Cannabidiol Treatment Might Promote Resilience to Cocaine and Methamphetamine Use Disorders: A Review of Possible Mechanisms
Abstract
1. Introduction
2. Studies About the Influence of CBD on the Effects of Psychostimulant Drugs
2.1. Preclinical Studies
2.1.1. Pharmacokinetic Interactions and Cocaine-Induced Toxicity
2.1.2. Motor Activity and Behavioural Sensitisation
2.1.3. Intracranial Self-Stimulation (ICSS) Paradigm
2.1.4. Self-Administration Paradigm
2.1.5. Conditioned Place Preference (CPP) Paradigm
2.2. Human Studies
2.2.1. Observational Studies
2.2.2. Clinical Trials
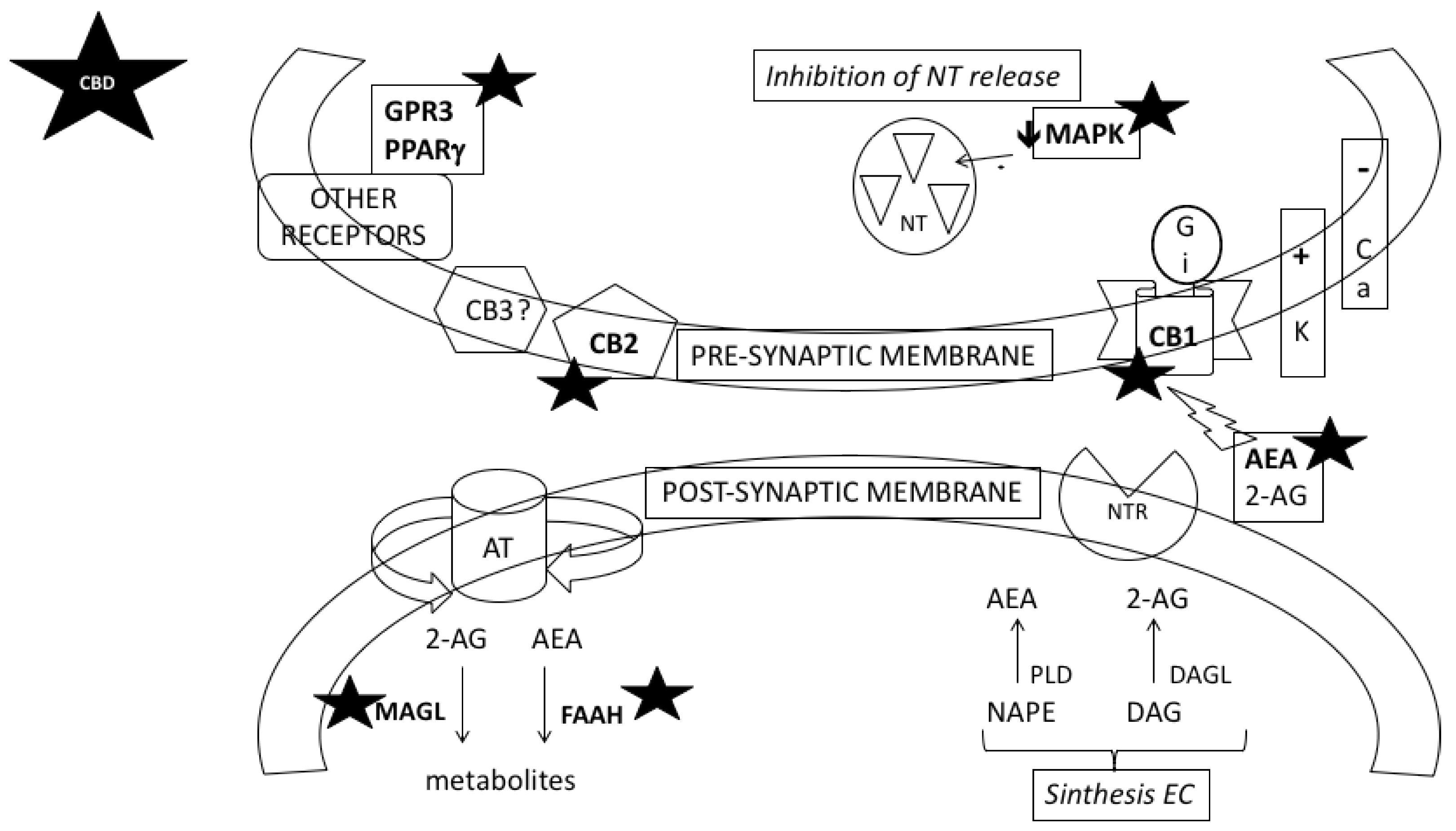
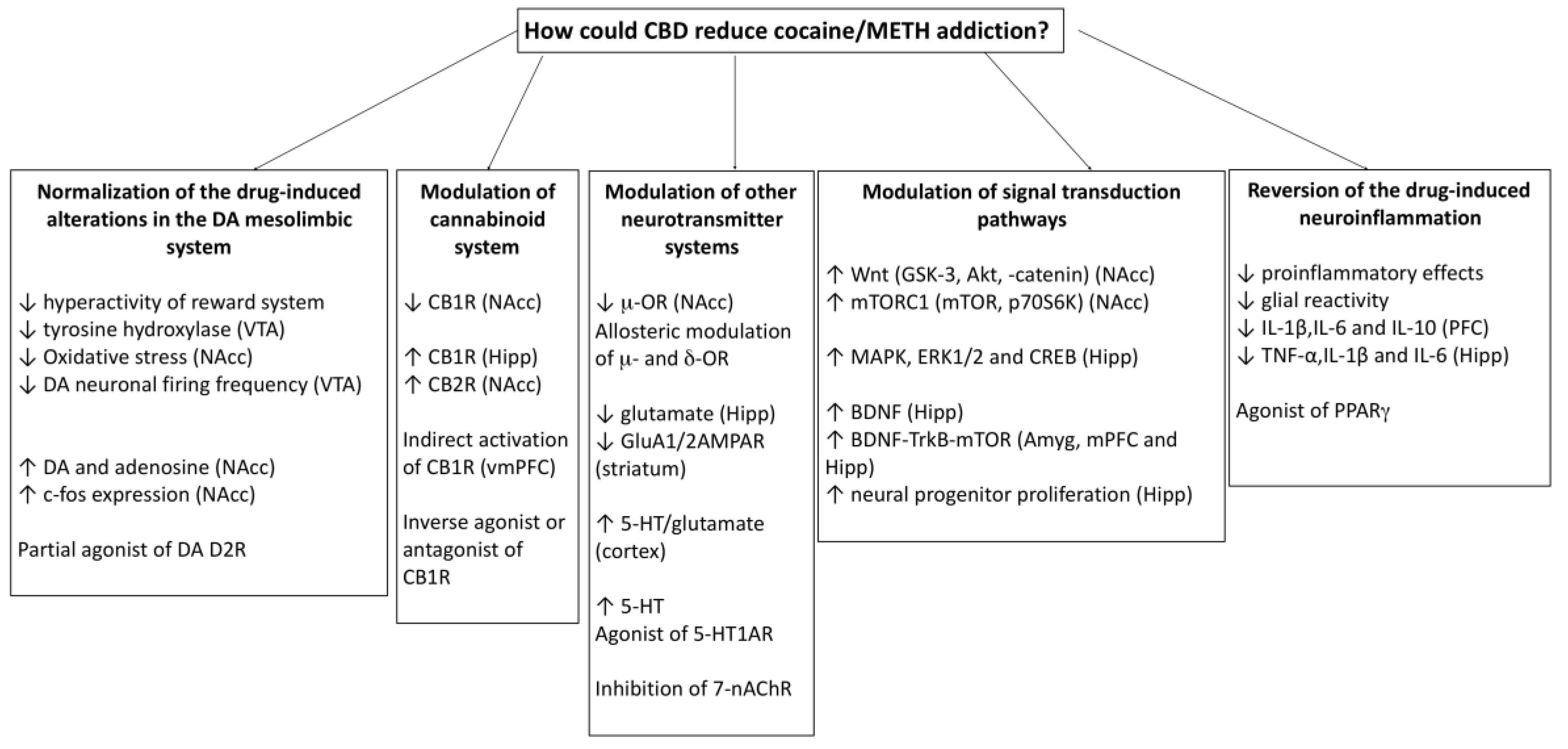
3. Possible Mechanisms Involved in the Protective Effects of CBD on Addiction to Psychostimulant Drugs
3.1. CBD Could Prevent Neuroadaptations Induced by Psychostimulant Drugs
3.2. CBD Could Reverse the Behavioural Effects of Psychostimulant Drugs through the Action on Different Neurotransmitter Systems and Intracellular Signalling Pathways
3.3. CBD could Reverse the Alterations in the Immune System and the Neuroinflammation Induced by Psychostimulant Drugs
3.4. CBD could Erase the Aberrant Drug-Related Memories Affecting the Reconsolidation Process
3.5. CBD could Reverse the Cognitive Deficits Induced by Psychostimulant Drugs
3.6. CBD could Alleviate the Mental Disorders Comorbid with Psychostimulants Abuse
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- European Monitoring Centre for Drugs and Drug Addiction, European Drug Report 2018: Trends and Developments, Publications Office of the European Union, Luxembourg. Available online: http://www.emcdda.europa.eu/system/files/publications/8585/20181816_TDAT18001ENN_PDF.pdf (accessed on 5 April 2019).
- World Drug Report 2018 (United Nations publication, Sales No. E.18.XI.9). Available online: https://www.unodc.org/wdr2018/prelaunch/WDR18_Booklet_4_YOUTH.pdf (accessed on 5 April 2019).
- Cao, D.N.; Shi, J.J.; Hao, W.; Wu, N.; Li, J. Advances and challenges in pharmacotherapeutics for amphetamine-type stimulants addiction. Eur. J. Pharmacol. 2016, 780, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Morley, K.C.; Cornish, J.L.; Faingold, A.; Wood, K.; Haber, P.S. Pharmacotherapeutic agents in the treatment of methamphetamine dependence. Expert Opin. Investig. Drugs 2017, 26, 563–578. [Google Scholar] [CrossRef] [PubMed]
- Buchholz, J.; Saxon, A.J. Medications to treat cocaine use disorders: Current options. Curr. Opin. Psychiatry 2019, 32, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Mechoulam, R.; Hanus, L. Cannabidiol: An overview of some chemical and pharmacological aspects. Part I: Chemical aspects. Chem. Phys. Lipids 2002, 121, 35–43. [Google Scholar] [CrossRef]
- Mechoulam, R.; Parker, L.A.; Gallily, R. Cannabidiol: An overview of some pharmacological aspects. J. Clin. Pharmacol. 2002, 42, 11S–19S. [Google Scholar] [CrossRef] [PubMed]
- Pertwee, R.G. Cannabinoid pharmacology: The first 66 years. Br. J. Pharmacol. 2006, 147, S163–S171. [Google Scholar] [CrossRef] [PubMed]
- Pertwee, R.G. The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids: delta9-tetrahydrocannabinol, cannabidiol and delta9-tetrahydrocannabivarin. Br. J. Pharmacol. 2008, 153, 199–215. [Google Scholar] [CrossRef] [PubMed]
- Izzo, A.; Borrelli, F.; Capasso, R.; Di Marzo, V.; Mechoulam, R. Non-psychotropic plant cannabinoids: New therapeutic opportunities from an ancient herb. Trends Pharmacol. Sci. 2009, 30, 515–527. [Google Scholar] [CrossRef] [PubMed]
- Thomas, A.; Baillie, G.L.; Phillips, A.M.; Razdan, R.K.; Ross, R.A.; Pertwee, R.G. Cannabidiol displays unexpectedly high potency as an antagonist of CB1 and CB2 receptor agonists in vitro. Br. J. Pharmacol. 2007, 150, 613–623. [Google Scholar] [CrossRef] [PubMed]
- Laprairie, R.B.; Bagher, A.M.; Kelly, M.E.M.; Denovan-Wright, E.M. Cannabidiol is a negative allosteric modulator of the cannabinoid CB 1 receptor. Br. J. Pharmacol. 2015, 172, 4790–4805. [Google Scholar] [CrossRef]
- Tham, M.; Yilmaz, O.; Alaverdashvili, M.; Kelly, M.E.M.; Denovan-wright, E.M.; Laprairie, R.B. Allosteric and orthosteric pharmacology of cannabidiol and cannabidiol-dimethylheptyl at the type 1 and type 2 cannabinoid receptors. Br. J. Pharmacol. 2019, 176, 1455–1469. [Google Scholar] [CrossRef] [PubMed]
- Campos, A.C.; Ortega, Z.; Palazuelos, J.; Fogaça, M.V.; Aguiar, D.C. The anxiolytic effect of cannabidiol on chronically stressed mice depends on hippocampal neurogenesis: Involvement of the endocannabinoid system. Int. J. Neuropsychopharmacol. 2013, 16, 1407–1419. [Google Scholar] [CrossRef] [PubMed]
- Leweke, F.M.; Piomelli, D.; Pahlisch, F.; Muhl, D.; Gerth, C.W.; Hoyer, C.; Koethe, D. Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia. Transl. Psychiatry 2012, 2, e94. [Google Scholar] [CrossRef] [PubMed]
