Ultrasonic Processing Induced Activity and Structural Changes of Polyphenol Oxidase in Orange (Citrus sinensis Osbeck)
Abstract
1. Introduction
2. Results and Discussion
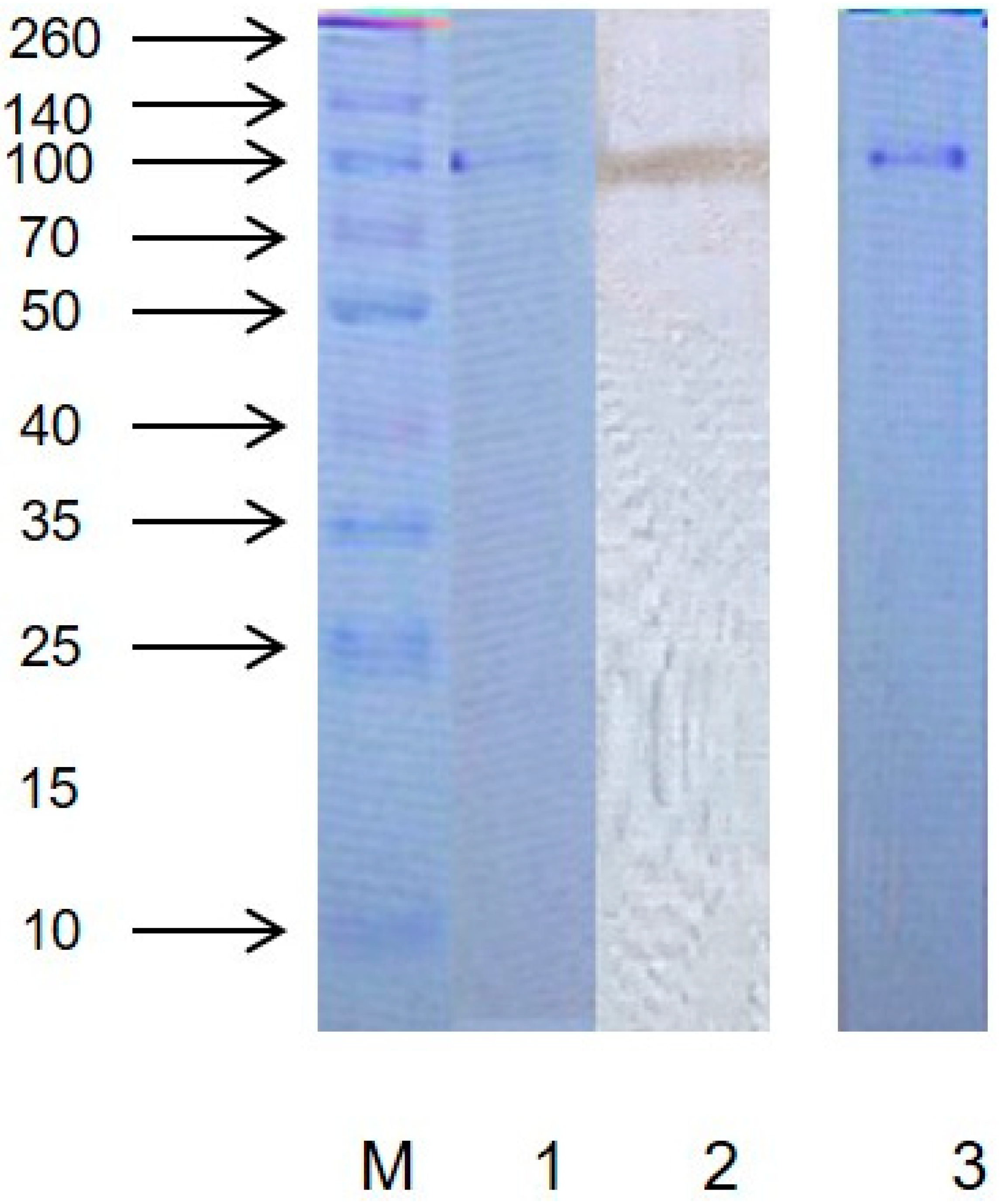
2.1. Purity and Molecular Weight
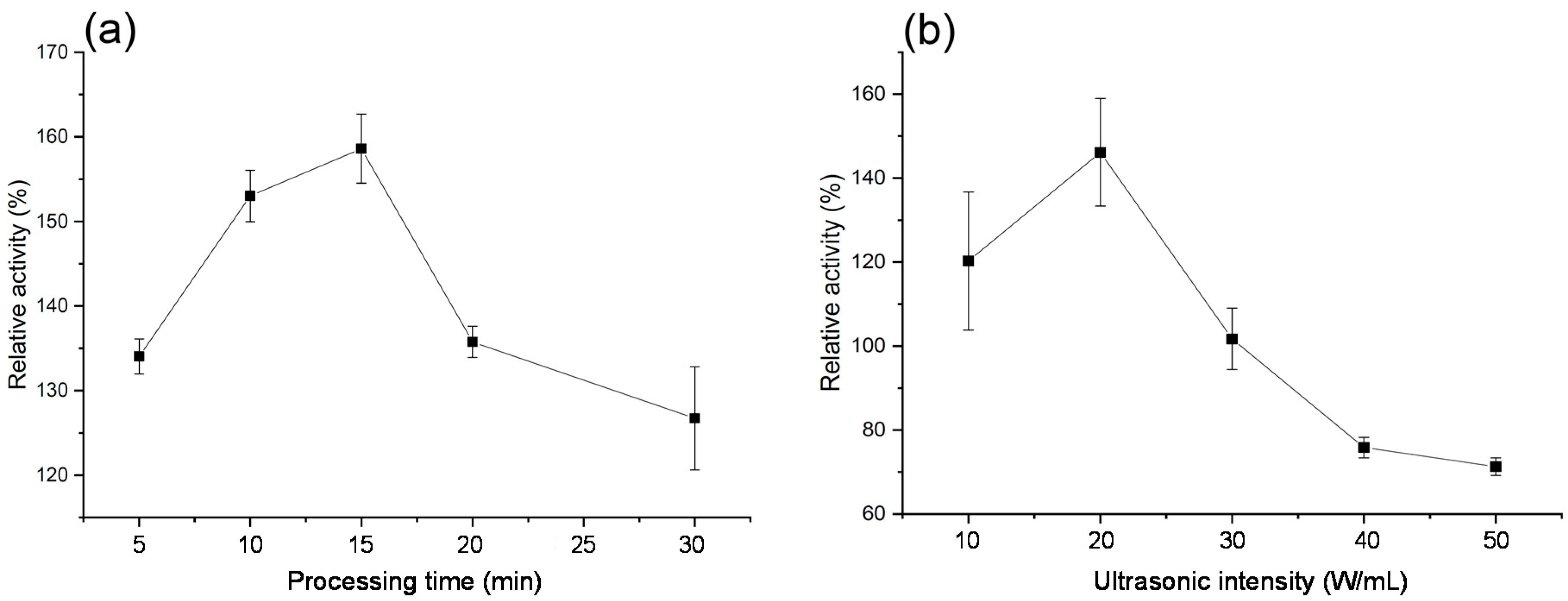
2.2. Effect of Ultrasonication on PPO Activity
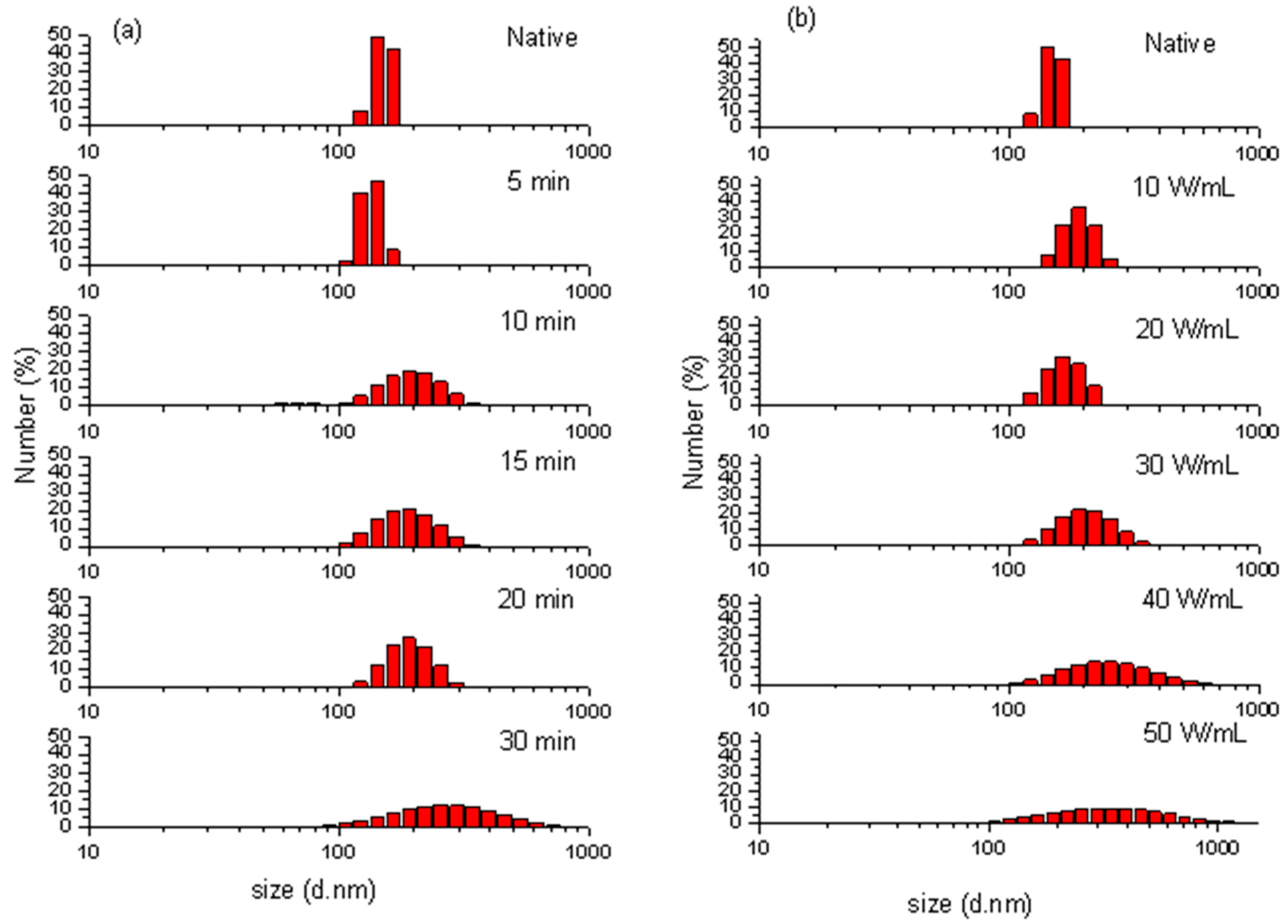
2.3. Particle Size Distribution
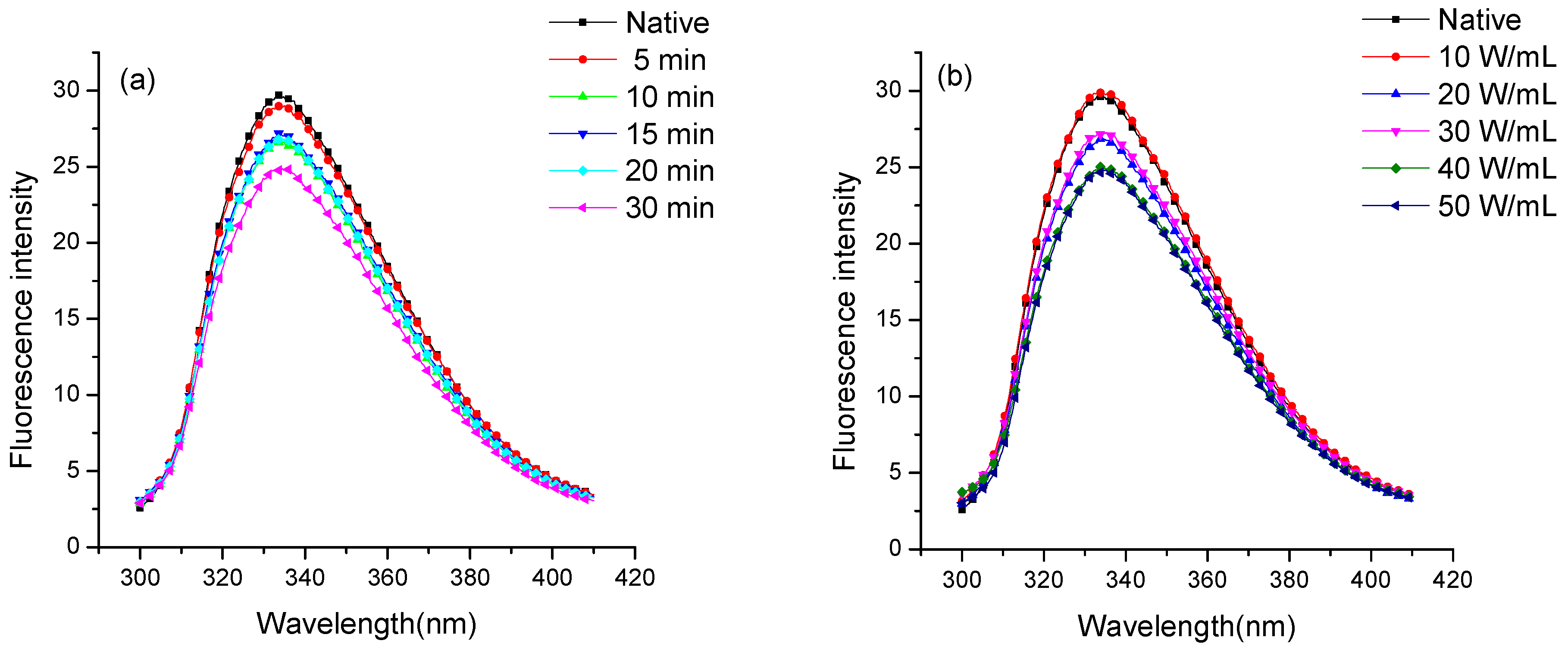
2.4. Fluorescence Spectroscopic Analysis
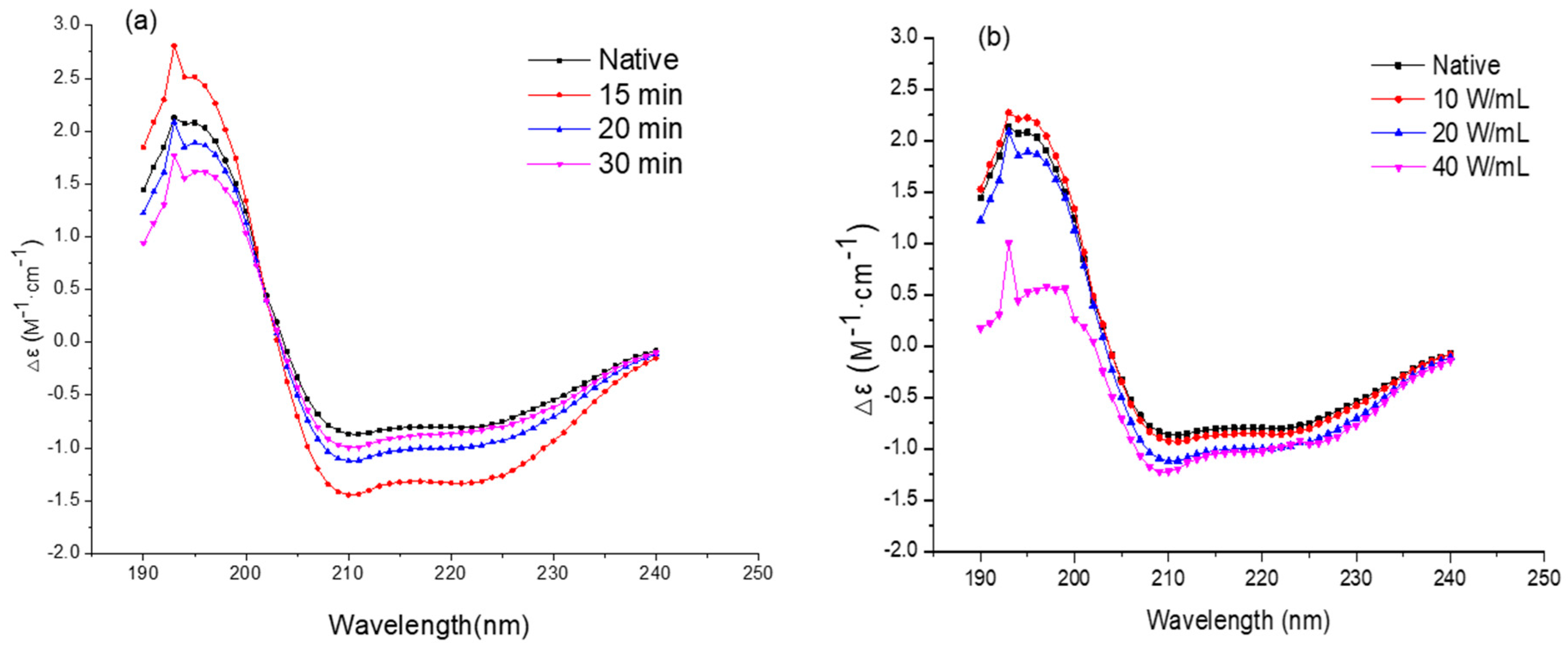
2.5. Circular Dichroism Spectroscopy Analysis
3. Materials and Methods
3.1. Materials and Chemicals
3.2. Extraction and Partial Purification of Protein
3.3. Electrophoresis Assay
3.4. Protein Content
3.5. Ultrasonic Processing
3.6. PPO Activity Assay
3.7. Particle Size Distribution Analysis
3.8. CD Spectral Measurement
3.9. Fluorescence Spectral Measurement
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Kacem, B.; Matthews, R.F.; Crandall, P.G.; Cornell, J.A. Nonenzymatic browning in aseptically packaged orange juice and orange drinks. Effect of amino acids, deaeration, and anaerobic storage. J. Food Sci. 1987, 52, 1665–1667. [Google Scholar] [CrossRef]
