A New Valuable Synthesis of Polyfunctionalized Furans Starting from β-Nitroenones and Active Methylene Compounds
Abstract
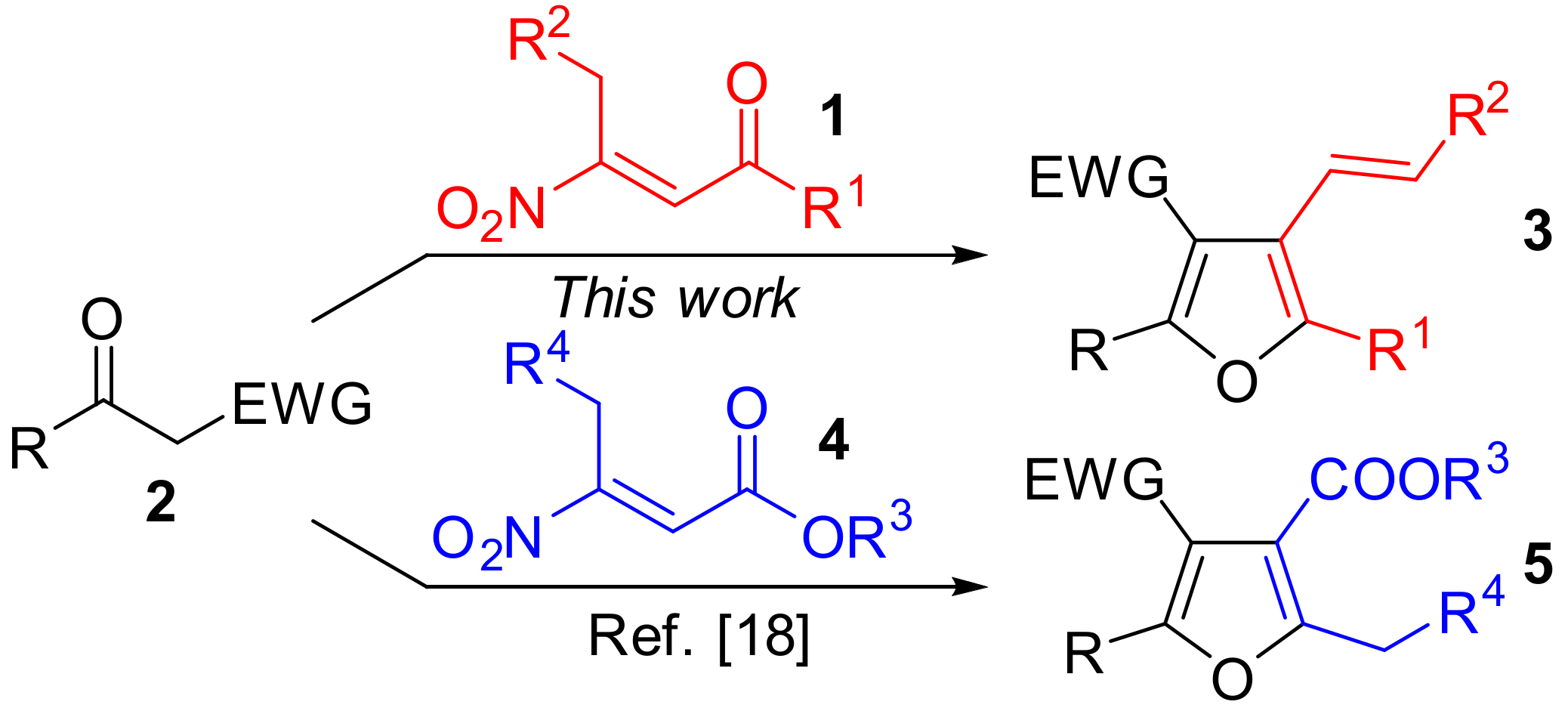
:1. Introduction
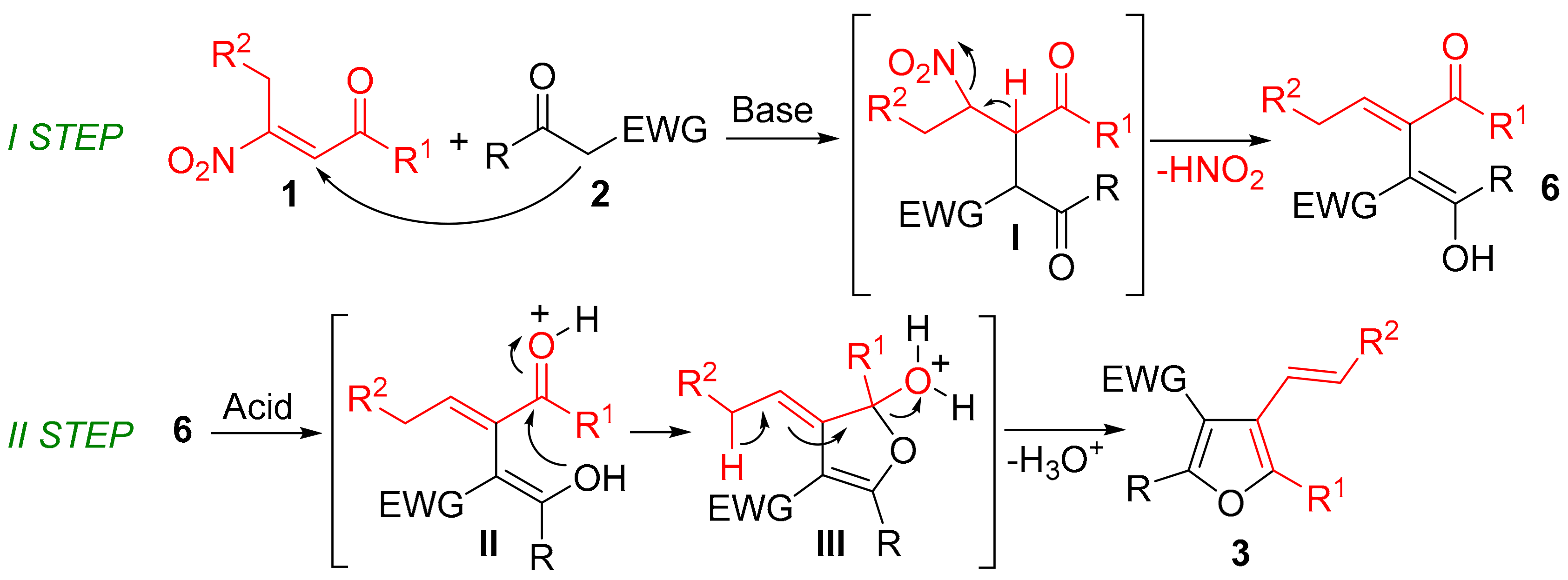
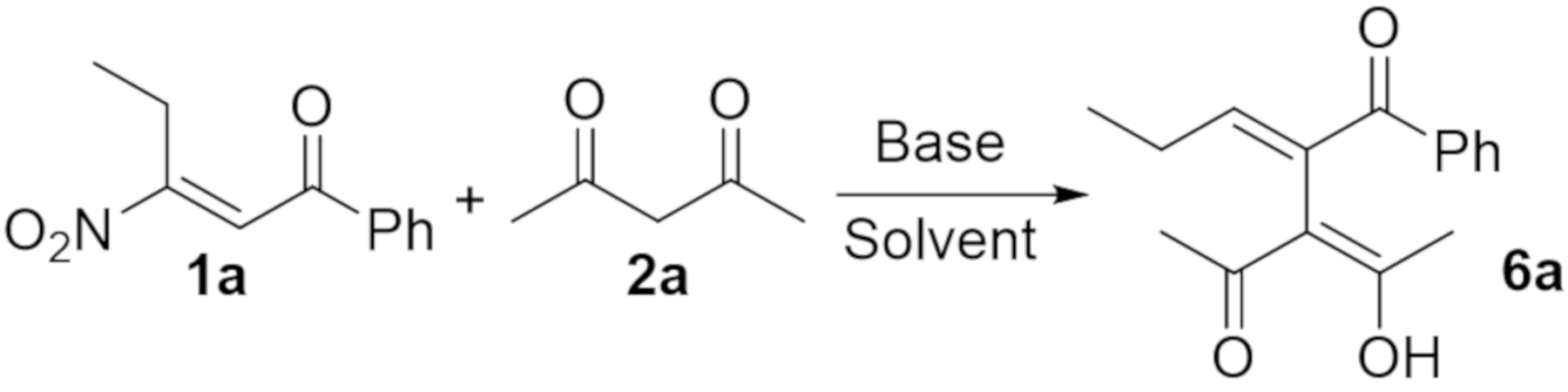
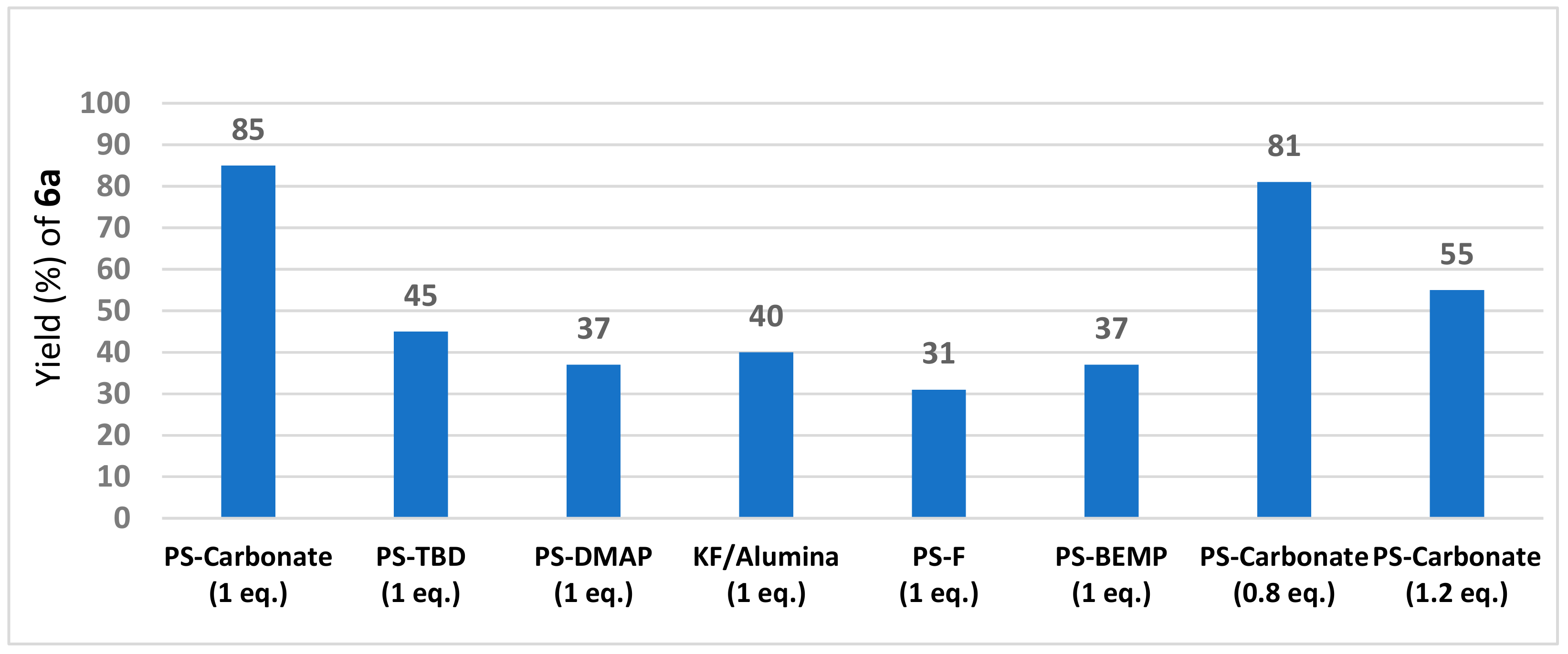
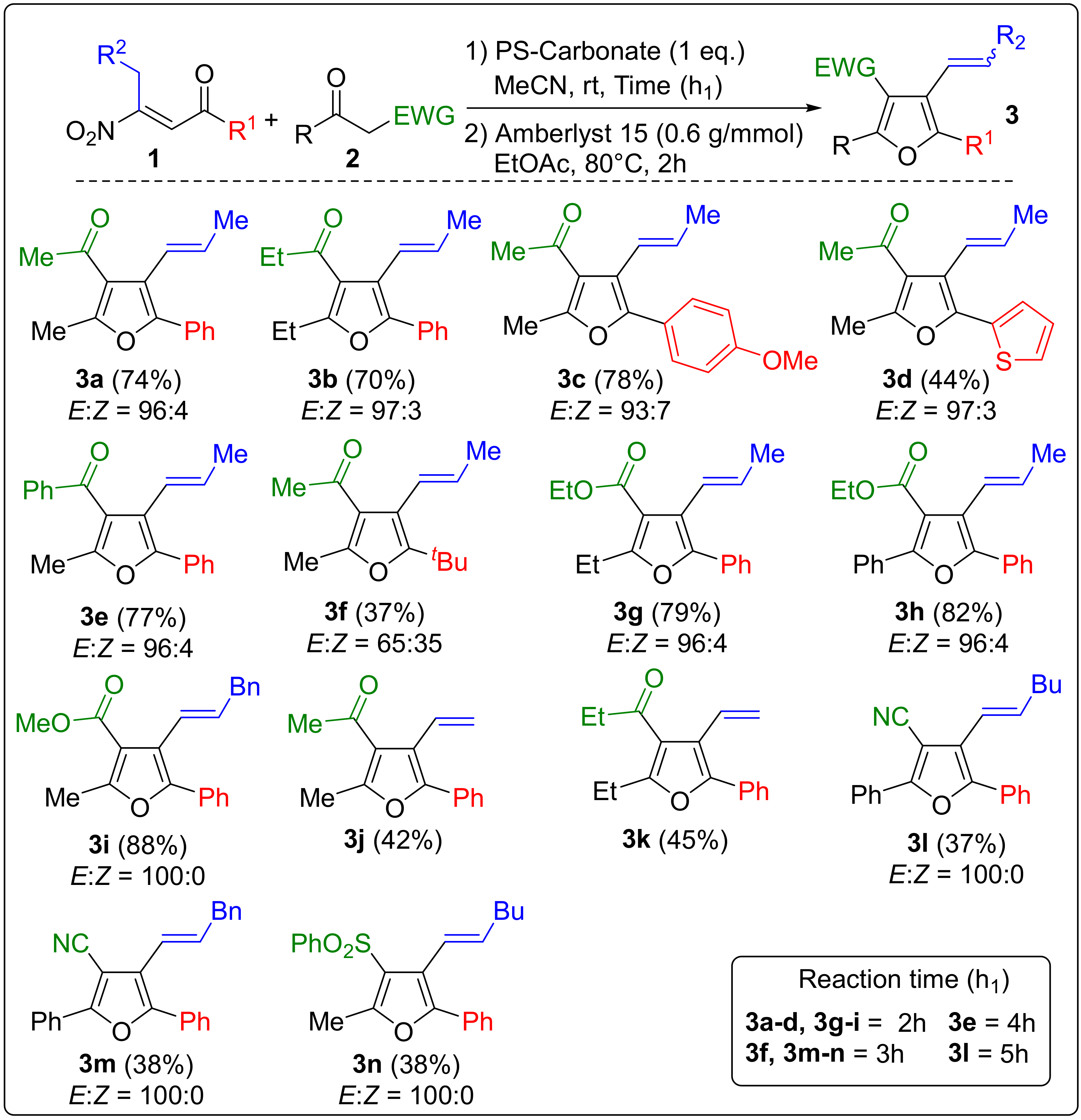
2. Results and Discussion
3. Conclusions
4. Materials and Methods
4.1. General Section
4.2. Chemistry Section
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PS-carbonate | carbonate on polymer support (Sigma-Aldrich code: 21850: loading: 3.5 mmol/g) |
| PS-DMAP | 4-(dimethylamino)pyridine, polymer-bound (Sigma-Aldrich code: 39410, loading: 3 mmol/g) |
| PS-BEMP | 2-tert-butylimino-2-diethylamino-1,3-dimethylperhydro-1,3,2-diazaphosphorine, polymer-bound (Sigma-Aldrich code: 20026, loading: 2.2 mmol/g) |
| PS-TBD | 1,5,7-Triazabicyclo[4.4.0]dec-5-ene bound to polystyrene (Sigma-Aldrich code: 01961, loading: 3.0 mmol/g) |
