GC-MS and HS-SPME-GC×GC-TOFMS Determination of the Volatile Composition of Essential Oils and Hydrosols (By-Products) from Four Eucalyptus Species Cultivated in Tuscany
Abstract
1. Introduction
2. Results and Discussion
2.1. Chemical Composition of Essential Oils and Hydrolats by GC-MS
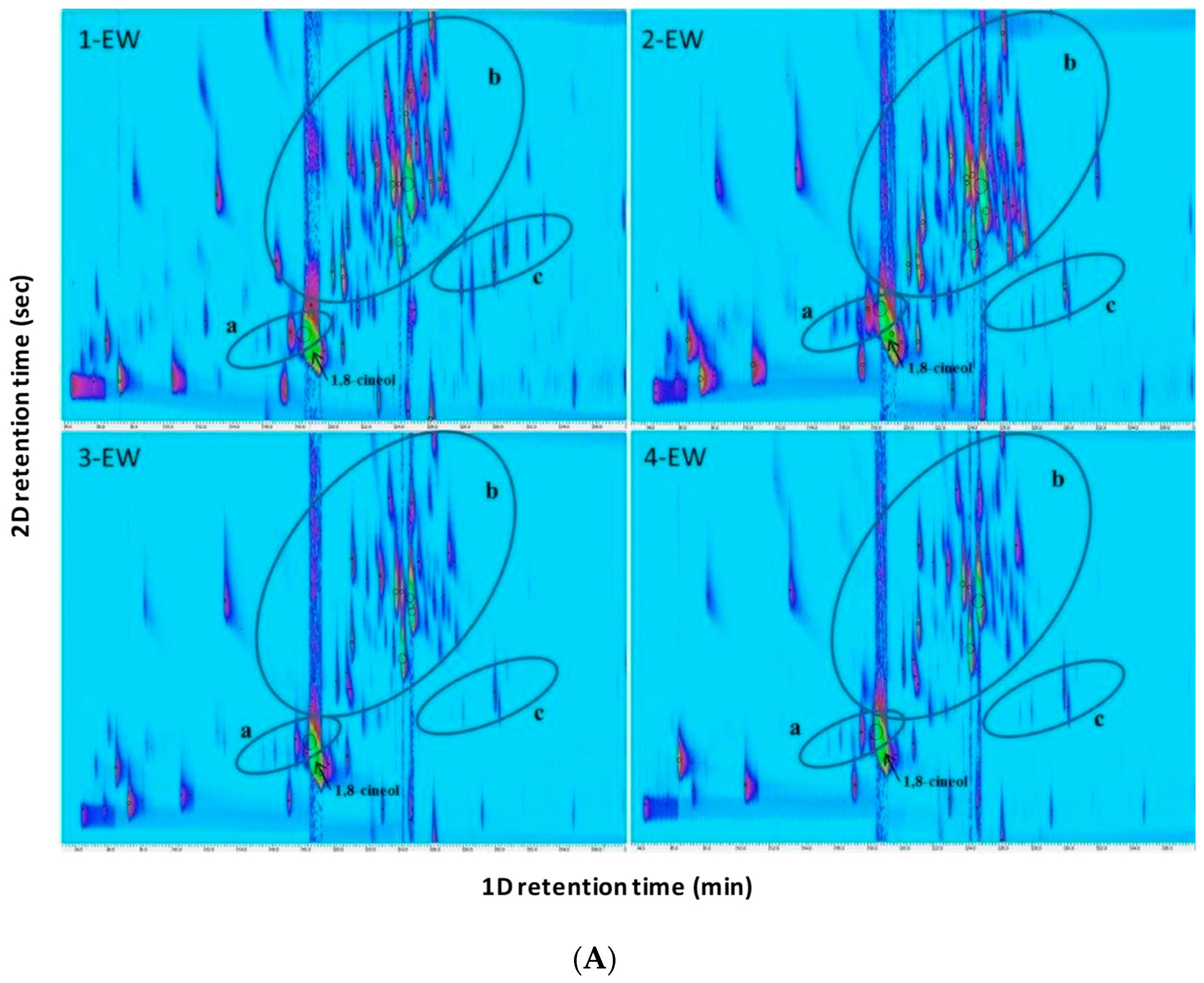
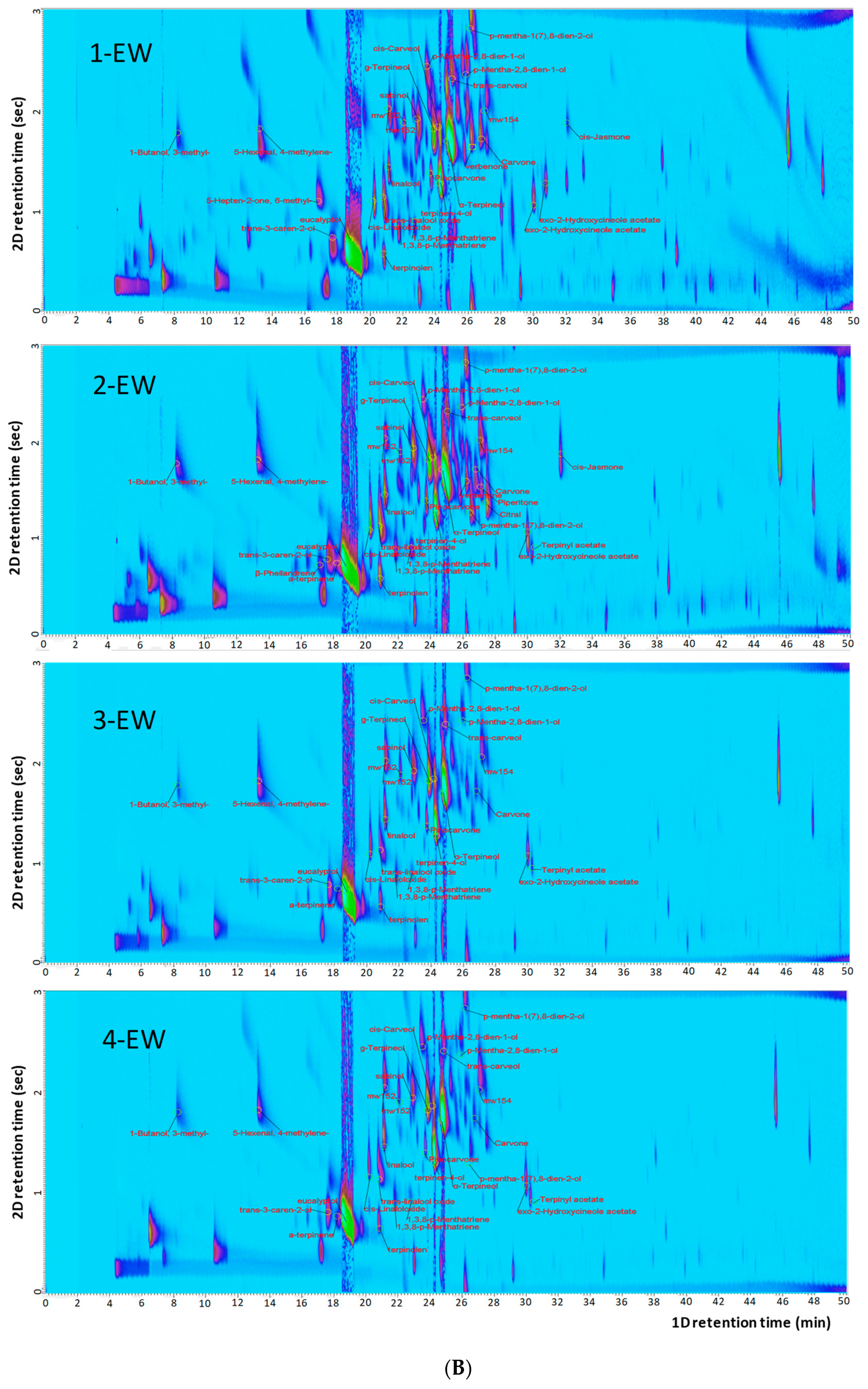
2.2. Fingerprint Analysis by HS-SPME-GC×GC-TOFMS
3. Materials and Methods
3.1. Chemicals
3.2. Plant Material
3.3. Obtaining of the Essential Oils and Hydrosols
3.4. Analysis of Essential Oils and Aromatic Waters
3.4.1. GC-MS Analysis
3.4.2. HS-SPME-GC×GC-TOFMS Analysis
3.5. Statistical Analysis
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Coppen, J.J.W. Eucalyptus: The Genus Eucalyptus; Taylor & Francis: London, UK, 2002. [Google Scholar]
- Ramezani, H.; Singh, H.P.; Batish, D.R.; Kohli, R.K. Antifungal activity of the volatile oil of Eucalyptus citriodora. Fitoterapia 2002, 73, 261–262. [Google Scholar] [CrossRef]
- Cermelli, C.; Fabio, A.; Fabio, G.; Quaglio, P. Effect of eucalyptus essential oil on respiratory bacteria and viruses. Curr. Microbiol. 2008, 56, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Mulyaningsih, S.; Sporer, F.; Zimmermann, S.; Reichling, J.; Wink, M. Synergistic properties of the terpenoids aromadendrene and 1,8-cineole from the essential oil of Eucalyptus globulus against antibiotic-susceptible and antibiotic-resistant pathogens. Phytomedicine 2010, 17, 1061–1066. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, A.K.; Malik, A. Antimicrobial potential and chemical composition of Eucalyptus globulus oil in liquid and vapour phase against food spoilage microorganisms. Food Chem. 2011, 26, 228–235. [Google Scholar] [CrossRef]
- Sadlon, A.E.; Lamson, D.W. Immune-Modifying and Antimicrobical Effects of Eucalyptus Oil and simple Inhalation Devices. Altern. Med. Rev. 2010, 15, 33–47. [Google Scholar]
- Bachir Raho, G.; Benali, M. Antibacterial activity of the essential oils from the leaves of Eucalyptus globulus against Escherichia coli and Staphylococcus aureus. Asian Pac. J. Trop. Biomed. 2012, 2, 739–742. [Google Scholar] [CrossRef]
