Bee Venom: Overview of Main Compounds and Bioactivities for Therapeutic Interests
Abstract
1. Generalities about Honeybees
2. Main Compounds of Bee Venom
2.1. Melittin
2.2. Apamin
2.3. Mast Cell Degranulating (MCD) Peptide
2.4. Adolapin
2.5. Phospholipase A2
2.6. Hyaluronidase
3. Bioactivities and Therapeutic Applications of Bee Venom and Its Major Compounds
3.1. Anti-Inflammatory Potential
3.2. BV Application for the Treatment of Neurodegenerative Diseases
3.2.1. Parkinson’s Disease
3.2.2. Alzheimer’s Disease
3.2.3. Amyotrophic Lateral Sclerosis
3.3. BV and/or Melittin Applications in Cancer
3.4. Antiviral and Antibacterial Properties
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Greenleaf, S.S.; Kremen, C. Wild bees enhance honey bees’ pollination of hybrid sunflower. Proc. Natl. Acad. Sci. USA 2006, 103, 13890–13895. [Google Scholar] [CrossRef] [PubMed]
- Fratellone, P.M. Apitherapy Products for Medicinal Use. J. Nutr. Food Sci. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.A.A.-S.M. Studies on bee venom and its medical uses. Int. J. Adv. Res. Technol. 2012, 1, 69–83. [Google Scholar]
- El-Wahab, S.D.A.; Eita, L.H. The effectiveness of live bee sting acupuncture on depression. J. Nurs. Health Sci. 2015, 4, 19–27. [Google Scholar]
- Trumbeckaite, S.; Dauksiene, J.; Bernatoniene, J.; Janulis, V. Knowledge, attitudes, and usage of apitherapy for disease prevention and treatment among undergraduate pharmacy students in Lithuania. Evid.-Based Complement. Altern. Med. 2015, 9. [Google Scholar] [CrossRef] [PubMed]
- Hellner, M.; von Georgi, D.W.R.; Münstedt, K. Apitherapy: Usage and experience in German beekeepers. Evid.-Based Complement. Altern. Med. 2007, 5, 475–479. [Google Scholar] [CrossRef]
- Lee, J.A.; Son, M.J.; Choi, J.; Yun, K.J.; Jun, J.H.; Lee, M.S. Bee venom acupuncture for rheumatoid arthritis: A systematic review protocol. Bmj Open 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Liu, Y.; Ye, Y.; Wang, X.R.; Lin, L.T.; Xiao, L.Y.; Zhou, P.; Shi, G.X.; Liu, C.Z. Bee venom therapy: Potential mechanisms and therapeutic applications. Toxicon 2018, 148, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Moreno, M.; Giralt, E. Three Valuable Peptides from Bee and Wasp Venoms for Therapeutic and Biotechnological Use: Melittin, Apamin and Mastoparan. Toxins 2015, 7, 1126–1150. [Google Scholar] [CrossRef]
- Bellik, Y. Bee Venom: Its potential use in alternative medicine. Anti-Infect. Agents 2015, 13, 3–16. [Google Scholar] [CrossRef]
- Jo, M.; Park, M.H.; Kollipara, P.S.; An, B.J.; Song, H.S.; Han, S.B.; Kim, J.H.; Song, M.J.; Hong, J.T. Anti-cancer effect of bee venom toxin and melittin in ovarian cancer cells through induction of death receptors and inhibition of JAK2/STAT3 pathway. Toxicol. Appl. Pharmacol. 2012, 258, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Rady, I.; Siddiqui, I.A.; Rady, M.; Mukhtar, H. Melittin, a major peptide component of bee venom, and its conjugates in cancer therapy. Cancer Lett. 2017, 402, 16–31. [Google Scholar] [CrossRef] [PubMed]