- Zlebnik, N.E.; Cheer, J.F. Beyond the CB1 receptor: Is cannabidiol the answer for disorders of motivation? Annu. Rev. Neurosci. 2016, 39, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Straiker, A.; Dvorakova, M.; Zimmowitch, A.; Mackie, K. Cannabidiol inhibits endocannabinoid signaling in autaptic hippocampal neurons. Mol. Pharmacol. 2018, 94, 743–748. [Google Scholar] [CrossRef]
- Bergamaschi, M.M.; Queiroz, R.H.C.; Zuardi, A.W.; Crippa, J.A.S. Safety and side effects of cannabidiol, a Cannabis sativa constituent. Curr. Drug Saf. 2011, 6, 237–249. [Google Scholar] [CrossRef]
- Pisanti, S.; Malfitano, A.M.; Ciaglia, E.; Lamberti, A.; Ranieri, R.; Cuomo, G.; Abate, M.; Faggiana, G.; Proto, M.C.; Fiore, D.; et al. Cannabidiol: State of the art and new challenges for therapeutic applications. Pharmacol. Ther. 2017, 175, 133–150. [Google Scholar] [CrossRef]
- Russo, E.B.; Burnett, A.; Hall, B.; Parker, K.K. Agonistic properties of cannabidiol at 5-HT1a receptors. Neurochem. Res. 2005, 30, 1037–1043. [Google Scholar] [CrossRef]
- Campos, A.C.; Guimarães, F.S. Involvement of 5HT1A receptors in the anxiolytic-like effects of cannabidiol injected into the dorsolateral periaqueductal gray of rats. Psychopharmacology 2008, 199, 223. [Google Scholar] [CrossRef]
- Yang, K.H.; Galadari, S.; Isaev, D.; Petroianu, G.; Shippenberg, T.S.; Oz, M. The nonpsychoactive cannabinoid cannabidiol inhibits 5-hydroxytryptamine3A receptor-mediated currents in Xenopus laevisoocytes. J. Pharmacol. Exp. Ther. 2010, 333, 547–554. [Google Scholar] [CrossRef]
- Al Kury, L.T.; Mahgoub, M.; Howarth, F.C.; Oz, M. Natural Negative Allosteric Modulators of 5-HT₃ Receptors. Molecules 2018, 23, 3186. [Google Scholar] [CrossRef] [PubMed]
- Mahgoub, M.; Keun-Hang, S.Y.; Sydorenko, V.; Ashoor, A.; Kabbani, N.; Al Kury, L. Effects of cannabidiol on the function of alpha7-nicotinic acetylcholine receptors. Eur. J. Pharmacol. 2013, 720, 310–319. [Google Scholar] [CrossRef] [PubMed]
- Kathmann, M.; Flau, K.; Redmer, A.; Tränkle, C.; Schlicker, E. Cannabidiol is an allosteric modulator at mu- and delta-opioid receptors. Arch. Pharmacol. 2006, 372, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Seeman, P. Cannabidiol is a partial agonist at dopamine D2High receptors, predicting its antipsychotic clinical dose. Transl. Psychiatry 2016, 6, e920. [Google Scholar] [CrossRef]
- Bisogno, T.; Hanuš, L.; De Petrocellis, L.; Tchilibon, S.; Ponde, D.E.; Brandi, I.; Moriello, A.S.; Davis, J.B.; Mechoulam, R.; Di Marzo, V. Molecular targets for cannabidiol and its synthetic analogues: Effect on vanilloid VR1 receptors and on the cellular uptake and enzymatic hydrolysis of anandamide. Br. J. Pharmacol. 2011, 134, 845–852. [Google Scholar] [CrossRef] [PubMed]
- Qin, N.; Neeper, M.P.; Liu, Y.; Hutchinson, T.L.; Lubin, M.L.; Flores, C.M. TRPV2 Is Activated by Cannabidiol and Mediates CGRP Release in Cultured Rat Dorsal Root Ganglion Neurons. J. Neurosci. 2008, 28, 6231–6238. [Google Scholar] [CrossRef] [PubMed]
- Muller, C.; Morales, P.; Reggio, P.H. Cannabinoid Ligands Targeting TRP Channels. Front. Mol. Neurosci. 2019, 11, 487. [Google Scholar] [CrossRef] [PubMed]
- Ahrens, J.; Demir, R.; Leuwer, M.; de la Roche, J.; Krampfl, K.; Foadi, N.; Karst, M.; Haeseler, G. The nonpsychotropic cannabinoid cannabidiol modulates and directly activates alpha-1 and alpha-1-Beta glycine receptor function. Pharmacology 2009, 83, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Ryberg, E.; Sims, N.A.; Ridge, S.A.; Mackie, K.; Greasley, P.J.; Rogers, M.J. The putative cannabinoid receptor GPR55 affects osteoclast function in vitro and bone mass in vivo. Proc. Natl. Acad. Sci. USA 2009, 106, 16511–16516. [Google Scholar] [CrossRef]
- Laun, A.S.; Song, Z.H. GPR3 and GPR6, novel molecular targets for cannabidiol. Biochem. Biophys. Res. Commun. 2017, 490, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Laun, A.S.; Shrader, S.H.; Brown, K.J.; Song, Z.H. GPR3, GPR6, and GPR12 as novel molecular targets: Their biological functions and interaction with cannabidiol. Acta Pharmacol. Sin. 2019, 40, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Iffland, K.; Grotenhermen, F. An update on safety and side effects of cannabidiol: A review of clinical data and relevant animal studies. Cannabis Cannabinoid Res. 2017, 2, 139–154. [Google Scholar] [CrossRef] [PubMed]
- Taylor, L.; Gidal, B.; Blakey, G.; Tayo, B.; Morrison, G. A phase I, randomized, double-blind, placebo-controlled, single ascending dose, multiple dose, and food effect trial of the safety, tolerability and pharmacokinetics of highly purified cannabidiol in healthy subjects. CNS Drugs 2018, 32, 1053–1067. [Google Scholar] [CrossRef] [PubMed]
