Neuroprotective Potential of Secondary Metabolites from Melicope lunu-ankenda (Rutaceae)
Abstract
1. Introduction
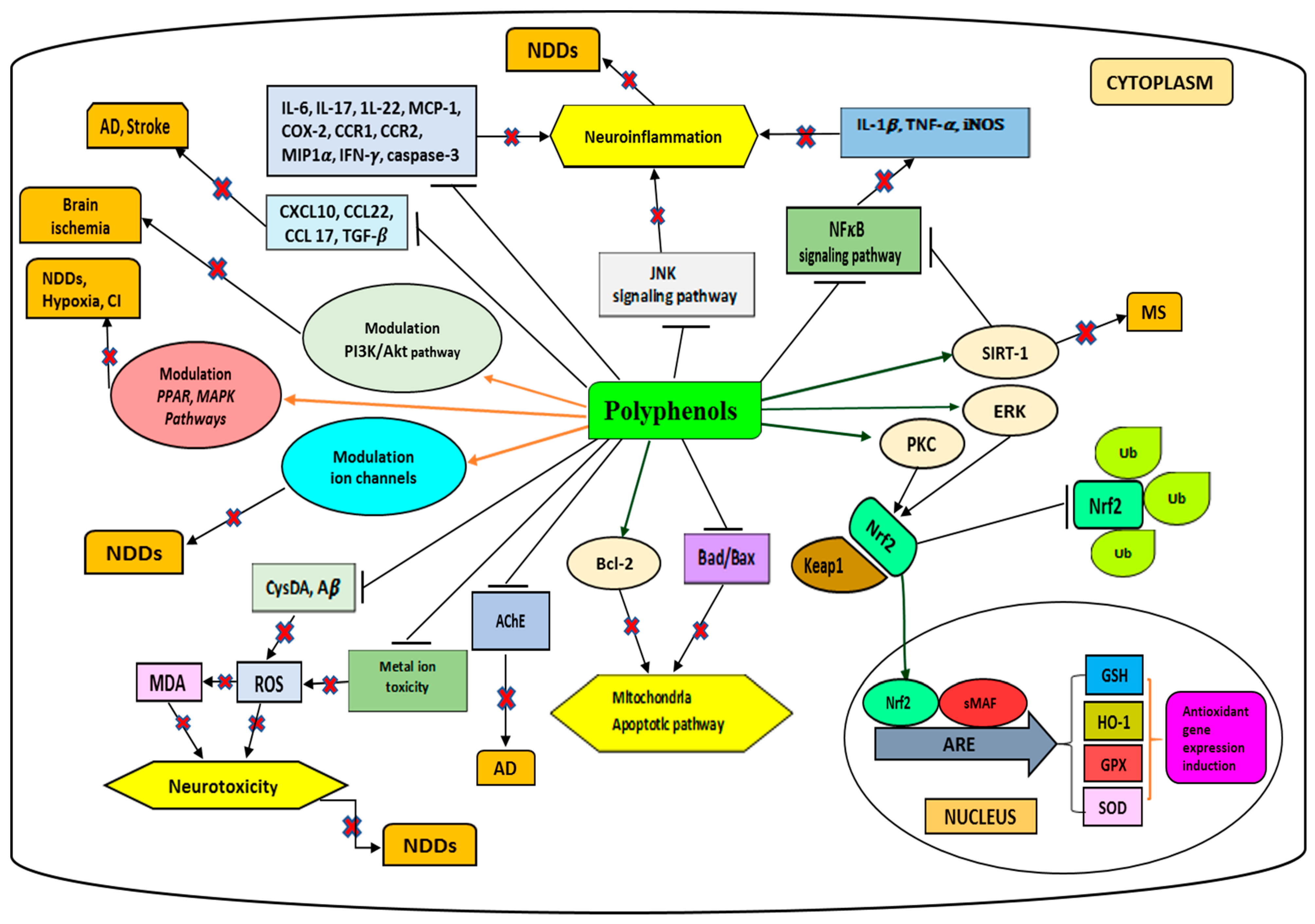
2. Polyphenols
2.1. Phenolic Acids
2.1.1. Cinnamic Acid Derivatives
Caffeic Acid
Caffeoylquinic Acid
Coumaric Acid
Ferulic Acid
Sinapic Acid
2.1.2. Benzoic Acid Derivatives
Gallic Acid
2.2. Flavonoids
2.2.1. Quercetin
2.2.2. Isorhamnetin
2.2.3. Catechin
3. Coumarins
3.1. Scopoletin
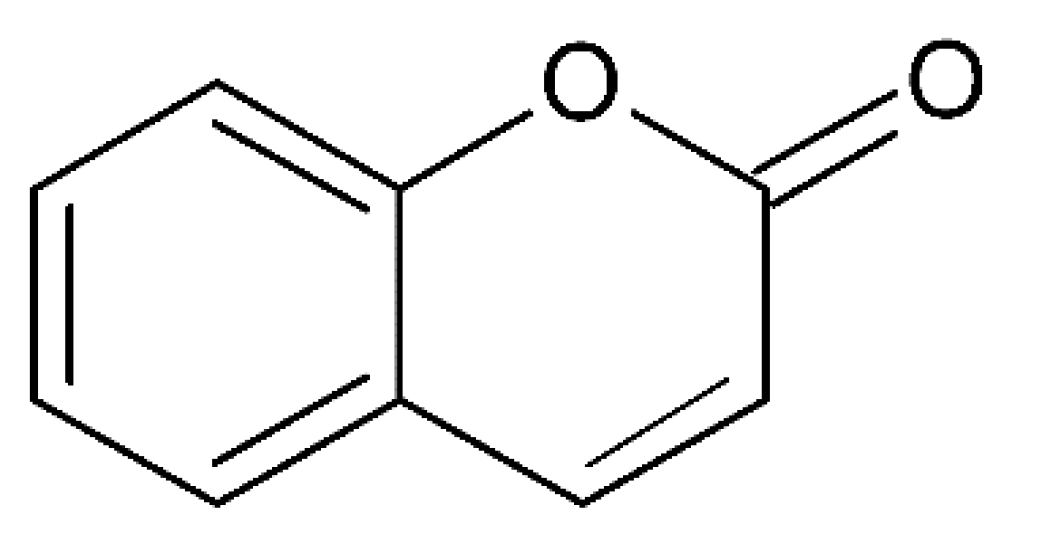
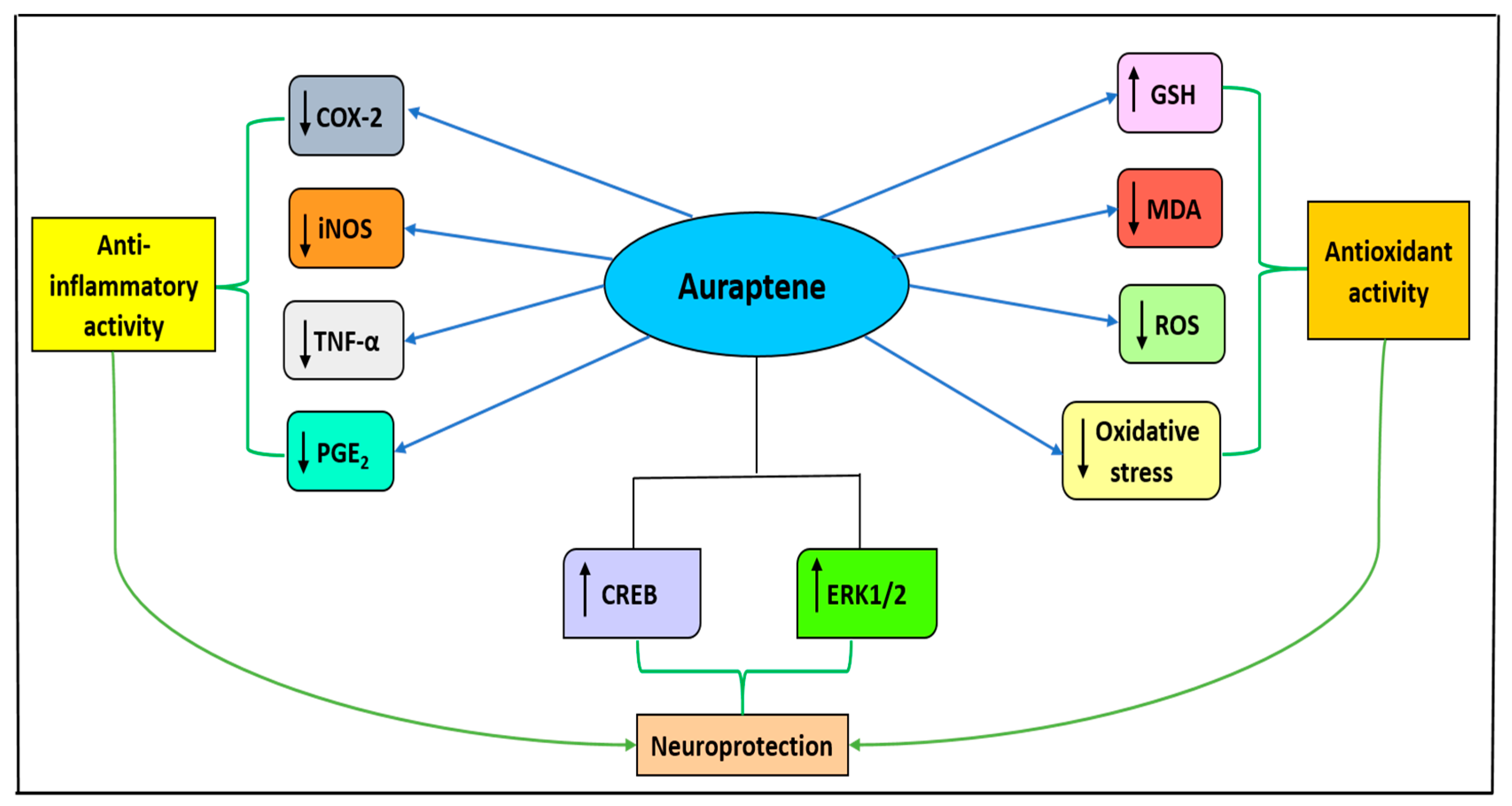
3.2. Auraptene
3.3. Esculetin
4. Alkaloids
4.1. Skimmianine
4.2. Evolitrin
5. Chromenes
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Perez-Hernandez, J.; Zaldivar-Machorro, V.J.; Villanueva-Porras, D.; Vega-Avila, E.; Chavarria, A. A potential alternative against neurodegenerative diseases: Phytodrugs. Oxid. Med. Cell. Longev. 2016, 8378613. [Google Scholar] [CrossRef] [PubMed]
- Ratheesh, G.; Tian, L.; Venugopal, J.R.; Ezhilarasu, H.; Sadiq, A.; Fan, T.P.; Ramakrishna, S. Role of medicinal plants in neurodegenerative diseases. Biomanuf. Rev. 2017, 2, 2. [Google Scholar] [CrossRef]
- Al-Zuaidy, M.H.; Hamid, A.A.; Ismail, A.; Mohamed, S.; Abdul Razis, A.F.; Mumtaz, M.W.; Salleh, S.Z. Potent antidiabetic activity and metabolite profiling of Melicope lunu-ankenda leaves. J. Food Sci. 2016, 81, C1080–C1090. [Google Scholar] [CrossRef] [PubMed]
- Eliaser, M.E.; Hui Ho, J.; Hashim, N.M.; Rukayadi, Y.; Lian Ee, G.C.; Abdull Razis, A.F. Phytochemical constituents and biological activities of Melicope lunu-ankenda. Molecules 2018, 23, 2708. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.J.; Kumar, R.A.; Rasheed, S.A.; Chandrika, S.P.; Chandrasekhar, A.; Baby, S.; Subramoniam, A. Antipyretic, analgesic, anti-inflammatory and antioxidant activities of two major chromenes from Melicope lunu-ankenda. J. Ethnopharmacol. 2010, 130, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.P.; Khanum, F. Neuroprotective potential of phytochemicals. Pharmacogn. Rev. 2012, 6, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Singh, B.; Singh, N. A review on coumarins as acetylcholinesterase inhibitors for Alzheimer’s disease. Bioorg. Med. Chem. 2012, 20, 1175–1180. [Google Scholar] [CrossRef]
- Soobrattee, M.A.; Neergheen, V.S.; Luximon-Ramma, A.; Aruoma, O.I.; Bahorun, T. Phenolics as potential antioxidant therapeutic agents: Mechanism and actions. Mutat. Res.-Fund. Mol. M. 2005, 579, 200–213. [Google Scholar] [CrossRef]
- Othman, A.; Mukhtar, N.J.; Ismail, N.S.; Chang, S.K. Phenolics, flavonoids content and antioxidant activities of 4 Malaysian herbal plants. Int. Food Res. J. 2014, 21, 759–766. [Google Scholar]
