Fluorine-18 Labeled Fluorofuranylnorprogesterone ([18F]FFNP) and Dihydrotestosterone ([18F]FDHT) Prepared by “Fluorination on Sep-Pak” Method
Abstract
1. Introduction
2. Results and Discussion
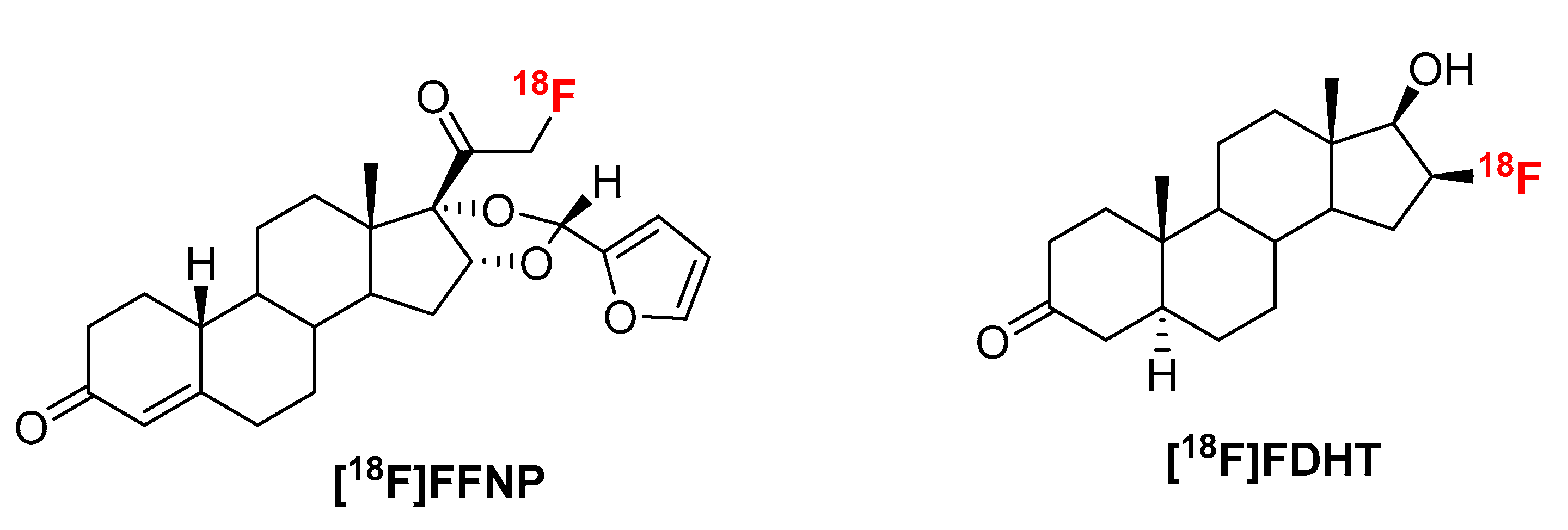
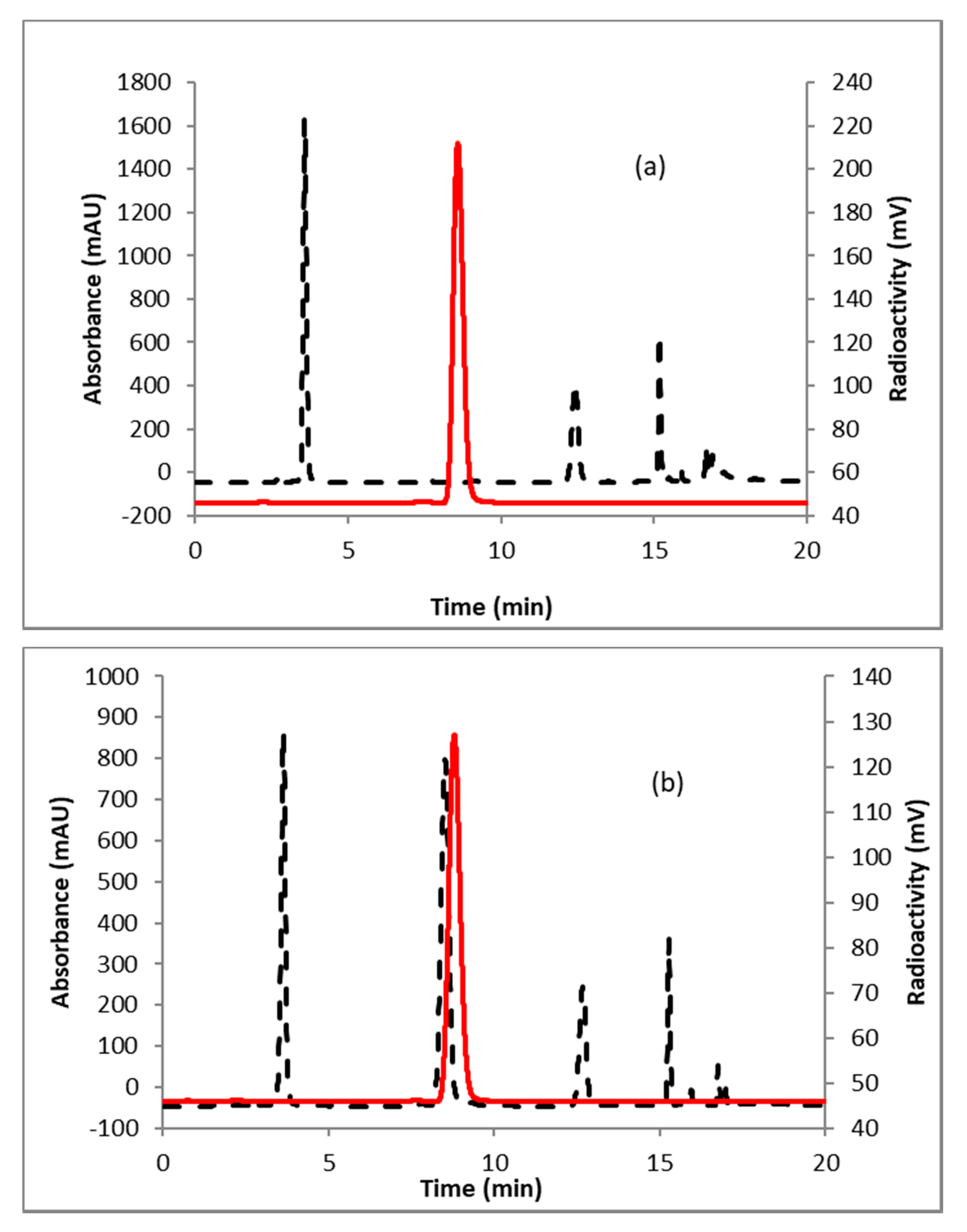
2.1. [18F]FFNP
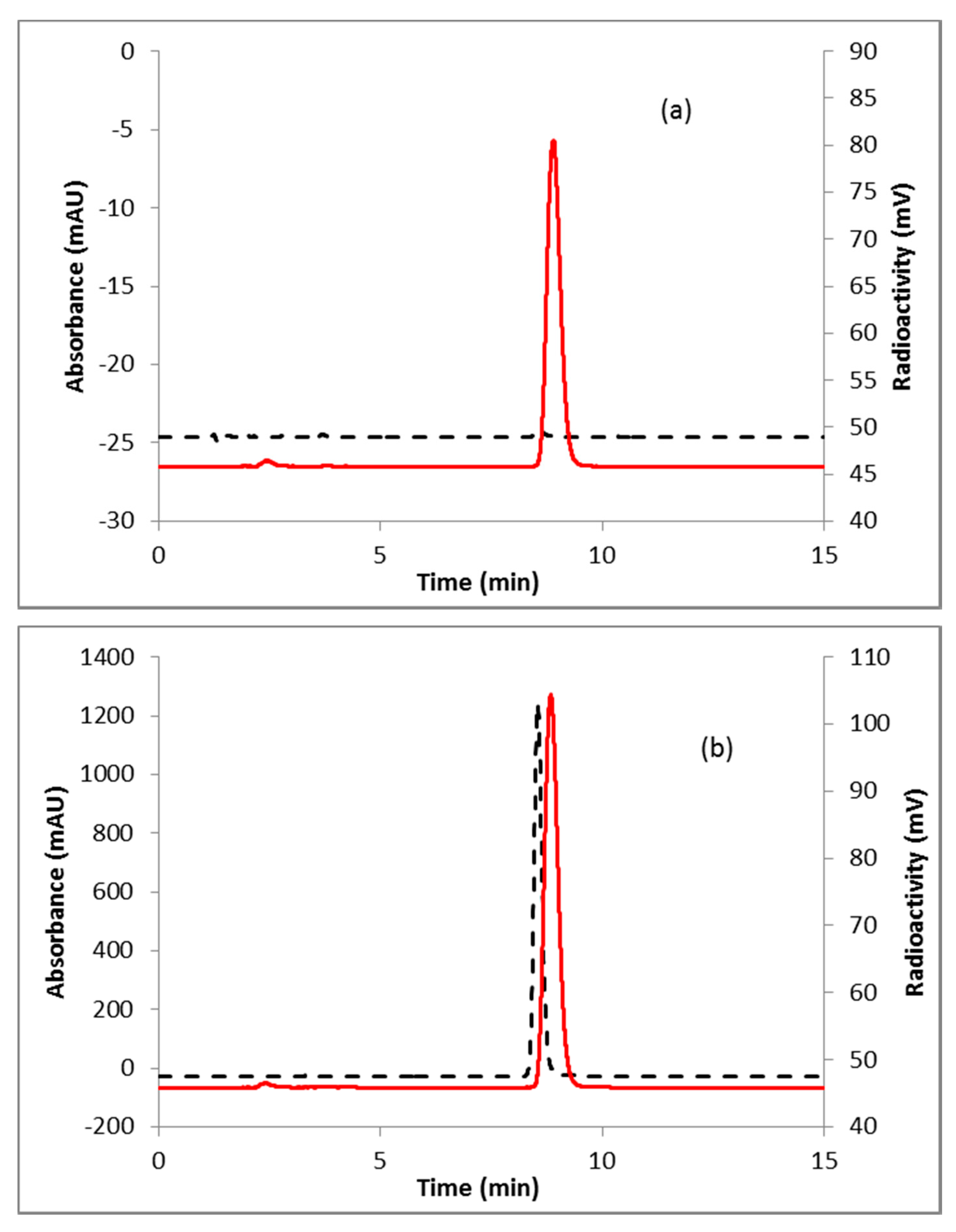
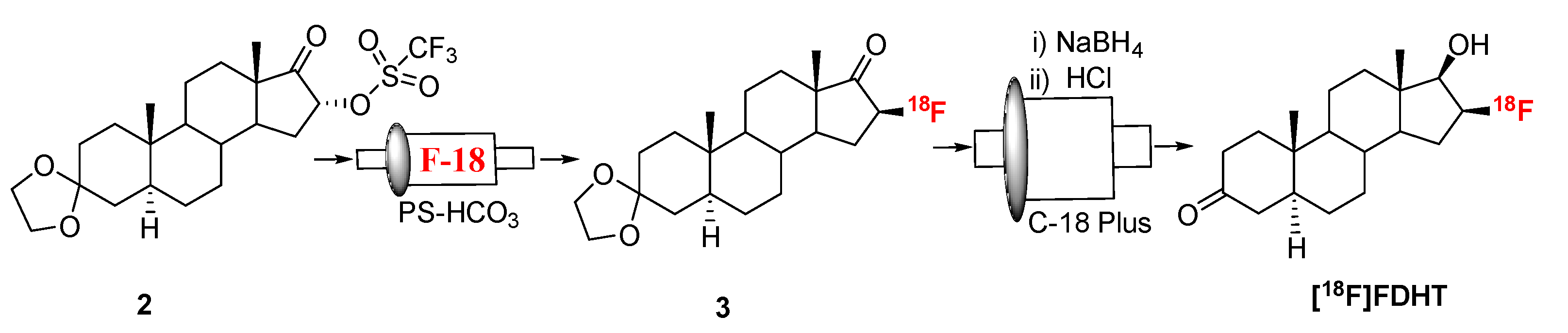
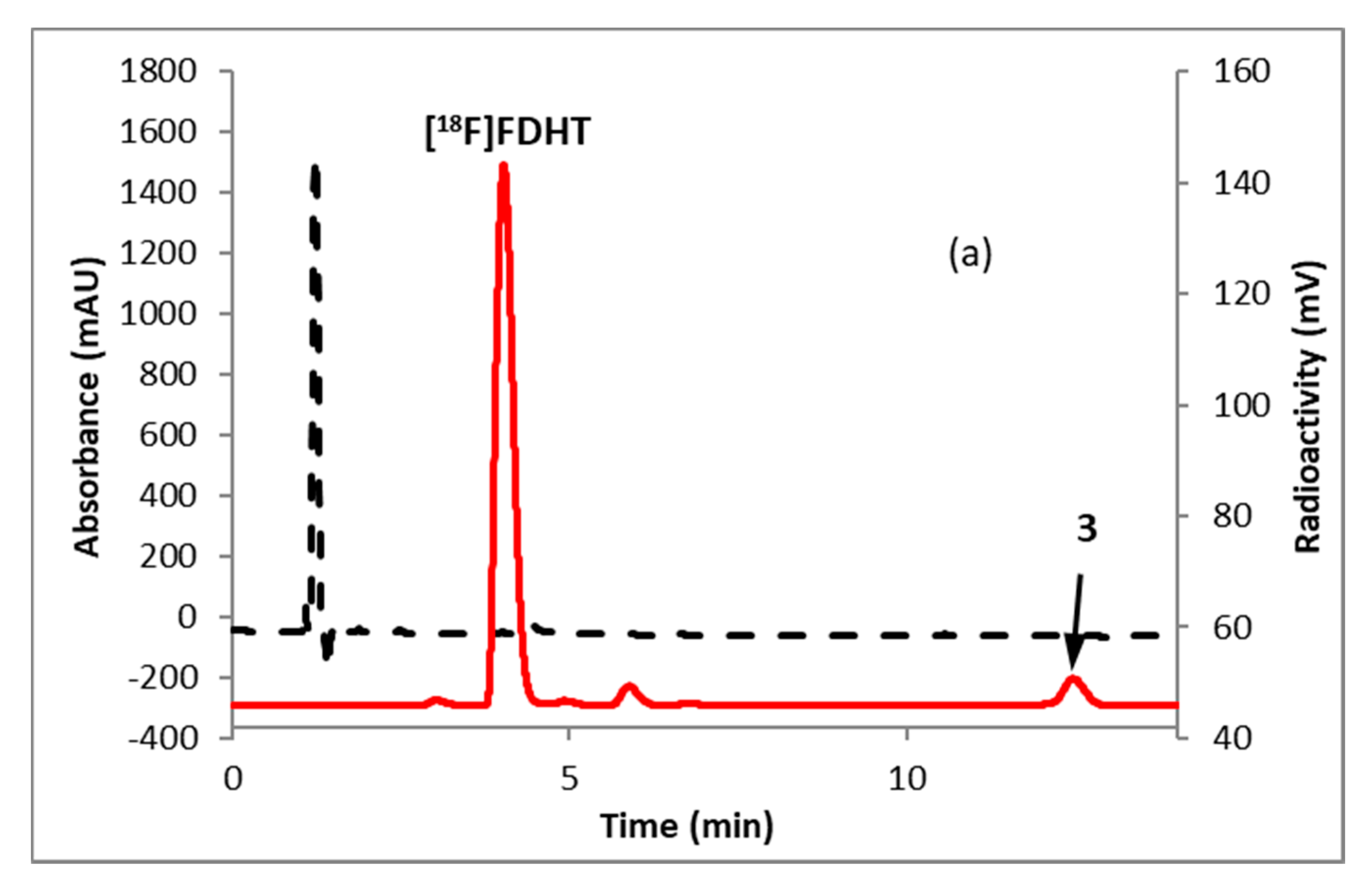
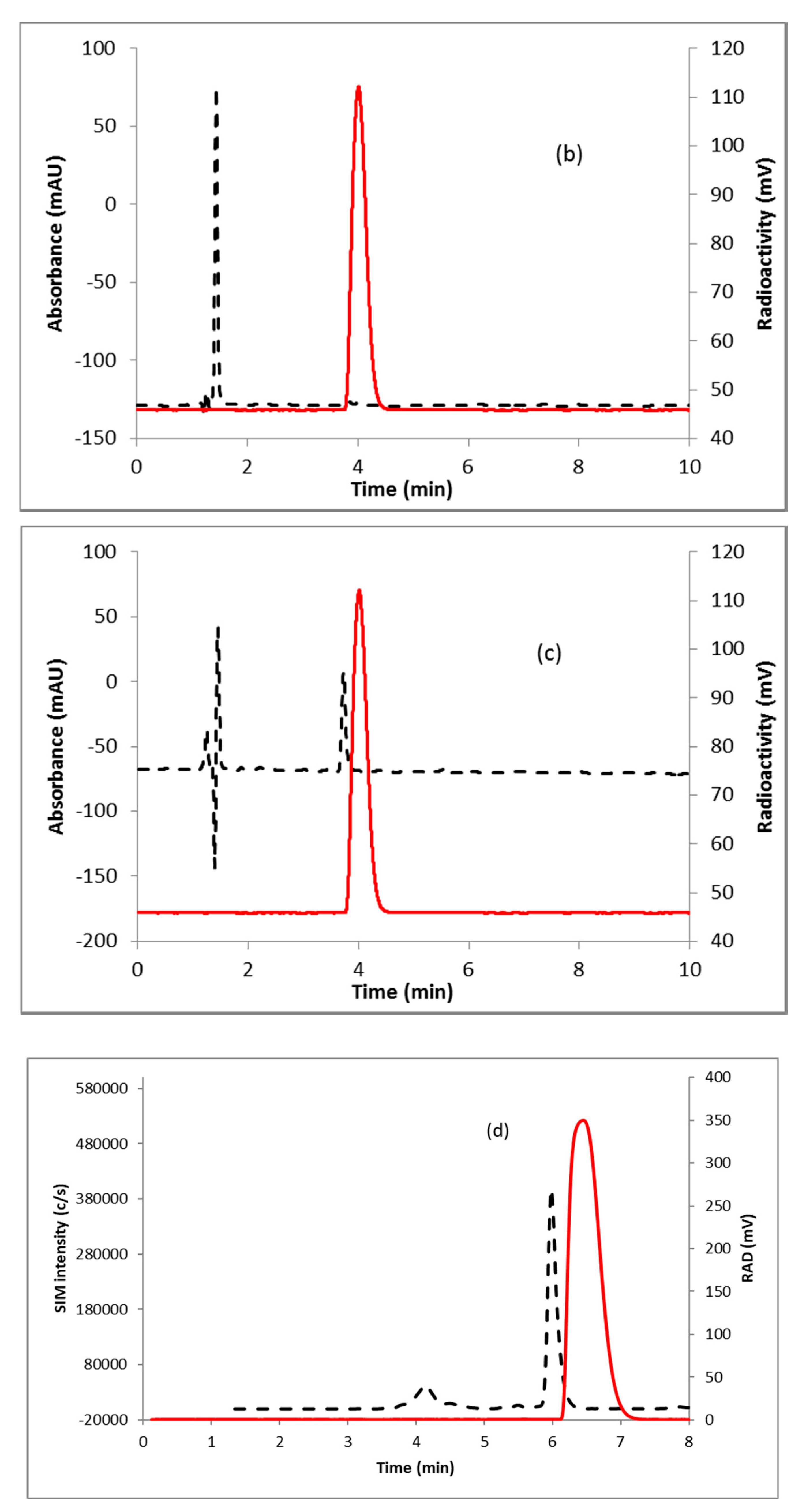
2.2. [18F]FDHT
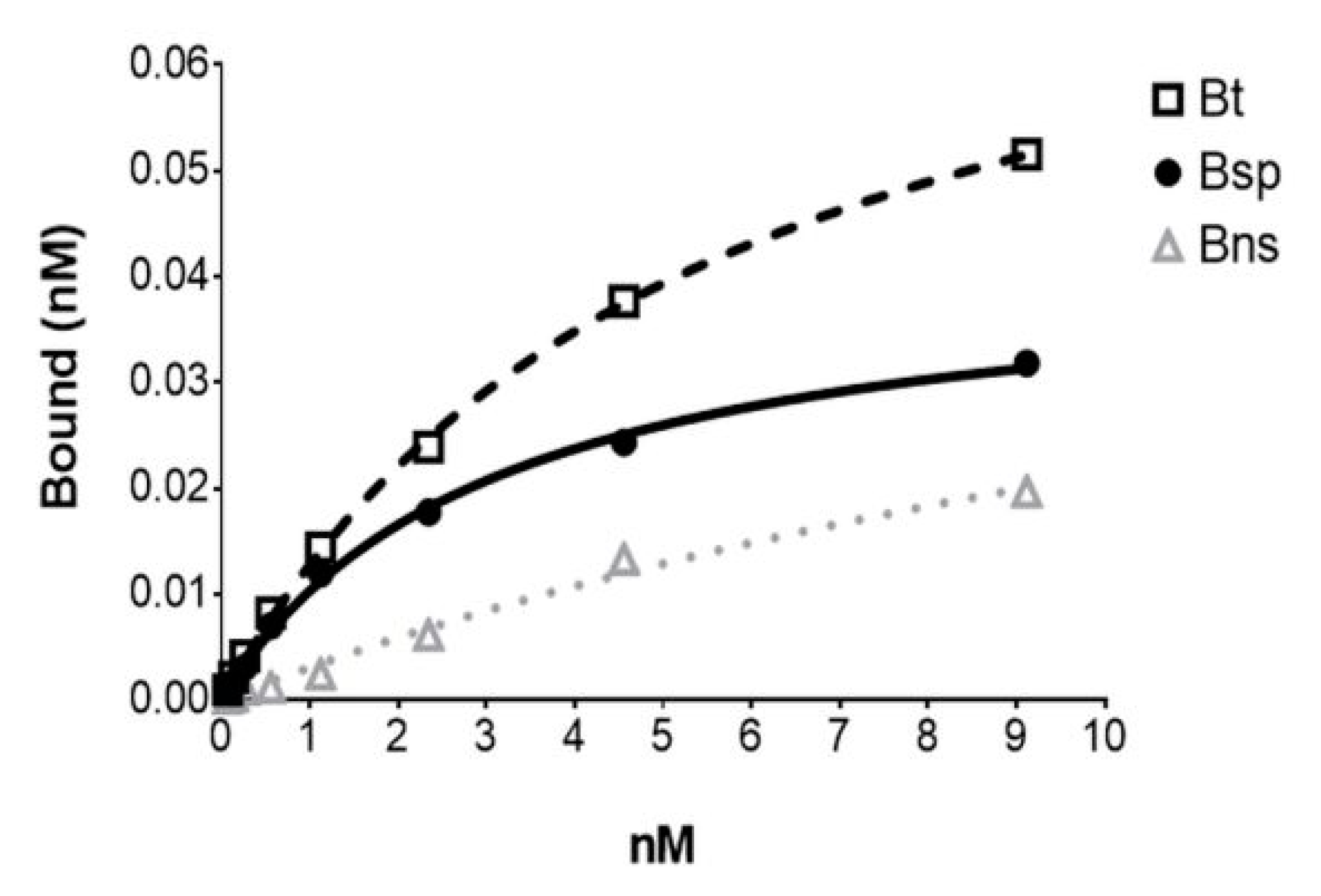
2.3. In Vitro Binding Assays
3. Materials and Methods
3.1. Materials, Chemicals, and Methods
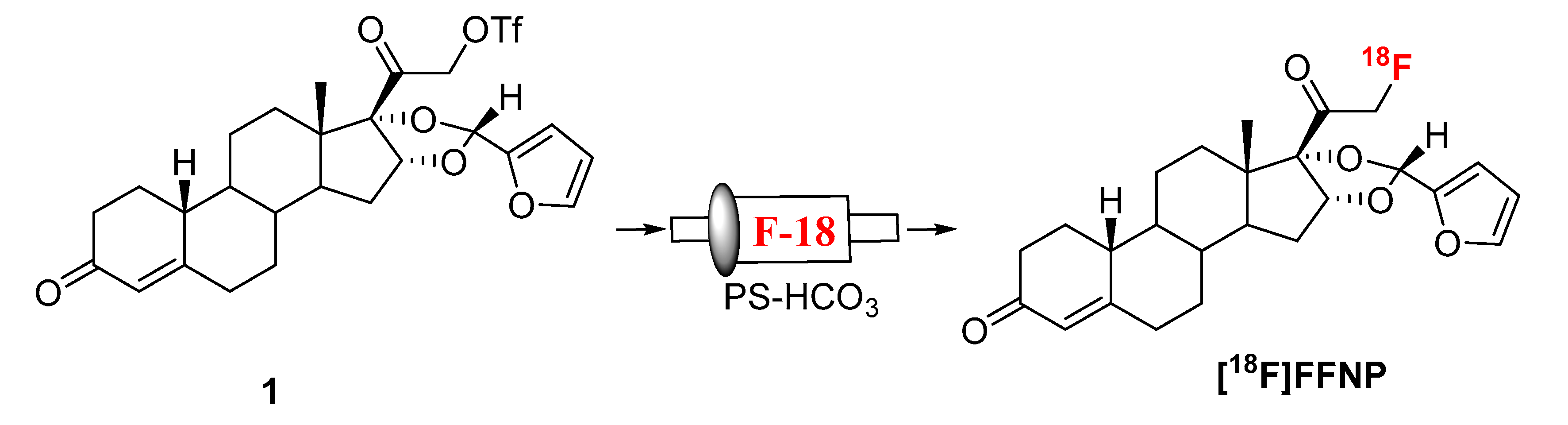
3.2. Radiosynthesis of [18F]FFNP
3.3. Radiosynthesis of [18F]FDHT
3.4. Preparation of Calibration Curves for Molar Activity Determination
3.5. In Vitro Studies
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Basuli, F.; Zhang, X.; Jagoda, E.M.; Choyke, P.L.; Swenson, R.E. Facile room temperature synthesis of fluorine-18 labeled fluoronicotinic acid-2,3,5,6-tetrafluorophenyl ester without azeotropic drying of fluorine-18. Nucl. Med. Biol. 2016, 43, 770–772. [Google Scholar] [CrossRef] [PubMed]
