Anti-Allergic and Anti-Inflammatory Effects of Kuwanon G and Morusin on MC/9 Mast Cells and HaCaT Keratinocytes
Abstract
1. Introduction
2. Results
2.1. High-Performance Liquid Chromatography (HPLC) Analysis of the Two Bioactive Marker Compounds in M. alba L.
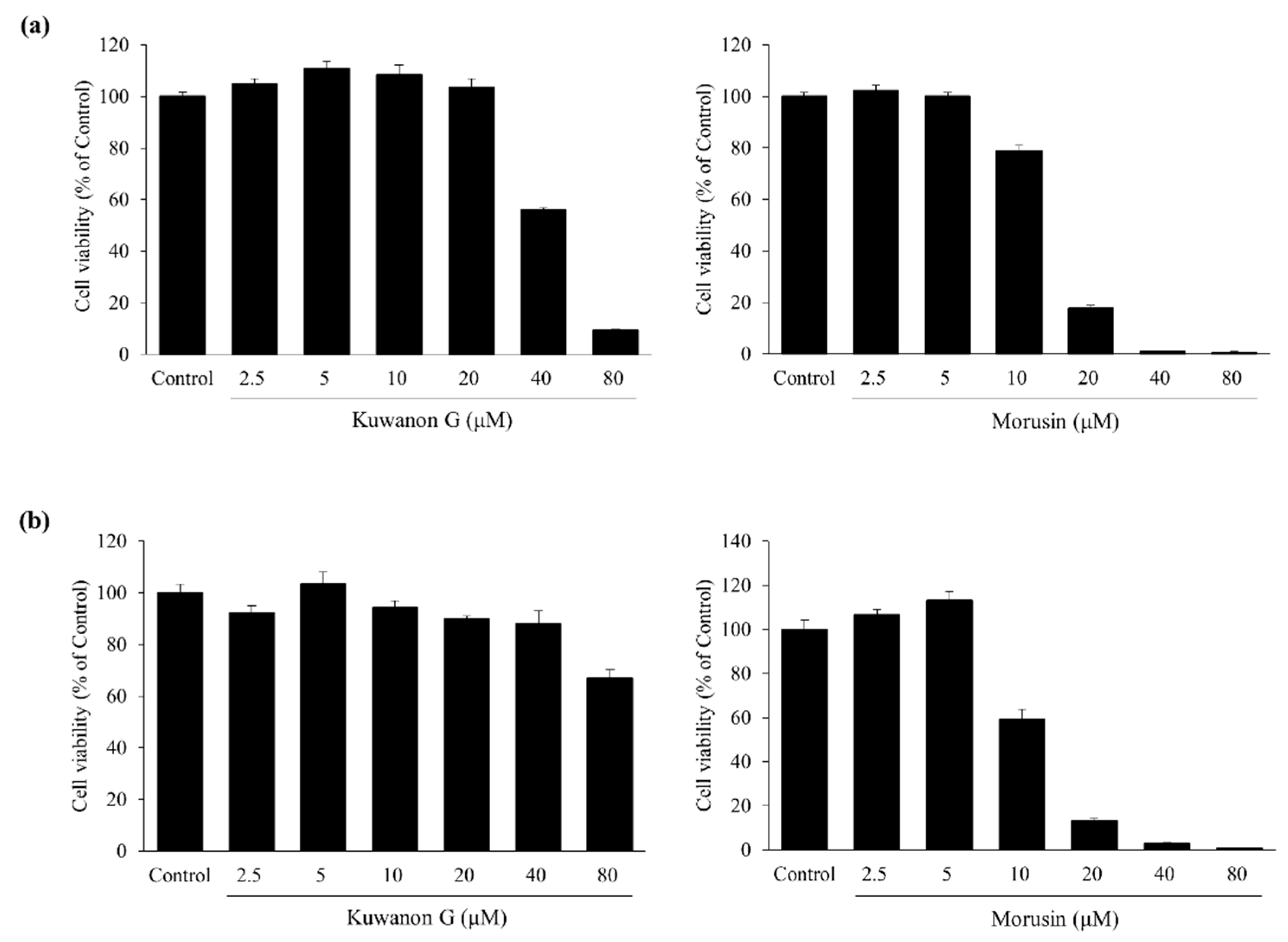
2.2. Effects of Kuwanon G and Morusin on HaCaT and MC/9 Cell Viability
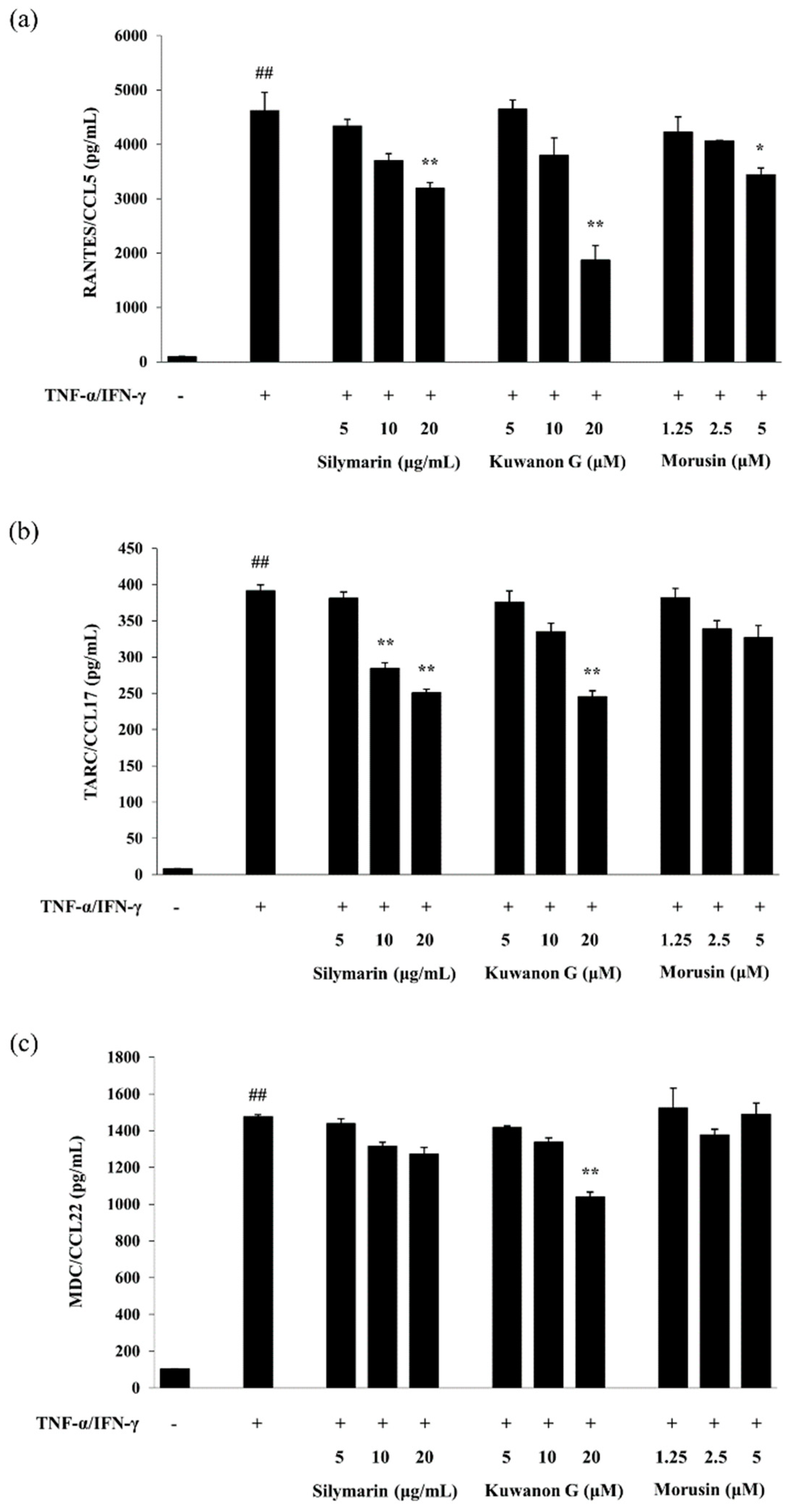
2.3. Effects of Kuwanon G and Morusin on the Chemokine Production in HaCaT Keratinocytes