- Katsidoni, V.; Anagnostou, I.; Panagis, G. Cannabidiol inhibits the reward-facilitating effect of morphine: Involvement of 5-HT1A receptors in the dorsal raphe nucleus. Addict. Biol. 2013, 2, 286–296. [Google Scholar] [CrossRef] [PubMed]
- Babalonis, S.; Haney, M.; Malcolm, R.J.; Lofwall, M.R.; Votaw, V.R.; Sparenborg, S. Oral cannabidiol does not produce a signal for abuse liability in frequent marijuana smokers. Drug Alcohol Depend. 2017, 172, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Parker, L.A.; Burton, P.; Sorge, R.E.; Yakiwchuk, C.; Mechoulam, R. Effect of low doses of delta9-tetrahydrocannabinol and cannabidiol on the extinction of cocaine-induced and amphetamine-induced conditioned place preference learning in rats. Psychopharmacology 2004, 175, 360–366. [Google Scholar] [CrossRef]
- Viudez-Martínez, A.; García-Gutiérrez, M.S.; Medrano-Relinque, J.; Navarrón, C.M.; Navarrete, F.; Manzanares, J. Cannabidiol does not display drug abuse potential in mice behavior. Acta Pharmacol. Sin. 2019, 40, 358–364. [Google Scholar] [CrossRef]
- Corroon, J.; Phillips, J.A. A Cross-Sectional Study of Cannabidiol Users. Cannabis Cannabinoid Res. 2018, 3, 152–161. [Google Scholar] [CrossRef]
- Crippa, J.A.; Guimarães, F.S.; Campos, A.C.; Zuardi, A.W. Translational Investigation of the therapeutic potential of cannabidiol (CBD): Toward a new age. Front. Immunol. 2018, 9, 2009. [Google Scholar] [CrossRef]
- Prud’homme, M.; Cata, R.; Jutras-Aswad, D. Cannabidiol as an Intervention for Addictive Behaviors: A Systematic Review of the Evidence. Subst. Abuse 2015, 9, 33–38. [Google Scholar] [CrossRef]
- dos Santos, R.G.; Hallak, J.E.C.; Zuardi, A.W.; de Souza Crippa, J.A. Cannabidiol for the treatment of drug use disorders. In Handbook of Cannabis and Related Pathologies; Preedy, V.P., Ed.; Elsevier: London, UK, 2017; pp. 939–946. [Google Scholar]
- Hurd, Y.L. Cannabidiol: Swinging the marijuana pendulum from ‘weed’ to medication to treat the opioid epidemic. Trends Neurosci. 2017, 40, 124–127. [Google Scholar] [CrossRef] [PubMed]
- Chye, Y.; Christensen, E.; Solowij, N.; Yücel, M. The Endocannabinoid System and Cannabidiol’s Promise for the Treatment of Substance Use Disorder. Front. Psychiatry 2019, 10, 63. [Google Scholar] [CrossRef] [PubMed]
- Turna, J.; Syan, S.K.; Frey, B.N.; Rush, B.; Costello, M.J.; Weiss, M.; MacKillop, J. Cannabidiol as a Novel Candidate Alcohol Use Disorder Pharmacotherapy: A Systematic Review. Alcohol. Clin. Exp. Res. 2019, 43, 550–563. [Google Scholar] [CrossRef] [PubMed]
- Pellinen, P.; Honkakoski, P.; Stenbäck, F.; Niemitz, M.; Alhava, E.; Pelkonen, O.; Lang, M.A.; Pasanen, M. Cocaine N-demethylation and the metabolism-related hepatotoxicity can be prevented by cytochrome P450 3A inhibitors. Eur. J. Pharmacol. 1994, 270, 35–43. [Google Scholar] [CrossRef]
- Bornheim, L.M. Effect of cytochrome P450 inducers on cocaine-mediated hepatotoxicity. Toxicol. Appl. Pharmacol. 1998, 150, 158–165. [Google Scholar] [CrossRef]
- Reid, M.J.; Bornheim, L.M. Cannabinoid-induced alterations in brain disposition of drugs of abuse. Biochem. Pharmacol. 2001, 61, 1357–1367. [Google Scholar] [CrossRef]
- Leibman, D.; Smolen, A.; Smolen, T.N. Strain, sex and developmental profiles of cocaine metabolizing enzymes in mice. Pharmacol. Biochem. Behav. 1990, 37, 161–165. [Google Scholar] [CrossRef]
- Vilela, L.R.; Gomides, L.F.; David, B.A.; Antunes, M.M.; Diniz, A.B.; Moreira Fde, A.; Menezes, G.B. Cannabidiol rescues acute hepatic toxicity and seizure induced by cocaine. Mediat. Inflamm. 2015, 2015, 523418. [Google Scholar] [CrossRef]
- Gobira, P.H.; Vilela, L.R.; Gonçalves, B.D.; Santos, R.P.; de Oliveira, A.C.; Vieira, L.B.; Aguiar, D.C.; Crippa, J.A.; Moreira, F.A. Cannabidiol, a Cannabis sativa constituent, inhibits cocaine-induced seizures in mice: Possible role of the mTOR pathway and reduction in glutamate release. Neurotoxicology 2015, 50, 116–121. [Google Scholar] [CrossRef]
- Gerdeman, G.L.; Schechter, J.B.; French, E.D. Context-specific reversal of cocaine sensitization by the CB1 cannabinoid receptor antagonist rimonabant. Neuropsychopharmacology 2008, 33, 2747–2759. [Google Scholar] [CrossRef]
- Luján, M.Á.; Castro-Zavala, A.; Alegre-Zurano, L.; Valverde, O. Repeated Cannabidiol treatment reduces cocaine intake and modulates neural proliferation and CB1R expression in the mouse hippocampus. Neuropharmacology 2018, 143, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Filev, R.; Engelke, D.S.; Da Silveira, D.X.; Mello, L.E.; Santos-Junior, J.G. THC inhibits the expression of ethanol-induced locomotor sensitization in mice. Alcohol 2017, 65, 31–35. [Google Scholar] [CrossRef] [PubMed]
