Some Properties of Electron Beam-Irradiated Sheep Wool Linked to Cr(III) Sorption
Abstract
1. Introduction
2. Results and Discussion
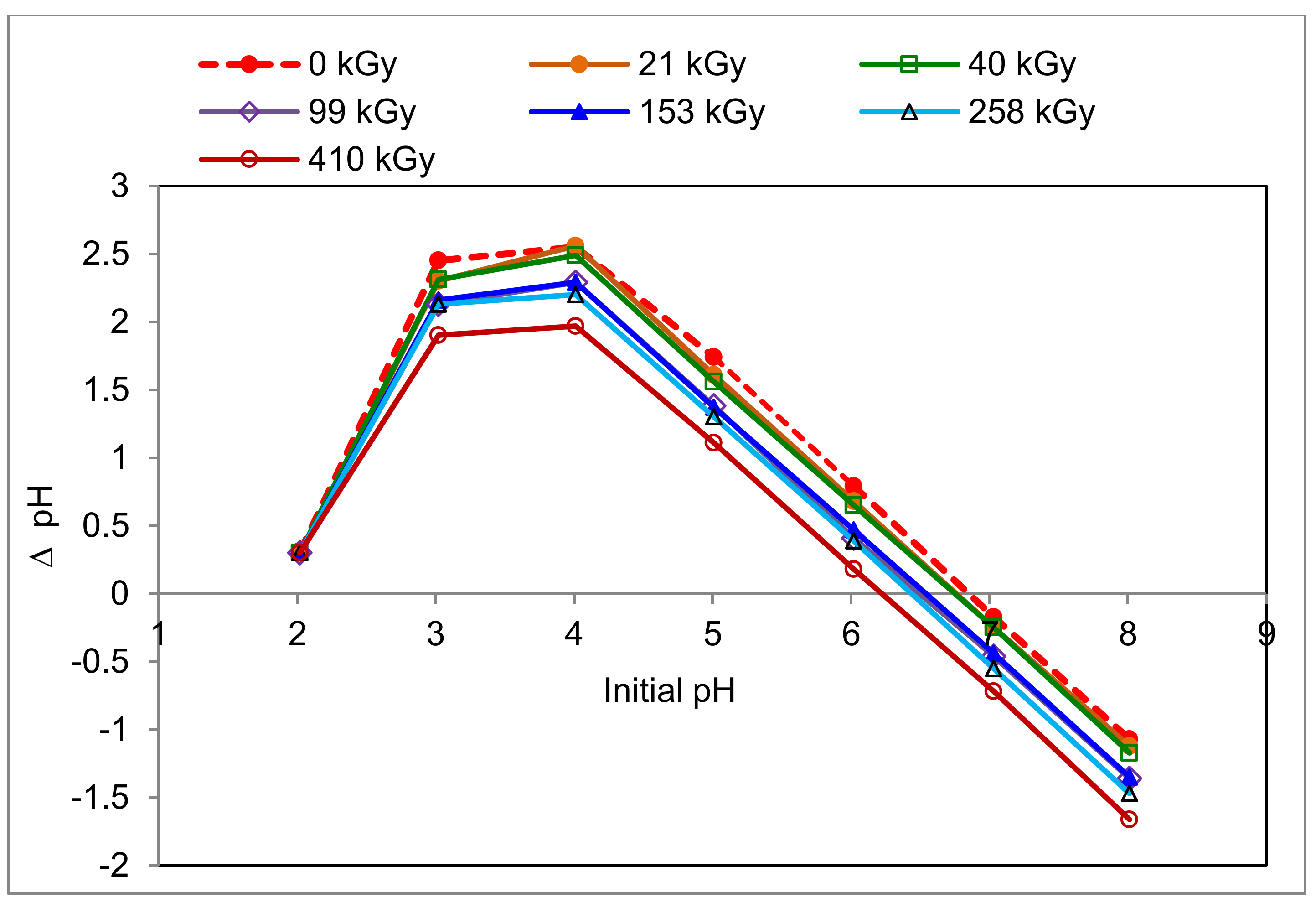
2.1. Impact of Absorbed Dose on Surface Properties of Wool (IEP/PZC)
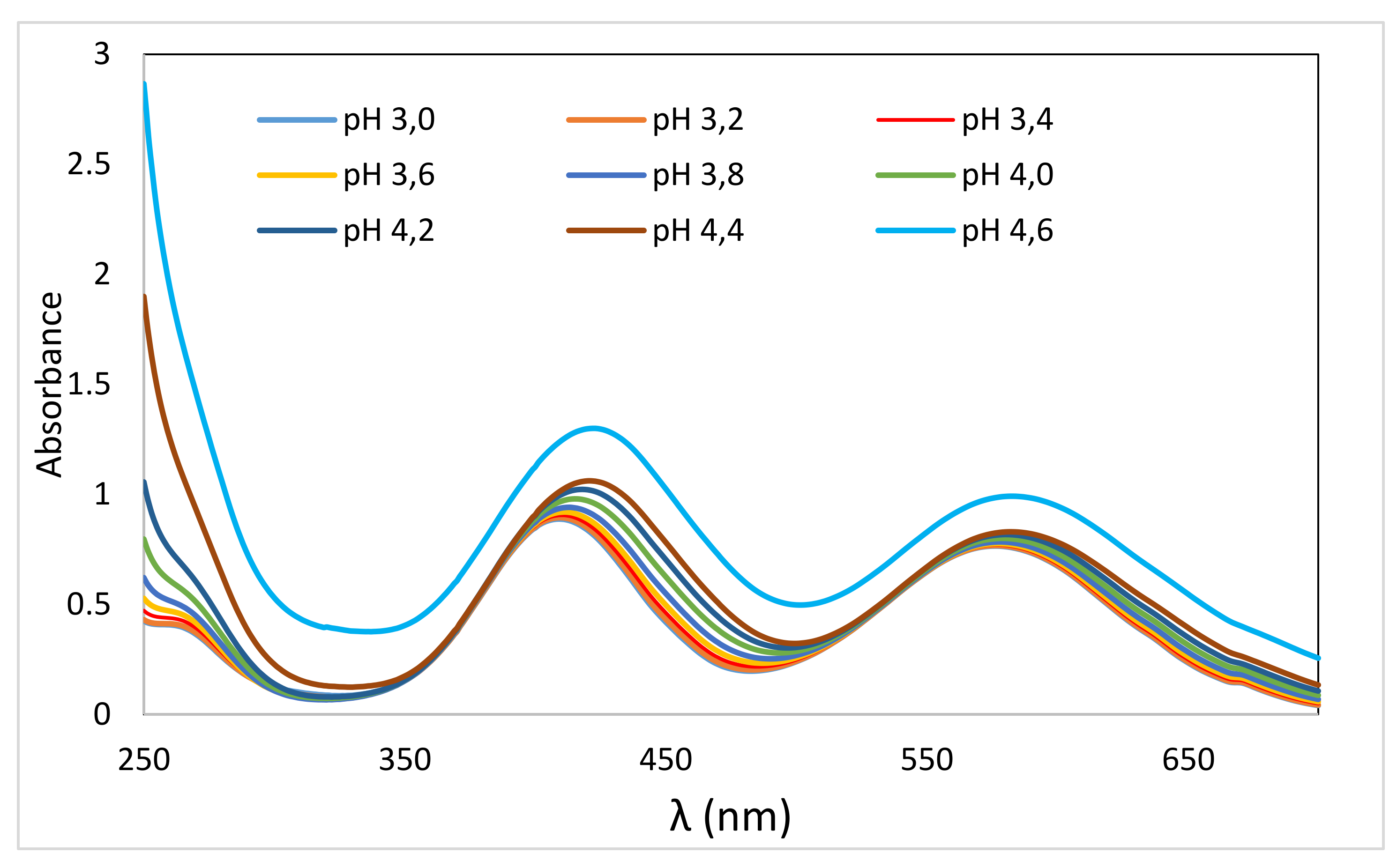
2.2. Effect of pH on VIS Spektra of Cr(III)
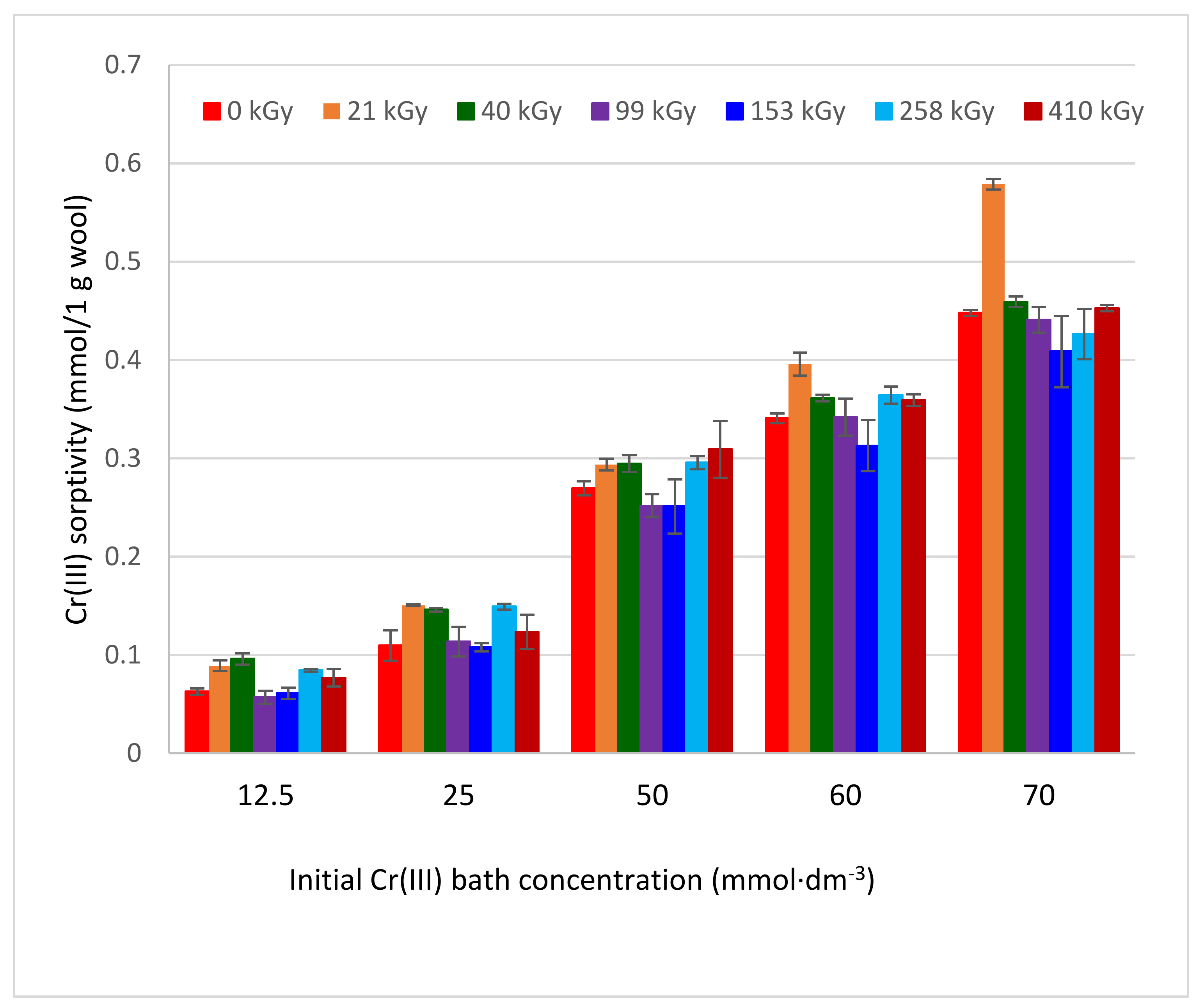
2.3. Sorptivity
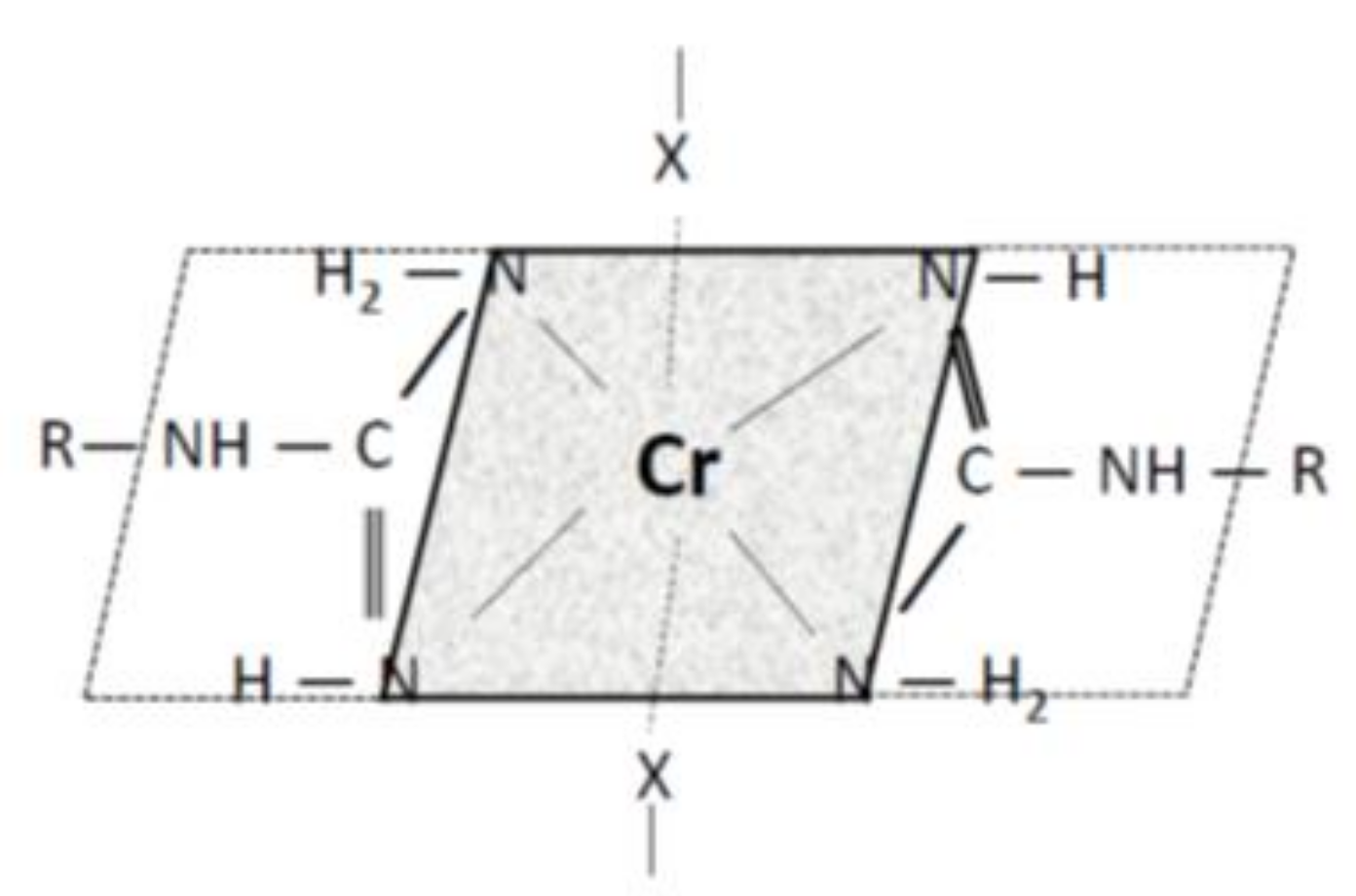
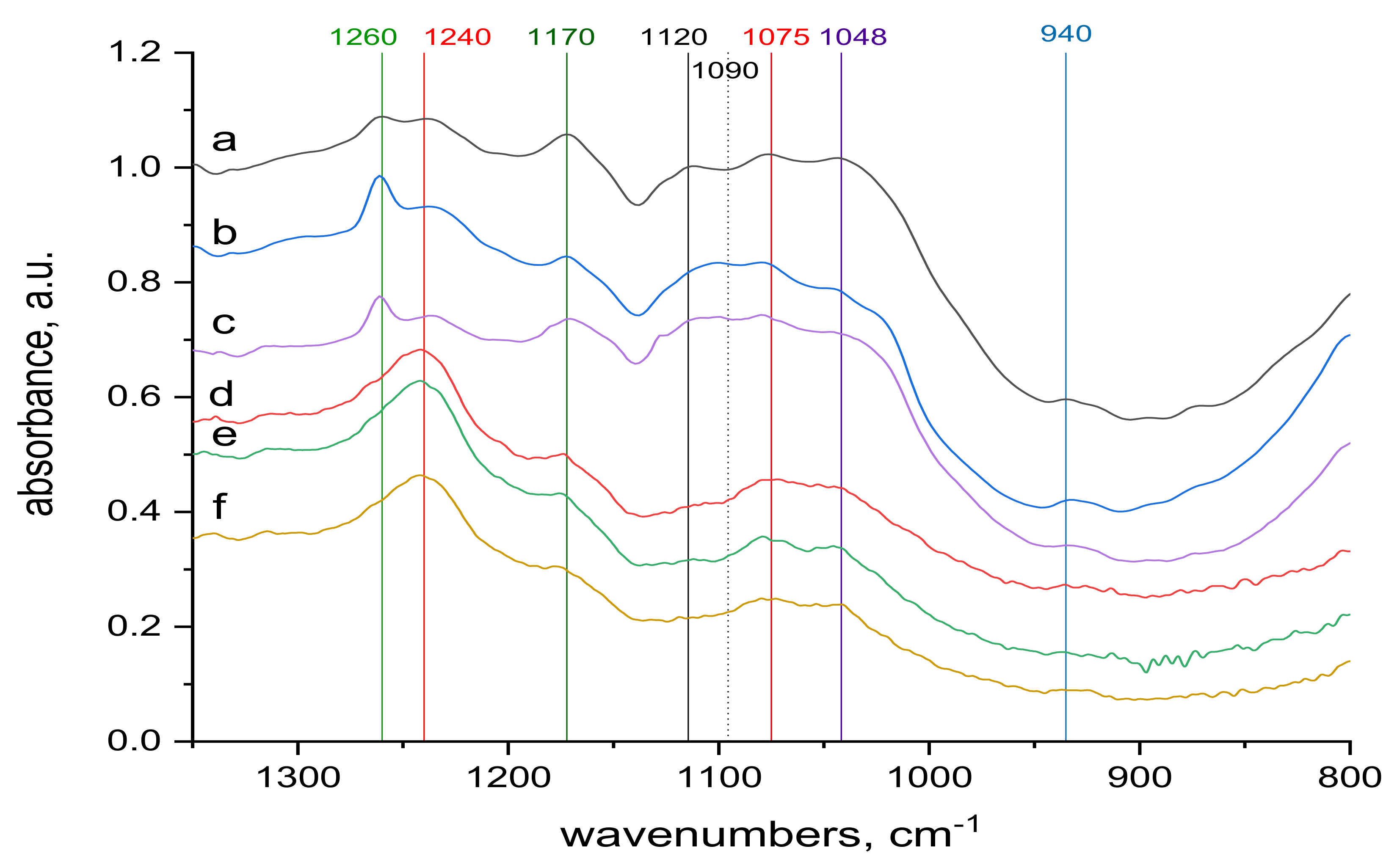
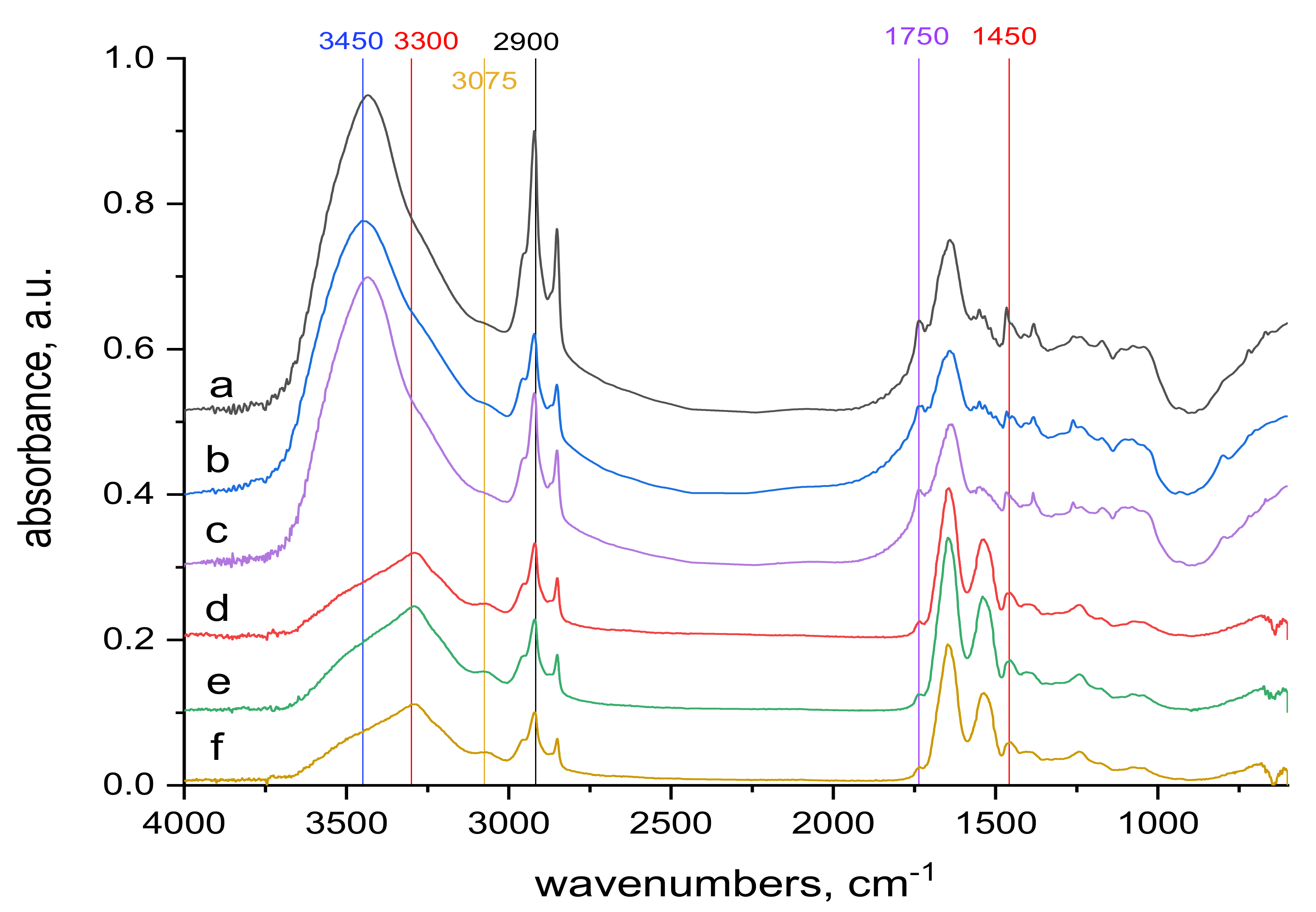
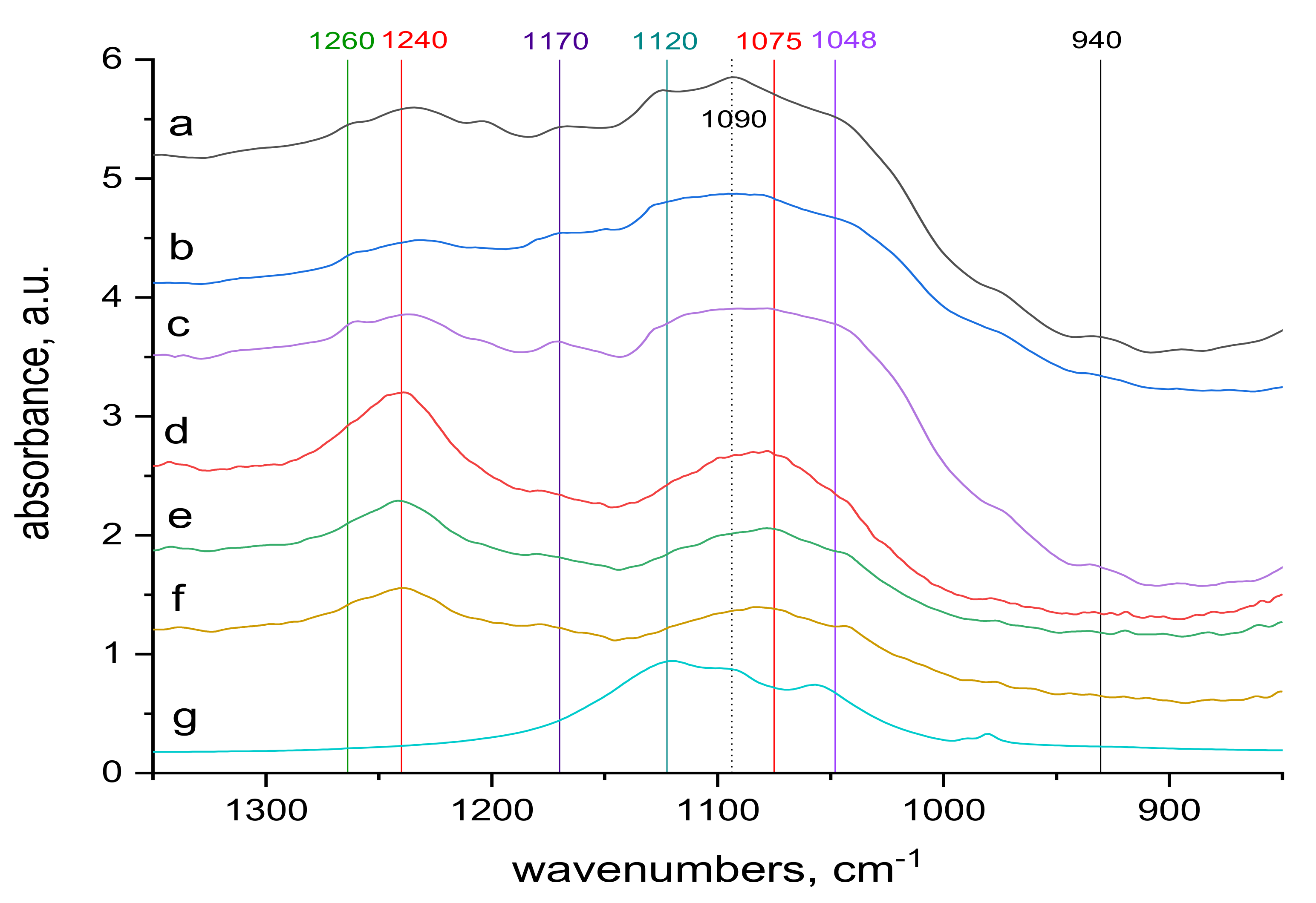
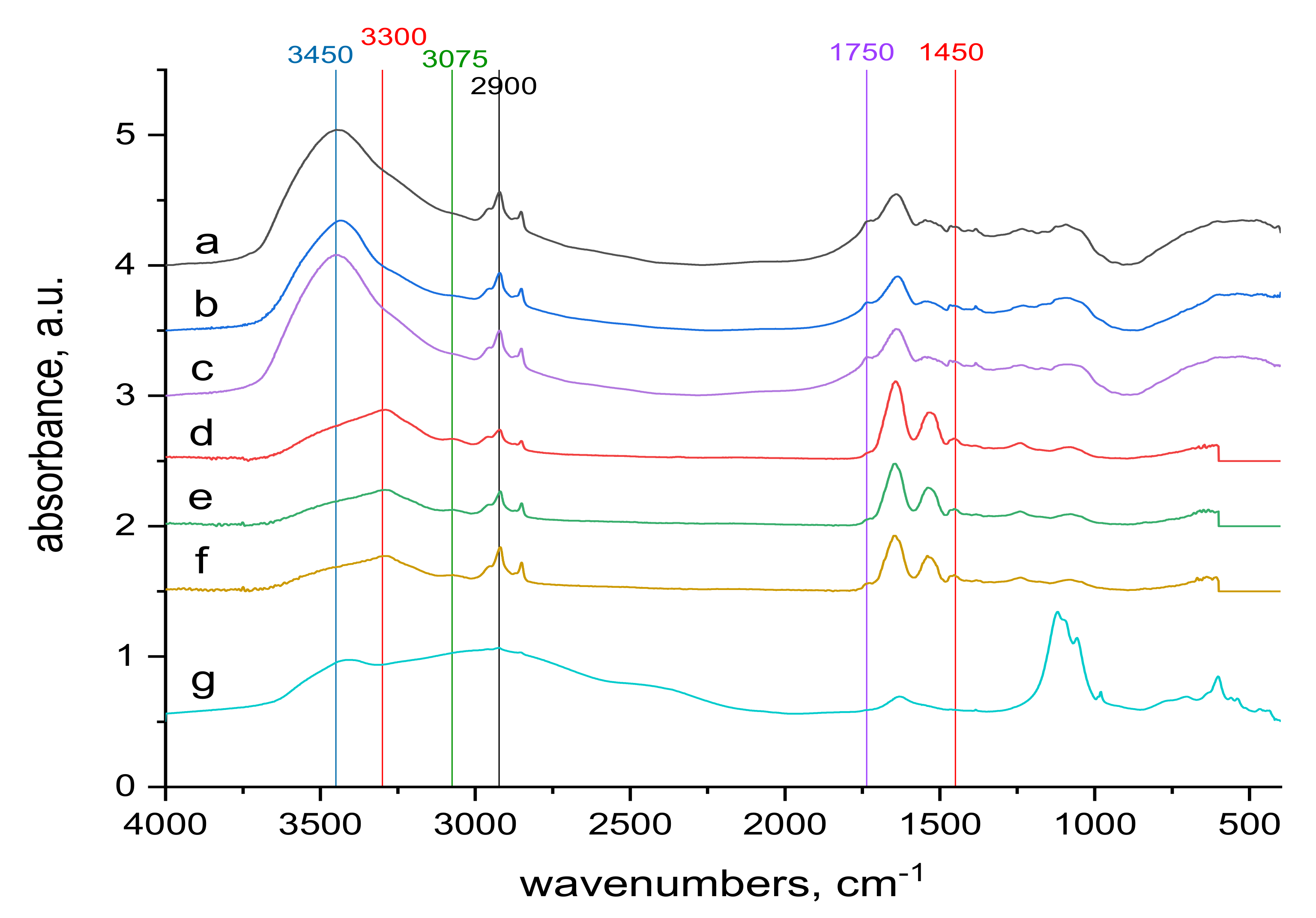
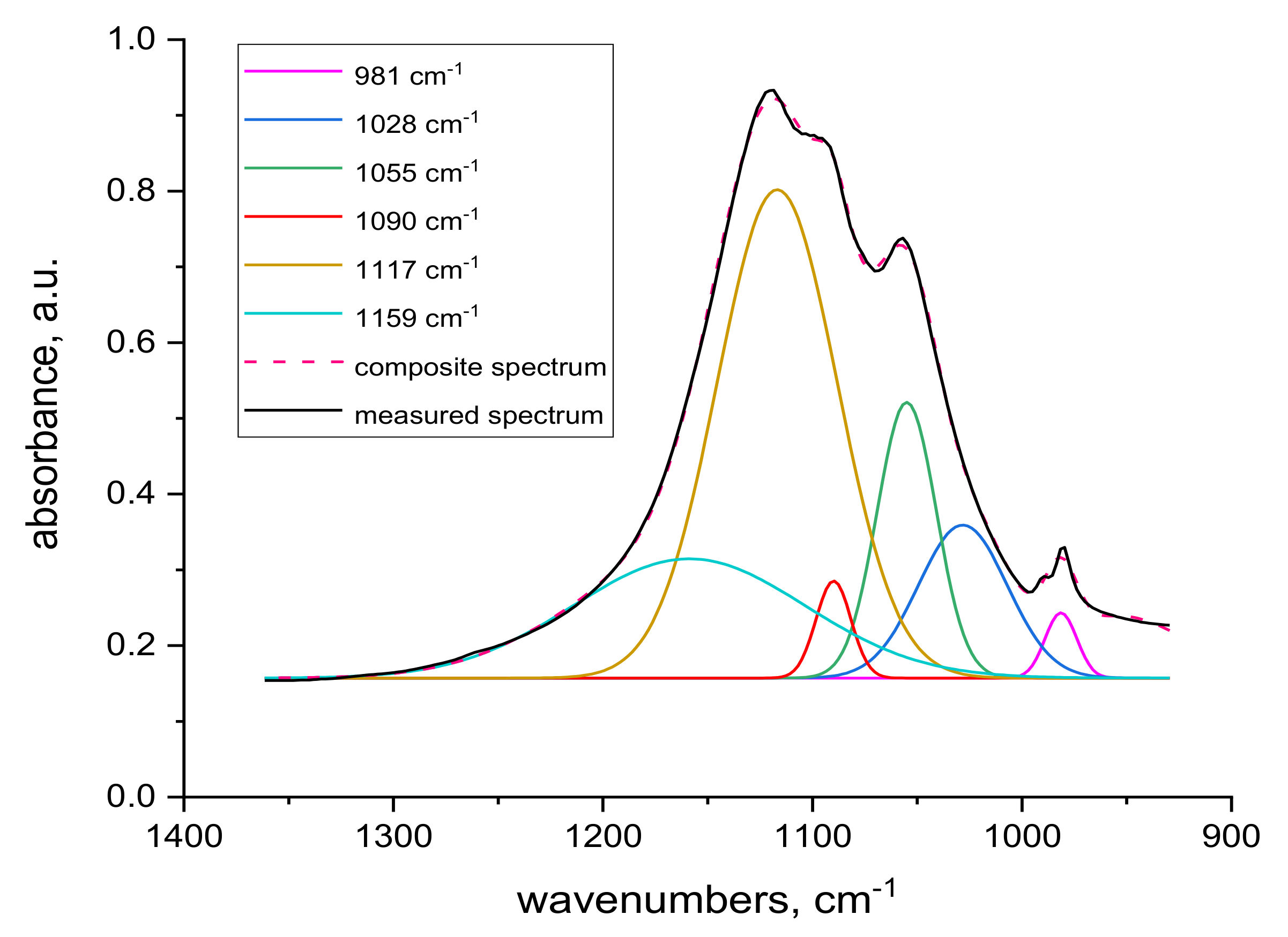
2.4. FTIR Spectra Measurements
3. Materials and Methods
3.1. Chemicals
3.2. Preparation of Adsorbent
3.3. Procedure of PZC and IEP Estimation
3.4. Spectral Measurements
3.5. Batch Sorption Experiments
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bedemo, A.; Chandravanshi, B.S.; Zewge, F. Removal of trivalent chromium from aqueous solution using aluminum oxide hydroxide. SpringerPlus 2016, 5, 1288. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Park, J.; Kang, J.; Han, Y.-S.; Jeong, H.Y. Removal of hexavalent chromium using mackinawite (FeS)-coated sand. J. Hazard. Mater. 2018, 360, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Dong, K.; Liu, Q.; Wei, G.; Hu, T.; Yao, J.; Zhang, X.; Gao, T. Mussel-inspired magnetic adsorbent: Adsorption/reduction treatment for the toxic Cr(VI) from simulated wastewater. J. Appl. Polym. Sci. 2018, 135, 46530. [Google Scholar] [CrossRef]