- Lu, H.; Shao, X.; Cao, J.; Ou, C.; Pan, D. Antimicrobial activity of eucalyptus essential oil against Pseudomonas in vitro and potential application in refrigerated storage of pork meat. Int. J. Food Sci. Technol. 2016, 51, 994–1001. [Google Scholar] [CrossRef]
- Vilela, G.R.; de Almeida, G.S.; D’Arce, M.A.B.R.; Moraes, M.H.D.; Brito, J.O.; da Silva, M.F.G.F.; Silva, S.C.; de Stefano Piedade, S.M.; Calori-Domingues, M.A.; da Gloria, E.M. Activity of essential oil and its major compound, 1,8-cineole, from Eucalyptus globulus Labill., against the storage fungi Aspergillus flavus Link and Aspergillus parasiticus Speare. J. Stored Prod. Res. 2009, 45, 108–111. [Google Scholar] [CrossRef]
- Ormancey, X.; Sisalli, S.; Coutiere, P. Formulation of essential oils in functional perfumery. Parfum. Cosmet. Actual. 2001, 157, 30–40. [Google Scholar]
- Sacchetti, G.; Maietti, S.; Muzzoli, M.; Scaglianti, M.; Manfredini, S.; Radice, M.; Bruni, R. Comparative evaluation of 11 essential oils of different origin as functional antioxidants, antiradicals and antimicrobials in foods. Food Chem. 2005, 91, 621–632. [Google Scholar] [CrossRef]
- Paolini, J.; Leandri, C.; Desjobert, J.M.; Barboni, T.; Costa, J. Comparison of liquid–liquid extraction with headspace methods for the characterization of volatile fractions of commercial hydrolats from typically Mediterranean species. J. Chromatogr. A 2008, 1193, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Edris, A.E. Identification and absolute quantification of the major water-soluble aroma components isolated from the hydrosols of some aromatic plants. J. Essent. Oil-Bear. Plants 2009, 12, 155–161. [Google Scholar] [CrossRef]
- Ndiaye, H.B.; Diop, M.B.; Gueye, M.T.; Ndiaye, I.; Diop, S.M.; Fauconnier, M.L.; Lognay, G. Characterization of essential oils and hydrosols from senegalese Eucalyptus camaldulensis Dehnh. J. Essent. Oil-Bear. Plants 2018, 30, 1–11. [Google Scholar] [CrossRef]
- Price, L.; Price, S. Understanding Hydrolats: The Specific Hydrosols for Aromatherapy; Churchill Livingstone/Elsevier: Amsterdam, The Nederlands, 2004; p. 96. [Google Scholar]
- Zrira, S.; Bessiere, J.M.; Menut, C.; Elamrani, A.; Benjilali, B. Chemical composition of the essential oil of nine Eucalyptus species growing in Morocco. Flavour Fragr. J. 2004, 19, 172–175. [Google Scholar] [CrossRef]
