Characterization of Composition and Antifungal Properties of Leaf Secondary Metabolites from Thirteen Cultivars of Chrysanthemum morifolium Ramat
Abstract
:1. Introduction
2. Results and Discussion
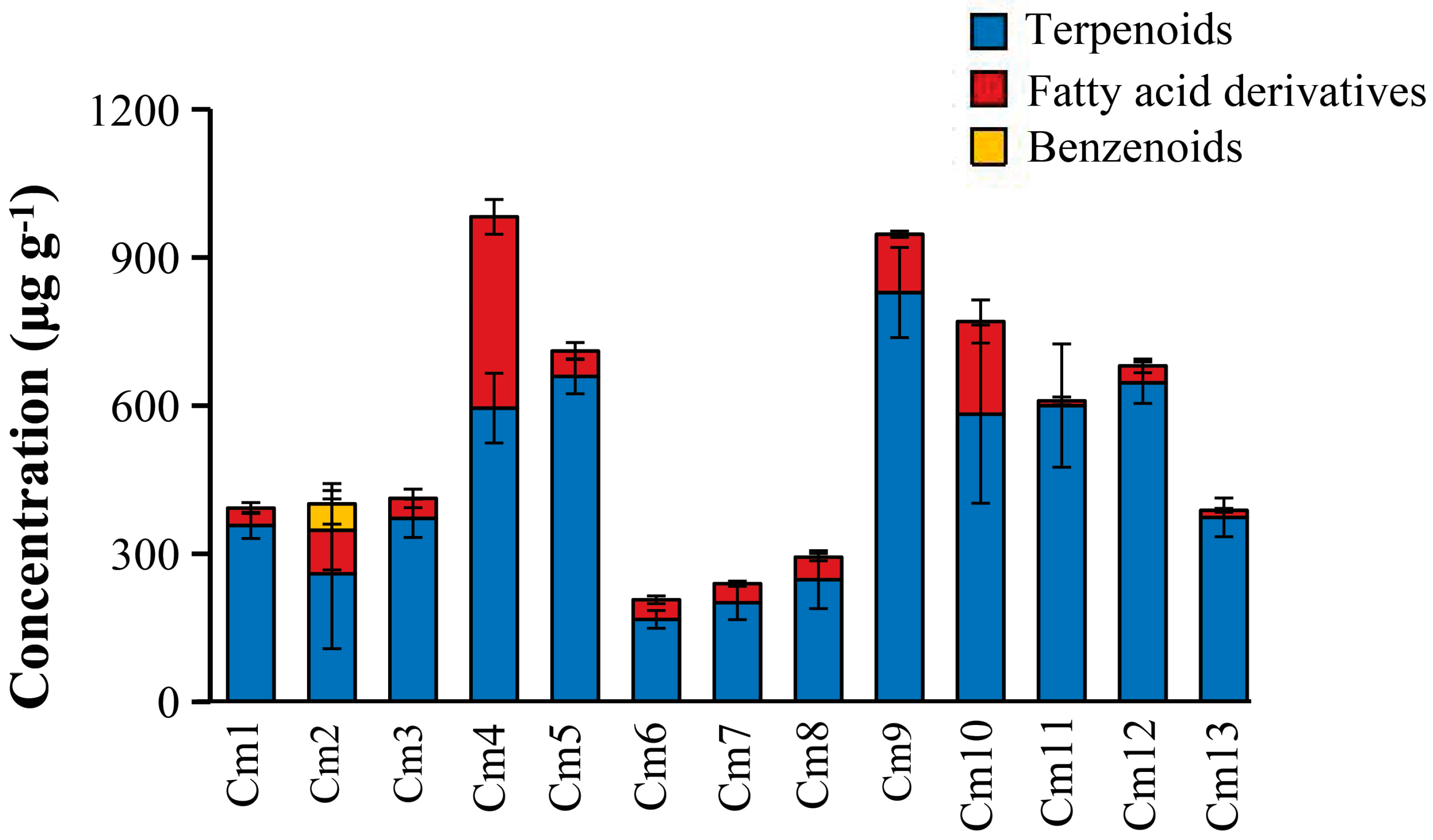
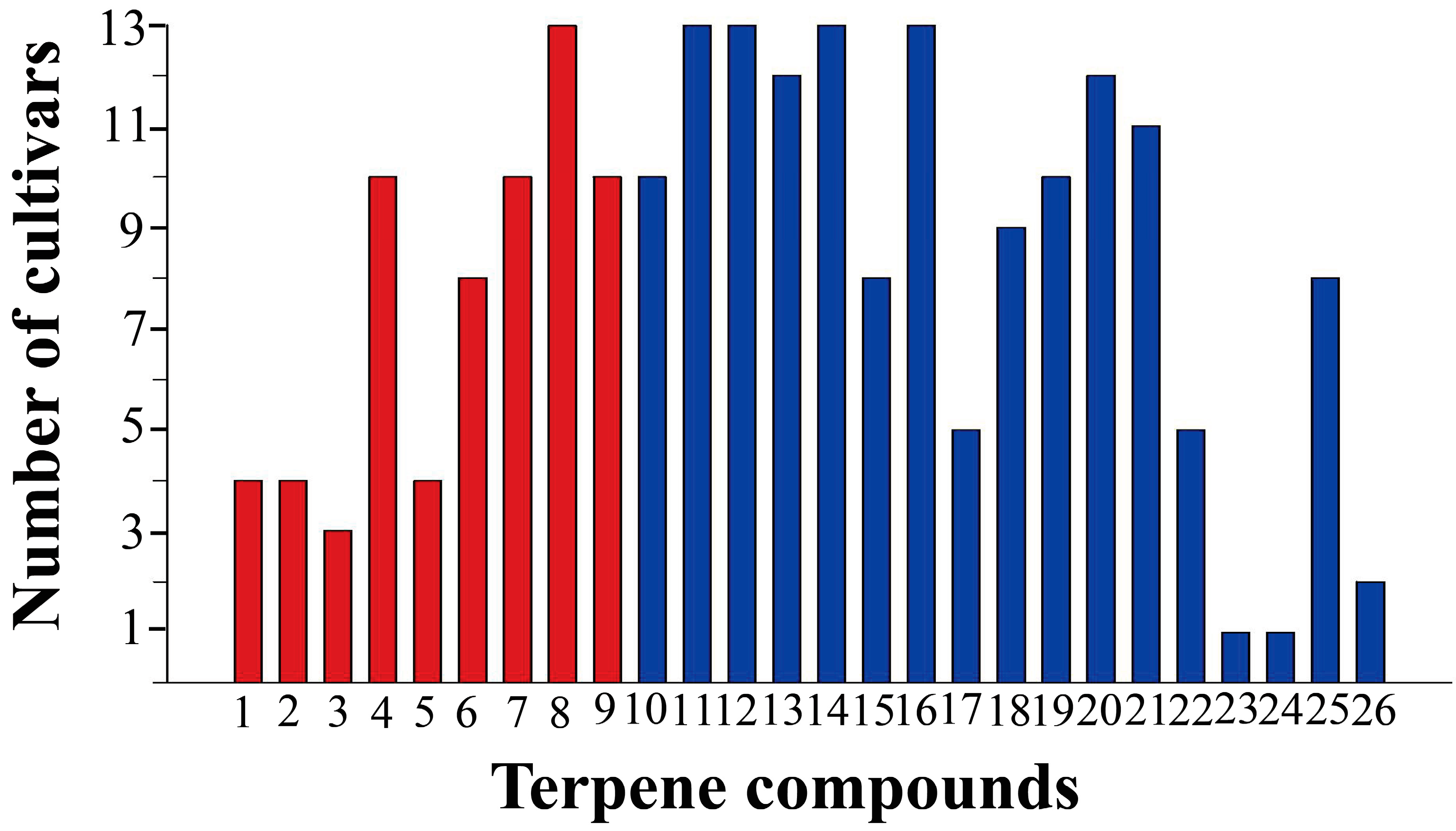
2.1. Chemical Composition of Apolar Secondary Metabolites of C. morifolium Leaves
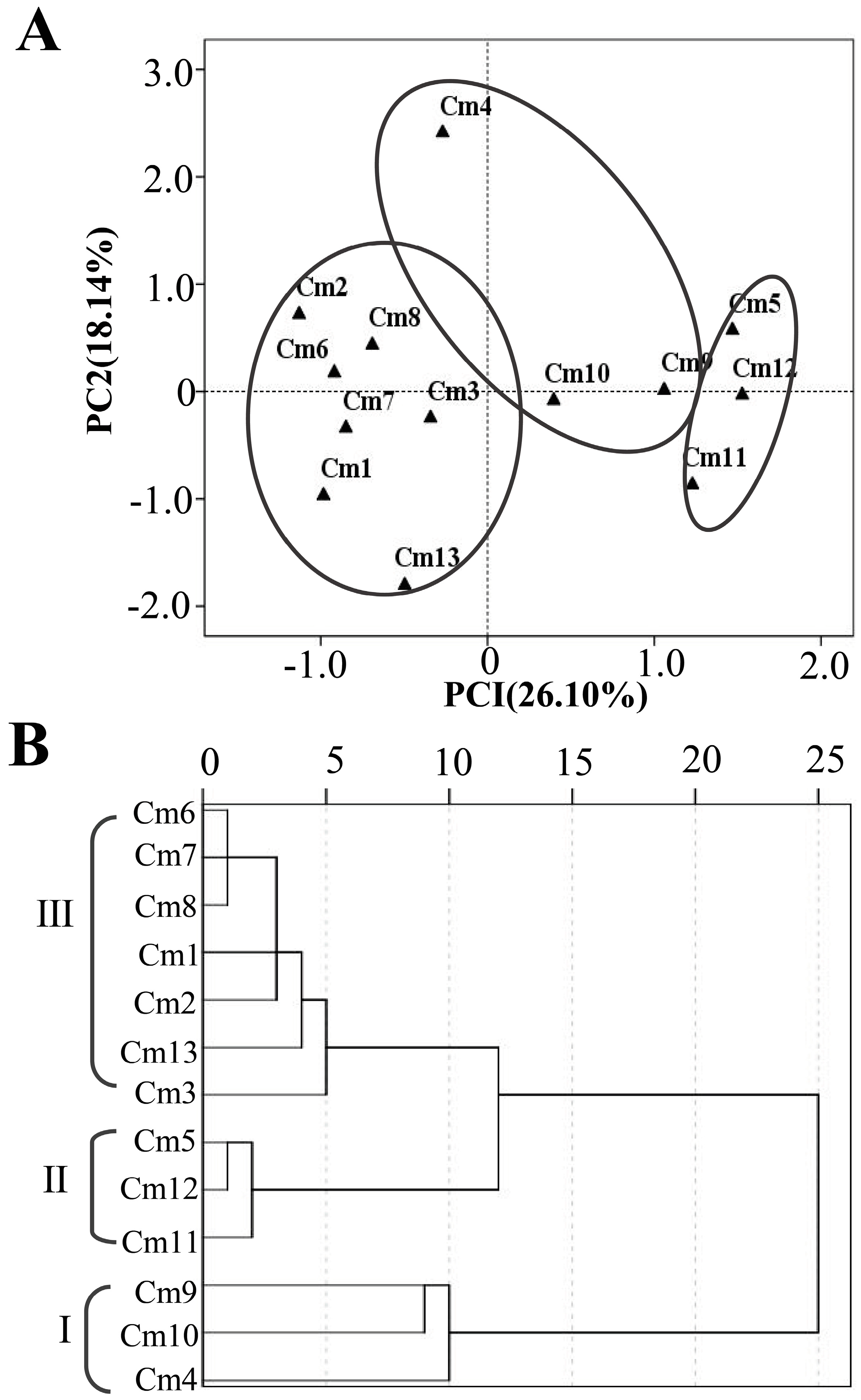
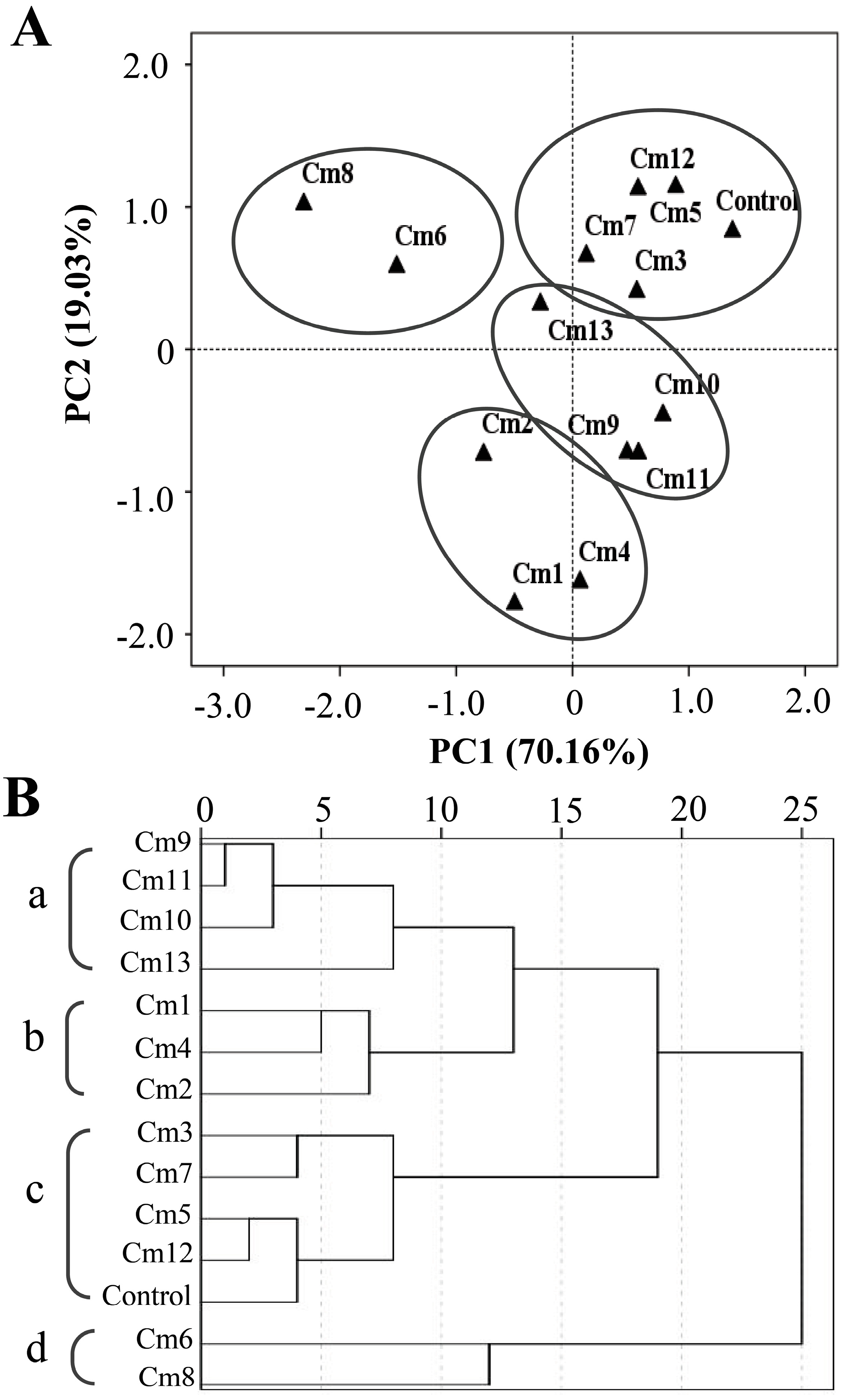
2.2. PCA and HCA Analysis Based on the Terpenoid Contents
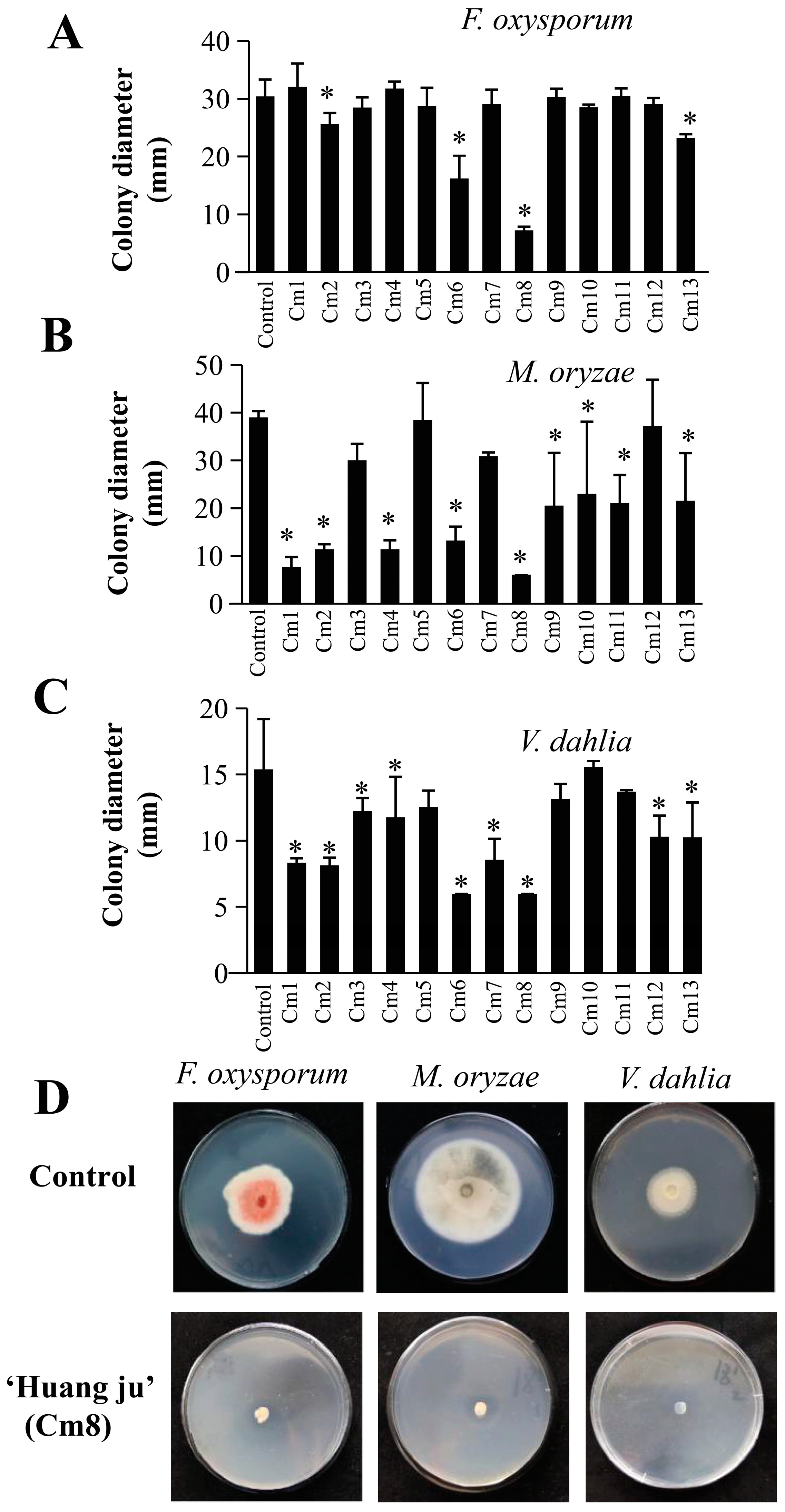
2.3. Antifungal Activity of Apolar Secondary Metabolites from C. morifolium Leaves
2.4. PCA and HCA Analysis Based on the Antifungal Effect
2.5. Conclusions
3. Materials and Methods
3.1. Plant Material
3.2. Organic Extraction of Leaves of C. morifolium
3.3. GC-MS Analysis and Identification of Extract Constituents
3.4. Assessment of Antifungal Activity
3.5. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Zhao, N.; Wang, G.; Norris, A.; Chen, X.; Chen, F. Studying plant secondary metabolism in the age of genomics. Crit. Rev. Plant Sci. 2013, 32, 369–382. [Google Scholar] [CrossRef]
- Chen, F.; Liu, C.J.; Tschaplinski, T.J.; Zhao, N. Genomics of secondary metabolism in Populus. Interactions with biotic and abiotic environments. Crit. Rev. Plant Sci. 2009, 28, 375–392. [Google Scholar] [CrossRef]
- McCaskill, D.; Croteau, R. Prospects for the bioengineering of isoprenoid biosynthesis. Adv. Biochem. Eng. Biotechnol. 1997, 55, 107–146. [Google Scholar] [PubMed]
