilic Organophosphorus-Catalyzed Intramolecular Csp2-H Amination: Evidence for a Nitrenoid in Catalytic Cadogan Cyclizations. J. Am. Chem. Soc. 2018, 140, 3103–3113. [Google Scholar] [CrossRef]
- White, P.B.; Rijpkema, S.J.; Bunschoten, R.P.; Mecinovic, J. Mechanistic Insight into the Catalytic Staudinger Ligation. Org. Lett. 2019, 21, 1011–1014. [Google Scholar]
- Nykaza, T.V.; Cooper, J.C.; Li, G.; Mahieu, N.; Ramirez, A.; Luzung, M.R.; Radosevich, A.T. Intermolecular Reductive C-N Cross Coupling of Nitroarenes and Boronic Acids by PIII/PV=O Catalysis. J. Am. Chem. Soc. 2018, 140, 15200–15205. [Google Scholar] [CrossRef]
- Hamstra, D.F.J.; Lenstra, D.C.; Koenders, T.J.; Rutjes, F.P.J.T.; Mecinovic, J. Poly(methylhydrosiloxane) as a green reducing agent in organophosphorus-catalyzed amide bond formation. Org. Biomol. Chem. 2017, 15, 6426–6432. [Google Scholar] [CrossRef]
- Fourmy, K.; Voituriez, A. Catalytic Cyclization Reactions of Huisgen Zwitterion with α-Ketoesters by in Situ Chemoselective Phosphine Oxide Reduction. Org. Lett. 2015, 17, 1537–1540. [Google Scholar] [CrossRef]
- Zhao, W.; Yan, P.K.; Radosevich, A.T. A Phosphetane Catalyzes Deoxygenative Condensation of α-Keto Esters and Carboxylic Acids via PIII/PV=O Redox Cycling. J. Am. Chem. Soc. 2015, 137, 616–619. [Google Scholar] [CrossRef] [PubMed]
- Kosal, A.D.; Wilson, E.E.; Ashfeld, B.L. Phosphine-Based Redox Catalysis in the Direct Traceless Staudinger Ligation of Carboxylic Acids and Azides. Angew. Chem. Int. Ed. 2012, 51, 12036–12040. [Google Scholar] [CrossRef] [PubMed]
- Rotstein, B.H.; Zaretsky, S.; Rai, V.; Yudin, A.K. Small Heterocycles in Multicomponent Reactions. Chem. Rev. 2014, 114, 8323–8359. [Google Scholar] [CrossRef] [PubMed]
- Estevez, V.; Villacampa, M.; Menendez, J.C. Recent advances in the synthesis of pyrroles by multicomponent reactions. Chem. Soc. Rev. 2014, 43, 4633–4657. [Google Scholar] [CrossRef]
- Cioc, R.C.; Ruijter, E.; Orru, R.V.A. Multicomponent reactions: Advanced tools for sustainable organic synthesis. Green Chem. 2014, 16, 2958–2975. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, R.; He, Z.-R.; He, Z. Phosphane-Mediated Domino Synthesis of Tetrasubstituted Furans from Simple Terminal Activated Olefins. Eur. J. Org. Chem. 2012, 6033–6041. [Google Scholar] [CrossRef]
- Lawrence, N.J.; Crump, J.P.; McGown, A.T.; Hadfield, J.A. Reaction of Baylis–Hillman products with Swern and Dess–Martin oxidants. Tetrahedron Lett. 2001, 42, 3939–3941. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 3 are not available from the authors. |
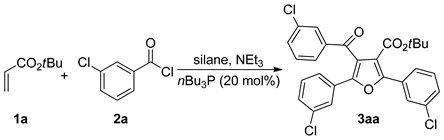
| Entry | Silane (mmol) | Solvent | Temp (°C) | 3 (%) b |