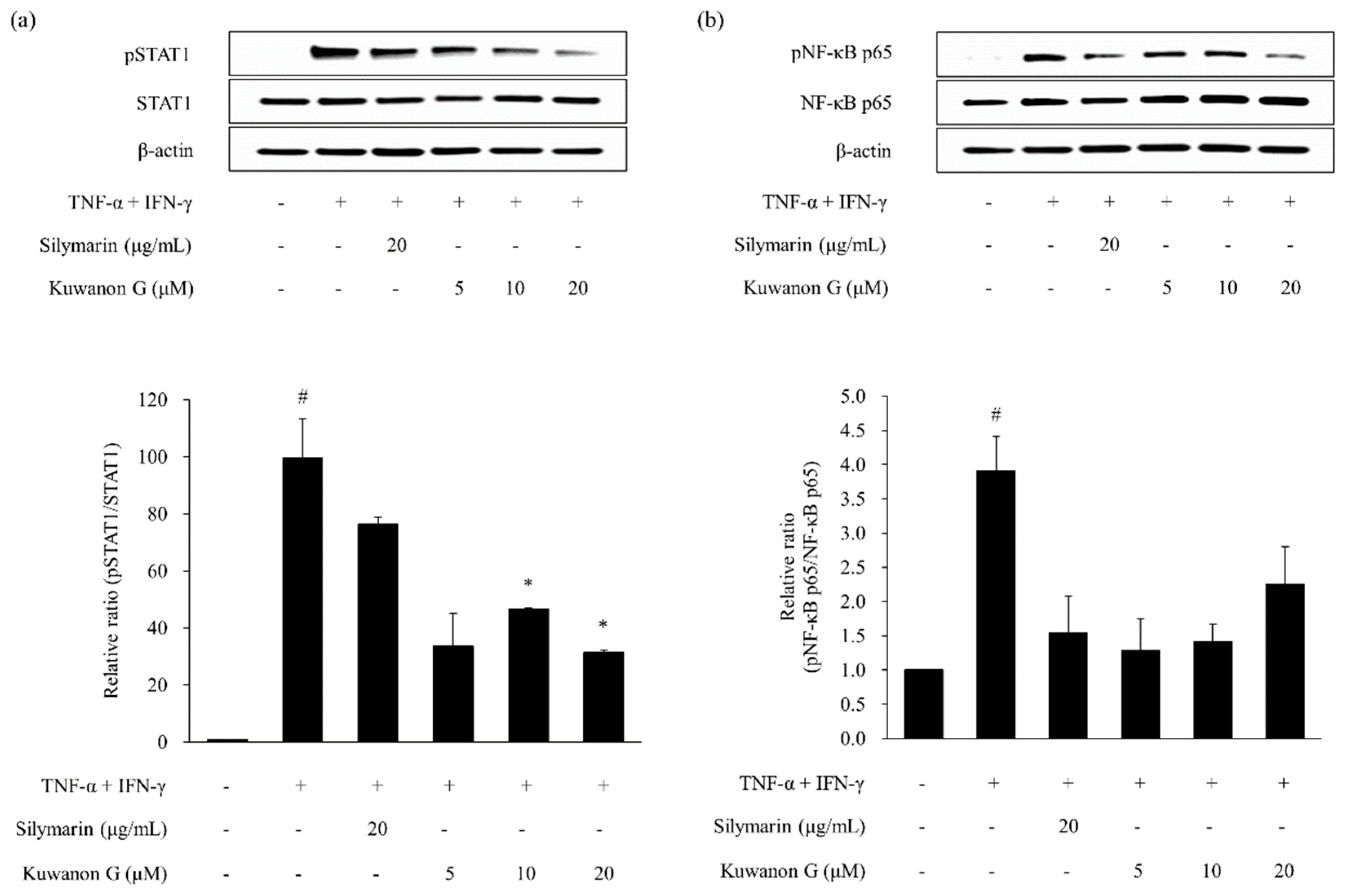
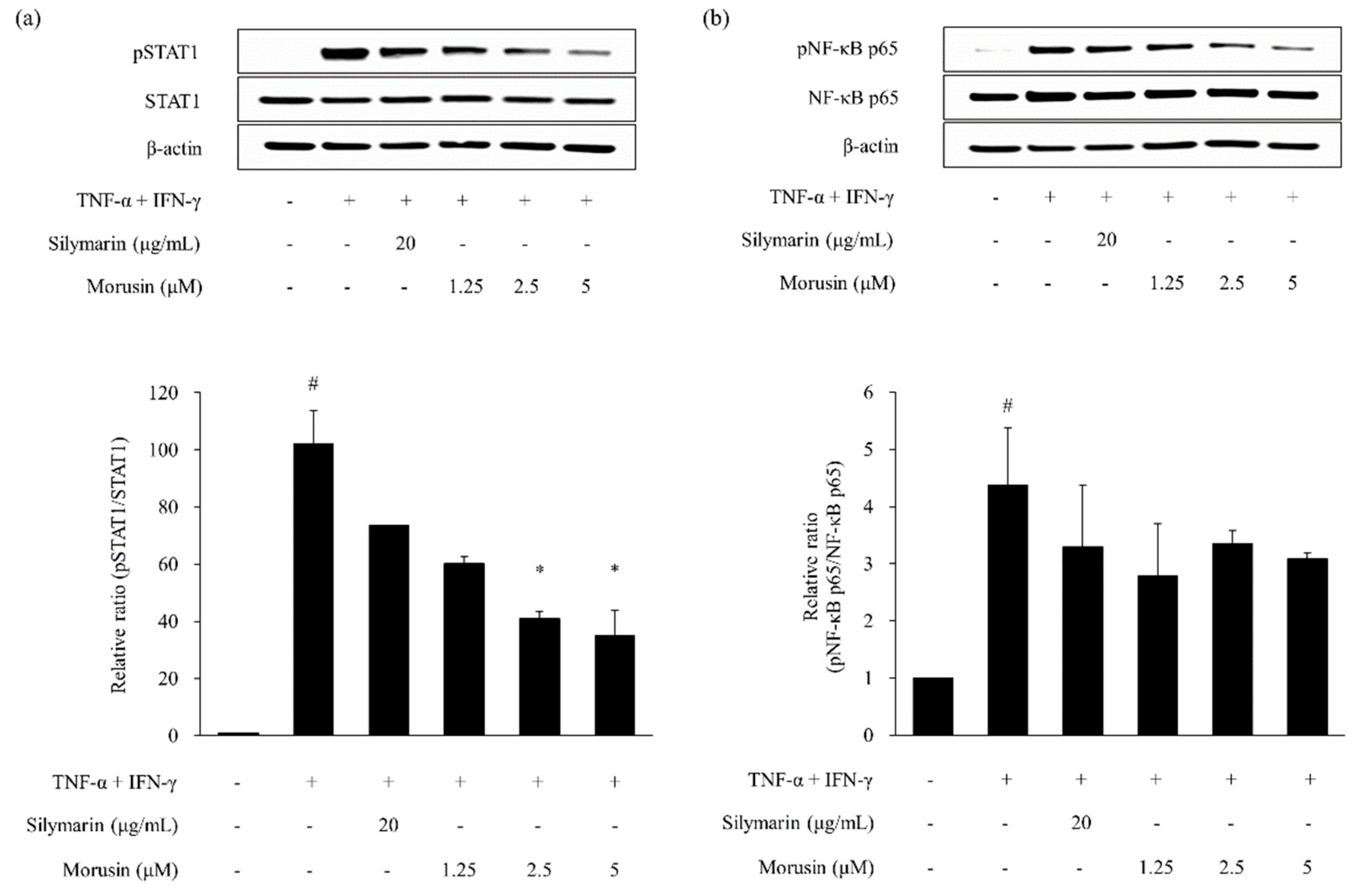
2.4. Effects of Kuwanon G and Morusin on Signal Transducer and Activator of Transcription 1 (STAT1) and Nuclear Transcription Factor-κB (NF-κB) Phosphorylation in HaCaT Keratinocytes
