Nano-and Micromotors Designed for Cancer Therapy
Abstract
1. Introduction
- Reduced drug concentration due to specific targeting possibilities, which results in less side effects and in lower costs.
- A reduced amount of active pharmaceuticals consequentially leads to a reduced amount of waste products. Generally, pharmaceuticals are metabolized or simply excreted by the body after their use, leading to high concentrations in sewage waters, which are very often difficult to remediate in water treatment plants. This is not only the case for cancer medicine, but due to the high cytotoxicity of these compounds are especially dangerous for living organisms in aquatic systems [39].
- Compared to passive drug delivery agents, the use of nano- and micromotors bears the advantage, that propulsion is independent from the blood flow. Smart engineered drug carriers display additional advantages, especially when combined with other approaches, such as encapsulation, targeting moieties on the surface, cell or gene delivery. Drug delivery with on-demand site-specific release becomes especially useful for pharmaceuticals causing severe side effects, such as those used in cancer therapy.
- The high variability among nano-and micromotors allows the incorporation of different features provided by the drug delivery community, such as drug protection by encapsulation [40,41,42], selectivity by combination with selective biomarkers and, in case of nano-scaled motors, the ability to penetrate tissue.
- A remote trigger mechanism to release the drug at the desired location can be implemented using micromotors. This has been introduced into micromotion using piezo induced changes [43] but also in strategies like molecular valves are envisioned [44,45]. The full overview of demonstrated examples of controlled drug release for cancer therapy by nano-and micromotors is displayed in Table 1, Table 2 and Table 3.
- The first point to list here is often (also in many current publications) the use of highly reactive fuels, such as hydrogen peroxide or hydrazine or ultraviolet (UV) light to achieve propulsion. Even though alternative strategies are being developed, many propulsion mechanisms are not fully compatible with the use in body fluids, but this will be discussed in detail in Section 2.
- Particles require nanoscale dimensions for passage through tissues. For achieving an enhanced permeability and retention (EPR) effect usually dimensions up to 180 nm are assumed. According to a study of the Fischer group for penetrating mucus a particle size not larger than 100 nm seems to be beneficial for passing through hydrogel networks [46]. Additionally, the particle size plays an important role in biologically relevant processes such as the circulation and biodistribution of nanoparticles. Currently, most balistically moving nano-and micromotors are in the micrometer scale rather than lower nanometer scale.
- As an opposing fact to the previous point, Brownian rotation dominates for particle sizes below 800 nm. Here, the particle orientations are randomized and only Brownian motion or random walks are expected. Motion control is, however, crucial for targeted drug delivery, but fabrication, integration, modification and motor-cargo integration are still rather challenging below 100 nm.
- The materials employed for nano- and micromotor design are mostly chosen for functionality rather than biocompatibility, which can often lead to toxic components in motors. For clinical applications these need to be adapted and optimized concerning their non-toxicity, as well as their ability to be scaled up to allow high through-put fabrication.
- Most nano- and micromotors are still rather slow, so that large distances in the body would require long action times or, if applicable, high fuel concentrations. Further, careful navigation to avoid tissue damage might still be recommendable or injection to areas close to the tumor site is necessary.
2. Propulsion Strategies
2.1. Chemically Based Strategies
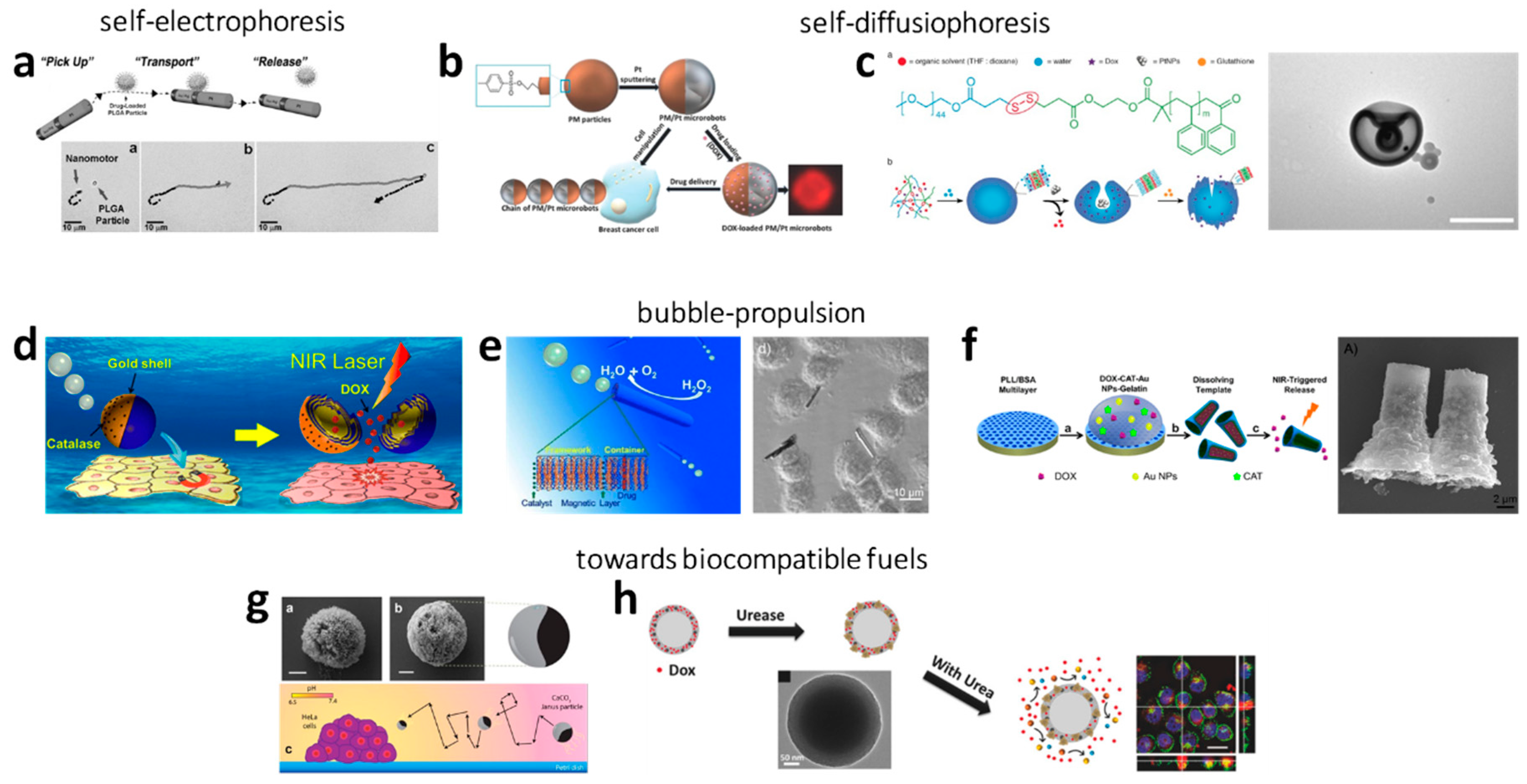
2.1.1. Electrophoresis
2.1.2. Self-Diffusiophoresis
H2O2 as Fuel
Developments towards More Biocompatible Fuels
2.1.3. Bubble Propulsion
H2O2 as Fuel
Developments towards More Biocompatible Fuels
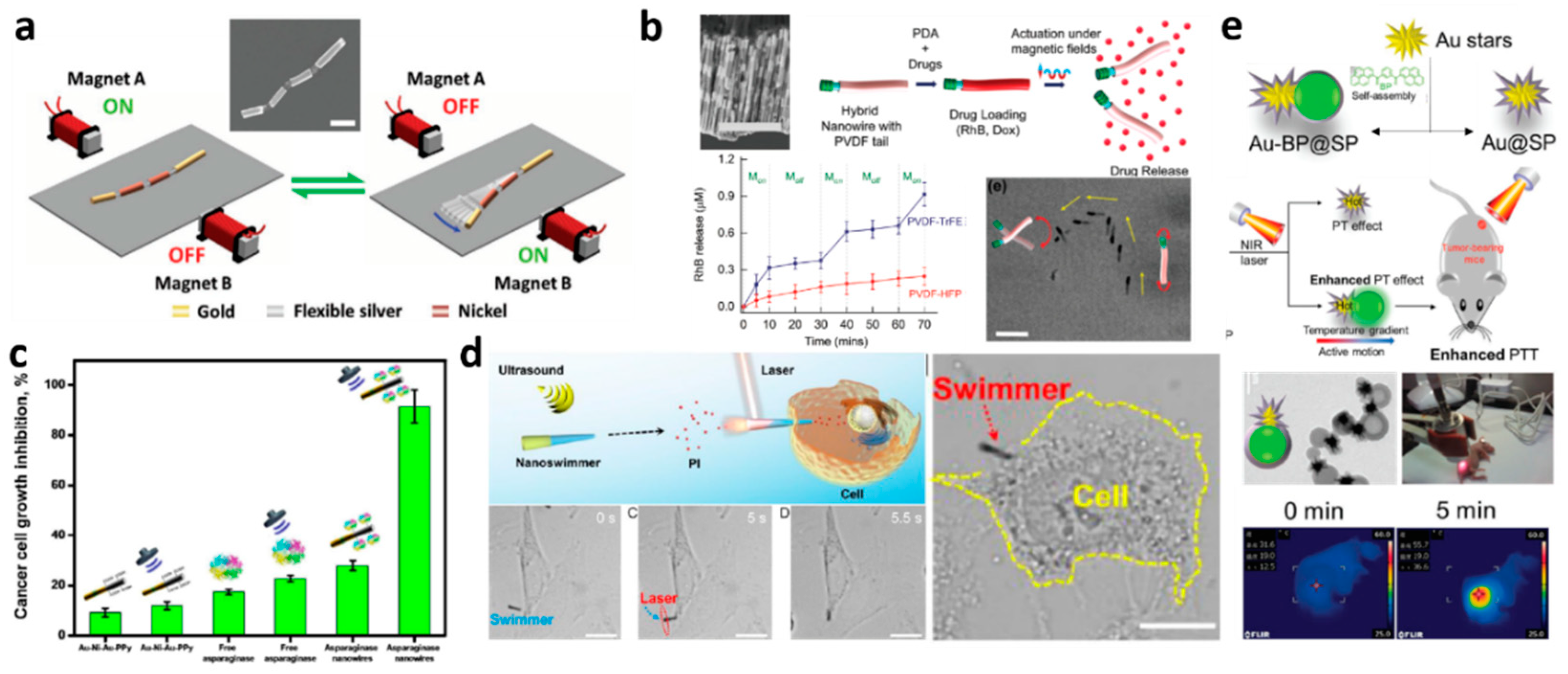
2.2. Energy Transferred by External Fields
2.2.1. Electric Fields
2.2.2. Magnetic Fields
2.2.3. Acoustically Driven Systems
2.2.4. Light
2.3. Biological Constituents for Nano- and Micromotors
2.3.1. Structural Units
2.3.2. Loading Units
Blood Cells
Viruses
Spores
2.3.3. Propulsion Units
Bacteria
Sperm Cells
Algae
2.4. Shape-Changing Nano-and Micromotors for Cancer Therapy
3. Performance Optimization
3.1. Power Transmission
3.2. Loading
3.3. Guidance
3.4. Release
3.5. In Vivo Imaging
3.6. Retrieval of Micromotors
4. Outlook
Funding
Acknowledgments
Conflicts of Interest
References
- Gao, W.; Wang, J. The environmental impact of Micro/Nanomachines: A Review. ACS Nano 2014, 8, 3170–3180. [Google Scholar] [CrossRef]
- Wang, H.; Pumera, M. Micro/Nanomachines and Living Biosystems: From Simple Interactions to Microcyborgs. Adv. Funct. Mater. 2018, 28, 1705421. [Google Scholar] [CrossRef]
- Bente, K.; Codutti, A.; Bachmann, F.; Faivre, D. Biohybrid and Bioinspired Magnetic Microswimmers. Small 2018, 14, 1704374. [Google Scholar] [CrossRef]
- Bastos-Arrieta, J.; Revilla-Guarinos, A.; Uspal, W.E.; Simmchen, J. Bacterial Biohybrid Microswimmers. Front. Robot. AI 2018, 5, 97. [Google Scholar] [CrossRef]
- Ricotti, L.; Trimmer, B.; Feinberg, A.W.; Raman, R.; Parker, K.K.; Bashir, R.; Sitti, M.; Martel, S.; Dario, P.; Menciassi, A. Biohybrid actuators for robotics: A review of devices actuated by living cells Biohybrid actuators for robotics: A review of devices actuated by living cells. Sci. Robot. 2017, 2, eaaq0495. [Google Scholar] [CrossRef]
- Magdanz, V.; Medina-Sánchez, M.; Schwarz, L.; Xu, H.; Elgeti, J.; Schmidt, O.G. Spermatozoa as Functional Components of Robotic Microswimmers. Adv. Mater. 2017, 29, 1606301. [Google Scholar] [CrossRef]
- Purcell, E.M. Life at low Reynolds number. Am. J. Phys. 1977, 45, 3–11. [Google Scholar] [CrossRef]
- Anderson, J. Colloid Transport By Interfacial Forces. Annu. Rev. Fluid Mech. 1989, 21, 61–99. [Google Scholar] [CrossRef]
- Moran, J.L.; Posner, J.D. Phoretic Self-Propulsion. Annu. Rev. Fluid Mech. 2017, 49, 511–540. [Google Scholar] [CrossRef]
- Erkoc, P.; Yasa, I.C.; Ceylan, H.; Yasa, O.; Alapan, Y.; Sitti, M. Mobile Microrobots for Active Therapeutic Delivery. Adv. Ther. 2019, 2, 1800064. [Google Scholar] [CrossRef]
- Gao, C.; Lin, Z.; Lin, X.; He, Q. Cell Membrane—Camouflaged Colloid Motors for Biomedical Applications. Adv. Ther. 2018, 1, 1800056. [Google Scholar] [CrossRef]
- Luo, M.; Feng, Y.; Wang, T.; Guan, J. Micro-/Nanorobots at Work in Active Drug Delivery. Adv. Funct. Mater. 2018, 28, 1706100. [Google Scholar] [CrossRef]
- Wang, S.; Liu, K.; Wang, F.; Peng, F.; Tu, Y. The Application of Micro- and Nanomotors in Classified Drug Delivery. Chem.–An Asian J. 2019, 14. [Google Scholar] [CrossRef] [PubMed]
- Medina-Sánchez, M.; Xu, H.; Schmidt, O.G. Micro-and nano-motors: The new generation of drug carriers. Ther. Deliv. 2018, 9, 303–316. [Google Scholar] [CrossRef] [PubMed]