- Park, M.H.; Choi, M.S.; Kwak, D.H.; Oh, K.W.; Yoon, D.Y.; Han, S.B.; Song, H.S.; Song, M.J.; Hong, J.T. Anti-cancer effect of bee venom in prostate cancer cells through activation of caspase pathway via inactivation of NF-κB. Prostate 2011, 71, 801–812. [Google Scholar] [CrossRef] [PubMed]
- Hood, J.L.; Jallouk, A.P.; Campbell, N.; Ratner, L.; Wickline, S.A. Cytolytic nanoparticles attenuate HIV-1 infectivity. Antivir. Ther. 2013, 18, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Bilò, M.B.; Bonifazi, F. The natural history and epidemiology of insect venom allergy: Clinical implications. Clin. Exp. Allergy. 2009, 39, 1467–1476. [Google Scholar] [CrossRef] [PubMed]
- Golden, D.B.K. Insect sting anaphylaxis. Immunol. Allergy Clin. North. Am. 2007, 27, 261. [Google Scholar] [CrossRef] [PubMed]
- Mingomataj, E.Ç.; Bakiri, A.H. Episodic hemorrhage during honeybee venom anaphylaxis: Potential mechanisms. J. Investig. Allergol. Clin. Immunol. 2012, 22, 237–244. [Google Scholar] [PubMed]
- Kaplan, A.P. Kinins, airway obstruction, and anaphylaxis. Chem. Immunol. Allergy. 2010, 95, 67–84. [Google Scholar]
- Mingomataj, E.; Bakiri, A. The inhibition of kallikrein-bradykinin pathway may be useful in the reduction of allergic reactions during honeybee venom immunotherapy. J. Med. Hypotheses Ideas 2009, 3, 10. [Google Scholar]
- Ziai, M.R.; Russek, S.; Wang, H.C.; Beer, B.; Blume, A.J. Mast cell degranulating peptide: A multi-functional neurotoxin. J. Pharm. Pharmacol. 1990, 42, 457–461. [Google Scholar] [CrossRef]
- Chen, J.; Guan, S.M.; Sun, W.; Fu, H. Melittin, the Major Pain-Producing Substance of Bee Venom. Neurosci Bull. 2016, 32. [Google Scholar] [CrossRef]
- Raghuraman, H.; Chattopadhyay, A. Melittin: A membrane-active peptide with diverse functions. Biosci. Rep. 2007, 27, 189–223. [Google Scholar] [CrossRef]
- Pino-Angeles, A.; Lazaridis, T. Effects of peptide charge, orientation, and concentration on melittin transmembrane pores. Biophysj. J. 2018, 114, 2865–2874. [Google Scholar] [CrossRef]
- Lee, M.T.; Sun, T.L.; Hung, W.C.; Huang, H.W. Process of inducing pores in membranes by melittin. PNAS 2013, 110, 14243–14248. [Google Scholar] [CrossRef]
- Zarrinnahad, H.; Mahmoodzadeh, A.; Hamidi, M.P.; Mahdavi, M.; Moradi, A.; Bagheri, K.P.; Shahbazzadeh, D. Apoptotic effect of melittin purified from iranian honey bee venom on human cervical cancer heLa cell line. Intj. Pept Res. Ther. 2018, 24, 563–570. [Google Scholar] [CrossRef]
- Keith, D.J.; Eshleman, A.J.; Janowsky, A. Melittin stimulates fatty acid release through non-phospholipase-mediated mechanisms and interacts with the dopamine transporter and other membrane spanning proteins. Eur. J. Pharmacol. 2011, 650, 501–510. [Google Scholar] [CrossRef]