- Sroka, Z.; Cisowski, W. Hydrogen peroxide scavenging, antioxidant and anti-radical activity of some phenolic acids. Food Chem. Toxicol. 2003, 41, 753–758. [Google Scholar] [CrossRef]
- Bhullar, K.S.; Rupasinghe, H.P.V. Polyphenols: Multipotent therapeutic agents in neurodegenerative diseases. Oxid. Med. Cell Longev. 2013, 2013, 891748. [Google Scholar] [CrossRef]
- Song, J.X.; Sun, Y.R.; Peluso, I.; Zeng, Y.; Yu, X.; Lu, J.H.; Xu, Z.; Wang, M.Z.; Liu, L.F.; Huang, Y.Y.; et al. A novel curcumin analog binds to and activates TFEB in vitro and in vivo independent of MTOR inhibition. Autophagy 2016, 12, 1372–1389. [Google Scholar] [CrossRef]
- Squillaro, T.; Schettino, C.; Sampaolo, S.; Galderisi, U.; Di Iorio, G.; Giordano, A.; Melone, M.A.B. Adult-onset brain tumors and neurodegeneration: Are polyphenols protective? Cell. Physiol. 2018, 233, 3955–3967. [Google Scholar] [CrossRef]
- Squillaro, T.; Cimini, A.; Peluso, G.; Giordano, A.; Melone, M.A.B. Nano-delivery systems for encapsulation of dietary polyphenols: An experimental approach for neurodegenerative diseases and brain tumors. Biochem. Pharmacol. 2018, 154, 303–317. [Google Scholar] [CrossRef]
- Finicelli, M.; Squillaro, T.; Di Cristo, F.; Di Salle, A.; Melone, M.A.B.; Galderisi, U.; Peluso, G. Metabolic syndrome, Mediterranean diet, and polyphenols: Evidence and perspectives. J. Cell. Physiol. 2019, 234, 5807–5826. [Google Scholar] [CrossRef]
- Natella, F.; Nardini, M.; Di Felice, M.; Scaccini, C. Benzoic and cinnamic acid derivatives as antioxidants: Structure− activity relation. J. Agric. Food Chem. 1999, 47, 1453–1459. [Google Scholar] [CrossRef]
- Lan, J.S.; Hou, J.W.; Liu, Y.; Ding, Y.; Zhang, Y.; Li, L.; Zhang, T. Design, synthesis and evaluation of novel cinnamic acid derivatives bearing N-benzyl pyridinium moiety as multifunctional cholinesterase inhibitors for Alzheimer’s disease. J. Enzyme Inhib. Med. Chem. 2017, 32, 776–788. [Google Scholar] [CrossRef]
- Sul, D.; Kim, H.-S.; Lee, D.; Joo, S.S.; Hwang, K.W.; Park, S.-Y. Protective effect of caffeic acid against beta-amyloid-induced neurotoxicity by the inhibition of calcium influx and tau phosphorylation. Life Sci. 2009, 84, 257–262. [Google Scholar] [CrossRef]
- Jeong, C.-H.; Jeong, H.R.; Choi, G.N.; Kim, D.-O.; Lee, U.; Heo, H.J. Neuroprotective and anti-oxidant effects of caffeic acid isolated from Erigeron annuus leaf. Chin. Med. 2011, 6, 25. [Google Scholar] [CrossRef]
- Vauzour, D.; Corona, G.; Spencer, J.P. Caffeic acid, tyrosol and p-coumaric acid are potent inhibitors of 5-S-cysteinyl-dopamine induced neurotoxicity. Arch. Biochem. Biophys. 2010, 501, 106–111. [Google Scholar] [CrossRef]
