The Synthesis, Characterization, Cytotoxic Activity Assessment and Structure-Activity Relationship of 4-Aryl-6-(2,5-dichlorothiophen-3-yl)-2-methoxypyridine-3-carbonitriles
Abstract
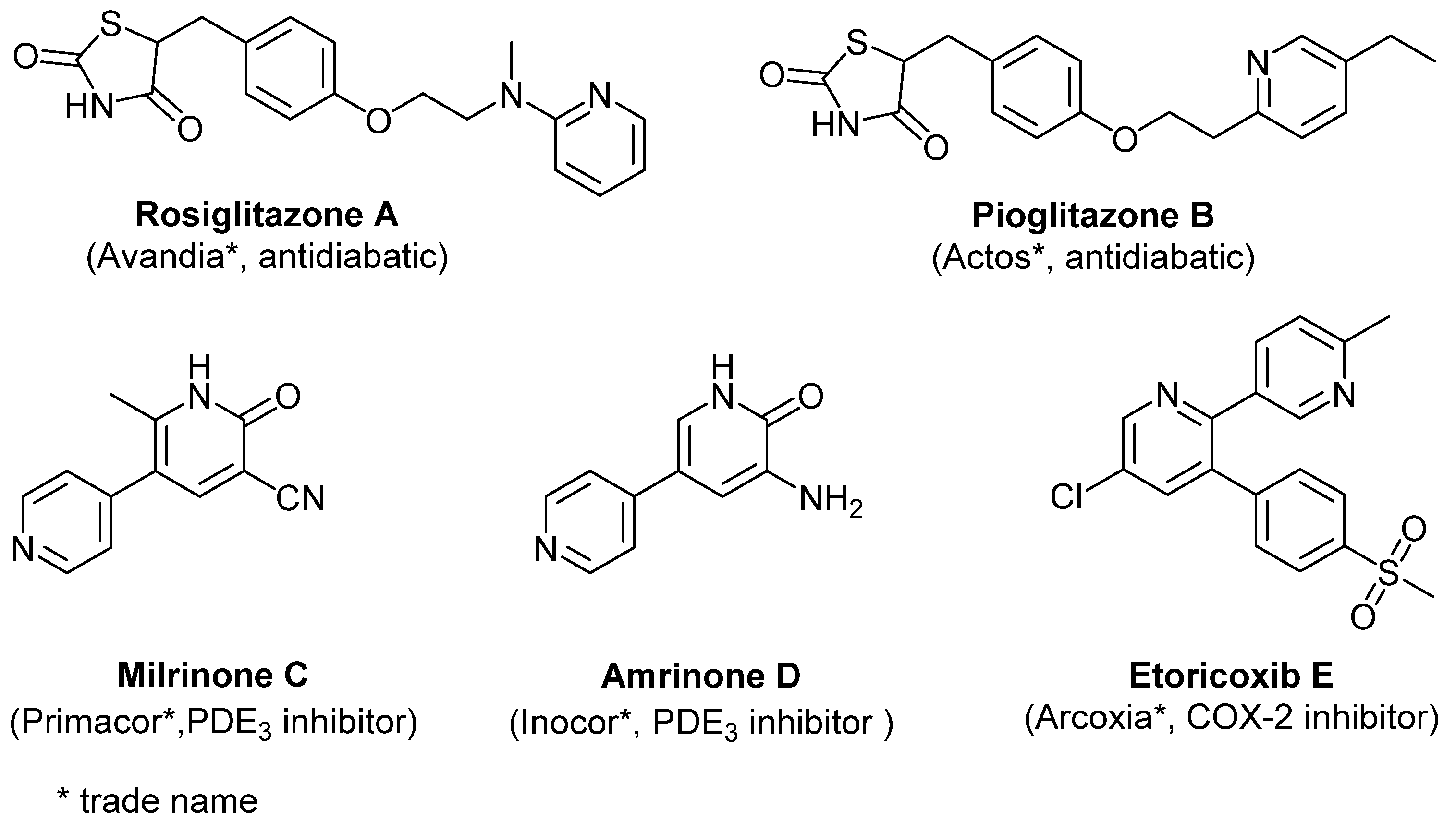
1. Introduction
2. Results and Discussion
2.1. Synthesis and Characterization
2.2. Cytotoxicity Properties
2.3. Structure-Activity Relationship (SAR)
3. Materials and Methods
3.1. Materials
3.2. Instrumentation
3.3. Synthesis
3.3.1. General procedure for the Synthesis 6-(2,5-dichlorothiophen-3-yl)-2-methoxy-4-(4-methoxyphenyl)pyridines (5a–i) and (6a–g)
3.3.2. Characterization Data for Products
3.4. Cell Culture and Maintenance
3.5. Cytotoxicity Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Fossa, P.; Menozzi, G.; Dorigo, P.; Floreani, M.; Mosti, L. Synthesis and pharmacological characterization of functionalized 2-pyridones structurally related to the cardiotonic agent milrinone. Bioorg. Med. Chem. 2003, 11, 4749–4759. [Google Scholar] [CrossRef]
- Kishi, T.; Nakahashi, K.; Ito, H.; Taniguchi, S.; Takaki, M. Effects Of Milrinone On Left Ventricular End-Systolic Pressure–Volume Relationship of Rat Hearts In Situ. Clin. Exp. Pharmacol. Physiol. 2001, 28, 737–742. [Google Scholar] [CrossRef] [PubMed]
- Kikura, M.; Levy, J.H. New cardiac drugs. Int. Anesthesiol. Clin. 1995, 33, 21–37. [Google Scholar] [CrossRef] [PubMed]
- Friesen, R.W.; Brideau, C.; Chan, C.C.; Charleson, S.; Deschênes, D.; Dubé, D.; Ethier, D.; Fortin, R.; Gauthier, J.Y.; Girard, Y.; et al. 2-Pyridinyl-3-(4-methylsulfonyl)phenylpyridines: Selective and orally active cyclooxygenase-2 inhibitors. Bioorg. Med. Chem. Lett. 1998, 8, 2777–2782. [Google Scholar] [CrossRef]
- Vieira De Almeida, M.; Vinicius de Nora Souza, M.; Rezende Barbosa, N.; Pittella Silva, F.; Wilson Amarante, G.; Helena Cardoso, S. Synthesis and Antimicrobial Activity of Pyridine Derivatives Substituted at C-2 and C-6 Positions. Lett. Drug Des. Discov. 2007, 4, 149–153. [Google Scholar] [CrossRef]
- Stevens, M.; Pannecouque, C.; De Clercq, E.; Balzarini, J. Inhibition of Human Immunodeficiency Virus by a New Class of Pyridine Oxide Derivatives. Antimicrob. Agents Chemother. 2003, 47, 2951–2957. [Google Scholar] [CrossRef]
- Xue, J.; Diao, J.; Cai, G.; Deng, L.; Zheng, B.; Yao, Y.; Song, Y. Antimalarial and Structural Studies of Pyridine-Containing Inhibitors of 1-Deoxyxylulose-5-phosphate Reductoisomerase. ACS Med. Chem. Lett. 2013, 4, 278–282. [Google Scholar] [CrossRef]
