Eugenol Exerts Apoptotic Effect and Modulates the Sensitivity of HeLa Cells to Cisplatin and Radiation
Abstract
1. Introduction
2. Results
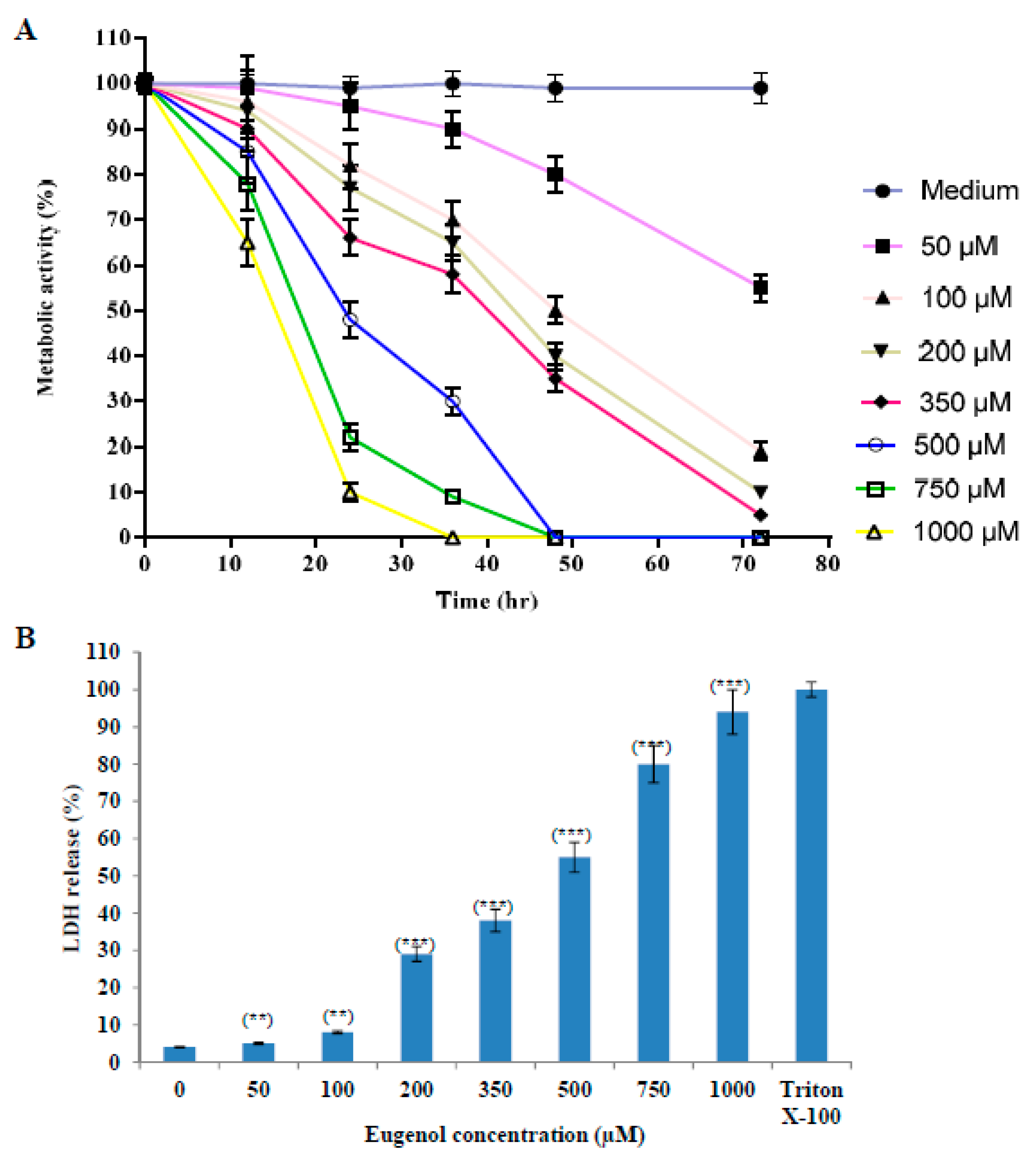
2.1. Eugenol Cytotoxicity on HeLa Cells
2.1.1. Effect of Eugenol on Survival of HeLa Cells
2.1.2. Effect of Eugenol on Lactate Dehydrogenase (LDH) Release from HeLa Cells
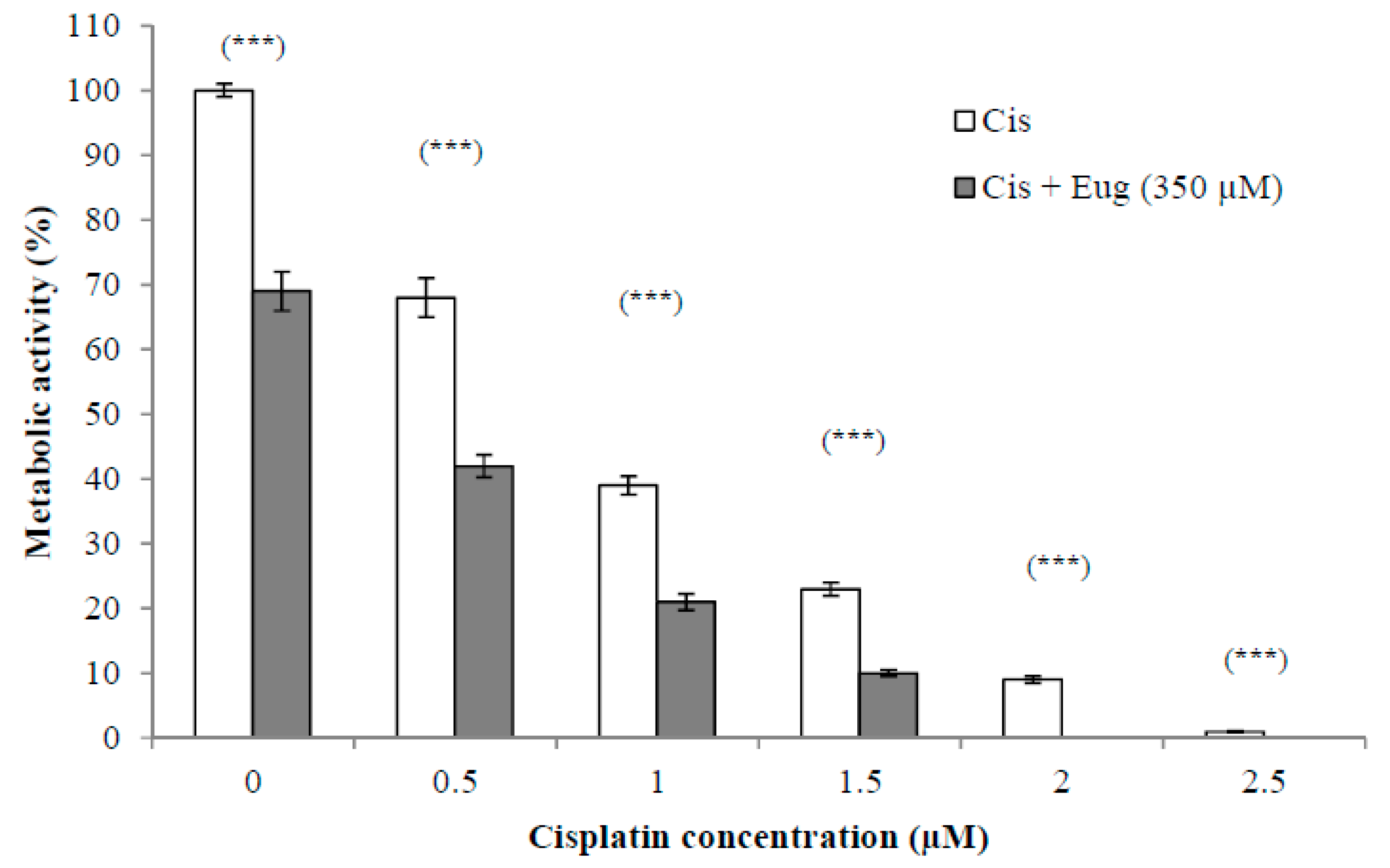
2.2. Effect of Eugenol on Sensitivity of HeLa Cells to Cisplatin
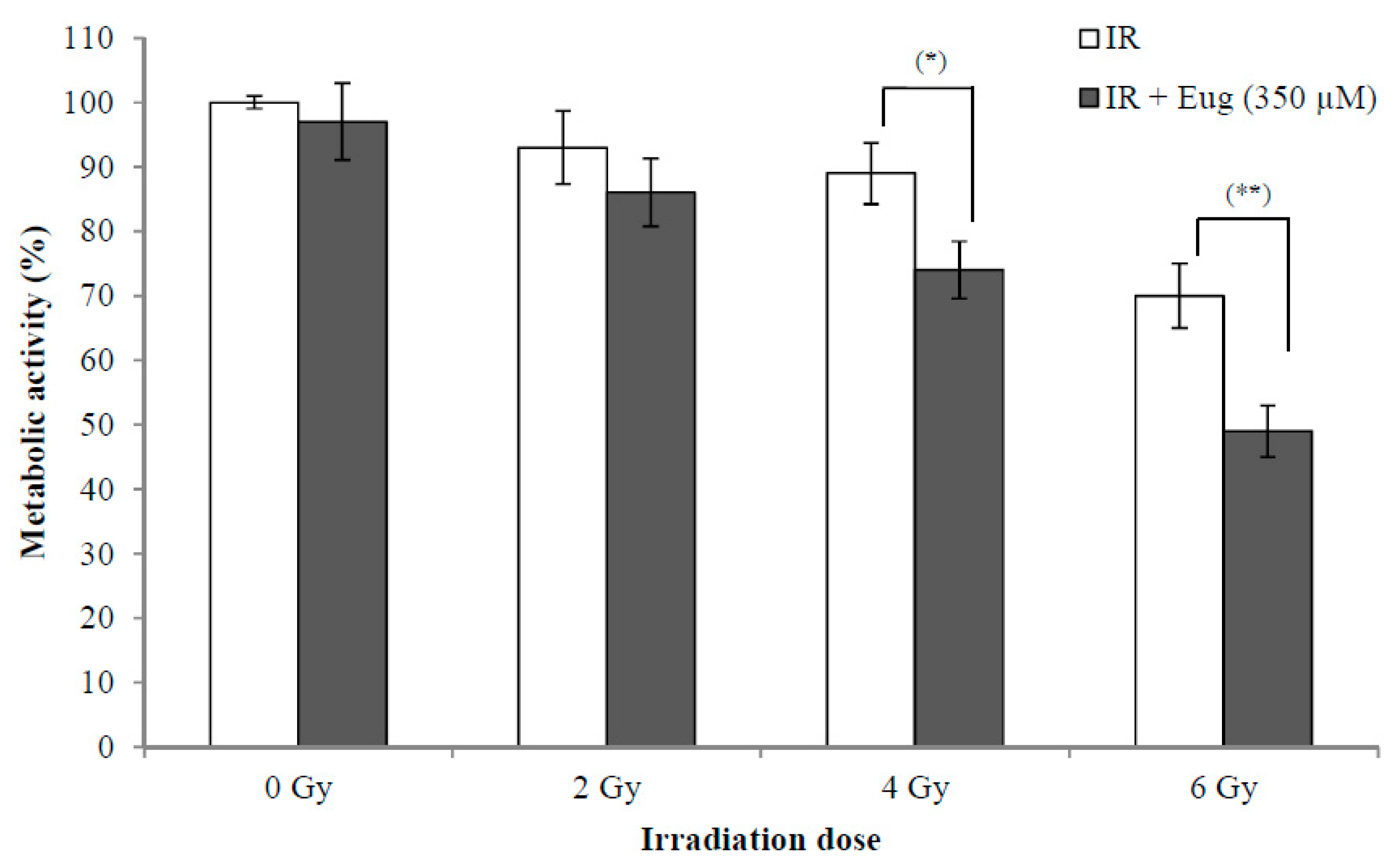
2.3. Effect of Eugenol on Sensitivity of HeLa Cells to radiation
2.4. Effect of Eugenol on Cell Signaling of HeLa Cells
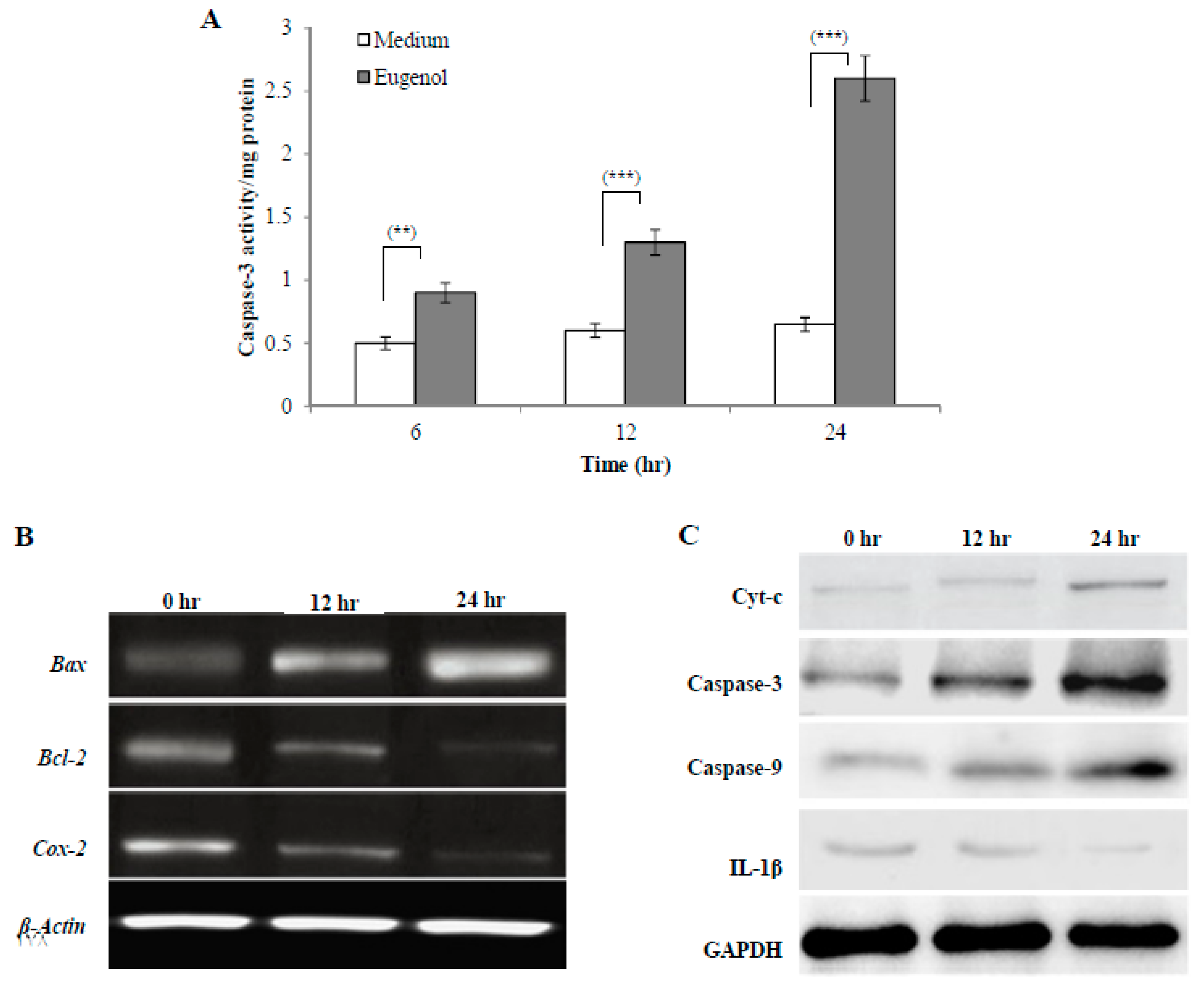
2.4.1. Effect of Eugenol on Caspase-3 Activity in HeLa Cells
2.4.2. Effect of Eugenol on Bax, Bcl-2, and Cox-2 Gene Expression in HeLa Cells
2.4.3. Effect of Eugenol on Cytochrome c (Cyt-c), Caspase-3, Caspase-9, and Interleukin-1 Beta (IL-1β) Protein Expression in HeLa Cells
3. Discussion
4. Materials and Methods
4.1. Chemicals, Reagents, and Antibodies
4.2. Cell Culture and Reagents
4.3. Cell Viability Assay
4.4. Cytotoxicity Assay
4.5. Chemotherapy Sensitivity Assay
4.6. Radiotherapy Sensitivity Assay
4.7. Caspase-3 Activity Analysis
4.8. Expression Analysis of Bax, Bcl-2, and Cox-2 Genes by Reverse Transcription-PCR
4.9. Western Blot Assay of Cyt-c, Caspase-3, Caspase-9, and IL-1β Protein Expression
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sklirou, A.; Papanagnou, E.D.; Fokialakis, N.; Trougakos, I.P. Cancer chemoprevention via activation of proteostatic modules. Cancer Lett. 2018, 413, 110–121. [Google Scholar] [CrossRef] [PubMed]
- Couffignal, A.L.; Lapeyre-Mestre, M.; Bonhomme, C.; Bugat, R.; Montastruc, J.L. Adverse effects of anticancer drugs: Apropos of a pharmacovigilance study at a specialized oncology institution. Therapie 2000, 55, 635–641. [Google Scholar] [PubMed]
- Amereh, Z.; Hatami, N.; Shirazi, F.H.; Gholami, S.; Hosseini, S.H.; Noubarani, M.; Kamalinejad, M.; Andalib, S.; Keyhanfar, F.; Eskandari, M.R. Cancer chemoprevention by oleaster (Elaeagnus angustifoli L.) fruit extract in a model of hepatocellular carcinoma induced by diethylnitrosamine in rats. EXCLI J. 2017, 16, 1046–1056. [Google Scholar] [CrossRef] [PubMed]