- Li, Q.; Liu, Y.; Yu, X.; Li, L.; Zhang, X.; Lu, Z.; Lin, J.; Yang, X.; Huang, Y. Removal of Cr(iii)/Cr(vi) from wastewater using defective porous boron nitride: A DFT study. Inorg. Chem. Front. 2018, 5, 1933–1940. [Google Scholar] [CrossRef]
- Zhu, F.; He, S.; Liu, T. Effect of pH, temperature and co-existing anions on the Removal of Cr(VI) in groundwater by green synthesized nZVI/Ni. Ecotox. Environ. Saf. 2018, 163, 544–550. [Google Scholar] [CrossRef]
- Chand, R.; Watari, T.; Inoue, K.; Luitel, H.N.; Torikai, T.; Yada, M. Chemical modification of carbonized wheat and barley straw using HNO3 and the adsorption of Cr(III). J. Hazard. Mater. 2009, 167, 319–324. [Google Scholar] [CrossRef]
- Lyubchik, S.I.; Lyubchik, A.I.; Galushko, O.L.; Tikhonova, L.P.; Vital, J.; Fonseca, I.M.; Lyubchik, S.B. Kinetics and thermodynamics of the Cr(III) adsorption on the activated carbon from co-mingled wastes. Colloid Surf. A 2004, 242, 151–158. [Google Scholar] [CrossRef]
- Su, M.; Fang, Y.; Li, B.; Yin, W.; Gu, J.; Liang, H.; Li, P.; Wu, J. Enhanced hexavalent chromium removal by activated carbon modified with micro-sized goethite using a facile impregnation method. Sci. Total. Environ. 2019, 647, 47–56. [Google Scholar] [CrossRef]
