Characterization of Volatile Compounds in Four Different Rhododendron Flowers by GC×GC-QTOFMS
Abstract
:1. Introduction
2. Results and Discussion
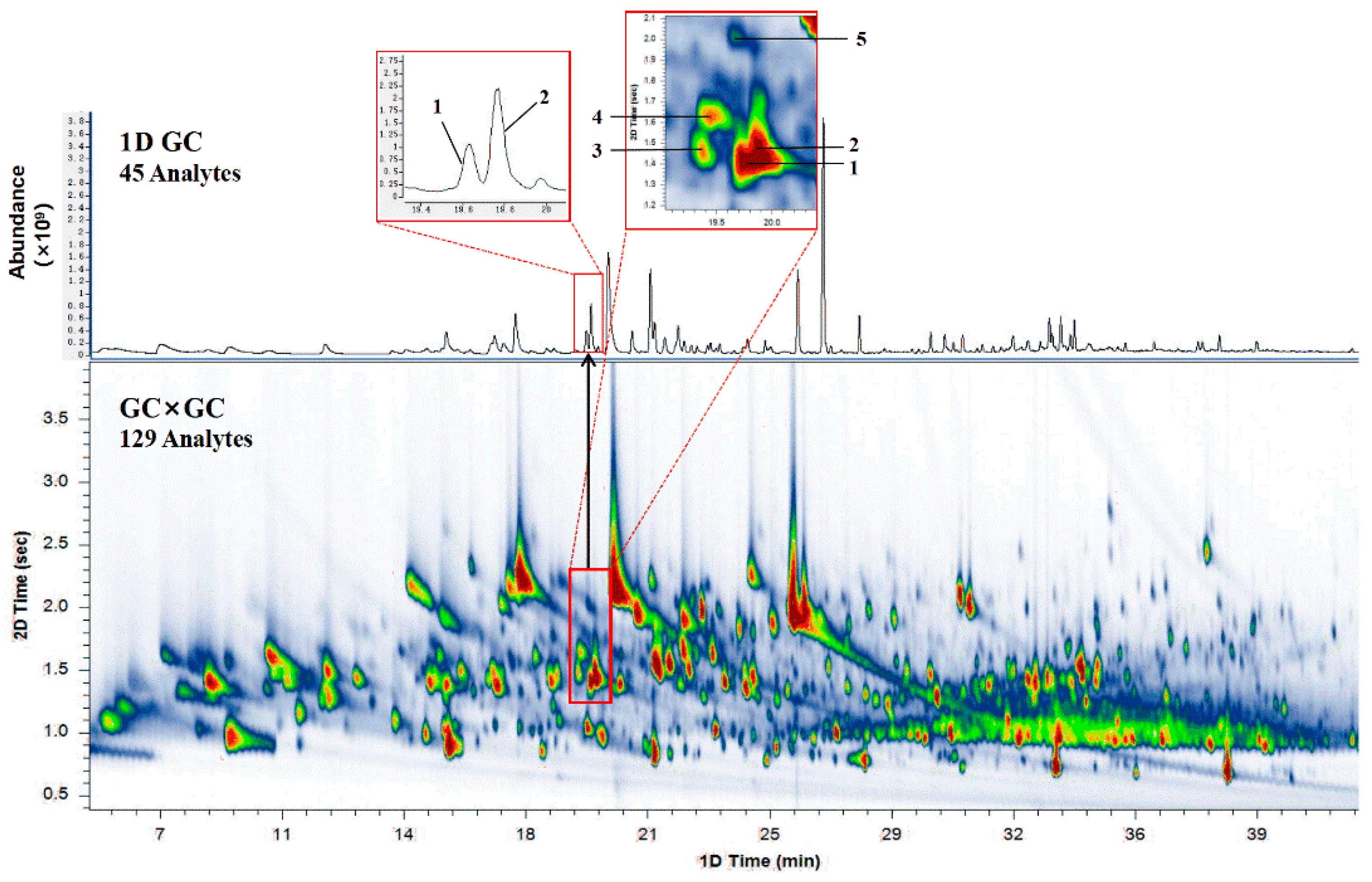
2.1. Comparison of 1DGC and GC×GC
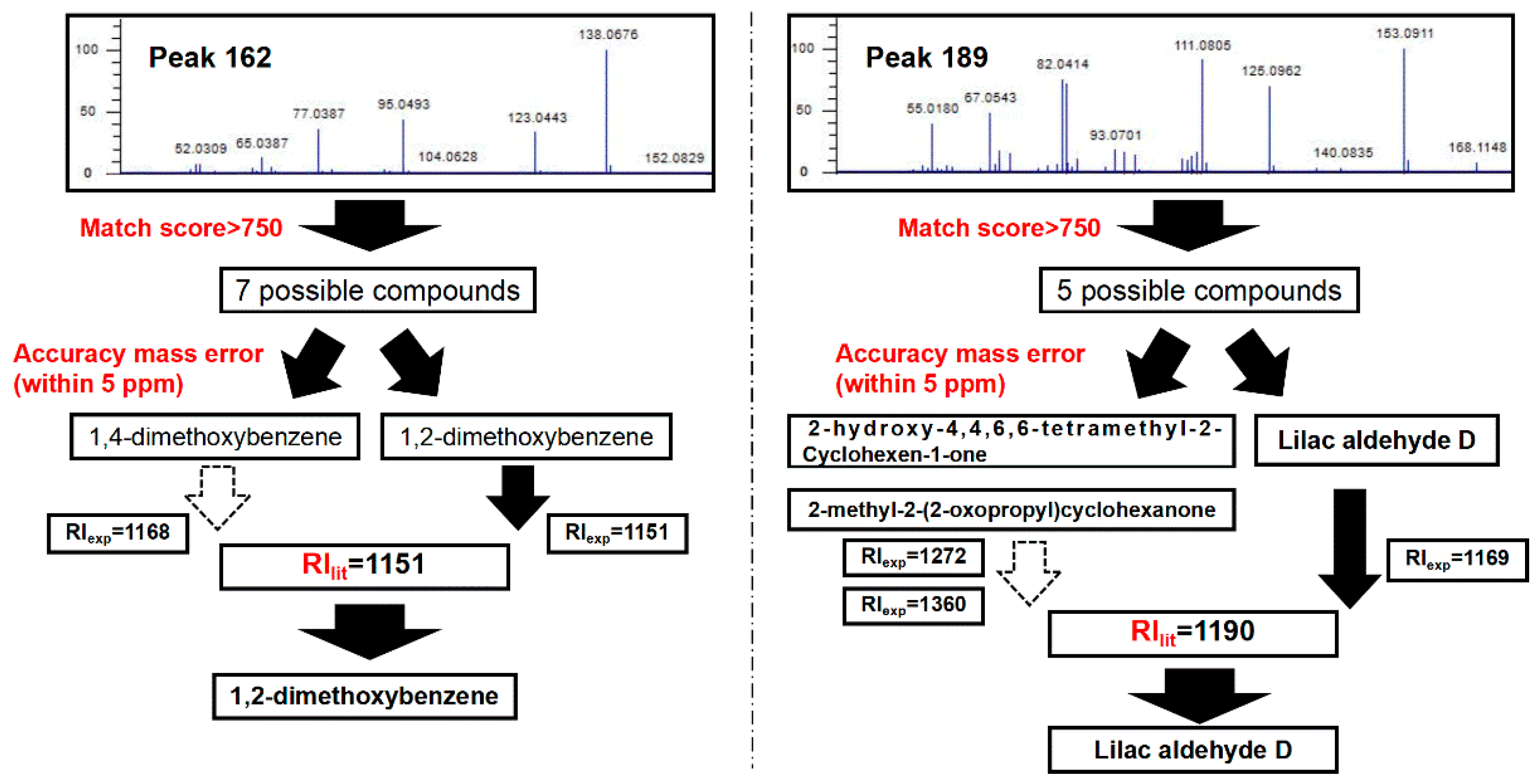
2.2. Identification of Common Volatile Components
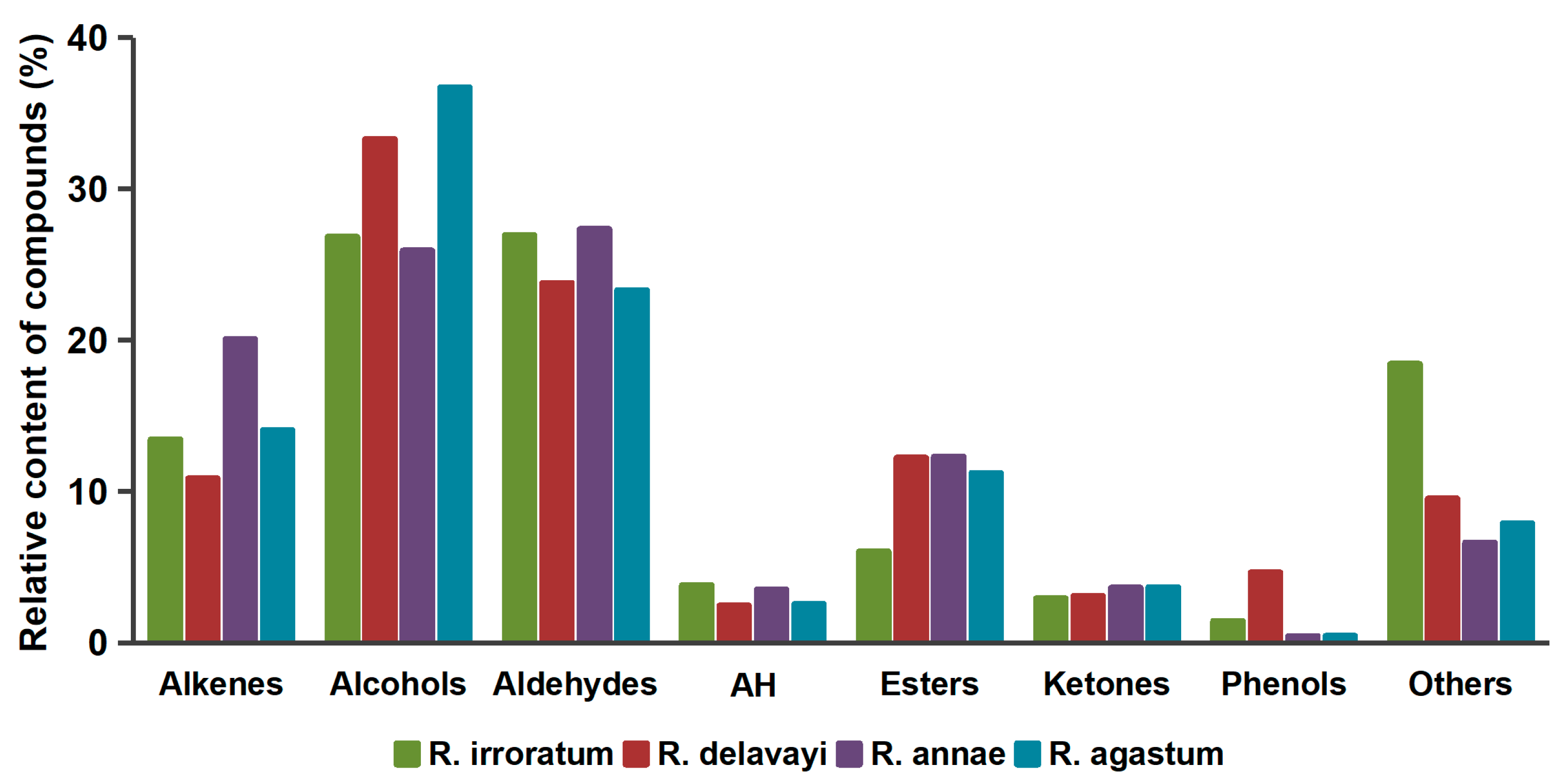
2.3. Volatile Component Analysis
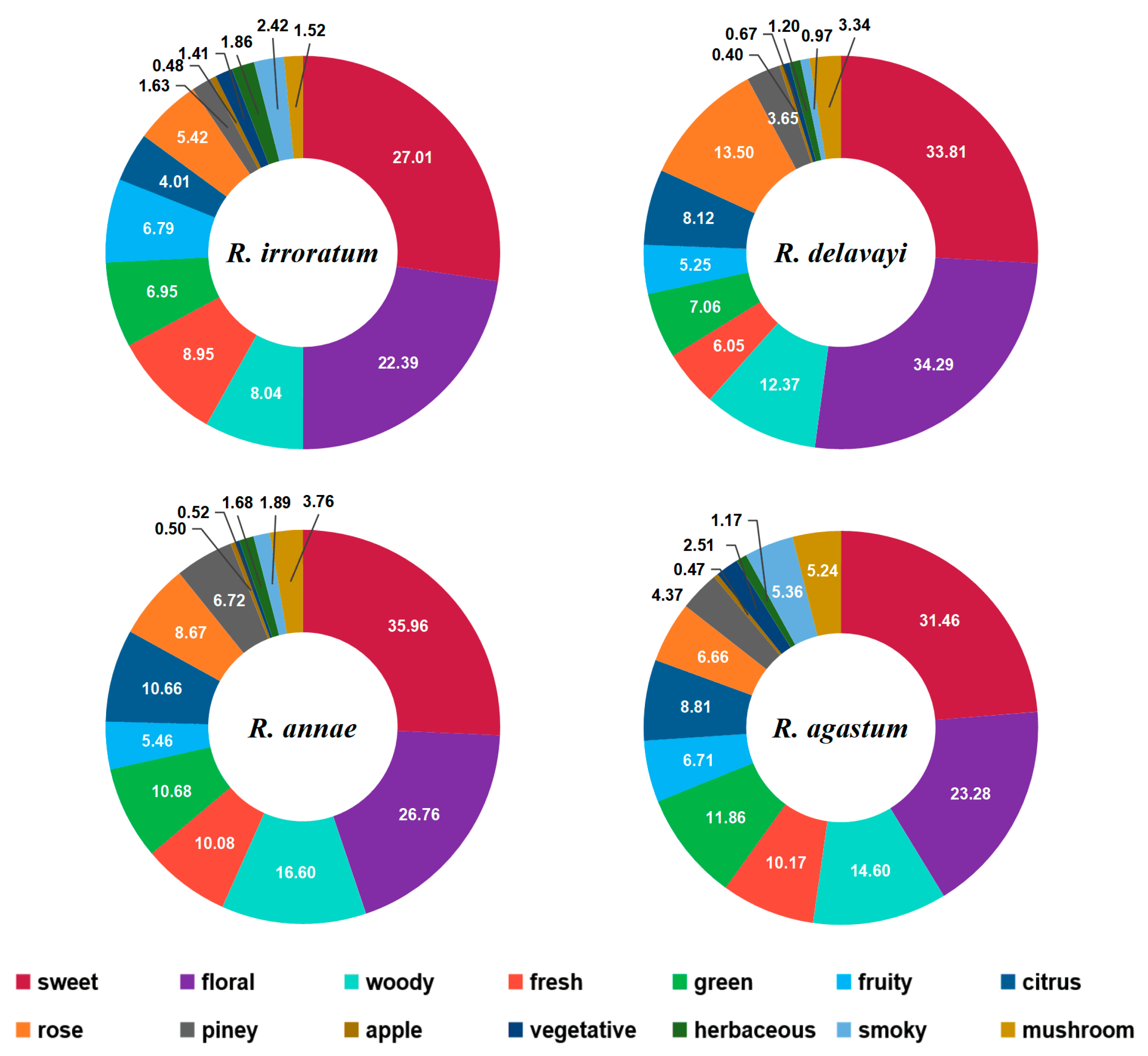
2.4. Odor Analysis
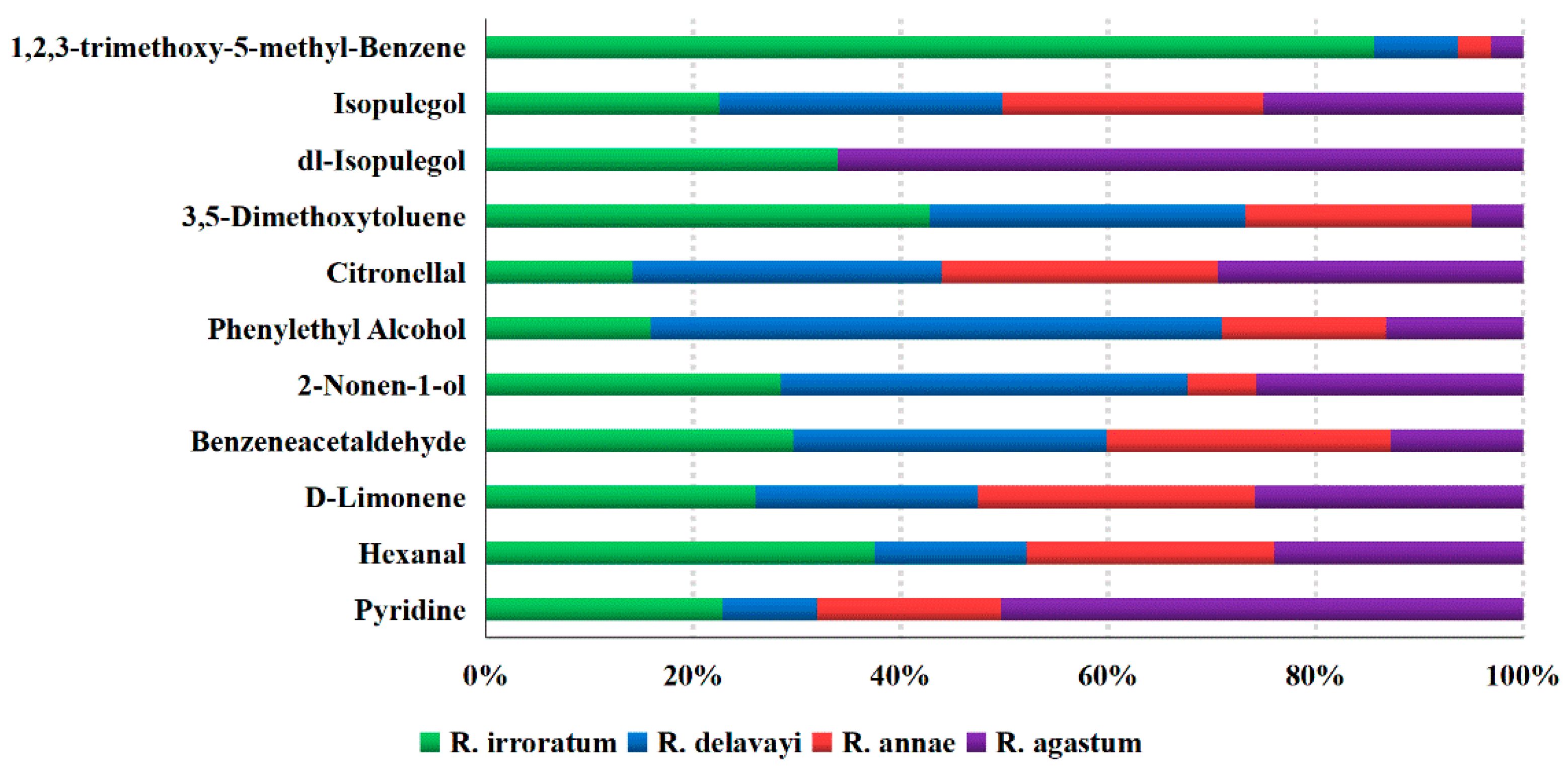
2.4.1. Floral and Woody Odor
2.4.2. Green and Fresh Odor
2.4.3. Sweet and Fruity Odor
2.4.4. Total Odor Description
3. Materials and Methods
3.1. Sample Pretreatment
3.2. SPME Methodology
3.3. Analytical Instrumentation
3.4. Data Method
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Zhou, W.; Oh, J.; Li, W.; Kim, D.W.; Yang, M.H.; Jang, J.H.; Ahn, J.S.; Lee, S.H.; Na, M.K. Chemical constituents of the Korean endangered species Rhododendron Brachycarpum. Biochem. Syst. Ecol. 2014, 56, 231–236. [Google Scholar] [CrossRef]
- Zhang, L.; Xu, P.W.; Cai, Y.F.; Ma, L.L.; Li, S.F.; Li, S.F.; Xie, W.J.; Song, J.; Peng, L.C.; Yan, H.J.; et al. The draft genome assembly of Rhododendron delavayi Franch. var. delavayi. GigaScience 2017, 6, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.Z.; Yao, S.; Tang, C.P.; Ke, C.Q.; Li, L.; Lin, G.; Ye, Y. Diterpenoids from the Flowers of Rhododendron Molle. J. Nat. Prod. 2014, 775, 1185–1192. [Google Scholar] [CrossRef]