- Basuli, F.; Zhang, X.; Woodroofe, C.C.; Jagoda, E.M.; Choyke, P.L.; Swenson, R.E. Fast indirect fluorine-18 labeling of protein/peptide using the useful 6-fluoronicotinic acid-2,3,5,6-tetrafluorophenyl prosthetic group: A method comparable to direct fluorination. J. Labelled Comp. Radiopharm. 2017, 60, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Basuli, F.; Zhang, X.; Jagoda, E.M.; Choyke, P.L.; Swenson, R.E. Rapid synthesis of maleimide functionalized fluorine-18 labeled prosthetic group using “radio-fluorination on the Sep-Pak” method. J. Labelled Comp. Radiopharm. 2018, 61, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Basuli, F.; Swenson, R.E. An azeotropic drying-free approach for copper-mediated radiofluorination without addition of base. J. Labelled Comp. Radiopharm. 2019, 62, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed]
- Jatoi, I.; Chen, B.E.; Anderson, W.F.; Rosenberg, P.S. Breast cancer mortality trends in the United States according to estrogen receptor status and age at diagnosis. J. Clin. Oncol. 2007, 25, 1683–1690. [Google Scholar] [CrossRef] [PubMed]
- Dasgupta, S.; Lonard, D.M.; O’Malley, B.W. Nuclear receptor coactivators: Master regulators of human health and disease. Annu. Rev. Med. 2014, 65, 279–292. [Google Scholar] [CrossRef]