- Choi, K.; Lee, S.; Park, J.O.; Park, J.-A.; Cho, S.-H.; Lee, S.Y.; Lee, J.H.; Choi, J.-W. Chromium removal from aqueous solution by a PEI-silica nanocomposite. Sci. Rep. UK 2018, 8, 1438. [Google Scholar] [CrossRef]
- Mortazavian, S.; An, H.; Chun, D.; Moon, J. Activated carbon impregnated by zero-valent iron nanoparticles (AC/nZVI) optimized for simultaneous adsorption and reduction of aqueous hexavalent chromium: Material characterizations and kinetic studies. Chem. Eng. J. 2018, 353, 781–795. [Google Scholar] [CrossRef]
- Yang, Y.; Chen, N.; Feng, C.; Li, M.; Gao, Y. Chromium removal using a magnetic corncob biochar/polypyrrole composite by adsorption combined with reduction: Reaction pathway and contribution degree. Colloids Surf. A 2018, 556, 201–209. [Google Scholar] [CrossRef]
- Tyagi, U.; Khandegar, V. Biosorption potential of Vetiveria zizanioides for the Removal of chromium(VI) from synthetic wastewater. J. Hazard. Toxic. Radioactive Waste 2018, 22, 04018014. [Google Scholar] [CrossRef]
- Surendran, G.; Baral, S.S. Biosorption of Cr(VI) from wastewater using Sorghastrum Nutans, L. Nash. Chem. Ecol. 2018, 34, 762–785. [Google Scholar] [CrossRef]
