Mesoporous Silica-Based Materials for Electronics-Oriented Applications
Abstract
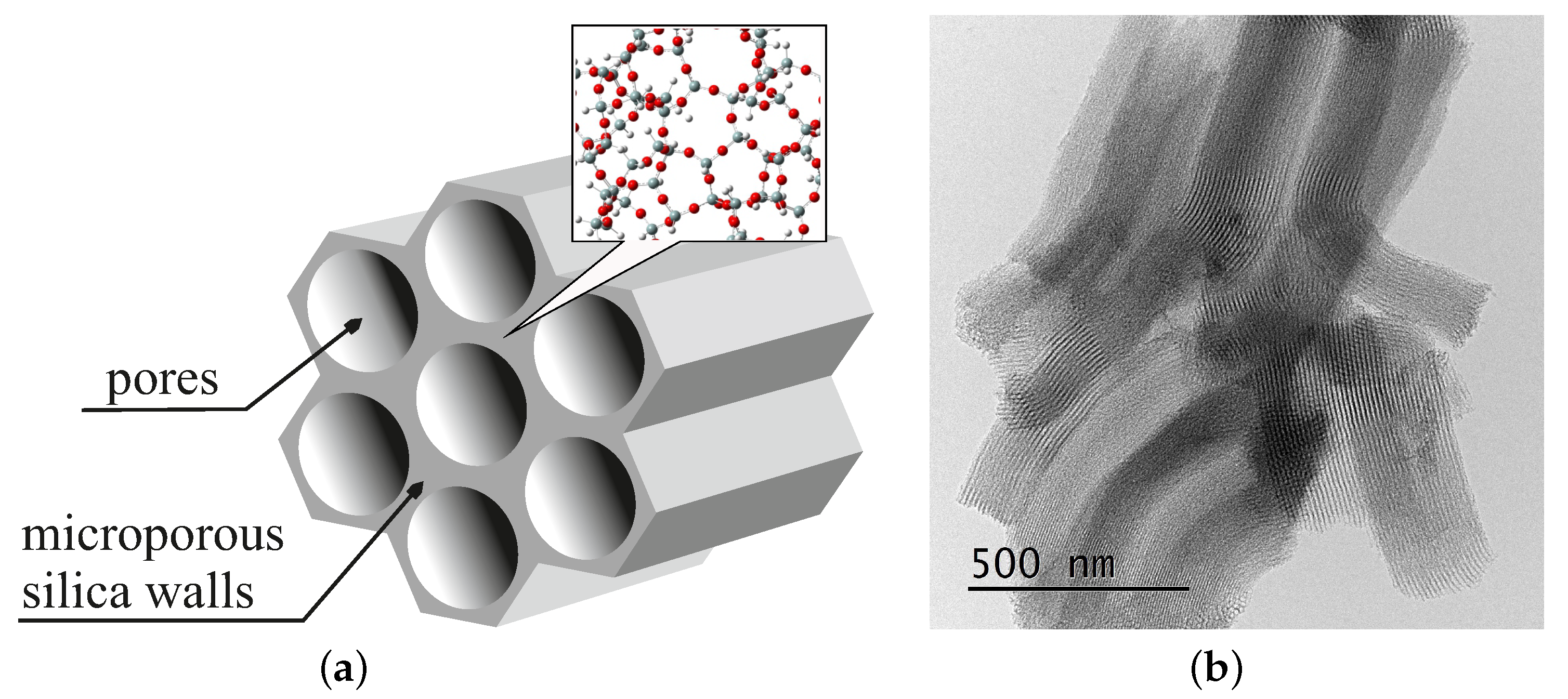
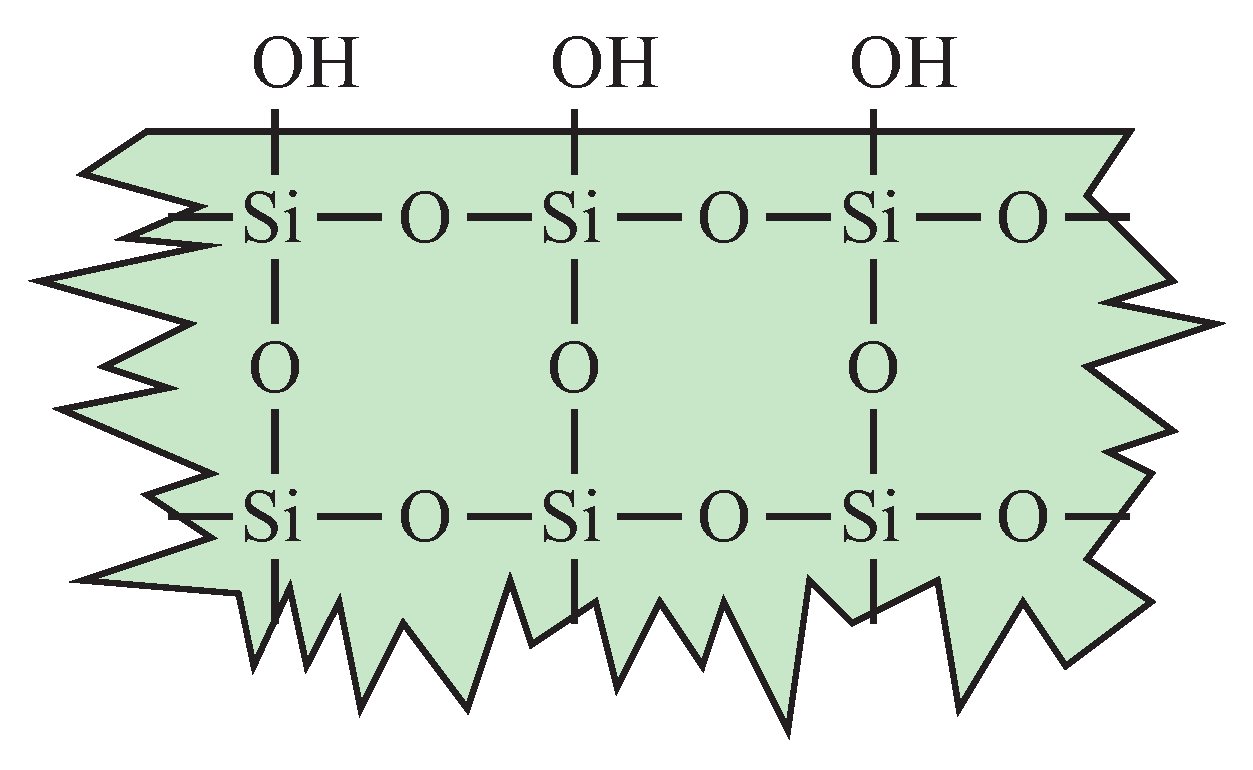
1. Introduction
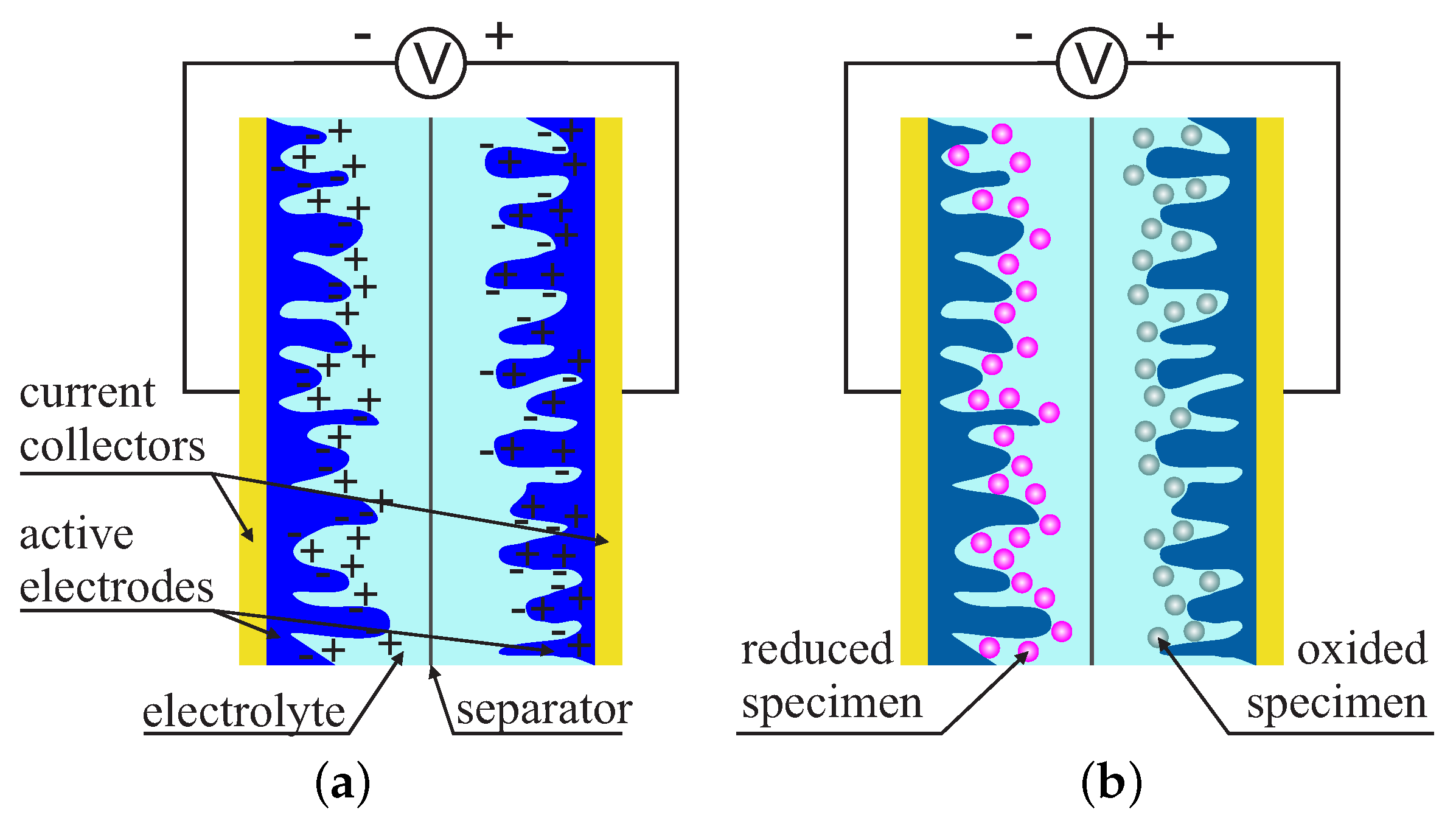
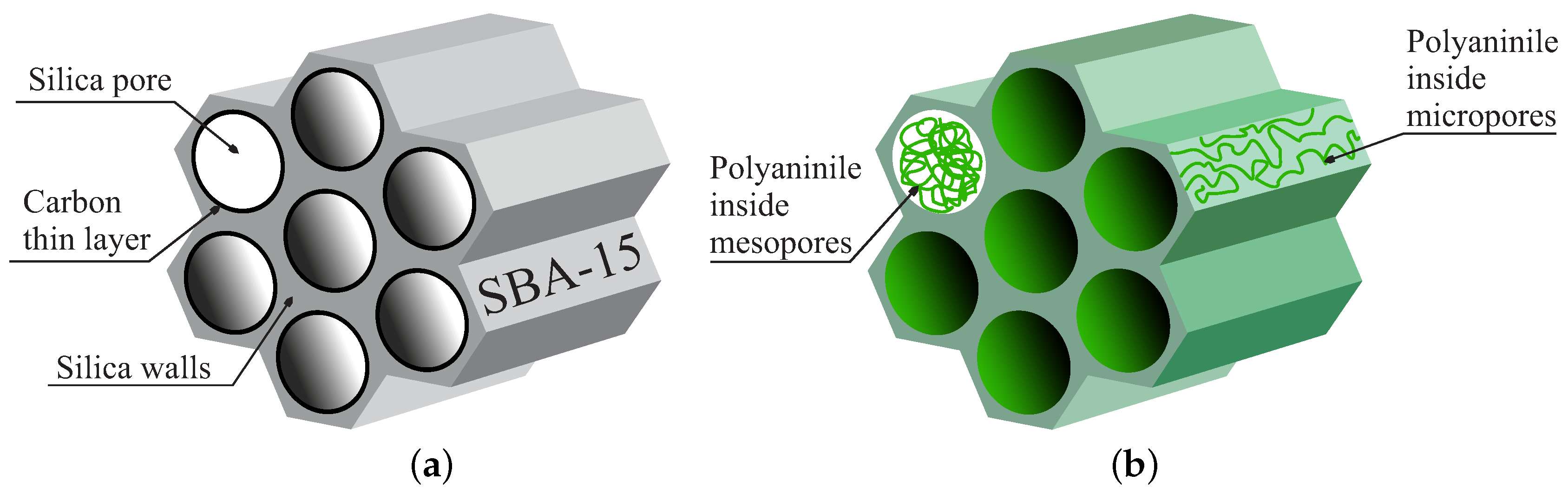
2. Supercapacitors
3. Low-k Dielectrics for Electronic Devices
4. Redox-Active Silica-Based Organic-Inorganic Hybrids
5. Perspectives
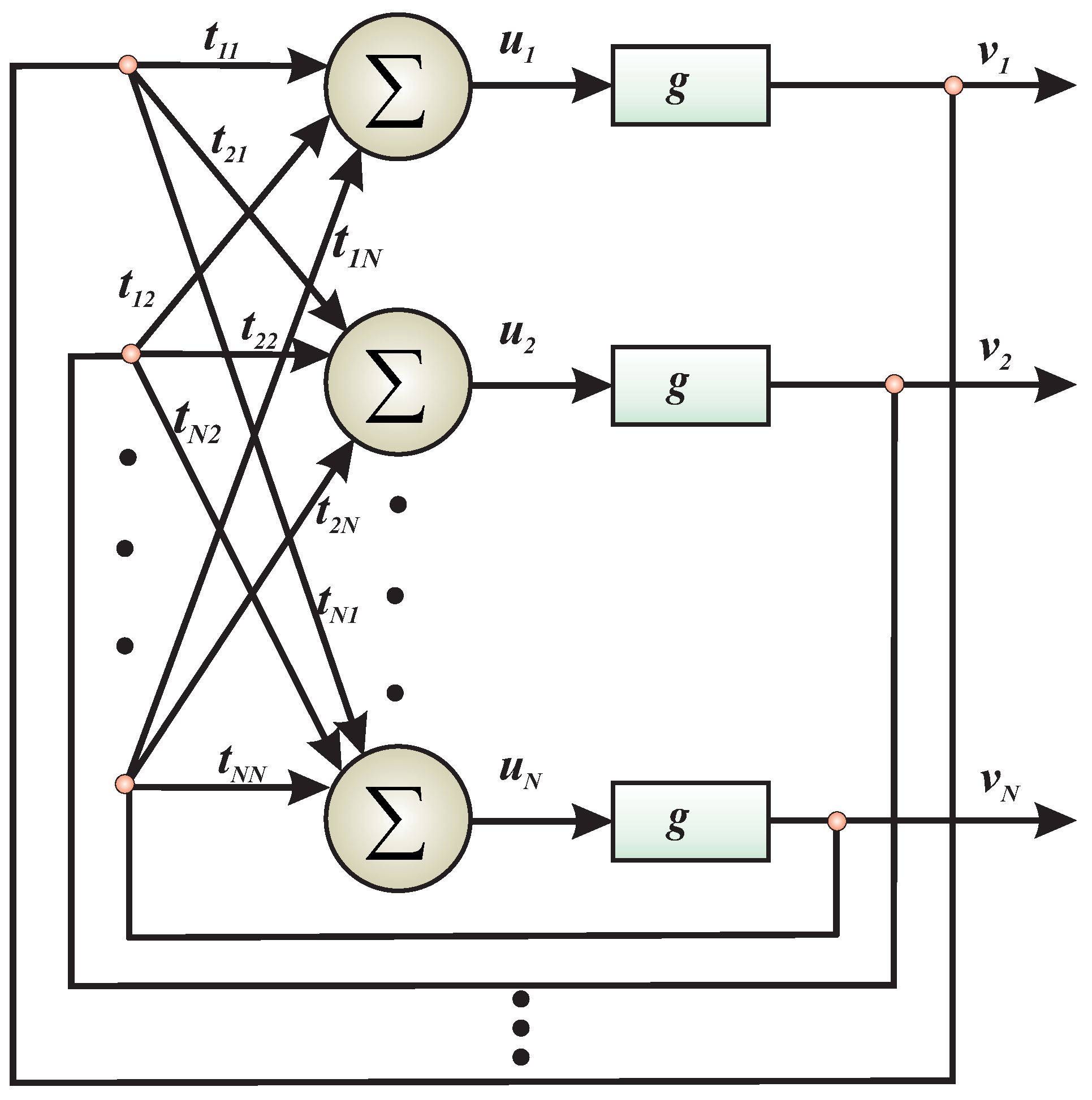
- The problem, must be formulated in such a way that each syntactically correct solution corresponds to the local energy minima.
- A lower energy minimum corresponds to s better solution.
- The best solution corresponds to the global minimum.
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| TEOS | tetraethoxy silane |
| TMOS | tetramethoxy silane |
| TMES | trimethylethoxysilane |
| MTMS | methyltrimethoxy silane |
| OTMS | octyltrimethoxy silane |
| APTMS | 3-aminopropyl trimethoxy silane |
| Me | methyl units |
| Et | ethyl units |
| CTAB | |
| CTACl | |
| Brij76 | |
| Brij56 | |
| Brij30 | |
| Pluronic F127 | poly(ethylene oxide)-poly(propylene oxide)-poly(ethylene oxide) triblock copolymer: |
| PEO106-PPO70-PEO106 | |
| Pluronic P123 | poly(ethylene oxide)-poly(propylene oxide)-poly(ethylene oxide) triblock copolymer: |
| PEO20-PPO70-PEO20 | |
| ALD | atomic layer deposition |
| OTES-POS | octa(triethoxysilylethyl)polyhedral oligomeric silsesquioxane |
| MFM | magnetic force microscopy |
| XMCD | X-ray magnetic circular dichroism |
| SMM | single-molecule magnet |
| Mn12-ac16 | |
| Mn12-st | derivative of Mn12 containing strearic acid ligands |
| MSF | multi-step functionalization procedure |
References
- Horowitz, P.; Hill, W. The Art of Electronics; Cambridge University Press: Cambridge, MA, USA, 1989. [Google Scholar]
- Tietze, U.; Schenk, C.; Schmid, E. Advanced Electronic Circuits; Springer: New York, NY, USA, 1991. [Google Scholar]
- Tietze, U.; Schenk, C.; Gamm, E. Electronic Circuits: Handbook for Design and Application; Springer: New York, NY, USA, 2015. [Google Scholar]
- De Forest, L. The audion: A new receiver for wireless telegraphy. Proc. Am. Inst. Electr. Eng. 1906, 25, 719–747. [Google Scholar] [CrossRef]
- Boot, H.; Randall, J. The cavity magnetron. J. Inst. Electr. Eng. Part IIIA Radiolocat. 1946, 93, 928–938. [Google Scholar] [CrossRef]
- Blanchard, Y.; Galati, G.; Van Genderen, P. The cavity magnetron: Not just a british invention [Historical Corner]. IEEE Antennas Propag. Mag. 2013, 55, 244–254. [Google Scholar] [CrossRef]
- Wolff, C. Radar Tutorial (Book 1): Radar Basics. Radartutorial Webpage. Available online: www.radartutorial.eu (accessed on 28 June 2019).
- Copeland, J.; Haeff, A.A. The true history of the traveling wave tube. IEEE Spectr. 2015, 52, 38–43. [Google Scholar] [CrossRef]
- Gilmour, A. Klystrons, Traveling Wave Tubes, Magnetrons, Crossed-Field Amplifiers, and Gyrotrons; Artech House: London, UK, 2011. [Google Scholar]
- Gilmour, A. Principles of Traveling Wave Tubes; Artech House: London, UK, 1994. [Google Scholar]
- Vardalas, J. Twists and Turns in the Development of the Transistor. In IEEE-USA Today’s Engineer; IEEE: Piscataway, NJ, USA, 2003. [Google Scholar]
- Riordan, M.; Hoddeson, L.; Herring, C. The invention of the transistor. In More Things in Heaven and Earth; Springer: New York, NY, USA, 1999; pp. 563–578. [Google Scholar]
- Brinkman, W.F.; Haggan, D.E.; Troutman, W.W. A history of the invention of the transistor and where it will lead us. IEEE J. Solid-State Circuits 1997, 32, 1858–1865. [Google Scholar] [CrossRef]
- Gray, P.R.; Hurst, P.; Meyer, R.G.; Lewis, S. Analysis and Design of Analog Integrated Circuits; Wiley: New York, NY, USA, 2001. [Google Scholar]
- Schaller, R.R. Moore’s law: Past, present and future. IEEE Spectr. 1997, 34, 52–59. [Google Scholar] [CrossRef]
- Meindl, J.D. Beyond Moore’s law: The interconnect era. Comput. Sci. Eng. 2003, 5, 20–24. [Google Scholar] [CrossRef]
- Zalasiński, M.; Cpałka, K. Novel algorithm for the on-line signature verification. In International Conference on Artificial Intelligence and Soft Computing; Springer: Berlin, Germany, 2012; pp. 362–367. [Google Scholar]
- Cpalka, K.; Łapa, K.; Przybył, A. Genetic Programming Algorithm for Designing of Control Systems. Inf. Technol. Control 2018, 47, 668–683. [Google Scholar] [CrossRef]
- Wu, X.; Lee, I.; Dong, Q.; Yang, K.; Kim, D.; Wang, J.; Peng, Y.; Zhang, Y.; Saliganc, M.; Yasuda, M.; et al. A 0.04 MM 3 16NW Wireless and Batteryless Sensor System with Integrated Cortex-M0+ Processor and Optical Communication for Cellular Temperature Measurement. In Proceedings of the 2018 IEEE Symposium on VLSI Circuits, Honolulu, HI, USA, 18–22 June 2018; pp. 191–192. [Google Scholar]
- Salahuddin, S.; Ni, K.; Datta, S. The era of hyper-scaling in electronics. Nat. Electron. 2018, 1, 442. [Google Scholar] [CrossRef]
- Deleonibus, S. Electronic Devices Architectures for the NANO-CMOS Era; Pan Stanford Publishing: Singapore, 2009. [Google Scholar]
- Waldrop, M.M. The chips are down for Moore’s law. Nat. News 2016, 530, 144. [Google Scholar] [CrossRef]
- Beck, J.S.; Vartuli, J.; Roth, W.J.; Leonowicz, M.; Kresge, C.; Schmitt, K.; Chu, C.; Olson, D.H.; Sheppard, E.; McCullen, S.; et al. A new family of mesoporous molecular sieves prepared with liquid crystal templates. J. Am. Chem. Soc. 1992, 114, 10834–10843. [Google Scholar] [CrossRef]
- Chen, C.Y.; Li, H.X.; Davis, M.E. Studies on mesoporous materials: I. Synthesis and characterization of MCM-41. Microporous Mater. 1993, 2, 17–26. [Google Scholar] [CrossRef]
- Jehng, J.M.; Tung, W.C.; Kuo, C.H. The formation mechanisms of multi-wall carbon nanotubes over the Ni modified MCM-41 catalysts. J. Porous Mater. 2008, 15, 43–51. [Google Scholar] [CrossRef]