- Dalm, S.U.; Verzijlbergen, J.F.; De Jong, M. Review: Receptor Targeted Nuclear Imaging of Breast Cancer. Int. J. Mol. Sci. 2017, 18, 260. [Google Scholar] [CrossRef]
- Buckman, B.O.; Bonasera, T.A.; Kirschbaum, K.S.; Welch, M.J.; Katzenellenbogen, J.A. Fluorine-18-labeled progestin 16α, 17α-dioxolanes: Development of high-affinity ligands for the progesterone receptor with high in vivo target site selectivity. J. Med. Chem. 1995, 38, 328–337. [Google Scholar] [CrossRef]
- Salem, K.; Kumar, M.; Yan, Y.; Jeffery, J.J.; Kloepping, K.C.; Michel, C.J.; Powers, G.L.; Mahajan, A.M.; Fowler, A.M. Sensitivity and Isoform Specificity of 18F-Fluorofuranylnorprogesterone for Measuring Progesterone Receptor Protein Response to Estradiol Challenge in Breast Cancer. J. Nucl. Med. 2019, 60, 220–226. [Google Scholar] [CrossRef]
- Poelaert, F.; Van Praet, C.; Beerens, A.-S.; De Meerleer, G.; Fonteyne, V.; Ost, P.; Lumen, N. The Role of Androgen Receptor Expression in the Curative Treatment of Prostate Cancer with Radiotherapy: A Pilot Study. Biomed. Res. Int. 2015, 2015, 812815. [Google Scholar] [CrossRef] [PubMed]
- Dehdashti, F.; Laforest, R.; Gao, F.; Aft, R.L.; Dence, C.S.; Zhou, D.; Shoghi, K.I.; Siegel, B.A.; Katzenellenbogen, J.A.; Welch, M.J. Assessment of progesterone receptors in breast