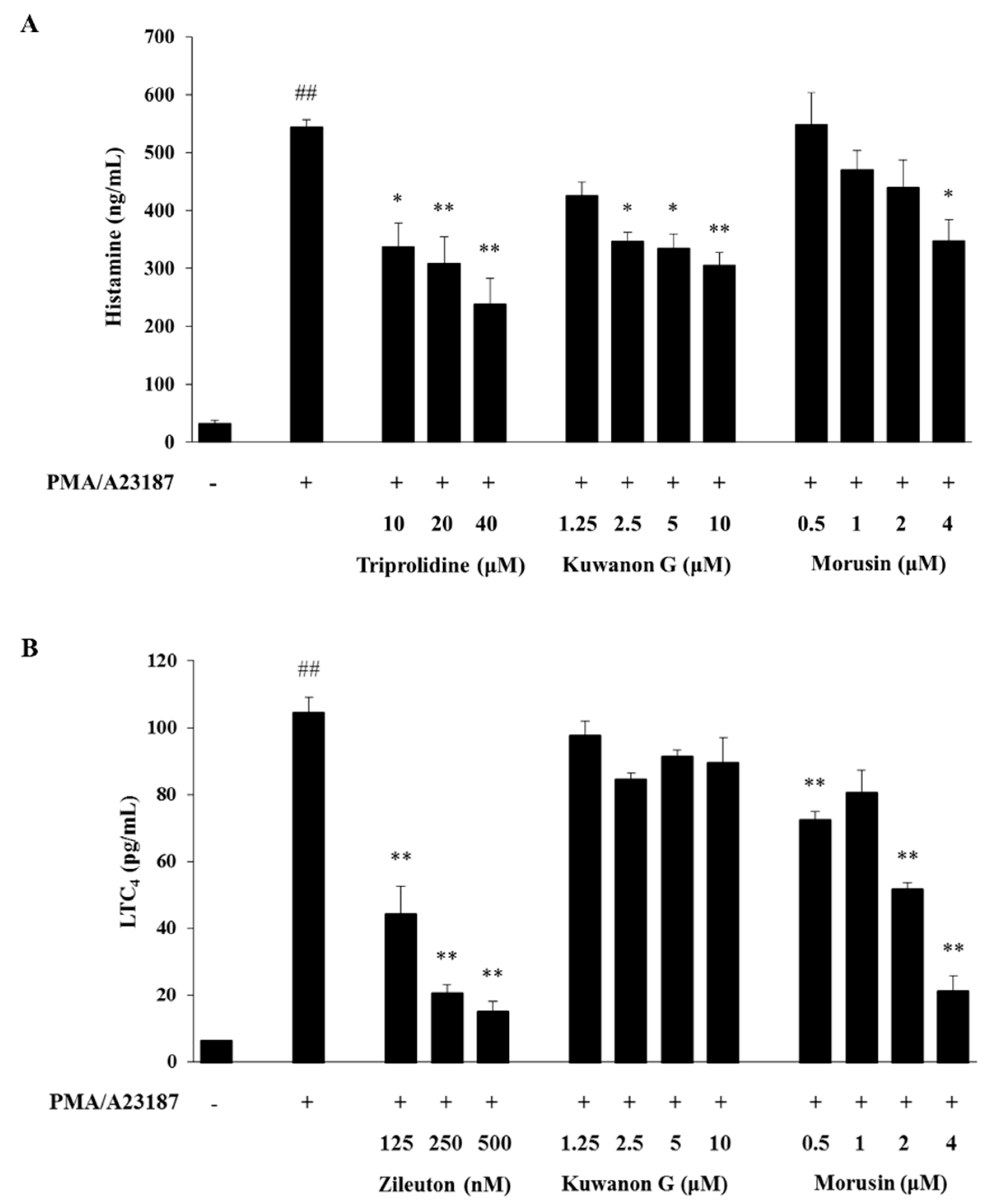
2.5. Effects of Kuwanon G and Morusin on the Production of Histamine and LTC4 in MC/9 Mast Cells
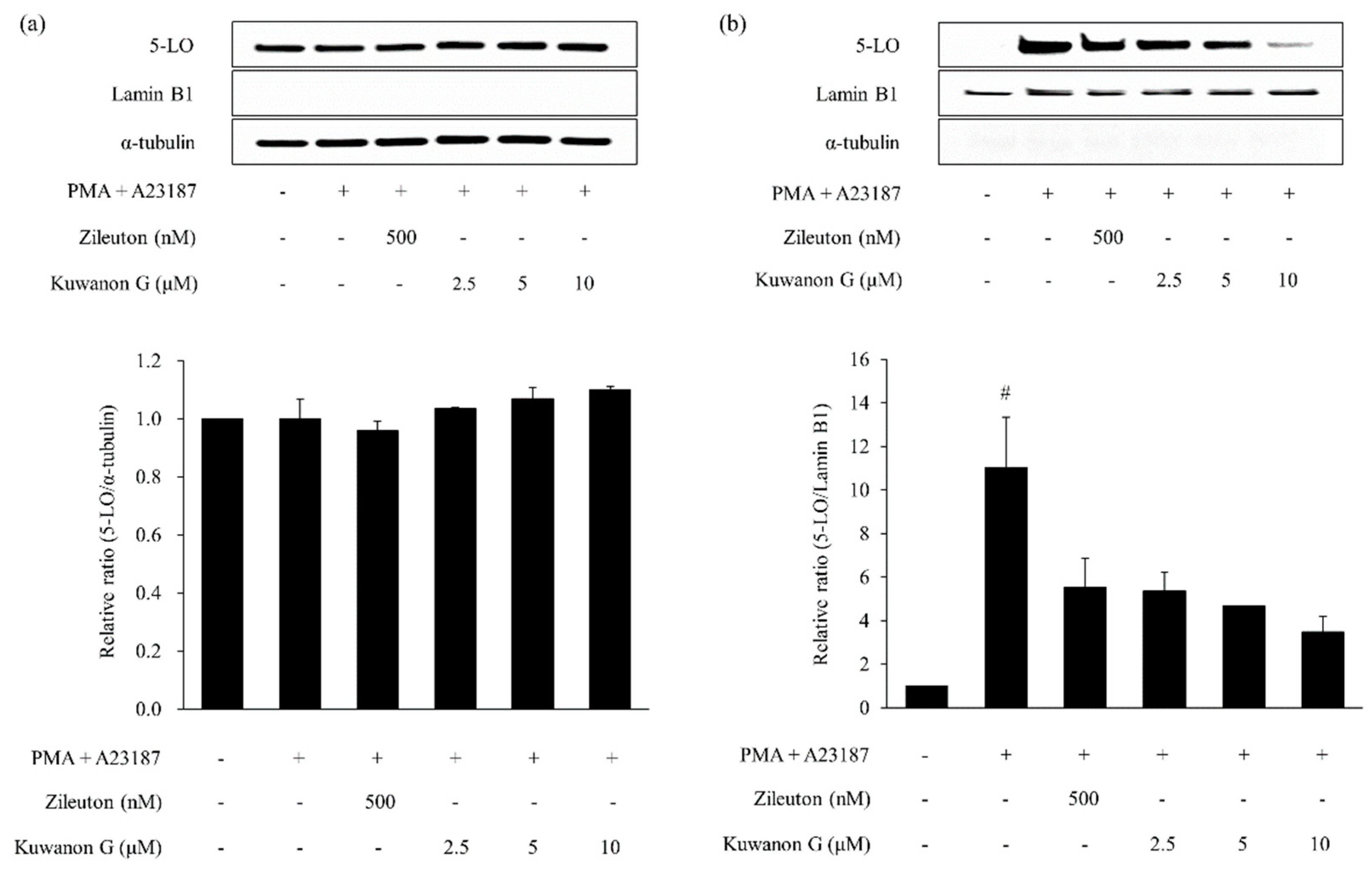
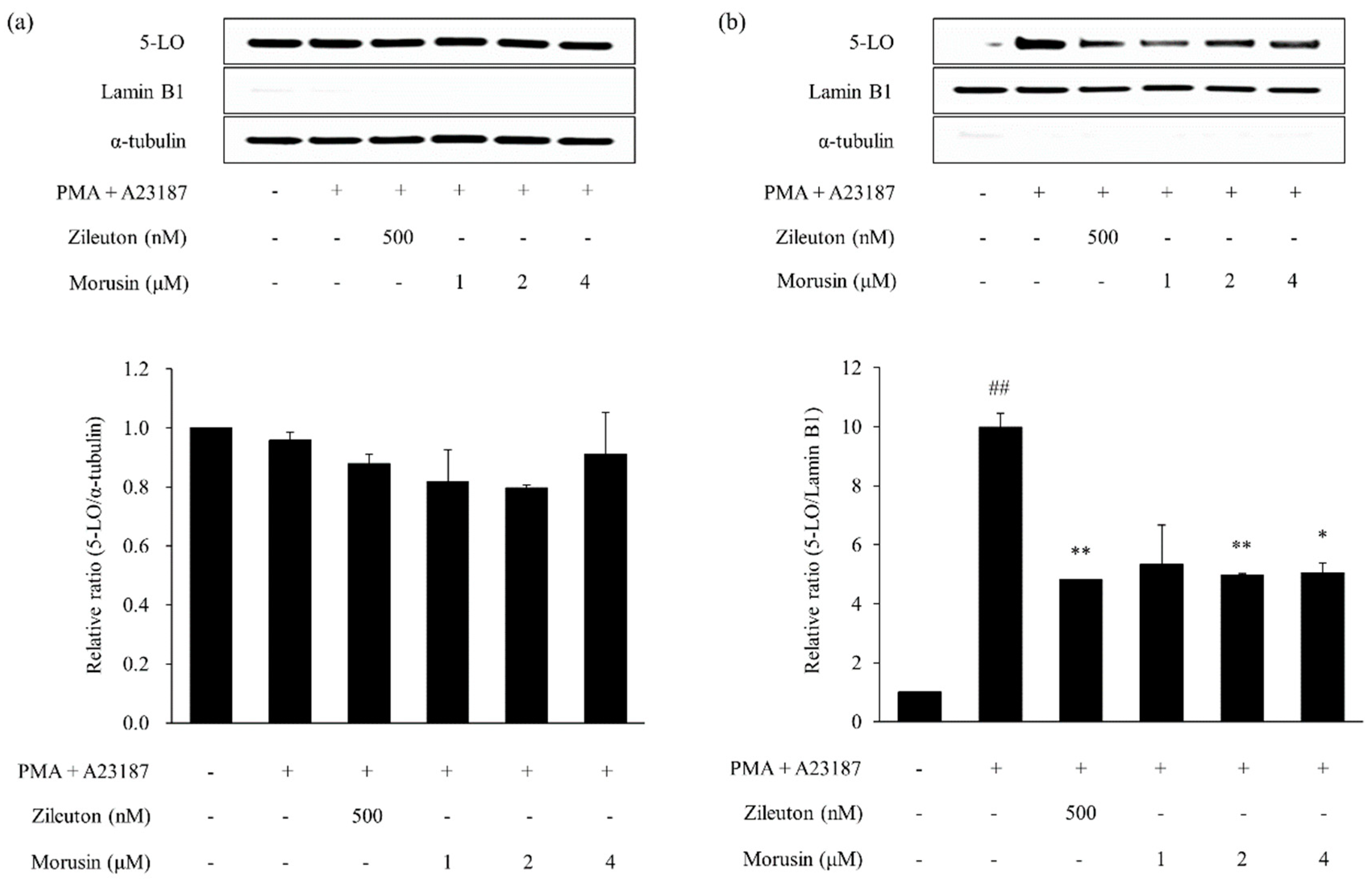
2.6. Effects of Kuwanon G and Morusin on the 5-lipoxygenase (5-LO) Activation in MC/9 Mast Cells
3. Discussion
4. Materials and Methods
4.1. Plant Materials
4.2. Chemicals and Reagents
4.3. Preparation of 70% Ethanol Extract of M. alba L.
4.4. HPLC Analysis of the Two Bioactive Marker Compounds in M. alba L.
4.5. Cell Culture
4.6. Cytotoxicity Assay
4.7. Measurement of Chemokine Levels
4.8. Measurement of Histamine and Leukotriene C4 Levels
4.9. Western Blotting
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lee, K.S.; Chun, S.Y.; Lee, M.G.; Kim, S.; Jang, T.J.; Nam, K.S. The prevention of TNF-α/IFN-γ mixture-induced inflammation in human keratinocyte and atopic dermatitis-like skin lesions in Nc/Nga mice by mineral-balanced deep sea water. Biomed. Pharmacother. 2018, 97, 1331–1340. [Google Scholar] [CrossRef] [PubMed]
- D’Auria, E.; Banderali, G.; Barberi, S.; Gualandri, L.; Pietra, B.; Riva, E.; Cerri, A. Atopic dermatitis: Recent insight on pathogenesis and novel therapeutic target. Asian Pac. J. Allergy Immunol. 2016, 34, 98–108. [Google Scholar] [PubMed]
- Bieber, T. Atopic dermatitis. N. Engl. J. Med. 2008, 358, 1483–1494. [Google Scholar] [CrossRef] [PubMed]
- Weidinger, S.; Novak, N. Atopic dermatitis. Lancet 2016, 387, 1109–1122. [Google Scholar] [CrossRef]
- Pease, J.E.; Williams, T.J. Chemokines and their receptors in allergic disease. J. Allergy Clin. Immunol. 2006, 118, 305–318. [Google Scholar] [CrossRef] [PubMed]
- Jahnz-Rozyk, K.; Targowski, T.; Paluchowska, E.; Owczarek, W.; Kucharczyk, A. Serum thymus and activation-regulated chemokine, macrophage-derived chemokine and eotaxin as markers of severity of atopic dermatitis. Allergy 2005, 60, 685–688. [Google Scholar] [CrossRef]
- Jung, M.R.; Lee, T.H.; Bang, M.H.; Kim, H.; Son, Y.; Chung, D.K.; Kim, J. Suppression of thymus- and activation-regulated chemokine (TARC/CCL17) production by 3-O-β-D-glucopyanosylspinasterol via blocking NF-κB and STAT1 signaling pathways in TNF-α and IFN-γ-induced HaCaT keratinocytes. Biochem. Biophys. Res. Commun. 2012, 427, 236–241. [Google Scholar] [CrossRef]
- Bernard, F.X.; Morel, F.; Camus, M.; Pedretti, N.; Barrault, C.; Garnier, J.; Lecron, J.C. Keratinocytes under Fire of Proinflammatory Cytokines: Bona Fide Innate Immune Cells Involved in the Physiopathology of Chronic Atopic Dermatitis and Psoriasis. J. Allergy (Cairo) 2012, 2012, 718725. [Google Scholar] [CrossRef]
