Immunomodulatory Effects of the Meretrix Meretrix Oligopeptide (QLNWD) on Immune-Deficient Mice
Abstract
:1. Introduction
2. Results and Discussion
2.1. Comparison of Body Weight
2.2. Thymus and Spleen Indexes
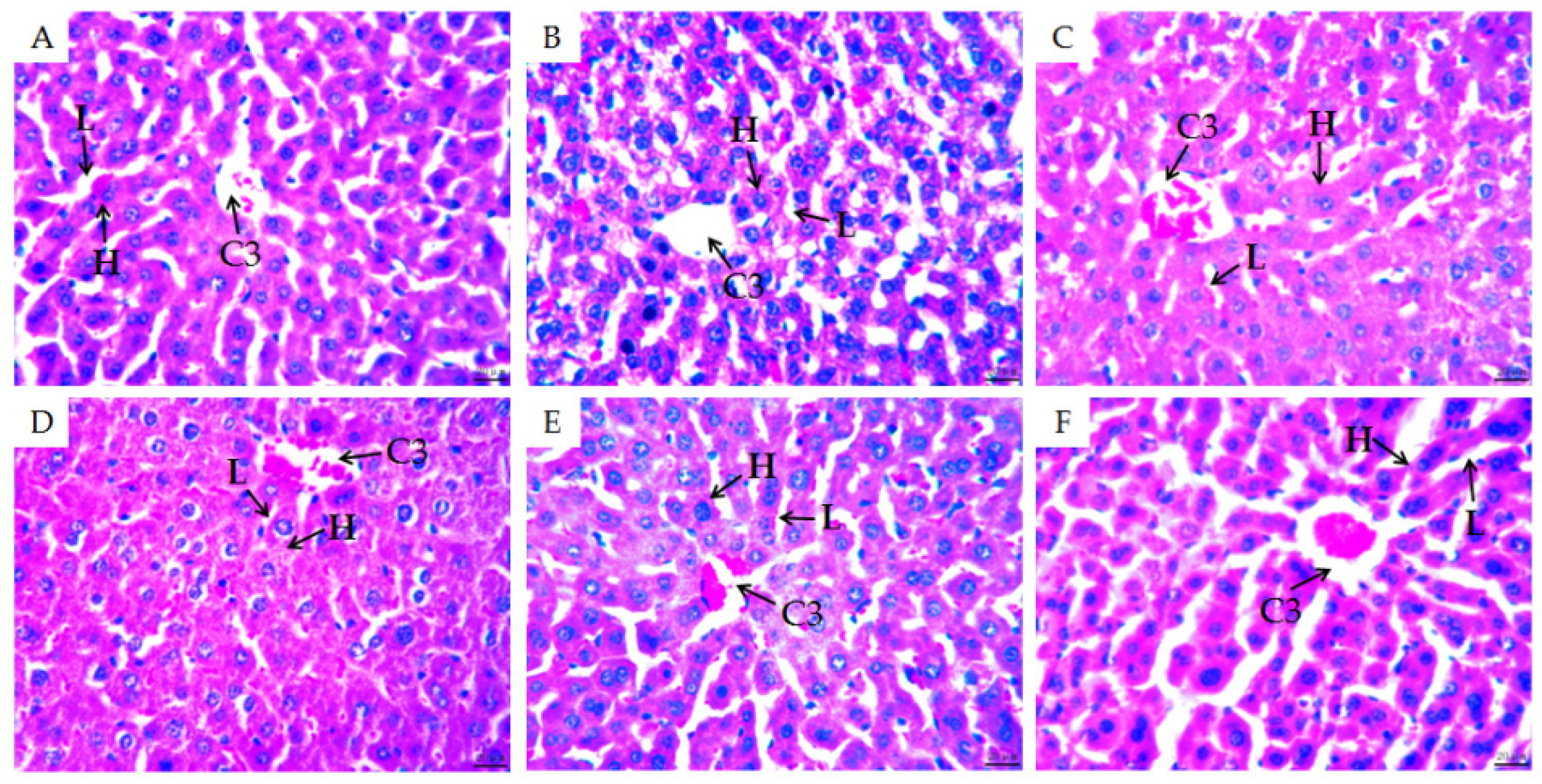
2.3. Morphological Observations of Mouse Organs
2.4. Serum Immunoglobulin G (IgG) Levels
2.5. Serum Hemolysin
2.6. T Lymphocyte Assessment
3. Materials and Methods
3.1. Animals
3.2. Materials and Chemical Reagents
3.3. Animal Groupings and Treatments
3.4. Body Weight and Immune Organ Index Changes
3.5. Histomorphological Observation
3.6. Determination of IgG Serum Content
3.7. Detection of Serum Hemolysin
3.8. Proliferation of Spleen T-Lymphocytes
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Deguine, J. New Flavors in Immunomodulation. Cell 2018, 173, 1553–1555. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gein, S.V.; Sharav’eva, I.L. Immunomodulating Effects of Cold Stress. Biol. Bull. Rev. 2018, 8, 482–488. [Google Scholar] [CrossRef]
- Yahfoufi, N.; Mallet, J.F.; Graham, E.; Matar, C. Role of probiotics and prebiotics in immunomodulation. Curr. Opin. Food Sci. 2018, 20, 82–91. [Google Scholar] [CrossRef]
- Yao, W.; Wang, F.; Wang, H. Immunomodulation of artemisinin and its derivatives. Sci. Bull. 2016, 61, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Turowski, R.C.; Triozzi, P.L. Application of chemical immunomodulators to the treatment of cancer and AIDS. Cancer Invest. 1994, 12, 620–643. [Google Scholar] [CrossRef] [PubMed]
- Backus, K.M.; Cao, J.; Maddox, S.M. Opportunities and challenges for the development of covalent chemical immunomodulators. Bioorg. Med. Chem. Lett. 2019, 27, 3421–3439. [Google Scholar] [CrossRef]
- Kanterman, J.; Sade-Feldman, M.; Baniyash, M. New insights into chronic inflammation-induced immunosuppression. Semin. Cancer Biol. 2012, 22, 307–318. [Google Scholar] [CrossRef]
- Seyed, M.A. A comprehensive review on Phyllanthus derived natural products as potential chemotherapeutic and immunomodulators for a wide range of human disease. Biocatal. Agr. Biotechnol. 2019, 17, 529–537. [Google Scholar] [CrossRef]
- Tarnawski, M.; Depta, K.; Grejciun, D.; Szelepin, B. HPLC determination of phenolic acids and antioxidant activity in concentrated peat extract—a natural immunomodulator. J. Pharmaceut. Biomed. 2006, 41, 182–188. [Google Scholar] [CrossRef]
- Hong, S.H.; Ku, J.M.; In Kim, H.; Ahn, C.-W.; Park, S.-H.; Seo, H.S.; Shin, Y.C.; Ko, S.-G. The immune-enhancing activity of Cervus nippon mantchuricus extract (NGE) in RAW264.7 macrophage cells and immunosuppressed mice. Food Res. Int. 2017, 99, 623–629. [Google Scholar] [CrossRef]