- Vilardi, G.; Ochando-Pulido, J.M.; Verdone, N.; Stoller, M.; Di Palma, L. On the removal of hexavalent chromium by olive stones coated by iron-based nanoparticles: Equilibrium study and chromium recovery. J. Clean. Prod. 2018, 190, 200–210. [Google Scholar] [CrossRef]
- Quiton, K.G.; Doma, B., Jr.; Futalan, C.M.; Wan, M.-W. Removal of chromium(VI) and zinc(II) from aqueous solution using kaolin-supported bacterial biofilms of Gram-negative E. coli and Gram-positive Staphylococcus epidermidis. Sustain. Environ. Res. 2018, 28, 206–213. [Google Scholar] [CrossRef]
- Xiao, R.; Wang, J.J.; Li, R.; Park, J.; Meng, Y.; Zhou, B.; Pensky, S.; Zhang, Z. Enhanced sorption of hexavalent chromium [Cr(VI)] from aqueous solutions by diluted sulfuric acid-assisted MgO-coated biochar composite. Chemosphere 2018, 208, 408–416. [Google Scholar] [CrossRef]
- Samuel, M.S.; Shah, S.S.; Subramaniyan, V.; Qureshi, T.; Bhattacharya, J.; Pradeep Singh, N.D. Preparation of graphene oxide/chitosan/ferrite nanocomposite for Chromium(VI) removal from aqueous solution. Int. J. Biol. Macromol. 2018, 119, 540–547. [Google Scholar] [CrossRef]
- Balkaya, N.; Bektas, N. Chromium(VI) sorption from dilute aqueous solutions using wool. Desalin. Water Treat. 2009, 3, 43–49. [Google Scholar] [CrossRef]
- Balköse, D.; Baltacioǧlu, H. Adsorption of heavy metal cations from aqueous solutions by wool fibers. J. Chem. Technol. Biot. 1992, 54, 393–397. [Google Scholar]