- Reinišová, L.; Hermanová, S.; Pumera, M. Micro/nanomachines: What is needed for them to become a real force in cancer therapy? Nanoscale 2019, 11, 6519–6532. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Guo, J.; Liang, Z.; Fan, D. Artificial Micro/Nanomachines for Bioapplications: Biochemical Delivery and Diagnostic Sensing. Adv. Funct. Mater. 2018, 28, 1705867. [Google Scholar] [CrossRef]
- Gao, W.; de Ávila, B.E.F.; Zhang, L.; Wang, J. Targeting and isolation of cancer cells using micro/nanomotors. Adv. Drug Deliv. Rev. 2018, 125, 94–101. [Google Scholar] [CrossRef]
- Srivastava, S.K.; Clergeaud, G.; Andresen, T.L.; Boisen, A. Micromotors for drug delivery in vivo: The road ahead. Adv. Drug Deliv. Rev. 2019, 138, 41–55. [Google Scholar] [CrossRef]
- Gao, W.; Wang, J. Synthetic micro/nanomotors in drug delivery. Nanoscale 2014, 6, 10486–10494. [Google Scholar] [CrossRef]
- Khalil, I.S.M.; Tabak, A.F.; Sadek, K.; Member, S. Rubbing Against Blood Clots using Helical Robots: Modeling and In Vitro Experimental Validation. IEEE Robot. Automa. Lett. 2017, 2. [Google Scholar] [CrossRef]
- Xi, W.; Solovev, A.A.; Ananth, A.N.; Gracias, D.H.; Sanchez, S.; Schmidt, O.G. Rolled-up magnetic microdrillers: Towards remotely controlled minimally invasive surgery. Nanoscale 2013, 5, 1294–1297. [Google Scholar] [CrossRef] [PubMed]
- Ismagilov, R.F.; Schwartz, A.; Bowden, N.; Whitesides, G.M. Autonomous movement and self-assembly. Angew. Chem. Int. Ed. 2002, 41, 652–654. [Google Scholar] [CrossRef]
- Paxton, W.F.; Kistler, K.C.; Olmeda, C.C.; Sen, A.; St. Angelo, S.K.; Cao, Y.; Mallouk, T.E.; Lammert, P.E.; Crespi, V.H. Catalytic Nanomotors: Autonomous Movement of Striped Nanorods. J. Am. Chem. Soc. 2004, 126, 13424–13431. [Google Scholar] [CrossRef] [PubMed]
- Palagi, S.; Mark, A.G.; Reigh, S.Y.; Melde, K.; Qiu, T.; Zeng, H.; Parmeggiani, C.; Martella, D.; Sanchez-Castillo, A.; Kapernaum, N.; et al. Structured light enables biomimetic swimming and versatile locomotion of photoresponsive soft microrobots. Nat. Mater. 2016, 15, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Mourran, A.; Zhang, H.; Vinokur, R.; Möller, M.; Mourran, A.; Zhang, H.; Vinokur, R.; Möller, M. Soft Microrobots Employing Nonequilibrium Actuation via Plasmonic Heating. Adv. Mater. 2016, 29, 1604825. [Google Scholar] [CrossRef] [PubMed]
- Safdar, M.; Simmchen, J.; Jänis, J. Light-driven micro- and nanomotors for environmental remediation. Environ. Sci. Nano 2017, 4, 1602–1616. [Google Scholar] [CrossRef]
- Jurado-Sánchez, B.; Wang, J. Micromotors for environmental applications: A review. Environ. Sci. Nano 2018, 5, 1530–1544. [Google Scholar] [CrossRef]
- Parmar, J.; Vilela, D.; Villa, K.; Wang, J.; Sánchez, S. Micro- and Nanomotors as Active Environmental Microcleaners and Sensors. J. Am. Chem. Soc. 2018, 140, 9317–9331. [Google Scholar] [CrossRef]
- Guix, M.; Mayorga-Martinez, C.C.; Merkoçi, A. Nano/Micromotors in (Bio)chemical Science Applications. Chem. Rev. 2014, 114, 6285–6322. [Google Scholar] [CrossRef]
- Zarei, M.; Zarei, M. Self-Propelled Micro/Nanomotors for Sensing and Environmental Remediation. Small 2018, 14, 1800912. [Google Scholar] [CrossRef]
- Li, J.; Angsantikul, P.; Liu, W.; Esteban-Fernández de Ávila, B.; Thamphiwatana, S.; Xu, M.; Sandraz, E.; Wang, X.; Delezuk, J.; Gao, W.; et al. Micromotors Spontaneously Neutralize Gastric Acid for pH-Responsive Payload Release. Angew. Chem. Int. Ed. 2017, 56, 2156–2161. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Dong, R.; Thamphiwatana, S.; Li, J.; Gao, W.; Zhang, L.; Wang, J. Artificial Micromotors in the Mouse’s Stomach: A Step toward in Vivo Use of Synthetic Motors. ACS Nano 2015, 9, 117–123. [Google Scholar] [CrossRef] [PubMed]
- De Ávila, B.E.F.; Angsantikul, P.; Li, J.; Angel Lopez-Ramirez, M.; Ramírez-Herrera, D.E.; Thamphiwatana, S.; Chen, C.; Delezuk, J.; Samakapiruk, R.; Ramez, V.; et al. Micromotor-enabled active drug delivery for in vivo treatment of stomach infection. Nat. Commun. 2017, 8, 272. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Hou, T.; Li, J.; Yu, S.; Xu, Z.; Yin, M.; Wang, J.; Wang, X. Self-Propelled and Targeted Drug Delivery of Poly(aspartic acid)/Iron–Zinc Microrocket in the Stomach. ACS Nano 2019, 13, 1324–1332. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Mou, F.; Gong, H.; Luo, M.; Guan, J. Light-driven micro/nanomotors: From fundamentals to applications. Chem. Soc. Rev. 2017, 46, 6905–6926. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Rozen, I.; Wang, J. Rocket Science at the Nanoscale. ACS Nano 2016, 10, 5619–5634. [Google Scholar] [CrossRef]
- Karshalev, E.; Esteban-Fernández De Ávila, B.; Beltrán-Gastélum, M.; Angsantikul, P.; Tang, S.; Mundaca-Uribe, R.; Zhang, F.; Zhao, J.; Zhang, L.; Wang, J. Micromotor pills as a dynamic oral delivery platform. ACS Nano 2018, 12, 8397–8405. [Google Scholar] [CrossRef]
- Chen, H.; Zhao, Q.; Du, X. Light-powered micro/nanomotors. Micromachines 2018, 9, 41. [Google Scholar] [CrossRef]
- Ferrando-Climent, L.; Rodriguez-Mozaz, S.; Barceló, D. Incidence of anticancer drugs in an aquatic urban system: From hospital effluents through urban wastewater to natural environment. Environ. Pollut. 2014, 193, 216–223. [Google Scholar] [CrossRef]
- Felfoul, O.; Mohammadi, M.; Taherkhani, S.; de Lanauze, D.; Zhong Xu, Y.; Loghin, D.; Essa, S.; Jancik, S.; Houle, D.; Lafleur, M.; et al. Magneto-aerotactic bacteria deliver drug-containing nanoliposomes to tumour hypoxic regions. Nat. Nanotechnol. 2016, 11, 941–947. [Google Scholar] [CrossRef]
- Champion, J.A.; Katare, Y.K.; Mitragotri, S. Particle Shape: A New Design Parameter for Micro- and Nanoscale Drug Delivery Carriers. J. Control Release 2014, 121, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Cunha, D.; Ben Yahia, M.; Hall, S.; Miller, S.R.; Chevreau, H.; Elkaïm, E.; Maurin, G.; Horcajada, P.; Serre, C. Rationale of Drug Encapsulation and Release from Biocompatible Porous Metal–Organic Frameworks. Chem. Mater. 2013, 25, 2767–2776. [Google Scholar] [CrossRef]
- Mushtaq, F.; Torlakcik, H.; Hoop, M.; Jang, B.; Carlson, F.; Grunow, T.; Läubli, N.; Ferreira, A.; Chen, X.; Nelson, B.J.; et al. Motile Piezoelectric Nanoeels for Targeted Drug Delivery. Adv. Funct. Mater. 2019, 29, 1808135. [Google Scholar] [CrossRef]
- Ruiz-Hernández, E.; Baeza, A.; Vallet-Regí, M. Smart Drug Delivery through DNA/Magnetic Nanoparticle Gates. ACS Nano 2011, 5, 1259–1266. [Google Scholar] [CrossRef] [PubMed]
- Schlossbauer, A.; Warncke, S.; Gramlich, P.M.E.; Kecht, J.; Manetto, A.; Carell, T.; Bein, T. A Programmable DNA-Based Molecular Valve for Colloidal Mesoporous Silica. Angew. Chem. Int. Ed. 2010, 49, 4734–4737. [Google Scholar] [CrossRef]
- Walker, D.; Käsdorf, B.T.; Jeong, H.-H.; Lieleg, O.; Fischer, P. Enzymatically active biomimetic micropropellers for the penetration of mucin gels. Sci. Adv. 2015, 1, e1500501. [Google Scholar] [CrossRef] [PubMed]
- Golestanian, R.; Liverpool, T.B.; Ajdari, A. Propulsion of a molecular machine by asymmetric distribution of reaction products. Phys. Rev. Lett. 2005, 94, 220801. [Google Scholar] [CrossRef]
- Hong, Y.; Diaz, M.; Córdova-Fteueroa, U.M.; Sen, A. Light-driven titanium-dioxide-based reversible microfireworks and micromotor/micropump systems. Adv. Funct. Mater. 2010, 20, 1568–1576. [Google Scholar] [CrossRef]
- Simmchen, J.; Magdanz, V.; Sanchez, S.; Chokmaviroj, S.; Ruiz-Molina, D.; Baeza, A.; Schmidt, O.G. Effect of surfactants on the performance of tubular and spherical micromotors-a comparative study. RSC Adv. 2014, 4, 20334–20340. [Google Scholar] [CrossRef]
