Analysis of Potent Odour-Active Volatile Thiols in Foods and Beverages with a Focus on Wine
Abstract
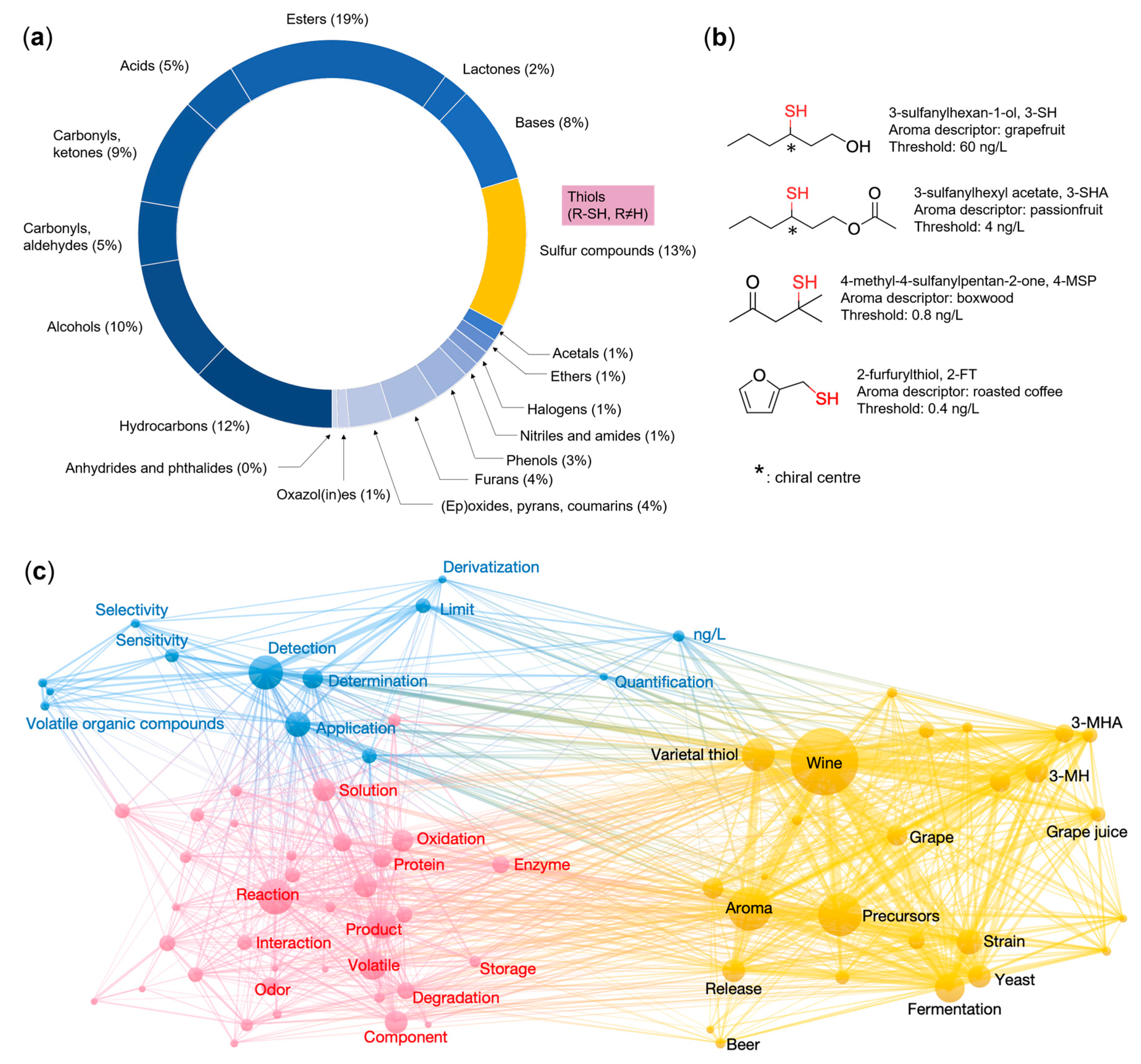
1. Introduction—Importance of Thiols to the Aroma of Foods and Beverages
2. Analytical Challenges and Requirements
- matrix complexity;
- properties of thiols.
3. Thiol Isolation—Extraction and Derivatisation
- non-specific extraction, e.g., straightforward application of headspace solid-phase microextraction (HS–SPME), solid phase extraction (SPE), liquid-liquid extraction (LLE), purge and trap (P+T), and vacuum distillation;
- selective extraction with metal ions (e.g., Hg+ or Ag+);
- derivatisation (coupled with LLE, HS–SPME, SPE, or gas purge microsyringe extraction (GP–MSE)).