- Landi, M.; Degl’innocenti, E.; Guglielminetti, L.; Guidi, L. Role of ascorbic acid in the inhibition of polyphenol oxidase and the prevention of browning in different browning-sensitive Lactuca sativa var. capitata (L.) and Eruca sativa (Mill.) stored as fresh-cut produce. J. Sci. Food Agric. 2013, 93. [Google Scholar] [CrossRef]
- Huang, N.; Cheng, X.; Hu, W.; Pan, S. Inactivation, aggregation, secondary and tertiary structural changes of germin-like protein in Satsuma mandarine with high polyphenol oxidase activity induced by ultrasonic processing. Biophys. Chem. 2015, 197, 18–24. [Google Scholar] [CrossRef]
- Zhao, G.Y.; Li, B. Studies on the occurrence of non-enzymatic browning during the storage of cloudy apple juice. Proc. 2007 Int. Conf. Agric. Eng. 2007, 32, 634–643. [Google Scholar]
- Cheng, X.; Huang, X.; Liu, S.; Tang, M.; Hu, W.; Pan, S. Characterization of germin-like protein with polyphenol oxidase activity from Satsuma mandarine. Biochem. Biophys. Res. Commun. 2014, 449, 313–318. [Google Scholar] [CrossRef]
- Vamos-Vigyázó, L. Polyphenol Oxidase and Peroxidase in Fruits and Vegetables. CRC Crit. Rev. Food Sci. Nutr. 1981, 15, 49–127. [Google Scholar] [CrossRef]
- Jang, J.H.; Moon, K.D. Inhibition of polyphenol oxidase and peroxidase activities on fresh-cut apple by simultaneous treatment of ultrasound and ascorbic acid. Food Chem. 2011, 124, 444–449. [Google Scholar] [CrossRef]