- Paxton, W.F.; Baker, P.T.; Kline, T.R.; Wang, Y.; Mallouk, T.E.; Sen, A. Catalytically induced electrokinetics for motors and micropumps. J. Am. Chem. Soc. 2006, 128, 14881–14888. [Google Scholar] [CrossRef]
- Würger, A. Thermophoresis in colloidal suspensions driven by Marangoni forces. Phys. Rev. Lett. 2007, 98, 138301. [Google Scholar] [CrossRef] [PubMed]
- Braun, M.; Cichos, F. Optically controlled thermophoretic trapping of single nano-objects. ACS Nano 2013, 7, 11200–11208. [Google Scholar] [CrossRef] [PubMed]
- Behkam, B.; Sitti, M. Effect of quantity and configuration of attached bacteria on bacterial propulsion of microbeads. Appl. Phys. Lett. 2008, 93, 223901. [Google Scholar] [CrossRef]
- Magdanz, V.; Sanchez, S.; Schmidt, O.G. Development of a sperm-flagella driven micro-bio-robot. Adv. Mater. 2013, 25, 6581–6588. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Martel, S. Preliminary investigation of bio-carriers using magnetotactic bacteria. In Proceedings of the 2006 International Conference of the IEEE Engineering in Medicine and Biology Society, New York, NY, USA, 30 August–3 September 2006; pp. 3415–3418. [Google Scholar]
- Stanton, M.M.; Simmchen, J.; Ma, X.; Miguel-López, A.; Sánchez, S. Biohybrid Janus Motors Driven by Escherichia coli. Adv. Mater. Interfaces 2016, 3, 1500505. [Google Scholar] [CrossRef]
- Kagan, D.; Laocharoensuk, R.; Zimmerman, M.; Clawson, C.; Balasubramanian, S.; Kang, D.; Bishop, D.; Sattayasamitsathit, S.; Zhang, L.; Wang, J. Rapid delivery of drug carriers propelled and navigated by catalytic nanoshuttles. Small 2010, 6, 2741–2747. [Google Scholar] [CrossRef] [PubMed]
- Howse, J.R.; Jones, R.A.L.; Ryan, A.J.; Gough, T.; Vafabakhsh, R.; Golestanian, R. Self-Motile Colloidal Particles: From Directed Propulsion to Random Walk. Phys. Rev. Lett. 2007, 99, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.A.; Nolte, R.J.M.; van Hest, J.C.M. Autonomous movement of platinum-loaded stomatocytes. Nat. Chem. 2012, 4, 268–274. [Google Scholar] [CrossRef]
- Tu, Y.; Peng, F.; Sui, X.; Men, Y.; White, P.B.; van Hest, J.C.M.; Wilson, D.A. Self-propelled supramolecular nanomotors with temperature-responsive speed regulation. Nat. Chem. 2017, 9, 480–486. [Google Scholar] [CrossRef]
- Tu, Y.; Peng, F.; White, P.B.; Wilson, D.A. Redox-Sensitive Stomatocyte Nanomotors: Destruction and Drug Release in the Presence of Glutathione. Angew. Chem. Int. Ed. 2017, 56, 7620–7624. [Google Scholar] [CrossRef]
- Villa, K.; Krejčová, L.; Novotný, F.; Heger, Z.; Sofer, Z.; Pumera, M. Cooperative Multifunctional Self-Propelled Paramagnetic Microrobots with Chemical Handles for Cell Manipulation and Drug Delivery. Adv. Funct. Mater. 2018, 28, 1804343. [Google Scholar] [CrossRef]
- Imlay, J.; Chin, S.; Linn, S. Toxic DNA damage by hydrogen peroxide through the Fenton reaction in vivo and in vitro. Science 1988, 240, 640–642. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, B.C.; Chang, C.J. Chemistry and biology of reactive oxygen species in signaling or stress responses. Nat. Chem. Biol. 2011, 7, 504–511. [Google Scholar] [CrossRef] [PubMed]
- Szatrowski, T.P.; Nathan, C.F. Production of Large Amounts of Hydrogen Peroxide by Human Tumor Cells. Cancer Res. 1991, 51, 794–798. [Google Scholar] [PubMed]
- Peng, F.; Tu, Y.; van Hest, J.C.M.; Wilson, D.A. Self-Guided Supramolecular Cargo-Loaded Nanomotors with Chemotactic Behavior towards Cells. Angew. Chem. Int. Ed. 2015, 54, 11662–11665. [Google Scholar] [CrossRef]
- López-Lázaro, M. Dual role of hydrogen peroxide in cancer: Possible relevance to cancer chemoprevention and therapy. Cancer Lett. 2007, 252, 1–8. [Google Scholar] [CrossRef] [PubMed]
- MacKay, E.M.; MacKay, L.L. The concentration of urea in the blood of normal individuals 1. J. Clin. Invest. 1927, 4, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Engelgau, M.M.; Narayan, K.M.; Herman, W.H. Screening for type 2 diabetes. Diabetes Care 2000, 23, 1563–1580. [Google Scholar] [CrossRef] [PubMed]
- Hortelão, A.C.; Patiño, T.; Perez-Jiménez, A.; Blanco, À.; Sánchez, S. Enzyme-Powered Nanobots Enhance Anticancer Drug Delivery. Adv. Funct. Mater. 2018, 28, 1705086. [Google Scholar] [CrossRef]
- Ma, X.; Jannasch, A.; Albrecht, U.-R.; Hahn, K.; Miguel-López, A.; Schäffer, E.; Sánchez, S. Enzyme-Powered Hollow Mesoporous Janus Nanomotors. Nano Lett. 2015, 15, 7043–7050. [Google Scholar] [CrossRef]
- Ma, X.; Wang, X.; Hahn, K.; Sánchez, S. Motion Control of Urea-Powered Biocompatible Hollow Microcapsules. ACS Nano 2016, 10, 3597–3605. [Google Scholar] [CrossRef] [PubMed]
- Muddana, H.S.; Sengupta, S.; Mallouk, T.E.; Sen, A.; Butler, P.J. Substrate Catalysis Enhances Single-Enzyme Diffusion. J. Am. Chem. Soc. 2010, 132, 2110–2111. [Google Scholar] [CrossRef] [PubMed]
- Hortelão, A.C.; Carrascosa, R.; Murillo-Cremaes, N.; Patiño, T.; Sánchez, S. Targeting 3D Bladder Cancer Spheroids with Urease-Powered Nanomotors. ACS Nano 2019, 13, 429–439. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Xia, T.; Zhang, Z.; Xie, S.; Wang, T.; Li, X. Enzyme-powered Janus nanomotors launched from intratumoral depots to address drug delivery barriers. Chem. Eng. J. 2019, 375, 122109. [Google Scholar] [CrossRef]
- Schattling, P.; Thingholm, B.; Städler, B. Enhanced Diffusion of Glucose-Fueled Janus Particles. Chem. Mater. 2015, 27, 7412–7418. [Google Scholar] [CrossRef]
- Nijemeisland, M.; Abdelmohsen, L.K.E.A.; Huck, W.T.S.; Wilson, D.A.; van Hest, J.C.M. A Compartmentalized Out-of-Equilibrium Enzymatic Reaction Network for Sustained Autonomous Movement. ACS Cent. Sci. 2016, 2, 843–849. [Google Scholar] [CrossRef] [PubMed]
- Schattling, P.S.; Ramos-Docampo, M.A.; Salgueiriño, V.; Städler, B. Double-Fueled Janus Swimmers with Magnetotactic Behavior. ACS Nano 2017, 11, 3973–3983. [Google Scholar] [CrossRef]
- Wu, Y.; Lin, X.; Wu, Z.; Möhwald, H.; He, Q. Self-Propelled Polymer Multilayer Janus Capsules for Effective Drug Delivery and Light-Triggered Release. ACS Appl. Mater. Interfaces 2014, 6, 10476–10481. [Google Scholar] [CrossRef]
- Xuan, M.; Shao, J.; Lin, X.; Dai, L.; He, Q. Self-Propelled Janus Mesoporous Silica Nanomotors with Sub-100 nm Diameters for Drug Encapsulation and Delivery. ChemPhysChem 2014, 15, 2255–2260. [Google Scholar] [CrossRef]
- Tejeda-Rodríguez, J.A.; Núñez, A.; Soto, F.; García-Gradilla, V.; Cadena-Nava, R.; Wang, J.; Vazquez-Duhalt, R. Virus-Based Nanomotors for Cargo Delivery. ChemNanoMat 2019, 5, 194–200. [Google Scholar] [CrossRef]
- Solovev, A.A.; Mei, Y.; Bermúdez Ureña, E.; Huang, G.; Schmidt, O.G. Catalytic Microtubular Jet Engines Self-Propelled by Accumulated Gas Bubbles. Small 2009, 5, 1688–1692. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Huang, G.; Ye, M.; Li, M.; Liu, R.; Mei, Y. Dynamics of catalytic tubular microjet engines: Dependence on geometry and chemical environment. Nanoscale 2011, 3, 5083. [Google Scholar] [CrossRef] [PubMed]
- Manjare, M.; Yang, B.; Zhao, Y.-P. Bubble-Propelled Microjets: Model and Experiment. J. Phys. Chem. C 2013, 117, 4657–4665. [Google Scholar] [CrossRef]
- Li, L.; Wang, J.; Li, T.; Song, W.; Zhang, G. A unified model of drag force for bubble-propelled catalytic micro/nano-motors with different geometries in low Reynolds number flows. J. Appl. Phys. 2015, 117, 104308. [Google Scholar] [CrossRef]