- Park, J.H.; Kim, M.S.; Jeong, G.S.; Yoon, J. Xanthii fructus extract inhibits TNF-α/IFN-γ-induced Th2-chemokines production via blockade of NF-κB, STAT1 and p38-MAPK activation in human epidermal keratinocytes. J. Ethnopharmacol. 2015, 171, 85–93. [Google Scholar] [CrossRef]
- Ju, S.M.; Song, H.Y.; Lee, S.J.; Seo, W.Y.; Sin, D.H.; Goh, A.R.; Kang, Y.H.; Kang, I.J.; Won, M.H.; Yi, J.S.; et al. Suppression of thymus- and activation-regulated chemokine (TARC/CCL17) production by 1,2,3,4,6-penta-O-galloyl-beta-D-glucose via blockade of NF-kappaB and STAT1 activation in the HaCaT cells. Biochem. Biophys. Res. Commun. 2009, 387, 115–120. [Google Scholar] [CrossRef]
- Kwon, D.J.; Bae, Y.S.; Ju, S.M.; Goh, A.R.; Youn, G.S.; Choi, S.Y.; Park, J. Casuarinin suppresses TARC/CCL17 and MDC/CCL22 production via blockade of NF-κB and STAT1 activation in HaCaT cells. Biochem. Biophys. Res. Commun. 2012, 417, 1254–1259. [Google Scholar] [CrossRef] [PubMed]
- Yano, C.; Saeki, H.; Komine, M.; Kagami, S.; Tsunemi, Y.; Ohtsuki, M.; Nakagawa, H. Mechanism of Macrophage-Derived Chemokine/CCL22 Production by HaCaT Keratinocytes. Ann. Dermatol. 2015, 27, 152–156. [Google Scholar] [CrossRef] [PubMed]
- Barrett, N.A.; Austen, K.F. Innate cells and T helper 2 cell immunity in airway inflammation. Immunity 2009, 31, 425–437. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, T.; Ando, T.; Kimura, M.; Wilson, B.S.; Kawakami, Y. Mast cells in atopic dermatitis. Curr. Opin. Immunol. 2009, 21, 666–678. [Google Scholar] [CrossRef] [PubMed]
- Ikoma, A.; Rukwied, R.; Ständer, S.; Steinhoff, M.; Miyachi, Y.; Schmelz, M. Neuronal sensitization for histamine-induced itch in lesional skin of patients with atopic dermatitis. Arch. Dermatol. 2003, 139, 1455–1458. [Google Scholar] [CrossRef] [PubMed]
- Carr, W.W. Topical calcineurin inhibitors for atopic dermatitis: Review and treatment recommendations. Paediatr. Drugs 2013, 15, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Flohr, C.; Irvine, A.D. Systemic therapies for severe atopic dermatitis in children and adults. J. Allergy Clin. Immunol. 2013, 132, 774. [Google Scholar] [CrossRef]
- Lim, H.S.; Ha, H.; Lee, H.; Lee, J.K.; Lee, M.Y.; Shin, H.K. Morus alba L. suppresses the development of atopic dermatitis induced by the house dust mite in NC/Nga mice. BMC Complement. Altern. Med. 2014, 14, 139. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, D.; Mao, J.; Ke, X.X.; Zhang, R.; Yin, C.; Gao, N.; Cui, H. Morusin inhibits cell proliferation and tumor growth by down-regulating c-Myc in human gastric cancer. Oncotarget 2017, 8, 57187–57200. [Google Scholar]