- Garrido, J.; Gaspar, A.; Garrido, E.M.; Miri, R.; Tavakkoli, M.; Pourali, S.; Saso, L.; Borges, F.; Firuzi, O. Alkyl esters of hydroxycinnamic acids with improved antioxidant activity and lipophilicity protect PC12 cells against oxidative stress. Biochimie 2012, 94, 961–967. [Google Scholar] [CrossRef]
- Scapagnini, G.; Foresti, R.; Calabrese, V.; Stella, A.G.; Green, C.J.; Motterlini, R. Caffeic acid phenethyl ester and curcumin: A novel class of heme oxygenase-1 inducers. Mol. Pharmacol. 2002, 61, 554–561. [Google Scholar] [CrossRef]
- Morroni, F.; Sita, G.; Graziosi, A.; Turrini, E.; Fimognari, C.; Tarozzi, A.; Hrelia, P. Neuroprotective effect of caffeic acid phenethyl ester in a mouse model of Alzheimer’s disease involves Nrf2/HO-1 pathway. Aging Dis. 2018, 9, 605. [Google Scholar] [CrossRef]
- Zaitone, S.A.; Ahmed, E.; Elsherbiny, N.M.; Mehanna, E.T.; El-Kherbetawy, M.K.; ElSayed, M.H.; Moustafa, Y.M. Caffeic acid improves locomotor activity and lessens inflammatory burden in a mouse model of rotenone-induced nigral neurodegeneration: Relevance to Parkinson’s disease therapy. Pharmacol. Rep. 2019, 71, 32–41. [Google Scholar] [CrossRef]
- Han, J.; Miyamae, Y.; Shigemori, H.; Isoda, H. Neuroprotective effect of 3, 5-di-O-caffeoylquinic acid on SH-SY5Y cells and senescence-accelerated-prone mice 8 through the up-regulation of phosphoglycerate kinase-1. Neuroscience 2010, 169, 1039–1045. [Google Scholar] [CrossRef]
- Tian, X.; Gao, L.; An, L.; Jiang, X.; Bai, J.; Huang, J.; Zhao, Q. Pretreatment of MQA, a caffeoylquinic acid derivative compound, protects against H2O2-induced oxidative stress in SH-SY5Y cells. Neurol. Res. 2016, 38, 1079–1087. [Google Scholar] [CrossRef]
- Chen, L.; Liu, D.N.; Wang, Y.; Liu, X.Y.; Han, S.; Zhang, K.; Wang, J.H. Treatment with MQA, a Derivative of Caffeoylquinic Acid, Provides Neuroprotective Effects against Cerebral Ischemia Through Suppression of the p38 Pathway and Oxidative Stress in Rats. J. Mol. Neurosci. 2019, 67, 604–612. [Google Scholar] [CrossRef]
- Guven, M.; Sehitoglu, M.H.; Yuksel, Y.; Tokmak, M.; Aras, A.B.; Akman, T.; Cosar, M. The neuroprotective effect of coumaric acid on spinal cord ischemia/reperfusion injury in rats. Inflammation 2015, 38, 1986–1995. [Google Scholar] [CrossRef]
- Hong, S.Y.; Jeong, W.S.; Jun, M. Protective effects of the key compounds isolated from Corni fructus against β-amyloid-induced neurotoxicity in PC12 cells. Molecules 2012, 17, 10831–10845. [Google Scholar] [CrossRef]