- Fomin, V.M.; Hippler, M.; Magdanz, V.; Soler, L.; Sanchez, S.; Schmidt, O.G. Propulsion Mechanism of Catalytic Microjet Engines. IEEE Trans. Robot. 2014, 30, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Moo, J.G.S.; Pumera, M. From Nanomotors to Micromotors: The Influence of the Size of an Autonomous Bubble-Propelled Device upon Its Motion. ACS Nano 2016, 10, 5041–5050. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wang, J.; Li, T.; Song, W.; Zhang, G. Hydrodynamics and propulsion mechanism of self-propelled catalytic micromotors: Model and experiment. Soft Matter 2014, 10, 7511–7518. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Wu, Y.; He, W.; Lin, X.; Sun, J.; He, Q. Self-Propelled Polymer-Based Multilayer Nanorockets for Transportation and Drug Release. Angew. Chem. Int. Ed. 2013, 52, 7000–7003. [Google Scholar] [CrossRef]
- Wu, Z.; Lin, X.; Zou, X.; Sun, J.; He, Q. Biodegradable Protein-Based Rockets for Drug Transportation and Light-Triggered Release. ACS Appl. Mater. Interfaces 2015, 7, 250–255. [Google Scholar] [CrossRef]
- Beladi-Mousavi, S.M.; Khezri, B.; Krejčová, L.; Heger, Z.; Sofer, Z.; Fisher, A.C.; Pumera, M. Recoverable Bismuth-Based Microrobots: Capture, Transport, and On-Demand Release of Heavy Metals and an Anticancer Drug in Confined Spaces. ACS Appl. Mater. Interfaces 2019, 11, 13359–13369. [Google Scholar] [CrossRef]
- Peng, F.; Tu, Y.; Men, Y.; van Hest, J.C.M.; Wilson, D.A. Supramolecular Adaptive Nanomotors with Magnetotaxis Behavior. Adv. Mater. 2017, 29, 1604996. [Google Scholar] [CrossRef] [PubMed]
- Tu, Y.; Peng, F.; André, A.A.M.; Men, Y.; Srinivas, M.; Wilson, D.A. Biodegradable Hybrid Stomatocyte Nanomotors for Drug Delivery. ACS Nano 2017, 11, 1957–1963. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhu, H.; Shi, Y.; Ge, Y.; Feng, X.; Liu, R.; Li, Y.; Ma, Y.; Wang, L. Novel catalytic micromotor of porous zeolitic imidazolate framework-67 for precise drug delivery. Nanoscale 2018, 10, 11384–11391. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Sattayasamitsathit, S.; Orozco, J.; Wang, J. Highly Efficient Catalytic Microengines: Template Electrosynthesis of Polyaniline/Platinum Microtubes. J. Am. Chem. Soc. 2011, 133, 11862–11864. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Karshalev, E.; Guan, J.; Wang, J. Magnesium-Based Micromotors: Water-Powered Propulsion, Multifunctionality, and Biomedical and Environmental Applications. Small 2018, 14, 1704252. [Google Scholar] [CrossRef] [PubMed]
- Mou, F.; Chen, C.; Ma, H.; Yin, Y.; Wu, Q.; Guan, J. Self-Propelled Micromotors Driven by the Magnesium-Water Reaction and Their Hemolytic Properties. Angew. Chem. Int. Ed. 2013, 52, 7208–7212. [Google Scholar] [CrossRef] [PubMed]
- Mou, F.; Chen, C.; Zhong, Q.; Yin, Y.; Ma, H.; Guan, J. Autonomous Motion and Temperature-Controlled Drug Delivery of Mg/Pt-Poly(N-isopropylacrylamide) Janus Micromotors Driven by Simulated Body Fluid and Blood Plasma. ACS Appl. Mater. Interfaces 2014, 6, 9897–9903. [Google Scholar] [CrossRef]
- Gerweck, L.E.; Seetharaman, K. Cellular pH Gradient in Tumor versus Normal Tissue: Potential Exploitation for the Treatment of Cancer. Cancer Res. 1996, 56, 1194. [Google Scholar]
- Guix, M.; Meyer, A.K.; Koch, B.; Schmidt, O.G. Carbonate-based Janus micromotors moving in ultra-light acidic environment generated by HeLa cells in situ. Sci. Rep. 2016, 6, 21701. [Google Scholar] [CrossRef]
- Wan, M.; Chen, H.; Wang, Q.; Niu, Q.; Xu, P.; Yu, Y.; Zhu, T.; Mao, C.; Shen, J. Bio-inspired nitric-oxide-driven nanomotor. Nat. Commun. 2019, 10, 966. [Google Scholar] [CrossRef]
- Kong, L.; Chen, C.; Mou, F.; Feng, Y.; You, M.; Yin, Y.; Guan, J. Magnesium Particles Coated with Mesoporous Nanoshells as Sustainable Therapeutic-Hydrogen Suppliers to Scavenge Continuously Generated Hydroxyl Radicals in Long Term. Part. Part. Syst. Charact. 2019, 36, 1800424. [Google Scholar] [CrossRef]
- Walker, V. Ammonia toxicity and its prevention in inherited defects of the urea cycle. Diabetes, Obes. Metab. 2009, 11, 823–835. [Google Scholar] [CrossRef] [PubMed]
- Pittman, R. Regulation of Tissue Oxygenation. In Colloquium Series on Integrated Systems Physiology: From Molecule to Function; Morgan & Claypool Life Sciences: San Rafael, CA, USA, 2011. [Google Scholar]
- Levett, D.Z.H.; Millar, I.L. Bubble trouble: A review of diving physiology and disease. Postgrad. Med. J. 2008, 84, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Gangwal, S.; Cayre, O.J.; Bazant, M.Z.; Velev, O.D. Induced-charge electrophoresis of metallodielectric particles. Phys. Rev. Lett. 2008, 100, 58302. [Google Scholar] [CrossRef] [PubMed]
- Boymelgreen, A.M.; Miloh, T. Induced-charge electrophoresis of uncharged dielectric spherical Janus particles. Electrophoresis 2012, 33, 870–879. [Google Scholar] [CrossRef] [PubMed]
- Boymelgreen, A.M.; Balli, T.; Miloh, T.; Yossifon, G. Active colloids as mobile microelectrodes for unified label-free selective cargo transport. Nat. Commun. 2018, 9, 760. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Go, G.; Ko, S.Y.; Park, J.; Park, S. Magnetic actuated pH-responsive hydrogel- based soft micro-robot for targeted drug delivery. Smart Mater. Struct. 2016, 25, 27001. [Google Scholar] [CrossRef]
- Ren, Z.; Hu, W.; Dong, X.; Sitti, M. Multi-functional soft-bodied jellyfish-like swimming. Nat. Commun. 2019, 10, 2703. [Google Scholar] [CrossRef]
- Xin, C.; Yang, L.; Li, J.; Hu, Y.; Qian, D.; Fan, S.; Hu, K.; Cai, Z.; Wu, H.; Wang, D.; et al. Conical Hollow Microhelices with Superior Swimming Capabilities for Targeted Cargo Delivery. Adv. Mater. 2019, 31, 1808226. [Google Scholar] [CrossRef]
- Sheng, L.; He, Z.; Yao, Y.; Liu, J. Transient State Machine Enabled from the Colliding and Coalescence of a Swarm of Autonomously Running Liquid Metal Motors. Small 2015, 5253–5261. [Google Scholar] [CrossRef]
- Tan, S.; Gui, H.; Yuan, B.; Liu, J.; Tan, S.; Gui, H.; Yuan, B.; Liu, J. Magnetic trap effect to restrict motion of self-powered tiny liquid metal motors Magnetic trap effect to restrict motion of self-powered tiny liquid metal motors. Appl. Phys. Lett. 2015, 107, 071904. [Google Scholar] [CrossRef]
- Qiu, T.; Lee, T.C.; Mark, A.G.; Morozov, K.I.; Münster, R.; Mierka, O.; Turek, S.; Leshansky, A.M.; Fischer, P. Swimming by reciprocal motion at low Reynolds number. Nat. Commun. 2014, 5, 5119. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Li, J.; Zhang, H.; Chang, X.; Song, W.; Hu, Y. Magnetically Propelled Fish-Like Nanoswimmers. Small 2016, 12, 6098–6105. [Google Scholar] [CrossRef] [PubMed]
- Ishiyama, K.; Sendoh, M.; Yamazaki, A.; Arai, K.I. Swimming micro-machine driven by magnetic torque. Sens. Actuators A Phys. 2001, 91, 141–144. [Google Scholar] [CrossRef]
- Bell, D.J.; Leutenegger, S.; Hammar, K.M.; Dong, L.X.; Nelson, B.J. Flagella-like propulsion for microrobots using a nanocoil and a rotating electromagnetic field. In Proceedings of the 2007 IEEE International Conference on Robotics and Automation, Roma, Italy, 10–14 April 2007; pp. 1128–1133. [Google Scholar]
- Zhang, L.; Abbott, J.J.; Dong, L.; Kratochvil, B.E.; Bell, D.; Nelson, B.J.; Zhang, L.; Abbott, J.J.; Dong, L.; Kratochvil, B.E.; et al. Artificial bacterial flagella: Fabrication and magnetic control Artificial bacterial flagella: Fabrication and magnetic control. Appl. Phys. Lett. 2009, 94, 064107. [Google Scholar] [CrossRef]
- Ghosh, A.; Fischer, P. Controlled Propulsion of Artificial Magnetic Nanostructured Propellers. Nano Lett. 2009, 9, 2243–2245. [Google Scholar] [CrossRef] [PubMed]
