Organocatalytic Asymmetric Conjugate Addition of Aldehydes to Maleimides and Nitroalkenes in Deep Eutectic Solvents
Abstract
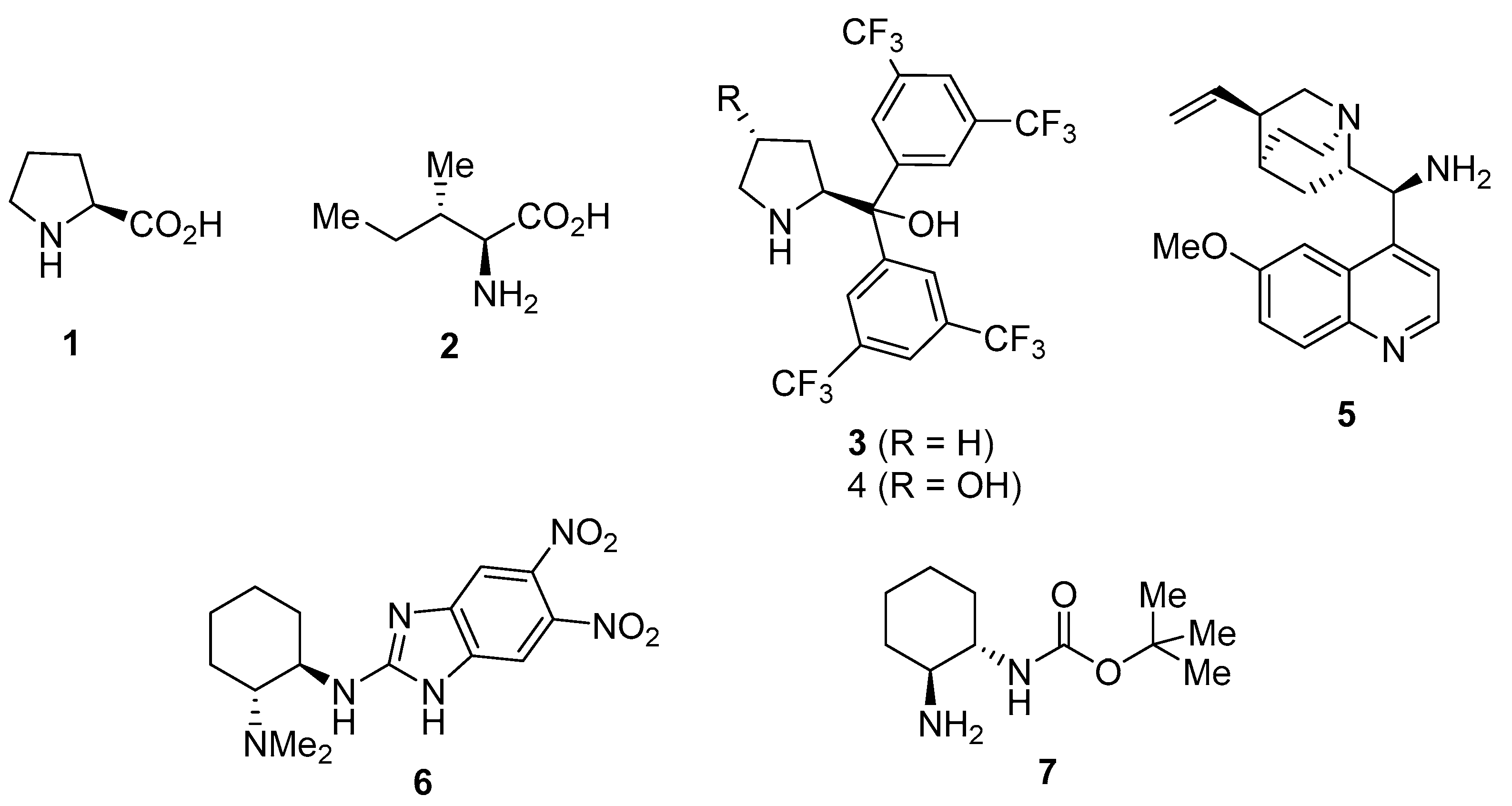
1. Introduction
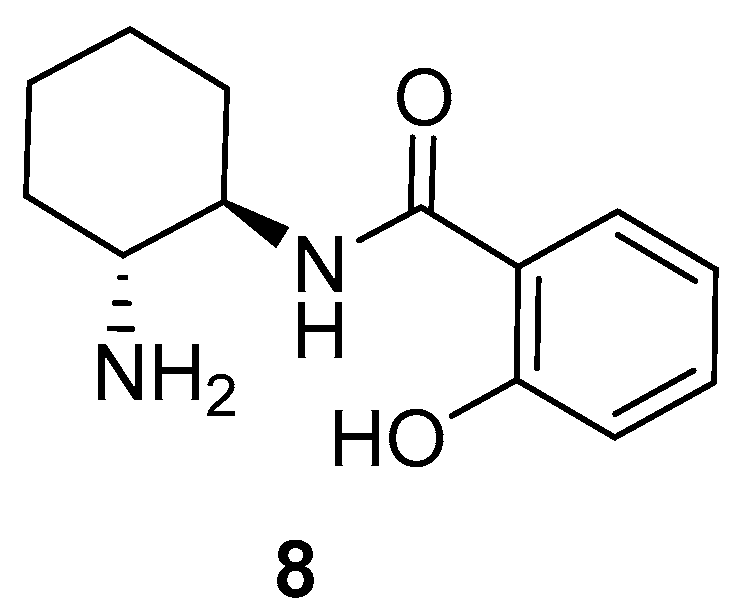
2. Results and Discussion
3. Experimental Section
3.1. General Information
3.2. General Procedure for the Preparation of DESs
3.3. General Procedure for the Enantioselective Conjugate Addition of Aldehydes to Maleimides
3.4. General Procedure for the Enantioselective Conjugate Addition of Isobutyraldehyde to Nitroalkenes
3.5. General Procedure for Recycling Experiments
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Clark, J.H.; Hunt, A.; Topi, C.; Paggiola, G.; Sherwood, J. Sustainable Solvents: Perspectives from Research, Business and International Policy; Royal Society of Chemistry: London, UK, 2017. [Google Scholar]
- Clarke, C.J.; Tu, W.-C.; Levers, O.; Brohl, A.; Hallett, J.P. Green and sustainable solvents in chemical processes. Chem. Rev. 2018, 118, 747–800. [Google Scholar] [CrossRef] [PubMed]
- Marcus, Y. Deep Eutectic Solvents; Springer: Cham, Switzerland, 2019. [Google Scholar]
- García-Alvarez, J. Deep eutectic mixtures: Promising sustainable solvents for metal-catalyzed and metal-mediated organic reactions. Eur. J. Inorg. Chem. 2015, 5147–5157. [Google Scholar] [CrossRef]
- Liu, P.; Hao, J.-W.; Mo, L.-P.; Zhang, Z.-H. Recent advances in the application of deep eutectic solvents as sustainable media as well as catalysts in organic reactions. RSC Adv. 2015, 5, 48675–48704. [Google Scholar] [CrossRef]
- Alonso, D.A.; Baeza, A.; Chinchilla, R.; Guillena, G.; Pastor, I.M.; Ramón, D.J. Deep eutectic solvents: The organic reaction medium of the century. Eur. J. Org. Chem. 2016, 612–632. [Google Scholar] [CrossRef]
- Guajardo, N.; Müller, C.R.; Schrebler, R.; Carlesi, C.; Domínguez de María, P. Deep eutectic solvents for organocatalysis, biotransformations, and multistep organocatalyst/enzyme combinations. ChemCatChem 2016, 8, 1020–1027. [Google Scholar] [CrossRef]