carcinoma by PET with 21-18F-fluoro-16α,17α -[(R)-(1′-α -furylmethylidene)dioxy]-19-norpregn- 4-ene-3,20-dione. J. Nucl. Med. 2012, 53, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Lin, M.; Yasui, N.; Al-Qahtani, M.H.; Dence, C.S.; Schwarz, S.; Katzenellenbogen, J.A. Optimization of the preparation of fluorine-18-labeled steroid receptor ligands 16α-[18F]fluoroestradiol (FES), [18F]fluoro furanyl norprogesterone (FFNP), and 16β -[18F]fluoro-5α-dihydrotestosterone (FDHT) as radiopharmaceuticals. J. Labelled Comp. Radiopharm. 2014, 57, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Salem, K.; Kumar, M.; Kloepping, K.C.; Michel, C.J.; Yan, Y.; Fowler, A.M. Determination of binding affinity of molecular imaging agents for steroid hormone receptors in breast cancer. Am. J. Nucl. Med. Mol. Imaging 2018, 8, 119–126. [Google Scholar] [PubMed]
- Joseph, D.R. Structure, function, and regulation of androgen-binding protein/sex hormone-binding globulin. Vitam. Horm. 1994, 49, 197–280. [Google Scholar] [PubMed]
- The Veterans Administration Co-operative Urological Research Group. Treatment and survival of patients with cancer of the prostate. Surg. Gynecol. Obstet. 1967, 124, 1011–1017. [Google Scholar]