- Jung, S.J.; Kim, D.H.; Hong, Y.H.; Lee, J.H.; Song, H.N.; Rho, Y.D.; Baek, N.I. Flavonoids from the flower of Rhododendron yedoense var. Poukhanense and their antioxidant activities. Arch. Pharm. Res. 2007, 30, 146–150. [Google Scholar] [CrossRef] [PubMed]
- Kiruba, S.; Mahesh, M.; Nisha, S.R.; Miller Paul, Z.; Jeeva, S. Phytochemical analysis of the flower extracts of Rhododendron arboreum Sm. ssp. nilagiricum (Zenker) Tagg. Asian Pac. J. Trop. Biomed. 2011, 1, S284–S286. [Google Scholar] [CrossRef]
- Zhu, Y.X.; Zhang, Z.X.; Yan, H.M.; Lu, D.; Zhang, H.P.; Li, L.; Liu, Y.B.; Li, Y. Antinociceptive diterpenoids from the leaves and twigs of Rhododendron Decorum. J. Nat. Prod. 2018, 81, 1183–1192. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.F.; Liu, T.T.; Zhang, H.Q.; Zheng, G.J.; Qiu, Y.; Deng, M.Y.; Zhang, C.; Yao, G.M. Anti-inflammatory grayanane diterpenoids from the leaves of Rhododendron molle. J. Nat. Prod. 2018, 81, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.J.; Lin, S.; Zhu, C.G.; Yang, Y.C.; Li, S.; Zhang, J.J.; Chen, X.G.; Shi, J.G. Highly acylated diterpenoids with a New 3,4-Secograyanane Skeleton from the Flower Buds of\r Rhododendron Molle. Org. Lett. 2010, 12, 1560–1563. [Google Scholar] [CrossRef]
- Li, Y.; Liu, Y.B.; Yu, S.S. Grayanoids from the Ericaceae family: Structures, biological activities and mechanism of action. Phytochem. Rev. 2013, 12, 305–325. [Google Scholar] [CrossRef]
- Zeng, L.T.; Wang, X.Q.; Dong, F.; Watanabe, N.; Yang, Z.Y. Increasing postharvest high-temperatures lead to increased volatile phenylpropanoids/benzenoids accumulation in cut rose (Rosa hybrida) flowers. Postharvest Biol. Tec. 2019, 148, 68–75. [Google Scholar] [CrossRef]