- Kanski, J.; Aksenova, M.; Stoyanova, A.; Butterfield, D.A. Ferulic acid antioxidant protection against hydroxyl and peroxyl radical oxidation in synaptosomal and neuronal cell culture systems in vitro: Structure-activity studies. J. Nutr. Biochem. 2002, 13, 273–281. [Google Scholar] [CrossRef]
- Mancuso, C.; Santangelo, R. Ferulic acid: Pharmacological and toxicological aspects. Food Chem. Toxicol. 2014, 65, 185–195. [Google Scholar] [CrossRef]
- Sgarbossa, A.; Giacomazza, D.; Di Carlo, M. Ferulic acid: A hope for Alzheimer’s disease therapy from plants. Nutrients 2015, 7, 5764–5782. [Google Scholar] [CrossRef]
- Ono, K.; Hirohata, M.; Yamada, M. Ferulic acid destabilizes preformed β-amyloid fibrils in vitro. Biochem. Biophys. Res. Commun. 2005, 336, 444–449. [Google Scholar] [CrossRef]
- Nićiforović, N.; Abramovič, H. Sinapic acid and its derivatives: Natural sources and bioactivity. Compr. Rev. Food Sci. F. 2014, 13, 34–51. [Google Scholar] [CrossRef]
- Lee, H.E.; Kim, D.H.; Park, S.J.; Kim, J.M.; Lee, Y.W.; Jung, J.M.; Lee, C.H.; Hong, J.G.; Liu, X.; Cai, M.; et al. Neuroprotective effect of sinapic acid in a mouse model of amyloid β1–42 protein-induced Alzheimer’s disease. Pharmacol. Biochem. Behav. 2012, 103, 260–266. [Google Scholar] [CrossRef]
- Zare, K.; Eidi, A.; Roghani, M.; Rohani, A.H. The neuroprotective potential of sinapic acid in the 6-hydroxydopamine-induced hemi-parkinsonian rat. Metab. Brain Dis. 2015, 30, 205–213. [Google Scholar] [CrossRef]
- Mansouri, M.T.; Farbood, Y.; Sameri, M.J.; Sarkaki, A.; Naghizadeh, B.; Rafeirad, M. Neuroprotective effects of oral gallic acid against oxidative stress induced by 6-hydroxydopamine in rats. Food Chem. 2013, 138, 1028–1033. [Google Scholar] [CrossRef]
- Lu, Z.; Nie, G.; Belton, P.S.; Tang, H.; Zhao, B. Structure–activity relationship analysis of antioxidant ability and neuroprotective effect of gallic acid derivatives. Neurochem. Int. 2006, 48, 263–274. [Google Scholar] [CrossRef]
- Mirshekar, M.A.; Sarkaki, A.; Farbood, Y.; Naseri, M.K.G.; Badavi, M.; Mansouri, M.T.; Haghparast, A. Neuroprotective effects of gallic acid in a rat model of traumatic brain injury: Behavioral, electrophysiological, and molecular studies. Iran. J. Basic Med. Sci. 2018, 21, 1056. [Google Scholar]
- Maya, S.; Prakash, T.; Madhu, K. Assessment of neuroprotective effects of Gallic acid against glutamate-induced neurotoxicity in primary rat cortex neuronal culture. Neurochem. Int. 2018, 121, 50–58. [Google Scholar] [CrossRef]
- Spagnuolo, C.; Moccia, S.; Russo, G.L. Anti-inflammatory effects of flavonoids in neurodegenerative disorders. Eur. J. Med. Chem. 2018, 153, 105–115. [Google Scholar] [CrossRef]
