Heat Capacities and Thermodynamic Properties of Hungchaoite and Mcallisterite
Abstract
:1. Introduction
2. Experimental
2.1. Chemicals
2.2. Synthesis
2.3. Identification and Analytical Methods
2.4. Calorimetry and Experiment Method
3. Results and Discussion
3.1. Heat Capacities
3.2. Enthalpy, Entropy, and Gibbs Free Energy
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Li, J.G.; Wang, F.F.; Zhang, Y.J. Microstructure and mechanical properties of magnesium matrix composite reinforced with magnesium borate whisker. J. Compos. Mater. 2012, 24, 3011–3016. [Google Scholar] [CrossRef]
- Shao, S.N.; Xiong, X.X. Discussion on main Ore collection area of boron ore in China and its resource potential. Chem. Miner. Geol. 2010, 32, 65–74. [Google Scholar]
- Han, J.W.; Li, F.Q.; Wang, X.W. Advances in the extraction of boron from liquid mines. Salt Lake Res. 2007, 15, 57–61. [Google Scholar]
- Zhao, W.; Guo, Y.F.; Gao, J. The general situation of boron resources and the research progress of boron extraction in China. World Sci. Technol. Res. Dev. 2011, 33, 29–32. [Google Scholar]
- Han, X.N.; Zhao, M.L.; Wang, S.Q. Phase Equilibria of the Aqueous Systems Containing Magnesium and Borate Ions. Trans. Tech. 2014, 1015, 409–412. [Google Scholar] [CrossRef]
- Yuan, G.H. Structural Characteristics and Classification of Boric Acid Salt; Dalian University of Technology: Liaoning, China, 2007. [Google Scholar]
- Lin, F.; Dong, Y.P.; Peng, J.Y. Synthesis and formation mechanism of pinnoite by the phase transition process. Phase Transit. 2015, 89, 558–567. [Google Scholar] [CrossRef]
- Xu, G.M. Shortening the reaction time is the only way to produce the high yield and low consumption of borax by carbon-alkali method. Dandong Chem. 1995, 12, 1–3. [Google Scholar]
- Meng, L.Z.; Deng, T.L.; Duan, C.W. Study on synthesis method of three-way boron magnesium stone. World Sci. Technol. Res. Dev. 2010, 32, 825–826. [Google Scholar]
- Derun, E.M.; Senberber, F.T. Characterization and thermal dehydration kinetics of highly crystalline mcallisterite, synthesized at low temperatures. Sci. World J. 2014, 2014, 985185. [Google Scholar]
- Frost, R.L.; López, A.; Xi, Y.F. The molecular structure of the borate mineral inderite Mg(H4B3O7)(OH)5H2O—A vibrational spectroscopic study. Mol. Biomol. Spectrosc. 2013, 116, 160–164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qu, Y.H.; Han, W.T.; Qian, Z.Q. Three ways boron magnesium stone a new borate mineral. Geology 1965, 45, 298–305. [Google Scholar]
- Marochkina, M.N.; Smirnova, N.N.; Knyazev, A.V. The low-temperature heat capacity of alkali and alkaline-earth metal uranoborates. Russ. J. Phys. Chem. 2008, 82, 1516–1520. [Google Scholar] [CrossRef]
- Zhang, S.S. Thermochemical Properties of Aqueous System Containing Alkalis Borates; Tianjin University of Science and Technology: Tianjin, China, 2017. [Google Scholar]
- Bespyatov, M.A.; Chernyaikin, I.S.; Naumov, V.N. Low-temperature heat capacity of Al(C11H19O2)3. Thermochim. Acta 2014, 596, 40–41. [Google Scholar] [CrossRef]
- Gao, S.Y.; Xu, K.F.; Li, G. The behavior of boric acid in the chemical v-of boron-containing concentrated halogen dilution of halogen borate. J. Chem. 1986, 44, 1229–1233. [Google Scholar]
- Calvar, N.; Gómez, E.; Macedo, E.A. Thermal analysis and heat capacities of pyridinium and imidazolium ionic liquids. Thermochim. Acta 2013, 565, 178–182. [Google Scholar] [CrossRef]
- Hu, R.Z.; Zhao, F.Q.; Gao, H.X. Fundamentals and Applications of Quantitative Thermodynamics; Science Press: Beijing, China, 2011. [Google Scholar]
- Ge, H.W. Study on Multi-Warm Mechanical Properties of MgB4O7-H2O System; University of Technology: Chengdu, China, 2011. [Google Scholar]
- Qinghai Institute of Salt Lakes, Chinese Academy of Sciences. Analysis Methods for Brines and Salts, 2nd ed.; Science Press: Beijing, China, 1998.