- Bueno, C.A.; Barquero, A.A.; Di Cónsoli, H.; Maier, M.S.; Alché, L.E. A natural tetranortriterpenoid with immunomodulating properties as a potential anti-HSV agent. Virus Res. 2009, 141, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Song, R.; Wei, R.; Zhang, B.; Yang, Z.; Wang, D. Antioxidant and Antiproliferative Activities of Heated Sterilized Pepsin Hydrolysate Derived from Half-Fin Anchovy (Setipinna taty). Mar. Drugs 2011, 9, 1142–1156. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Zhao, S.; Yu, F.; Yang, Z.; Ding, G. Protective Effects and Mechanism of Meretrix meretrix Oligopeptides against Nonalcoholic Fatty Liver Disease. Mar. Drugs 2017, 15, 31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, F.; Zhang, Y.; Ye, L.; Tang, Y.; Ding, G.; Zhang, X.; Yang, Z. A novel anti-proliferative pentapeptide (ILYMP) isolated fromCyclina sinensisprotein hydrolysate induces apoptosis of DU-145 prostate cancer cells. Mol. Med. Rep. 2018, 18, 771–778. [Google Scholar] [PubMed] [Green Version]
- Pang, G.C.; Chen, Q.S.; Hu, Z.-H.; Xie, J.B. Bioactive Peptides:Absorption, Utilization and Functionality. Food Sci. 2013, 34, 375–391. [Google Scholar]
- Yang, R.Y.; Zhang, Z.F.; Pei, X.Y.; Han, X.L.; Wang, J.B.; Wang, L.L.; Long, Z.; Shen, X.Y.; Li, Y.i. Immunomodulatory effects of marine oligopeptide preparation from Chum Salmon (Oncorhynchus keta) in mice. Food Chem. 2009, 113, 464–470. [Google Scholar] [CrossRef]
- Gao, S.; Hong, H.; Zhang, C.Y.; Wang, K.; Zhang, B.H.; Han, Q.A.; Liu, H.G.; Luo, Y.K. Immunomodulatory effects of collagen hydrolysates from yak (Bos grunniens) bone on cyclophosphamide-induced immunosuppression in BALB/c mice. J. Funct. Foods 2019, 60, 103420. [Google Scholar] [CrossRef]
- Li, W.; Ye, S.W.; Zhang, Z.W.; Tang, J.C.; Jin, H.X.; Huang, F.F.; Yang, Z.S.; Tang, Y.P.; Chen, Y.; Ding, G.F.; et al. Purification and Characterization of a Novel Pentadecapeptide from Protein Hydrolysates of Cyclina sinensis and Its Immunomodulatory Effects on RAW264.7 Cells. Mar. Drugs 2019, 17, 30. [Google Scholar] [CrossRef] [Green Version]
- Yu, F.M.; Zhang, Z.W.; Ye, S.W.; Hong, X.X.; Jin, H.X.; Huang, F.F.; Yang, Z.S.; Tang, Y.P.; Chen, Y.; Ding, G.F. Immunoenhancement effects of pentadecapeptide derived from Cyclina sinensis on immune-deficient mice induced by Cyclophosphamide. J. Funct. Foods. 2019, 60, 103408. [Google Scholar] [CrossRef]
- Huang, F.F.; Wang, J.J.; Yu, F.M.; Tang, Y.P.; Ding, G.F.; Yang, Z.S.; Sun, Y. Protective Effect of Meretrix meretrix Oligopeptides on High-Fat-Diet-Induced Non-Alcoholic Fatty Liver Disease in Mice. Mar. Drugs 2018, 16, 39. [Google Scholar] [CrossRef] [Green Version]