- Aljbory, Z.; Chen, M.S. Indirect plant defense against insect herbivores a review. Insect Sci. 2018, 25, 2–23. [Google Scholar] [CrossRef] [PubMed]
- Ashour, H.M. Antibacterial, antifungal, and anticancer activities of volatile oils and extracts from stems, leaves, and flowers of Eucalyptus sideroxylon and Eucalyptus torquata. Cancer Biol Ther. 2008, 7, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Daíse, L.L.; Alviano, D.S.; Alviano, C.S.; Kolodziejczyk, P.P. Screening of chemical composition, antimicrobial and antioxidant activities of Artemisia essential oils. Phytochemistry 2008, 69, 1732–1738. [Google Scholar]
- Griffin, S.G.; Wyllie, S.G.; Markham, J.L.; Leach, D.N. The role of structure and molecular properties of terpenoids in determining their antimicrobial activity. Flavour Fragr. J. 2015, 14, 322–332. [Google Scholar] [CrossRef]
- Shen, Q.; Zhang, L.; Liao, Z.; Wang, S.; Yan, T.; Shi, P.; Liu, M.; Fu, X.; Pan, Q.; Wang, Y.; et al. The Genome of Artemisia annua Provides insight into the evolution of Asteraceae family and artemisinin biosynthesis. Mol. Plant 2018, 11, 776–788. [Google Scholar] [CrossRef]
- Kim, S.; Park, M.; Yeom, S.I.; Kim, Y.M.; Lee, J.M.; Lee, H.A.; Seo, E.; Choi, J.; Cheong, K.; Kim, K.T. Genome sequence of the hot pepper provides insights into the evolution of pungency in Capsicum species. Nat. Genet. 2014, 46, 270–276. [Google Scholar] [CrossRef]