- Murtaza, A.; Muhammad, Z.; Iqbal, A.; Ramzan, R.; Liu, Y.; Pan, S.; Hu, W. Aggregation and Conformational Changes in Native and Thermally Treated Polyphenol Oxidase From Apple Juice (Malus domestica). Front. Chem. 2018, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Fonteles, T.V.; Costa, M.G.M.; de Jesus, A.L.T.; de Miranda, M.R.A.; Fernandes, F.A.N.; Rodrigues, S. Power ultrasound processing of cantaloupe melon juice: Effects on quality parameters. Food Res. Int. 2012, 48, 41–48. [Google Scholar] [CrossRef]
- Liu, S.; Liu, Y.; Huang, X.; Yang, W.; Hu, W.; Pan, S. Effect of ultrasonic processing on the changes in activity, aggregation and the secondary and tertiary structure of polyphenol oxidase in oriental sweet melon (Cucumis melo var. makuwa Makino). J. Sci. Food Agric. 2017, 97, 1326–1334. [Google Scholar] [CrossRef] [PubMed]
- Illera, A.E.; Sanz, M.T.; Benito-Román, O.; Varona, S.; Beltrán, S.; Melgosa, R.; Solaesa, A.G. Effect of thermosonication batch treatment on enzyme inactivation kinetics and other quality parameters of cloudy apple juice. Innov. Food Sci. Emerg. Technol. 2018, 47, 71–80. [Google Scholar] [CrossRef]
- Yang, C.; Fujita, S.; Nakamura, N. Purification and Characterization of Polyphenol Oxidase from Banana (Musa sapientum L.) Pulp. J. Agric. Food Chem. 2000, 48, 2732–2735. [Google Scholar] [CrossRef] [PubMed]