- Pichersky, E.; Gershenzon, J. The formation and function of plant volatiles: Perfumes for pollinator attraction and defense. Curr. Opin. Plant. Biol. 2002, 5, 237–243. [Google Scholar] [CrossRef]
- Junker, R.R.; Blüthgen, N. Floral scents repel potentially nectar-thieving ants. Evol. Ecol. Res. 2008, 10, 295–308. [Google Scholar]
- Pichersky, E.; Dudareva, N. Scent engineering: Toward the goal of controlling how flowers smell. Trends Biotechnol. 2007, 25, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.Y.; Yan, H.; Zhu, Y.; Liu, X.; Lv, H.P.; Zhang, Y.; Dai, W.D.; Guo, L.; Tan, J.F.; Peng, Q.H.; et al. Identification and quantification of key odorants in the world’s four most famous black teas. Food Res. Int. 2019, 121, 73–83. [Google Scholar] [CrossRef]
- Qian, C.Y.; Quan, W.X.; Li, C.C.; Xiang, Z.M. Analysis of volatile terpenoid compounds in Rhododendron species by multidimensional gas chromatography with quadrupole time-of-flight mass spectrometry. Microchem. J. 2019, 149, 104064. [Google Scholar] [CrossRef]
- Zha, H.G.; Milne, R.I.; Sun, H. Asymmetric hybridization in Rhododendron agastum: A hybrid taxon comprising mainly F1 s in Yunnan, China. Ann. Bot. 2010, 105, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Amanpour, A.; Guclu, G.; Kelebek, H.; Selli, S. Characterization of key aroma compounds in fresh and roasted terebinth fruits using aroma extract dilution analysis and GC-MS-Olfactometry. Microchem. J. 2019, 145, 96–104. [Google Scholar] [CrossRef]