- Xu, H.; Moghe, G.D.; Wiegert-Rininger, K.; Schilmiller, A.L.; Barry, C.S.; Last, R.L.; Pichersky, E. Coexpression analysis identifies two oxidoreductases involved in the biosynthesis of the monoterpene acid moiety of natural pyrethrin insecticides in Tanacetum cinerariifolium. Plant Physiol. 2018, 176, 524–537. [Google Scholar] [CrossRef]
- Magallon, S.; Herendeen, C.P.S. Phylogenetic pattern, diversity, and diversification of Eudicots. Ann. Mo. Bot. Garden 1999, 86, 297–372. [Google Scholar] [CrossRef]
- Zhu, S.; Yang, Y.; Yu, H.; Ying, Y.; Zou, G. Chemical composition and antimicrobial activity of the essential oils of Chrysanthemum indicum. J. Ethnopharmacol 2005, 96, 151–158. [Google Scholar]
- Woo, K.; Yu, J.; Hwang, I.; Lee, Y.; Lee, C.; Yoon, H.; Lee, J.; Jeong, H. Antioxidative activity of volatile compounds in flower of Chrysanthemum indicum, C. morifolium, and C. zawadskii. J. Korean Soc. Food Sci. Nutr. 2008, 37, 805–809. [Google Scholar] [CrossRef]
- Lee, H.J.; Hwang, Y.I.; Park, E.J.; Choi, S.U. Antihepatotoxic and antigenotoxic effects of herb tea composed of Chrysanthemum morifolium ramat. J. Korean Soc. Food Sci. Nutr. 2011, 40, 78–83. [Google Scholar] [CrossRef]
- Han, X.B.; Zhao, J.; Cao, J.M.; Zhang, C.S. Essential oil of Chrysanthemum indicum L.: Potential biocontrol agent against plant pathogen Phytophthora nicotianae. Environ. Sci. Pollut. Res. Int. 2019, 26, 7013–7023. [Google Scholar] [CrossRef] [PubMed]