- Agus, D.B.; Cordon-Cardo, C.; Fox, W.; Drobnjak, M.; Koff, A.; Golde, D.W.; Scher, H.I. Prostate cancer cell cycle regulators: Response to androgen withdrawal and development of androgen independence. J. Natl. Cancer. Inst. 1999, 91, 1869–1876. [Google Scholar] [CrossRef]
- Isaacs, J.T.; Coffey, D.S. Adaptation versus selection as the mechanism responsible for the relapse of prostatic cancer to androgen ablation therapy as studied in the Dunning R-3327-H adenocarcinoma. Cancer Res. 1981, 41, 5070–5075. [Google Scholar]
- Dehdashti, F.; Picus, J.; Michalski, J.M.; Dence, C.S.; Siegel, B.A.; Katzenellenbogen, J.A.; Welch, M.J. Positron tomographic assessment of androgen receptors in prostatic carcinoma. Eur. J. Nucl. Med. Mol. Imaging 2005, 32, 344–350. [Google Scholar] [CrossRef]
- Larson, S.M.; Morris, M.; Gunther, I.; Beattie, B.; Humm, J.L.; Akhurst, T.A.; Finn, R.D.; Erdi, Y.; Pentlow, K.; Dyke, J.; et al. Tumor localization of 16β-18F-fluoro-5α-dihydrotestosterone versus 18F-FDG in patients with progressive, metastatic prostate cancer. J. Nucl. Med. 2004, 45, 366–373. [Google Scholar]
- Zanzonico, P.B.; Finn, R.; Pentlow, K.S.; Erdi, Y.; Beattie, B.; Akhurst, T.; Squire, O.; Morris, M.; Scher, H.; McCarthy, T.; et al. PET-based radiation dosimetry in man of 18F-fluorodihydrotestosterone, a new radiotracer for imaging prostate cancer. J. Nucl. Med. 2004, 45, 1966–1971. [Google Scholar] [PubMed]