- Dok-Go, H.; Lee, K.H.; Kim, H.J.; Lee, E.H.; Lee, J.; Song, Y.S.; Lee, Y.-H.; Jin, C.; Lee, Y.S.; Cho, J. Neuroprotective effects of antioxidative flavonoids, quercetin, (+)-dihydroquercetin and quercetin 3-methyl ether, isolated from Opuntia ficus-indica var. saboten. Brain Res. 2003, 965, 130–136. [Google Scholar] [CrossRef]
- Zhang, Z.J.; Cheang, L.C.V.; Wang, M.W.; Lee, S.M.Y. Quercetin exerts a neuroprotective effect through inhibition of the iNOS/NO system and pro-inflammation gene expression in PC12 cells and in zebrafish. Int. J. Mol. Med. 2011, 27, 195–203. [Google Scholar]
- Dajas, F. Life or death: Neuroprotective and anticancer effects of quercetin. J. Ethnopharmacol. 2012, 143, 383–396. [Google Scholar] [CrossRef]
- Jembrek, M.J.; Vlainić, J.; Čadež, V.; Šegota, S. Atomic force microscopy reveals new biophysical markers for monitoring subcellular changes in oxidative injury: Neuroprotective effects of quercetin at the nanoscale. PLoS ONE 2018, 13, e0200119. [Google Scholar]
- Zhao, J.J.; Song, J.Q.; Pan, S.Y.; Wang, K. Treatment with isorhamnetin protects the brain against ischemic injury in mice. Neurochem. Res. 2016, 41, 1939–1948. [Google Scholar] [CrossRef]
- Ishola, I.O.; Osele, M.O.; Chijioke, M.C.; Adeyemi, O.O. Isorhamnetin enhanced cortico-hippocampal learning and memory capability in mice with scopolamine-induced amnesia: Role of antioxidant defense, cholinergic and BDNF signaling. Brain Res. 2019, 1712, 188–196. [Google Scholar] [CrossRef]
- Mandel, S.; Youdim, M.B.H. Catechin polyphenols: Neurodegeneration and neuroprotection in neurodegenerative diseases. Free Radic. Biol. Med. 2004, 37, 304–317. [Google Scholar] [CrossRef]
- Zhao, X.; Li, R.; Jin, H.; Jin, H.; Wang, Y.; Zhang, W.; Chen, W. Epigallocatechin-3-gallate confers protection against corticosterone-induced neuron injuries via restoring extracellular signal-regulated kinase 1/2 and phosphatidylinositol-3 kinase/protein kinase B signaling pathways. PLoS ONE 2018, 13, e0192083. [Google Scholar] [CrossRef]
- Jameel, E.; Umar, T.; Kumar, J.; Hoda, N. Coumarin: A privileged scaffold for the design and development of antineurodegenerative agents. Chem. Biol. Drug Des. 2016, 87, 21–38. [Google Scholar] [CrossRef]
- Epifano, F.; Curini, M.; Menghini, L.; Genovese, S. Natural coumarins as a novel class of neuroprotective agents. Mini-Rev. Med. Chem. 2009, 9, 1262–1271. [Google Scholar] [CrossRef]
- Venugopala, K.N.; Rashmi, V.; Odhav, B. Review on natural coumarin lead compounds for their pharmacological activity. Biomed. Res. Int. 2013, 2013, 963248. [Google Scholar] [CrossRef]