- Servant, A.; Qiu, F.; Mazza, M.; Kostarelos, K.; Nelson, B.J. Controlled In Vivo Swimming of a Swarm of Bacteria-Like Microrobotic Flagella. Adv. Mater. 2015, 27, 2981–2988. [Google Scholar] [CrossRef] [PubMed]
- Qiu, F.; Mhanna, R.; Zhang, L.; Ding, Y.; Fujita, S.; Nelson, B.J. Sensors and Actuators B: Chemical Artificial bacterial flagella functionalized with temperature-sensitive liposomes for controlled release. Sens. Actuators, B. Chem. 2014, 196, 676–681. [Google Scholar] [CrossRef]
- Chen, X.-Z.; Hoop, M.; Shamsudhin, N.; Huang, T.; Özkale, B.; Li, Q.; Siringil, E.; Mushtaq, F.; Di Tizio, L.; Nelson, B.J.; et al. Hybrid Magnetoelectric Nanowires for Nanorobotic Applications: Fabrication, Magnetoelectric Coupling, and Magnetically Assisted In Vitro Targeted Drug Delivery. Adv. Mater. 2017, 29, 1605458. [Google Scholar] [CrossRef]
- Jeon, S.; Kim, S.; Ha, S.; Lee, S.; Kim, E.; Kim, S.Y.; Park, S.H.; Jeon, J.H.; Kim, S.W.; Moon, C.; et al. Magnetically actuated microrobots as a platform for stem cell transplantation. Sci. Robot. 2019, 4, eaav4317. [Google Scholar] [CrossRef]
- Srivastava, S.K.; Ajalloueian, F.; Boisen, A. TRAP Technology: Thread-Like Radical-Polymerization via Autonomously Propelled (TRAP) Bots (Adv. Mater. 30/2019). Adv. Mater. 2019, 31, 1970213. [Google Scholar] [CrossRef]
- Chatzipirpiridis, G.; Ergeneman, O.; Pokki, J.; Ullrich, F.; Fusco, S.; Ortega, J.A.; Sivaraman, K.M.; Nelson, B.J.; Pané, S. Electroforming of Implantable Tubular Magnetic Microrobots for Wireless Ophthalmologic Applications. Adv. Healthc. Mater. 2014, 4, 209–214. [Google Scholar] [CrossRef]
- Ceylan, H.; Yasa, I.C.; Yasa, O.; Tabak, A.F.; Giltinan, J.; Sitti, M. 3D-Printed Biodegradable Microswimmer for Theranostic Cargo Delivery and Release. ACS Nano 2019, 13, 3353–3362. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Zhou, Q.; Vincent, M.; Deng, Y.; Yu, J.; Xu, J.; Xu, T.; Tang, T.; Bian, L.; Wang, Y.-X.J.; et al. Multifunctional biohybrid magnetite microrobots for imaging-guided therapy. Sci. Robot. 2017, 2, eaaq1155. [Google Scholar] [CrossRef]
- Srivastava, S.K.; Medina-Sánchez, M.; Koch, B.; Schmidt, O.G. Medibots: Dual-Action Biogenic Microdaggers for Single-Cell Surgery and Drug Release. Adv. Mater. 2015, 28, 832–837. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, L.; Yang, L.; Vong, C.I.; Chan, K.F.; Wu, W.K.K.; Kwong, T.N.Y.; Lo, N.W.S.; Ip, M.; Wong, S.H.; et al. Real-time tracking of fluorescent magnetic spore–based microrobots for remote detection of C. diff toxins. Sci. Adv. 2019, 5, eaau9650. [Google Scholar] [CrossRef] [PubMed]
- Magdanz, V.; Gebauer, J.; Mahdi, D.; Simmchen, J.; Khalil, I.K.M. Sperm-templated magnetic microrobots. In Proceedings of the International Conference on Manipulation, Automation and Robotics at Small Scales (MARSS), Helsinki, Finland, 1–5 July 2019. [Google Scholar]
- Nadal, F.; Lauga, E. Asymmetric steady streaming as a mechanism for acoustic propulsion of rigid bodies. Phys. Fluids 2014, 26, 82001. [Google Scholar] [CrossRef]
- Soto, F.; Wagner, G.L.; Garcia-Gradilla, V.; Gillespie, K.T.; Lakshmipathy, D.R.; Karshalev, E.; Angell, C.; Chen, Y.; Wang, J. Acoustically propelled nanoshells. Nanoscale 2016, 8, 17788–17793. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Gradilla, V.; Sattayasamitsathit, S.; Soto, F.; Kuralay, F.; Yardımcı, C.; Wiitala, D.; Galarnyk, M.; Wang, J. Ultrasound-Propelled Nanoporous Gold Wire for Efficient Drug Loading and Release. Small 2014, 10, 4154–4159. [Google Scholar] [CrossRef]
- Wang, W.; Wu, Z.; Lin, X.; Si, T.; He, Q.; Wang, W.; Wu, Z.; Lin, X.; Si, T.; He, Q. Article Gold Nanoshell-Functionalized Polymer Nanoswimmer for Photomechanical Poration of Single Cell Membrane Gold Nanoshell-Functionalized Polymer Nanoswimmer for Photomechanical Poration of Single Cell Membrane. J. Am. Chem. Soc. 2019, 141, 6601–6608. [Google Scholar] [CrossRef]
- Wang, W.; Duan, W.; Ahmed, S.; Mallouk, T.E.; Sen, A. Small power: Autonomous nano- and micromotors propelled by self-generated gradients. Nano Today 2013, 8, 531–554. [Google Scholar] [CrossRef]
- Wang, W.; Li, S.; Mair, L.; Ahmed, S.; Huang, T.J.; Mallouk, T.E. Acoustic Propulsion of Nanorod Motors Inside Living Cells. Angew. Chem. Int. Ed. 2014, 53, 3201–3204. [Google Scholar] [CrossRef] [PubMed]
- Uygun, M.; Jurado-Sánchez, B.; Uygun, D.A.; Singh, V.V.; Zhang, L.; Wang, J. Ultrasound-propelled nanowire motors enhance asparaginase enzymatic activity against cancer cells. Nanoscale 2017, 9, 18423–18429. [Google Scholar] [CrossRef]
- Jiang, H.R.; Yoshinaga, N.; Sano, M. Active motion of a Janus particle by self-thermophoresis in a defocused laser beam. Phys. Rev. Lett. 2010, 105, 268302. [Google Scholar] [CrossRef] [PubMed]
- Bregulla, A.P.; Cichos, F. Size dependent efficiency of photophoretic swimmers. Faraday Discuss. 2015, 184, 381–391. [Google Scholar] [CrossRef]
- Xuan, M.; Wu, Z.; Shao, J.; Dai, L.; Si, T.; He, Q. Near Infrared Light-Powered Janus Mesoporous Silica Nanoparticle Motors. J. Am. Chem. Soc. 2016, 138, 6492–6497. [Google Scholar] [CrossRef]
- Wu, Z.; de Ávila, B.; Martín, A.; Christianson, C.; Gao, W.; Thamphiwatana, S.K.; Escarpa, A.; He, Q.; Zhang, L.; Wang, J. RBC micromotors carrying multiple cargos towards potential theranostic applications. Nanoscale 2015, 7, 13680–13686. [Google Scholar] [CrossRef]
- Yang, P.P.; Zhai, Y.G.; Qi, G.B.; Lin, Y.X.; Luo, Q.; Yang, Y.; Xu, A.P.; Yang, C.; Li, Y.S.; Wang, L.; et al. NIR Light Propulsive Janus-like Nanohybrids for Enhanced Photothermal Tumor Therapy. Small 2016, 12, 5423–5430. [Google Scholar] [CrossRef]
- Xuan, M.; Shao, J.; Gao, C.; Wang, W.; Dai, L.; He, Q. Self-Propelled Nanomotors for Thermomechanically Percolating Cell Membranes. Angew. Chem. Int. Ed. 2018, 57, 12463–12467. [Google Scholar] [CrossRef]
- Chen, C.; Chang, X.; Angsantikul, P.; Li, J.; de Ávila, B.; Karshalev, E.; Liu, W.; Mou, F.; He, S.; Castillo, R.; et al. Chemotactic Guidance of Synthetic Organic/Inorganic Payloads Functionalized Sperm Micromotors. Adv. Biosyst. 2018, 2, 1700160. [Google Scholar] [CrossRef]
- Xu, H.; Medina-Sánchez, M.; Magdanz, V.; Schwarz, L.; Hebenstreit, F.; Schmidt, O.G. Sperm-Hybrid Micromotor for Targeted Drug Delivery. ACS Nano 2018, 12, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Yasa, O.; Erkoc, P.; Alapan, Y.; Sitti, M. Microalga-Powered Microswimmers toward Active Cargo Delivery. Adv. Mater. 2018, 30, 1804130. [Google Scholar] [CrossRef]
- Alapan, Y.; Yasa, O.; Schauer, O.; Giltinan, J.; Tabak, A.F.; Sourjik, V.; Sitti, M. Soft erythrocyte-based bacterial microswimmers for cargo delivery. Sci. Robot. 2018, 3, eaar4423. [Google Scholar] [CrossRef]
- Park, B.-W.; Zhuang, J.; Yasa, O.; Sitti, M. Multifunctional Bacteria-Driven Microswimmers for Targeted Active Drug Delivery. ACS Nano 2017, 11, 8910–8923. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.D.; Han, J.-W.; Choi, Y.J.; Cho, S.; Zheng, S.; Ko, S.Y.; Park, J.-O.; Park, S. Active tumor-therapeutic liposomal bacteriobot combining a drug (paclitaxel)-encapsulated liposome with targeting bacteria (Salmonella Typhimurium). Sens. Actuators B Chem. 2016, 224, 217–224. [Google Scholar] [CrossRef]