- Zhao, D.; Feng, J.; Huo, Q.; Melosh, N.; Fredrickson, G.H.; Chmelka, B.F.; Stucky, G.D. Triblock copolymer syntheses of mesoporous silica with periodic 50 to 300 angstrom pores. Science 1998, 279, 548–552. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Huo, Q.; Feng, J.; Chmelka, B.F.; Stucky, G.D. Nonionic triblock and star diblock copolymer and oligomeric surfactant syntheses of highly ordered, hydrothermally stable, mesoporous silica structures. J. Am. Chem. Soc. 1998, 120, 6024–6036. [Google Scholar] [CrossRef]
- Xu, R.; Pang, W.; Yu, J.; Huo, Q.; Chen, J. Chemistry of Zeolites and Related Porous Materials: Synthesis and Structure; John Wiley & Sons: Singapore, 2009. [Google Scholar]
- Imperor-Clerc, M.; Davidson, P.; Davidson, A. Existence of a microporous corona around the mesopores of silica-based SBA-15 materials templated by triblock copolymers. J. Am. Chem. Soc. 2000, 122, 11925–11933. [Google Scholar] [CrossRef]
- Ryoo, R.; Ko, C.H.; Kruk, M.; Antochshuk, V.; Jaroniec, M. Block-copolymer-templated ordered mesoporous silica: Array of uniform mesopores or mesopore- micropore network? J. Phys. Chem. B 2000, 104, 11465–11471. [Google Scholar] [CrossRef]
- Dos Santos, S.M.L.; Nogueira, K.A.B.; de Souza Gama, M.; Lima, J.D.F.; da Silva Júnior, I.J.; de Azevedo, D.C.S. Synthesis and characterization of ordered mesoporous silica (SBA-15 and SBA-16) for adsorption of biomolecules. Microporous Mesoporous Mater. 2013, 180, 284–292. [Google Scholar] [CrossRef]
- Thielemann, J.P.; Girgsdies, F.; Schlögl, R.; Hess, C. Pore structure and surface area of silica SBA-15: Influence of washing and scale-up. Beilstein J. Nanotechnol. 2011, 2, 110–118. [Google Scholar] [CrossRef]
- Kruk, M.; Jaroniec, M.; Ko, C.H.; Ryoo, R. Characterization of the porous structure of SBA-15. Chem. Mater. 2000, 12, 1961–1968. [Google Scholar] [CrossRef]
- Guari, Y.; Thieuleux, C.; Mehdi, A.; Reyé, C.; Corriu, R.J.; Gomez-Gallardo, S.; Philippot, K.; Chaudret, B. In situ formation of gold nanoparticles within thiol functionalized HMS-C16 and SBA-15 type materials via an organometallic two-step approach. Chem. Mater. 2003, 15, 2017–2024. [Google Scholar] [CrossRef]
- Maria Chong, A.; Zhao, X. Functionalization of SBA-15 with APTES and characterization of functionalized materials. J. Phys. Chem. B 2003, 107, 12650–12657. [Google Scholar] [CrossRef]
- Boury, B.; Corriu, R. Auto-Organization in Sol-Gel Type Polycondensation: A Door to the Nanosciences. Chem. Rec. 2003, 3, 120–132. [Google Scholar] [CrossRef] [PubMed]
- Yeh, Y.Q.; Lin, H.P.; Tang, C.Y.; Mou, C.Y. Mesoporous silica SBA-15 sheet with perpendicular nanochannels. J. Colloid Interface Sci. 2011, 362, 354–366. [Google Scholar] [CrossRef] [PubMed]
- Johansson, E.M.; Córdoba, J.M.; Odén, M. Synthesis and characterization of large mesoporous silica SBA-15 sheets with ordered accessible 18 nm pores. Mater. Lett. 2009, 63, 2129–2131. [Google Scholar] [CrossRef]
- Hao, N.; Tang, F.; Li, L. MCM-41 mesoporous silica sheet with ordered perpendicular nanochannels for protein delivery and the assembly of Ag nanoparticles in catalytic applications. Microporous Mesoporous Mater. 2015, 218, 223–227. [Google Scholar] [CrossRef]
- Walcarius, A.; Sibottier, E.; Etienne, M.; Ghanbaja, J. Electrochemically assisted self-assembly of mesoporous silica thin films. Nat. Mater. 2007, 6, 602. [Google Scholar] [CrossRef] [PubMed]
- Bardeau, J.F.; Gourbil, A.; Dutreilh-Colas, M.; Dourdain, S.; Mehdi, A.; Gibaud, A. X-ray reflectivity study of acid–base post-synthesis treatments of mesoporous thin films templated by P123. Thin Solid Film. 2006, 495, 191–196. [Google Scholar] [CrossRef]
- Teng, Z.; Zheng, G.; Dou, Y.; Li, W.; Mou, C.Y.; Zhang, X.; Asiri, A.M.; Zhao, D. Highly ordered mesoporous silica films with perpendicular mesochannels by a simple stöber-solution growth approach. Angew. Chem. Int. Ed. 2012, 51, 2173–2177. [Google Scholar] [CrossRef]
- Lu, M. Supercapacitors: Materials, Systems, and Applications; John Wiley & Sons: Singapore, 2013. [Google Scholar]
- Yu, A.; Chabot, V.; Zhang, J. Electrochemical Supercapacitors for Energy Storage and Delivery: Fundamentals and Applications; CRC Press: New York, NY, USA, 2013. [Google Scholar]
- Conway, B.E. Electrochemical Supercapacitors: Scientific Fundamentals and Technological Applications; Springer Science & Business Media: New York, NY, USA, 2013. [Google Scholar]
- Moussa, M.; El-Kady, M.F.; Zhao, Z.; Majewski, P.; Ma, J. Recent progress and performance evaluation for polyaniline/graphene nanocomposites as supercapacitor electrodes. Nanotechnology 2016, 27, 442001. [Google Scholar] [CrossRef]
- Jiang, Y.; Liu, J. Definitions of Pseudocapacitive Materials: A Brief Review. Energy Environ. Mater. 2019, 2, 30–37. [Google Scholar] [CrossRef]
- Lu, W.; Dai, L. Carbon nanotube supercapacitors. In Carbon Nanotubes; IntechOpen: London, UK, 2010. [Google Scholar]
- Ma, W.; Chen, S.; Yang, S.; Chen, W.; Weng, W.; Zhu, M. Bottom-up fabrication of activated carbon fiber for all-solid-state supercapacitor with excellent electrochemical performance. ACS Appl. Mater. Interfaces 2016, 8, 14622–14627. [Google Scholar] [CrossRef] [PubMed]
- Divyashree, A.; Hegde, G. Activated carbon nanospheres derived from bio-waste materials for supercapacitor applications—A review. RSC Adv. 2015, 5, 88339–88352. [Google Scholar]
- Du, C.; Pan, N. High power density supercapacitor electrodes of carbon nanotube films by electrophoretic deposition. Nanotechnology 2006, 17, 5314. [Google Scholar] [CrossRef]
- Du, C.; Yeh, J.; Pan, N. High power density supercapacitors using locally aligned carbon nanotube electrodes. Nanotechnology 2005, 16, 350. [Google Scholar] [CrossRef]
- Du, C.; Pan, N. Carbon nanotube-based supercapacitors. Nanotechnol. Law Bus. 2007, 4, 3. [Google Scholar]