- Wititsuwannakul, D.; Chareonthiphakorn, N.; Pace, M.; Wititsuwannakul, R. Polyphenol oxidases from latex of Hevea brasiliensis: Purification and characterization. Phytochemistry 2002, 61, 115–121. [Google Scholar] [CrossRef]
- Xu, J.; Zheng, T.; Meguro, S.; Kawachi, S. Purification and characterization of polyphenol oxidase from Henry chestnuts (Castanea henryi). J. Wood Sci. 2004, 50, 260–265. [Google Scholar] [CrossRef]
- Cheng, X.F.; Zhang, M.; Adhikari, B. The inactivation kinetics of polyphenol oxidase in mushroom (Agaricus bisporus) during thermal and thermosonic treatments. Ultrason. Sonochem. 2013, 20, 674–679. [Google Scholar] [CrossRef]
- Ma, H.; Huang, L.; Jia, J.; He, R.; Luo, L.; Zhu, W. Effect of energy-gathered ultrasound on Alcalase. Ultrason. Sonochem. 2011, 18, 419–424. [Google Scholar] [CrossRef]
- Li, R.; Wang, Y.; Hu, W.; Liao, X. Changes in the activity, dissociation, aggregation, and the secondary and tertiary structures of a thaumatin-like protein with a high polyphenol oxidase activity induced by high pressure CO2. Innov. Food Sci. Emerg. Technol. 2014, 23, 68–78. [Google Scholar] [CrossRef]
- Jambrak, A.R.; Mason, T.J.; Lelas, V.; Paniwnyk, L.; Herceg, Z. Effect of ultrasound treatment on particle size and molecular weight of whey proteins. J. Food Eng. 2014, 121, 15–23. [Google Scholar] [CrossRef]