- Renard, J.; Loureiro, M.; Rosen, L.G.; Zunder, J.; de Oliveira, C.; Schmid, S. Cannabidiol counteracts amphetamine-induced neuronal and behavioral sensitization of the mesolimbic dopamine pathway through a novel mTOR/p70S6 kinase signaling pathway. J. Neurosci. 2016, 36, 5160–5169. [Google Scholar] [CrossRef] [PubMed]
- Mahmud, A.; Gallant, S.; Sedki, F.; D’Cunha, T.; Shalev, U. Effects of an acute cannabidiol treatment on cocaine self-administration and cue-induced cocaine seeking in male rats. J. Psychopharmacol. 2017, 31, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Hay, G.L.; Baracz, S.J.; Everett, N.A.; Roberts, J.; Costa, P.A.; Arnold, J.C.; McGregor, I.S.; Cornish, J.L. Cannabidiol treatment reduces the motivation to self-administer methamphetamine and methamphetamine-primed relapse in rats. J. Psychopharmacol. 2018, 32, 1369–1378. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Cuevas, G.; Martin-Fardon, R.; Kerr, T.M.; Stouffer, D.G.; Parsons, L.H.; Hammell, D.C.; Banks, S.L.; Stinchcomb, A.L.; Weiss, F. Unique treatment potential of cannabidiol for the prevention of relapse to drug use: Preclinical proof of principle. Neuropsychopharmacology 2018, 43, 2036–2045. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho, C.R.; Takahashi, R.N. Cannabidiol disrupts the reconsolidation of contextual drug-associated memories in Wistar rats. Addict. Biol. 2017, 22, 742–751. [Google Scholar] [CrossRef] [PubMed]
- Karimi-Haghighi, S.; Haghparast, A. Cannabidiol inhibits priming-induced reinstatement of methamphetamine in REM sleep deprived rats. Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 82, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Fischer, B.; Kuganesan, S.; Gallassi, A.; Malcher-Lopes, R.; van den Brink, W.; Wood E6. Addressing the stimulant treatment gap: A call to investigate the therapeutic benefits potential of cannabinoids for crack-cocaine use. Int. J. Drug Policy 2015, 26, 1177–1182. [Google Scholar] [CrossRef] [PubMed]
- Socías, M.E.; Kerr, T.; Wood, E.; Dong, H.; Lake, S.; Hayashi, K. Intentional cannabis use to reduce crack cocaine use in a Canadian setting: A longitudinal analysis. Addict. Behav. 2017, 72, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Giasson-Gariépy, K.; Potvin, S.; Ghabrash, M.; Bruneau, J.; Jutras-Aswad, D. Cannabis and cue-induced craving in cocaine-dependent individuals: A pilot study. Addict. Behav. 2017, 73, 4–8. [Google Scholar] [CrossRef] [PubMed]
- Murillo-Rodríguez, E.; Palomero-Rivero, M.; Millán-Aldaco, D.; Mechoulam, R.; Drucker-Colín, R. Effects on sleep and dopamine levels of microdialysis perfusion of cannabidiol into the lateral hypothalamus of rats. Life Sci. 2011, 88, 504–511. [Google Scholar] [CrossRef] [PubMed]
- Mijangos-Moreno, S.; Poot-Aké, A.; Arankowsky-Sandoval, G.; Murillo-Rodríguez, E. Intrahypothalamic injection of cannabidiol increases the extracellular levels of adenosine in nucleus accumbens in rats. Neurosci. Res. 2014, 84, 60–63. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, V.M.; Zuardi, A.W.; Del Bel, E.A.; Guimarães, F.S. Cannabidiol increases Fos expression in the nucleus accumbens but not in the dorsal striatum. Life Sci. 2004, 75, 633–638. [Google Scholar] [CrossRef] [PubMed]
- Viudez-Martínez, A.; García-Gutiérrez, M.S.; Fraguas-Sánchez, A.I.; Torres-Suárez, A.I.; Manzanares, J. Effects of cannabidiol plus naltrexone on motivation and ethanol consumption. Br. J. Pharmacol. 2018, 75, 3369–3378. [Google Scholar] [CrossRef] [PubMed]
- Pedrazzi, J.F.C.; Issy, A.C.; Gomes, F.V. Cannabidiol effects in the prepulse inhibition disruption induced by amphetamine. Psychopharmacology 2015, 232, 3057. [Google Scholar] [CrossRef] [PubMed]
- Renard, J.; Norris, C.; Rushlow, W.; Laviolette, S.R. Neuronal and molecular effects of cannabidiol on the mesolimbic dopamine system: Implications for novel schizophrenia treatments. Neurosci. Biobehav. Rev. 2017, 75, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Valvassori, S.S.; Elias, G.; de Souza, B.; Petronilho, F.; Dal-Pizzol, F.; Kapczinski, F.; Crippa, J.A. Effects of cannabidiol on amphetamine-induced oxidative stress generation in an animal model of mania. J. Psychopharmacol. 2011, 25, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Whittard, J.; Higuera-Matas, A.; Morris, C.V.; Hurd, Y.L. Cannabidiol, a nonpsychotropic component of cannabis, inhibits cue-induced heroin seeking and normalizes discrete mesolimbic neuronal disturbances. J. Neurosci. 2009, 29, 14764–14769. [Google Scholar] [CrossRef]
- McPartland, J.M.; Duncan, M.; Di Marzo, V.; Pertwee, R.G. Are cannabidiol and Δ(9) -tetrahydrocannabivarin negative modulators of the endocannabinoid system? A systematic review. Br. J. Pharmacol. 2015, 172, 737–753. [Google Scholar] [CrossRef] [PubMed]