- Suh, S.B.; Jo, A.; Traore, M.A.; Zhan, Y.; Coutermarsh-Ott, S.L.; Ringel-Scaia, V.M.; Allen, I.C.; Davis, R.M.; Behkam, B. Nanoscale Bacteria-Enabled Autonomous Drug Delivery System (NanoBEADS) Enhances Intratumoral Transport of Nanomedicine. Adv. Sci. 2019, 6, 1801309. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Feng, X.; Pei, A.; Kane, C.R.; Tam, R.; Hennessy, C.; Wang, J. Bioinspired helical microswimmers based on vascular plants. Nano Lett. 2013, 14, 305–310. [Google Scholar] [CrossRef]
- Santomauro, G.; Singh, A.V.; Park, B.-W.; Mohammadrahimi, M.; Erkoc, P.; Goering, E.; Schütz, G.; Sitti, M.; Bill, J. Incorporation of Terbium into a Microalga Leads to Magnetotactic Swimmers. Adv. Biosyst. 2018, 2, 1800039. [Google Scholar] [CrossRef]
- Guillerey, C.; Huntington, N.D.; Smyth, M.J. Targeting natural killer cells in cancer immunotherapy. Nat. Immunol. 2016, 17, 1025. [Google Scholar] [CrossRef]
- Villa, C.H.; Anselmo, A.C.; Mitragotri, S.; Muzykantov, V. Red blood cells: Supercarriers for drugs, biologicals, and nanoparticles and inspiration for advanced delivery systems. Adv. Drug Deliv. Rev. 2016, 106, 88–103. [Google Scholar] [CrossRef]
- Zhang, H. Erythrocytes in nanomedicine: An optimal blend of natural and synthetic materials. Biomater. Sci. 2016, 4, 1024–1031. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.-M.J.; Fang, R.H.; Zhang, L. Erythrocyte-Inspired Delivery Systems. Adv. Healthc. Mater. 2012, 1, 537–547. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Wang, C.; Liu, Z. Red Blood Cells as Smart Delivery Systems. Bioconjug. Chem. 2018, 29, 852–860. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Li, T.; Li, J.; Gao, W.; Xu, T.; Christianson, C.; Gao, W.; Galarnyk, M.; He, Q.; Zhang, L.; et al. Turning Erythrocytes into Functional Micromotors. ACS Nano 2014, 8, 12041–12048. [Google Scholar] [CrossRef] [PubMed]
- Gao, C.; Lin, Z.; Wang, D.; Wu, Z.; Xie, H.; He, Q. Red Blood Cell-Mimicking Micromotor for Active Photodynamic Cancer Therapy. ACS Appl. Mater. Interfaces 2019, 11, 23392–23400. [Google Scholar] [CrossRef]
- Sun, X.; Wang, C.; Gao, M.; Hu, A.; Liu, Z. Remotely Controlled Red Blood Cell Carriers for Cancer Targeting and Near-Infrared Light-Triggered Drug Release in Combined Photothermal–Chemotherapy. Adv. Funct. Mater. 2015, 25, 2386–2394. [Google Scholar] [CrossRef]
- Mu, Q.; Wang, H.; Gu, X.; Stephen, Z.R.; Yen, C.; Chang, F.-C.; Dayringer, C.J.; Zhang, M. Biconcave Carbon Nanodisks for Enhanced Drug Accumulation and Chemo-Photothermal Tumor Therapy. Adv. Healthc. Mater. 2019, 8, 1801505. [Google Scholar] [CrossRef]
- Wan, G.; Chen, B.; Li, L.; Wang, D.; Shi, S.; Zhang, T.; Wang, Y.; Zhang, L.; Wang, Y. Nanoscaled red blood cells facilitate breast cancer treatment by combining photothermal/photodynamic therapy and chemotherapy. Biomaterials 2018, 155, 25–40. [Google Scholar] [CrossRef]
- Osuka, S.; Van Meir, E.G. Neutrophils traffic in cancer nanodrugs. Nat. Nanotechnol. 2017, 12, 616. [Google Scholar] [CrossRef]
- Kennedy, L.C.; Bear, A.S.; Young, J.K.; Lewinski, N.A.; Kim, J.; Foster, A.E.; Drezek, R.A. T cells enhance gold nanoparticle delivery to tumors in vivo. Nanoscale Res. Lett. 2011, 6, 283. [Google Scholar] [CrossRef]
- Zhang, F.; Mundaca-Uribe, R.; Gong, H.; Esteban-Fernández de Ávila, B.; Beltrán-Gastélum, M.; Karshalev, E.; Nourhani, A.; Tong, Y.; Nguyen, B.; Gallot, M.; et al. A Macrophage–Magnesium Hybrid Biomotor: Fabrication and Characterization. Adv. Mater. 2019, 31, 1901828. [Google Scholar] [CrossRef] [PubMed]
- Basel, M.T.; Balivada, S.; Wang, H.; Shrestha, T.B.; Seo, G.M.; Pyle, M.; Abayaweera, G.; Dani, R.; Koper, O.B.; Tamura, M.; et al. Cell-delivered magnetic nanoparticles caused hyperthermia-mediated increased survival in a murine pancreatic cancer model. Int. J. Nanomed. 2012, 7, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Marín-Caba, L.; Chariou, P.L.; Pesquera, C.; Correa-Duarte, M.A.; Steinmetz, N.F. Tobacco Mosaic Virus-Functionalized Mesoporous Silica Nanoparticles, a Wool-Ball-like Nanostructure for Drug Delivery. Langmuir 2019, 35, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Steele, J.F.C.; Peyret, H.; Saunders, K.; Castells-Graells, R.; Marsian, J.; Meshcheriakova, Y.; Lomonossoff, G.P. Synthetic plant virology for nanobiotechnology and nanomedicine. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2017, 9, e1447. [Google Scholar] [CrossRef] [PubMed]
- Hovlid, M.L.; Steinmetz, N.F.; Laufer, B.; Lau, J.L.; Kuzelka, J.; Wang, Q.; Hyypiä, T.; Nemerow, G.R.; Kessler, H.; Manchester, M.; et al. Guiding plant virus particles to integrin-displaying cells. Nanoscale 2012, 4, 3698–3705. [Google Scholar] [CrossRef] [PubMed]
- Bareford, L.M.; Swaan, P.W. Endocytic mechanisms for targeted drug delivery. Adv. Drug Deliv. Rev. 2007, 59, 748–758. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Wong, S.M.; Lim, L.-Y. Folic acid-conjugated protein cages of a plant virus: A novel delivery platform for doxorubicin. Bioconjug. Chem. 2007, 18, 836–843. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Potroz, M.G.; Jackman, J.A.; Khezri, B.; Marić, T.; Cho, N.J.; Pumera, M. Bioinspired Spiky Micromotors Based on Sporopollenin Exine Capsules. Adv. Funct. Mater. 2017, 27, 1–9. [Google Scholar] [CrossRef]
- Li, D.; Choi, H.; Cho, S.; Jeong, S.; Jin, Z.; Lee, C.; Ko, S.Y.; Park, J.O.; Park, S. A hybrid actuated microrobot using an electromagnetic field and flagellated bacteria for tumor-targeting therapy. Biotechnol. Bioeng. 2015, 112, 1623–1631. [Google Scholar] [CrossRef]
- Schauer, O.; Mostaghaci, B.; Colin, R.; Hürtgen, D.; Kraus, D.; Sitti, M.; Sourjik, V. Motility and chemotaxis of bacteria-driven microswimmers fabricated using antigen 43-mediated biotin display. Sci. Rep. 2018, 8, 9801. [Google Scholar] [CrossRef]
- Kim, D.; Liu, A.; Diller, E.; Sitti, M. Chemotactic steering of bacteria propelled microbeads. Biomed. Microdevices 2012, 14, 1009–1017. [Google Scholar] [CrossRef] [PubMed]
- Martel, S.; Mohammadi, M.; Felfoul, O.; Lu, Z.; Pouponneau, P. Flagellated Magnetotactic Bacteria as Controlled MRI-trackable Propulsion and Steering Systems for Medical Nanorobots Operating in the Human Microvasculature. Int. J. Rob. Res. 2009, 28, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, J.; Carlsen, R.W.; Sitti, M. pH-taxis of biohybrid microsystems. Sci. Rep. 2015, 5, 11403. [Google Scholar] [CrossRef] [PubMed]
- Behkam, B.; Sitti, M. Bacterial flagella-based propulsion and on/off motion control of microscale objects. Appl. Phys. Lett. 2007, 90, 23902. [Google Scholar] [CrossRef]
- Park, S.J.; Park, S.-H.; Cho, S.; Kim, D.-M.; Lee, Y.; Ko, S.Y.; Hong, Y.; Choy, H.E.; Min, J.-J.; Park, J.-O.; et al. New paradigm for tumor theranostic methodology using bacteria-based microrobot. Sci. Rep. 2013, 3, 3394. [Google Scholar] [CrossRef] [PubMed]
- Akin, D.; Sturgis, J.; Ragheb, K.; Sherman, D.; Burkholder, K.; Robinson, J.P.; Bhunia, A.K.; Mohammed, S.; Bashir, R. Bacteria-mediated delivery of nanoparticles and cargo into cells. Nat. Nanotechnol. 2007, 2, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.V.; Hosseinidoust, Z.; Park, B.-W.; Yasa, O.; Sitti, M. Microemulsion-Based Soft Bacteria-Driven Microswimmers for Active Cargo Delivery. ACS Nano 2017, 11, 9759–9769. [Google Scholar] [CrossRef] [PubMed]
- Mostaghaci, B.; Yasa, O.; Zhuang, J.; Sitti, M. Bioadhesive Bacterial Microswimmers for Targeted Drug Delivery in the Urinary and Gastrointestinal Tracts. Adv. Sci. 2017, 4, 1700058. [Google Scholar] [CrossRef] [PubMed]
- Stanton, M.M.; Park, B.-W.; Miguel-López, A.; Ma, X.; Sitti, M.; Sánchez, S. Biohybrid Microtube Swimmers Driven by Single Captured Bacteria. Small 2017, 13, 1603679. [Google Scholar] [CrossRef] [PubMed]