- Hosni, K.; Hassen, I.; Sebei, H.; Casabianca, H. Secondary metabolites from Chrysanthemum coronarium (Garland) flowerheads: Chemical composition and biological activities. Ind. Crops Prod. 2013, 44, 263–271. [Google Scholar] [CrossRef]
- Kuang, C.L.; Lv, D.; Shen, G.H.; Li, S.S.; Luo, Q.Y.; Zhang, Z.Q. Chemical composition and antimicrobial activities of volatile oil extracted from Chrysanthemum morifolium ramat. J. Food Sci. Technol. 2018, 55, 2786–2794. [Google Scholar] [CrossRef]
- Zhang, C.; Qin, M.J.; Shu, P.; Hong, J.L.; Lü, L.; He, D.X. Chemical variations of the essential oils in flower heads of Chrysanthemum indicum L. from China. Chem. Biodivers. 2011, 7, 2951–2962. [Google Scholar] [CrossRef]
- Shafaghat, A.; Sadeghi, H.; Oji, K. Composition and antibacterial activity of essential oils from leaf, stem and root of Chrysanthemum parthenium (L.) bernh. from iran. Nat. Prod. Commun. 2009, 4, 859–860. [Google Scholar] [CrossRef]
- Chen, F.; Tholl, D.; Bohlmann, J.; Pichersky, E. The family of terpene synthases in plants: A mid-size familyof genes for specialized metabolism that is highly diversified throughout the kingdom. Plant J. 2011, 66, 212–229. [Google Scholar] [CrossRef]
- Jiang, Y.; Ownley, B.; Chen, F. Terpenoids from Weedy ricefield Flatsedge (Cyperus iria L.) are developmentally regulated and stress-Induced, and have antifungal properties. Molecules 2018, 23, 3149. [Google Scholar] [CrossRef] [PubMed]