3.1. Selective Extraction with Metal Ions
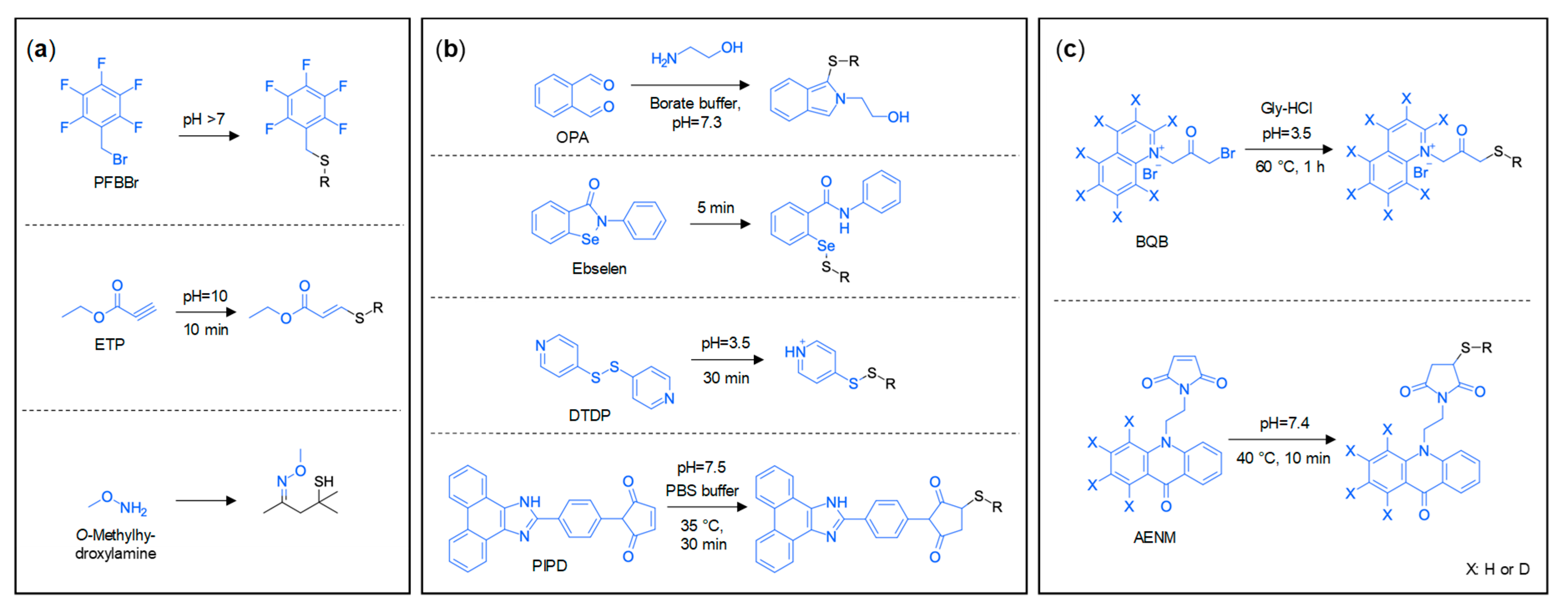
3.2. Derivatisation Approaches
3.2.1. Derivatisations for GC Analysis of Thiols
3.2.2. Derivatisations for LC Analysis of Thiols
4. Analytical Instrumentation
4.1. Analysis by GC
4.2. Analysis by LC
4.3. LC vs. GC
4.4. Other Instruments
4.5. Matrix Effects and Quantitative Analysis
5. Conclusions and Outlook
Author Contributions
Funding
Conflicts of Interest
References
- VCF Online. Available online: http://www.vcf-online.nl/VcfHome.cfm (accessed on 5 May 2019).
- McGorrin, R.J. The Significance of Volatile Sulfur Compounds in Food Flavors. In Volatile Sulfur Compounds in Food; Qian, M.C., Fan, X., Mahattanatawee, K., Eds.; American Chemical Society: Washington, DC, USA, 2011; Volume 1068, pp. 3–31. [Google Scholar]
- Mussinan, C.J.; Keelan, M.E. Sulfur compounds in foods. In Sulfur Compounds in Foods; Mussinan, C.J., Keelan, M.E., Eds.; American Chemical Society: Washington, DC, USA, 1994; Volume 564, pp. 1–6. [Google Scholar]
- Roland, A.; Schneider, R.; Razungles, A.; Cavelier, F. Varietal thiols in wine: Discovery, analysis and applications. Chem. Rev. 2011, 111, 7355–7376. [Google Scholar] [CrossRef] [PubMed]
- Waltman, L.; van Eck, N.J.; Noyons, E.C.M. A unified approach to mapping and clustering of bibliometric networks. J. Informetr. 2010, 4, 629–635. [Google Scholar] [CrossRef]
- Dubourdieu, D.; Tominaga, T. Polyfunctional thiol compounds. In Wine Chemistry and Biochemistry; Moreno-Arribas, M.V., Polo, C., Eds.; Springer: Berlin, Germany, 2009; pp. 275–293. [Google Scholar]
- Capone, D.L.; Ristic, R.; Pardon, K.H.; Jeffery, D.W. Simple quantitative determination of potent thiols at ultratrace levels in wine by derivatization and high-performance liquid chromatography–tandem mass spectrometry (HPLC-MS/MS) Analysis. Anal. Chem. 2015, 87, 1226–1231. [Google Scholar] [CrossRef] [PubMed]
- Buettner, A.; Schieberle, P. Evaluation of key aroma compounds in hand-squeezed grapefruit juice (Citrus paradisi Macfayden) by quantitation and flavor reconstitution experiments. J. Agric. Food Chem. 2001, 49, 1358–1363. [Google Scholar] [CrossRef] [PubMed]
- Tamura, H.; Fujita, A.; Steinhaus, M.; Takahisa, E.; Watanabe, H.; Schieberle, P. Identification of novel aroma-active thiols in pan-roasted white sesame seeds. J. Agric. Food Chem. 2010, 58, 7368–7375. [Google Scholar] [CrossRef] [PubMed]
- Steinhaus, M.; Sinuco, D.; Polster, J.; Osorio, C.; Schieberle, P. Characterization of the aroma-active compounds in pink guava (Psidium guajava, L.) by application of the aroma extract dilution analysis. J. Agric. Food Chem. 2008, 56, 4120–4127. [Google Scholar] [CrossRef] [PubMed]
- Coetzee, C.; du Toit, W.J. A comprehensive review on Sauvignon blanc aroma with a focus on certain positive volatile thiols. Food Res. Int. 2012, 45, 287–298. [Google Scholar] [CrossRef]
- Vermeulen, C.; Lejeune, I.; Tran, T.T.H.; Collin, S. Occurrence of polyfunctional thiols in fresh lager beers. J. Agric. Food Chem. 2006, 54, 5061–5068. [Google Scholar] [CrossRef]
- Dulsat-Serra, N.; Quintanilla-Casas, B.; Vichi, S. Volatile thiols in coffee: A review on their formation, degradation, assessment and influence on coffee sensory quality. Food Res. Int. 2016, 89, 982–988. [Google Scholar] [CrossRef]
- Vermeulen, C.; Gijs, L.; Collin, S. Sensorial contribution and formation pathways of thiols in foods: A review. Food Rev. Int. 2005, 21, 69–137. [Google Scholar] [CrossRef]
- Souza-Silva, E.A.; Gionfriddo, E.; Pawliszyn, J. A critical review of the state of the art of solid-phase microextraction of complex matrices II. Food analysis. TrAC Trends Anal. Chem. 2015, 71, 236–248. [Google Scholar] [CrossRef]
- Liem-Nguyen, V.; Bouchet, S.; Björn, E. Determination of sub-nanomolar levels of low molecular mass thiols in natural waters by liquid chromatography tandem mass spectrometry after derivatization with p-(hydroxymercuri) benzoate and online preconcentration. Anal. Chem. 2015, 87, 1089–1096. [Google Scholar] [CrossRef]
- Block, E.; Calvey, E.M. Facts and artifacts in Allium chemistry. In Sulfur Compounds in Foods; Keelan, M.E., Mussinan, C.J., Eds.; American Chemical Society: Washington, DC, USA, 1994; Volume 564, pp. 63–79. [Google Scholar]
- Mateo-Vivaracho, L.; Ferreira, V.; Cacho, J. Automated analysis of 2-methyl-3-furanthiol and 3-mercaptohexyl acetate at ngL−1 level by headspace solid-phase microextracion with on-fibre derivatisation and gas chromatography–negative chemical ionization mass spectrometric determination. J. Chromatogr. A 2006, 1121, 1–9. [Google Scholar] [CrossRef]