- Bi, X.; Hemar, Y.; Balaban, M.O.; Liao, X. The effect of ultrasound on particle size, color, viscosity and polyphenol oxidase activity of diluted avocado puree. Ultrason. Sonochem. 2015, 27, 567–575. [Google Scholar] [CrossRef]
- Gülseren, I.; Güzey, D.; Bruce, B.D.; Weiss, J. Structural and functional changes in ultrasonicated bovine serum albumin solutions. Ultrason. Sonochem. 2007, 14, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Chandrapala, J.; Zisu, B.; Palmer, M.; Kentish, S.; Ashokkumar, M. Effects of ultrasound on the thermal and structural characteristics of proteins in reconstituted whey protein concentrate. Ultrason. Sonochem. 2011, 18, 951–957. [Google Scholar] [CrossRef] [PubMed]
- Vivian, J.T.; Callis, P.R. Mechanisms of tryptophan fluorescence shifts in proteins. Biophys. J. 2001, 80, 2093–2109. [Google Scholar] [CrossRef]
- Carvalho, A.S.L.; Ferreira, B.S.; Neves-Petersen, M.T.; Petersen, S.B.; Aires-Barros, M.R.; Melo, E.P. Thermal denaturation of HRPA2: pH-dependent conformational changes. Enzyme Microb. Technol. 2007, 40, 696–703. [Google Scholar] [CrossRef]
- Hu, W.; Zhang, Y.; Wang, Y.; Zhou, L.; Leng, X.; Liao, X.; Hu, X. Aggregation and homogenization, surface charge and structural change, and inactivation of mushroom tyrosinase in an aqueous system by subcritical/supercritical carbon dioxide. Langmuir 2011, 27, 909–916. [Google Scholar] [CrossRef] [PubMed]