- Fathy, M.; Nikaido, T. In Vivo modulation of iNOS pathway in hepatocellular carcinoma by Nigella sativa. Environ. Health Prev. Med. 2013, 18, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, V.; Shrivastava, R.; Hussain, S.; Ganguly, C.; Bharadwaj, M. Comparative anticancer potential of clove (Syzygium aromaticum)-an Indian spice-against cancer cell lines of various anatomical origin. Asian Pac. J. Cancer Prev. 2011, 12, 1989–1993. [Google Scholar]
- Liu, H.; Schmitz, J.C.; Wei, J.; Cao, S.; Beumer, J.H.; Strychor, S.; Cheng, L.; Liu, M.; Wang, C.; Wu, N.; et al. Clove extract inhibits tumor growth and promotes cell cycle arrest and apoptosis. Oncol. Res. 2014, 21, 247–259. [Google Scholar] [CrossRef]
- Jaganathan, S.K.; Supriyanto, E. Antiproliferative and molecular mechanism of eugenol-induced apoptosis in cancer cells. Molecules 2012, 17, 6290–6304. [Google Scholar] [CrossRef]
- Rastogi, S.C.; Johansen, J.D.; Menne, T. Natural ingredients based cosmetics. Content of selected fragrance sensitizers. Contact Dermat. 1996, 34, 423–426. [Google Scholar] [CrossRef]
- Kayaoglu, G.; Erten, H.; Alacam, T.; Orstavik, D. Short-term antibacterial activity of root canal sealers towards enterococcus faecalis. Int. Endod. J. 2005, 38, 483–488. [Google Scholar] [CrossRef]
- de Souza Romaneli, R.; Boaratti, A.Z.; Tellechea Rodrigues, A.; de Almeida Ueiroz, D.M.; Khan, K.U.; Nascimento, T.M.T.; Fernandes, J.B.K. Efficacy of Benzocaine, Eugenol and Menthol as Anesthetics for Freshwater Angelfish (Pterophyllum scalare). J. Aquat. Anim. Health 2018. [Google Scholar] [CrossRef]
- Benencia, F.; Courreges, M.C. In Vitro and in vivo activity of eugenol on human herpesvirus. Phytother. Res. 2000, 14, 495–500. [Google Scholar] [CrossRef]
- Pisano, M.; Pagnan, G.; Loi, M.; Mura, M.E.; Tilocca, M.G.; Palmieri, G.; Fabbri, D.; Dettori, M.A.; Delogu, G.; Ponzoni, M.; et al. Antiproliferative and pro-apoptotic activity of eugenol-related biphenyls on malignant melanoma cells. Mol. Cancer 2007, 6, 8. [Google Scholar] [CrossRef] [PubMed]
- Wieczynska, J.; Cavoski, I. Antimicrobial, antioxidant and sensory features of eugenol, carvacrol and trans-anethole in active packaging for organic ready-to-eat iceberg lettuce. Food Chem. 2018, 259, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Soundran, V.; Namagiri, T.; Manonayaki, S.; Vanithakumari, G. Hepatotoxicity of eugenol. Anc. Sci. Life 1994, 13, 213–217. [Google Scholar] [PubMed]