- Herbst-Johnstone, M.; Piano, F.; Duhamel, N.; Barker, D.; Fedrizzi, B. Ethyl propiolate derivatisation for the analysis of varietal thiols in wine. J. Chromatogr. A 2013, 1312, 104–110. [Google Scholar] [CrossRef]
- Hart, H.; Schuetz, R.D. Organic Chemistry: A Short Course; Houghton Mifflin: Boston, MA, USA, 1978; p. 148. [Google Scholar]
- Darriet, P.; Tominaga, T.; Lavigne, V.; Boidron, J.-N.; Dubourdieu, D. Identification of a powerful aromatic component of Vitis vinifera L. var. Sauvignon wines: 4-mercapto-4-methylpentan-2-one. Flavour Fragance J. 1995, 10, 385–392. [Google Scholar] [CrossRef]
- Tominaga, T.; Murat, M.-L.; Dubourdieu, D. Development of a method for analyzing the volatile thiols involved in the characteristic aroma of wines made from Vitis vinifera L. Cv. Sauvignon blanc. J. Agric. Food Chem. 1998, 46, 1044–1048. [Google Scholar] [CrossRef]
- Schoenauer, S.; Schieberle, P. Screening for novel mercaptans in 26 fruits and 20 wines using a thiol-selective isolation procedure in combination with three detection methods. J. Agric. Food Chem. 2019, 67, 4553–4559. [Google Scholar] [CrossRef]
- Tominaga, T.; Blanchard, L.; Darriet, P.; Dubourdieu, D. A powerful aromatic volatile thiol, 2-furanmethanethiol, exhibiting roast coffee aroma in wines made from several Vitis vinifera grape varieties. J. Agric. Food Chem. 2000, 48, 1799–1802. [Google Scholar] [CrossRef]
- Schneider, R.; Kotseridis, Y.; Ray, J.-L.; Augier, C.; Baumes, R. Quantitative determination of sulfur-containing wine odorants at sub parts per billion levels. 2. Development and application of a stable isotope dilution assay. J. Agric. Food Chem. 2003, 51, 3243–3248. [Google Scholar] [CrossRef]
- Sourabié, A.M.; Spinnler, H.-E.; Bonnarme, P.; Saint-Eve, A.; Landaud, S. Identification of a powerful aroma compound in Munster and Camembert cheeses: Ethyl 3-mercaptopropionate. J. Agric. Food Chem. 2008, 56, 4674–4680. [Google Scholar] [CrossRef]
- Reglitz, K.; Steinhaus, M. Quantitation of 4-methyl-4-sulfanylpentan-2-one (4MSP) in hops by a stable isotope dilution assay in combination with GC×GC-TOFMS: Method development and application to study the influence of variety, provenance, harvest year, and processing on 4MSP concentrations. J. Agric. Food Chem. 2017, 65, 2364–2372. [Google Scholar]
- Takazumi, K.; Takoi, K.; Koie, K.; Tuchiya, Y. Quantitation method for polyfunctional thiols in hops (Humulus lupulus L.) and beer using specific extraction of thiols and gas chromatography–tandem mass spectrometry. Anal. Chem. 2017, 89, 11598–11604. [Google Scholar] [CrossRef]
- Mateo-Vivaracho, L.; Cacho, J.; Ferreira, V. Quantitative determination of wine polyfunctional mercaptans at nanogram per liter level by gas chromatography–negative ion mass spectrometric analysis of their pentafluorobenzyl derivatives. J. Chromatogr. A 2007, 1146, 242–250. [Google Scholar] [CrossRef]
- Mateo-Vivaracho, L.; Cacho, J.; Ferreira, V. Improved solid-phase extraction procedure for the isolation and in-sorbent pentafluorobenzyl alkylation of polyfunctional mercaptans: Optimized procedure and analytical applications. J. Chromatogr. A 2008, 1185, 9–18. [Google Scholar] [CrossRef]
- Rodríguez-Bencomo, J.J.; Schneider, R.; Lepoutre, J.P.; Rigou, P. Improved method to quantitatively determine powerful odorant volatile thiols in wine by headspace solid-phase microextraction after derivatization. J. Chromatogr. A 2009, 1216, 5640–5646. [Google Scholar] [CrossRef]
- Capone, D.L.; Sefton, M.A.; Jeffery, D.W. Application of a modified method for 3-mercaptohexan-1-ol determination to investigate the relationship between free thiol and telated conjugates in grape juice and wine. J. Agric. Food Chem. 2011, 59, 4649–4658. [Google Scholar] [CrossRef]
- Musumeci, L.E.; Ryona, I.; Pan, B.S.; Loscos, N.; Feng, H.; Cleary, M.T.; Sacks, G.L. Quantification of polyfunctional thiols in wine by HS-SPME-GC-MS following extractive alkylation. Molecules 2015, 20, 12280–12299. [Google Scholar] [CrossRef]
- Ochiai, N.; Sasamoto, K.; Kishimoto, T. Development of a method for the quantitation of three thiols in beer, hop, and wort samples by stir bar sorptive extraction with in situ derivatization and thermal desorption gas chromatography–tandem mass spectrometry. J. Agric. Food Chem. 2015, 63, 6698–6706. [Google Scholar] [CrossRef]
- Dagan, L.; Reillon, F.; Roland, A.; Schneider, R. Development of a routine analysis of 4-mercapto-4-methylpentan-2-one in wine by stable isotope dilution assay and mass tandem spectrometry. Anal. Chim. Acta 2014, 821, 48–53. [Google Scholar] [CrossRef]