- Lee, H.M.; Lee, K.; Kim, C.K. Electrodeposition of manganese-nickel oxide films on a graphite sheet for electrochemical capacitor applications. Materials 2014, 7, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Liew, C.W.; Ramesh, S. Comparing triflate and hexafluorophosphate anions of ionic liquids in polymer electrolytes for supercapacitor applications. Materials 2014, 7, 4019–4033. [Google Scholar] [CrossRef]
- Yun, Y.S.; Im, C.; Park, H.H.; Hwang, I.; Tak, Y.; Jin, H.J. Hierarchically porous carbon nanofibers containing numerous heteroatoms for supercapacitors. J. Power Sources 2013, 234, 285–291. [Google Scholar] [CrossRef]
- Yang, X.; Li, Z.; Zhi, J.; Ma, J.; Hu, A. Synthesis of Ultrathin Mesoporous Carbon through Bergman Cyclization of Enediyne Self-Assembled Monolayers in SBA-15. Langmuir 2010, 26, 11244–11248. [Google Scholar] [CrossRef]
- Zhi, J.; Song, D.; Li, Z.; Lei, X.; Hu, A. Palladium nanoparticles in carbon thin film-lined SBA-15 nanoreactors: Efficient heterogeneous catalysts for Suzuki–Miyaura cross coupling reaction in aqueous media. Chem. Commun. 2011, 47, 10707–10709. [Google Scholar] [CrossRef] [PubMed]
- Zhi, J.; Wang, Y.; Deng, S.; Hu, A. Study on the relation between pore size and supercapacitance in mesoporous carbon electrodes with silica-supported carbon nanomembranes. RSC Adv. 2014, 4, 40296–40300. [Google Scholar] [CrossRef]
- Zhi, J.; Deng, S.; Wang, Y.; Hu, A. Highly Ordered Metal Oxide Nanorods inside Mesoporous Silica Supported Carbon Nanomembranes: High Performance Electrode Materials for Symmetrical Supercapacitor Devices. J. Phys. Chem. C 2015, 119, 8530–8536. [Google Scholar] [CrossRef]
- Wang, W.; Shan, W.; Ru, H.; Wu, N. A facile and versatile partitioned cooperative self-assembly process to prepare SBA-15s with larger mesopores, high microporosity and tunable particle sizes. J. Mater. Chem. 2011, 21, 12059–12067. [Google Scholar] [CrossRef]
- Fulvio, P.F.; Pikus, S.; Jaroniec, M. Tailoring properties of SBA-15 materials by controlling conditions of hydrothermal synthesis. J. Mater. Chem. 2005, 15, 5049–5053. [Google Scholar] [CrossRef]
- Numao, S.; Judai, K.; Nishijo, J.; Mizuuchi, K.; Nishi, N. Synthesis and characterization of mesoporous carbon nano-dendrites with graphitic ultra-thin walls and their application to supercapacitor electrodes. Carbon 2009, 47, 306–312. [Google Scholar] [CrossRef]
- Huang, Y.; Li, Y.; Hu, Z.; Wei, G.; Guo, J.; Liu, J. A carbon modified MnO2 nanosheet array as a stable high-capacitance supercapacitor electrode. J. Mater. Chem. A 2013, 1, 9809–9813. [Google Scholar] [CrossRef]
- Mu, J.; Chen, B.; Guo, Z.; Zhang, M.; Zhang, Z.; Shao, C.; Liu, Y. Tin oxide (SnO2) nanoparticles/electrospun carbon nanofibers (CNFs) heterostructures: Controlled fabrication and high capacitive behavior. J. Colloid Interface Sci. 2011, 356, 706–712. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Sarkar, D.; Khan, G.G.; Mandal, K. Unique hydrogenated Ni/NiO core/shell 1D nano-heterostructures with superior electrochemical performance as supercapacitors. J. Mater. Chem. A 2013, 1, 12759–12767. [Google Scholar] [CrossRef]
- Hu, Z.; Zu, L.; Jiang, Y.; Lian, H.; Liu, Y.; Wang, X.; Cui, X. High performance nanocomposite electrodes of mesoporous silica platelet-polyaniline synthesized via impregnation polymerization. Polym. Compos. 2017, 38, 1616–1623. [Google Scholar] [CrossRef]
- Silva, R.; Asefa, T. Noble metal-free oxidative electrocatalysts: Polyaniline and Co(II)-polyaniline nanostructures hosted in nanoporous silica. Adv. Mater. 2012, 24, 1878–1883. [Google Scholar] [CrossRef] [PubMed]
- Javed, M.; Abbas, S.M.; Siddiq, M.; Han, D.; Niu, L. Mesoporous silica wrapped with graphene oxide-conducting PANI nanowires as a novel hybrid electrode for supercapacitor. J. Phys. Chem. Solids 2018, 113, 220–228. [Google Scholar] [CrossRef]
- Wang, Y.G.; Xia, Y.Y. Electrochemical capacitance characterization of NiO with ordered mesoporous structure synthesized by template SBA-15. Electrochim. Acta 2006, 51, 3223–3227. [Google Scholar] [CrossRef]
- Ghimbeu, C.M.; Malak-Polaczyk, A.; Frackowiak, E.; Vix-Guterl, C. Template-derived high surface area λ-MnO 2 for supercapacitor applications. J. Appl. Electrochem. 2014, 44, 123–132. [Google Scholar] [CrossRef]
- Jun, S.; Joo, S.H.; Ryoo, R.; Kruk, M.; Jaroniec, M.; Liu, Z.; Ohsuna, T.; Terasaki, O. Synthesis of New, Nanoporous Carbon with Hexagonally Ordered Mesostructure. J. Am. Chem. Soc. 2000, 122, 10712–10713. [Google Scholar] [CrossRef]
- Wang, D.W.; Feng, L.I.; Min LI, U.; Cheng, H.M. Improved capacitance of SBA-15 templated mesoporous carbons after modification with nitric acid oxidation. New Carbon Mater. 2007, 22, 307–314. [Google Scholar] [CrossRef]
- Singh, D.K.; Krishna, K.S.; Harish, S.; Sampath, S.; Eswaramoorthy, M. No More HF: Teflon-Assisted Ultrafast Removal of Silica to Generate High-Surface-Area Mesostructured Carbon for Enhanced CO2 Capture and Supercapacitor Performance. Angew. Chem. Int. Ed. 2016, 55, 2032–2036. [Google Scholar] [CrossRef]
- Zhi, J.; Deng, S.; Zhang, Y.; Wang, Y.; Hu, A. Embedding CO3O4 nanoparticles in SBA-15 supported carbon nanomembrane for advanced supercapacitor materials. J. Mater. Chem. A 2013, 1, 3171–3176. [Google Scholar] [CrossRef]
- Huang, H.S.; Chang, K.H.; Suzuki, N.; Yamauchi, Y.; Hu, C.C.; Wu, K.C.W. Evaporation-Induced Coating of Hydrous Ruthenium Oxide on Mesoporous Silica Nanoparticles to Develop High-Performance Supercapacitors. Small 2013, 9, 2520–2526. [Google Scholar] [CrossRef]
- Chiang, Y.D.; Lian, H.Y.; Leo, S.Y.; Wang, S.G.; Yamauchi, Y.; Wu, K.C.W. Controlling particle size and structural properties of mesoporous silica nanoparticles using the Taguchi method. J. Phys. Chem. C 2011, 115, 13158–13165. [Google Scholar] [CrossRef]