- Li, H.X.; Dong, O.Y.; Yao, Y. The mass titration analytical method and its application. J. Salt Lake Res. 2011, 19, 31–36. [Google Scholar]
- Speight, J.G. Lange’s Handbook of Chemistry, 16th ed.; McGraw-Hill: New York, NY, USA, 2005. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |
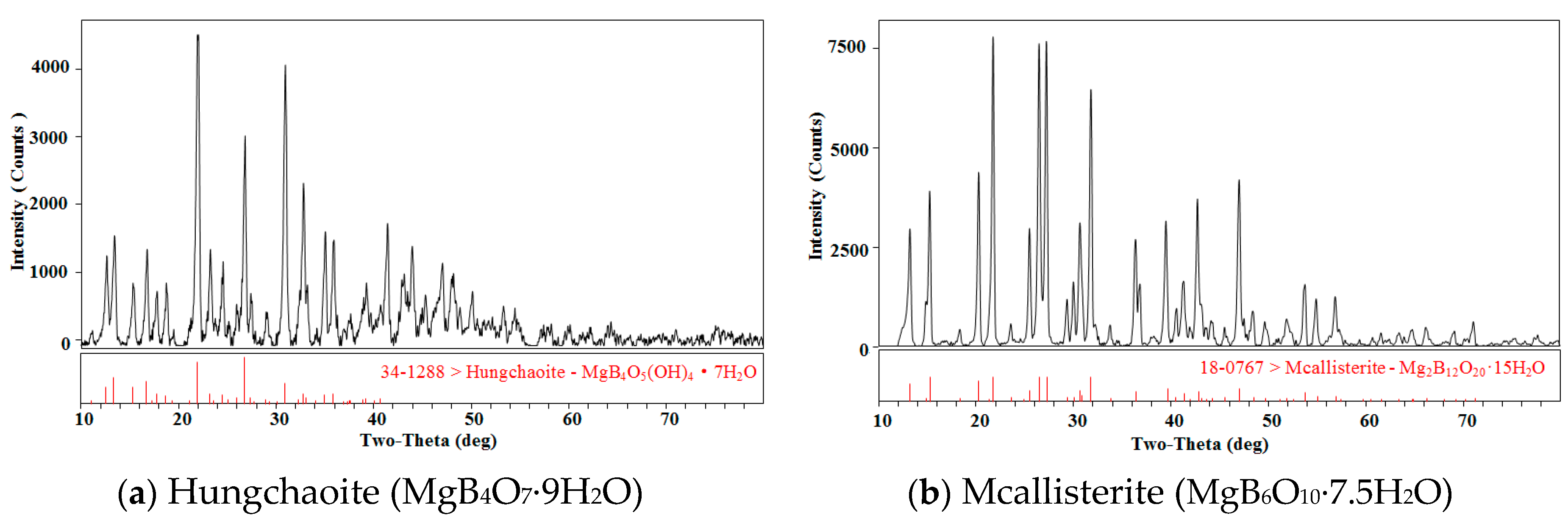
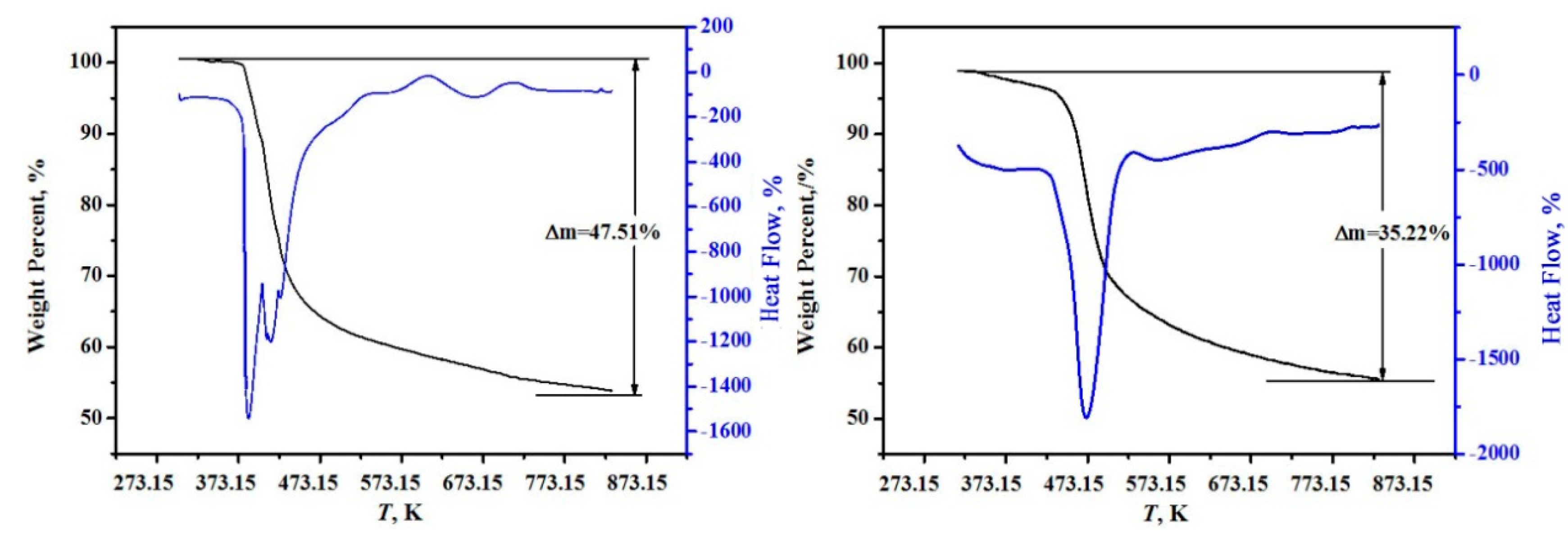
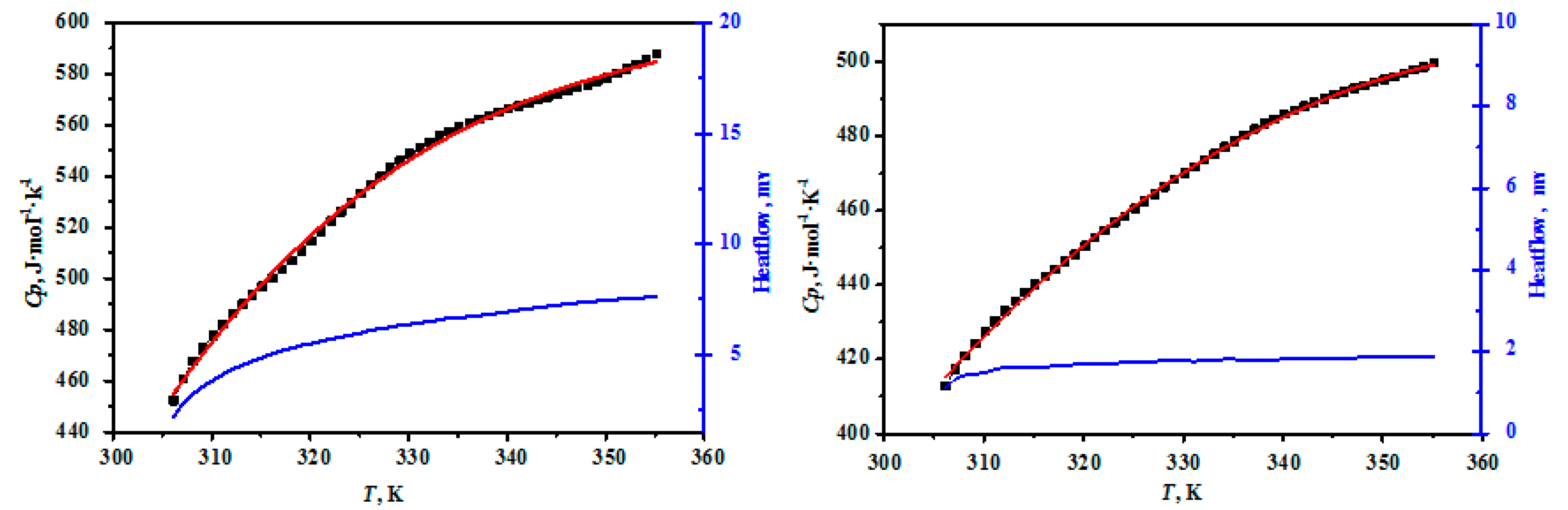
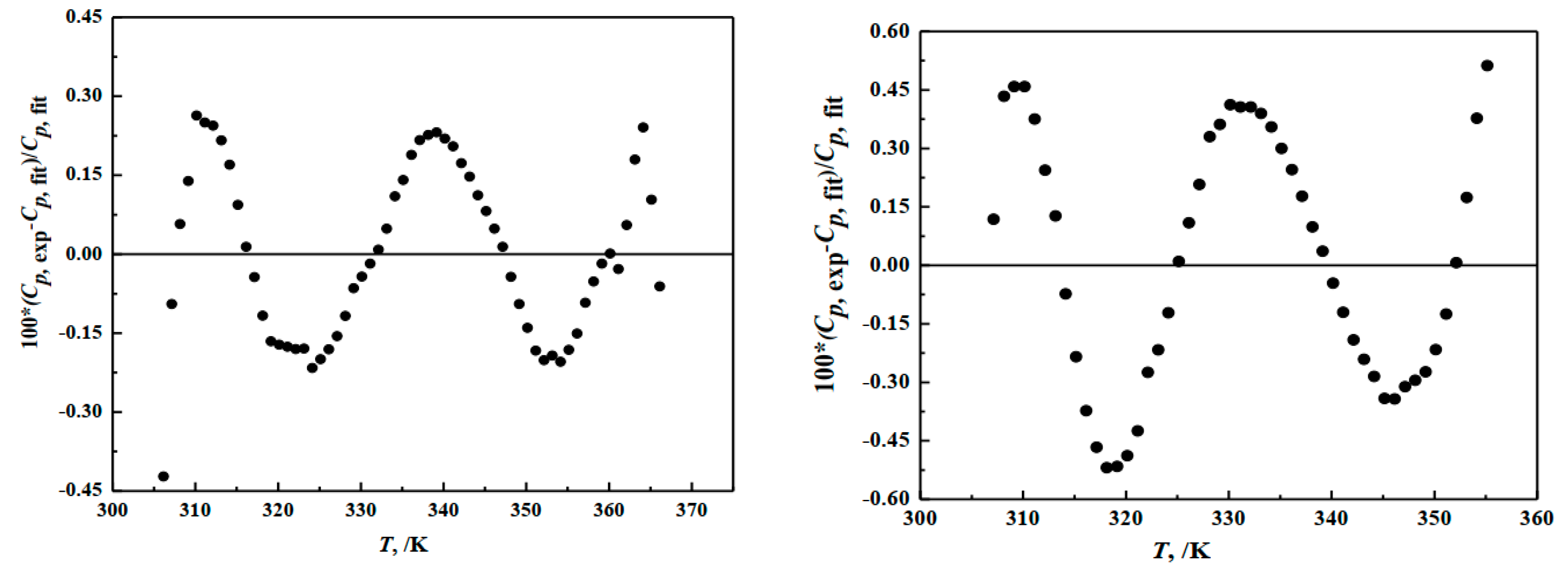
| Component Content | MgO/wt % | B2O3/wt % | H2O/wt % | n (MgO:B2O3:H2O) |
|---|---|---|---|---|
| Hungchaoite, MgB4O7·9H2O | ||||
| Experimental value | 11.86 | 40.79 | 47.35 | 1:2.04:8.95 |
| Theoretical value | 11.80 | 40.75 | 47.45 | 1:2.00:9.00 |
| Relative error (%) | 0.51 | 0.05 | 0.20 | - |
| Mcallisterite, MgB6O10·7.5H2O | ||||
| Experimental value | 10.50 | 54.60 | 34.87 | 1:3.00:7.42 |
| Theoretical value | 10.49 | 54.35 | 35.16 | 1:3.00:7.50 |
| Relative error (%) | 0.10 | 0.46 | 0.82 | - |
| Hungchaoite, MgB4O7·9H2O | |||||