- Sakagami, H.; Sugimoto, M.; Kanda, Y.; Murakami, Y.; Amano, O.; Saitoh, J.; Kochi, A. Changes in metabolic profiles of human oral cells by benzylidene ascorbates and eugenol. Medicines 2018, 5. [Google Scholar] [CrossRef]
- Slamenova, D.; Horvathova, E.; Wsolova, L.; Sramkova, M.; Navarova, J. Investigation of anti-oxidative, cytotoxic, DNA-damaging and DNA-protective effects of plant volatiles eugenol and borneol in human-derived HepG2, Caco-2 and VH10 cell lines. Mutat. Res. 2009, 677, 46–52. [Google Scholar] [CrossRef]
- Bezerra, D.P.; Militao, G.C.G.; de Morais, M.C.; de Sousa, D.P. The dual antioxidant/prooxidant effect of eugenol and its action in cancer development and treatment. Nutrients 2017, 9, 1367. [Google Scholar] [CrossRef]
- Fangjun, L.; Zhijia, Y. Tumor suppressive roles of eugenol in human lung cancer cells. Thorac. Cancer 2018, 9, 25–29. [Google Scholar] [CrossRef]
- Islam, S.S.; Al-Sharif, I.; Sultan, A.; Al-Mazrou, A.; Remmal, A.; Aboussekhra, A. Eugenol potentiates cisplatin anti-cancer activity through inhibition of ALDH-positive breast cancer stem cells and the NF-kappaB signaling pathway. Mol. Carcinog. 2018, 57, 333–346. [Google Scholar] [CrossRef]
- Jaganathan, S.K.; Mazumdar, A.; Mondhe, D.; Mandal, M. Apoptotic effect of eugenol in human colon cancer cell lines. Cell Biol. Int. 2011, 35, 607–615. [Google Scholar] [CrossRef]
- Sisakhtnezhad, S.; Heidari, M.; Bidmeshkipour, A. Eugenol enhances proliferation and migration of mouse bone marrow-derived mesenchymal stem cells in vitro. Environ. Toxicol. Pharmacol. 2018, 57, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Yi, J.L.; Shi, S.; Shen, Y.L.; Wang, L.; Chen, H.Y.; Zhu, J.; Ding, Y. Myricetin and methyl eugenol combination enhances the anticancer activity, cell cycle arrest and apoptosis induction of cis-platin against HeLa cervical cancer cell lines. Int. J. Clin. Exp. Pathol. 2015, 8, 1116–1127. [Google Scholar] [PubMed]