- Ali, M.Y.; Jannat, S.; Jung, H.A.; Choi, R.J.; Roy, A.; & Choi, J.S. Anti-Alzheimer’s disease potential of coumarins from Angelica decursiva and Artemisia capillaris and structure-activity analysis. Asian Pac. J. Trop. Med. 2016, 9, 103–111. [Google Scholar] [CrossRef]
- Matos, M.J.; Santana, L.; Uriarte, E.; Abreu, O.A.; Molina, E.; Yordi, E.G. Coumarins—An Important Class of Phytochemicals. In Phytochemicals-Isolation, Characterisation and Role in Human Health; Rao, A.V., Rao, L.G., Eds.; InTech: Rijeka, Croatia, 2015. [Google Scholar]
- Kostova, I.; Bhatia, S.; Grigorov, P.; Balkansky, S.; Parmar, V.S.; Prasad, A.K.; Saso, L. Coumarins as antioxidants. Curr. Med. Chem. 2011, 18, 3929–3951. [Google Scholar] [CrossRef]
- Lee, B.; Weon, J.B.; Eom, M.R.; Jung, Y.S.; Ma, C.J. Neuroprotective compounds of Tilia amurensis. Pharmacogn. Mag. 2015, 11, S303. [Google Scholar]
- Shaw, C.-Y.; Chen, C.-H.; Hsu, C.-C.; Chen, C.-C.; Tsai, Y.-C. Antioxidant properties of scopoletin isolated from Sinomonium acutum. Phytother. Res. 2003, 17, 823–825. [Google Scholar] [CrossRef]
- Kaur, M.; Prakash, A.; Kalia, A.N. Neuroprotective potential of antioxidant potent fractions from Convolvulus pluricaulis Chois. in 3-nitropropionic acid challenged rats. Nutr. Neurosci. 2016, 19, 70–78. [Google Scholar] [CrossRef]
- García-Morales, G.; Huerta-Reyes, M.; González-Cortazar, M.; Zamilpa, A.; Jiménez-Ferrer, E.; Silva-García, R.; Aguilar-Rojas, A. Anti-inflammatory, antioxidant and anti-acetylcholinesterase activities of Bouvardia ternifolia: Potential implications in Alzheimer’s disease. Arch. Pharm. Res. 2015, 38, 1369–1379. [Google Scholar] [CrossRef]
- Sahebkar, A. Citrus auraptene: A potential multifunctional therapeutic agent for nonalcoholic fatty liver disease. Ann. Hepatol. 2011, 10, 575–577. [Google Scholar] [CrossRef]
- Okuyama, S.; Minami, S.; Shimada, N.; Makihata, N.; Nakajima, M.; Furukawa, Y. Anti-inflammatory and neuroprotective effects of auraptene, a citrus coumarin, following cerebral global ischemia in mice. Eur. J. Pharmacol. 2013, 699, 118–123. [Google Scholar] [CrossRef]
- Furukawa, Y.; Watanabe, S.; Okuyama, S.; Nakajima, M. Neurotrophic Effect of Citrus Auraptene: Neuritogenic Activity in PC12 Cells. Int. J. Mol. Sci. 2012, 13, 5338–5347. [Google Scholar] [CrossRef]
- Ghanbarabadi, M.; Iranshahi, M.; Amoueian, S.; Mehri, S.; Motamedshariaty, V.S.; Mohajeri, S.A. Neuroprotective and memory enhancing effects of auraptene in a rat model of vascular dementia: Experimental study and histopathological evaluation. Neurosci. Lett. 2016, 623, 13–21. [Google Scholar] [CrossRef]