- Bae, G.; Bae, H. Anti-Inflammatory applications of melittin, a major component of bee venom: Detailed mechanism of action and adverse effects. Molecules 2016, 21, 616. [Google Scholar]
- Son, D.J.; Lee, J.W.; Lee, Y.H.; Song, H.S.; Lee, C.K.; Hong, J.T. Therapeutic application of anti-arthritis, pain-releasing, and anti-cancer effects of bee venom and its constituent compounds. Pharm. Ther. 2007, 115, 246–270. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kim, K.H.; Lee, W.; An, H.J.; Lee, S.J.; Han, S.M.; Lee, K.G.; Park, Y.Y.; Kim, K.S.; Lee, Y.S.; et al. Apamin inhibits PDGF-BB-induced vascular smooth muscle cell proliferation and migration through suppressions of activated Akt and Erk signaling pathway. Vasc. Pharmacol. 2015, 70. [Google Scholar] [CrossRef]
- Modzelewska, B.; Kostrzewska, A.; Sipowicz, M.; Kleszczewski, T.; Batra, S. Apamin inhibits NO-induced relaxation of the spontaneous contractile activity of the myometrium from non-pregnant women. Reprod. Biol. Endocrinol. 2003, 1, 8. [Google Scholar]
- Hanson, J.M.; Morley, J. Anti-inflammatory property of 401 (MCD-peptide), a peptide from the venom of the bee Apis mellifera (L.). Br. J. Pharmacol. 1974, 50, 383–392. [Google Scholar] [CrossRef]
- Banks, B.E.; Dempsey, C.E.; Vernon, C.A.; Warner, J.A.; Ymey, J. Anti-inflammatory activity of bee venom peptide 401 (mast cell degranulating peptide) and compound 48/80 results from mast cell degranulation in vivo. Br. J. Pharmacol. 1990, 99, 350–354. [Google Scholar] [CrossRef]
- Cherniack, E.P.; Govorushko, S. To bee or not to bee: The potential efficacy and safety of bee venom acupuncture in humans. Toxicon 2018, 154, 74–78. [Google Scholar] [CrossRef]
- Jung, S.Y.; Lee, K.W.; Choi, S.M.; Yang, E.J. Bee Venom Protects against Rotenone-Induced Cell Death in NSC34 Motor Neuron Cells. Toxins 2015, 7, 3715–3726. [Google Scholar] [CrossRef]
- Samel, M.; Vija, H.; Kurvet, I.; Künnis-Beres, K.; Trummal, K.; Subbi, J.; Kahru, A.; Siigur, J. Interactions of PLA2-s from Vipera lebetina, Vipera berus berus and Naja naja oxiana Venom with Platelets, Bacterial and Cancer Cells. Toxins 2013, 5, 203–223. [Google Scholar] [CrossRef]
- Leandro, L.F.; Mendes, C.A.; Casemiro, L.A.; Vinholis, A.H.; Cunha, W.R.; De Almeida, R.; Martins, C.H. Antimicrobial activity of apitoxin, melittin and phospholipase A₂ of honey bee (Apis mellifera) venom against oral pathogens. An. Acad. Bras. Cienc. 2015, 87, 147–155. [Google Scholar] [CrossRef]
- Frangieh, J.; Salma, Y.; Haddad, K.; Mattei, C.; Legros, C.; Fajloun, Z.; El Obeid, D. First Characterization of The Venom from Apis mellifera syriaca, A Honeybee from The Middle East Region. Toxins 2019, 11, 191. [Google Scholar] [CrossRef]
- Lu, Z.M.; Xie, F.; Fu, H.; Liu, M.G.; Cao, F.L.; Hao, J.; Chen, J. Roles of peripheral P2X and P2Y receptors in the development of melittin-induced nociception and hypersensitivity. Neurochem. Res. 2008, 33, 2085–2091. [Google Scholar] [CrossRef]