- Piano, F.; Fracassetti, D.; Buica, A.; Stander, M.; du Toit, W.J.; Borsa, D.; Tirelli, A. Development of a novel liquid/liquid extraction and ultra-performance liquid chromatography tandem mass spectrometry method for the assessment of thiols in South African Sauvignon Blanc wines. Aust. J. Grape Wine Res. 2015, 21, 40–48. [Google Scholar] [CrossRef]
- Vichi, S.; Cortés-Francisco, N.; Caixach, J. Determination of volatile thiols in lipid matrix by simultaneous derivatization/extraction and liquid chromatography–high resolution mass spectrometric analysis. Application to virgin olive oil. J. Chromatogr. A 2013, 1318, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Vichi, S.; Cortés-Francisco, N.; Caixach, J. Analysis of volatile thiols in alcoholic beverages by simultaneous derivatization/extraction and liquid chromatography-high resolution mass spectrometry. Food Chem. 2015, 175, 401–408. [Google Scholar] [CrossRef]
- Quintanilla-Casas, B.; Dulsat-Serra, N.; Cortés-Francisco, N.; Caixach, J.; Vichi, S. Thiols in brewed coffee: Assessment by fast derivatization and liquid chromatography–high resolution mass spectrometry. LWT Food Sci. Technol. 2015, 64, 1085–1090. [Google Scholar] [CrossRef]
- Vichi, S.; Jerí, Y.; Cortés-Francisco, N.; Palacios, O.; Caixach, J. Determination of volatile thiols in roasted coffee by derivatization and liquid chromatography–high resolution mass spectrometric analysis. Food Res. Int. 2014, 64, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Capone, D.L.; Jeffery, D.W. Chiral analysis of 3-sulfanylhexan-1-ol and 3-sulfanylhexyl acetate in wine by high-performance liquid chromatography–tandem mass spectrometry. Anal. Chim. Acta 2018, 998, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Mafata, M.; Stander, M.; Thomachot, B.; Buica, A. Measuring thiols in single cultivar South African red wines using 4,4-dithiodipyridine (DTDP) derivatization and ultraperformance convergence chromatography-tandem mass spectrometry. Foods 2018, 7, 138. [Google Scholar] [CrossRef]
- Liu, P.; Huang, Y.-Q.; Cai, W.-J.; Yuan, B.-F.; Feng, Y.-Q. Profiling of thiol-containing compounds by stable isotope labeling double precursor ion scan mass spectrometry. Anal. Chem. 2014, 86, 9765–9773. [Google Scholar] [CrossRef]
- Lv, Z.; You, J.; Lu, S.; Sun, W.; Ji, Z.; Sun, Z.; Song, C.; Chen, G.; Li, G.; Hu, N.; et al. Sensitive determination of thiols in wine samples by a stable isotope-coded derivatization reagent d0/d4-acridone-10-ethyl-N-maleimide coupled with high-performance liquid chromatography-electrospray ionization-tandem mass spectrometry analysis. J. Chromatogr. A 2017, 1491, 98–107. [Google Scholar] [CrossRef]
- Zhuang, J.; You, J.; Zhang, S.; Sun, Z.; Ji, Z.; Liu, J.; Yu, Y. Determination of thiols by gas purge microsyringe extraction coupled with chemical derivatization by high performance liquid chromatography-fluorescence detection with mass spectrometry identification. J. Liq. Chromatogr. Relat. Technol. 2018, 41, 794–803. [Google Scholar] [CrossRef]
- Kumazawa, K.; Kubota, K.; Masuda, H. Influence of manufacturing conditions and crop season on the formation of 4-mercapto-4-methyl-2-pentanone in Japanese green tea (Sen-cha). J. Agric. Food Chem. 2005, 53, 5390–5396. [Google Scholar] [CrossRef]
- Takoi, K.; Degueil, M.; Shinkaruk, S.; Thibon, C.; Maeda, K.; Ito, K.; Bennetau, B.; Dubourdieu, D.; Tominaga, T. Identification and characteristics of new volatile thiols derived from the hop (Humulus luplus L.) cultivar Nelson Sauvin. J. Agric. Food Chem. 2009, 57, 2493–2502. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Armbruster, M.R.; Coulton, J.B.; Edwards, J.L. Chemical tagging in mass spectrometry for systems biology. Anal. Chem. 2019, 91, 109–125. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.-J.; Wang, Y.-L.; Liu, P.; Zhang, Z.; Yu, L.; Yuan, B.-F.; Feng, Y.-Q. Stable isotope labeling-solid phase extraction-mass spectrometry analysis for profiling of thiols and aldehydes in beer. Food Chem. 2017, 237, 399–407. [Google Scholar] [CrossRef]
- Zuman, P. Reactions of orthophthalaldehyde with nucleophiles. Chem. Rev. 2004, 104, 3217–3238. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Zhang, Y.; Tang, B.; Laskin, J.; Roach, P.J.; Chen, H. Study of highly selective and efficient thiol derivatization using selenium reagents by mass spectrometry. Anal. Chem. 2010, 82, 6926–6932. [Google Scholar] [CrossRef]
- Chiu, F.C.K.; Lo, C.M.Y. Observation of amide anions in solution by electrospray ionization mass spectrometry. J. Am. Soc. Mass Spectrom. 2000, 11, 1061–1064. [Google Scholar] [CrossRef][Green Version]