- Elaissi, A.; Hadj Salah, K.; Mabrouk, S.; Larb, K.K.; Chemli, R.; Harzallah-Skhiri, F. Antibacterial activity and chemical composition of 20 Eucalyptus species essential oils. Food Chem. 2011, 129, 1427–1434. [Google Scholar] [CrossRef]
- Kahla, Y.; Zouari-Bouassida, K.; Rezgui, F.; Trigui, M.; Tounsi, S. Efficacy of Eucalyptus cinerea as a Source of Bioactive Compounds for Curative Biocontrol of Crown Gall Caused by Agrobacterium tumefaciens Strain B6. BioMed Res. Int. 2017. [Google Scholar] [CrossRef] [PubMed]
- Ieri, F.; Cecchi, L.; Vignolini, P.; Belcaro, M.F.; Romani, A. HPLC/DAD, GC/MS and GC/GC/TOF analysis of Lemon balm (Melissa officinalis L.) sample as standardized raw material for food and nutraceuticals uses. Adv. Hortic. Sci. 2017, 3, 141–147. [Google Scholar] [CrossRef]
- Wong, Y.F.; Perlmutter, P.; Marriott, P.J. Untargeted metabolic profiling of Eucalyptus spp. leaf oils using comprehensive two-dimensional gas chromatography with high resolution mass spectrometry: Expanding the metabolic profile. Metabolomics 2017, 13, 46. [Google Scholar] [CrossRef]
- Calamai, L.; Villanelli, F.; Bartolucci, G.; Pieraccini, G.; Moneti, G. Sample preparation for direct ms analysis of food. In Comprehensive sampling and sample preparation; Elsevier: Amsterdam, The Nederlands, 2012. [Google Scholar]
- Oliver-Pozo, C.; Aparicio-Ruiz, R.; Romero, I.; Garcia-Gonzalez, D.L. Analysis of volatile markers for virgin olive oil aroma defects by SPME-GC/FID: Possible sources of incorrect data. J. Agric. Food Chem. 2015, 63, 10477–10483. [Google Scholar] [CrossRef]
- Fortini, M.; Migliorini, M.; Cherubini, C.; Cecchi, L.; Calamai, L. Multiple internal standard normalization for improving HS-SPME-GC-MS quantitation in virgin olive oil volatile compounds (VOO-VOCs) profile. Talanta 2017, 165, 641–652. [Google Scholar] [CrossRef]
- May, F.; Ash, J. An assessment of the allelopathic potential of Eucalyptus. Aust. J. Botany 1990, 38, 245–254. [Google Scholar] [CrossRef]
- Kohli, R.K.; Batish, D.R.; Singh, H.P. Eucalypt oils for the control of Parthenium (Perthenium hysterophorus L.). Crop Prot. 1998, 17, 119–122. [Google Scholar] [CrossRef]