- Park, S.; Baek, H.; Jung, K.H.; Lee, G.; Lee, H.; Kang, G.H.; Lee, G.; Bae, H. Bee venom phospholipase A2 suppresses allergic airway inflammation in an ovalbumin-induced asthma model through the induction of regulatory T cells. Immun. Inflamm. Dis. 2015, 3, 386–397. [Google Scholar] [CrossRef]
- Ye, M.; Chung, H.S.; Lee, C.; Yoon, M.S.; Yu, A.R.; Kim, J.S.; Hwang, D.S.; Shim, I.; Bae, H. Neuroprotective effects of bee venomphospholipase A2 in the 3xTg AD mousemodel of Alzheimer’s disease. J. Neuroinflamm. 2016, 13, 10. [Google Scholar]
- Kim, K.H.; Lee, S.Y.; Shin, J.; Hwang, J.T.; Jeon, H.N.; Bae, H. Dose-Dependent Neuroprotective Effect of Standardized Bee Venom Phospholipase A2 Against MPTP-Induced Parkinson’s Disease in Mice. Front. Aging Neurosci. 2019, 11. [Google Scholar] [CrossRef]
- Chung, E.S.; Lee, G.; Lee, C.; Ye, M.; Chung, H.S.; Kim, H.; Bae, S.J.; Hwang, D.S.; Bae, H. Bee venom phospholipase A2, a novel Foxp3+ regulatory T cell inducer, protects dopaminergic neurons by modulating neuroinflammatory responses in a mouse model of Parkinson’s Disease. J. Immunol. 2015, 195, 4853–4860. [Google Scholar] [CrossRef]
- Topchiyeva, T.; Mammadova, F.Z. The seasonal activity of hyaluronidase in venom of a honey bee (Apis mellifera L. caucasica) in various regions of Azerbaijan. J. Entomol. Zool. Stud. 2016, 4, 1388–1391. [Google Scholar]
- Hossen, M.S.; Shapla, U.M.; Gan, S.H.; Khalil, M.I. Impact of Bee Venom Enzymes on Diseases and Immune Responses. Molecules 2017, 22, 25. [Google Scholar] [CrossRef]
- Glass, C.K.; Saijo, K.; Winner, B.; Marchetto, M.C.; Gage, F.H. Mechanisms Underlying Inflammation in Neurodegeneration. Cell 2010, 140, 918–934. [Google Scholar] [CrossRef]
- Khalil, W.S.; Khalil, E.A.G. Immune response modifying effects of bee venom protein [Melittin]/Autoclaved, L. donovani complex in CD1 Mice: The search for new vaccine adjuvants. J. Vaccines Vaccin. 2017, 8, 2. [Google Scholar]
- Park, H.J.; Son, D.J.; Oh, K.W.; Kim, K.H.; Song, H.S.; Kim, G.J.; Oh, G.T.; Yoon, D.Y.; Hong, J.T. Inhibition of inflammation mediator generation by suppression of NF-kB through interaction with the p50 subunit. Arthritis Rheum. 2004, 50, 504–3515. [Google Scholar] [CrossRef]
- Kim, W.H.; An, H.J.; Kim, J.Y.; Gwon, M.G.; Gu, H.; Jeon, M.; Kim, M.K.; Han, S.M.; Park, K.K. Anti-Inflammatory Effect of Melittin on Porphyromonas Gingivalis LPS-Stimulated Human Keratinocytes. Molecules 2018, 23, 332. [Google Scholar] [CrossRef]
- Ong, P.Y.; Leung, D.Y.M. Bacterial and viral infections in atopic dermatitis: A comprehensive review. Clin. Rev. Allergy Immunol. 2016, 51, 329–337. [Google Scholar] [CrossRef]
- Kim, Y.; Lee, Y.W.; Kim, H.; Chung, D.K. Bee venom alleviates atopic dermatitis symptoms through the upregulation of decay-accelerating factor (DAF/CD55). Toxins 2019, 11, 239. [Google Scholar] [CrossRef]