- Ye, L.; Yan, J.; Zhang, W.; Zou, S.S.; Ye, S.W.; Yang, Z.S.; Yu, F.M.; Ding, G.F. Immunomodulatory effects of Meretrix meretrix oligopeptides on RAW264.7 cells. J. Fish. China 2019, 43, 24–32. [Google Scholar]
- Kyung, D.; Sung, H.; Kim, Y. Global transcriptome analysis identifies weight regain-induced activation of adaptive immune responses in white adipose tissue of mice(Article). Int. J. Obesity 2018, 42, 755–764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sadasivan, S.; Vasamsetti, B.; Singh, J.; Marikunte, V.; Oommen, A.; Jagannath, M.; Rao, R. Exogenous administration of spermine improves glucose utilization and decreases bodyweight in mice. Eur. J. Pharmacol. 2014, 729, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Lee, S.; Lee, S. Rutecarpine ameliorates bodyweight gain through the inhibition of orexigenic neuropeptides NPY and AgRP in mice. Biochem. Bioph. Res. Co. 2009, 389, 437–442. [Google Scholar] [CrossRef]
- Deng, Y.; Wang, L.; Wang, C. Tolerance-like innate immunity and spleen injury: A novel discovery via the weekly administrations and consecutive injections of PEGylated emulsions. Int. J. Nanomed. 2014, 9, 3645–3657. [Google Scholar] [CrossRef] [Green Version]
- Mebius, R.E.; Kraal, G. Structure and function of the spleen. Nat. Rev. Immunol. 2005, 5, 606–616. [Google Scholar] [CrossRef]
- Guo, L.; Sun, Y.; Wang, A. Effect of polysaccharides extract ofrhizoma atractylodis macrocephalaeon thymus, spleen and cardiac indexes, caspase-3 activity ratio, Smac/DIABLO and HtrA2/Omi protein and mRNA expression levels in aged rats. Mol. Biol. Rep. 2012, 39, 9285–9290. [Google Scholar] [CrossRef]
- Zhe, W.; Yu, Z.; Yan, Y. Lipopolysaccharide Preconditioning Increased the Level of Regulatory B cells in the Spleen after Acute Ischaemia/Reperfusion in Mice. Brain Res. 2018, 1701, 46–57. [Google Scholar]
- Salem, M.L.; Al-Khami, A.A.; El-Nagaar, S.A.; Zidan, A.A.; Al-Sharkwi, I.M.; Díaz-Montero, C.M.; Cole, D.J. Kinetics of rebounding of lymphoid and myeloid cells in mouse peripheral blood, spleen and bone marrow after treatment with cyclophosphamide. Cell. Immunol. 2012, 276, 67–74. [Google Scholar] [CrossRef] [Green Version]
- Brian, J.; Jason, C.; John, C. Reduction of MHC-I expression limits T-lymphocyte-mediated killing of Cancer-initiating cells. BMC Cancer 2018, 18, 469. [Google Scholar]