- Park, B.S.; Song, Y.S.; Yee, S.B.; Lee, B.G.; Seo, S.Y.; Park, Y.C.; Kim, J.M.; Kim, H.M.; Yoo, Y.H. Phospho-ser 15-p53 translocates into mitochondria and interacts with Bcl-2 and Bcl-xL in eugenol-induced apoptosis. Apoptosis 2005, 10, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Junior, P.L.; Camara, D.A.; Costa, A.S.; Ruiz, J.L.; Levy, D.; Azevedo, R.A.; Pasqualoto, K.F.; de Oliveira, C.F.; de Melo, T.C.; Pessoa, N.D.; et al. Apoptotic effect of eugenol envolves G2/M phase abrogation accompanied by mitochondrial damage and clastogenic effect on cancer cell in vitro. Phytomedicine 2016, 23, 725–735. [Google Scholar] [CrossRef]
- Yoo, C.B.; Han, K.T.; Cho, K.S.; Ha, J.; Park, H.J.; Nam, J.H.; Kil, U.H.; Lee, K.T. Eugenol isolated from the essential oil of eugenia caryophyllata induces a reactive oxygen species-mediated apoptosis in HL-60 human promyelocytic leukemia cells. Cancer Lett. 2005, 225, 41–52. [Google Scholar] [CrossRef]
- Vidhya, N.; Devaraj, S.N. Induction of apoptosis by eugenol in human breast cancer cells. Indian J. Exp. Biol. 2011, 49, 871–878. [Google Scholar]
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef]
- Bellone, S.; Pecorelli, S.; Cannon, M.J.; Santin, A.D. Advances in dendritic-cell-based therapeutic vaccines for cervical cancer. Expert Rev. Anticancer Ther. 2007, 7, 1473–1486. [Google Scholar] [CrossRef]
- Fathy, M.; Awale, S.; Nikaido, T. Phosphorylated akt protein at Ser473 enables HeLa cells to tolerate nutrient-deprived conditions. Asian Pac. J. Cancer Prev. 2017, 18, 3255–3260. [Google Scholar] [CrossRef]
- Friedlander, M.; Grogan, M. Guidelines for the treatment of recurrent and metastatic cervical cancer. Oncologist 2002, 7, 342–347. [Google Scholar]
- Derks, M.; Van der Velden, J.; De Kroon, C.D.; Nijman, H.W.; Van Lonkhuijzen, L.; Van der Zee, A.G.J.; Zwinderman, A.H.; Kenter, G.G. Surgical treatment of early-stage cervical cancer: A multi-institution experience in 2124 cases in The Netherlands over a 30-year period. Int. J. Gynecol. Cancer 2018, 28, 757–763. [Google Scholar] [CrossRef] [PubMed]
- Mapanga, W.; Chipato, T.; Feresu, S.A. Treatment of cervical cancer in HIV-seropositive women from developing countries: A protocol for a systematic review. Syst. Rev. 2018, 7, 22. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Maheshwari, A.; Parab, P.; Mahantshetty, U.; Hawaldar, R.; Sastri Chopra, S.; Kerkar, R.; Engineer, R.; Tongaonkar, H.; Ghosh, J.; et al. Neoadjuvant chemotherapy followed by radical surgery versus concomitant chemotherapy and radiotherapy in patients with stage IB2, IIA, or IIB squamous cervical cancer: A randomized controlled trial. J. Clin. Oncol. 2018, 36, 1548–1555. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.B.; Kim, Y.S.; Lee, J.M. Oncologic outcomes of adjuvant chemotherapy alone after radical surgery for stage IB-IIA cervical cancer patients. J. Gynecol. Oncol. 2018, 29. [Google Scholar] [CrossRef] [PubMed]
- Yip-Schneider, M.T.; Wu, H.; Ralstin, M.; Yiannoutsos, C.; Crooks, P.A.; Neelakantan, S.; Noble, S.; Nakshatri, H.; Sweeney, C.J.; Schmidt, C.M. Suppression of pancreatic tumor growth by combination chemotherapy with sulindac and LC-1 is associated with cyclin D1 inhibition in vivo. Mol. Cancer Ther. 2007, 6, 1736–1744. [Google Scholar] [CrossRef]
- You, M.; Savaraj, N.; Wangpaichitr, M.; Wu, C.; Kuo, M.T.; Varona-Santos, J.; Nguyen, D.M.; Feun, L. The combination of ADI-PEG20 and TRAIL effectively increases cell death in melanoma cell lines. Biochem. Biophys. Res. Commun. 2010, 394, 760–766. [Google Scholar] [CrossRef]
- Bi, Y.L.; Min, M.; Shen, W.; Liu, Y. Genistein induced anticancer effects on pancreatic cancer cell lines involves mitochondrial apoptosis, G0/G1cell cycle arrest and regulation of STAT3 signalling pathway. Phytomedicine 2018, 39, 10–16. [Google Scholar] [CrossRef]
- Burnett, J.P.; Lim, G.; Li, Y.; Shah, R.B.; Lim, R.; Paholak, H.J.; McDermott, S.P.; Sun, L.; Tsume, Y.; Bai, S.; et al. Sulforaphane enhances the anticancer activity of taxanes against triple negative breast cancer by killing cancer stem cells. Cancer Lett. 2017, 394, 52–64. [Google Scholar] [CrossRef]
- Carlson, A.; Alderete, K.S.; Grant, M.K.O.; Seelig, D.M.; Sharkey, L.C.; Zordoky, B.N.M. Anticancer effects of resveratrol in canine hemangiosarcoma cell lines. Vet. Comp. Oncol. 2018, 16, 253–261. [Google Scholar] [CrossRef]
- Fathy, M.; Nikaido, T. In Vivo attenuation of angiogenesis in hepatocellular carcinoma by Nigella sativa. Turk. J. Med. Sci. 2018, 48, 178–186. [Google Scholar] [CrossRef]
- Chatterjee, D.; Bhattacharjee, P. Use of eugenol-lean clove extract as a flavoring agent and natural antioxidant in mayonnaise: Product characterization and storage study. J. Food Sci. Technol. 2015, 52, 4945–4954. [Google Scholar] [CrossRef] [PubMed]