- Adamczyk, P.; Miszkiel, J.; McCreary, A.C.; Filip, M.; Papp, M.; Przegalinski, E. The effects of cannabinoid CB1, CB2 and vanilloid TRPV1 receptor antagonists on cocaine addictive behavior in rats. Brain Res. 2012, 1444, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Linge, R.; Jiménez-Sánchez, L.; Campa, L.; Pilar-Cuéllar, F.; Vidal, R.; Pazos, A.; Adell, A.; Díaz, Á. Cannabidiol induces rapid-acting antidepressant-like effects and enhances cortical 5-HT/glutamate neurotransmission: Role of 5-HT1A receptors. Neuropharmacology 2016, 103, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.P.; Homberg, J.R. The role of serotonin in drug use and addiction. Behav. Brain Res. 2015, 277, 146–192. [Google Scholar] [CrossRef] [PubMed]
- Shelton, K.L.; Hendrick, E.S.; Beardsley, P.M. Efficacy of buspirone for attenuating cocaine and methamphetamine reinstatement in rats. Drug Alcohol Depend. 2013, 129, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Ago, Y.; Nakamura, S.; Uda, M.; Kajii, Y.; Abe, M.; Baba, A.; Matsuda, T. Attenuation by the 5-HT1A receptor agonist osemozotan of the behavioral effects of single and repeated methamphetamine in mice. Neuropharmacology 2006, 51, 914–922. [Google Scholar] [CrossRef] [PubMed]
- Picard, M.; Morisset, S.; Cloix, J.F.; Bizot, J.C.; Guerin, M.; Beneteau, V.; Guillaumet, G.; Hevor, T.K. Pharmacological, neurochemical, and behavioral profile of JB-788, a new 5-HT1A agonist. Neuroscience 2010, 169, 1337–1346. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.P.; Carey, R.J.; Huston, J.P.; De Souza Silva, M.A. Serotonin and psychostimulant addiction: Focus on 5-HT1A-receptors. Prog. Neurobiol. 2007, 81, 133–178. [Google Scholar] [CrossRef]
- Feltenstein, M.W.; Altar, C.A.; See, R.E. Aripiprazole blocks reinstatement of cocaine seeking in an animal model of relapse. Biol. Psychiatry 2007, 61, 582–590. [Google Scholar] [CrossRef]
- Feltenstein, M.W.; Do, P.H.; See, R.E. Repeated aripiprazole administration attenuates cocaine seeking in a rat model of relapse. Psychopharmacology 2009, 207, 401–411. [Google Scholar] [CrossRef]
- Sørensen, G.; Sager, T.N.; Petersen, J.H.; Brennum, L.T.; Thøgersen, P.; Hee Bengtsen, C.; Thomsen, M.; Wörtwein, G.; Fink-Jensen, A.; Woldbye, D.P. Aripiprazole blocks acute self-administration of cocaine and is not self-administered in mice. Psychopharmacology 2008, 199, 37–46. [Google Scholar] [CrossRef]
- Pulvirenti, L.; Balducci, C.; Piercy, M.; Koob, G.F. Characterization of the effects of the partial dopamine agonist terguride on cocaine self-administration in the rat. J. Pharmacol. Exp. Ther. 1998, 286, 1231–1238. [Google Scholar] [PubMed]
- Bäckström, P.; Etelälahti, T.J.; Hyytiä, P. Attenuation of reinforcing and psychomotor stimulant effects of amphetamine by aripiprazole. Addict. Biol. 2011, 16, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Wee, S.; Wang, Z.; Woolverton, W.L.; Pulvirenti, L.; Koob, G.F. Effect of aripiprazole, a partial dopamine D2 receptor agonist, on increased rate of methamphetamine self-administration in rats with prolonged session duration. Neuropsychopharmacology 2007, 32, 2238–2247. [Google Scholar] [CrossRef] [PubMed]
- Panagis, G.; Kastellakis, A.; Spyraki, C.; Nomikos, G. Effects of methyllycaconitine (MLA), an alpha 7 nicotinic receptor antagonist, on nicotine- and cocaine-induced potentiation of brain stimulation reward. Psychopharmacology 2000, 149, 388–396. [Google Scholar] [CrossRef]
- Zanetti, L.; de Kerchove D’Exaerde, A.; Zanardi, A.; Changeux, J.P.; Picciotto, M.R.; Zoli, M. Inhibition of both alpha7* and beta2* nicotinic acetylcholine receptors is necessary to prevent development of sensitization to cocaine-elicited increases in extracellular dopamine levels in the ventral striatum. Psychopharmacology 2006, 187, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Escubedo, E.; Chipana, C.; Pérez-Sánchez, M.; Camarasa, J.; Pubill, D. Methyllycaconitine prevents methamphetamine-induced effects in mouse striatum: Involvement of alpha7 nicotinic receptors. J. Pharmacol. Exp. Ther. 2005, 315, 658–667. [Google Scholar] [CrossRef]
- Tourino, C.; Valjent, E.; Ruiz-Medina, J.; Herve, D.; Ledent, C.; Valverde, O. The orphan receptor GPR3 modulates the early phases of cocaine reinforcement. Br. J. Pharmacol. 2012, 167, 892–904. [Google Scholar] [CrossRef]
- O’Sullivan, S.E.; Kendall, D.A. Cannabinoid activation of peroxisome proliferator-activated receptors: Potential for modulation of inflammatory disease. Immunobiology 2010, 215, 611–666. [Google Scholar] [CrossRef]
- O’Sullivan, S.E. An update on PPAR activation by cannabinoids. Br. J. Pharmacol. 2016, 173, 1899–1910. [Google Scholar] [CrossRef]