- Jonscher, A.K. Dielectric relaxation in solids. J. Phys. D Appl. Phys. 1999, 32, R57. [Google Scholar] [CrossRef]
- Kremer, F.; Schönhals, A. Broadband Dielectric Spectroscopy; Springer Science & Business Media: New York, NY, USA, 2012. [Google Scholar]
- Maier, G. Low dielectric constant polymers for microelectronics. Prog. Polym. Sci. 2001, 26, 3–65. [Google Scholar] [CrossRef]
- Ho, P.S.; Leu, J. Low Dielectric Constant Materials for IC Applications; Springer Science & Business Media: New York, NY, USA, 2003; Volume 9. [Google Scholar]
- Croes, K.; Pantouvaki, M.; Carbonell, L.; Zhao, L.; Beyer, G.; Tőkei, Z. Comparison between intrinsic and integrated reliability properties of low-k materials. In Proceedings of the 2011 International Reliability Physics Symposium, Monterey, CA, USA, 10–14 April 2011; pp. 2–3. [Google Scholar]
- Gupta, T. Copper Interconnect Technology; Springer Science & Business Media: New York, NY, USA, 2010. [Google Scholar]
- Böttcher, C.J.F.; van Belle, O.C.; Bordewijk, P.; Rip, A. Theory of Electric Polarization; Elsevier Science Ltd.: Munich, Germany, 1978; Volume 2. [Google Scholar]
- Ferradini, C.; Jay-Gerin, J.P. Excess Electrons in Dielectric Media; CRC Press: New York, NY USA, 1991. [Google Scholar]
- Shamiryan, D.; Abell, T.; Iacopi, F.; Maex, K. Low-k dielectric materials. Mater. Today 2004, 7, 34–39. [Google Scholar] [CrossRef]
- Prakash, S.S.; Brinker, C.J.; Hurd, A.J.; Rao, S.M. Silica aerogel films prepared at ambient pressure by using surface derivatization to induce reversible drying shrinkage. Nature 1995, 374, 439. [Google Scholar] [CrossRef]
- Prakash, S.S.; Brinker, C.J.; Hurd, A.J. Silica aerogel films at ambient pressure. J. Non-Cryst. Solids 1995, 190, 264–275. [Google Scholar] [CrossRef]
- Jin, C.; Luttmer, J.; Smith, D.M.; Ramos, T.A. Nanoporous silica as an ultralow-k dielectric. MRS Bull. 1997, 22, 39–42. [Google Scholar] [CrossRef]
- Lu, Y.; Ganguli, R.; Drewien, C.A.; Anderson, M.T.; Brinker, C.J.; Gong, W.; Guo, Y.; Soyez, H.; Dunn, B.; Huang, M.H.; et al. Continuous formation of supported cubic and hexagonal mesoporous films by sol–gel dip-coating. Nature 1997, 389, 364. [Google Scholar] [CrossRef]
- Baskaran, S.; Liu, J.; Domansky, K.; Kohler, N.; Li, X.; Coyle, C.; Fryxell, G.E.; Thevuthasan, S.; Williford, R.E. Low dielectric constant mesoporous silica films through molecularly templated synthesis. Adv. Mater. 2000, 12, 291–294. [Google Scholar] [CrossRef]
- Bruinsma, P.J.; Kim, A.Y.; Liu, J.; Baskaran, S. Mesoporous silica synthesized by solvent evaporation: Spun fibers and spray-dried hollow spheres. Chem. Mater. 1997, 9, 2507–2512. [Google Scholar] [CrossRef]
- Zhao, D.; Yang, P.; Melosh, N.; Feng, J.; Chmelka, B.F.; Stucky, G.D. Continuous mesoporous silica films with highly ordered large pore structures. Adv. Mater. 1998, 10, 1380–1385. [Google Scholar] [CrossRef]
- Wirnsberger, G.; Yang, P.; Scott, B.J.; Chmelka, B.F.; Stucky, G.D. Mesostructured materials for optical applications: From low-k dielectrics to sensors and lasers. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2001, 57, 2049–2060. [Google Scholar] [CrossRef]
- Cho, A.; Pan, F.; Chao, K.; Liu, P.; Chen, J. The preparation of mesoporous silica ultra-low-k film using ozone ashing treatment. Thin Solid Film. 2005, 483, 283–286. [Google Scholar] [CrossRef]
- Tanaka, S.; Tada, H.; Maruo, T.; Nishiyama, N.; Egashira, Y.; Ueyama, K. Vapor infiltration techniques for spin-on mesoporous silica films. Thin Solid Film. 2006, 495, 186–190. [Google Scholar] [CrossRef]
- Maruo, T.; Tanaka, S.; Hillhouse, H.W.; Nishiyama, N.; Egashira, Y.; Ueyama, K. Disordered mesoporous silica low-k thin films prepared by vapor deposition into a triblock copolymer template film. Thin Solid Film. 2008, 516, 4771–4776. [Google Scholar] [CrossRef]
- Min, C.K.; Wu, T.B.; Yang, W.T.; Chen, C.L. Functionalized mesoporous silica/polyimide nanocomposite thin films with improved mechanical properties and low dielectric constant. Compos. Sci. Technol. 2008, 68, 1570–1578. [Google Scholar] [CrossRef]
- Vengatesan, M.; Devaraju, S.; Dinakaran, K.; Alagar, M. SBA-15 filled polybenzoxazine nanocomposites for low-k dielectric applications. J. Mater. Chem. 2012, 22, 7559–7566. [Google Scholar] [CrossRef]
- Gunasekaran, S.; Rajakumar, K.; Alagar, M.; Dharmendirakumar, M. Design and development of mesoporous silica reinforced skeletal modified triaryl pyridine core based polybenzoxazine (SBA-15/PBZ) nanocomposites. Int. J. Plast. Technol. 2015, 19, 309–332. [Google Scholar] [CrossRef]
- Selvaraj, V.; Jayanthi, K.; Lakshmikandhan, T.; Alagar, M. Development of a polybenzoxazine/TSBA-15 composite from the renewable resource cardanol for low-k applications. RSC Adv. 2015, 5, 48898–48907. [Google Scholar] [CrossRef]
- Purushothaman, R. Amine functionalized SBA-15/terpolyimide composites with low dielectric constant. J. Porous Mater. 2015, 22, 585–594. [Google Scholar] [CrossRef]
- Devaraju, S.; Vengatesan, M.; Selvi, M.; Song, J.; Alagar, M. Mesoporous silica reinforced cyanate ester nanocomposites for low k dielectric applications. Microporous Mesoporous Mater. 2013, 179, 157–164. [Google Scholar] [CrossRef]
- de Theije, F.K.; Balkenende, A.R.; Verheijen, M.A.; Baklanov, M.R.; Mogilnikov, K.P.; Furukawa, Y. Structural characterization of mesoporous organosilica films for ultralow-k dielectrics. J. Phys. Chem. B 2003, 107, 4280–4289. [Google Scholar] [CrossRef]
- Li, Z.; Li, S.; Luo, H.; Yan, Y. Effects of Crystallinity in Spin-On Pure-Silica-Zeolite MFI Low-Dielectric-Constant Films. Adv. Funct. Mater. 2004, 14, 1019–1024. [Google Scholar] [CrossRef]