- Mahboub, R.; Memmou, F. Antioxidant activity and kinetics studies of eugenol and 6-bromoeugenol. Nat. Prod. Res. 2015, 29, 966–971. [Google Scholar] [CrossRef] [PubMed]
- Fathy, M.; Khalifa, E.M.; Fawzy, M.A. Modulation of inducible nitric oxide synthase pathway by eugenol and telmisartan in carbon tetrachloride-induced liver injury in rats. Life Sci. 2019, 216, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Al-Sharif, I.; Remmal, A.; Aboussekhra, A. Eugenol triggers apoptosis in breast cancer cells through E2F1/survivin down-regulation. BMC Cancer 2013, 13, 600. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.; Brahmbhatt, K.; Priyani, A.; Ahmed, M.; Rizvi, T.A.; Sharma, C. Eugenol enhances the chemotherapeutic potential of gemcitabine and induces anticarcinogenic and anti-inflammatory activity in human cervical cancer cells. Cancer Biother. Radiopharm. 2011, 26, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.; Priyani, A.; Sadrieh, L.; Brahmbhatt, K.; Ahmed, M.; Sharma, C. Concurrent sulforaphane and eugenol induces differential effects on human cervical cancer cells. Integr. Cancer Ther. 2012, 11, 154–165. [Google Scholar] [CrossRef] [PubMed]
- Girdhani, S.; Bhosle, S.M.; Thulsidas, S.A.; Kumar, A.; Mishra, K.P. Potential of radiosensitizing agents in cancer chemo-radiotherapy. J. Cancer Res. Ther. 2005, 1, 129–131. [Google Scholar]
- Lee, J.H.; Lee, H.H.; Kim, K.N.; Kim, K.M. Cytotoxicity and anti-inflammatory effects of zinc ions and eugenol during setting of ZOE in immortalized human oral keratinocytes grown as three-dimensional spheroids. Dent. Mater. 2016, 32, e93–e104. [Google Scholar] [CrossRef]
- Duensing, T.D.; Watson, S.R. Assessment of apoptosis (Programmed cell death) by flow cytometry. Cold Spring Harb. Protoc. 2018, 1. [Google Scholar] [CrossRef]
- Chou, H.L.; Fong, Y.; Wei, C.K.; Tsai, E.M.; Chen, J.Y.; Chang, W.T.; Wu, C.Y.; Huang, H.W.; Chiu, C.C. A Quinone-Containing Compound Enhances Camptothecin-Induced Apoptosis of Lung Cancer Through Modulating Endogenous ROS and ERK Signaling. Arch. Immunol. Ther. Exp. 2017, 65, 241–252. [Google Scholar] [CrossRef]