- Maeda, T.; Kiguchi, N.; Fukazawa, Y.; Yamamoto, A.; Ozaki, M.; Kishioka, S. Peroxisome proliferator-activated receptor gamma activation relieves expression of behavioral sensitization to methamphetamine in mice. Neuropsychopharmacology 2007, 32, 1133–1140. [Google Scholar] [CrossRef]
- Schmitz, J.M.; Green, C.E.; Hasan, K.M.; Vincent, J.; Suchting, R.; Weaver, M.F.; Moeller, F.G.; Narayana, P.A.; Cunningham, K.A.; Dineley, K.T.; et al. PPAR-gamma agonist pioglitazone modifies craving intensity and brain white matter integrity in patients with primary cocaine use disorder: A double-blind randomized controlled pilot trial. Addiction 2017, 112, 1861–1868. [Google Scholar] [CrossRef] [PubMed]
- Bachtell, R.K.; Jones, J.D.; Heinzerling, K.G.; Beardsley, P.M.; Comer, S.D. Glial and neuroinflammatory targets for treating substance use disorders. Drug Alcohol. Depend. 2017, 180, 156–170. [Google Scholar] [CrossRef] [PubMed]
- Crews, F.T.; Zou, J.; Qin, L. Induction of innate immune genes in brain create the neurobiology of addiction. Brain Behav. Immun. 2011, 25, S4–S12. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.; Levis, S.; O’Neill, C.; Northcutt, A.; Fabisiak, T.; Watkins, L.; Bachtell, R. Innate immune signaling in the ventral tegmental area contributes to drug-primed reinstatement of cocaine seeking. Brain Behav. Immun. 2018, 67, 130–138. [Google Scholar] [CrossRef]
- Karimi-Haghighi, S.; Dargahi, L.; Haghparast, A. Cannabidiol modulates the expression of neuroinflammatory factors in stress -and drug- induced reinstatement of methamphetamine in extinguished rats. Addict. Biol. 2019. [Google Scholar] [CrossRef]
- Kozela, E.; Juknat, A.; Vogel, Z. Modulation of Astrocyte Activity by Cannabidiol, a Nonpsychoactive Cannabinoid. Int. J. Mol. Sci. 2017, 18, 8. [Google Scholar] [CrossRef]
- Saravia, R.; Ten-Blanco, M.; Grande, M.T.; Maldonado, R.; Berrendero, F. Anti-inflammatory agents for smoking cessation? Focus on cognitive deficits associated with nicotine withdrawal in male mice. Brain Behav. Immun. 2019, 75, 228–239. [Google Scholar] [CrossRef]
- Lee, J.L.C.; Bertoglio, L.J.; Guimarães, F.S.; Stevenson, C.W. Cannabidiol regulation of emotion and emotional memory processing: Relevance for treating anxiety-related and substance abuse disorders. Br. J. Pharmacol. 2017, 174, 3242–3256. [Google Scholar] [CrossRef]
- Stern, C.A.J.; de Carvalho, C.R.; Bertoglio, L.J.; Takahashi, R.N. Effects of Cannabinoid Drugs on Aversive or Rewarding Drug-Associated Memory Extinction and Reconsolidation. Neuroscience 2018, 370, 62–80. [Google Scholar] [CrossRef]
- Milton, A.L. Drink, drugs and disruption: Memory manipulation for the treatment of addiction. Curr. Opin. Neurobiol. 2013, 23, 706–712. [Google Scholar] [CrossRef]
- Bitencourt, R.M.; Takahashi, R.N. Cannabidiol as a Therapeutic Alternative for Post-traumatic Stress Disorder: From Bench Research to Confirmation in Human Trials. Front. Neurosci. 2018, 12, 502. [Google Scholar] [CrossRef] [PubMed]
- Stern, C.A.; Gazarini, L.; Takahashi, R.N.; Guimarães, F.S.; Bertoglio, L.J. On disruption of fear memory by reconsolidation blockade: Evidence from cannabidiol treatment. Neuropsychopharmacology 2012, 37, 2132–2142. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Stevenson, C.W.; Guimaraes, F.S.; Lee, J.L. Bidirectional Effects of Cannabidiol on Contextual Fear Memory Extinction. Front. Pharmacol. 2016, 7, 493. [Google Scholar] [CrossRef] [PubMed]
- Jurkus, R.; Day, H.L.; Guimarães, F.S.; Lee, J.L.; Bertoglio, L.J.; Stevenson, C.W. Cannabidiol Regulation of Learned Fear: Implications for Treating Anxiety-Related Disorders. Front. Pharmacol. 2016, 7, 454. [Google Scholar] [CrossRef] [PubMed]
- Gazarini, L.; Stern, C.A.; Piornedo, R.R.; Takahashi, R.N.; Bertoglio, L.J. PTSD-like memory generated through enhanced noradrenergic activity is mitigated by a dual step pharmacological intervention targeting its reconsolidation. Int. J. Neuropsychopharmacol. 2014, 18. [Google Scholar] [CrossRef]
- Potvin, S.; Stavro, K.; Rizkallah, E.; Pelletier, J. Cocaine and cognition: A systematic quantitative review. J. Addict. Med. 2014, 8, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Sabrini, S.; Wang, G.Y.; Lin, J.C.; Ian, J.K.; Curley, L.E. Methamphetamine use and cognitive function: A systematic review of neuroimaging research. Drug Alcohol Depend. 2019, 194, 75–87. [Google Scholar] [CrossRef]
- Sofuoglu, M.; DeVito, E.E.; Waters, A.J.; Carroll, K.M. Cognitive Function as a Transdiagnostic Treatment Target in Stimulant Use Disorders. J. Dual Diagn. 2016, 12, 90–106. [Google Scholar] [CrossRef]