- Li, Z.; Lew, C.M.; Li, S.; Medina, D.I.; Yan, Y. Pure-silica-zeolite MEL low-k films from nanoparticle suspensions. J. Phys. Chem. B 2005, 109, 8652–8658. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Johnson, M.C.; Sun, M.; Ryan, E.T.; Earl, D.J.; Maichen, W.; Martin, J.I.; Li, S.; Lew, C.M.; Wang, J.; et al. Mechanical and dielectric properties of pure-silica-zeolite low-k materials. Angew. Chem. Int. Ed. 2006, 45, 6329–6332. [Google Scholar] [CrossRef] [PubMed]
- Eslava, S.; Baklanov, M.R.; Neimark, A.V.; Iacopi, F.; Kirschhock, C.E.; Maex, K.; Martens, J.A. Evidence of Large Voids in Pure-Silica-Zeolite Low-k Dielectrics Synthesized by Spin-on of Nanoparticle Suspensions. Adv. Mater. 2008, 20, 3110–3116. [Google Scholar] [CrossRef]
- Baklanov, M.; Green, M.; Maex, K. Dielectric Films for Advanced Microelectronics; Wiley Online Library: Singapore, 2007; Volume 12. [Google Scholar]
- Liu, Y.; Sun, M.; Lew, C.M.; Wang, J.; Yan, Y. MEL-type Pure-Silica Zeolite Nanocrystals Prepared by an Evaporation-Assisted Two-Stage Synthesis Method as Ultra-Low-k Materials. Adv. Funct. Mater. 2008, 18, 1732–1738. [Google Scholar] [CrossRef]
- Lu, Y.; Fan, H.; Doke, N.; Loy, D.A.; Assink, R.A.; LaVan, D.A.; Brinker, C.J. Evaporation-induced self-assembly of hybrid bridged silsesquioxane film and particulate mesophases with integral organic functionality. J. Am. Chem. Soc. 2000, 122, 5258–5261. [Google Scholar] [CrossRef]
- Hatton, B.D.; Landskron, K.; Whitnall, W.; Perovic, D.D.; Ozin, G.A. Spin-Coated Periodic Mesoporous Organosilica Thin Films-Towards a New Generation of Low-Dielectric-Constant Materials. Adv. Funct. Mater. 2005, 15, 823–829. [Google Scholar] [CrossRef]
- Yang, S.; Mirau, P.A.; Pai, C.S.; Nalamasu, O.; Reichmanis, E.; Pai, J.C.; Obeng, Y.S.; Seputro, J.; Lin, E.K.; Lee, H.J.; et al. Nanoporous ultralow dielectric constant organosilicates templated by triblock copolymers. Chem. Mater. 2002, 14, 369–374. [Google Scholar] [CrossRef]
- De Rouffignac, P.; Li, Z.; Gordon, R.G. Sealing porous low-k dielectrics with silica. Electrochem. Solid-State Lett. 2004, 7, G306–G308. [Google Scholar] [CrossRef]
- Peters, L. Is pore sealing key to ultralow-K adoption? Semicond. Int. 2005, 28, 49–53. [Google Scholar]
- Jiang, Y.B.; Liu, N.; Gerung, H.; Cecchi, J.L.; Brinker, C.J. Nanometer-thick conformal pore sealing of self-assembled mesoporous silica by plasma-assisted atomic layer deposition. J. Am. Chem. Soc. 2006, 128, 11018–11019. [Google Scholar] [CrossRef] [PubMed]
- Seino, M.; Wang, W.; Lofgreen, J.E.; Puzzo, D.P.; Manabe, T.; Ozin, G.A. Low-k periodic mesoporous organosilica with air walls: POSS-PMO. J. Am. Chem. Soc. 2011, 133, 18082–18085. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Abbenhuis, H.C.; Yang, Q.; Wang, Y.M.; Magusin, P.C.; Mezari, B.; van Santen, R.A.; Li, C. Mesoporous organic–inorganic hybrid materials built using polyhedral oligomeric silsesquioxane blocks. Angew. Chem. Int. Ed. 2007, 46, 5003–5006. [Google Scholar] [CrossRef] [PubMed]
- Tsionsky, M.; Gun, G.; Glezer, V.; Lev, O. Sol-gel-derived ceramic-carbon composite electrodes: Introduction and scope of applications. Anal. Chem. 1994, 66, 1747–1753. [Google Scholar] [CrossRef]
- Lev, O.; Wu, Z.; Bharathi, S.; Glezer, V.; Modestov, A.; Gun, J.; Rabinovich, L.; Sampath, S. Sol- gel materials in electrochemistry. Chem. Mater. 1997, 9, 2354–2375. [Google Scholar] [CrossRef]
- Rabinovich, L.; Lev, O. Sol-Gel Derived Composite Ceramic Carbon Electrodes. Electroanal. Int. J. Devoted Fundam. Pract. Asp. Electroanal. 2001, 13, 265–275. [Google Scholar] [CrossRef]
- Bharathi, S.; Nogami, M.; Lev, O. Electrochemical organization of gold nanoclusters in three dimensions as thin films from an aminosilicate-stabilized gold sol and their characterization. Langmuir 2001, 17, 2602–2609. [Google Scholar] [CrossRef]
- Walcarius, A. Electroanalysis with Pure, Chemically Modified and Sol-Gel-Derived Silica-Based Materials. Electroanal. Int. J. Devoted Fundam. Pract. Asp. Electroanal. 2001, 13, 701–718. [Google Scholar] [CrossRef]
- Mousty, C.; Walcarius, A. Electrochemically assisted deposition by local pH tuning: A versatile tool to generate ordered mesoporous silica thin films and layered double hydroxide materials. J. Solid State Electrochem. 2015, 19, 1905–1931. [Google Scholar] [CrossRef]
- Gangopadhyay, R.; De, A. Conducting polymer nanocomposites: A brief overview. Chem. Mater. 2000, 12, 608–622. [Google Scholar] [CrossRef]
- Walcarius, A. Electrochemical applications of silica-based organic- inorganic hybrid materials. Chem. Mater. 2001, 13, 3351–3372. [Google Scholar] [CrossRef]
- Onoda, M.; Moritake, T.; Matsuda, T.; Nakayama, H. Physical properties and application of conducting polypyrrole-silica glass composite films prepared by electrochemical polymerization. Synth. Met. 1995, 71, 2255–2256. [Google Scholar] [CrossRef]
- Li, J.; Tan, S.; Oh, J. Silica sol-gel immobilized amperometric enzyme electrode for peroxide determination in the organic phase. J. Electroanal. Chem. 1998, 448, 69–77. [Google Scholar] [CrossRef]
- Verghese, M.; Ramanathan, K.; Ashraf, S.; Kamalasanan, M.; Malhotra, B. Electrochemical Growth of Polyaniline in Porous Sol- Gel Films. Chem. Mater. 1996, 8, 822–824. [Google Scholar] [CrossRef]
- Gamero-Quijano, A.; Huerta, F.; Salinas-Torres, D.; Morallón, E.; Montilla, F. Enhancement of the Electrochemical Performance of SWCNT dispersed in a Silica Sol-gel Matrix by Reactive Insertion of a Conducting Polymer. Electrochim. Acta 2014, 135, 114–120. [Google Scholar] [CrossRef]
- Farghaly, A.A.; Collinson, M.M. Mesoporous hybrid polypyrrole-silica nanocomposite films with a strata-like structure. Langmuir 2016, 32, 5925–5936. [Google Scholar] [CrossRef]
- Corriu, R.J.; Moreau, J.J.; Thepot, P.; Wong Chi Man, M.; Chorro, C.; Lere-Porte, J.P.; Sauvajol, J.L. Trialkoxysilyl Mono-, Bi-, and terthiophenes as molecular precursors of hybrid organic-inorganic materials. Chem. Mater. 1994, 6, 640–649. [Google Scholar] [CrossRef]