- Hummelbrunner, L.A.; Isman, M.B. Acute, sublethal, antifeedant, and synergistic effects of monoterpenoid essential oil compounds on the tobacco cutworm, Spodoptera litura (Lep., Noctuidae). J. Agric. Food Chem. 2001, 49, 715–720. [Google Scholar] [CrossRef] [PubMed]
- Nerio, L.S.; Olivero-Verbel, J.; Stashenko, E. Repellent activity of essential oils: A review. Bioresour. Technol. 2010, 101, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Spietelun, A.; Marcinkowski, L.; De la Guardia, M.; Namieśnik, J. Review recent developments and future trends in solid phase microextraction techniques towards green analytical chemistry. J. Chromatogr. A 2013, 1321, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Hantao, W.L.; Toledo, B.R.; de Lima Ribeiro, F.A.; Pizetta, M.; Geraldi Pierozzi, C.; Luiz Furtado, E.; Augusto, F. Comprehensive two-dimensional gas chromatography combined to multivariate data analysis for detection of disease-resistant clones of Eucalyptus. Talanta 2013, 116, 1079–1084. [Google Scholar] [CrossRef]
- Cordero, C.; Bicchi, C.; Rubiol, P. Group-type and fingerprint analysis of roasted food matrices (coffee and hazelnut samples) by comprehensive two-dimensional gas chromatography. J. Agric. Food Chem. 2008, 56, 7655–7666. [Google Scholar] [CrossRef]
- Von Mühlen, C.; Alcaraz Zini, C.; Bastos Caramaob, E.; Marriott, P.J. Comparative study of Eucalyptus dunnii volatile oil composition using retention indices and comprehensive two-dimensional gas chromatography coupled to time-of-flight and quadrupole mass spectrometry. J. Chromatogr. A 2008, 1200, 34–42. [Google Scholar] [CrossRef]
- Cordero, C.; Atsbaha Zebelo, S.; Gnavi, G.; Griglione, A.; Bicchi, C.; Maffei, M.E.; Biolo, P. HS-SPME-GC×GC-qMS volatile metabolite profiling of Chrysolina herbacea frass and Mentha spp. Leaves. Anal. Bioanal. Chem. 2012, 402, 1941–1952. [Google Scholar] [CrossRef]
- Van den Dool, H.; Kratz, P.D. A generalization of the retention index system including linear temperature programmed gas-liquid partition chromatography. J. Chromatogr. 1963, 11, 463–471. [Google Scholar] [CrossRef]
- Chemistry WebBook. Available online: http://www.nist.gov/index.html (accessed on 1 December 2018).