- Battista, J.; Tallmadge, R.; Stokol, T.; Felippe, M. Hematopoiesis in the equine fetal liver suggests immune preparedness. Immunogenetics 2014, 66, 635–649. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vidarsson, G.; Dekkers, G.; Rispens, T. IgG Subclasses and Allotypes: From Structure to Effector Functions. Front. Immuno. 2014, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, H.W.; Shen, X.H.; Chen, Z.Y.; Liu, F.; Wang, T.; Xie, Y.K.; Ma, C. Nociceptive neuronal Fc-gamma receptor I is involved in IgG immune complex induced pain in the rat. Brain Behav. Immun. 2017, 62, 351–361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sangwan, V.; Tomar, S.K.; Ali, B.; Singh, R.R.B.; Singh, A.K. Galactooligosaccharides reduce infection caused by Listeria monocytogenes and modulate IgG and IgA levels in mice. Int. Dairy J. 2015, 41, 58–63. [Google Scholar] [CrossRef]
- Pan, D.D.; Wu, Z.; Liu, J. Immunomodulatory and hypoallergenic properties of milk protein hydrolysates in ICR mice, Journal of Dairy Science. J. Dairy Sci. 2013, 96, 4958–4964. [Google Scholar] [CrossRef]
- Chen, Y.; Tang, J.B.; Wang, X.K.; Sun, F.X.; Liang, S.J. An immunostimulatory polysaccharide (SCP-IIa) from the fruit of Schisandra chinensis (Turcz.). Int. J. Biol. Macromol. 2012, 50, 844–848. [Google Scholar] [CrossRef]
- Liu, J.C.; Sun, H.; Nie, C.X.; Ge, W.X.; Wang, Y.Q.; Zhang, W.J. Oligopeptide derived from solid-state fermented cottonseed meal significantly affect the immunomodulatory in balb/c mice treated with cyclophosphamide. Food Sci. Biotechnol. 2018, 27, 1791–1799. [Google Scholar] [CrossRef] [Green Version]
- Karayannopoulou, M.; Anagnostou, T.; Margariti, A. Evaluation of blood T-lymphocyte subpopulations involved in host cellular immunity in dogs with mammary cancer. Vet. Immunol. Immunop. 2017, 186, 45–50. [Google Scholar] [CrossRef]
- Actor, J. T Lymphocytes: Ringleaders of Adaptive Immune Function. In Introductory Immunology, 1st ed.; Academic Press: Cambridge, MA, USA, 2014; pp. 42–58. [Google Scholar]
- Nikš, M.; Otto, M.; Bušová, B.; Stefanovic, J. Quantification of proliferative and suppressive responses of human T lymphocytes following ConA stimulation. J. Immunol. Methods 1990, 126, 263–271. [Google Scholar] [CrossRef]
- Mustafa, W.; Al-Saleem, F.H.; Nasser, Z. Immunization of mice with the non-toxic HC50 domain of botulinum neurotoxin presented by rabies virus particles induces a strong immune response affording protection against high-dose botulinum neurotoxin challenge. Vaccine 2011, 29, 4638–4645. [Google Scholar] [CrossRef] [Green Version]