- Khandelwal, S.; Tailor, Y.K.; Kumar, M. Deep eutectic solvents (DESs) as eco-friendly and sustainable solvent/catalyst systems in organic transformations. J. Mol. Liq. 2016, 215, 345–386. [Google Scholar] [CrossRef]
- Longo, L.S.; Craveiro, M.V. Deep eutectic solvents as unconventional media for multicomponent reactions. J. Braz. Chem. Soc. 2018, 29, 1999–2025. [Google Scholar] [CrossRef]
- Florindo, C.; Branco, L.C.; Marrucho, I.M. Quest for green-solvent design: From hydrophilic to hydrophobic (deep) eutectic solvents. ChemSusChem 2019, 12, 1549–1559. [Google Scholar] [CrossRef]
- Peng, L.; Hu, Z.; Lu, Q.; Tang, Z.; Jiao, Y.; Xu, X. DESs: Green solvents for transition metal catalyzed organic reactions. Chin. Chem. Lett. 2019. [Google Scholar] [CrossRef]
- Dalko, P.I. Comprehensive Enantioselective Organocatalysis: Catalysts, Reactions, and Applications; Wiley-VCH: Weinheim, Germany, 2013. [Google Scholar]
- Karame, I.; Srour, H. Recent Advances in Organocatalysis; IntechOpen: London, UK, 2016. [Google Scholar]
- Hernández, J.G.; Juaristi, E. Recent efforts directed to the development of more sustainable asymmetric organocatalysis. Chem. Commun. 2012, 48, 5396–5409. [Google Scholar] [CrossRef]
- Martínez, R.; Berbegal, L.; Guillena, G.; Ramón, D.J. Bio-renewable enantioselective aldol reaction in natural deep eutectic solvents. Green Chem. 2016, 18, 1724–1730. [Google Scholar] [CrossRef]
- Brenna, D.; Massolo, E.; Puglisi, A.; Rossi, S.; Celentano, G.; Benaglia, M.; Capriati, V. Towards the development of continuous, organocatalytic, and stereoselective reactions in deep eutectic solvents. Beilstein J. Org. Chem. 2016, 12, 2620–2626. [Google Scholar] [CrossRef]
- Fanjul-Mosteirin, N.; Concellón, C.; del Amo, V. L-Isoleucine in a choline chloride/ethylene glycol deep eutectic solvent: A reusable reaction kit for the asymmetric cross-aldol carboligation. Org. Lett. 2016, 18, 4266–4269. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.R.; Meiners, I.; Domínguez de María, P. Highly enantioselective tandem enzyme-organocatalyst crossed aldol reactions with acetaldehyde in deep-eutectic-solvents. RSC Adv. 2014, 4, 46097–46101. [Google Scholar] [CrossRef]