- Ghosh, A.; Collie, S.R. Keratinous materials as novel absorbent systems for toxic pollutants. Defence Sci. J. 2014, 64, 209–221. [Google Scholar] [CrossRef]
- Porubská, M.; Hanzlíková, Z.; Braniša, J.; Kleinová, A.; Hybler, P.; Fülöp, M.; Ondruška, J.; Jomová, K. The effect of electron beam on sheep wool. Polym. Degrad. Stabil. 2015, 111, 151–158. [Google Scholar] [CrossRef]
- Hanzlíková, Z.; Lawson, M.K.; Hybler, P.; Fülöp, M.; Porubská, M. Time-Dependent Variations in Structure of Sheep Wool Irradiated by Electron Beam. Adv. Mater. Sci. Eng. 2017, 2017. [Google Scholar] [CrossRef]
- Hanzlíková, Z.; Braniša, J.; Hybler, P.; Šprinclová, I.; Jomová, K.; Porubská, M. Sorption properties of sheep wool irradiated by accelerated electron beam. Chem. Pap. 2016, 70, 1299–1308. [Google Scholar] [CrossRef]
- Hanzlíková, Z.; Braniša, J.; Jomová, K.; Fülöp, M.; Hybler, P.; Porubská, M. Electron beam irradiated sheep wool – Prospective sorbent for heavy metals in wastewater. Sep. Purif. Technol. 2018, 193, 345–350. [Google Scholar] [CrossRef]
- Porubská, M.; Kleinová, A.; Hybler, P.; Braniša, J. Why Natural or Electron Irradiated Sheep Wool Show Anomalous Sorption of Higher Concentrations of Copper(II). Molecules 2018, 23, 3180. [Google Scholar] [CrossRef]