- Rezapour, T.; DeVito, E.E.; Sofuoglu, M.; Ekhtiari, H. Perspectives on neurocognitive rehabilitation as an adjunct treatment for addictive disorders: From cognitive improvement to relapse prevention. Prog. Brain Res. 2016, 224, 345–369. [Google Scholar] [CrossRef]
- Aso, E.; Fernández-Dueñas, V.; López-Cano, M.; Taura, J.; Watanabe, M.; Ferrer, I.; Luján, R.; Ciruela, F. Adenosine A2A-Cannabinoid CB1 Receptor Heteromers in the Hippocampus: Cannabidiol Blunts Δ9-Tetrahydrocannabinol-Induced Cognitive Impairment. Mol. Neurobiol. 2019. [Google Scholar] [CrossRef]
- Solowij, N.; Broyd, S.J.; Beale, C.; Prick, J.A.; Greenwood, L.; van Hell, H. Therapeutic effects of prolonged cannabidiol treatment on psychological symptoms and cognitive function in regular cannabis users: A pragmatic open-label clinical trial. Cannabis Cannabinoid Res. 2018, 3, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Hindocha, C.; Freeman, T.P.; Grabski, M.; Crudgington, H.; Davies, A.C.; Stroud, J.B.; Das, R.K.; Lawn, W.; Morgan, C.J.A.; Curran, H.V. The effects of cannabidiol on impulsivity and memory during abstinence in cigarette dependent smokers. Sci. Rep. 2018, 8, 7568. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, S.; Morrison, P.D.; Fusar-Poli, P.; Martin-Santos, R.; Borgwardt, S.; Winton-Brown, T. Opposite effects of 1-9-tetrahydrocannabinol and cannabidiol on human brain function and psychopathology. Neuropsychopharmacology 2010, 35, 764–774. [Google Scholar] [CrossRef]
- Greenwald, M.K. Anti-stress neuropharmacological mechanisms and targets for addiction treatment: A translational framework. Neurobiol. Stress 2018, 9, 84–104. [Google Scholar] [CrossRef] [PubMed]
- Blessing, E.M.; Steenkamp, M.M.; Manzanares, J.; Marmar, C.R. Cannabidiol as a Potential Treatment for Anxiety Disorders. Neurotherapeutics 2015, 12, 825–836. [Google Scholar] [CrossRef]
- Soares, V.P.; Campos, A.C. Evidences for the Anti-panic Actions of Cannabidiol. Curr. Neuropharmacol. 2017, 15, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Mandolini, G.M.; Lazzaretti, M.; Pigoni, A.; Oldani, L.; Delvecchio, G.; Brambilla, P. Pharmacological properties of cannabidiol in the treatment of psychiatric disorders: A critical overview. Epidemiol. Psychiatr. Sci. 2018, 27, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Campos, A.C.; Moreira, F.A.; Gomes, F.V.; Del Bel, E.A.; Guimarães, F.S. Multiple mechanisms involved in the large-spectrum therapeutic potential of cannabidiol in psychiatric disorders. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2012, 367, 3364–3378. [Google Scholar] [CrossRef]
- Lisboa, S.F.; Vila-Verde, C.; Rosa, J.; Uliana, D.L.; Stern, C.A.; Bertoglio, L.J.; Resstel, L.B.; Guimaraes, F.S. Tempering aversive/traumatic memories with cannabinoids: A review of evidence from animal and human studies. Psychopharmacology 2019, 236, 201–226. [Google Scholar] [CrossRef]
- Deiana, S.; Watanabe, A.; Yamasaki, Y.; Amada, N.; Arthur, M.; Fleming, S.; Woodcock, H.; Dorward, P.; Pigliacampo, B.; Close, S.; et al. Plasma and brain pharmacokinetic profile of cannabidiol (CBD), cannabidivarine (CBDV), Δ⁹-tetrahydrocannabivarin (THCV) and cannabigerol (CBG) in rats and mice following oral and intraperitoneal administration and CBD action on obsessive-compulsive behaviour. Psychopharmacology 2012, 219, 859–873. [Google Scholar] [CrossRef]
- Martín-González, E.; Prados-Pardo, Á.; Mora, S.; Flores, P.; Moreno, M. Do psychoactive drugs have a therapeutic role in compulsivity? Studies on schedule-induced polydipsia. Psychopharmacology 2018, 235, 419–432. [Google Scholar] [CrossRef] [PubMed]
- Alexander, P.D.; Gicas, K.M.; Willi, T.S.; Kim, C.N.; Boyeva, V.; Procyshyn, R.M.; Smith, G.N.; Thornton, A.E.; Panenka, W.J.; Jones, A.A.; et al. A comparison of psychotic symptoms in subjects with methamphetamine versus cocaine dependence. Psychopharmacology 2017, 234, 1535–1547. [Google Scholar] [CrossRef] [PubMed]
- Rohleder, C.; Müller, J.K.; Lange, B.; Leweke, F.M. Cannabidiol as a potential new type of an antipsychotic. A critical review of the evidence. Front. Pharmacol. 2016, 7, 422. [Google Scholar] [CrossRef] [PubMed]
- Gomes, F.V.; Llorente, R.; Del Bel, E.A.; Viveros, M.P.; López-Gallardo, M.; Guimarães, F.S. Decreased glial reactivity could be involved in the antipsychotic-like effect of cannabidiol. Schizophr. Res. 2015, 164, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Hudson, R.; Rushlow, W.; Laviolette, S.R. Phytocannabinoids modulate emotional memory processing through interactions with the ventral hippocampus and mesolimbic dopamine system: Implications for neuropsychiatric pathology. Psychopharmacology 2018, 235, 447–458. [Google Scholar] [CrossRef]