Sample Availability: Samples of the compounds are not available from the authors. |
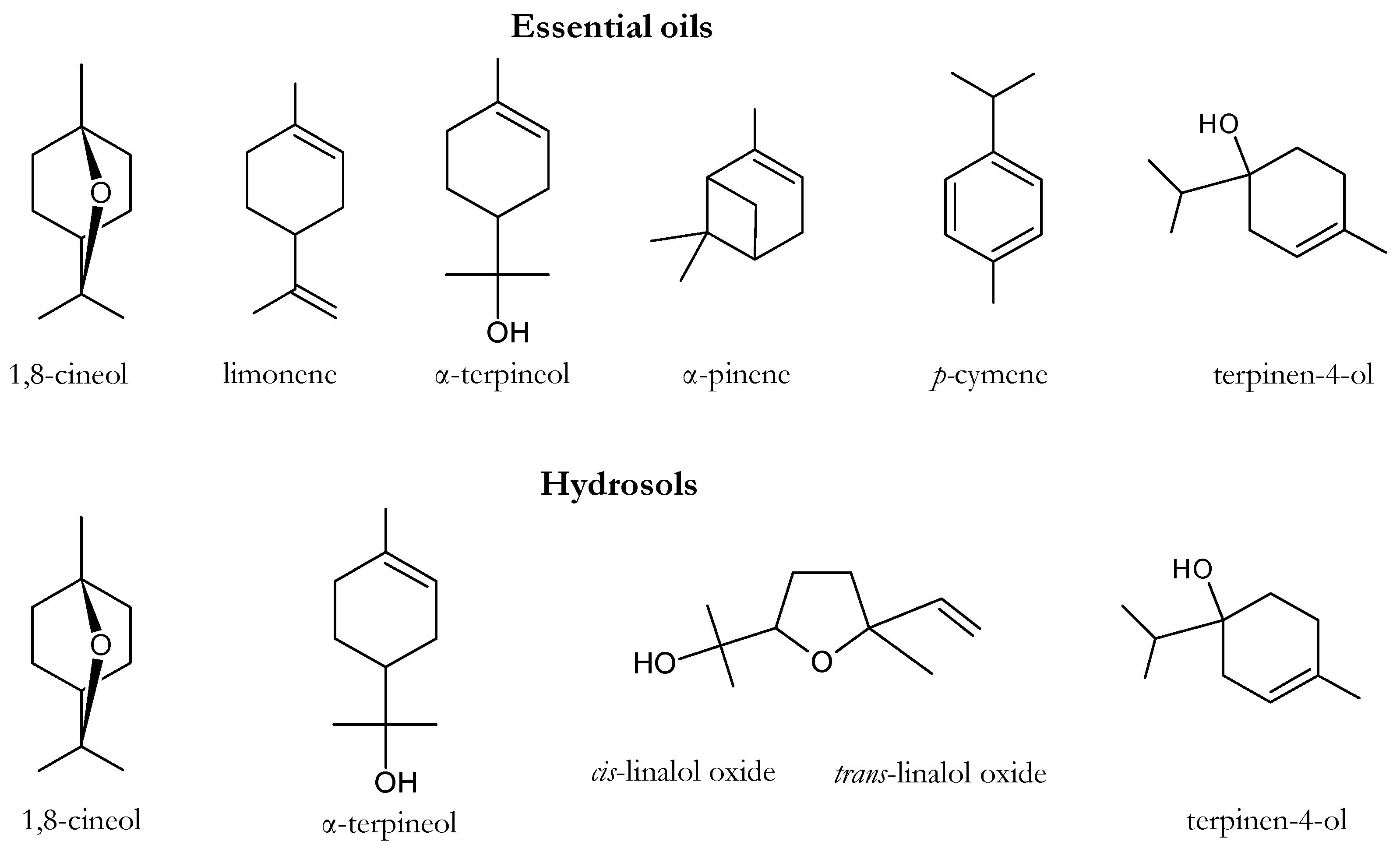

| n° | Compound | RIcal | RIref | Hydrosols (Area %) | Essential Oils (Area %) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1-16-EW | 2-16-EW | 3-16-EW | 4-16-EW | 1-17-EW | 1-1617-EW | 1-16-EO | 2-16-EO | 3-16-EO | 4-16-EO | 1-17-EO | 1-1617-EO | ||||
| Monoterpene Hydrocarbons | 0.22 | 0.02 | 0.00 | 0.02 | 0.01 | 0.00 | 8.05 | 10.76 | 7.10 | 8.34 | 6.74 | 7.54 | |||
| 1 | α-pinene | 1030 | 1026 | 0.09 | - | - | - | - | - | 1.20 b | 4.45 e | 2.38 d | 2.15 c | 0.95 a | 1.12 ab |
| 2 | camphene | 1067 | 1065 | - | - | - | - | - | - | 0.03 bc | 0.04 c | 0.02 ab | 0.02 ab | 0.01 a | 0.02 ab |
| 3 | β-pinene | 1122 | 1118 | - | - | - | - | - | - | 0.11 ab | 0.17 cd | 0.14 bc | 0.19 d | 0.09 a | 0.10 a |
| 7 | β-myrcene | 1168 | 1167 | - | - | - | tr | tr | - | 0.12 a | 0.22 c | 0.17 b | 0.25 c | 0.09 a | 0.11 a |
| 8 | α-phellandrene | 1177 | 1177 | - | - | - | - | - | - | 0.15 b | 0.13 b | 0.04 a | 0.01 a | 0.15 b | 0.14 b |
| 12 | Limonene | 1214 | 1210 | 0.08 y | 0.02 z | tr | 0.02 z | 0.01 z | - | 3.60 b | 4.65 d | 3.66 b | 4.29 c | 3.00 a | 3.41 b |
| 14 | Z-ocimene | 1241 | 1242 | - | - | - | - | - | - | 0.36 c | 0.17 b | 0.07 a | 0.07 a | 0.45 d | 0.39 c |
| 15 | γ-terpinene | 1259 | 1254 | tr | - | - | - | - | Tr | 0.54 b | 0.13 a | 0.10 a | 0.07 a | 0.48b | 0.50 b |
| 16 | p-cymene | 1286 | 1281 | 0.05 | - | - | tr | tr | - | 1.78 e | 0.73 b | 0.50 a | 1.23 c | 1.42 cd | 1.63 de |
| 20 | alloocimene | 1383 | 1377 | - | - | - | - | - | - | 0.15 d | 0.07 b | 0.03 a | 0.04 a | 0.12 c | 0.13 c |
| Oxygenated Monoterpenes | 98.62 | 98.50 | 98.29 | 97.64 | 98.91 | 98.80 | 91.30 | 88.74 | 91.99 | 90.60 | 92.71 | 91.89 | |||
| 11 | 2,3-dehydro-1,8-cineole | 1202 | 1197 | - | - | - | - | - | - | - | 0.09 c | 0.08 bc | 0.07 b | 0.05 a | 0.05 a |
| 13 | 1,8-cineol | 1225 | 1221 | 89.53 y | 88.40 z | 90.78 x | 89.17 zy | 90.22 yx | 89.35 zy | 87.06 b | 83.80 a | 87.72 bc | 85.14 a | 88.66 c | 87.75 bc |
| 23 | cis-linalool oxide (furanoid) | 1453 | 1453 | 0.5 zy | 0.77 x | 0.47 zy | 0.44 z | 0.52 zy | 0.55 y | - | - | - | - | - | - |
| 25 | trans-linalool oxide (furanoid) | 1481 | 1482 | 0.49 y | 0.68 x | 0.36 z | 0.43 zy | 0.46 zy | 0.52 y | - | - | - | - | - | - |