| 306.15 | 452.21 | 323.15 | 525.99 | 340.15 | 566.32 |
| 307.15 | 460.92 | 324.15 | 529.57 | 341.15 | 567.48 |
| 308.15 | 467.61 | 325.15 | 533.26 | 342.15 | 568.58 |
| 309.15 | 472.84 | 326.15 | 536.68 | 343.15 | 569.74 |
| 310.15 | 477.79 | 327.15 | 539.99 | 344.15 | 570.90 |
| 311.15 | 482.20 | 328.15 | 543.34 | 345.15 | 571.92 |
| 312.15 | 486.20 | 329.15 | 546.07 | 346.15 | 573.19 |
| 313.15 | 490.12 | 330.15 | 548.84 | 347.15 | 574.59 |
| 314.15 | 493.50 | 331.15 | 551.19 | 348.15 | 575.85 |
| 315.15 | 496.89 | 332.15 | 553.48 | 349.15 | 577.12 |
| 316.15 | 500.27 | 333.15 | 555.60 | 350.15 | 578.55 |
| 317.15 | 503.72 | 334.15 | 557.54 | 351.15 | 580.12 |
| 318.15 | 507.23 | 335.15 | 559.25 | 352.15 | 581.90 |
| 319.15 | 510.92 | 336.15 | 560.93 | 353.15 | 583.84 |
| 320.15 | 514.61 | 337.15 | 562.39 | 354.15 | 586.00 |
| 321.15 | 518.37 | 338.15 | 563.76 | 355.15 | 587.70 |
| 322.15 | 522.47 | 339.15 | 565.13 | ||
| Mcallisterite, MgB6O10·7.5H2O | |||||
| 306.15 | 412.82 | 323.15 | 456.70 | 340.15 | 485.82 |