- Shin, D.; Choi, W.; Bae, H. Bee venom Phospholipase A2 alleviate house dust mite-induced atopic dermatitis-like skin lesions by the CD206 mannose receptor. Toxins 2018, 10, 146. [Google Scholar] [CrossRef]
- Goldman, J.G.; Williams-Gray, C.; Barker, R.A.; Duda, J.E.; Galvin, J.E. The spectrum of cognitive impairment in Lewy body diseases. Mov. Disord. 2014, 29, 217–231. [Google Scholar] [CrossRef]
- Aarsland, D.; Creese, B.; Politis, M.; Chaudhuri, K.R.; Ffytche, D.H.; Weintraub, D.; Ballard, C. Cognitive decline in Parkinson disease. Nat. Rev. Neurol. 2017, 13, 217–231. [Google Scholar] [CrossRef]
- Iakovakis, D.; Hadjidimitriou, S.; Charisis, V.; Bostantzopoulou, S.; Katsarou, Z.; Hadjileontiadis, L.J. Touchscreen typing-pattern analysis for detecting fine motor skills decline in early-stage Parkinson’s disease. Sci. Rep. 2018. [Google Scholar] [CrossRef]
- Tanner, C.M.; Kamel, F.; Ross, G.W.; Hoppin, J.A.; Goldman, S.M.; Korell, M.; Marras, C.; Bhudhikanok, G.S.; Kasten, M.; Chade, A.R.; et al. Rotenone, paraquat, and Parkinson’s disease. Environ. Health Perspect. 2011, 119, 866–872. [Google Scholar] [CrossRef]
- Khalil, W.K.B.; Assaf, N.; ElShebiney, S.A.; Salem, N.A. Neuroprotective effects of bee venom acupuncture therapy against rotenone-induced oxidative stress and apoptosis. Neurochem. Int. 2015, 80, 79–86. [Google Scholar] [CrossRef]
- Alvarez-Fischer, D.; Noelker, C.; Vulinović, F.; Grünewald, A.; Chevarin, C.; Klein, C.; Oertel, W.H.; Hirsch, E.C.; Michel, P.P.; Hartmann, A. Bee Venom and Its Component Apamin as Neuroprotective Agents in a Parkinson Disease Mouse Model. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Aksoz, E.; Gocmez, S.; Sahin, T.D.; Aksit, D.; Aksit, H.; Utkan, T. The protective effect of metformin in scopolamine-induced learning and memory impairment in rats. Pharm. Rep. 2019, 71, 818–825. [Google Scholar] [CrossRef]
- Sabri, O.; Sabbagh, M.N.; Seibyl, J.; Barthel, H.; Akatsu, H.; Ouchi, Y.; Senda, K.; Murayama, S.; Ishii, K.; Takao, M.; et al. Florbetaben PET imaging to detect amyloid beta plaques in Alzheimer’s disease: Phase 3 study. Alzheimers Dement. 2015, 11, 964–974. [Google Scholar] [CrossRef]
- Baek, H.; Lee, C.; Choi, D.B.; Kim, N.S.; Kim, Y.S.; Ye, Y.J.; Kim, Y.S.; Kim, J.S.; Shim, I.; Bae, H. Bee venom phospholipase A2 ameliorates Alzheimer’s disease pathology in Aβ vaccination treatment without inducing neuro-inflammation in a 3xTg-AD mouse model. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef]
- Kakuda, N.; Miyasaka, T.; Iwasaki, N.; Nirasawa, T.; Wada-Kakuda, S.; Takahashi-Fujigasaki, J.; Murayama, S.; Ihara, Y.; Ikegawa, M. Distinct deposition of amyloid-β species in brains with Alzheimer’s disease pathology visualized with MALDI imaging mass spectrometry. Acta Neuropathol. Commun. 2017, 5. [Google Scholar] [CrossRef]