- Pandit-Taskar, N.; Veach, D.R.; Fox, J.J.; Scher, H.I.; Morris, M.J.; Larson, S.M. Evaluation of Castration-Resistant Prostate Cancer with Androgen Receptor–Axis Imaging. J. Nucl. Med. 2016, 57, 73S–78S. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Liu, A.; Dence, C.S.; Welch, M.J.; Katzenellenbogen, J.A. Fluorine-18-labeled androgens: Radiochemical synthesis and tissue distribution studies on six fluorine-substituted androgens, potential imaging agents for prostatic cancer. J. Nucl. Med. 1992, 33, 724–734. [Google Scholar]
- Ackermann, U.; Lewis, J.S.; Young, K.; Morris, M.J.; Weickhardt, A.; Davis, I.D.; Scott, A.M. Fully automated synthesis of [18F]fluoro-dihydrotestosterone ([18F]FDHT) using the FlexLab module. J. Labelled Comp. Radiopharm. 2016, 59, 424–428. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Dunlow, R.; Blackman, B.N.; Swenson, R.E. Optimization of 18F-syntheses using 19F-reagents at tracer-level concentrations and liquid chromatography/tandem mass spectrometry analysis: Improved synthesis of [18F]MDL100907. J. Labelled Comp. Radiopharm. 