- Saletti, P.G.; Tomaz, C. Cannabidiol effects on prepulse inhibition in nonhuman primates. Rev. Neurosci. 2018, 30, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Long, L.E.; Malone, D.T.; Taylor, D.A. Cannabidiol reverses MK-801-induced disruption of prepulse inhibition in mice. Neuropsychopharmacology 2006, 31, 795–803. [Google Scholar] [CrossRef]
- El-Alfy, A.T.; Ivey, K.; Robinson, K.; Ahmed, S.; Radwan, M.; Slade, D.; Khan, I.; ElSohly, M.; Ross, S. Antidepressant-like effect of delta9-tetrahydrocannabinol and other cannabinoids isolated from Cannabis sativa L. Pharmacol. Biochem. Behav. 2010, 95, 434–442. [Google Scholar] [CrossRef]
- Sales, A.J.; Crestani, C.C.; Guimarães, F.S.; Joca, S.R.L. Antidepressant-like effect induced by Cannabidiol is dependent on brain serotonin levels. Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 86, 255–261. [Google Scholar] [CrossRef]
- Sales, A.J.; Fogaça, M.V.; Sartim, A.G.; Pereira, V.S.; Wegener, G.; Guimarães, F.S.; Joca, S.R.L. Cannabidiol Induces Rapid and Sustained Antidepressant-Like Effects Through Increased BDNF Signaling and Synaptogenesis in the Prefrontal Cortex. Mol. Neurobiol. 2019, 56, 1070–1081. [Google Scholar] [CrossRef]
- Sartim, A.G.; Sales, A.J.; Guimarães, F.S.; Joca, S.R. Hippocampal mammalian target of rapamycin is implicated in stress-coping behavior induced by cannabidiol in the forced swim test. J. Psychopharmacol. 2018, 32, 922–931. [Google Scholar] [CrossRef] [PubMed]
- Zanelati, T.V.; Biojone, C.; Moreira, F.A.; Guimarães, F.S.; Joca, S.R. Antidepressant-like effects of cannabidiol in mice: Possible involvement of 5-HT1A receptors. Br. J. Pharmacol. 2010, 159, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Réus, G.Z.; Stringari, R.B.; Ribeiro, K.F.; Luft, T.; Abelaira, H.M.; Fries, G.R.; Aguiar, B.W.; Kapczinski, F.; Hallak, J.E.; Zuardi, A.W.; et al. Administration of cannabidiol and imipramine induces antidepressant-like effects in the forced swimming test and increases brain-derived neurotrophic factor levels in the rat amygdala. Acta Neuropsychiatr. 2011, 23, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Sartim, A.G.; Guimarães, F.S.; Joca, S.R. Antidepressant-like effect of cannabidiol injection into the ventral medial prefrontal cortex-Possible involvement of 5-HT1A and CB1 receptors. Behav. Brain Res. 2016, 303, 218–227. [Google Scholar] [CrossRef] [PubMed]
- Schiavon, A.P.; Bonato, J.M.; Milani, H.; Guimarães, F.S.; Weffort de Oliveira, R.M. Influence of single and repeated cannabidiol administration on emotional behavior and markers of cell proliferation and neurogenesis in non-stressed mice. Prog. Neuropsychopharmacol. Biol. Psychiatry 2016, 64, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Shbiro, L.; Hen-Shoval, D.; Hazut, N.; Rapps, K.; Dar, S.; Zalsman, G.; Mechoulam, R.; Weller, A.; Shoval, G. Effects of cannabidiol in males and females in two different rat models of depression. Physiol. Behav. 2019, 201, 59–63. [Google Scholar] [CrossRef] [PubMed]
- de Mello Schier, A.R.; de Oliveira Ribeiro, N.P.; Coutinho, D.S.; Machado, S.; Arias-Carrión, O.; Crippa, J.A.; Zuardi, A.W.; Nardi, A.E.; Silva, A.C. Antidepressant-like and anxiolytic-like effects of cannabidiol: A chemical compound of Cannabis sativa. CNS Neurol. Disord. Drug Targets 2014, 13, 953–960. [Google Scholar] [CrossRef]
- Augustin, S.M.; Lovinger, D.M. Functional Relevance of Endocannabinoid-Dependent Synaptic Plasticity in the Central Nervous System. ACS Chem. Neurosci. 2018, 9, 2146–2161. [Google Scholar] [CrossRef]
- Bobadilla, A.C.; Heinsbroek, J.A.; Gipson, C.D.; Griffin, W.C.; Fowler, C.D.; Kenny, P.J.; Kalivas, P.W. Corticostriatal plasticity, neuronal ensembles, and regulation of drug-seeking behavior. Prog. Brain Res. 2017, 235, 93–112. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calpe-López, C.; García-Pardo, M.P.; Aguilar, M.A. Cannabidiol Treatment Might Promote Resilience to Cocaine and Methamphetamine Use Disorders: A Review of Possible Mechanisms. Molecules 2019, 24, 2583. https://doi.org/10.3390/molecules24142583
Calpe-López C, García-Pardo MP, Aguilar MA. Cannabidiol Treatment Might Promote Resilience to Cocaine and Methamphetamine Use Disorders: A Review of Possible Mechanisms. Molecules. 2019; 24(14):2583. https://doi.org/10.3390/molecules24142583
Chicago/Turabian StyleCalpe-López, Claudia, M. Pilar García-Pardo, and Maria A. Aguilar. 2019. "Cannabidiol Treatment Might Promote Resilience to Cocaine and Methamphetamine Use Disorders: A Review of Possible Mechanisms" Molecules 24, no. 14: 2583. https://doi.org/10.3390/molecules24142583
APA StyleCalpe-López, C., García-Pardo, M. P., & Aguilar, M. A. (2019). Cannabidiol Treatment Might Promote Resilience to Cocaine and Methamphetamine Use Disorders: A Review of Possible Mechanisms. Molecules, 24(14), 2583. https://doi.org/10.3390/molecules24142583