- Forbes, N.S. Engineering the perfect (bacterial) cancer therapy. Nat. Rev. Cancer 2010, 10, 785. [Google Scholar] [CrossRef]
- Zhou, S.; Gravekamp, C.; Bermudes, D.; Liu, K. Tumour-targeting bacteria engineered to fight cancer. Nat. Rev. Cancer 2018, 18, 727–743. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.; Zhao, L.; Song, X.; Tang, M.; Mo, C.; Li, X. Doxorubicin-conjugated Escherichia coli Nissle 1917 swimmers to achieve tumor targeting and responsive drug release. J. Control. Release 2017, 268, 390–399. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Medina-Sánchez, M.; Brison, D.R.; Edmondson, R.J.; Taylor, S.S.; Nelson, L.; Zeng, K.; Bagley, S.; Ribeiro, C.; Restrepo, L.P.; et al. Human Spermbots for Cancer-Relevant Drug Delivery. arXiv 2019, arXiv:1904.12684. [Google Scholar]
- Machines, P.; Mould, S.; Scientific, W.; Co, P. From reaction—diffusion to Physarum computing. In Physarum Machines; World Scientific Publishing Co. Pte. Ltd.: Singapore, 2010; pp. 1–13. [Google Scholar]
- Nawroth, J.C.; Lee, H.; Feinberg, A.W.; Ripplinger, C.M.; McCain, M.L.; Grosberg, A.; Dabiri, J.O.; Parker, K.K. A tissue-engineered jellyfish with biomimetic propulsion. Nat. Biotechnol. 2012, 30, 792–797. [Google Scholar] [CrossRef] [PubMed]
- Medina-Sánchez, M.; Magdanz, V.; Guix, M.; Fomin, V.M.; Schmidt, O.G. Swimming Microrobots: Soft, Reconfigurable, and Smart. Adv. Funct. Mater. 2018, 28, 1707228. [Google Scholar] [CrossRef]
- Ghosh, A.; Yoon, C.; Ongaro, F.; Scheggi, S.; Selaru, F.M.; Misra, S.; Gracias, D.H. Stimuli-Responsive Soft Untethered Grippers for Drug Delivery and Robotic Surgery. Front. Mech. Eng. 2017, 3, 7. [Google Scholar] [CrossRef] [PubMed]
- Erol, O.; Pantula, A.; Liu, W.; Gracias, D.H. Transformer Hydrogels: A Review. Adv. Mater. Technol. 2019, 4, 1–27. [Google Scholar] [CrossRef]
- Malachowski, K.; Breger, J.; Kwag, H.R.; Wang, M.O.; Fisher, J.P.; Selaru, F.M.; Gracias, D.H. Stimuli-responsive theragrippers for chemomechanical controlled release. Angew. Chem. Int. Ed. Engl. 2014, 53, 8045–8049. [Google Scholar] [CrossRef]
- Fusco, S.; Sakar, M.S.; Kennedy, S.; Peters, C.; Bottani, R.; Starsich, F.; Mao, A.; Sotiriou, G.A.; Pané, S.; Pratsinis, S.E.; et al. An Integrated Microrobotic Platform for On-Demand, Targeted Therapeutic Interventions. Adv. Mater. 2014, 26, 952–957. [Google Scholar] [CrossRef]
- Kobayashi, K.; Yoon, C.; Oh, S.H.; Pagaduan, J.V.; Gracias, D.H. Biodegradable Thermomagnetically Responsive Soft Untethered Grippers. ACS Appl. Mater. Interfaces 2019, 11, 151–159. [Google Scholar] [CrossRef]
- Sitti, M.; Ceylan, H.; Hu, W.; Giltinan, J.; Turan, M.; Yim, S.; Diller, E. Biomedical Applications of Untethered Mobile Milli/Microrobots. Proc. IEEE Inst. Electr. Eletron. Eng. 2015, 103, 205–224. [Google Scholar] [CrossRef] [PubMed]
- Jayson, M.; Darwin, Z.; Gl, J. Light-actuated microrobots for biomedical science. Spie Newsroom 2017. [Google Scholar] [CrossRef]
- Imaz, I.; Hernando, J.; Ruiz-Molina, D.; Maspoch, D. Metal–Organic Spheres as Functional Systems for Guest Encapsulation. Angew. Chem. Int. Ed. 2009, 48, 2325–2329. [Google Scholar] [CrossRef] [PubMed]
- Simmchen, J.; Katuri, J.; Uspal, W.E.; Popescu, M.N.; Tasinkevych, M.; Sánchez, S. Topographical pathways guide chemical microswimmers. Nat. Commun. 2016, 7, 10598. [Google Scholar] [CrossRef] [PubMed]
- You, M.; Chen, C.; Xu, L.; Mou, F.; Guan, J. Intelligent Micro/nanomotors with Taxis. Acc. Chem. Res. 2018, 51, 3006–3014. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Blackman, N.M.K.; Kopp, N.D.; Sen, A.; Velegol, D. Chemotaxis of nonbiological colloidal rods. Phys. Rev. Lett. 2007, 99, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.H.; Huang, C.T.; Su, C.H.; Yeh, C.S. Bacteria-Mediated Hypoxia-Specific Delivery of Nanoparticles for Tumors Imaging and Therapy. Nano Lett. 2016, 16, 3493–3499. [Google Scholar] [CrossRef]
- Bozuyuk, U.; Yasa, O.; Yasa, I.C.; Ceylan, H.; Kizilel, S.; Sitti, M. Light-Triggered Drug Release from 3D-Printed Magnetic Chitosan Microswimmers. ACS Nano 2018, 12, 9617–9625. [Google Scholar] [CrossRef]
- Xu, X.; Kim, K.; Fan, D. Tunable Release of Multiplex Biochemicals by Plasmonically Active Rotary Nanomotors. Angew. Chem. Int. Ed. 2015, 54, 2525–2529. [Google Scholar] [CrossRef] [PubMed]
- Garnacho, C. Intracellular Drug Delivery: Mechanisms for Cell Entry. Curr. Pharm. Des. 2016, 22, 1210–1226. [Google Scholar] [CrossRef] [PubMed]
- Medina-Sanchez, M.; Schmidt, O.G. Medical microbots need better imaging and control. Nat. News 2017, 545, 406–408. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Zhang, Y.; Zhang, L. Recent progress on micro- and nano-robots: Towards in vivo tracking and localization. Quant. Imaging Med. Surg. 2018, 8, 461–479. [Google Scholar] [CrossRef] [PubMed]
- Pané, S.; Puigmartí-Luis, J.; Bergeles, C.; Chen, X.; Pellicer, E.; Sort, J.; Počepcová, V.; Ferreira, A.; Nelson, B.J. Imaging Technologies for Biomedical Micro-and Nanoswimmers. Adv. Mater. Technol. 2019, 4, 1800575. [Google Scholar] [CrossRef]
- Bruns, O.T.; Bischof, T.S.; Harris, D.K.; Franke, D.; Shi, Y.; Riedemann, L.; Bartelt, A.; Jaworski, F.B.; Carr, J.A.; Rowlands, C.J.; et al. Next-generation in vivo optical imaging with short-wave infrared quantum dots. Nat. Biomed. Eng. 2017, 1, 56. [Google Scholar] [CrossRef] [PubMed]
- Carr, J.A.; Franke, D.; Caram, J.R.; Perkinson, C.F.; Saif, M.; Askoxylakis, V.; Datta, M.; Fukumura, D.; Jain, R.K.; Bawendi, M.G.; et al. Shortwave infrared fluorescence imaging with the clinically approved near-infrared dye indocyanine green. 2018, 115, 4465–4470. [Google Scholar] [CrossRef]
- Iacovacci, V.; Blanc, A.; Huang, H.; Ricotti, L.; Schibli, R.; Menciassi, A.; Behe, M.; Pané, S.; Nelson, B.J. High-Resolution SPECT Imaging of Stimuli-Responsive Soft Microrobots. Small 2019, 15, 1900709. [Google Scholar] [CrossRef] [PubMed]
- Aziz, A.; Medina-Sánchez, M.; Claussen, J.; Schmidt, O.G. Real-time optoacoustic tracking of single moving micro-objects in deep tissue-mimicking phantoms. BioRxiv 2019, 640045. [Google Scholar] [CrossRef]
- Sanchez, A.; Magdanz, V.; Schmidt, O.G.; Misra, S. Magnetic control of self-propelled microjets under ultrasound image guidance. In Proceedings of the 5th IEEE RAS/EMBS International Conference on Biomedical Robotics and Biomechatronics, Sao Paulo, Brazil, 12–15 August 2014; pp. 169–174. [Google Scholar]
- Khalil, I.S.M.; Ferreira, P.; Eleuterio, R.; de Korte, C.L.; Misra, S. Magnetic-based closed-loop control of paramagnetic microparticles using ultrasound feedback. In Proceedings of the 2014 IEEE International Conference on Robotics and Automation (ICRA), Hong Kong, China, 31 May–7 June 2014; pp. 3807–3812. [Google Scholar]
- Wu, Z.; Li, L.; Yang, Y.; Hu, P.; Li, Y.; Yang, S.-Y.; Wang, L.V.; Gao, W. A microrobotic system guided by photoacoustic computed tomography for targeted navigation in intestines in vivo. Sci. Robot. 2019, 4, eaax0613. [Google Scholar] [CrossRef]
- Iacovacci, V.; Ricotti, L.; Sinibaldi, E.; Signore, G.; Vistoli, F.; Menciassi, A. An Intravascular Magnetic Catheter Enables the Retrieval of Nanoagents from the Bloodstream. Adv. Sci. 2018, 5, 1800807. [Google Scholar] [CrossRef] [PubMed]