- Kepley, L.J.; Bard, A.J. Polymer Films on Electrodes XXVII. Electrochemical and Ellipsometric Measurements of a Viologen-Siloxane Polymer Film: Deposition, Solvent Swelling, Oxidation-State-Dependent Thickness, and Charge Transport. J. Electrochem. Soc. 1995, 142, 4129–4138. [Google Scholar] [CrossRef]
- Audebert, P.; Calas, P.; Cerveau, G.; Corriu, R.J.; Costa, N. Modified electrodes from organic-inorganic hybrid gels containing ferrocene units covalently bonded inside a silica network. J. Electroanal. Chem. 1994, 372, 275–277. [Google Scholar] [CrossRef]
- Audebert, P.; Cerveau, G.; Corriu, R.J.; Costa, N. Modified electrodes from organic-inorganic hybrid gels formed by hydrolysis-polycondensation of some trimethoxysilylferrocenes. J. Electroanal. Chem. 1996, 413, 89–96. [Google Scholar] [CrossRef]
- Gun, J.; Leva, O. Wiring of Glucose Oxidase to Carbon Matrices VIA Sol-Gel Derived Redox Modified Silicate. Anal. Lett. 1996, 29, 1933–1938. [Google Scholar] [CrossRef]
- Luka, M.; Karreman, S.; Polarz, S. Plug and play synthesis of an organic/inorganic hybrid electrode with adjustable porosity: Redox-active organosilica confined in mesoporous carbon. J. Mater. Chem. A 2015, 3, 22017–22020. [Google Scholar] [CrossRef]
- Bharathi, S.; Wu, Z.; Lev, O. Redox-modified silicate films: Anthraquinone- and Prussian-blue-modified sol-gel electrodes. SPIE Proc. 1997, 3136, 127–134. [Google Scholar]
- Wei, L.; Hu, N.; Zhang, Y. Synthesis of Polymer-Mesoporous Silica Nanocomposites. Materials 2010, 3, 4066–4079. [Google Scholar] [CrossRef] [PubMed]
- Cardin, D. Encapsulated Conducting Polymers. Adv. Mater. 2002, 14, 553–563. [Google Scholar] [CrossRef]
- Cheng, Q.; Pavlinek, V.; Li, C.; Lengalova, A.; He, Y.; Saha, P. Synthesis and characterization of new mesoporous material with conducting polypyrrole confined in mesoporous silica. Mater. Chem. Phys. 2006, 98, 504–508. [Google Scholar] [CrossRef]
- Cheng, Q.; Pavlinek, V.; Lengalova, A.; Li, C.; He, Y.; Saha, P. Conducting polypyrrole confined in ordered mesoporous silica SBA-15 channels: Preparation and its electrorheology. Microporous Mesoporous Mater. 2006, 93, 263–269. [Google Scholar] [CrossRef]
- Li, N.; Li, X.; Geng, W.; Zhang, T.; Zuo, Y.; Qiu, S. Synthesis and humidity sensitivity of conducting polyaniline in SBA-15. J. Appl. Polym. Sci. 2004, 93, 1597–1601. [Google Scholar] [CrossRef]
- Sasidharan, M.; Mal, N.K.; Bhaumik, A. In-situ polymerization of grafted aniline in the channels of mesoporous silica SBA-15. J. Mater. Chem. 2007, 17, 278–283. [Google Scholar] [CrossRef]
- Zeng, C.; Han, S.; Yan, X.; Yu, X.; Che, H.; Zhao, X. Ordered mesoporous silica/polyaniline composites. Prog. Chem. 2008, 20, 26–32. [Google Scholar]
- Li, Y.; Zhang, Q.; Zhao, X.; Yu, P.; Wu, L.; Chen, D. Enhanced electrochemical performance of polyaniline/sulfonated polyhedral oligosilsesquioxane nanocomposites with porous and ordered hierarchical nanostructure. J. Mater. Chem. 2012, 22, 1884–1892. [Google Scholar] [CrossRef]
- Montilla, F.; Cotarelo, M.A.; Morallón, E. Hybrid sol–gel–conducting polymer synthesised by electrochemical insertion: Tailoring the capacitance of polyaniline. J. Mater. Chem. 2009, 19, 305–310. [Google Scholar] [CrossRef]
- Salinas-Torres, D.; Montilla, F.; Huerta, F.; Morallón, E. All electrochemical synthesis of polyaniline/silica sol–gel materials. Electrochim. Acta 2011, 56, 3620–3625. [Google Scholar] [CrossRef]
- Djelad, H.; Huerta, F.; Morallón, E.; Montilla, F. Modulation of the electrocatalytic performance of PEDOT-PSS by reactive insertion into a sol-gel silica matrix. Eur. Polym. J. 2018, 105, 323–330. [Google Scholar] [CrossRef]
- Rivero, O.; Huerta, F.; Montilla, F.; Sanchis, C.; Morallón, E. Electrocatalytic oxidation of ascorbic acid on mesostructured SiO2-conducting polymer composites. Eur. Polym. J. 2015, 69, 201–207. [Google Scholar] [CrossRef]
- Domenech, A.; Alvaro, M.; Ferrer, B.; Garcia, H. Electrochemistry of Mesoporous Organosilica of MCM-41 Type Containing 4,4’-Bipyridinium Units: Voltammetric Response and Electrocatalytic Effect on 1,4-Dihydrobenzoquinone Oxidation. J. Phys. Chem. B 2003, 107, 12781–12788. [Google Scholar] [CrossRef]
- Guo, Y.; Mylonakis, A.; Zhang, Z.; Yang, G.; Lelkes, P.; Che, S.; Lu, Q.; Wei, Y. Templated Synthesis of Electroactive Periodic Mesoporous Organosilica Bridged with Oligoaniline. Chem. Eur. J. 2008, 14, 2909–2917. [Google Scholar] [CrossRef]
- Fattakhova Rohlfing, D.; Rathouský, J.; Rohlfing, Y.; Bartels, O.; Wark, M. Functionalized Mesoporous Silica Films as a Matrix for Anchoring Electrochemically Active Guests. Langmuir 2005, 21, 11320–11329. [Google Scholar] [CrossRef]
- Sel, O.; Sallard, S.; Brezesinski, T.; Rathouský, J.; Dunphy, D.; Collord, A.; Smarsly, B. Periodically Ordered Meso- and Macroporous SiO2 Thin Films and Their Induced Electrochemical Activity as a Function of Pore Hierarchy. Adv. Funct. Mater. 2007, 17, 3241–3250. [Google Scholar] [CrossRef]
- Zhou, Z.; Franz, A.W.; Bay, S.; Sarkar, B.; Seifert, A.; Yang, P.; Wagener, A.; Ernst, S.; Pagels, M.; Müller, T.; et al. Redox Active Mesoporous Hybrid Materials by In situ Syntheses with Urea-linked Triethoxysilylated Phenothiazines. Chem. Asian J. 2010, 5, 2001–2015. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Villemure, G. Electrode modification with spin-coated films of mesoporous molecular sieve silicas. Microporous Mesoporous Mater. 2001, 44, 679–689. [Google Scholar] [CrossRef]
- Luka, M.; Polarz, S. Wiring functional groups in mesoporous organosilica materials. J. Mater. Chem. C 2015, 3, 2195–2203. [Google Scholar] [CrossRef][Green Version]
- Delacote, C.; Bouillon, J.P.; Walcarius, A. Voltammetric response of ferrocene-grafted mesoporous silica. Electrochim. Acta 2006, 51, 6373–6383. [Google Scholar] [CrossRef]