- Qia, D.D.; Miaoa, A.Q.; Cao, J.X.; Wang, W.W.; Chen, W.; Pang, S.; He, X.G.; Ma, C.Y. Study on the effects of rapid aging technology on the aroma quality of whitetea using GC-MS combined with chemometrics: In comparison with naturalaged and fresh white tea. Food Chem. 2018, 265, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Lv, H.P.; Dai, W.D.; Guo, L.; Tan, J.F.; Zhang, Y.; Yu, F.L.; Shao, C.Y.; Peng, Q.H.; Lin, Z. Separation of aroma components in Xihu Longjing tea using simultaneous distillation extraction with comprehensive two-dimensional gas chromatography-time-of-flight mass spectrometry. Sep. Purif. Technol. 2016, 164, 146–154. [Google Scholar] [CrossRef]
- Huang, X.H.; Zheng, X.; Chen, Z.H.; Zhang, Y.Y.; Du, M.; Dong, X.P.; Qin, L.; Zhu, B.W. Fresh and grilled eel volatile fingerprinting by e-Nose, GC-O, GC-MS and GC×GC-QTOF combined with purge and trap and solvent-assisted flavor evaporation. Food Res. Int. 2019, 115, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.T.; Li, S.J.; Gao, J.H.; Cheng, K.; Yuan, F. Changes of terpenoids and other volatiles during alcoholic fermentation of blueberry wines made from two southern highbush cultivars. LWT 2019, 109, 233–240. [Google Scholar] [CrossRef]
- Egert, B.; Weinert, C.H.; Kulling, S.E. A peaklet-based generic strategy for the untargeted analysis of comprehensive two-dimensional gas chromatography mass spectrometry data sets. J. Chromatogr. A. 2015, 1405, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Lukić, I.; Carlin, S.; Horvat, I.; Vrhovsek, U. Combined targeted and untargeted profiling of volatile aroma compounds with comprehensive two-dimensional gas chromatography for differentiation of virgin olive oils according to variety and geographical origin. Food Chem. 2019, 270, 403–414. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zeng, Z.D.; Zhao, C.X.; Kong, H.W.; Lu, X.; Xu, G.W. A comparative study of volatile components in green, oolong and black teas by using comprehensive two-dimensional gas chromatography-time-of-flight mass spectrometry and multivariate data analysis. J. Chromatogr. A. 2013, 1313, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Kupska, M.; Chmiel, T.; Jedrkiewicz, R.; Wardencki, W.; Namies´nik, J. Comprehensive two-dimensional gas chromatography for determination of the terpenes profile of blue honeysuckle berries. Food Chem. 2014, 152, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Schwanz, T.G.; Bokowski, L.V.V.; Marcelo, M.C.A.; Jandrey, A.C.; Dias, J.C.; Maximiano, D.H.; Canova, L.S.; Pontes, O.F.S.; Sabin, G.P.; Kaiser, S. Analysis of chemosensory markers in cigarette smoke from different tobacco varieties by GC×GC-TOFMS and chemometrics. Talanta 2019, 202, 74–89. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.X.; Li, X.N.; Liang, Y.Z.; Fang, H.Z.; Huang, L.F.; Guo, F.Q. Comparative analysis of chemical components of essential oils from different samples of Rhododendron with the help of chemometrics methods. Chemom. Intell. Lab. Syst. 2006, 82, 218–228. [Google Scholar] [CrossRef]