| PS-F | fluoride on polymer support (Sigma-Aldrich code: 47060, loading: 3.0 mmol/g) |
| KF/alumina | potassium fluoride on aluminum oxide (Sigma-Aldrich code: 60244, loading: 5.5 mmol/g) |
References
- Lipshutz, B.H. Five-membered heteroaromatic rings as intermediates in organic synthesis. Chem. Rev. 1986, 865, 795–819. [Google Scholar] [CrossRef]
- Merritt, A.T.; Ley, S.V. Clerodane diterpenoids. Nat. Prod. Rep. 1992, 9, 243–287. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, D.S.; Rodriguez, A.L.; Carlson, K.E.; Sun, J.; Katzenellenbogen, B.S.; Katzenellenbogen, J.A. Synthesis and biological evaluation of a novel series of furans: ligands selective for estrogen receptor α. J. Med. Chem. 2001, 44, 3838–3848. [Google Scholar] [CrossRef] [PubMed]
- Gandini, A.; Lacerda, T.L.; Carvalho, A.J.F.; Trovatti, E. Progress of polymers from renewable resources: Furans, vegetable oils, and polysaccharides. Chem. Rev. 2016, 116, 1637–1669. [Google Scholar] [CrossRef] [PubMed]
- Dhiman, S.; Ramasastry, S.S.V. Taming furfuryl cations for the synthesis of privileged structures and novel scaffolds. Org. Biomol. Chem. 2013, 11, 4299–4303. [Google Scholar] [CrossRef] [PubMed]
- Yet, L. Privileged Structures in Drug Discovery: Medicinal Chemistry and Synthesis, 1st ed.; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2018. [Google Scholar] [CrossRef]
- Hou, X.L.; Cheung, H.Y.; Hon, T.Y.; Kwan, P.L.; Lo, T.H.; Tong, S.Y.; Wong, H.N.C. Regioselective syntheses of substituted furans. Tetrahedron 1998, 51, 1955–2020. [Google Scholar] [CrossRef]
- Melzig, L.; Rauhut, C.B.; Knochel, P. 2,3-Functionalization of furans, benzofurans and thiophenes via magnesiation and sulfoxide-magnesium exchange. Chem. Commun. 2009, 3536–3538. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bach, T.; Krüger, L. The preparation of 2,3,5-tri- and 2,3-disubstituted furans by regioselective palladium(0)-catalyzed coupling reactions: Application to the syntheses of rosefuran and the F5 furan fatty acid. Eur. J. Org. Chem. 1999, 2045–2057. [Google Scholar] [CrossRef]
- Moran, W.J.; Rodríguez, A. Metal-catalyzed furan synthesis. A review. Org. Prep. Proced. Int. 2012, 44, 103–130. [Google Scholar] [CrossRef] [Green Version]
- Palmieri, A.; Gabrielli, S.; Ballini, R. Efficient two-step sequence for the synthesis of 2,5-disubstituted furanderivatives from functionalized nitroalkanes: Successive AmberlystA21- and Amberlyst 15-catalyzed processes. Chem. Commun. 2010, 46, 6165–6167. [Google Scholar] [CrossRef] [PubMed]
- Minkler, S.R.K.; Isley, N.A.; Lippincott, D.J.; Krause, N.; Lipshutz, B.H. Leveraging the micellar effect: Gold-catalyzed dehydrative cyclizations in water at room temperature. Org. Lett. 2014, 16, 724–726. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Palisse, A.; Kirsch, S.F. Synthesis of furans through silver-catalyzed propargyl–Claisen rearrangement followed by cyclocondensation. Eur. J. Org. Chem. 2014, 32, 7095–7098. [Google Scholar] [CrossRef]
- Sampaolesi, S.; Gabrielli, S.; Ballini, R.; Palmieri, A. Two-Step synthesis of polysubstituted 6-nitroindoles under flow chemical and microwave conditions. Adv. Synth. Catal. 2017, 359, 3407–3413. [Google Scholar] [CrossRef]
- Palmieri, A.; Gabrielli, S.; Parlapiano, M.; Ballini, R. One-pot synthesis of alkyl pyrrole-2-carboxylates starting from β-nitroacrylates and primary amines. RSC Adv. 2015, 5, 4210–4213. [Google Scholar] [CrossRef]
- Chiurchiù, E.; Patehebieke, Y.; Gabrielli, S.; Ballini, R.; Palmieri, A. β-Nitroacrylates as starting materials of thiophene-2-carboxylates under continuous flow conditions. Adv. Synth. Catal. 2019, 361, 2042–2047. [Google Scholar] [CrossRef]