- Hansen, R.E.; Roth, D.; Winther, J.R. Quantifying the global cellular thiol–disulfide status. Proc. Natl. Acad. Sci. USA 2009, 106, 422–427. [Google Scholar] [CrossRef]
- Zhang, S.; Yu, Q.; Sheng, C.; You, J. Gas purge microextraction goupled with stable isotope labeling–liquid chromatography/mass spectrometry for the analysis of bromophenols in aquatic products. J. Agric. Food Chem. 2016, 64, 9452–9458. [Google Scholar] [CrossRef]
- Bruheim, P.; Kvitvang, H.F.N.; Villas-Boas, S.G. Stable isotope coded derivatizing reagents as internal standards in metabolite profiling. J. Chromatogr. A 2013, 1296, 196–203. [Google Scholar] [CrossRef]
- Cooper, G.; Negrusz, A. Clarke′s Analytical Forensic Toxicology; Pharmaceutical Press: London, UK, 2013; pp. 483–489. [Google Scholar]
- Gros, J.; Lavigne, V.; Thibaud, F.; Gammacurta, M.; Moine, V.; Dubourdieu, D.; Darriet, P.; Marchal, A. Toward a molecular understanding of the typicality of chardonnay wines: Identification of powerful aromatic compounds reminiscent of hazelnut. J. Agric. Food Chem. 2017, 65, 1058–1069. [Google Scholar] [CrossRef]
- Block, E.; Batista, V.S.; Matsunami, H.; Zhuang, H.; Ahmed, L. The role of metals in mammalian olfaction of low molecular weight organosulfur compounds. Nat. Prod. Rep. 2017, 34, 529–557. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Ahmed, L.; Zhang, R.; Pan, Y.; Matsunami, H.; Burger, J.L.; Block, E.; Batista, V.S.; Zhuang, H. Smelling sulfur: Copper and silver regulate the response of human odorant receptor OR2T11 to low-molecular-weight thiols. J. Am. Chem. Soc. 2016, 138, 13281–13288. [Google Scholar] [CrossRef] [PubMed]
- Fedrizzi, B.; Versini, G.; Lavagnini, I.; Nicolini, G.; Magno, F. Gas chromatography–mass spectrometry determination of 3-mercaptohexan-1-ol and 3-mercaptohexyl acetate in wine: A comparison of headspace solid phase microextraction and solid phase extraction methods. Anal. Chim. Acta 2007, 596, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Fedrizzi, B.; Versini, G.; Lavagnini, I.; Badocco, D.; Nicolini, G.; Magno, F. Hyphenated gas chromatography–mass spectrometry analysis of 3-mercaptohexan-1-ol and 3-mercaptohexyl acetate in wine: Comparison with results of other sampling procedures via a robust regression. Anal. Chim. Acta 2008, 621, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Mateo-Vivaracho, L.; Cacho, J.; Ferreira, V. Selective preconcentration of volatile mercaptans in small SPE cartridges: Quantitative determination of trace odor-active polyfunctional mercaptans in wine. J. Sep. Sci. 2009, 32, 3845–3853. [Google Scholar] [CrossRef] [PubMed]
- Thibon, C.; Pons, A.; Mouakka, N.; Redon, P.; Méreau, R.; Darriet, P. Comparison of electron and chemical ionization modes for the quantification of thiols and oxidative compounds in white wines by gas chromatography–tandem mass spectrometry. J. Chromatogr. A 2015, 1415, 123–133. [Google Scholar] [CrossRef]
- Floch, M.; Shinkaruk, S.; Darriet, P.; Pons, A. Identification and organoleptic contribution of vanillylthiol in wines. J. Agric. Food Chem. 2016, 64, 1318–1325. [Google Scholar] [CrossRef]
- Gaines, K.K.; Chatham, W.H.; Farwell, S.O. Comparison of the SCD and FPD for HRGC determination of atmospheric sulfur gases. J. High Resolut. Chromatogr. 1990, 13, 489–493. [Google Scholar] [CrossRef]
- Meng, Q.; Kakuta, T.; Sugawara, E. Quantification and odor contribution of volatile thiols in Japanese soy sauce. Food Sci. Technol. Res. 2012, 18, 429–436. [Google Scholar] [CrossRef]
- National Institute of Standards and Technology. Available online: https://www.nist.gov/srd/nist-standard-reference-database-1a-v17 (accessed on 26 July 2019).
- Wiley Registry 11th Edition/NIST 2017 Mass Spectral Library. Available online: https://www.wiley.com/en-us/Wiley+Registry+11th+Edition+NIST+2017+Mass+Spectral+Library-p-9781119412236 (accessed on 26 June 2019).
- Kankolongo Cibaka, M.-L.; Gros, J.; Nizet, S.; Collin, S. Quantitation of selected terpenoids and mercaptans in the dual-purpose hop varieties Amarillo, Citra, Hallertau Blanc, Mosaic, and Sorachi Ace. J. Agric. Food Chem. 2015, 63, 3022–3030. [Google Scholar] [CrossRef]
- Chen, W.; Zhao, Y.; Seefeldt, T.; Guan, X. Determination of thiols and disulfides via HPLC quantification of 5-thio-2-nitrobenzoic acid. J. Pharm. Biomed. Anal. 2008, 48, 1375–1380. [Google Scholar] [CrossRef] [PubMed]
- Mopper, K.; Delmas, D. Trace determination of biological thiols by liquid chromatography and precolumn fluorometric labeling with o-phthalaldehyde. Anal. Chem. 1984, 56, 2557–2560. [Google Scholar] [CrossRef] [PubMed]