- Yim, S.; Gultepe, E.; Gracias, D.H.; Sitti, M. Biopsy using a magnetic capsule endoscope carrying, releasing, and retrieving untethered microgrippers. IEEE Trans. Biomed. Eng. 2014, 61, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Peters, C.; Costanza, V.; Pané, S.; Nelson, B.J.; Hierold, C. Superparamagnetic hydrogels for Two-Photon Polymerization and their application for the fabrication of swimming microrobots. In Proceedings of the 2015 Transducers—2015 18th International Conference on Solid-State Sensors, Actuators and Microsystems (TRANSDUCERS), Anchorage, AK, USA, 21–25 June 2015; pp. 764–767. [Google Scholar]
- Wang, X.; Yu, G.; Han, X.; Zhang, H.; Ren, J.; Wu, X.; Qu, Y. Biodegradable and Multifunctional Polymer Micro-Tubes for Targeting Photothermal Therapy. Int. J. Mol. Sci. 2014, 15, 11730–11741. [Google Scholar] [CrossRef] [PubMed]
- Pena-Francesch, A.; Giltinan, J.; Sitti, M. Multifunctional and biodegradable self-propelled protein motors. Nat. Commun. 2019, 10, 3188. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, Y.; Maeda, H. A New Concept for Macromolecular Therapeutics in Cancer Chemotherapy: Mechanism of Tumoritropic Accumulation of Proteins and the Antitumor Agent Smancs. Cancer Res. 1986, 46, 6387–6392. [Google Scholar] [PubMed]
- Robertson, B.; Huang, M.J.; Chen, J.X.; Kapral, R. Synthetic Nanomotors: Working Together through Chemistry. Acc. Chem. Res. 2018, 51, 2355–2364. [Google Scholar] [CrossRef] [PubMed]
- Elgeti, J.; Winkler, R.G.; Gompper, G. Physics of microswimmers—single particle motion and collective behavior: A review. Rep. Prog. Phys. 2015, 78, 056601. [Google Scholar] [CrossRef] [PubMed]
- Martel, S.; Mohammadi, M. Using a swarm of self-propelled natural microrobots in the form of flagellated bacteria to perform complex micro-assembly tasks. In Proceedings of the 2010 IEEE International Conference on Robotics and Automation, Anchorage, AK, USA, 3–7 May 2010; pp. 500–505. [Google Scholar] [CrossRef]
- Singh, D.P.; Choudhury, U.; Fischer, P.; Mark, A.G. Non-Equilibrium Assembly of Light-Activated Colloidal Mixtures. Adv. Mater. 2017, 29, 1701328. [Google Scholar] [CrossRef]
- Zhang, J.; Salehizadeh, M.; Diller, E. Parallel Pick and Place Using Two Independent Untethered Mobile Magnetic Microgrippers. In Proceedings of the 2018 IEEE International Conference on Robotics and Automation (ICRA), Brisbane, QLD, Australia, 21–25 May 2018. [Google Scholar]
- Jurado-Sánchez, B.; Escarpa, A. Janus Micromotors for Electrochemical Sensing and Biosensing Applications: A Review. Electroanalysis 2017, 29, 14–23. [Google Scholar] [CrossRef]
- Kong, L.; Guan, J.; Pumera, M. Micro- and nanorobots based sensing and biosensing. Curr. Opin. Electrochem. 2018, 10, 174–182. [Google Scholar] [CrossRef]

| Carrier Material | Drug | Target | Propulsion | Guidance | Release | Speed [µm/s] | Ref. |
|---|---|---|---|---|---|---|---|
| phoretic propulsion | |||||||
| Ni/(Au50/Ag50)/Ni/Pt nanowire | DOX on PLGA particles and liposomes | - | self-electrophoresis (5% H2O2) | magnetic | drag force | 9 | [57] |
| polymersome stomatocyte/Pt | DOX | HeLa cells | self-electrophoresis (4.98 mM H2O2) | chemotactic, H2O2 gradient | glutathione assisted | 35 | [61] |
| PM/Pt microparticles | DOX | T47D cells | diffusiophoresis (2.5wt% H2O2) | magnetic | diffusion | 1.25 | [62] |
| mesoporous SiO2/urease | DOX | HeLa cells | diffusiophoresis (urea) | - | urea supported | - | [70] |
| SiO2/PEG/urease | anti-FGFR3 | bladder cancer cells | diffusiophoresis (urea) | via anti-FGFR3 | [74] | ||
| bubble propulsion | |||||||
| (PSS-PAH)5/Ni/Au/CAT | DOX | - | O2 (0.5% H2O2) | magnetic | heat (NIR light) | 25 | [79] |
| mesoporous SiO2/Cr/Pt | DOX | HeLa cells | O2 (0.2% H2O2) | - | catalytic hydrolysis of lipid bilayers | 3.6 | [80] |
| Virus/Pt | tamoxifen | MDA-231 | O2 (1.5% H2O2) | acid-supported | - | 4.15 | [81] |
| Pt NP/(CHI/ALG)18 nanotube | DOX | HeLa cells | O2 (H2O2) | magnetic | ultrasound | 22 | [89] |
| (PLL/BSA)10 -CAT-AuNPs | DOX | HeLa cells | O2 (0.5% H2O2) | magnetic | NIR light, gelatin melting | 4 | [90] |
| polymersome stomatocyte/Pt/Ni | DOX | HeLa cells | O2 (1.5% v/v H2O2) | magnetic | - | 12.4 | [92] |
| polymersome stomatocyte/Pt | DOX | HeLa cells | O2 (4.98 mM H2O2) | acid buffer | 39 | [93] | |
| ZIF-67 | DOX | - | O2 (1% H2O2) | magnetic | through H2O2 | 15.32 | [94] |
| CaCO3/Co | - | HeLa cells | CO2 (acid) | - | - | 0.544 | [100] |
| Polyamide/L-arginine (HLA) | particle itself | MCF-7 cells, HUVECs | NO (L-arginine and 20% H2O2) | 3 | [101] | ||
| Carrier Material | Drug | Loading Mechanism | Target | Propulsion | Guidance | Release | Speed [µm/s] | Ref. |
|---|---|---|---|---|---|---|---|---|
| FeGa@P(VDF-TrFE) nanowires | Paclitaxel | polydopamine functionalization | MDA MB 231 | magnetically | piezoelectric | alternating magnetic field | - | [122] |
| Hydrogel grippers and polycaprolactone particles | docetaxel (DTX) | Closing the gripper | / | magnetically | magnetic | Opening the gripper | - | [109] |
| Au-Ni-Au nanorods with nanoporous segments | DOX | Electrostatic interaction with the drug | HeLa cells | ultrasound | magnetic | Photothermal (NIR) | 60 | [133] |
| Carrier material | Drug | Target | Propulsion | Guidance | Release | Speed[µm/s] | Ref |
|---|---|---|---|---|---|---|---|
| Structural units | |||||||
| plant-based microtubes | camptothecin | HeLa cells | magnetic | magnetic | drilling into cells, diffusion | [128] | |
| Loading units | |||||||
| red blood cells | DOX | - | ultrasound | magnetic | photothermal | 50 | |
| Propulsion units | |||||||
| MC-1 magnetotactic bacteria | SN-38 | HCT116 colorectal xenografts | bacterial flagella | magnetic aerotaxis | endocytosis of liposomes | [40] | |
| S. typhimurium bacteria | paclitaxel | breast cancer cells | bacterial flagella | - | endocytosis of liposomes | 3 | [149] |
| E. coli bacteria | DOX | 4Z T1 breast cancer cells | bacterial flagella | magnetic, chemotaxis | pH-dependent diffusion | 10 | [148] |
| E. coli bioadhesive bacteriabot | PMMA | mannose expressing cell line (HTB-9 cells) | bacterial flagella | - | adhesion to mannose-expressing cell surfaces | [182] | |
| E. coli with red blood cells | DOX | - | bacterial flagella | magnetic | pH-dependent drug release from blood cells, NIR-triggered termination of bacteria | 10 | [147] |
| S. typhimurium NanoBEADS | - | tumor spheroids | bacterial flagella | bacteria translocation and proliferation | - | - | [150] |
| bovine sperm | DOX | tumor spheroids | sperm flagellum | magnetic | cell-cell-fusion | 41 | [145] |
| sea squirt sperm | DOX | ovarian cancer cells | sperm flagellum | chemotaxis | pH-induced death of sperm cells | 200 | [144] |
| human sperm | DOX | HeLa cells & human ovarian cancer tissue | sperm flagella | magnetic | cell-cell fusion | - | [187] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sonntag, L.; Simmchen, J.; Magdanz, V. Nano-and Micromotors Designed for Cancer Therapy. Molecules 2019, 24, 3410. https://doi.org/10.3390/molecules24183410
Sonntag L, Simmchen J, Magdanz V. Nano-and Micromotors Designed for Cancer Therapy. Molecules. 2019; 24(18):3410. https://doi.org/10.3390/molecules24183410
Chicago/Turabian StyleSonntag, Luisa, Juliane Simmchen, and Veronika Magdanz. 2019. "Nano-and Micromotors Designed for Cancer Therapy" Molecules 24, no. 18: 3410. https://doi.org/10.3390/molecules24183410
APA StyleSonntag, L., Simmchen, J., & Magdanz, V. (2019). Nano-and Micromotors Designed for Cancer Therapy. Molecules, 24(18), 3410. https://doi.org/10.3390/molecules24183410