- Küçük, S.; Kürkçüoğlu, M.; Başer, K.H.C. Morphological, Indumentum and Chemical Characteristics and Analysis of the Volatile Components of the Flowers of Rhododendron ponticum L. subsp. ponticum (Ericaceae) of Turkish Origin. Rec. Nat. Prod. 2018, 12, 498–507. [Google Scholar]
- Park, C.H.; Yeo, H.J.; Kim, N.S.; Park, Y.E.; Park, S.Y.; Kim, J.K.; Park, S.U. Metabolomic profiling of the white, violet, and red flowers of Rhododendron schlippenbachii Maxim. Molecules 2018, 23, 827. [Google Scholar] [CrossRef]
- Purcaro, G.; Cordero, C.; Liberto, E.; Bicchi, C.; Conte, L.S. Toward a definition of blueprint of virgin olive oil by comprehensive two-dimensional gas chromatography. J. Chromatogr. A. 2014, 1334, 101–111. [Google Scholar] [CrossRef]
- Jiang, M.; Kulsing, C.; Marriott, P.J. Comprehensive 2D gas chromatography-time-of-flight mass spectrometry with 2D retention indices for analysis of volatile compounds in frankincense (Boswellia papyrifera). Anal. Bioanal. Chem. 2018, 410, 3185–3196. [Google Scholar] [CrossRef]
- Feng, S.; Huang, M.Y.; Crane, J.H.; Wang, Y. Characterization of key aroma-active compounds in lychee (Litchi chinensis Sonn.). J. Food Drug Anal. 2018, 26, 497–503. [Google Scholar] [CrossRef]
- Mo, E.K.; Sung, C.K. Phenylethyl alcohol (PEA) application slows fungal growth and maintains aroma in strawberry. Postharvest Biol. Technol. 2007, 45, 234–239. [Google Scholar] [CrossRef]
- An, K.J.; Liu, H.C.; Fu, M.Q.; Qian, M.C.; Yu, Y.S.; Wu, J.J.; Xiao, G.S.; Xu., Y.J. Identification of the cooked off-flavor in heat-sterilized lychee (Litchi chinensis Sonn.) juice by means of molecular sensory science. Food Chem. 2019, 301, 125282. [Google Scholar] [CrossRef]