- Yin, X.L.; Lv, Y.; Wang, S.; Zhang, Y.Q. Morusin suppresses A549 cell migration and induces cell apoptosis by downregulating the expression of COX-2 and VEGF genes. Oncol. Rep. 2018, 40, 504–510. [Google Scholar] [CrossRef]
- Gao, L.; Wang, L.; Sun, Z.; Li, H.; Wang, Q.; Yi, C.; Wang, X. Morusin shows potent antitumor activity for human hepatocellular carcinoma in vitro and in vivo through apoptosis induction and angiogenesis inhibition. Drug Des. Devel. Ther. 2017, 11, 1789–1802. [Google Scholar] [CrossRef]
- Li, H.; Wang, Q.; Dong, L.; Liu, C.; Sun, Z.; Gao, L.; Wang, X. Morusin suppresses breast cancer cell growth in vitro and in vivo through C/EBPβ and PPARγ mediated lipoapoptosis. J. Exp. Clin. Cancer Res. 2015, 34, 137. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.L.; Park, S.Y.; Kang, S.; Park, D.; Kim, S.H.; Um, J.Y.; Jang, H.J.; Lee, J.H.; Jeong, C.H.; Jang, J.H.; et al. Morusin induces cell death through inactivating STAT3 signaling in prostate cancer cells. Am. J. Cancer Res. 2014, 5, 289–299. [Google Scholar] [PubMed]
- Liu, X.X.; Zhang, X.W.; Wang, K.; Wang, X.Y.; Ma, W.L.; Cao, W.; Mo, D.; Sun, Y.; Li, X.Q. Kuwanon G attenuates atherosclerosis by upregulation of LXRα-ABCA1/ABCG1 and inhibition of NFκB activity in macrophages. Toxicol. Appl. Pharmacol. 2018, 341, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.W.; Kang, S.Y.; Kang, J.S.; Kim, A.R.; Woo, E.R.; Park, Y.K. Effect of Kuwanon G isolated from the root bark of Morus alba on ovalbumin-induced allergic response in a mouse model of asthma. Phytother. Res. 2014, 28, 1713–1719. [Google Scholar] [CrossRef] [PubMed]
- Park, K.M.; You, J.S.; Lee, H.Y.; Baek, N.I.; Hwang, J.K. Kuwanon G: An antibacterial agent from the root bark of Morus alba against oral pathogens. J. Ethnopharmacol. 2003, 84, 181–185. [Google Scholar] [CrossRef]
- Spergel, J.M.; Paller, A.S. Atopic dermatitis and the atopic march. J. Allergy Clin. Immunol. 2003, 112, 118–127. [Google Scholar] [CrossRef]
- Skibola, C.F.; Smith, M.T. Potential health impacts of excessive flavonoid intake. Free Radic. Biol. Med. 2000, 29, 375–383. [Google Scholar] [CrossRef]
- Arellano, F.M.; Wentworth, C.E.; Arana, A.; Fernández, C.; Paul, C.F. Risk of lymphoma following exposure to calcineurin inhibitors and topical steroids in patients with atopic dermatitis. J. Invest. Dermatol. 2007, 127, 808–816. [Google Scholar] [CrossRef] [PubMed]
- Sumiyoshi, K.; Nakao, A.; Setoguchi, Y.; Tsuboi, R.; Okumura, K.; Ogawa, H. TGF-beta/Smad signaling inhibits IFN-gamma and TNF-alpha-induced TARC (CCL17) production in HaCaT cells. J. Dermatol. Sci. 2003, 31, 53–58. [Google Scholar] [CrossRef]