- Easmon, J.; Pürstinger, G.; Thies, K.-S.; Heinisch, G.; Hofmann, J. Synthesis, Structure–Activity Relationships, and Antitumor Studies of 2-Benzoxazolyl Hydrazones Derived from Alpha-(N)-acyl Heteroaromatics. J. Med. Chem. 2006, 49, 6343–6350. [Google Scholar] [CrossRef]
- Altaf, A.A.; Shahzad, A.; Gul, Z.; Rasool, N.; Badshah, A.; Lal, B.; Khan, E. A Review on the Medicinal Importance of Pyridine Derivatives. J. Drug Des. Med. Chem. 2015, 1, 1–11. [Google Scholar] [CrossRef]
- Matsumoto, A.K.; Cavanaugh, P.F. Etoricoxib. Drugs Today 2004, 40, 395–414. [Google Scholar] [CrossRef]
- Gouda, M.A.; Hussein, B.H.M.; Helal, M.H.; Salem, M.A. A Review: Synthesis and Medicinal Importance of Nicotinonitriles and Their Analogous. J. Heterocycl. Chem. 2018, 55, 1524–1553. [Google Scholar] [CrossRef]
- Shishoo, C.J.; Devani, M.B.; Bhadti, V.S.; Ananthan, S.; Ullas, G.V. Reaction of nitriles under acidic conditions: A novel, direct formation of condensed 4-chloropyrimidines. Tetrahedron Lett. 1983, 24, 4611–4612. [Google Scholar] [CrossRef]
- Deo, K.; Avasthi, K.; Pratap, R.; Bhakuni, D.; Joshi, M. Studies in nucleosides. 27. Synthesis of 2, 4-bis, (methylthio)-5-methyl-7-substituted-7h-pyrrolo [2, 3-d] pyrimidines and their biological-activity. Indian J. Chem. B 1990, 29, 459–463. [Google Scholar] [CrossRef]
- Al-Refai, M. Synthesis, Characterization and Biological Evaluation of New 4-Aryl-6-(2,5-dichlorothiophen-3-yl)-1,2-dihydro-2-oxopyridine-3-carbonitrile. Asian J. Chem. 2015, 27, 725–728. [Google Scholar] [CrossRef]
- Ibrahim, M.M. One-Pot Synthesis, Characterization and Antimicrobial Activity of New 3-Cyano-4-Alkyl-6-(2,5-Dichlorothiophen-3-yl)-2(1H)-Pyridones. Jordan J. Chem. 2015, 10, 98–107. [Google Scholar] [CrossRef]
- Ibrahim, M.M.; Al-Refai, M.; Al-Fawwaz, A.; Ali, B.F.; Geyer, A.; Harms, K.; Marsch, M.; Krüger, M.; Osman, H.; Azmi, M.N. Synthesis of Fluorescent 1-(3-Amino-4-(4-(tert-butyl)phenyl)-6-(p-tolyl)furo [2,3-b]pyridin-2-yl)ethan-1-one: Crystal Structure, Fluorescence Behavior, Antimicrobial and Antioxidant Studies. J. Fluoresc. 2018, 28, 655–662. [Google Scholar] [CrossRef]
- Al-Refai, M.; Ibrahim, M.; Al-Fawwaz, A.; Geyer, A. Synthesis and characterization of new 4-aryl-2-(2-oxopropoxy)-6-(2, 5-dichlorothiophene) nicotinonitrile and their furo [2,3-b] pyridine derivatives. Eur. J. Chem. 2018, 9, 375–381. [Google Scholar] [CrossRef]
- Ibrahim, M.M.; Al-Refai, M.; Azmi, M.N.; Osman, H.; Abu Bakar, M.H.; Geyer, A. Synthesis, characterization and cytotoxicity of new nicotinonitriles and their furo[2,3-b]pyridine derivatives. J. Iran. Chem. Soc. 2019, 16, 715–722. [Google Scholar] [CrossRef]