- Müller, C.R.; Rosen, A.; Domínguez de María, P. Multi-step enzyme-organocatalyst C-C bond forming reactions in deep-eutectic-solvents: Towards improved performances by organocatalyst design. Sustain. Chem. Process. 2015, 3, 1–8. [Google Scholar] [CrossRef]
- Massolo, E.; Palmieri, S.; Benaglia, M.; Capriati, V.; Perna, F.M. Stereoselective organocatalysed reactions in deep eutectic solvents: Highly tunable and biorenewable reaction media for sustainable organic synthesis. Green Chem. 2016, 792–797. [Google Scholar] [CrossRef]
- Ñiguez, D.R.; Guillena, G.; Alonso, D.A. Chiral 2-Aminobenzimidazoles in Deep Eutectic Mixtures: Recyclable Organocatalysts for the Enantioselective Michael Addition of 1,3-Dicarbonyl Compounds to β-Nitroalkenes. ACS Sustain. Chem. Eng. 2017, 5, 10649–10656. [Google Scholar] [CrossRef]
- Flores-Ferrándiz, J.; Chinchilla, R. Organocatalytic enantioselective conjugate addition of aldehydes to maleimides in deep eutectic solvents. Tetrahedron Asymmetry 2017, 28, 302–306. [Google Scholar] [CrossRef]
- Ñiguez, D.R.; Khazaeli, P.; Alonso, D.A.; Guillena, G. Deep eutectic mixtures as reaction media for the enantioselective organocatalyzed α-amination of 1,3-dicarbonyl compounds. Catalysts 2018, 8, 217. [Google Scholar] [CrossRef]
- Torregrosa-Chinillach, A.; Moragues, A.; Pérez-Furundarena, H.; Chinchilla, R.; Gómez-Bengoa, E.; Guillena, G. Enantioselective Michael addition of aldehydes to maleimides organocatalyzed by a chiral primary amine-salicylamide. Molecules 2018, 23, 3299. [Google Scholar] [CrossRef]
- Martínez-Guillén, J.R.; Flores-Ferrándiz, J.; Gómez, C.; Gómez-Bengoa, E.; Chinchilla, R. Asymmetric conjugate addition of α,α-disubstituted aldehydes to nitroalkenes organocatalyzed by chiral monosalicylamides from trans-cyclohexane-1,2-diamines. Molecules 2018, 23, 141. [Google Scholar] [CrossRef] [PubMed]
- Freiberg, C.; Brunner, N.A.; Schiffer, G.; Lampe, T.; Pohlmann, J.; Brands, M.; Raabe, M.; Haebich, D.; Ziegelbauer, K. Identification and characterization of the first class of potent bacterial Acetyl-CoA carboxylase inhibitors with antibacterial activity. J. Biol. Chem. 2004, 279, 26066–26073. [Google Scholar] [CrossRef] [PubMed]