- Ye, F.; Yan, S.; Xu, L. Tr1 regulatory T cells induced by ConA pretreatment prevent mice from ConA-induced hepatitis. Immunol. Lett. 2009, 122, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X. Immunization with Pseudomonas aeruginosa outer membrane vesicles stimulates protective immunity in mice. Vaccine 2018, 36, 1047–1054. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Huang, S.; Ye, Q.H.; Zeng, X.F.; Yu, H.T.; Qi, D.S.; Qiao, S.Y. Prevention of Cyclophosphamide-Induced Immunosuppression in Mice with the Antimicrobial Peptide Sublancin. Clin. Dev. Immunol. 2018, 2018, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meng, Y.Y.; Li, B.L.; Jin, D.; Zhan, M.; Lu, J.J.; Huo, G.C. Immunomodulatory activity ofLactobacillus plantarumKLDS1.0318 in cyclophosphamide-treated mice. Food Nutr. Res. 2018, 62, 1296. [Google Scholar] [CrossRef]
- Cai, G.D.; Sun, K.; Wang, T. Mechanism and effects of Zearalenone on mouse T lymphocytes activation in vitro. Ecotox. Environ. Safe 2018, 162, 208–217. [Google Scholar] [CrossRef]
- Ye, Y.; Zhang, Y.; Lu, X.; Huang, X.Y.; Zeng, X.F.; Lai, X.Q.; Zeng, Y.Y. The anti-inflammatory effect of the SOCC blocker SK&F 96365 on mouse lymphocytes after stimulation by ConA or PMA/ionomycin. Immunobiology 2011, 216, 1044–1053. [Google Scholar]






| Group | HC50 | Proliferation Rate % |
|---|---|---|
| Negative Control | 87.58 ± 0.05 | 0 |
| Diseased Model | 86.90 ± 0.05 | −0.78 ± 0.05 |
| Positive Control | 91.39 ± 0.03 * | 4.35 ± 0.03 * |
| Low-dose | 87.84 ± 0.10 | 0.29 ± 0.10 |
| Medium-dose | 89.27 ± 0.07 | 1.92 ± 0.07 |
| High-dose | 90.78 ± 0.05 * | 3.65 ± 0.05 * |
| Group | Pre-Treatment (10 days) | Treatment (5 days) |
|---|---|---|
| Dose (0.2 mL) | Dose (0.2 mL) | |
| Negative Control | NS | NS |
| Disease Model | NS | CTX (80 mg/kg BW) |
| Positive Control | Levamisole (25 mg/kg BW) | CTX (80 mg/kg BW) |
| Low Dose | MMO (50 mg/kg BW) | CTX (80 mg/kg BW) |
| Medium Dose | MMO (100 mg/kg BW) | CTX (80 mg/kg BW) |
| High Dose | MMO (200 mg/kg BW) | CTX (80 mg/kg BW) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, W.; Ye, L.; Wang, F.; Zheng, J.; Tian, X.; Chen, Y.; Ding, G.; Yang, Z. Immunomodulatory Effects of the Meretrix Meretrix Oligopeptide (QLNWD) on Immune-Deficient Mice. Molecules 2019, 24, 4452. https://doi.org/10.3390/molecules24244452
Zhang W, Ye L, Wang F, Zheng J, Tian X, Chen Y, Ding G, Yang Z. Immunomodulatory Effects of the Meretrix Meretrix Oligopeptide (QLNWD) on Immune-Deficient Mice. Molecules. 2019; 24(24):4452. https://doi.org/10.3390/molecules24244452
Chicago/Turabian StyleZhang, Wen, Lei Ye, Fenglei Wang, Jiawen Zheng, Xiaoxiao Tian, Yan Chen, Guofang Ding, and Zuisu Yang. 2019. "Immunomodulatory Effects of the Meretrix Meretrix Oligopeptide (QLNWD) on Immune-Deficient Mice" Molecules 24, no. 24: 4452. https://doi.org/10.3390/molecules24244452