- Van Eldik, L.J.; Carrillo, M.C.; Cole, P.E.; Feuerbach, D.; Greenberg, B.; Hendrix, J.A.; Kennedy, M.; Kozauer, N.; Margolin, R.A.; Molinuevo, J.L.; et al. The roles of inflammation and immune mechanisms in Alzheimer’s disease. Alzheimers Dement. 2016, 2, 99–109. [Google Scholar] [CrossRef]
- Kinney, J.W.; Bemiller, S.M.; Murtishaw, A.S.; Leisgang, A.M.; Salazar, A.M.; Lamb, B.T. Inflammation as a central mechanism in Alzheimer’s disease. Alzheimers Dement. 2018, 4, 575–590. [Google Scholar] [CrossRef]
- Terry, A.V.; Buccafusco, J.J. The Cholinergic Hypothesis of Age and Alzheimer’s Disease-Related Cognitive Deficits: Recent Challenges and TheirImplications for Novel Drug Development. J. Pharm. Exp. Ther. 2003, 306, 821–827. [Google Scholar] [CrossRef]
- Rajagopalan, V.; Pioro, E.P. Unbiased MRI analyses identify micropathologic differences between upper motor neuron-predominant ALS phenotypes. Front. Neurosci. 2019, 13. [Google Scholar] [CrossRef]
- Boillée, S.; Yamanaka, K.; Lobsiger, C.S.; Copeland, N.G.; Jenkins, N.A.; Kassiotis, G.; Kollias, G.; Cleveland, D.W. Onset and progression in inherited ALS determined by motor neurons and microglia. Science 2006, 312, 1389–1392. [Google Scholar] [CrossRef]
- Jaarsma, D.; Haasdijk, E.D.; Grashorn, J.A.C.; Hawkins, R.; Van Duijn, W.; Verspaget, W.H.; London, J.; Holstege, J.C. Human Cu/Zn Superoxide Dismutase (SOD1) Overexpression in Mice Causes Mitochondrial Vacuolization, Axonal Degeneration, and Premature Motoneuron Death and Accelerates MotoneuronDisease in Mice Expressing a Familial AmyotrophicLateral Sclerosis Mutant SOD1. Neurobiol. Dis. 2000, 7, 623–643. [Google Scholar] [CrossRef]
- Yang, E.J.; Jiang, J.H.; Lee, S.M.; Yang, S.C.; Hwang, H.S.; Lee, M.S.; Choi, S.M. Bee venom attenuates neuroinflammatory events and extends survival in amyotrophic lateral sclerosis models. J. Neuroinflamm. 2010, 7. [Google Scholar] [CrossRef]
- Cai, M.D.; Choi, S.M.; Yang, E.J. The effects of bee venom acupuncture on the central nervous system and muscle in an animal hSOD1G93A mutant. Toxins 2015, 7, 846–858. [Google Scholar] [CrossRef]
- Jung, G.B.; Huh, J.-E.; Lee, H.J.; Kim, D.; Lee, G.J.; Park, H.K.; Lee, J.D. Anti-cancer effect of bee venom on human MDA-MB-231 breast cancer cells using Raman spectroscopy. Biomed. Opt. Express 2018, 9, 5703–5718. [Google Scholar] [CrossRef]
- Lim, H.N.; Baek, S.B.; Jung, H.J. Bee venom and its peptide component melittin suppress growth and migration of Melanoma Cells via inhibition of PI3K/AKT/mTOR and MAPK pathways. Molecules 2019, 24, 929. [Google Scholar] [CrossRef]
- Hong, J.; Lu, X.; Deng, Z.; Xiao, S.; Yuan, B.; Yang, X. How Melittin inserts into cell membrane: Conformational changes, inter-peptide cooperation, and disturbance on the membrane. Molecules 2019, 24, 1775. [Google Scholar] [CrossRef]