- Wang, C.; Pei, A.; Chen, J.; Yu, H.; Sun, M.-L.; Liu, C.-F.; Xu, X. A natural coumarin derivative esculetin offers neuroprotection on cerebral ischemia/reperfusion injury in mice. J. Neurochem. 2012, 121, 1007–1013. [Google Scholar] [CrossRef]
- Wansi, J.D.; Devkota, K.P.; Tshikalange, E.; Kuete, V. Alkaloids from the medicinal plants of Africa. In Medicinal Plant Research in Africa, 1st ed.; Kuete, V., Ed.; Elsevier: Amsterdam, The Netherlands, 2013; pp. 557–605. [Google Scholar]
- Hussain, G.; Rasul, A.; Anwar, H.; Aziz, N.; Razzaq, A.; Wei, W.; Ali, M.; Li, J.; Li, X. Role of Plant Derived Alkaloids and Their Mechanism in Neurodegenerative Disorders. Int. J. Biol. Sci. 2018, 14, 341–357. [Google Scholar] [CrossRef]
- Ratheesh, M.; Sindhu, G.; Helen, A. Anti-inflammatory effect of quinoline alkaloid skimmianine isolated from Ruta graveolens L. Inflamm. Res. 2013, 62, 367–376. [Google Scholar] [CrossRef]
- Lal, B.; Bhise, N.B.; Gidwani, R.M.; Lakdawala, A.D.; Joshi, K.; Patvardhan, S. Isolation, synthesis and biological activity of Evolitrine and analogs. ARKIVOC 2005, 11, 77–97. [Google Scholar]
- Ratnayake, W.M.K.M.; Suresh, T.S.; Abeysekera, A.M.; Salim, N.; Chandrika, U.G. Acute anti-inflammatory and anti-nociceptive activities of crude extracts, alkaloid fraction and evolitrine from Acronychia pedunculata leaves. J. Ethnopharmacol. 2019, 238, 111827. [Google Scholar] [CrossRef]
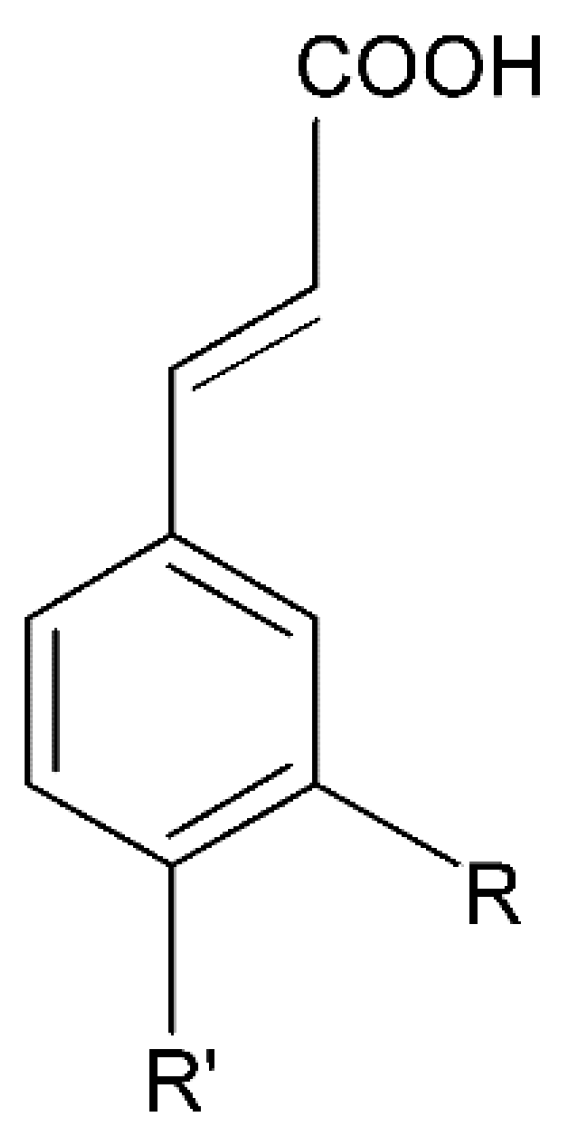
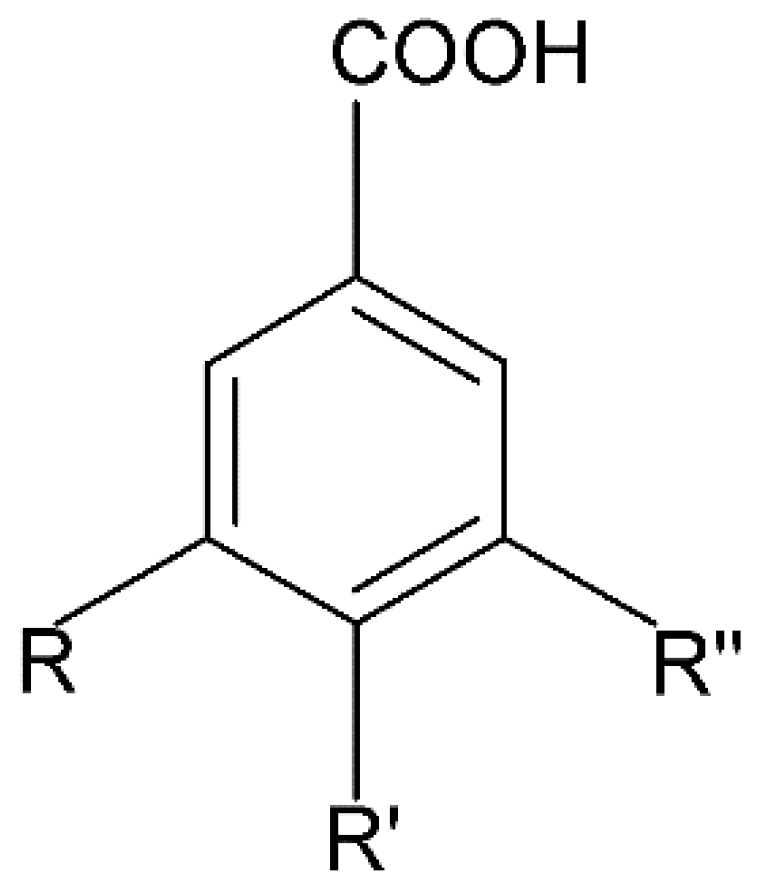
| Group | Compound | Chemical Structure | NDDs | Mechanism of Action | Ref. |
|---|---|---|---|---|---|
| Hydroxycinnamic acids | |||||
| Caffeic aid | 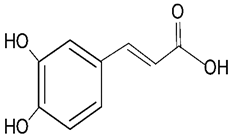 | AD PD | ↓oxidative stress, ↓calcium influx, ↓phosphorylation of GSK-3β (in vitro); PC12 cell line ↓formation of CysDA (in vitro); primary mouse cortical neurons | [18] [20] | |
| Ferulic acid | 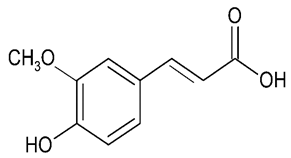 | NDDs particularly AD AD AD | ↓oxidative stress (in vitro); synaptosomal and neuronal cell culture ↓Aβ aggregation, ↓IL-1β (in vivo); APP/PS1 AD mouse model ↓ Aβ aggregation (in vivo); Tg2576 AD mouse model | [30] [31] [32] | |
| Caffeoylquinic acid (3,5-di-O-CQA) |  | NDDs | induction of PGK1 (in vitro); SH-SY5Y cell line | [25] | |
| Coumaric acid | 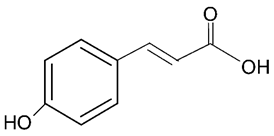 | NDDs particularly SCIR | ↓ MDA, ↑ SOD (in vivo); rat model | [28] | |
| Sinapic acid | 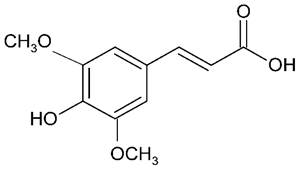 | NDDs particularly AD PD | ↓ Aβ1–42, ↓caspase-3, ↓apoptosis (in vivo); mouse model ↓oxidative stress, ↓MDA, ↑ SOD, ↓iron level (in vivo); rat model | [35] [36] | |