| 26 | linalool | 1545 | 1544 | - | - | - | - | - | - | 0.05 b | 0.09 c | 0.09 c | 0.11 d | 0.02 a | 0.03 a |
| 27 | fenchyl alcohol | 1595 | 1571 | tr | tr | - | tr | tr | - | 0.06 b | 0.08 c | 0.05 b | 0.06 b | 0.06 b | 0.03 a |
| 28 | pinocarvone | 1600 | 1575 | - | - | - | - | - | - | 0.06 b | 0.03 a | 0.05 b | 0.06 b | 0.06 b | 0.05 b |
| 29 | terpinen-4-ol | 1617 | 1612 | 0.92 y | 0.94 yx | 1.01 x | 1.37 w | 0.75 z | 0.87 y | 0.51 cd | 0.47 bc | 0.56 d | 0.72 e | 0.39 a | 0.44 b |
| 31 | cis-p-mentha-2,8-dienol | 1637 | 1642 | 0.07 zy | 0.06 z | 0.12 x | 0.08 y | 0.12 x | 0.08 y | 0.02 a | 0.03 ab | 0.06 c | 0.08 d | 0.04 b | 0.04 b |
| 33 | trans-pinocarveol | 1683 | 1659 | 0.17 z | 0.24 yx | 0.23 yx | 0.24 yx | 0.25 x | 0.21 y | 0.09 ab | 0.13 c | 0.11 bc | 0.08 a | 0.10 ab | 0.09 ab |
| 34 | trans-p-mentha-2,8-dienol | 1679 | 1670 | 0.11 z | 0.10 z | 0.11 z | 0.12 z | 0.13 z | 0.10 z | - | - | - | - | - | - |
| 35 | terpineol isomer | 1682 | - | 0.25 z | 0.40 y | 0.37 y | 0.42 y | 0.25 z | 0.24 z | 0.14 ab | 0.19 c | 0.17 bc | 0.23 d | 0.12 a | 0.14 ab |
| 36 | citral | 1698 | 1695 | 0.17 x | 0.27 w | 0.19 x | 0.10 z | 0.14 y | 0.19 x | 0.05 ab | 0.07 bc | 0.08 c | 0.05 ab | 0.04 a | 0.05 ab |
| 37 | α-terpineol | 1707 | 1704 | 6.02 x | 6.24 x | 4.19 z | 4.50 z | 5.53 y | 6.21 x | 3.11 c | 2.77 b | 2.00 a | 2.20 a | 3.02 bc | 3.08 bc |
| 38 | borneol | 1718 | 1715 | tr | 0.05 x | - | 0.04 y | 0.02 z | - | - | - | - | - | - | - |
| 39 | α-terpinyl acetate | 1722 | 1721 | - | - | - | - | - | - | 0.02 a | 0.90 b | 0.92 b | 1.71 c | 0.02 a | - |
| 40 | cis-carveol | 1850 | 1847 | 0.14 yx | 0.11 z | 0.12 zy | 0.10 z | 0.18 w | 0.16 xw | - | - | - | - | - | - |
| 42 | exo-2-hydroxycineole | 1870 | 1870 | 0.08 z | 0.13 y | 0.21 x | 0.52 w | 0.10 zy | 0.11 zy | - | - | - | - | - | - |
| 43 | cis-p-mentha-1(7),8-dien-2-ol | 1905 | 1888 | 0.17 y | 0.11 z | 0.13 z | 0.11 z | 0.24 w | 0.21 x | 0.12 bc | 0.09 ab | 0.09 ab | 0.07 a | 0.13 c | 0.14 c |
| Sesquiterpene Hydrocarbons | - | - | - | - | - | - | 0.09 | 0.10 | 0.47 | 0.63 | 0.02 | 0.03 | |||
| 30 | β-caryophyllene | 1631 | 1625 | - | - | - | - | - | - | 0.07 b | 0.07 b | 0.03 a | 0.03 a | 0.02 a | 0.03 a |
| 32 | alloaromandrene | 1640 | 1645 | - | - | - | - | - | - | 0.02 a | 0.02 a | 0.44 b | 0.60 c | - | - |