- Zhao, X.; Li, X.; Ren, Q.; Tian, J.; Chen, J. Calycosin induces apoptosis in colorectal cancer cells, through modulating the ERbeta/MiR-95 and IGF-1R, PI3K/Akt signaling pathways. Gene 2016, 591, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Halimah, E.; Diantini, A.; Destiani, D.P.; Pradipta, I.S.; Sastramihardja, H.S.; Lestari, K.; Subarnas, A.; Abdulah, R.; Koyama, H. Induction of caspase cascade pathway by kaempferol-3-O-rhamnoside in LNCaP prostate cancer cell lines. Biomed. Rep. 2015, 3, 115–117. [Google Scholar] [CrossRef] [PubMed]
- Manoharan, G.; Jaiswal, S.R.; Singh, J. Effect of alpha, beta momorcharin on viability, caspase activity, cytochrome c release and on cytosolic calcium levels in different cancer cell lines. Mol. Cell. Biochem. 2014, 388, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Patel, P.B.; Thakkar, V.R. L-carvone induces p53, caspase 3 mediated apoptosis and inhibits the migration of breast cancer cell lines. Nutr. Cancer 2014, 66, 453–462. [Google Scholar] [CrossRef]
- Fisher, T.C.; Milner, A.E.; Gregory, C.D.; Jackman, A.L.; Aherne, G.W.; Hartley, J.A.; Dive, C.; Hickman, J.A. bcl-2 modulation of apoptosis induced by anticancer drugs: Resistance to thymidylate stress is independent of classical resistance pathways. Cancer Res. 1993, 53, 3321–3326. [Google Scholar]
- Okada, N.; Hirata, A.; Murakami, Y.; Shoji, M.; Sakagami, H.; Fujisawa, S. Induction of cytotoxicity and apoptosis and inhibition of cyclooxygenase-2 gene expression by eugenol-related compounds. Anticancer Res. 2005, 25, 3263–3269. [Google Scholar]
- Atari-Hajipirloo, S.; Nikanfar, S.; Heydari, A.; Noori, F.; Kheradmand, F. The effect of celecoxib and its combination with imatinib on human HT-29 colorectal cancer cells: Involvement of COX-2, Caspase-3, VEGF and NF-kappaB genes expression. Cell. Mol. Biol. 2016, 62, 68–74. [Google Scholar]
- Bocca, C.; Ievolella, M.; Autelli, R.; Motta, M.; Mosso, L.; Torchio, B.; Bozzo, F.; Cannito, S.; Paternostro, C.; Colombatto, S.; et al. Expression of Cox-2 in human breast cancer cells as a critical determinant of epithelial-to-mesenchymal transition and invasiveness. Expert. Opin Ther. Targets. 2014, 18, 121–135. [Google Scholar] [CrossRef]
- Tsujii, M.; DuBois, R.N. Alterations in cellular adhesion and apoptosis in epithelial cells overexpressing prostaglandin endoperoxide synthase 2. Cell 1995, 83, 493–501. [Google Scholar] [CrossRef]