| 307.15 | 417.28 | 324.15 | 458.50 | 341.15 | 486.97 |
| 308.15 | 420.89 | 325.15 | 460.50 | 342.15 | 488.04 |
| 309.15 | 424.16 | 326.15 | 462.46 | 343.15 | 489.08 |
| 310.15 | 427.54 | 327.15 | 464.38 | 344.15 | 490.08 |
| 311.15 | 430.26 | 328.15 | 466.34 | 345.15 | 491.04 |
| 312.15 | 432.95 | 329.15 | 468.30 | 346.15 | 491.96 |
| 313.15 | 435.49 | 330.15 | 470.06 | 347.15 | 492.85 |
| 314.15 | 437.87 | 331.15 | 471.83 | 348.15 | 493.61 |
| 315.15 | 440.06 | 332.15 | 473.56 | 349.15 | 494.38 |
| 316.15 | 442.14 | 333.15 | 475.29 | 350.15 | 495.15 |
| 317.15 | 444.25 | 334.15 | 477.06 | 351.15 | 495.92 |
| 318.15 | 446.25 | 335.15 | 478.67 | 352.15 | 496.76 |
| 319.15 | 448.28 | 336.15 | 480.32 | 353.15 | 497.72 |
| 320.15 | 450.47 | 337.15 | 481.86 | 354.15 | 498.61 |
| 321.15 | 452.58 | 338.15 | 483.24 | 355.15 | 499.61 |
| 322.15 | 454.66 | 339.15 | 484.59 | ||
| Hungchaoite, MgB4O7·9H2O | ||||
| 298.15 | 400.86 | |||
| 300.15 | 415.14 | 0.8161 | 2.7281 | −0.0027 |
| 305.15 | 452.21 | 2.9743 | 9.8585 | −0.0340 |