| Aromatic Monoterpenes | - | - | - | - | - | - | 0.03 | 0.04 | 0.02 | 0.03 | 0.04 | 0.03 | |||
| 24 | p-cymenene | 1455 | 1455 | - | - | - | - | - | - | 0.03 b | 0.04 c | 0.02 a | 0.03 b | 0.04 c | 0.03 b |
| Oxygenated Aromatic Monoterpenes | 0.06 | 0.11 | 0.08 | 0.10 | 0.10 | 0.08 | - | - | - | - | - | - | |||
| 41 | p-cymen-8-ol | 1857 | 1869 | 0.06 z | 0.11 x | 0.08 zy | 0.10 yx | 0.10 yx | 0.08 zy | - | - | - | - | - | - |
| Ester | - | - | - | - | - | - | 0.04 | - | - | - | 0.03 | 0.05 | |||
| 4 | isoamyl acetate | 1125 | 1126 | - | - | - | - | - | - | 0.04 a | - | - | - | 0.03 a | 0.05 a |
| Ketones | 0.21 | 0.22 | 0.20 | 0.23 | 0.20 | 0.27 | 0.24 | 0.20 | 0.25 | 0.24 | 0.22 | 0.22 | |||
| 5 | heptan-4-one | 1132 | - | - | - | - | - | - | - | 0.06 c | 0.03 a | 0.04 ab | 0.04 ab | 0.04 ab | 0.05 bc |
| 6 | heptan-3-one | 1161 | 1163 | 0.05 z | 0.06 zy | 0.06 zy | 0.07 yx | 0.05 z | 0.08 x | 0.07 a | 0.06 a | 0.07 a | 0.06 a | 0.06 a | 0.07 a |
| 9 | heptan-2-one | 1190 | 1185 | 0.12 z | 0.16 z | 0.14 z | 0.16 z | 0.13 z | 0.16 z | 0.12 a | 0.11 a | 0.14 a | 0.14 a | 0.12 a | 0.11 a |
| 19 | 6-methylhept-5-en-2-one | 1346 | 1338 | 0.04 x | - | - | - | 0.02 z | 0.03 y | tr | - | - | - | - | - |
| aldehyde | - | - | tr | - | - | - | - | - | - | - | - | - | |||
| 22 | nonanal | 1405 | 1401 | - | - | tr | - | - | - | - | - | - | - | - | - |
| Alcohols | 0.89 | 1.15 | 1.43 | 2.01 | 0.78 | 0.85 | 0.14 | 0.14 | 0.15 | 0.14 | 0.14 | 0.16 | |||
| 10 | 3-methylbutanol | 1198 | 1210 | 0.46 z | 0.76 y | 0.68 y | 1.07 x | 0.45 z | 0.49 z | - | - | - | - | - | - |
| 17 | heptan-3-ol | 1290 | - | 0.07 zy | 0.08 yx | 0.06 z | 0.08 yx | 0.06 z | 0.09 x | 0.07 a | 0.06 a | 0.08 a | 0.07 a | 0.06 a | 0.08 a |
| 18 | heptan-2-ol | 1314 | 1318 | 0.06 z | 0.09 x | 0.08 yx | 0.09 x | 0.07 zy | 0.08 yx | 0.07 a | 0.08 a | 0.08 a | 0.07 a | 0.07 a | 0.08 a |
| 21 | Z-hex-3-en-1-ol | 1384 | 1384 | 0.14 x | 0.07 z | 0.09 zy | 0.24 w | 0.12 yx | 0.13 yx | - | - | - | - | - | - |
| 44 | 2-phenylethanol | 1928 | 1924 | 0.16 y | 0.15 y | 0.52 x | 0.53 x | 0.08 z | 0.06 z | - | - | - | - | - | - |
| Sample | Kind of Sample | 1,8-cineole (g/L) |
|---|---|---|
| 1-16-EW | hydrosol | 1.58 a |
| 2-16-EW | hydrosol | 1.45 b |
| 3-16-EW | hydrosol | 1.52 a |
| 4-16-EW | hydrosol | 0.74 e |
| 1-17-EW | hydrosol | 0.86 d |