- Dong, X.; Zhang, L.; Meng, Q.; Gao, Q. Association between interleukin-1A, interleukin-1B, and bridging integrator 1 polymorphisms and alzheimer’s disease: A standard and cumulative meta-analysis. Mol. Neurobiol. 2017, 54, 736–747. [Google Scholar] [CrossRef]
- Mattila, K.M.; Rinne, J.O.; Lehtimaki, T.; Roytta, M.; Ahonen, J.P.; Hurme, M. Association of an interleukin 1B gene polymorphism (-511) with Parkinson’s disease in finnish patients. J. Med. Genet. 2002, 39, 400–402. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not Available. |
| Primer | Sequence of the Primer | Annealing Temperature | Size (bp) |
|---|---|---|---|
| Bax | Forward: 5′-GTTTCATCCAGGATCGAGCAG-3′ | 53 °C | 487 |
| Reverse: 5′-CATCTTCTTCCAGATGGTGA-3′ | |||
| Bcl-2 | Forward: 5′-CCTGTGGATGACTGAGTACC-3′ | 53 °C | 127 |
| Reverse: 5′-GAGACAGCCAGGAGAAATCA-3′ | |||
| Cox-2 | Forward: 5′-ATACCAAAACCGCATTGCCG-3′ | 57 °C | 305 |
| Reverse: 5′-TCTAACTCCGCAGCCATTTC-3′ | |||
| β-Actin | Forward: 5′-CGGGACCTGACTGACTAC-3′ | 56 °C | 252 |
| Reverse: 5′-GAAGGAAGGCTGGAAGAG-3′ |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fathy, M.; Fawzy, M.A.; Hintzsche, H.; Nikaido, T.; Dandekar, T.; Othman, E.M. Eugenol Exerts Apoptotic Effect and Modulates the Sensitivity of HeLa Cells to Cisplatin and Radiation. Molecules 2019, 24, 3979. https://doi.org/10.3390/molecules24213979
Fathy M, Fawzy MA, Hintzsche H, Nikaido T, Dandekar T, Othman EM. Eugenol Exerts Apoptotic Effect and Modulates the Sensitivity of HeLa Cells to Cisplatin and Radiation. Molecules. 2019; 24(21):3979. https://doi.org/10.3390/molecules24213979
Chicago/Turabian StyleFathy, Moustafa, Michael Atef Fawzy, Henning Hintzsche, Toshio Nikaido, Thomas Dandekar, and Eman M. Othman. 2019. "Eugenol Exerts Apoptotic Effect and Modulates the Sensitivity of HeLa Cells to Cisplatin and Radiation" Molecules 24, no. 21: 3979. https://doi.org/10.3390/molecules24213979
APA StyleFathy, M., Fawzy, M. A., Hintzsche, H., Nikaido, T., Dandekar, T., & Othman, E. M. (2019). Eugenol Exerts Apoptotic Effect and Modulates the Sensitivity of HeLa Cells to Cisplatin and Radiation. Molecules, 24(21), 3979. https://doi.org/10.3390/molecules24213979