|---|---|---|---|---|
| 1 | Ph2SiH2 (0.6) | THF | rt | 11 |
| 2 | Ph2SiH2 (0.6) | THF | 60 | 12 |
| 3 | PhSiH3 (0.6) | Toluene | rt | trace |
| 4 | PhSiH3 (0.6) | Toluene | 110 | 74 |
| 5 | Ph2SiH2 (0.6) | Toluene | 110 | 42 |
| 6 | (MeO)3SiH (0.6) | Toluene | 110 | 33 |
| 7 | PMHS (0.034) | Toluene | 110 | 26 |
| 8 | PMHS (0.106) | Toluene | 110 | 30 |
| 9 | Ph3SiH (1.2) | Toluene | 110 | 18 |
| 10 | SiHCl3 (0.6) | Toluene | rt | trace |
| 11 | PhSiH3 (0.25) | Toluene | 110 | 65 |
| 12 | PhSiH3 (0.4) | Toluene | 110 | 84 |
| 13 | PhSiH3 (0.75) | Toluene | 110 | 49 |
| 14 | PhSiH3 (0.4) | Toluene | 80 | 56 |
| 15 | PhSiH3 (0.4) | Dioxane | 110 | 54 |
| 16 | PhSiH3 (0.4) | Xylene | 110 | 60 |
| 17 | PhSiH3 (0.4) | DMF | 110 | 11 |
| 18 | PhSiH3 (0.4) | CH3CN | 80 | trace |
| 19 c | PhSiH3 (0.4) | Toluene | 110 | 48 |

| Entry | EWG in 1 | R in 2 or 2′ | 3 (%) b |
|---|---|---|---|
| 1 | CO2tBu (1a) | 3-ClC6H4 (2a) | 3aa, 84 |
| 2 | CO2tBu (1a) | 4-ClC6H4 (2b) | 3ab, 75 |
| 3 | CO2tBu (1a) | 2-ClC6H4 (2c) | 3ac, 33 |
| 4 | CO2tBu (1a) | Ph(2d) | 3ad, 70 |
| 5 | CO2tBu (1a) | 4-MeC6H4 (2e) | 3ae, 90 |
| 6 | CO2tBu (1a) | 4-NO2C6H4 (2f) | 3af, 37 |
| 7 | CO2tBu (1a) | 2-thienyl (2g) | 3ag, 46 |
| 8 | CO2Me (1b) | 4-ClC6H4 (2b) | 3bb, 81 |
| 9 | CO2Me (1b) | 4-MeC6H4 (2e) | 3be, 84 |
| 10 | CO2Et (1c) | 4-ClC6H4 (2b) | 3cb, 71 |
| 11 | CO2Et (1c) | 4-MeC6H4 (2e) | 3ce, 84 |
| 12 | CO2nBu (1d) | 4-ClC6H4 (2b) | 3db, 85 |
| 13 | CO2nBu (1d) | 4-MeC6H4 (2e) | 3de, 83 |
| 14 | CO2Bn (1e) | Ph (2d) | 3ed, 87 |
| 15 | CN (1f) | 4-ClC6H4 (2b) | 3fb, 38 |
| 16 | CN (1f) | Ph (2d) | 3fd, 57 |
| 17 | CN (1f) | 4-MeC6H4 (2e) | 3fe, 44 |
| 18 | CO2tBu (1a) | Me (2′a) | 3aa’, 27 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, X.; Chen, R.; Han, J.; He, Z. Convergent Synthesis of Polysubstituted Furans via Catalytic Phosphine Mediated Multicomponent Reactions. Molecules 2019, 24, 4595. https://doi.org/10.3390/molecules24244595
Fan X, Chen R, Han J, He Z. Convergent Synthesis of Polysubstituted Furans via Catalytic Phosphine Mediated Multicomponent Reactions. Molecules. 2019; 24(24):4595. https://doi.org/10.3390/molecules24244595
Chicago/Turabian StyleFan, Xia, Rongshun Chen, Jie Han, and Zhengjie He. 2019. "Convergent Synthesis of Polysubstituted Furans via Catalytic Phosphine Mediated Multicomponent Reactions" Molecules 24, no. 24: 4595. https://doi.org/10.3390/molecules24244595
APA StyleFan, X., Chen, R., Han, J., & He, Z. (2019). Convergent Synthesis of Polysubstituted Furans via Catalytic Phosphine Mediated Multicomponent Reactions. Molecules, 24(24), 4595. https://doi.org/10.3390/molecules24244595