- Al-Refai, M.; Ibrahim, M.M.; Geyer, A.; Marsch, M.; Ali, B.F. Synthesis, Spectroscopic Characterization and X-ray Structure Analysis of 6-(2,5-Dichlorothiophen-3-yl)-2-methoxy-4-(4-methoxyphenyl)pyridine-3-carbonitrile. J. Chem. Cryst. 2016, 46, 331–340. [Google Scholar] [CrossRef]
- Hartough, H.D.; Conley, L.G. Acylation studies in the thiophene and furan series. V. Thiophene carboxylic acids and semicarbazones derived from acetylthiophenes. J. Amer. Chem. Soc. 1947, 69, 3096–3098. [Google Scholar] [CrossRef]
- Wu, Y.-C.; Liu, L.; Liu, Y.-L.; Wang, D.; Chen, Y.-J. TFA-Mediated Tandem Friedel–Crafts Alkylation/Cyclization/Hydrogen Transfer Process for the Synthesis of Flavylium Compounds. J. Org. Chem. 2007, 72, 9383–9386. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.; Li, Y.; Van der Eycken, E.V.; Peng, Y.; Song, G. Synthesis of polysubstituted pyridines under combined microwave and ultrasound irradiation: K2CO3-promoted tandem addition/cyclization/hydrogen shift process. Tetrahedron Lett. 2012, 53, 1160–1162. [Google Scholar] [CrossRef]
- Bahrami, K.; Khodaei, M.M.; Naali, F.; Yousefi, B.H. Synthesis of polysubstituted pyridines via reactions of chalcones and malononitrile in alcohols using Amberlite IRA-400, (OH−). Tetrahedron Lett. 2013, 54, 5293–5298. [Google Scholar] [CrossRef]
- Jain, R.; Roschangar, F.; Ciufolini, M.A. A one-step preparation of functionalized 3-cyano-2-pyridones. Tetrahedron Lett. 1995, 36, 3307–3310. [Google Scholar] [CrossRef]
- Al-Refai, M.; Ibrahim, M.M.; Alsohaili, S.; Geyer, A. Synthesis, characterization and antimicrobial activity of new chalcones and their 6-aryl-4-(2,5-dichlorothiophen-3-yl)-6H-1,3-thiazin-2-amine derivatives. Phosphorus Sulfur Silicon Relat. Elem. 2017, 192, 560–564. [Google Scholar] [CrossRef]
- Elgemeie, G.E.H.; Zohdi, H.F.; Sherif, S.M. Activated nitriles in heterocyclic synthesis: A novel synthetic route to furyl-and thienyl-substituted pyridine derivatives. Phosphorus Sulfur Silicon Relat. Elem. 1990, 54, 215–219. [Google Scholar] [CrossRef]
- Tyndall, D.V.; Al Nakib, T.; Meegan, M.J. A novel synthetic route to phenyl-substituted pyridines synthesis of [1] benzopyrano[4,3-b]pyridines, [1]benzothiopyrano[4,3-b]bpyridines and pyrido[3,2-b][1,4]benzothiazines(1-azaphenothiazines). Tetrahedron Lett. 1988, 29, 2703–2706. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 5a–i are available from the authors. |



| Tested cpd. | IC50 Values (µM) | ||||||
|---|---|---|---|---|---|---|---|
| DU145 | SI | HepG2 | SI | MDA-MB-231 | SI | HSF1184 | |
| 5a | >100 | ND | 49.55 ± 0.51 | 1.66 | 21.15 ± 0.37 | 3.90 | 82.39 ± 0.04 |
| 5b | >100 | ND | 9.48 ± 0.63 | 12.08 | >100 | ND | 114.54 ± 0.38 |