- Isaka, M.; Rugseree, N.; Maithip, P.; Kongsaeree, P.; Prabpai, S.; Thebtaranonth, Y. Hirsutellones A-E, antimycobacterial alkaloids from the insect pathogenic fungus Hirsutella nivea body centered cubic 2594. Tetrahedron 2005, 61, 5577–5583. [Google Scholar] [CrossRef]
- Uddin, J.; Ueda, K.; Siwu, E.R.O.; Kita, M.; Uemura, D. Cytotoxic labdane alkaloids from an ascidian Lissoclinum sp.: Isolation, structure elucidation, and structure-activity relationship. Bioorg. Med. Chem. 2006, 14, 6954–6961. [Google Scholar] [CrossRef] [PubMed]
- Nöth, J.; Frankowski, K.J.; Neuenswander, B.; Aubé, J.; Reiser, O. Efficient synthesis of γ-lactams by a tandem reductive amination/lactamization sequence. J. Comb. Chem. 2008, 10, 456–459. [Google Scholar] [CrossRef]
- Kazmierski, W.M.; Andrews, W.; Furfine, E.; Spaltenstein, A.; Wright, L. Discovery of potent pyrrolidone-based HIV-1 protease inhibitors with enhanced drug-like properties. Bioorg. Med. Chem. Lett. 2004, 14, 5689–5692. [Google Scholar] [CrossRef]
- Das Sarma, K.; Zhang, J.; Huang, Y.; Davidson, J.G. Amino acid esters and amides for reductive amination of mucochloric acid: Synthesis of novel γ-lactams, short peptides and antiseizure agent Levetiracetam (Keppra). Eur. J. Org. Chem. 2006, 3730–3737. [Google Scholar] [CrossRef]
- Tang, K.; Zhang, J.-T. The effects of (-)-clausenamide on functional recovery in transient focal cerebral ischemia. Neurol. Res. 2002, 24, 473–478. [Google Scholar] [CrossRef]
- Aboul-Enein, M.N.; El-Azzouny, A.A.; Saleh, O.A.; Maklad, Y.A. On chemical structures with potent antiepileptic/anticonvulsant profile. Mini Rev. Med. Chem. 2012, 12, 671–700. [Google Scholar] [CrossRef]
- Andresen, H.; Aydin, B.E.; Mueller, A.; Iwersen-Bergmann, S. An overview of gamma-hydroxybutyric acid: Pharmacodynamics, pharmacokinetics, toxic effects, addiction, analytical methods, and interpretation of results. Drug Test Anal. 2011, 3, 560–568. [Google Scholar] [CrossRef]