- Benito-román, Ó.; Sanz, M.T.; Melgosa, R.; de Paz, E.; Escudero, I. Studies of polyphenol oxidase inactivation by means of high pressure carbon dioxide (HPCD). J. Supercrit. Fluids 2019, 147, 310–321. [Google Scholar] [CrossRef]
- Kelly, S.M.; Jess, T.J.; Price, N.C. How to study proteins by circular dichroism. Biochim. Biophys. Acta - Proteins Proteomics 2005, 1751, 119–139. [Google Scholar] [CrossRef] [PubMed]
- Greenfield, N.J. Applications of circular dichroism in protein and peptide analysis. TrAC - Trends Anal. Chem. 1999, 18, 236–244. [Google Scholar] [CrossRef]
- Barteri, M.; Diociaiuti, M.; Pala, A.; Rotella, S. Low frequency ultrasound induces aggregation of porcine fumarase by free radicals production. Biophys. Chem. 2004, 111, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.L.; Zeng, W.C.; Lu, X.L. Influence of ultrasound to the activity of tyrosinase. Ultrason. Sonochem. 2013, 20, 805–809. [Google Scholar] [CrossRef] [PubMed]
- Davis, B.J. Disc Electrophoresis ? II Method and Application to Human Serum Proteins. Ann. N. Y. Acad. Sci. 1964, 121, 404–427. [Google Scholar] [CrossRef] [PubMed]
- Aydemir, T. Partial purification and characterization of polyphenol oxidase from artichoke (Cynara scolymus L.) heads. Food Chem. 2004, 87, 59–67. [Google Scholar] [CrossRef]