- Gulati, A.; Ravindranath, S.D. Seasonal variations in quality of Kangra tea (Camellia sinensis (L) O Kuntze) in Himachal Pradesh. J. Sci. Food Agr. 1996, 71, 231–236. [Google Scholar] [CrossRef]
- Zhu, J.C.; Chen, F.; Wang, L.Y.; Niu, Y.W.; Chen, H.X.; Wang, H.L.; Xiao, Z.B. Characterization of the key aroma volatile compounds in Cranberry (Vaccinium macrocarpon Ait.) using gas chromatography-olfactometry (GC-O) and odor activity value (OAV). J. Agr. Food Chem. 2016, 64, 4990–4999. [Google Scholar] [CrossRef]
- Zhu, J.C.; Wang, L.Y.; Xiao, Z.B.; Niu, Y.W. Characterization of the key aroma compounds in mulberry fruits by application of gas chromatography-olfactometry (GC-O), odor activity value (OAV), gas chromatography-mass spectrometry (GC-MS) and flame photometric detection (FPD). Food Chem. 2018, 245, 775–785. [Google Scholar] [CrossRef]
- Lv, H.P.; Zhong, Q.S.; Lin, Z.; Wang, L.; Tan, J.F.; Guo, L. Aroma characterisation of Pu-erh tea using headspace-solid phase microextraction combined with GC/MS and GC–olfactometry. Food Chem. 2012, 130, 1074–1081. [Google Scholar] [CrossRef]
- Feng, Z.H.; Li, Y.F.; Li, M.; Wang, Y.J.; Zhang, L.; Wan, X.C.; Yang, X.G. Tea aroma formation from six model manufacturing processes. Food Chem. 2019, 285, 347–354. [Google Scholar] [CrossRef]
- Silva, É.A.S.; Saboia, G.; Jorge, N.C.; Hoffmann, C.; dos Santos Isaias, R.M.; Soares, G.L.G.; Zini, C.A. Development of a HS-SPME-GC/MS protocol assisted by chemometric tools to study herbivore-induced volatiles in Myrcia splendens. Talanta 2017, 175, 9–20. [Google Scholar] [CrossRef]