- Urbanova, V.; Walcarius, A. Vertically-aligned Mesoporous Silica Films. Z. Anorg. Und Allg. Chem. 2014, 640, 537–546. [Google Scholar] [CrossRef]
- Goux, A.; Etienne, M.; Aubert, E.; Lecomte, C.; Ghanbaja, J.; Walcarius, A. Oriented Mesoporous Silica Films Obtained by Electro-Assisted Self-Assembly (EASA). Chem. Mater. 2009, 21, 731–741. [Google Scholar] [CrossRef]
- Yan, F.; Lin, X.; Su, B. Vertically ordered silica mesochannel films: Electrochemistry and analytical applications. Analyst 2016, 141, 3482–3495. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Guo, W.; Xu, L.; Yang, Q.; Su, B. Two orders-of-magnitude enhancement in the electrochemiluminescence of Ru(bpy)32+ by vertically ordered silica mesochannels. Anal. Chim. Acta 2015, 886, 48–55. [Google Scholar] [CrossRef]
- Karman, C.; Vilà, N.; Walcarius, A. Amplified Charge Transfer for Anionic Redox Probes through Oriented Mesoporous Silica Thin Films. ChemElectroChem 2016, 3, 2130–2137. [Google Scholar] [CrossRef]
- Nasir, T.; Herzog, G.; Hébrant, M.; Despas, C.; Liu, L.; Walcarius, A. Mesoporous Silica Thin Films for Improved Electrochemical Detection of Paraquat. ACS Sens. 2018, 3, 484–493. [Google Scholar] [CrossRef]
- Goux, A.; Ghanbaja, J.; Walcarius, A. Prussian Blue electrodeposition within an oriented mesoporous silica film: Preliminary observations. J. Mater. Sci. 2009, 44, 6601–6607. [Google Scholar] [CrossRef]
- Del Valle, M.A.; Gacitúa, M.; Díaz, F.R.; Armijo, F.; del Río, R. Electrosynthesis of polythiophene nanowires via mesoporous silica thin film templates. Electrochem. Commun. 2009, 11, 2117–2120. [Google Scholar] [CrossRef]
- Kang, H.S.; Lee, H.C.; Kwak, J.H. Electrodeposition of Polypyrrole Nanowires within Vertically Oriented Mesoporous Silica Template. J. Korean Electrochem. Soc. 2011, 14, 22–26. [Google Scholar] [CrossRef][Green Version]
- Gamero-Quijano, A.; Karman, C.; Vilà, N.; Herzog, G.; Walcarius, A. Vertically Aligned and Ordered One-Dimensional Mesoscale Polyaniline. Langmuir 2017, 33, 4224–4234. [Google Scholar] [CrossRef] [PubMed]
- Valle, M.; Hernández, L.; Díaz, F.; Ramos, A. Electrosynthesis and Characterization of Poly(3,4-ethylenedioxythiophene) Nanowires. Int. J. Electrochem. Sci. 2015, 10, 5152–5163. [Google Scholar]
- Valle, M.A.D.; Ramos, A.C.; Antilen, M.P.; Hernandez, L.A.; Arteaga, G.C.; DIAZ, F.R.; Louarn, G. Electro-synthesis and Characterization of Polymer Nanostructures from Terthiophene Using Silica Mesoporous Films as Template. Electrochemistry 2014, 82, 146–151. [Google Scholar] [CrossRef]
- Del Valle, M.A.; Hernández, L.A.; Ramírez, A.M.; Díaz, F.R. Electrosynthesis of polyquinone nanowires with dispersed platinum nanoparticles toward formic acid oxidation. Ionics 2017, 23, 191–199. [Google Scholar] [CrossRef]
- Hernández, L.A.; Del Valle, M.A.; Díaz, F.R.; Fermin, D.J.; Risbridger, T.A.G. Polymeric nanowires directly electrosynthesized on the working electrode. Electrochim. Acta 2015, 166, 163–167. [Google Scholar] [CrossRef]
- Ramírez AM, R.; Gacitúa, M.A.; Ortega, E.; Díaz, F.R.; del Valle, M.A. Electrochemical in situ synthesis of polypyrrole nanowires. Electrochem. Commun. 2019, 102, 94–98. [Google Scholar] [CrossRef]
- Del Valle, M.A.; Gacitua, M.; Diaz, F.R.; Armijo, F.; Soto, J.P. Electro-synthesis and characterization of polythiophene nano-wires/platinum nano-particles composite electrodes. Study of formic acid electro-catalytic oxidation. Electrochim. Acta 2012, 71, 277–282. [Google Scholar] [CrossRef]
- Salgado, R.; del Valle, M.A.; Duran, B.G.; Pardo, M.A.; Armijo, F. Optimization of dopamine determination based on nanowires PEDOT/polydopamine hybrid film modified electrode. J. Appl. Electrochem. 2014, 44, 1289–1294. [Google Scholar] [CrossRef]
- Ramírez, M.R.A.; del Valle, M.A.; Armijo, F.; Díaz, F.R.; Angélica Pardo, M.; Ortega, E. Enhancement of electrodes modified by electrodeposited PEDOT-nanowires with dispersed Pt nanoparticles for formic acid electro-oxidation. J. Appl. Polym. Sci. 2017, 134. [Google Scholar] [CrossRef]
- Guillemin, Y.; Etienne, M.; Aubert, E.; Walcarius, A. Electrogeneration of highly methylated mesoporous silica thin films with vertically-aligned mesochannels and electrochemical monitoring of mass transport issues. J. Mater. Chem. 2010, 20, 6799–6807. [Google Scholar] [CrossRef]
- Vodolazkaya, N.A.; Despas, C.; Lebeau, B.; Marichal, C.; Walcarius, A. One pot synthesis of ordered mesoporous organosilica particles bearing propyl-, octyl- and hexadecyl-chains. J. Sol-Gel Sci. Technol. 2012, 63, 587–594. [Google Scholar] [CrossRef]
- Herzog, G.; Sibottier, E.; Etienne, M.; Walcarius, A. Electrochemically assisted self-assembly of ordered and functionalized mesoporous silica films: Impact of the electrode geometry and size on film formation and properties. Faraday Discuss. 2013, 164, 259–273. [Google Scholar] [CrossRef]
- Etienne, M.; Goux, A.; Sibottier, E.; Walcarius, A. Oriented Mesoporous Organosilica Films on Electrode: A New Class of Nanomaterials for Sensing. J. Nanosci. Nanotechnol. 2009, 9, 2398–2406. [Google Scholar] [CrossRef] [PubMed]
- Vilà, N.; Ghanbaja, J.; Aubert, E.; Walcarius, A. Electrochemically Assisted Generation of Highly Ordered Azide-Functionalized Mesoporous Silica for Oriented Hybrid Films. Angew. Chem. Int. Ed. 2014, 53, 2945–2950. [Google Scholar] [CrossRef]
- Walcarius, N.V.A. Electrochemical response of vertically-aligned, ferrocene-functionalized mesoporous silica films: Effect of the supporting electrolyte. Electrochim. Acta 2015, 179, 304–314. [Google Scholar]
- Vilà, N.; Ghanbaja, J.; Walcarius, A. Clickable Bifunctional and Vertically Aligned Mesoporous Silica Films. Adv. Mater. Interfaces 2016, 3, 1500440. [Google Scholar] [CrossRef]