- Braniša, J.; Jomová, K.; Kovalčíková, R.; Hybler, P.; Porubská, M. Role of Post-Exposure Time in Co(II) Sorption of Higher Concentrations on Electron Irradiated Sheep Wool. Molecules 2019, 24, 2639. [Google Scholar] [CrossRef]
- Kosmulski, M. IEP as a parameter characterizing the pH-dependent surface charging of materials other than metal oxides. Adv. Colloid. Interfac 2012, 171–172, 77–86. [Google Scholar] [CrossRef]
- Nacke, H.; Gonçalves, A.C.; Coelho, G.F.; Schwantes, D.; Compagnolo, M.A.; Völz Leismann, E.A.; Conradi, J.É.; Miola, J.A. Removal of Cd (II) from water using the waste of jatropa fruit (Jatropha curcas L.). Appl. Water. Sci. 2017, 7, 3207–3222. [Google Scholar] [CrossRef]
- Capablanca, J.S.; Watt, L.C. Factors Affecting the Zeta Potential at Wool Fiber Surfaces. Text. Res. J. 1986, 56, 49–55. [Google Scholar] [CrossRef]
- Harris, M. The isoelectric point of wool. Bur. Stand. J. Res. 1932, 8, 779–785. [Google Scholar] [CrossRef]
- Pušić, T.; Boban, A.; Dekanić, T.; Soljačić, I. The sorption ability of textile fibres. Vlakna a Textil 2011, 18, 7–15. [Google Scholar]
- Hamada, Y.Z.; Makoni, N.; Hamada, H. Three Very Different UV-VIS Absorption Spectra of Three Different Transition Metals Found in Biological Solutions. Electron. J. Biol. 2016, 2, 6–9. [Google Scholar]