- Gabrielli, S.; Chiurchiù, E.; Palmieri, A. β-Nitroacrylates: A versatile and growing class of functionalized nitroalkenes. Adv. Synth. Catal. 2019, 361, 630–653. [Google Scholar] [CrossRef]
- Ballini, R.; Gabrielli, S.; Palmieri, A. β-Nitroacrylates as precursors of tetrasubstituted furans in a one-pot process and under acidic solvent-free conditions. Synlett 2010, 2468–2470. [Google Scholar] [CrossRef]
- Ballini, R.; Palmieri, A. Formation of carbon-carbon double bonds: Recent developments via nitrous acid elimination (NAE) from aliphatic nitro compounds. Adv. Synth. Catal. 2019, 361, 5070–5097. [Google Scholar] [CrossRef]
- Ballini, R.; Gabrielli, S.; Palmieri, A.; Petrini, M. A two-step synthesis of unsymmetrical 1,4-disubstituted carbazoles from sulfonylindoles under heterogeneous catalysis. Adv. Synth. Catal. 2010, 352, 2459–2462. [Google Scholar] [CrossRef]
- Kadota, J.; Chatani, N.; Murai, S. A platinum complex-catalyzed reaction of 3-chloro-1,3-diene monoepoxides with carbon nucleophiles involving nucleophilic substitution at the central carbon atom of the π-allyl ligand in the intermediate complex. Dependency of regioselectivity upon the added Lewis acids. Tetrahedron 2000, 56, 2231–2237. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 3a–n are available from the authors. |
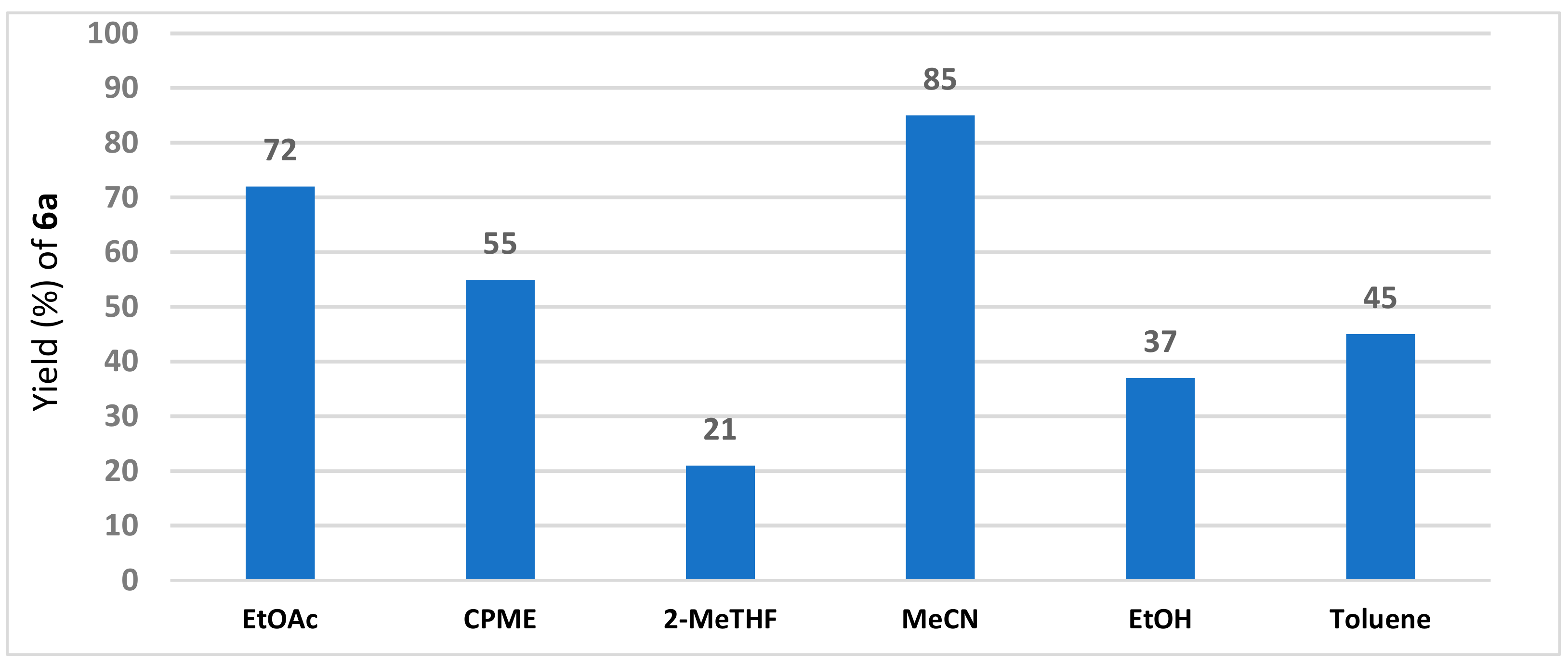
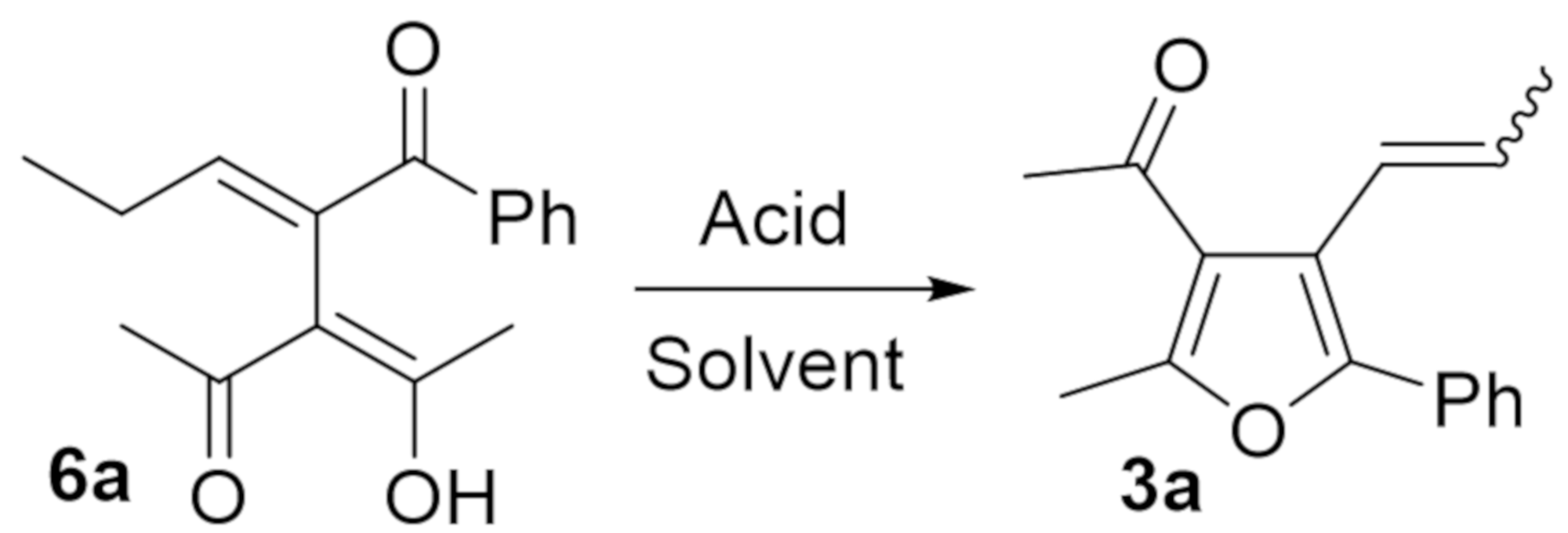
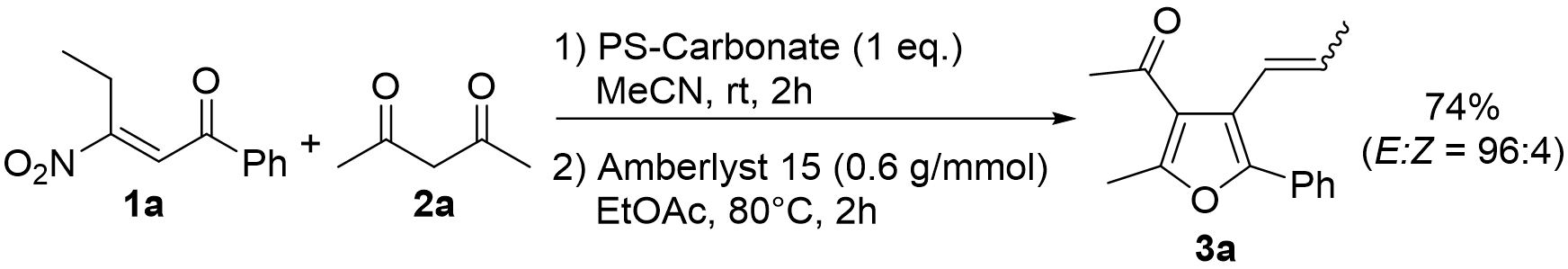
| Entry | Amberlyst 15 (g/mmol) | Solvent | Temperature (°C) 1 | Time (h) | Yield (%) of 3a 2 | E:Z |
|---|---|---|---|---|---|---|
| a | 1 | MeCN | 80 | 2 | 53 | 96:4 |
| b | 1 | EtOAc | 80 | 2 | 76 | 96:4 |
| c | 1 | Toluene | 80 | 2 | 68 | 82:18 |
| d | 1 | 2-MeTHF | 80 | 2 | 58 | 90:10 |
| e | 1 | EtOAc | 100 | 1 | 74 | 90:10 |
| f | 1 | EtOAc | 60 | 4 | 45 | 96:4 |
| g | 1.2 | EtOAc | 80 | 2 | 82 | 96:4 |
| h | 0.8 | EtOAc | 80 | 2 | 89 | 96:4 |
| i | 0.6 | EtOAc | 80 | 2 | 91 | 96:4 |
| j | 0.4 | EtOAc | 80 | 2 | 85 | 96:4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiurchiù, E.; Gabrielli, S.; Ballini, R.; Palmieri, A. A New Valuable Synthesis of Polyfunctionalized Furans Starting from β-Nitroenones and Active Methylene Compounds. Molecules 2019, 24, 4575. https://doi.org/10.3390/molecules24244575
Chiurchiù E, Gabrielli S, Ballini R, Palmieri A. A New Valuable Synthesis of Polyfunctionalized Furans Starting from β-Nitroenones and Active Methylene Compounds. Molecules. 2019; 24(24):4575. https://doi.org/10.3390/molecules24244575
Chicago/Turabian StyleChiurchiù, Elena, Serena Gabrielli, Roberto Ballini, and Alessandro Palmieri. 2019. "A New Valuable Synthesis of Polyfunctionalized Furans Starting from β-Nitroenones and Active Methylene Compounds" Molecules 24, no. 24: 4575. https://doi.org/10.3390/molecules24244575