- Pattnaik, S.; Subramanyam, V.R.; Bapaji, M.; Kole, C.R. Antibacterial and antifungal activity of aromatic constituents of essential oils. Microbios 1997, 89, 39–46. [Google Scholar] [PubMed]
- Scortichini, M.; Rossi, M.P. Preliminary in vitro evaluation of the antimicrobialactivity of terpenes and terpenoids towards Erwinia amylovora (Burrill). J. Appl. Bacteriol. 2010, 71, 109–112. [Google Scholar] [CrossRef]
- Núñez, Y.O.; Salabarria, I.S.; Collado, I.G.; Hernández-Galán, R. Antifungal activity of extracts and terpene constituents of aerial parts of Juniperus lucaiyana. Rev. Latinoam. Quím. 2010, 38, 145–152. [Google Scholar]
- de Macêdo, D.G.; Souza1, A.M.M.; Morais-Braga, B.M.F.; Coutinho, M.D.H.; dos Santos, L.T.; da Cruz, P.R.; da Costa, M.G.; Rodrigues, G.F.F.; Quintans-junior, J.L.; da Silva Almeida, G.J.R.; et al. Effect of seasonality on chemical profile and antifungal activity of essential oil isolated from leaves Psidium salutare (Kunth) O. Berg. PeerJ 2018, 6, e5476. [Google Scholar]
- Li, P.; Chen, J.; Li, Y.; Zhang, K.; Wang, H.L. Possible mechanisms of control of Fusarium wilt of cut chrysanthemum by Phanerochaete chrysosporium in continuous cropping fields: A case study. Sci. Rep. 2017, 7, 15994. [Google Scholar] [CrossRef]
- Matić, S.; Gilardi, G.; Gullino, M.L.; Garibaldi, A. Evidence for an expanded host range of Fusarium oxysporum f. sp. chrysanthemi. J. Plant Pathol. 2018, 100, 97–104. [Google Scholar]