- Gajcy, K.; Lochynski, S.; Librowski, T. A role of GABA analogues in the treatment of neurological diseases. Curr. Med. Chem. 2010, 17, 2338–2347. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.-W.; Liu, X.-F.; Liu, J.-T.; Liu, Z.-J.; Tao, J.-C. Highly enantioselective Michael addition of α,α-disubstituted aldehydes to maleimides catalyzed by new primary amine-squaramide bifunctional organocatalysts. Tetrahedron Lett. 2017, 58, 4487–4490. [Google Scholar] [CrossRef]
- Ma, Z.-W.; Liu, X.-F.; Sun, B.; Huang, X.-H.; Tao, J.-C. Chiral primary amine-squaramide catalyzed highly enantioselective Michael addition of isobutyraldehyde to nitroolefins. Synthesis 2017, 49, 1307–1314. [Google Scholar] [CrossRef]
- Rodríguez, J.M.; Pujol, M.D. Straightforward synthesis of nitroolefins by microwave- or ultrasound-assisted Henry reaction. Tetrahedron Lett. 2011, 52, 2629–2632. [Google Scholar] [CrossRef]
- García, G.; Aparicio, S.; Ullah, R.; Atilhan, M. Deep eutectic solvents: Physicochemical properties and gas separation applications. Energy Fuels 2015, 29, 2616–2644. [Google Scholar] [CrossRef]
- Miura, T.; Masuda, A.; Ina, M.; Nakashima, K.; Nishida, S.; Tada, N.; Itoh, A. Asymmetric Michael reactions of α,α-disubstituted aldehydes with maleimides using a primary amine thiourea organocatalyst. Tetrahedron Asymmetry 2011, 22, 1605–1609. [Google Scholar] [CrossRef]
- Avila-Ortiz, C.G.; Díaz-Corona, L.; Jiménez-González, E.; Juaristi, E. Asymmetric Michael addition organocatalyzed by α,β-dipeptides under solvent-free reaction conditions. Molecules 2017, 22, 1328. [Google Scholar] [CrossRef]
- Reyes-Rangel, G.; Vargas-Caporali, J.; Juaristi, E. In search of diamine analogs of the α,α-diphenyl prolinol privileged chiral organocatalyst. Synthesis of diamine derivatives of α,α-diphenyl-(S)-prolinol and their application as organocatalysts in the asymmetric Michael and Mannich reactions. Tetrahedron 2016, 72, 379–391. [Google Scholar] [CrossRef]
- Porta, R.; Coccia, F.; Annunziata, R.; Puglisi, A. Comparison of different polymer- and silica-supported 9-amino-9-deoxy-epi-quinines as recyclable organocatalysts. ChemCatChem 2015, 7, 1490–1499. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 7 and 8 are available from the authors. |