- Vilà, N.; Allain, C.; Audebert, P.; Walcarius, A. Tetrazine-functionalized and vertically-aligned mesoporous silica films with electrochemical activity and fluorescence properties. Electrochem. Commun. 2015, 59, 9–12. [Google Scholar] [CrossRef]
- Audebert, P.; Griesmar, P.; Hapiot, P.; Sanchez, C. Sol–gel–xerogel evolution investigated by electroactive probes in silica and transition-metal oxide based gels. J. Mater. Chem. 1992, 2, 1293–1300. [Google Scholar] [CrossRef]
- Gun, J.; Lev, O. Sol-gel derived, ferrocenyl-modified silicate-graphite composite electrode: Wiring of glucose oxidase. Anal. Chim. Acta 1996, 336, 95–106. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, T.; Gao, C.; Zhao, Y. Modifiying glassy carbon electrode with ferrocene-bridged polysilsesquioxanes. J. Sol-Gel Sci. Technol. 2010, 55, 293–298. [Google Scholar] [CrossRef]
- Vilà, N.; André, E.; Ciganda, R.; Ruiz, J.; Astruc, D.; Walcarius, A. Molecular Sieving with Vertically Aligned Mesoporous Silica Films and Electronic Wiring through Isolating Nanochannels. Chem. Mater. 2016, 28, 2511–2514. [Google Scholar] [CrossRef]
- Walcarius, A. Mesoporous materials and electrochemistry. Chem. Soc. Rev. 2013, 42, 4098–4140. [Google Scholar] [CrossRef] [PubMed]
- Melde, B.; Johnson, B.; Charles, P. Mesoporous silicate materials in sensing. Sensors 2008, 8, 5202–5228. [Google Scholar] [CrossRef] [PubMed]
- Walcarius, A. Mesoporous materials-based electrochemical sensors. Electroanalysis 2015, 27, 1303–1340. [Google Scholar] [CrossRef]
- Etienne, M.; Zhang, L.; Vilà, N.; Walcarius, A. Mesoporous Materials-Based Electrochemical Enzymatic Biosensors. Electroanalysis 2015, 27, 2028–2054. [Google Scholar] [CrossRef]
- Hopfield, J.J. Neural networks and physical systems with emergent collective computational abilities. Proc. Natl. Acad. Sci. USA 1982, 79, 2554–2558. [Google Scholar] [CrossRef]
- Hopfield, J.; Feinstein, D.; Palmer, R. Unlearning has a stabilizing effect in collective memories. Nature 1983, 304, 158–159. [Google Scholar] [CrossRef]
- Hopfield, J.J.; Tank, D.W. “Neural” computation of decisions in optimization problems. Biol. Cybern. 1985, 52, 141–152. [Google Scholar] [PubMed]
- Tank, D.; Hopfield, J. Simple “neural”optimization networks: An A/D converter, signal decision circuit, and a linear programming circuit. IEEE Trans. Circuits Syst. 1986, 33, 533–541. [Google Scholar] [CrossRef]
- Hopfield, J.; Tank, D. Computing with Neural Circuits: A Model. Science 1986, 233, 624–633. [Google Scholar] [CrossRef] [PubMed]
- Mézard, M.; Parisi, G.; Virasoro, M. Spin Glass Theory and Beyond: An Introduction to the Replica Method and Its Applications; World Scientific Publishing Company: Singapore, 1987; Volume 9. [Google Scholar]
- Laskowski, Ł. A novel hybrid-maximum neural network in stereo-matching process. Neural Comput. Appl. 2013, 23, 2435–2450. [Google Scholar] [CrossRef] [PubMed]
- Park, C.D.; Jeong, D.Y. Soluble Single-Molecule Magnet: Mn12-stearate. Bull. Korean Chem. Soc. 2001, 22, 611–615. [Google Scholar]
- Lis, T. Preparation, structure, and magnetic properties of a dodecanuclear mixed-valence manganese carboxylate. Acta Crystallogr. Sect. B Struct. Crystallogr. Cryst. Chem. 1980, 36, 2042–2046. [Google Scholar] [CrossRef]
- Christou, G.; Gatteschi, D.; Hendrickson, D.N.; Sessoli, R. Single-molecule magnets. MRS Bull. 2000, 25, 66–71. [Google Scholar] [CrossRef]
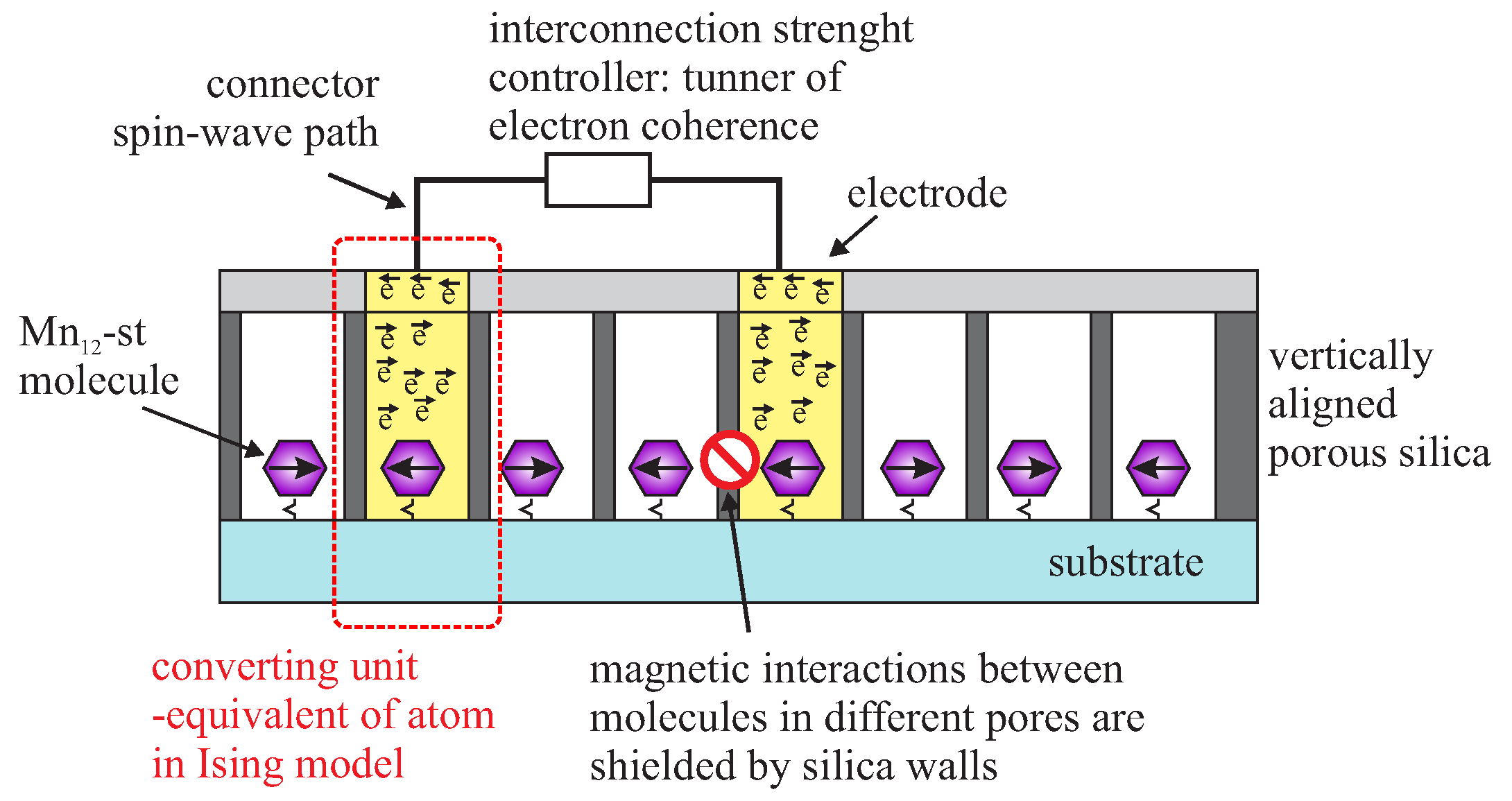
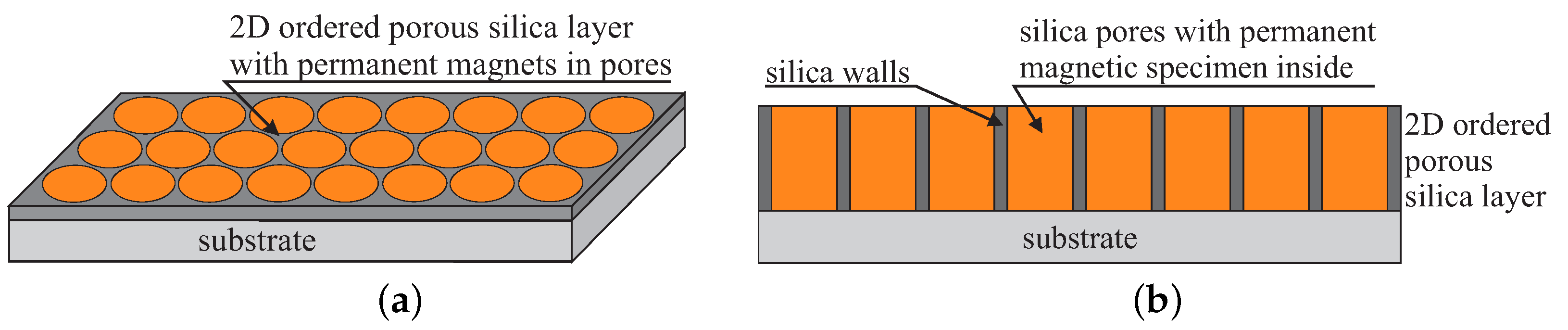
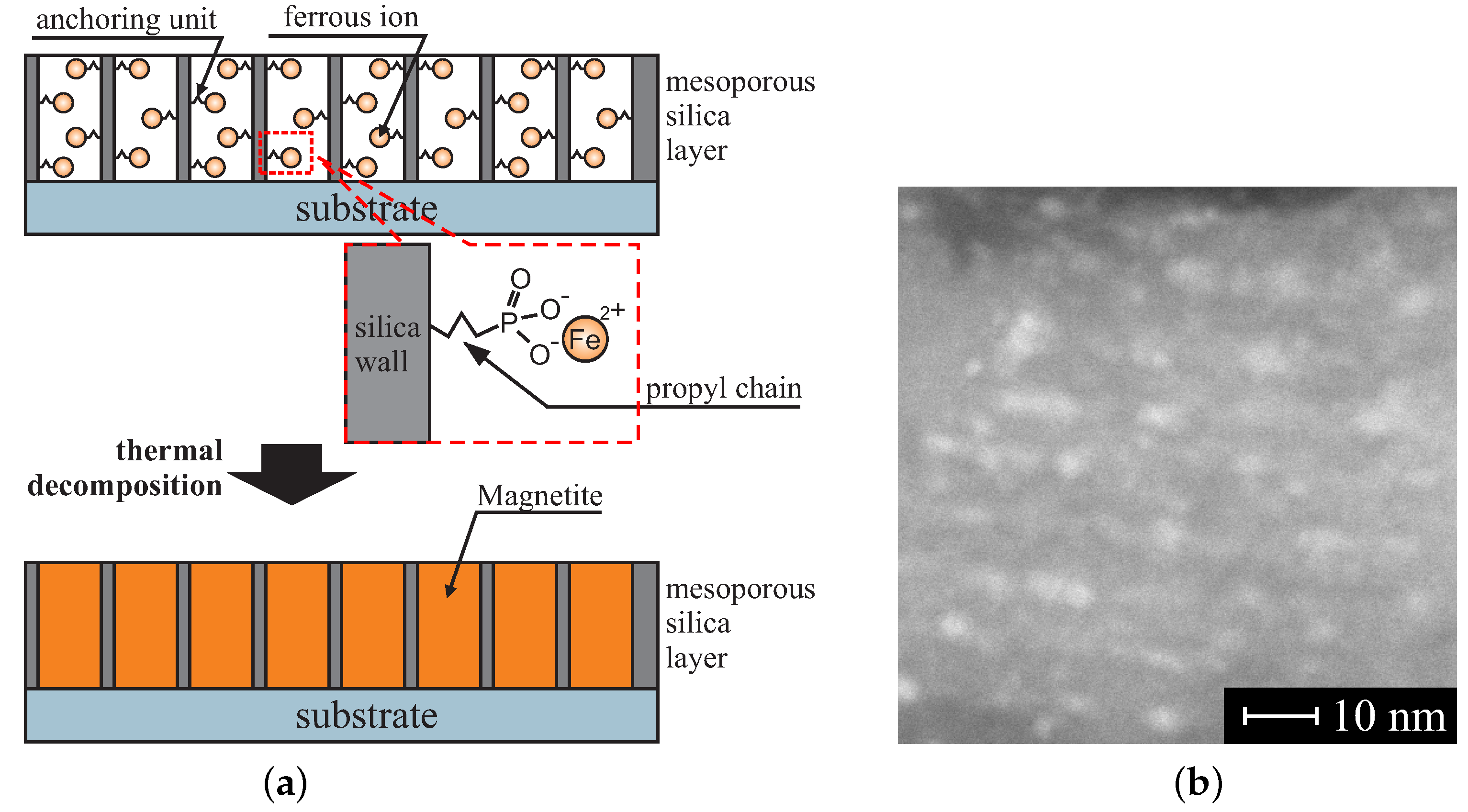
- Laskowski, Ł.; Laskowska, M.; Dulski, M.; Zubko, M.; Jelonkiewicz, J.; Perzanowski, M.; Vila, N.; Walcarius, A. Multi-step functionalization procedure for fabrication of vertically aligned mesoporous silica thin films with metal-containing molecules localized at the pores bottom. Microporous Mesoporous Mater. 2019, 274, 356–362. [Google Scholar] [CrossRef]
- Laskowski, L.; Kityk, I.; Konieczny, P.; Pastukh, O.; Schabikowski, M.; Laskowska, M. The Separation of the Mn12 Single-Molecule Magnets onto Spherical Silica Nanoparticles. Nanomaterials 2019, 9, 764. [Google Scholar] [CrossRef]
- Hueso, L.E.; Pruneda, J.M.; Ferrari, V.; Burnell, G.; Valdés-Herrera, J.P.; Simons, B.D.; Littlewood, P.B.; Artacho, E.; Fert, A.; Mathur, N.D. Transformation of spin information into large electrical signals using carbon nanotubes. Nature 2007, 445, 410–413. [Google Scholar] [CrossRef]
- Datta, S.; Das, B. Electronic analog of the electro-optic modulator. Appl. Phys. Lett. 1990, 56, 665–667. [Google Scholar] [CrossRef]
- Laskowski, Ł.; Laskowska, M.; Jelonkiewicz, J.; Galkowski, T.; Pawlik, P.; Piech, H.; Doskocz, M. Iron doped SBA-15 mesoporous silica studied by Mössbauer spectroscopy. J. Nanomater. 2016, 2016, 38. [Google Scholar]
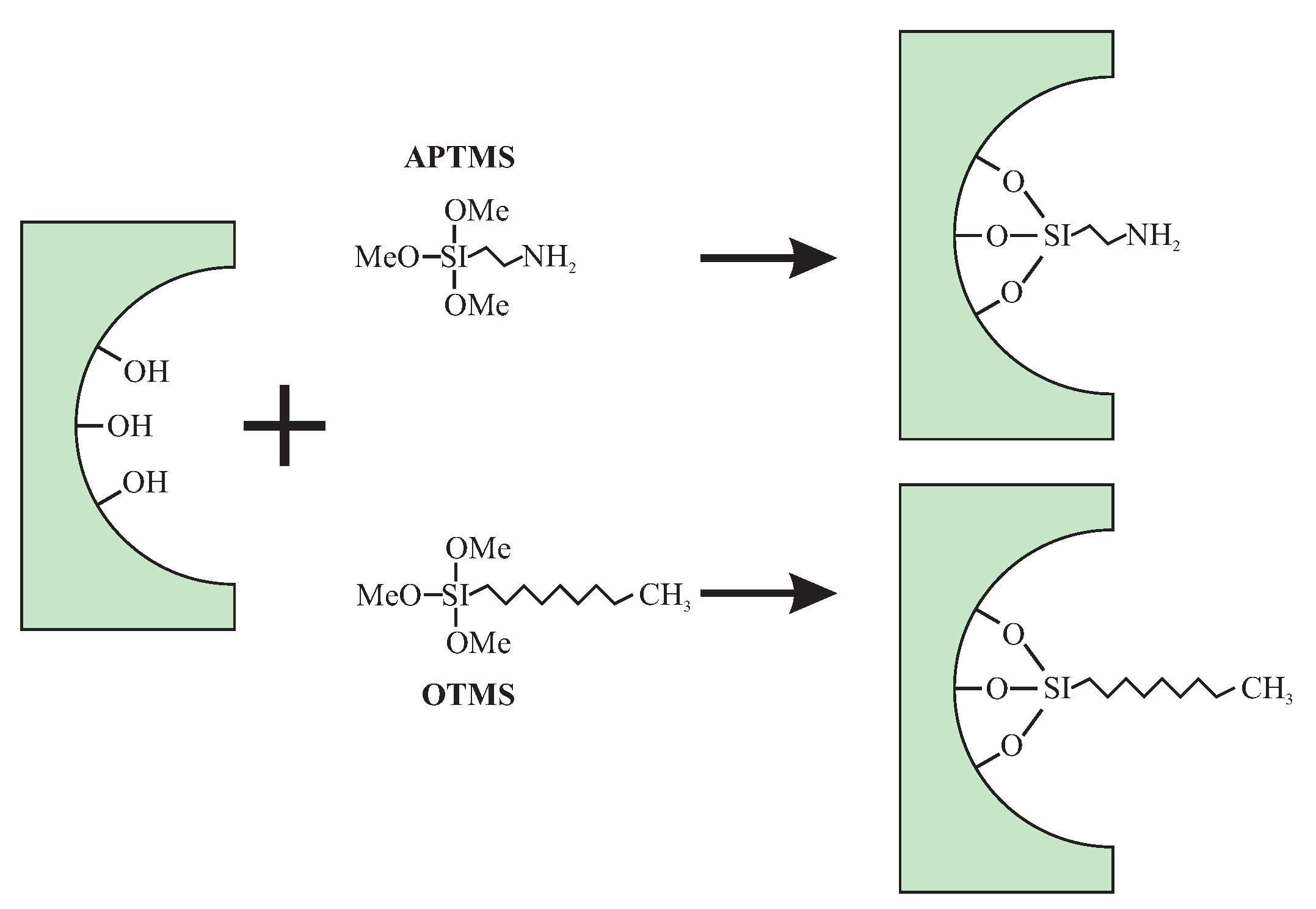
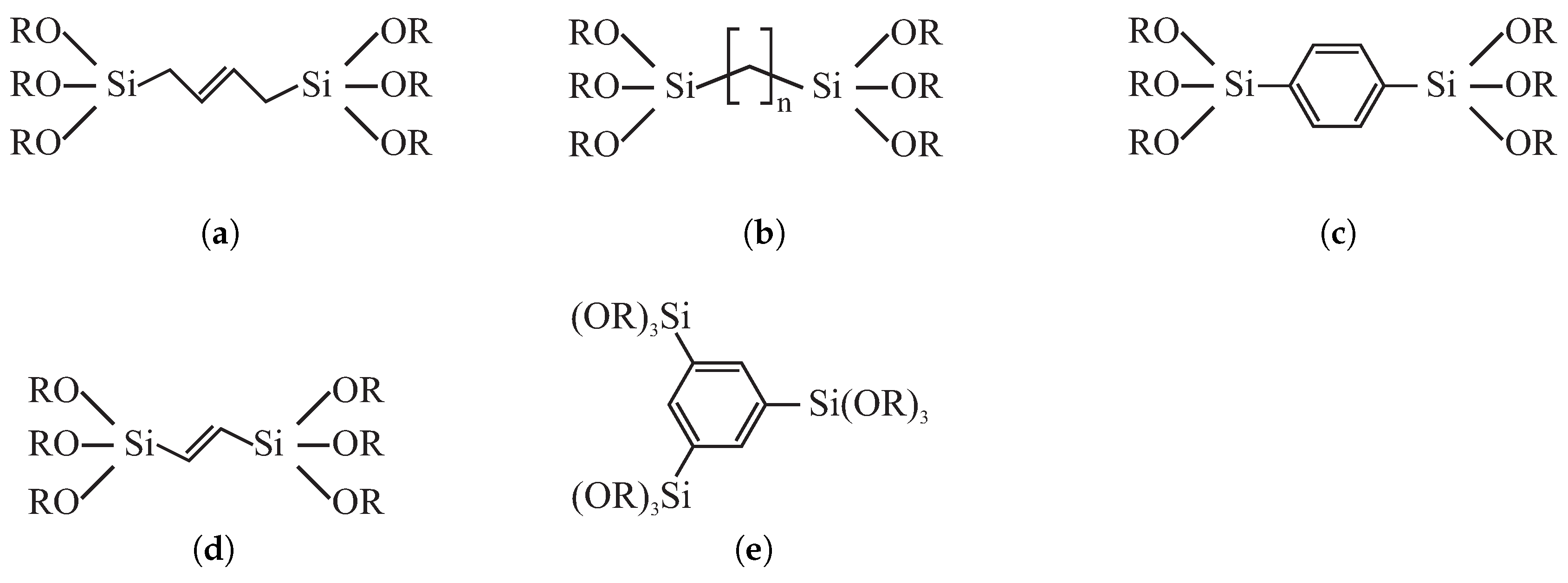
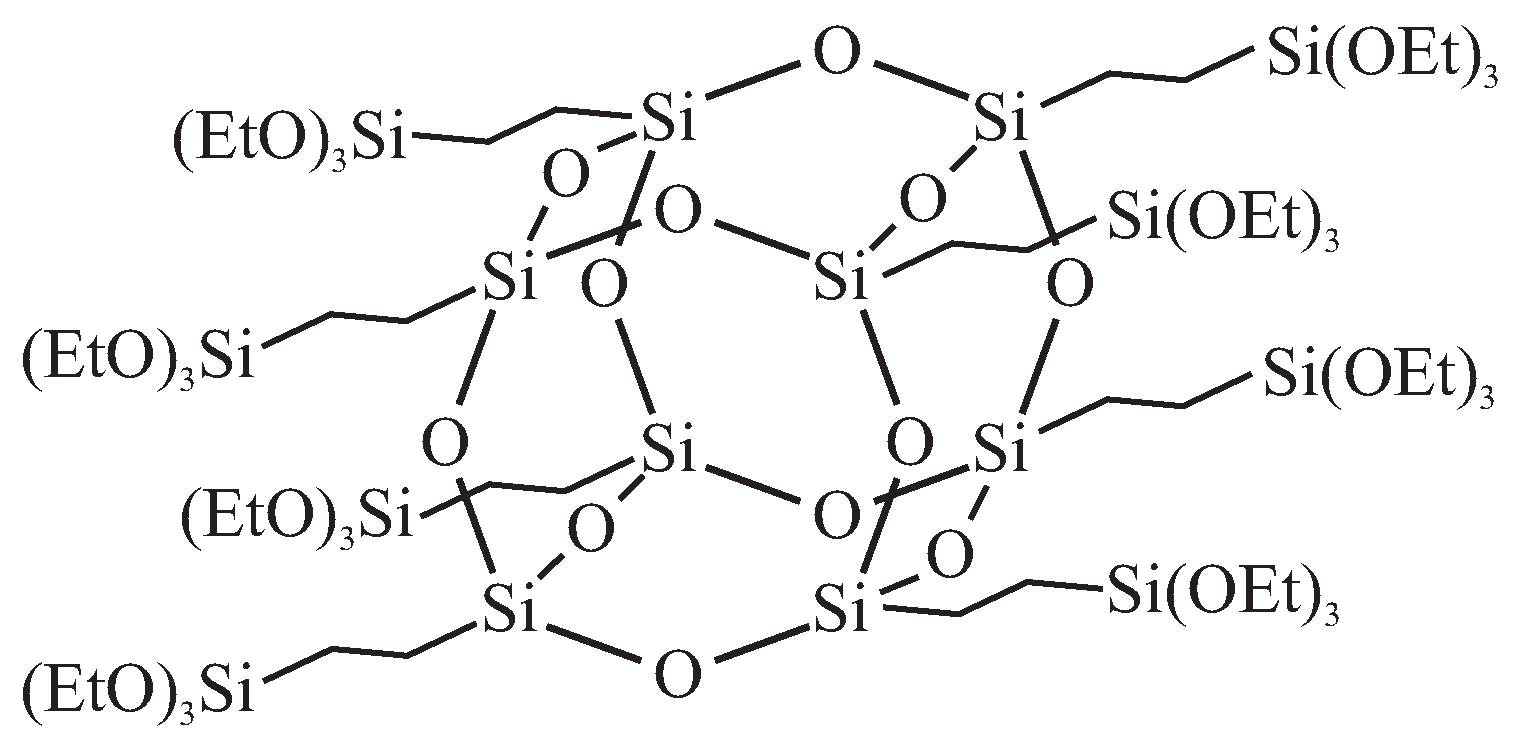

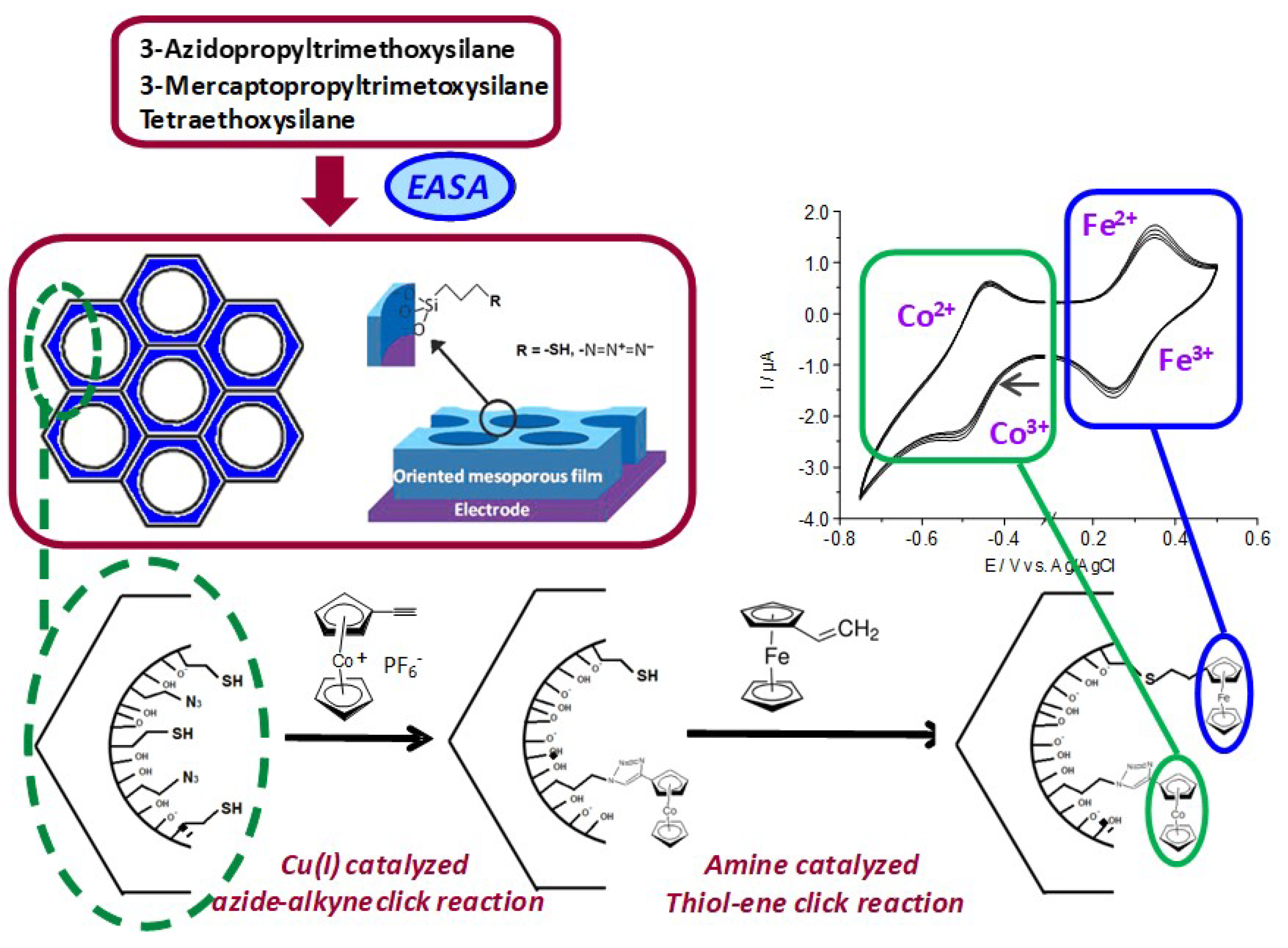


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laskowski, Ł.; Laskowska, M.; Vila, N.; Schabikowski, M.; Walcarius, A. Mesoporous Silica-Based Materials for Electronics-Oriented Applications. Molecules 2019, 24, 2395. https://doi.org/10.3390/molecules24132395
Laskowski Ł, Laskowska M, Vila N, Schabikowski M, Walcarius A. Mesoporous Silica-Based Materials for Electronics-Oriented Applications. Molecules. 2019; 24(13):2395. https://doi.org/10.3390/molecules24132395
Chicago/Turabian StyleLaskowski, Łukasz, Magdalena Laskowska, Neus Vila, Mateusz Schabikowski, and Alain Walcarius. 2019. "Mesoporous Silica-Based Materials for Electronics-Oriented Applications" Molecules 24, no. 13: 2395. https://doi.org/10.3390/molecules24132395
APA StyleLaskowski, Ł., Laskowska, M., Vila, N., Schabikowski, M., & Walcarius, A. (2019). Mesoporous Silica-Based Materials for Electronics-Oriented Applications. Molecules, 24(13), 2395. https://doi.org/10.3390/molecules24132395