- Terras, F.R.; Schoofs, H.M.; de Bolle, M.F.; van Leuven, F.; Rees, S.B.; Vanderleyden, J.; Cammue, B.P.; Broekaert, W.F. Analysis of two novel classes of plant antifungal proteins from radish (Raphanus sativus L.) seeds. J. Biol. Chem. 1992, 267, 15301–15309. [Google Scholar]
- Terras, F.R.; Eggermont, K.; Kovaleva, V.; Raikhel, N.V.; Osborn, R.W.; Kester, A.; Rees, S.B.; Torrekens, S.; van Leuven, F.; Vanderleyden, J.; et al. Small cysteine-rich antifungal proteins from radish: Their role in host defense. Plant Cell. 1995, 7, 573–588. [Google Scholar]
- Gao, A.G.; Hakimi, S.M.; Mittanck, C.A.; Wu, Y.; Woerner, B.M.; Stark, D.M.; Shah, D.M.; Liang, J.; Rommens, C.M. Fungal pathogen protection in potato by expression of a plant defensin peptide. Nat. Biotechnol. 2000, 18, 1307–1310. [Google Scholar] [CrossRef]
- Spelbrink, R.G.; Dilmac, N.; Allen, A.; Smith, T.J.; Shah, D.M.; Hockerman, G.H. Differential antifungal and calcium channel-blocking activity among structurally related plant defensins. Plant Physiol. 2004, 135, 2055–2067. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Liu, Y.F.; Song, A.P.; Dong, G.Q.; Zhao, H.B.; Sun, W.; Ramakrishnan, S.; Wang, Y.; Wang, S.B.; Li, T.Z.; et al. The Chrysanthemum nankingense genome provides insights into the evolution and diversification of Chrysanthemum flowers and medicinal traits. Mol. Plant 2018, 11, 1482–1491. [Google Scholar] [CrossRef] [PubMed]