- Bradford, M.M. A Rapid and Sensitive Method for the quantitation of Mocrigram Quantities of Protein Utilizing the Principle of Protein-Dye Binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Liu, S.; Murtaza, A.; Liu, Y.; Hu, W.; Xu, X.; Pan, S. Catalytic and Structural Characterization of a Browning-Related Protein in Oriental Sweet Melon (Cucumis Melo var. Makuwa Makino). Front. Chem. 2018, 6, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Murtaza, A.; Iqbal, A.; Linhu, Z.; Liu, Y.; Xu, X.; Pan, S.; Hu, W. Effect of high-pressure carbon dioxide on the aggregation and conformational changes of polyphenol oxidase from apple (Malus domestica) juice. Innov. Food Sci. Emerg. Technol. 2019. [Google Scholar] [CrossRef]
- Iqbal, A.; Murtaza, A.; Muhammad, Z.; Elkhedir, A.; Tao, M.; Xu, X. Inactivation, Aggregation and Conformational Changes of Polyphenol Oxidase from Quince (Cydonia oblonga Miller) Juice Subjected to Thermal and High-Pressure Carbon Dioxide Treatment. Molecules 2018, 23, 1743. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
| Samples | α-Helix | β-Sheet | β-Turn | Random Coil |
|---|---|---|---|---|
| Native | 76.20% | 0.00% | 23.60% | 0.10% |
| 10 W/mL/20 min | 76.00% | 0.00% | 24.00% | 0.00% |
| 20 W/mL/20 min | 53.30% | 0.00% | 29.30% | 17.50% |
| 40 W/mL/20 min | 32.80% | 0.00% | 33.60% | 33.60% |
| 20 W/mL/15 min | 54.60% | 0.00% | 26.50% | 18.90% |