- Vestergaard, C.; Bang, K.; Gesser, B.; Yoneyama, H.; Matsushima, K.; Larsen, C.G. A Th2 chemokine, TARC, produced by keratinocytes may recruit CLA+CCR4+ lymphocytes into lesional atopic dermatitis skin. J. Invest. Dermatol. 2000, 115, 640–646. [Google Scholar] [CrossRef] [PubMed]
- Han, E.H.; Hwang, Y.P.; Choi, J.H.; Yang, J.H.; Seo, J.K.; Chung, Y.C.; Jeong, H.G. Psidium guajava extract inhibits thymus and activation-regulated chemokine (TARC/CCL17) production in human keratinocytes by inducing heme oxygenase-1 and blocking NF-κB and STAT1 activation. Environ. Toxicol. Pharmacol. 2011, 32, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.H.; An, H.J.; Kim, J.Y.; Gwon, M.G.; Gu, H.; Lee, S.J.; Park, J.Y.; Park, K.D.; Han, S.M.; Kim, M.K.; et al. Apamin inhibits TNF-α- and IFN-γ-induced inflammatory cytokines and chemokines via suppressions of NF-κB signaling pathway and STAT in human keratinocytes. Pharmacol. Rep. 2017, 69, 1030–1035. [Google Scholar] [CrossRef]
- Ahn, S.; Siddiqi, M.H.; Aceituno, V.C.; Simu, S.Y.; Zhang, J.; Perez, Z.E.; Kim, Y.J.; Yang, D.C. Ginsenoside Rg5:Rk1 attenuates TNF-α/IFN-γ-induced production of thymus- and activation-regulated chemokine (TARC/CCL17) and LPS-induced NO production via downregulation of NF-κB/p38 MAPK/STAT1 signaling in human keratinocytes and macrophages. In Vitro Cell Dev. Biol. Anim. 2016, 52, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Jung, M.; Lee, T.H.; Oh, H.J.; Kim, H.; Son, Y.; Lee, E.H.; Kim, J. Inhibitory effect of 5,6-dihydroergosteol-glucoside on atopic dermatitis-like skin lesions via suppression of NF-κB and STAT activation. J. Dermatol. Sci. 2015, 79, 252–261. [Google Scholar] [CrossRef] [PubMed]
- Henderson, W.R., Jr. The role of leukotrienes in inflammation. Ann. Intern. Med. 1997, 121, 684–697. [Google Scholar] [CrossRef]
- Rezende, B.M.; Athayde, R.M.; Gonçalves, W.A.; Resende, C.B.; Teles de Tolêdo Bernardes, P.; Perez, D.A.; Esper, L.; Reis, A.C.; Rachid, M.A.; Castor, M.G.M.E.; et al. Inhibition of 5-lipoxygenase alleviates graft-versus-host disease. J. Exp. Med. 2017, 214, 3399–3415. [Google Scholar] [CrossRef]
- Byrum, R.S.; Goulet, J.L.; Griffiths, R.J.; Koller, B.H. Role of the 5-lipoxygenase-activating protein (FLAP) in murine acute inflammatory responses. J. Exp. Med. 1997, 185, 1065–1075. [Google Scholar] [CrossRef]
- Nedoszytko, B.; Sokołowska-Wojdyło, M.; Ruckemann-Dziurdzińska, K.; Roszkiewicz, J.; Nowicki, R.J. Chemokines and cytokines network in the pathogenesis of the inflammatory skin diseases: Atopic dermatitis, psoriasis and skin mastocytosis. Postepy. Dermatol. Alergol. 2014, 31, 84–91. [Google Scholar] [CrossRef]
- Brandt, E.B.; Sivaprasad, U. Th2 Cytokines and Atopic Dermatitis. J. Clin. Cell Immunol. 2011, 2, 110. [Google Scholar] [CrossRef]