- Leung, H.; Borromeo, E.S.; Bernardo, M.A.; Notteghem, J.L. Genetic Analysis of Virulence in the Rice Blast Fungus Magnaporthe grisea. Phytopathology 1988, 78, 1227–1233. [Google Scholar] [CrossRef]
- Xu, L.; Zhu, L.; Tu, L.; Guo, X.; Long, L.; Sun, L.; Gao, W.; Zhang, X. Differential Gene Expression in Cotton Defence Response to Verticillium dahliae by SSH. J. Phytopathol. 2011, 159, 606–615. [Google Scholar] [CrossRef]
- Bishop, P.; orochan, J.; Ownley, B.H.; Samples, T.J.; Windham, A.S.; Windham, M.T.; Trigiano, R.N. Resistance of Sclerotinia homeocarpa to iprodione, propiconazole, and thiophanate-methyl in Tennessee and northern Mississippi. Crop Sci. 2008, 48, 1615–1620. [Google Scholar] [CrossRef]
Sample Availability: Samples of the leaf extracts from 13 cultivars are available from the authors. |
| Code | Cultivar | Collection Locality |
|---|---|---|
| Cm1 | Chuju | Nanjing, Jiangsu province, China |
| Cm2 | Jin si huang ju | Nanjing, Jiangsu province, China |
| Cm3 | Hang bai ju | Nanjing, Jiangsu province, China |
| Cm4 | Xiao xiang ju | Nanjing, Jiangsu province, China |
| Cm5 | Da yang ju | Nanjing, Jiangsu province, China |
| Cm6 | Huang xiang li | Nanjing, Jiangsu province, China |
| Cm7 | Qi yue bai | Nanjing, Jiangsu province, China |
| Cm8 | Huang ju | Nanjing, Jiangsu province, China |
| Cm9 | Wan gong ju | Nanjing, Jiangsu province, China |
| Cm10 | Xiao huang ju | Nanjing, Jiangsu province, China |