| 20 W/mL/20 min | 53.30% | 0.00% | 29.30% | 17.50% |
| 20 W/mL/30 min | 51.80% | 0.00% | 31.10% | 17.20% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, L.; Zhu, L.; Murtaza, A.; Liu, Y.; Liu, S.; Li, J.; Iqbal, A.; Xu, X.; Pan, S.; Hu, W. Ultrasonic Processing Induced Activity and Structural Changes of Polyphenol Oxidase in Orange (Citrus sinensis Osbeck). Molecules 2019, 24, 1922. https://doi.org/10.3390/molecules24101922
Zhu L, Zhu L, Murtaza A, Liu Y, Liu S, Li J, Iqbal A, Xu X, Pan S, Hu W. Ultrasonic Processing Induced Activity and Structural Changes of Polyphenol Oxidase in Orange (Citrus sinensis Osbeck). Molecules. 2019; 24(10):1922. https://doi.org/10.3390/molecules24101922
Chicago/Turabian StyleZhu, Lijuan, Linhu Zhu, Ayesha Murtaza, Yan Liu, Siyu Liu, Junjie Li, Aamir Iqbal, Xiaoyun Xu, Siyi Pan, and Wanfeng Hu. 2019. "Ultrasonic Processing Induced Activity and Structural Changes of Polyphenol Oxidase in Orange (Citrus sinensis Osbeck)" Molecules 24, no. 10: 1922. https://doi.org/10.3390/molecules24101922
APA StyleZhu, L., Zhu, L., Murtaza, A., Liu, Y., Liu, S., Li, J., Iqbal, A., Xu, X., Pan, S., & Hu, W. (2019). Ultrasonic Processing Induced Activity and Structural Changes of Polyphenol Oxidase in Orange (Citrus sinensis Osbeck). Molecules, 24(10), 1922. https://doi.org/10.3390/molecules24101922