- Porterfield, W.W. Inorganic Chemistry: A Unified Approach, 2nd ed.; Academic Press: London, UK, 1993; p. 921. ISBN 0-12-562981-8. [Google Scholar]
- Drljaca, A.; Spiccia, L. Early stages of the hydrolysis of Chromium(III) in aqueous solution-X. Kinetics of formation of trimer from monomer and dimer. Polyhedron 1995, 14, 1653–1660. [Google Scholar] [CrossRef]
- Hiroishi, D.; Matsuura, C.; Ishigure, K. Hydrolysis of chromium(III) ion and solubility of chromium(III) oxide in high temperature water. Mineral. Mag. 1998, 62, 626–627. [Google Scholar] [CrossRef]
- Wang, B.; Yang, W.; McKittrick, J.; Meyer, M.A. Keratin: Structure, mechanical properties, occurrence in biological organisms, and efforts at bioinspiration. Prog. Mater. Sci. 2016, 76, 229–318. [Google Scholar] [CrossRef]
- Šima, J.; Koman, M.; Kotočová, A.; Segľa, P.; Tatarko, M.; Valigura, D. Anorganická Chémia, 2nd ed.; STU: Bratislava, Slovakia, 2011; pp. 375–378. ISBN 978-80-227-3579-7. (In Slovak) [Google Scholar]
- Thermo Scientific. Infrared Correlation Chart. 2015. Available online: http://ftirsearch.com (accessed on 2 May 2016).
- Milata, V.; Segľa, P.; Brezová, V.; GatiaL, A.; Kováčik, V.; Miglierini, M.; Stankovský, Š.; Šíma, J. Aplikovaná Molekulová Spektroskopia; STU: Bratislava, Slovakia, 2008; pp. 543–558. ISBN 978-80-227-2960-4. [Google Scholar]