| Cm11 | Su ju 9 | Nanjing, Jiangsu province, China |
| Cm12 | She yang hong xin ju | Nanjing, Jiangsu province, China |
| Cm13 | Bai xiang li | Nanjing, Jiangsu province, China |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xue, H.; Jiang, Y.; Zhao, H.; Köllner, T.G.; Chen, S.; Chen, F.; Chen, F. Characterization of Composition and Antifungal Properties of Leaf Secondary Metabolites from Thirteen Cultivars of Chrysanthemum morifolium Ramat. Molecules 2019, 24, 4202. https://doi.org/10.3390/molecules24234202
Xue H, Jiang Y, Zhao H, Köllner TG, Chen S, Chen F, Chen F. Characterization of Composition and Antifungal Properties of Leaf Secondary Metabolites from Thirteen Cultivars of Chrysanthemum morifolium Ramat. Molecules. 2019; 24(23):4202. https://doi.org/10.3390/molecules24234202
Chicago/Turabian StyleXue, Huanhuan, Yifan Jiang, Hongwei Zhao, Tobias G. Köllner, Sumei Chen, Fadi Chen, and Feng Chen. 2019. "Characterization of Composition and Antifungal Properties of Leaf Secondary Metabolites from Thirteen Cultivars of Chrysanthemum morifolium Ramat" Molecules 24, no. 23: 4202. https://doi.org/10.3390/molecules24234202
APA StyleXue, H., Jiang, Y., Zhao, H., Köllner, T. G., Chen, S., Chen, F., & Chen, F. (2019). Characterization of Composition and Antifungal Properties of Leaf Secondary Metabolites from Thirteen Cultivars of Chrysanthemum morifolium Ramat. Molecules, 24(23), 4202. https://doi.org/10.3390/molecules24234202