| 310.15 | 477.79 | 5.2814 | 17.3573 | −0.1019 |
| 315.15 | 496.89 | 7.7153 | 25.1415 | −0.2081 |
| 320.15 | 514.61 | 10.2557 | 33.1389 | −0.3537 |
| 325.15 | 533.26 | 12.8850 | 41.2877 | −0.5397 |
| 330.15 | 548.84 | 15.5875 | 49.5357 | −0.7667 |
| 335.15 | 559.25 | 18.3498 | 57.8397 | −1.0351 |
| 340.15 | 566.32 | 21.1609 | 66.1652 | −1.3452 |
| 345.15 | 571.92 | 24.0119 | 74.4855 | −1.6968 |
| 350.15 | 578.55 | 26.8960 | 82.7816 | −2.0900 |
| 355.15 | 587.70 | 29.8090 | 91.0418 | −2.5245 |
| Mcallisterite, MgB6O10·7.5H2O | ||||
| 298.15 | 387.81 | |||
| 300.15 | 395.16 | 0.7830 | 2.6175 | −0.0026 |
| 305.15 | 412.09 | 2.8020 | 9.2881 | −0.0323 |
| 310.15 | 427.54 | 4.9007 | 16.1098 | −0.0957 |
| 315.15 | 440.06 | 7.0701 | 23.0482 | −0.1936 |
| 320.15 | 450.47 | 9.3016 | 30.0732 | −0.3264 |
| 325.15 | 460.50 | 11.5876 | 37.1584 | −0.4944 |
| 330.15 | 470.06 | 13.9213 | 44.2810 | −0.6980 |
| 335.15 | 478.67 | 16.2966 | 51.4213 | −0.9373 |
| 340.15 | 485.82 | 18.7081 | 58.5632 | −1.2122 |