- Shefler, I.; Taube, Z.; Medalia, O.; Sagi-Eisenberg, R. Basic secretagogues activate protein tyrosine phosphorylation and release of arachidonic acid in mast cells via a novel protein kinase C and phosphatidylinositol 3-kinase-dependent mechanism. Eur. J. Immunol. 1998, 28, 3468–3478. [Google Scholar] [CrossRef]
- Peng, Z.; Beaven, M.A. An essential role for phospholipase D in the activation of protein kinase C and degranulation in mast cells. J. Immunol. 2005, 174, 5201–5208. [Google Scholar] [CrossRef] [PubMed]
- Baranes, D.; Razin, E. Protein kinase C regulates proliferation of mast cells and the expression of the mRNAs of fos and jun proto-oncogenes during activation by IgE-Ag or calcium ionophore A23187. Blood 1991, 78, 2354–2364. [Google Scholar] [PubMed]
- Nishida, K.; Yamasaki, S.; Ito, Y.; Kabu, K.; Hattori, K.; Tezuka, T.; Nishizumi, H.; Kitamura, D.; Goitsuka, R.; Geha, R.S.; et al. FcɛRI-mediated mast cell degranulation requires calcium-independent microtubule-dependent translocation of granules to the plasma membrane. J. Cell Biol. 2005, 170, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Kim, O.K.; Nam, D.E.; Lee, M.; Kwon, H.O.; Park, J.; You, Y.; Kim, S.I.; Lee, J.; Jun, W. The Effects of Costaria costata Extracts on Atopic Dermatitis in an In Vitro Model. J. Med. Food 2016, 19, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.S.; Ha, H.; Lee, M.Y.; Jin, S.E.; Jeong, S.J.; Jeon, W.Y.; Shin, N.R.; Sok, D.E.; Shin, H.K. Saussurea lappa alleviates inflammatory chemokine production in HaCaT cells and house dust mite-induced atopic-like dermatitis in Nc/Nga mice. Food Chem. Toxicol. 2014, 63, 212–220. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, S.E.; Ha, H.; Shin, H.-K.; Seo, C.-S. Anti-Allergic and Anti-Inflammatory Effects of Kuwanon G and Morusin on MC/9 Mast Cells and HaCaT Keratinocytes. Molecules 2019, 24, 265. https://doi.org/10.3390/molecules24020265
Jin SE, Ha H, Shin H-K, Seo C-S. Anti-Allergic and Anti-Inflammatory Effects of Kuwanon G and Morusin on MC/9 Mast Cells and HaCaT Keratinocytes. Molecules. 2019; 24(2):265. https://doi.org/10.3390/molecules24020265
Chicago/Turabian StyleJin, Seong Eun, Hyekyung Ha, Hyeun-Kyoo Shin, and Chang-Seob Seo. 2019. "Anti-Allergic and Anti-Inflammatory Effects of Kuwanon G and Morusin on MC/9 Mast Cells and HaCaT Keratinocytes" Molecules 24, no. 2: 265. https://doi.org/10.3390/molecules24020265
APA StyleJin, S. E., Ha, H., Shin, H.-K., & Seo, C.-S. (2019). Anti-Allergic and Anti-Inflammatory Effects of Kuwanon G and Morusin on MC/9 Mast Cells and HaCaT Keratinocytes. Molecules, 24(2), 265. https://doi.org/10.3390/molecules24020265