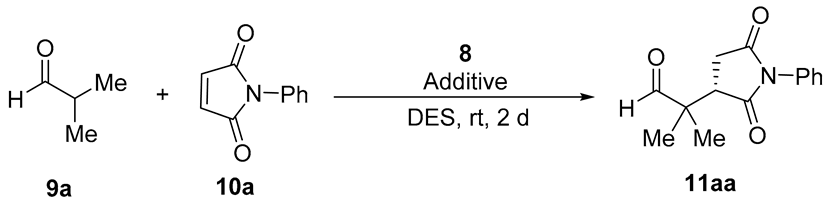
| Entry | 8 mol% | Additive (mol%) a | DES a,b | Conv. (%) c | ee (%) d |
|---|---|---|---|---|---|
| 1 | 10 | - | ChCl/Urea | 27 | 24 |
| 2 | 10 | - | ChCl/Gly | 40 | 63 |
| 3 | 10 | - | ChCl/EG | 99 | 80 |
| 4 | 10 | - | ChCl/H2O | 96 | 19 |
| 5 | 10 | - | Ph3MePBr/Gly | 96 | 79 |
| 6 | 10 | PhCO2H (10) | ChCl/EG | 99 | 82 |
| 7 | 10 | 4-MeOC6H4CO2H (10) | ChCl/EG | 99 | 81 |
| 8 | 10 | 4-O2NC6H4CO2H (10) | ChCl/EG | 99 | 88 |
| 9 | 10 | HDA | ChCl/EG | 99 | 86 |
| 10 | 10 | Imidazole (10) | ChCl/EG | 98 | 87 |
| 11 | 10 | DMAP (10) | ChCl/EG | dec. | n.d. |
| 12 | 10 | 4-O2NC6H4CO2H (20) | ChCl/EG | 99 | 78 |
| 13 | 5 | 4-O2NC6H4CO2H (5) | ChCl/EG | 63 | 81 |
| 14 | 20 | 4-O2NC6H4CO2H (20) | ChCl/EG | 99 | 82 |
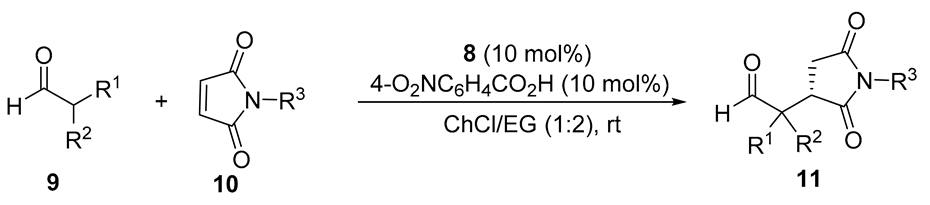
| Entry | Aldehyde | Maleimide | Time (day) | Succinimide | ||||
|---|---|---|---|---|---|---|---|---|
| R1,R2 | No. | R3 | No. | No. | Yield (%) a | eeb (%) b | ||
| 1 | Me,Me | 9a | Ph | 10a | 2 | 11aa | 98 | 88 |
| 2 | Me,Me | 9a | 4-MeC6H4 | 10b | 2 | 11ab | 88 | 66 |
| 3 | Me,Me | 9a | 4-MeOC6H4 | 10c | 2 | 11ac | 98 | 86 |
| 4 | Me,Me | 9a | 4-ClC6H4 | 10d | 2 | 11ad | 59 | 81 |
| 5 | Me,Me | 9a | 4-BrC6H4 | 10e | 2 | 11ae | 85 | 78 |
| 6 | Me,Me | 9a | Me | 10f | 2 | 11af | 98 | 78 |
| 7 | Me,Me | 9a | H | 10g | 2 | 11ag | 98 | 73 |
| 8 | -(CH2)5- | 9b | Ph | 10a | 4 | 11ba | 63 | 41 |
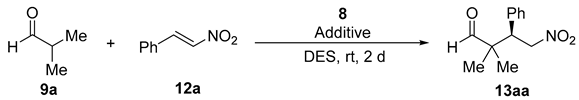
| Entry | 8 mol% | Additive (mol%) a | DES a,b | Conv. (%) c | ee (%) d |
|---|---|---|---|---|---|
| 1 | 10 | - | ChCl/Urea | 35 | 57 |
| 2 | 10 | - | ChCl/Gly | 7 | n.d. |
| 3 | 10 | - | ChCl/EG | 16 | 74 |
| 4 | 10 | - | ChCl/H2O | 18 | 15 |
| 5 | 10 | - | Ph3MePBr/Gly | 99 | 11 |
| 6 | 10 | Imidazole (10) | ChCl/H2O | 30 | 62 |
| 7 | 10 | DABCO (10) | ChCl/H2O | 99 | 55 |
| 8 | 10 | DMAP (10) | ChCl/H2O | 99 | 75 |
| 9 | 10 | PhCO2H (10) | ChCl/H2O | 3 | n.d. |
| 10 | 10 | HDA (10) | ChCl/H2O | 12 | n.d. |
| 11 | 5 | DMAP (5) | ChCl/H2O | 81 | 73 |
| 12 | 20 | DMAP (20) | ChCl/H2O | 99 | 64 |
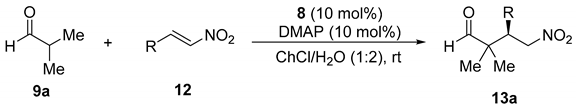