- Liu, S.; Yu, M.; He, Y.; Xiao, L.; Wang, F.; Song, C.; Sun, S.; Ling, C.; Xu, Z. Melittin prevents liver cancer cell metastasis through inhibition of the Rac1-dependent pathway. Hepatology 2008, 47, 1964–1973. [Google Scholar] [CrossRef]
- Saidemberg, D.M.; Baptista-Saidemberg, N.B.; Palma, M.S. Chemometric analysis of Hymenoptera toxins and defensins: A model for predicting the biological activity of novel peptides from venoms and hemolymph. Peptides 2011, 32, 1924–1933. [Google Scholar] [CrossRef]
- Liu, X.; Chen, D.; Xie, L.; Zhang, R. Effect of honey bee venom on proliferation of K1735M2 mouse melanoma cells in-vitro and growth of murine B16 melanomas in-vivo. J. Pharm Pharmacol. 2002, 54, 1083–1089. [Google Scholar] [CrossRef]
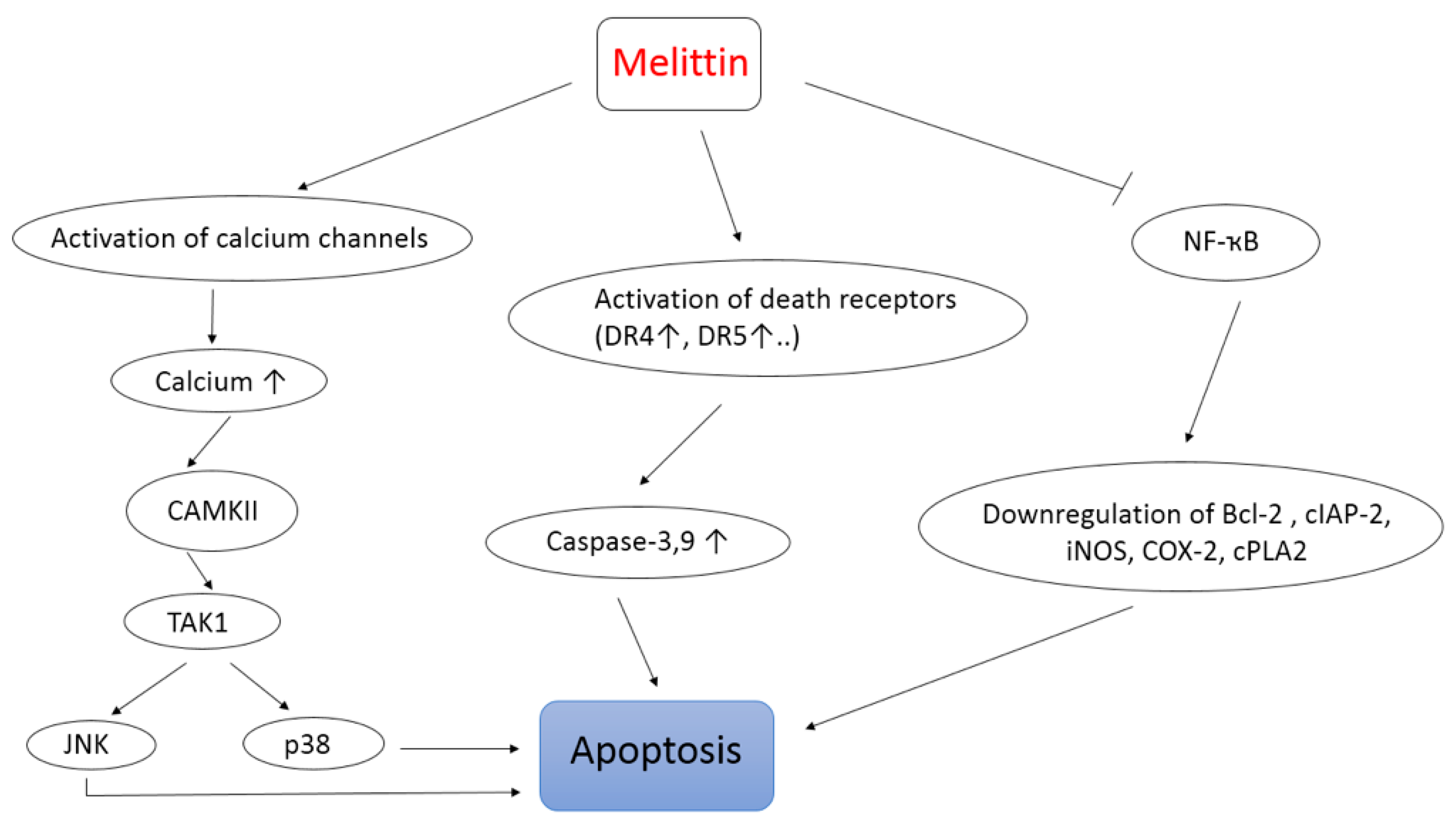
- Wang, C.; Chen, T.; Zhang, N.; Yang, M.; Li, B.; Lü, X.; Cao, X.; Ling, C. Melittin, a Major Component of Bee Venom, Sensitizes Human Hepatocellular Carcinoma Cells to Tumor Necrosis Factor-related Apoptosis-inducing Ligand (TRAIL)-induced. Apoptosis by Activating CaMKII-TAK1-JNK/p38 and Inhibiting IkB alpha Kinase-NFkB. J. Biol Chem. 2009, 284, 3804–3813. [Google Scholar] [CrossRef]
- Zheng, J.; Lee, H.L.; Ham, Y.W.; Song, H.S.; Song, M.J.; Hong, J.T. Anti-cancer effect of bee venom on colon cancer cell growth by activation of death receptors and inhibition of nuclear factor kappa B. Oncotarget 2015, 6, 44437–44451. [Google Scholar] [CrossRef]
- Zuazo-Gaztelu, I.; Casanovas, O. Unraveling the role of angiogenesis in cancer ecosystems. Front. Oncol. 2018, 8. [Google Scholar] [CrossRef]
- Rajabi, M.; Mousa, S.A. The role of angiogenesis in cancer treatment. Biomedicines 2017, 5, 34. [Google Scholar] [CrossRef]
- Gülmez, Y.; Aydın, A.; Can, I.; Tekin, S.; Cacan, E. Cellular toxicity and biological activities of honey bee (Apis mellifera L.) venom. Marmara Pharm. J. 2017, 21, 51–260. [Google Scholar]
- Soman, N.R.; Baldwin, S.L.; Hu, G.; Marsh, J.N.; Lanza, G.M.; Heuser, J.E.; Arbeit, J.M.; Wickline, S.A.; Schlesinger, P.H. Molecularly targeted nanocarriers deliver the cytolytic peptide melittin specifically to tumor cells in mice, reducing tumor growth. J. Clin. Investig. 2009, 119, 2830–2842. [Google Scholar] [CrossRef]
- Cheng, B.; Thapa, B.; Remant, K.C.; Xu, P. Dual secured nano-melittin for the safe and effective eradication of cancer cells. J. Mater. Chemb. 2015, 3, 25–29. [Google Scholar] [CrossRef]
- Jallouk, A.P.; Moley, K.H.; Omurtag, K.; Hu, G.; Lanza, G.M.; Wickline, S.A.; Hood, J.L. Nanoparticle Incorporation of Melittin Reduces Sperm and Vaginal Epithelium Cytotoxicity. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Hegazi, A.; Abdou, A.M.; EI-Moez, S.I.; Abd Allah, F. Evaluation of the antibacterial activity of bee venom from different sources. World Appl. Sci. J. 2014, 30, 266–270. [Google Scholar]
- Socarras, K.M.T.; Theophilus, P.A.S.; Torres, J.P.; Gupta, K.; Sapi, E. Antimicrobial Activity of Bee Venom and Melittin against Borrelia burgdorferi. Antibiot. (Basel) 2017, 6. [Google Scholar] [CrossRef]