| 5c | 42.36 ± 0.21 | 2.18 | >100 | ND | 28.34 ± 0.42 | 3.26 | 92.38 ± 0.38 |
| 5d | 3.63 ± 0.17 | 116.65 | 1.62 ± 0.15 | 261.39 | 2.59 ± 0.04 | 163.49 | 423.45 ± 0.49 |
| 5f | 27.45 ± 0.63 | 3.48 | 18.34 ± 0.31 | 5.21 | 43.49 ± 0.06 | 2.20 | 95.49 ± 0.29 |
| 5g | 4.97 ± 0.35 | 27.28 | 2.11 ± 0.26 | 64.26 | 1.93 ± 0.03 | 70.25 | 135.59 ± 0.55 |
| 5h | 1.77 ± 0.38 | 105.93 | 1.53 ± 0.38 | 122.54 | 2.14 ± 0.06 | 87.61 | 187.49 ± 0.14 |
| 5i | 1.82 ± 0.21 | 128.82 | 1.72 ± 0.42 | 136.31 | 1.38 ± 0.03 | 169.89 | 234.45 ± 0.27 |
| 6a | 53.49 ± 0.02 | 1.67 | 32.31 ± 0.23 | 2.77 | 15.34 ± 0.41 | 5.82 | 89.34 ± 0.55 |
| 6b | 64.59 ± 0.08 | 1.59 | 39.49 ± 0.58 | 2.59 | 52.39 ± 0.31 | 1.95 | 102.39 ± 0.30 |
| 6c | 21.65 ± 0.12 | 5.19 | 11.28 ± 0.22 | 9.96 | 10.34 ± 0. 37 | 10.87 | 112.38 ± 0.47 |
| 6d | 29.45 ± 0.88 | 3.14 | 64.59 ± 0.33 | 1.43 | 15.43 ± 0.48 | 6.00 | 92.55 ± 0.42 |
| 6f | 18.45 ± 0.02 | 5.68 | 38.45 ± 0.62 | 2.73 | 64.39 ± 0.28 | 1.63 | 104.83 ± 0.71 |
| 6g | 25.42 ± 0.38 | 3.33 | 39.43 ± 0.09 | 2.14 | 18.34 ± 0.34 | 4.61 | 84.56 ± 0.40 |
| 5-fluorouracil | 1.92 ± 0.24 | 96.60 | 1.65 ± 0.09 | 112.41 | 2.21 ± 0.28 | 83.93 | 185.48 ± 0.48 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Refai, M.; Ibrahim, M.M.; Nurul Azmi, M.; Osman, H.; Abu Bakar, M.H.; Geyer, A. The Synthesis, Characterization, Cytotoxic Activity Assessment and Structure-Activity Relationship of 4-Aryl-6-(2,5-dichlorothiophen-3-yl)-2-methoxypyridine-3-carbonitriles. Molecules 2019, 24, 4072. https://doi.org/10.3390/molecules24224072
Al-Refai M, Ibrahim MM, Nurul Azmi M, Osman H, Abu Bakar MH, Geyer A. The Synthesis, Characterization, Cytotoxic Activity Assessment and Structure-Activity Relationship of 4-Aryl-6-(2,5-dichlorothiophen-3-yl)-2-methoxypyridine-3-carbonitriles. Molecules. 2019; 24(22):4072. https://doi.org/10.3390/molecules24224072
Chicago/Turabian StyleAl-Refai, Mahmoud, Mohammad M. Ibrahim, Mohamad Nurul Azmi, Hasnah Osman, Mohamad Hafizi Abu Bakar, and Armin Geyer. 2019. "The Synthesis, Characterization, Cytotoxic Activity Assessment and Structure-Activity Relationship of 4-Aryl-6-(2,5-dichlorothiophen-3-yl)-2-methoxypyridine-3-carbonitriles" Molecules 24, no. 22: 4072. https://doi.org/10.3390/molecules24224072
APA StyleAl-Refai, M., Ibrahim, M. M., Nurul Azmi, M., Osman, H., Abu Bakar, M. H., & Geyer, A. (2019). The Synthesis, Characterization, Cytotoxic Activity Assessment and Structure-Activity Relationship of 4-Aryl-6-(2,5-dichlorothiophen-3-yl)-2-methoxypyridine-3-carbonitriles. Molecules, 24(22), 4072. https://doi.org/10.3390/molecules24224072