| Hydroxybenzoic acid | |||||
| Gallic acid | 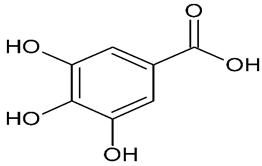 | PD | ↓ MDA, ↑ GPx (in vivo); rat model | [37] | |
| Flavonoids | |||||
| Quercetin |  | NDDs | ↓NO, ↓iNOS (in vitro); PC12 cell line ↓IL-1β, ↓COX-2, ↓TNF-α (in vivo); zebrafish model ↓ MDA, ↑ SOD (in vivo); mouse model | [43] [43] [44] | |
| Isorhamnetin | 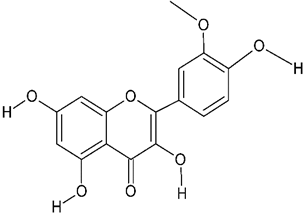 | Ischemic stroke | ↓NR1, ↓oxidative stress, ↑ Nrf2/HO-1 ↓ iNOS, ↓ NO (in vivo); mouse model | [46] | |
| Catechin | 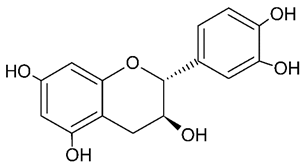 | Brain ischemia | ↓ NF-κB, ↓IL-1β, ↓TNF-α (in vivo); rat model | [11] | |
| Coumarins | |||||
| Scopoletin | 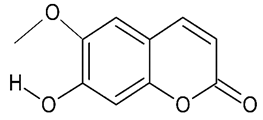 | AD | antioxidant (in vitro); HT22 cell line | [56] | |
| Auraptene |  | CI NDDs | ↓inflammatory response, ↓COX2 (in vivo); mouse model ↑ERK1/2, ↑CREB (in vitro); PC12 cell line | [61] [61] | |
| Esculetin |  | Cerebral I/R | ↓ apoptotic, ↑ Bcl-2, ↓ Bax (in vivo); mouse model | [64] | |
| Alkaloids | |||||
| Skimmianine |  | NNDs | ↓ TNF-α, ↓COX2, ↓ PGE2 (in vivo); rat model | [67] | |
| Evolitrine | 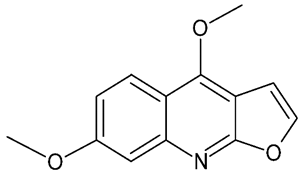 | NNDs | anti-inflammatory (in vivo); rat model | [68] | |
| Chromenes | |||||
| Evodione | 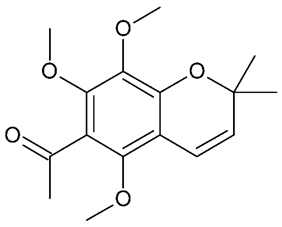 | Anti- inflammatory | anti-inflammatory | [5] | |
| Leptonol |  | Antioxidant anti-inflammatory | Antioxidant anti-inflammatory | [5] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdulwanis Mohamed, Z.; Mohamed Eliaser, E.; Mazzon, E.; Rollin, P.; Cheng Lian Ee, G.; Abdull Razis, A.F. Neuroprotective Potential of Secondary Metabolites from Melicope lunu-ankenda (Rutaceae). Molecules 2019, 24, 3109. https://doi.org/10.3390/molecules24173109
Abdulwanis Mohamed Z, Mohamed Eliaser E, Mazzon E, Rollin P, Cheng Lian Ee G, Abdull Razis AF. Neuroprotective Potential of Secondary Metabolites from Melicope lunu-ankenda (Rutaceae). Molecules. 2019; 24(17):3109. https://doi.org/10.3390/molecules24173109
Chicago/Turabian StyleAbdulwanis Mohamed, Zeinab, Enas Mohamed Eliaser, Emanuela Mazzon, Patrick Rollin, Gwendoline Cheng Lian Ee, and Ahmad Faizal Abdull Razis. 2019. "Neuroprotective Potential of Secondary Metabolites from Melicope lunu-ankenda (Rutaceae)" Molecules 24, no. 17: 3109. https://doi.org/10.3390/molecules24173109
APA StyleAbdulwanis Mohamed, Z., Mohamed Eliaser, E., Mazzon, E., Rollin, P., Cheng Lian Ee, G., & Abdull Razis, A. F. (2019). Neuroprotective Potential of Secondary Metabolites from Melicope lunu-ankenda (Rutaceae). Molecules, 24(17), 3109. https://doi.org/10.3390/molecules24173109