2018, 61, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Collier, T.L.; Dahl, K.; Stephenson, N.A.; Holland, J.P.; Riley, A.; Liang, S.H.; Vasdev, N. Recent applications of a single quadrupole mass spectrometer in 11C, 18F and radiometal chemistry. J. Fluor. Chem. 2018, 210, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Bauer, E.R.; Daxenberger, A.; Petri, T.; Sauerwein, H.; Meyer, H.H. Characterisation of the affinity of different anabolics and synthetic hormones to the human androgen receptor, human sex hormone binding globulin and to the bovine progestin receptor. APMIS 2000, 108, 838–846. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
| Tracer | Amount of Precursor 1 or 2 (mg) | Solvent (0.5 mL) | Eluted from the Sep-Pak (%) a |
|---|---|---|---|
| [18F]FFNP | 1.5 | Acetonitrile | 42 |
| 3 | Acetonitrile | 71–79 b | |
| 5 | Acetonitrile | 76 | |
| DMF | 40 | ||
| DMSO | 49 | ||
| THF | 36 |
| Compound | Amount of Precursor 1 or 2 (mg) | Solvent (0.5 mL) | Eluted from the Sep-Pak (%) a |
|---|---|---|---|
| 3 | 1.5 | Acetonitrile | 14 |
| 3 | Acetonitrile | 34 | |
| 5 | Acetonitrile | 52 | |
| DMF | 13 | ||
| DMSO | 27 | ||