- Xiang, Z.M.; Chen, X.T.; Zhao, Z.J.; Xiao, X.; Guo, P.R.; Song, H.C.; Yang, X.; Huang, M.H. Analysis of volatile components in Dalbergia cochinchinensis Pierre by a comprehensive two-dimensional gas chromatography with mass spectrometry method using a solid-state modulator. J. Sep. Sci. 2018, 41, 1–8. [Google Scholar] [CrossRef]
- Wang, C.C.; Zhang, W.J.; Li, H.D.; Mao, J.S.; Guo, C.Y.; Ding, R.Y.; Wang, Y.; Fang, L.P.; Chen, Z.L.; Yang, G.S. Analysis of Volatile Compounds in Pears by HS-SPME-GC×GC-TOFMS. Molecules 2019, 24, 1795. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qian, C.-Y.; Quan, W.-X.; Xiang, Z.-M.; Li, C.-C. Characterization of Volatile Compounds in Four Different Rhododendron Flowers by GC×GC-QTOFMS. Molecules 2019, 24, 3327. https://doi.org/10.3390/molecules24183327
Qian C-Y, Quan W-X, Xiang Z-M, Li C-C. Characterization of Volatile Compounds in Four Different Rhododendron Flowers by GC×GC-QTOFMS. Molecules. 2019; 24(18):3327. https://doi.org/10.3390/molecules24183327
Chicago/Turabian StyleQian, Chen-Yu, Wen-Xuan Quan, Zhang-Min Xiang, and Chao-Chan Li. 2019. "Characterization of Volatile Compounds in Four Different Rhododendron Flowers by GC×GC-QTOFMS" Molecules 24, no. 18: 3327. https://doi.org/10.3390/molecules24183327
APA StyleQian, C.-Y., Quan, W.-X., Xiang, Z.-M., & Li, C.-C. (2019). Characterization of Volatile Compounds in Four Different Rhododendron Flowers by GC×GC-QTOFMS. Molecules, 24(18), 3327. https://doi.org/10.3390/molecules24183327