- Ercal, N.; Yang, P.; Aykin, N. Determination of biological thiols by high-performance liquid chromatography following derivatization by ThioGlo maleimide reagents. J. Chromatogr. B Biomed. Sci. Appl. 2001, 753, 287–292. [Google Scholar] [CrossRef]
- Taylor, P.J. Matrix effects: The Achilles heel of quantitative high-performance liquid chromatography–electrospray–tandem mass spectrometry. Clin. Biochem. 2005, 38, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Schoenauer, S.; Schieberle, P. Structure–odor activity studies on monoterpenoid mercaptans synthesized by changing the structural motifs of the key food odorant 1-p-menthene-8-thiol. J. Agric. Food Chem. 2016, 64, 3849–3861. [Google Scholar] [CrossRef] [PubMed]
- Chin, S.-T.; Marriott, P.J. Multidimensional gas chromatography beyond simple volatiles separation. Chem. Commun. 2014, 50, 8819–8833. [Google Scholar] [CrossRef]
- Kromidas, S.; Kuss, H.-J. Quantification in LC and GC: A Practical Guide to Good Chromatographic Data; John Wiley & Sons: Hoboken, NJ, USA, 2009; pp. 237–238. [Google Scholar]
- Kruve, A.; Rebane, R.; Kipper, K.; Oldekop, M.-L.; Evard, H.; Herodes, K.; Ravio, P.; Leito, I. Tutorial review on validation of liquid chromatography–mass spectrometry methods: Part I. Anal. Chim. Acta 2015, 870, 29–44. [Google Scholar] [CrossRef]
- Wang, S.; Cyronak, M.; Yang, E. Does a stable isotopically labeled internal standard always correct analyte response?: A matrix effect study on a LC/MS/MS method for the determination of carvedilol enantiomers in human plasma. J. Pharm. Biomed. Anal. 2007, 43, 701–707. [Google Scholar] [CrossRef]
- Roland, A.; Cavelier, F.; Schneider, R. How organic and analytical chemistry contribute to knowledge of the biogenesis of varietal thiols in wine. A review. Flavour Fragance J. 2012, 27, 266–272. [Google Scholar] [CrossRef]
- Reyes-Garcés, N.; Gionfriddo, E.; Gómez-Ríos, G.A.; Alam, M.N.; Boyacı, E.; Bojko, B.; Singh, V.; Grandy, J.; Pawliszyn, J. Advances in solid phase microextraction and perspective on future directions. Anal. Chem. 2018, 90, 302–360. [Google Scholar] [CrossRef]
- Ali, I.; AL-Othman, Z.A.; Nagae, N.; Gaitonde, V.D.; Dutta, K.K. Recent trends in ultra-fast HPLC: New generation superficially porous silica columns. J. Sep. Sci. 2012, 35, 3235–3249. [Google Scholar] [CrossRef] [PubMed]
| Entry No. | Reference | No. of Analytes 1 | Matrix | Sample Amount | Isolation Overview 2 | Major Methodological Parameters 3 | Comments 4 |
|---|---|---|---|---|---|---|---|
| 1 | 1995 [21] | 1 [ID] | Wine | 1000 mL | LLE ⇒ selective extraction [p-HMB] | • LLE × 3 using p-HMB solution • Glutathione added at 20-fold the p-HMB amount | + Reversible tagging allows thiols to be analysed in native form by GC–O + Suitable for thiol screening with GC based methods − Large sample volume needed − High demand for organic solvents − p-HMB is highly toxic − Very time consuming ♦ cheese [26] |
| 2 | 1998 [22] | 5 [QT] | Wine | 500 mL | LLE ⇒ selective extraction [p-HMB] ⇒ strong anion exchange column ⇒ LLE | • 4-Methoxy-2-methyl-2-butane as IS • pH adjustments • Dowex 1X2-100 column • >45 min for column step | |
| 3 | 2003 [25] | 3 [QT] | Wine | 500 mL | LLE ⇒ selective extraction [p-HMB] | • SIDA • Wine protected in ice bath under N2 • LLE for 15 min • Affi-Gel 501 • 1,4-dithio-dl-threitol (DTT) elution | |
| 4 | 2017 [27] | 1 [QT] | Hops | 350 g | LLE ⇒ SAFE ⇒ selective extraction [p-HMB] ⇒ SAFE | • SIDA • Mercurated agarose gel prepared from Affi-Gel 10 • LLE duration > 3 h • SAFE at 40 °C • DTT elution • SAFE to remove DTT | |
| 5 | 2017 [28] | 6 [QT] | Beer, hops | 20 mL, 2 g | LLE ⇒ Ag+ resin based SPE | For beer: • LLE for 15 min • Centrifugation for 15 min • Meta-Sep IC-Ag SPE cartridge • Cartridge reversed • Salted eluate shaken for 15 min • Centrifugation for 15 min | + Novel SPE concept − Multiple extraction steps |
| 6 | 2006 [18] | 2 [QT] | Wine | 10 mL | HS–SPME with automated on-fibre derivatisation [PFBBr] | • PDMS/DVB SPME fibre • Wine bubbled with N2 at 4 °C | + Moderate amounts of sample required + Less solvents needed − Multiple steps for some − Hazardous PFBBr |
| 7 | 2007 [29] | 4 [QT] | Wine | 6 mL | LLE ⇒ derivatisation [PFBBr] | • Four IS • LLE with benzene • Sample bubbled with N2 • 35 min LLE and centrifugation • PFBBr reaction at 4 °C for 40 min | |
| 8 | 2008 [30] | 5 [QT] | Wine | 10 mL | Derivatisation [o-methylhydroxylamine for carbonyl of 4-MSP] ⇒ SPE with in-cartridge derivatisation [PFBBr] | • SIDA • Sample purged with N2 • o-methylhydroxylamine reaction at 55 °C for 45 min • SPE with Bond Elut-ENV | |