| THF | 57 b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basuli, F.; Zhang, X.; Blackman, B.; White, M.E.; Jagoda, E.M.; Choyke, P.L.; Swenson, R.E. Fluorine-18 Labeled Fluorofuranylnorprogesterone ([18F]FFNP) and Dihydrotestosterone ([18F]FDHT) Prepared by “Fluorination on Sep-Pak” Method. Molecules 2019, 24, 2389. https://doi.org/10.3390/molecules24132389
Basuli F, Zhang X, Blackman B, White ME, Jagoda EM, Choyke PL, Swenson RE. Fluorine-18 Labeled Fluorofuranylnorprogesterone ([18F]FFNP) and Dihydrotestosterone ([18F]FDHT) Prepared by “Fluorination on Sep-Pak” Method. Molecules. 2019; 24(13):2389. https://doi.org/10.3390/molecules24132389
Chicago/Turabian StyleBasuli, Falguni, Xiang Zhang, Burchelle Blackman, Margaret E. White, Elaine M. Jagoda, Peter L. Choyke, and Rolf E. Swenson. 2019. "Fluorine-18 Labeled Fluorofuranylnorprogesterone ([18F]FFNP) and Dihydrotestosterone ([18F]FDHT) Prepared by “Fluorination on Sep-Pak” Method" Molecules 24, no. 13: 2389. https://doi.org/10.3390/molecules24132389
APA StyleBasuli, F., Zhang, X., Blackman, B., White, M. E., Jagoda, E. M., Choyke, P. L., & Swenson, R. E. (2019). Fluorine-18 Labeled Fluorofuranylnorprogesterone ([18F]FFNP) and Dihydrotestosterone ([18F]FDHT) Prepared by “Fluorination on Sep-Pak” Method. Molecules, 24(13), 2389. https://doi.org/10.3390/molecules24132389