| 1-1617-EW | hydrosol | 1.20 c |
| Sample Name | Kind of Sample | Eucalyptus Species | Year | Yields % |
|---|---|---|---|---|
| 1-16-EW | aromatic water | E. parvula L.A.S. Johnson & K.D. Hill | 2016 | |
| 2-16-EW | aromatic water | E. cinerea F. Muell | 2016 | |
| 3-16-EW | aromatic water | E. pulverulenta Sims | 2016 | |
| 4-16-EW | aromatic water | E. pulverulenta baby blue Sims | 2016 | |
| 1-17-EW | aromatic water | E. parvula L.A.S. Johnson & K.D. Hill | 2017 | |
| 1-1617-EW | aromatic water | E. parvula L.A.S. Johnson & K.D. Hill | 2016–2017 | |
| 1-16-EO | essential oil | E. parvula L.A.S. Johnson & K.D. Hill | 2016 | 1.2 |
| 2-16-EO | essential oil | E. cinerea F. Muell | 2016 | 1.1 |
| 3-16-EO | essential oil | E. pulverulenta Sims | 2016 | 1.1 |
| 4-16-EO | essential oil | E. pulverulenta baby blue Sims | 2016 | 1.1 |
| 1-17-EO | essential oil | E. parvula L.A.S. Johnson & K.D. Hill | 2017 | 1.3 |
| 1-1617-EO | essential oil | E. parvula L.A.S. Johnson & K.D. Hill | 2016–2017 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ieri, F.; Cecchi, L.; Giannini, E.; Clemente, C.; Romani, A. GC-MS and HS-SPME-GC×GC-TOFMS Determination of the Volatile Composition of Essential Oils and Hydrosols (By-Products) from Four Eucalyptus Species Cultivated in Tuscany. Molecules 2019, 24, 226. https://doi.org/10.3390/molecules24020226
Ieri F, Cecchi L, Giannini E, Clemente C, Romani A. GC-MS and HS-SPME-GC×GC-TOFMS Determination of the Volatile Composition of Essential Oils and Hydrosols (By-Products) from Four Eucalyptus Species Cultivated in Tuscany. Molecules. 2019; 24(2):226. https://doi.org/10.3390/molecules24020226
Chicago/Turabian StyleIeri, Francesca, Lorenzo Cecchi, Elena Giannini, Clarissa Clemente, and Annalisa Romani. 2019. "GC-MS and HS-SPME-GC×GC-TOFMS Determination of the Volatile Composition of Essential Oils and Hydrosols (By-Products) from Four Eucalyptus Species Cultivated in Tuscany" Molecules 24, no. 2: 226. https://doi.org/10.3390/molecules24020226
APA StyleIeri, F., Cecchi, L., Giannini, E., Clemente, C., & Romani, A. (2019). GC-MS and HS-SPME-GC×GC-TOFMS Determination of the Volatile Composition of Essential Oils and Hydrosols (By-Products) from Four Eucalyptus Species Cultivated in Tuscany. Molecules, 24(2), 226. https://doi.org/10.3390/molecules24020226