- Perumal Samy, R.; Gopalakrishnakone, P.; Thwin, M.M.; Chow, T.K.; Bow, H.; Yap, E.H.; Thong, T.W. Antibacterial activity of snake, scorpion and bee venoms: A comparison with purified venom phospholipase A2 enzymes. J. Appl Microbiol. 2007, 102, 650–659. [Google Scholar] [CrossRef]
- Zolfagharian, H.; Babaie, M. Antimicrobial activity of bee venom and Melittin against Borrelia burgdorferi. J. Pharmacopuncture. 2016, 19, 225–230. [Google Scholar] [CrossRef]
- Uddin, M.B.; Lee, B.H.; Nikapitiya, C.; Kim, J.H.; Kim, T.H.; Lee, H.C.; Kim, C.G.; Lee, J.S.; Kim, C.J. Inhibitory effects of bee venom and its components against viruses in vitro and in vivo. J. Microbiol. 2016, 54, 853–866. [Google Scholar] [CrossRef]
- Bachis, A.; Cruz, M.I.; Mocchetti, I. M-tropic HIV envelope protein gp120 exhibits a different neuropathological profile than T-tropic gp120 in rat striatum. Eur. J. Neurosci. 2010, 32. [Google Scholar] [CrossRef]
- Cecilio, A.B.; Caldas, S.; Oliveira, R.A.; Santos, A.S.; Richardson, M.; Naumann, G.B.; Schneider, F.S.; Alvarenga, V.G.; Estevão-Costa, M.I.; Fuly, A.L.; et al. Molecular characterization of Lys49 and Asp49 phospholipases A₂from snake venom and their antiviral activities against Dengue virus. Toxins 2013, 5, 1780–1798. [Google Scholar] [CrossRef]
- Fenard, D.; Lambeau, G.; Valentin, E.; Lefebvre, J.C.; Lazdunski, M.; Doglio, A. Secreted phospholipases A2, a new class of HIV inhibitors that block virus entry into host cells. J. Clin. Investig. 1999, 104, 611–618. [Google Scholar] [CrossRef]
- Fenard, D.; Lambeau, G.; Maurin, T.; Lefebvre, J.C.; Doglio, A. A peptide derived from bee venom-secreted phospholipase A2 inhibits replication of T-cell tropic HIV-1 strains via interaction with the CXCR4 chemokine receptor. Mol. Pharmacol. 2001, 60, 341–347. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wehbe, R.; Frangieh, J.; Rima, M.; El Obeid, D.; Sabatier, J.-M.; Fajloun, Z. Bee Venom: Overview of Main Compounds and Bioactivities for Therapeutic Interests. Molecules 2019, 24, 2997. https://doi.org/10.3390/molecules24162997
Wehbe R, Frangieh J, Rima M, El Obeid D, Sabatier J-M, Fajloun Z. Bee Venom: Overview of Main Compounds and Bioactivities for Therapeutic Interests. Molecules. 2019; 24(16):2997. https://doi.org/10.3390/molecules24162997
Chicago/Turabian StyleWehbe, Rim, Jacinthe Frangieh, Mohamad Rima, Dany El Obeid, Jean-Marc Sabatier, and Ziad Fajloun. 2019. "Bee Venom: Overview of Main Compounds and Bioactivities for Therapeutic Interests" Molecules 24, no. 16: 2997. https://doi.org/10.3390/molecules24162997
APA StyleWehbe, R., Frangieh, J., Rima, M., El Obeid, D., Sabatier, J.-M., & Fajloun, Z. (2019). Bee Venom: Overview of Main Compounds and Bioactivities for Therapeutic Interests. Molecules, 24(16), 2997. https://doi.org/10.3390/molecules24162997