- Lyklema, J. Fundamentals of Interface and Colloid Science. In Solid-Liquid Interfaces, 1st ed.; Academic Press: San Diego, CA, USA, 1995; Volume 3, pp. 101–109. ISBN 0-12-460524-9. [Google Scholar]
- Debajyoti, D. Biochemistry, 1st ed.; Academic Publishers (Elsevier): London, UK, 1978; 634p, ISBN 9788187504825. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Braniša, J.; Kleinová, A.; Jomová, K.; Malá, R.; Morgunov, V.; Porubská, M. Some Properties of Electron Beam-Irradiated Sheep Wool Linked to Cr(III) Sorption. Molecules 2019, 24, 4401. https://doi.org/10.3390/molecules24234401
Braniša J, Kleinová A, Jomová K, Malá R, Morgunov V, Porubská M. Some Properties of Electron Beam-Irradiated Sheep Wool Linked to Cr(III) Sorption. Molecules. 2019; 24(23):4401. https://doi.org/10.3390/molecules24234401
Chicago/Turabian StyleBraniša, Jana, Angela Kleinová, Klaudia Jomová, Radka Malá, Volodymyr Morgunov, and Mária Porubská. 2019. "Some Properties of Electron Beam-Irradiated Sheep Wool Linked to Cr(III) Sorption" Molecules 24, no. 23: 4401. https://doi.org/10.3390/molecules24234401
APA StyleBraniša, J., Kleinová, A., Jomová, K., Malá, R., Morgunov, V., & Porubská, M. (2019). Some Properties of Electron Beam-Irradiated Sheep Wool Linked to Cr(III) Sorption. Molecules, 24(23), 4401. https://doi.org/10.3390/molecules24234401