| Entry | Nitroalkene | Time (day) | γ-Nitroaldehyde | |||
|---|---|---|---|---|---|---|
| R | No. | No. | Yield (%) a | ee (%)b | ||
| 1 | Ph | 12a | 2 | 13aa | 92 | 75 |
| 2 | 4-MeC6H4 | 12b | 1 | 13ab | 60 | 61 |
| 3 | 4-MeOC6H4 | 12c | 1 | 13ac | 74 | 63 |
| 4 | 3,4-(OCH2O)C6H3 | 12d | 2 | 13ad | 67 | 75 |
| 5 | 3,4,5-(MeO)3C6H2 | 12e | 1 | 13ae | 73 | 68 |
| 6 | 4-FC6H4 | 12f | 2 | 13af | 78 | 72 |
| 7 | 2-ClC6H4 | 12g | 1 | 13ag | 49 | 80 |
| 8 | 4-ClC6H4 | 12h | 2 | 13ah | 89 | 53 |
| 9 | 2-BrC6H4 | 12i | 2 | 13ai | 70 | 80 |
| 10 | 4-BrC6H4 | 12j | 1 | 13aj | 83 | 70 |
| 11 | 4-F3CC6H4 | 12k | 1 | 13ak | 85 | 70 |
| 12 | 2-Naphthyl | 12l | 1 | 13al | 67 | 75 |
| 13 | 2-Furanyl | 12m | 2 | 13am | 75 | 76 |
| Reaction Cycle | Conv. (%) a | ee (%) b |
|---|---|---|
| 1 | 99 | 88 |
| 2 | 97 | 88 |
| 3 | 53 | 85 |
| Reaction Cycle | Conv. (%) a | ee (%) b |
|---|---|---|
| 1 | 99 | 75 |
| 2 | 93 | 75 |
| 3 | 59 | 74 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torregrosa-Chinillach, A.; Sánchez-Laó, A.; Santagostino, E.; Chinchilla, R. Organocatalytic Asymmetric Conjugate Addition of Aldehydes to Maleimides and Nitroalkenes in Deep Eutectic Solvents. Molecules 2019, 24, 4058. https://doi.org/10.3390/molecules24224058
Torregrosa-Chinillach A, Sánchez-Laó A, Santagostino E, Chinchilla R. Organocatalytic Asymmetric Conjugate Addition of Aldehydes to Maleimides and Nitroalkenes in Deep Eutectic Solvents. Molecules. 2019; 24(22):4058. https://doi.org/10.3390/molecules24224058
Chicago/Turabian StyleTorregrosa-Chinillach, Alejandro, Alba Sánchez-Laó, Elisa Santagostino, and Rafael Chinchilla. 2019. "Organocatalytic Asymmetric Conjugate Addition of Aldehydes to Maleimides and Nitroalkenes in Deep Eutectic Solvents" Molecules 24, no. 22: 4058. https://doi.org/10.3390/molecules24224058
APA StyleTorregrosa-Chinillach, A., Sánchez-Laó, A., Santagostino, E., & Chinchilla, R. (2019). Organocatalytic Asymmetric Conjugate Addition of Aldehydes to Maleimides and Nitroalkenes in Deep Eutectic Solvents. Molecules, 24(22), 4058. https://doi.org/10.3390/molecules24224058