| 9 | 2009 [31] | 3 [QT] | Wine | 100 mL | Derivatisation [o-methylhydroxylamine for carbonyl of 4-MSP] ⇒ SPE with derivatisation [PFBBr] ⇒ HS–SPME | • Similar to [30] • SIDA • DVB/CAR/PDMS for 30 min at 100 °C | |
| 10 | 2011 [32] | 1 [QT] | Wine | 200 mL | LLE ⇒ derivatisation [PFBBr] ⇒ HS–SPME | • SIDA • LLE and back extraction with ice-cold aqueous NaOH • PFBBr reaction at room temperature for 20 min • pH adjustment • PDMS/DVB fibre • SPME for 30 min at 80 °C | |
| 11 | 2015 [33] | 3 [QT] | Wine | 40 mL | Simultaneous LLE extraction and derivatisation [PFBBr] ⇒ HS–SPME | • SIDA • pH adjustment • PFBBr reaction and LLE for 10 min at room temperature • PDMS/DVB fibre • SPME for 60 min at 70 °C | |
| 12 | 2013 [19] | 3 [QT] | Wine | 50 mL | Derivatisation [ETP] ⇒ SPE | • SIDA • pH adjustment • ETP reaction for 10 min under stirring • SPE with ENVI-18 | − Poor reaction efficiency with 4-MSP |
| 13 | 2015 [34] | 3 [QT] | Beer, wort, hops | 20 mL | Derivatisation [ETP] and SBSE | • SIDA • pH adjustment • PDMS stir bar • ETP reaction for 10 min at 25 °C • NaOH addition • SBSE for 180 min at 1500 rpm | − Long extraction time |
| 14 | 2014 [35] | 1 [QT] | Wine | 3 mL | Automated derivatisation of 4-MSP carbonyl [o-methylhydroxylamine] and HS–SPME | • SIDA • DVB/CAR/PDMS fibre • SPME for 45 min at 55 °C | + Easy automated extraction approach − Only one analyte assessed |
| 15 | 2015 [36] | 2 [QT] | Wine | 180 mL | LLE ⇒ derivatisation [OPA] | • SIDA • Add potassium metabisulfite and PVPP, stir for 10 min • Centrifugation for 10 min • pH adjustment and sodium borohydride addition • LLE for 20 min • Reaction for 5 min at room temperature | − Large sample volume − Complicated protocol |
| 16 | 2013 [37] | 7 [QT] | Olive oil | 2 g | Single step derivatisation [ebselen] | • 4-Methoxy-α-toluenethiol • Reaction maintained under N2 • 1 min reaction | + Simple and fast extraction – Requires high resolution MS ♦ wine [38], beer [38], brewed coffee [39], roasted coffee [40] |
| 17 | 2015 [7] | 5 [QT] | Wine | 20 mL | Derivatisation [DTDP] ⇒ SPE | • SIDA • Reaction for 30 min at room temperature • Bond Elut C18 SPE cartridge | + Simple extraction + Suitable for multiple thiols + Chiral analysis possible ♦ wines [41,42] |
| 18 | 2014 [43] | 1 [ID] | Beer | 100 µL | Single step stable isotope labelled chemical derivatisation [d0/d7-BQB] | • BQB dried under N2 • Gly-HCl buffer • Reaction for 1 h at 60 °C | + Stable isotope derivatisation applied + Precursor ion scan − Synthesis of reagents required |
| 19 | 2017 [44] | 6 [QT] | Wine | 100 mL | Selective extraction [p-HMB] & SPE ⇒ LLE ⇒ Stable isotope labelled chemical derivatisation [d0/d4-AENM] | • LiChrolut-EN SPE mercurated with p-HMB • Reaction at 40 °C for 10 min | |
| 20 | 2018 [45] | 4 [QT] | Coffee bean, cookies, fried nuts, biscuit | 2 g | GP–MSE ⇒ derivatisation [PIPD] | • Gas purge with N2 • 1.0 mL of syringe loaded with 0.5 mL of MeOH as extraction solvent • Sample heated for 30 min at 190 °C • Derivatisation for 10 min | + No pre-enrichment step – Customised extraction apparatus required – Sample subjected to high temp. – Synthesis of reagent required |
| Entry No. 1 | No. of Thiols | Year | Matrix | Analyte Form 2 | Analytical Instrumentation | Aim 3 | Major Separation Parameters 4 | ME 5 | LOD 6 | RSD 7 (%) | Recovery 8 (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 1 | 1995 [21] | Wine | Free | GC–O, –FPD, –EI–MS | ID | • Multiple columns | – | – | – | – |
| 2 | 5 | 1998 [22] | Wine | Free | GC–EI–MS | QT | • BP20 (50 m × 0.22 mm, 0.25 µm) | – | – | 4–10 | 75–80 |
| 3 | 3 | 2003 [25] | Wine | Free | GC–NCI–ITMS/MS, –AED | QT | • DB WAX (30 m × 0.25 mm, 0.5 µm) for MS • DB WAX (30 m × 0.53, 0.5 µm) for AED | – | ⇔ | <12 | – |
| 4 | 12 | 2006 [12] | Beer | Free | GC–O, –PFPD, –FID, –EI–MS | ID | • CP-Sil 5 CB (50 m × 0.32 mm, 1.2 µm) or FFAP CB (25 m × 0.32 mm, 0.3 µm) for O, FID, PFPD • DB CP-Sil 5 CB-MS (50 m × 0.32 mm, 1.2 µm) for MS | – | – | – | – |
| 5 | 2 | 2006 [18] | Wine | Deriv. | GC–ECD,–NCI–MS | QT | • VF-5ms (20 m × 0.15 mm, 0.15 µm) | Y | < | 10–20 | – |
| 6 | 2 | 2007 [60] | Wine | Free | GC–EI–MS | QT | • INNOwax (30 m × 0.32 mm, 0.25 µm) connected to HP-1 (10 m × 0.32 mm, 0.25 µm) | N | > | – | ≈100 |
| 7 | 4 | 2007 [29] | Wine | Deriv. | GC–CI–MS | QT | • VF-5ms (20 m × 0.15 mm, 0.15 µm) | Y/N | < | 10–17 | – |
| 8 | 5 | 2008 [30] | Wine | Deriv. | GC–CI–MS | QT | • VF-5ms (20 m × 0.15 mm, 0.15 µm) | Y | < | 1–20 | 47–123 |
| 9 | 2 | 2008 [61] | Wine | Free | GC–EI–MS | QT | • TR-5MS (30 m × 0.25 mm, 0.25 µm) | N | ⇔ | – | – |