| 345.15 | 491.04 | 21.1511 | 65.6933 | −1.5229 |
| 350.15 | 495.15 | 23.6221 | 72.8009 | −1.8691 |
| 355.15 | 499.61 | 26.1179 | 79.8781 | −2.2508 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, J.; Yuan, F.; Li, L.; Guo, Y.; Deng, T. Heat Capacities and Thermodynamic Properties of Hungchaoite and Mcallisterite. Molecules 2019, 24, 4470. https://doi.org/10.3390/molecules24244470
Song J, Yuan F, Li L, Guo Y, Deng T. Heat Capacities and Thermodynamic Properties of Hungchaoite and Mcallisterite. Molecules. 2019; 24(24):4470. https://doi.org/10.3390/molecules24244470
Chicago/Turabian StyleSong, Jiangtao, Fei Yuan, Long Li, Yafei Guo, and Tianlong Deng. 2019. "Heat Capacities and Thermodynamic Properties of Hungchaoite and Mcallisterite" Molecules 24, no. 24: 4470. https://doi.org/10.3390/molecules24244470
APA StyleSong, J., Yuan, F., Li, L., Guo, Y., & Deng, T. (2019). Heat Capacities and Thermodynamic Properties of Hungchaoite and Mcallisterite. Molecules, 24(24), 4470. https://doi.org/10.3390/molecules24244470