| 10 | 1 | 2008 [26] | Cheese | Free | GC–O, –PFPD, –EI–MS | ID, QT | • DB-XLB (30 m × 0.25 mm, 0. 5 µm) • HP-5ms (30 m × 0.25 mm, 0.25 µm) | – | – | – | >70 |
| 11 | 5 | 2009 [62] | Wine | Free | GC–EI–ITMS | QT | • DB-WAXetr (60 m × 0.25 mm, 0.25 µm) | – | ⇔ | 6.5–12.3 | 28–123 |
| 12 | 3 | 2009 [31] | Wine | Deriv. | GC–CI–MS | QT | • Optima Wax (30 m × 0.25 mm, 0.25 µm) | N | < | <10 | – |
| 13 | 1 | 2011 [32] | Wine | Deriv. | GC–EI–MS | QT | • DB-5ms (60 m × 0.25 mm, 0.25 µm) | N | < | <2.5 | – |
| 14 | 3 | 2013 [19] | Wine | Deriv. | GC–EI–MS | QT | • HP-INNOwax (60 m × 0.25 mm, 0.25 µm) | N | ⇔ | 1.9–17 | 94–112 |
| 15 | 7 | 2013 [37] | Olive oil | Deriv. | HPLC–ESI–Orbitrap MS | QT | • Luna C18 (150 mm × 2.1 mm, 5 µm) • A: 10 mM ammonium formate in water • B: 10 mM ammonium formate in MeOH | Y | < | ≈13 | 79–20 |
| 16 | 1 | 2014 [43] | Beer | Deriv. | LC–ESI–MS/MS LC–Q–TOF | ID | • VP-ODS column (150 mm × 2.0 mm, 5 μm) • A: 0.1% formic acid in water • B: 0.1% formic acid in MeOH | – | – | – | – |
| 17 | 1 | 2014 [35] | Wine | Deriv. | GC–EI–MS/MS | QT | • DB-WAX (60 m × 0.25 mm, 0.25 µm) | N | < | 15 | 99–102 |
| 18 | 3 | 2015 [33] | Wine | Deriv. | GC–EI–MS | QT | • DB-FFAP (30 m × 0.25 mm, 0.25 µm) | N | ⇔ | 5–11 | 90–109 |
| 19 | 5 | 2015 [63] | Wine | Free | GC–MS/MS(QqQ) | QT | • BP20 (2 m × 0.25 mm, 0.22 µm) connected to ZB-1ms (60 m × 0.25 mm, 1 µm) | N | < | 5–18 | 86–110 |
| 20 | 3 | 2015 [34] | Beer, hops, wort | Deriv. | GC–EI–Q–TOF–MS/SCD GC–EI–MS/MS(QqQ) | ID, QT | • DB-WAX (30 m × 0.25 mm, 0.25 µm) • DB-WAX (15 m × 0.25 mm, 0.25 µm) | – | < | 1.3–7.2 | 99–101 |
| 21 | 2 | 2015 [36] | Wine | Deriv. | UHPLC–ESI–MS/MS(QqQ) | QT | • Acquity UPLC BEH C18 (100 mm × 2.1 mm, 1.7 μm) • A: 10 mM ammonium acetate in water • B: MeOH:MeCN:isopropanol (49:49:2) | Y/N | < | 0.6–11.9 | 98–128 |
| 22 | 5 | 2015 [7] | Wine | Deriv. | HPLC–ESI– MS/MS(QqQ) | QT | • Alltima C18 (250 mm × 2.1 mm, 5 μm) • A: 0.5% aqueous formic acid • B: 0.5% formic acid in acetonitrile | – | ⇔ | <8.5 | 94–103 |
| 23 | 1 | 2016 [64] | Wine | Free | GC–EI–MS/MS (QqQ) | QT | • ZB-1ms (60 m × 0.25 mm, 1 µm) | – | < | 9 | – |
| 24 | 2 | 2017 [57] | Wine | Free | GC–EI–MS/MS (QqQ) | ID | • ZB-1ms (60 m × 0.25 mm, 1 µm) | – | – | – | – |
| 25 | 6 | 2017 [28] | Beer, hops | Free | GC–EI–MS/MS(QqQ) | QT | • InertCap Pure-WAX (30 m × 0.25 mm, 0.25 µm) | – | < | 2.8–8.4 | 74–113 |
| 26 | 6 | 2017 [44] | Wine | Deriv. | UHPLC–ESI–MS/MS(QqQ) | QT | • Eclipse Plus C18 column (50 mm × 2.1 mm, 1.8 µm) • A: 0.1% formic acid in 5% aqueous MeCN • B: 0.1% formic acid in MeCN | N | ⇔ | ≤3.5 | ≥78 |
| 27 | 1 | 2017 [27] | Hops | Free | GC×GC–Q–TOF Heart cut 2D-GC–CI–ITMS | QT | • Q–TOF: 1st GC: DB-FFAP (30 m × 0.25 mm, 0.25 µm), 2nd GC: DB-5 (2 m × 0.15 mm, 0.30 µm) • ITMS: 1st GC: FFAP (30 m × 0.32 mm, 0.25 µm), cool-on-column injection; 2nd GC: DB-1701 (30 m × 0.25 mm, 0.25 µm) | N | < | <15 | 109 ± 6 104 ± 4 |
| 28 | 2 | 2018 [41] | Wine | Deriv. | HPLC–ESI–MS/MS(QqQ) | QT | • Polysaccharide Amylose-1 column (150 mm × 2.0 mm, 3 µm) • A: 5 mM aqueous ammonium bicarbonate • B: MeCN | Y/N | < | <8 | 90–110 |
| 29 | 8 | 2018 [45] | Coffee bean, cookie, fried nut, biscuit | Deriv. | HPLC–FLD-APCI-MS | QT | • Eclipse XDB-C18 column (150 mm × 4.6 mm, 5 µm) • A: 30% aq. MeCN • B: MeCN | N | > | 4.98 | 86–97 |
| 30 | 4 | 2018 [42] | Wine | Deriv. | UPC2–MS/MS(QqQ) | QT | • BEH 2-EP column (100 mm × 3 mm, 1.7 µm) • Solvent: CO2 and MeOH | N | < | 8–18 | 94–119 |
| 31 | 11 | 2019 [23] | Fruit, wine | Free | GC–O, –FID, –SCD, GC×GC–Q–TOF | ID, QT | • DB-FFAP (30 m × 0.32 mm, 0.25 µm) • 1st GC: DB-FFAP (30 m × 0.25 mm, 0.25 µm), 2nd GC: DB-17ms (2 m × 0.18 mm, 0.18 µm) | – | – | – | – |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, L.; Capone, D.L.; Jeffery, D.W. Analysis of Potent Odour-Active Volatile Thiols in Foods and Beverages with a Focus on Wine. Molecules 2019, 24, 2472. https://doi.org/10.3390/molecules24132472
Chen L, Capone DL, Jeffery DW. Analysis of Potent Odour-Active Volatile Thiols in Foods and Beverages with a Focus on Wine. Molecules. 2019; 24(13):2472. https://doi.org/10.3390/molecules24132472
Chicago/Turabian StyleChen, Liang, Dimitra L. Capone, and David W. Jeffery. 2019. "Analysis of Potent Odour-Active Volatile Thiols in Foods and Beverages with a Focus on Wine" Molecules 24, no. 13: 2472. https://doi.org/10.3390/molecules24132472
APA StyleChen, L., Capone, D. L., & Jeffery, D. W. (2019). Analysis of Potent Odour-Active Volatile Thiols in Foods and Beverages with a Focus on Wine. Molecules, 24(13), 2472. https://doi.org/10.3390/molecules24132472






