Carboxyxanthones: Bioactive Agents and Molecular Scaffold for Synthesis of Analogues and Derivatives
Abstract
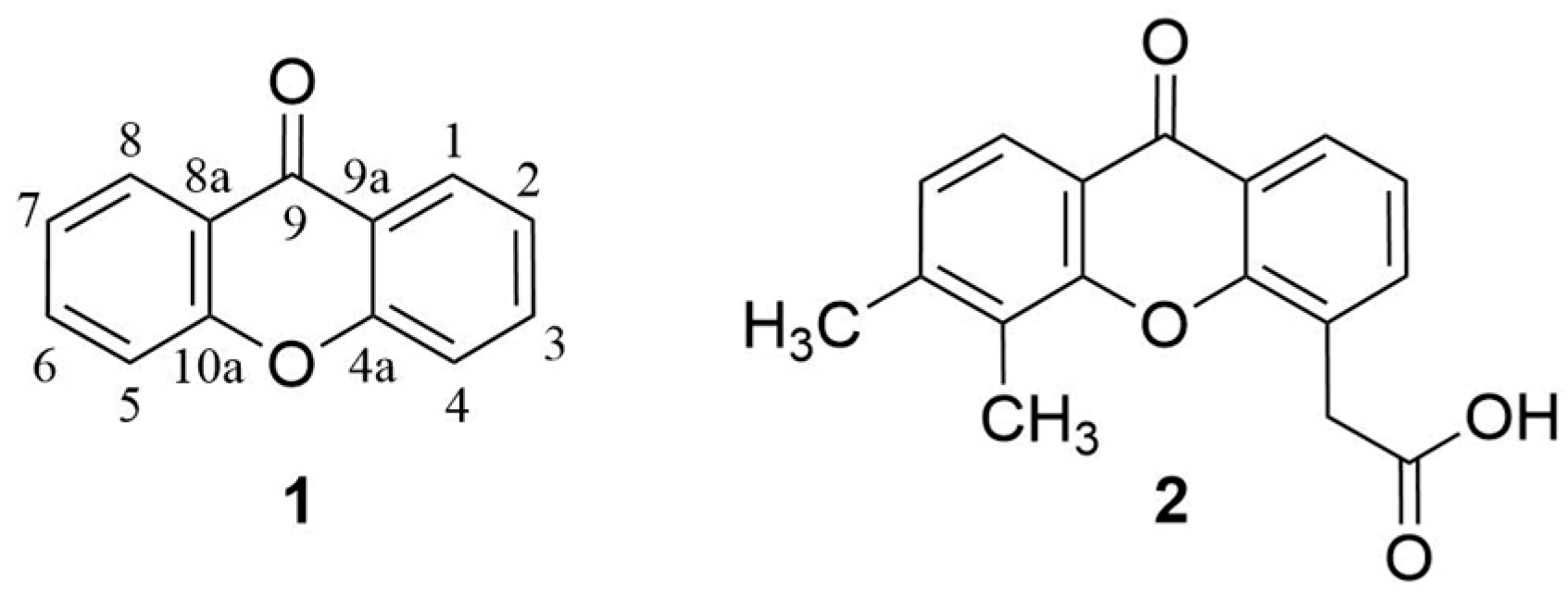
1. Introduction
2. Natural Carboxyxanthone Derivatives
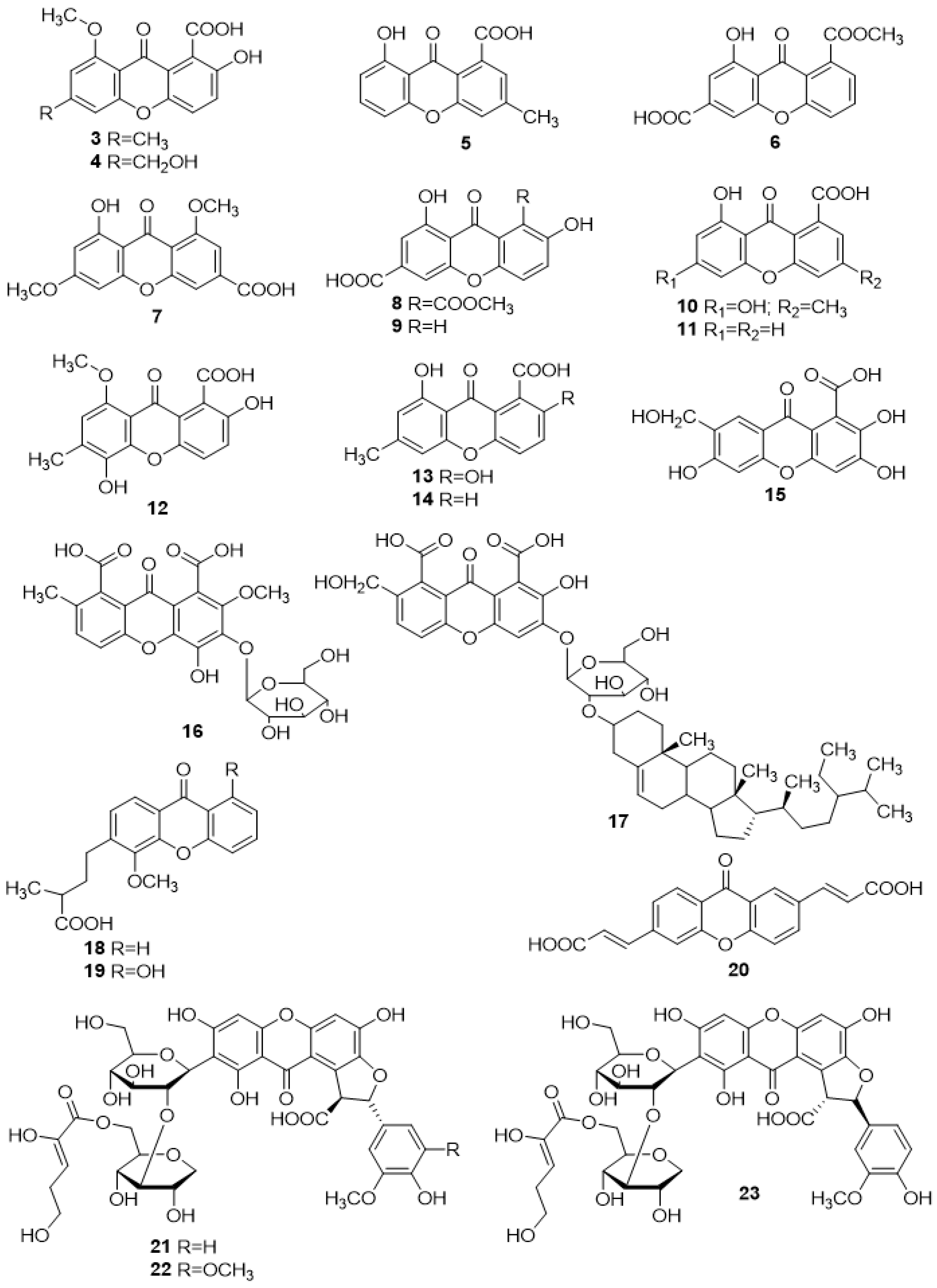
2.1. Simple Carboxyxanthone Derivatives
2.1.1. 2-Hydroxy-6-Methyl-8-Methoxy-9-oxo-9H-Xanthene-1-Carboxylic Acid (3) and 2-Hydroxy-6-Hydroxymethyl-8-Methoxy-9-Oxo-9H-Xanthene-1-Carboxylic Acid (4)
2.1.2. Monodictyxanthone (5)
2.1.3. 8-(Methoxycarbonyl)-1-Hydroxy-9-Oxo-9H-Xanthene-3-Carboxylic Acid (6)
2.1.4. Yicathin C (7)
2.1.5. 2,8-Dihydroxy-1-Methoxycarbonyl-9-Oxo-9H-Xanthene-6-Carboxylic Acid (8) and 2,8-Dihydroxy-9-Oxo-9H-Xanthene-6-Carboxylic acid (9)
2.1.6. 6,8-Dihydroxy-3-Methyl-9-Oxo-9H-Xanthene-1-Carboxylic Acid (10)
2.1.7. Globosuxanthone D (11)
2.1.8. 2,5-Dihydroxy-8-Methoxy-6-Methyl-9-Oxo-9H-Xanthene-1-Carboxylic Acid (12)
2.1.9. Pinselic Acid (13)
2.1.10. 8-Hydroxy-6-Methyl-9-Oxo-9H-Xanthene-1-Carboxylic Acid (14)
2.1.11. 2,3,6-Trihydroxy-7-Hydroxymethylene Xanthone-1-Carboxylic Acid (15) and Glycosilated Analogues (16–17)
2.1.12. Scriblitifolic Acid (18) and Teysmannic Acid (19)
2.1.13. (2E,2′E)-3,3′-(9-Oxo-9H-Xanthene-2,6-Diyl)Diacrylic Acid (20)
2.1.14. Glomexanthones A–C (21–23)
2.2. Prenylated Carboxyxanthone Derivatives
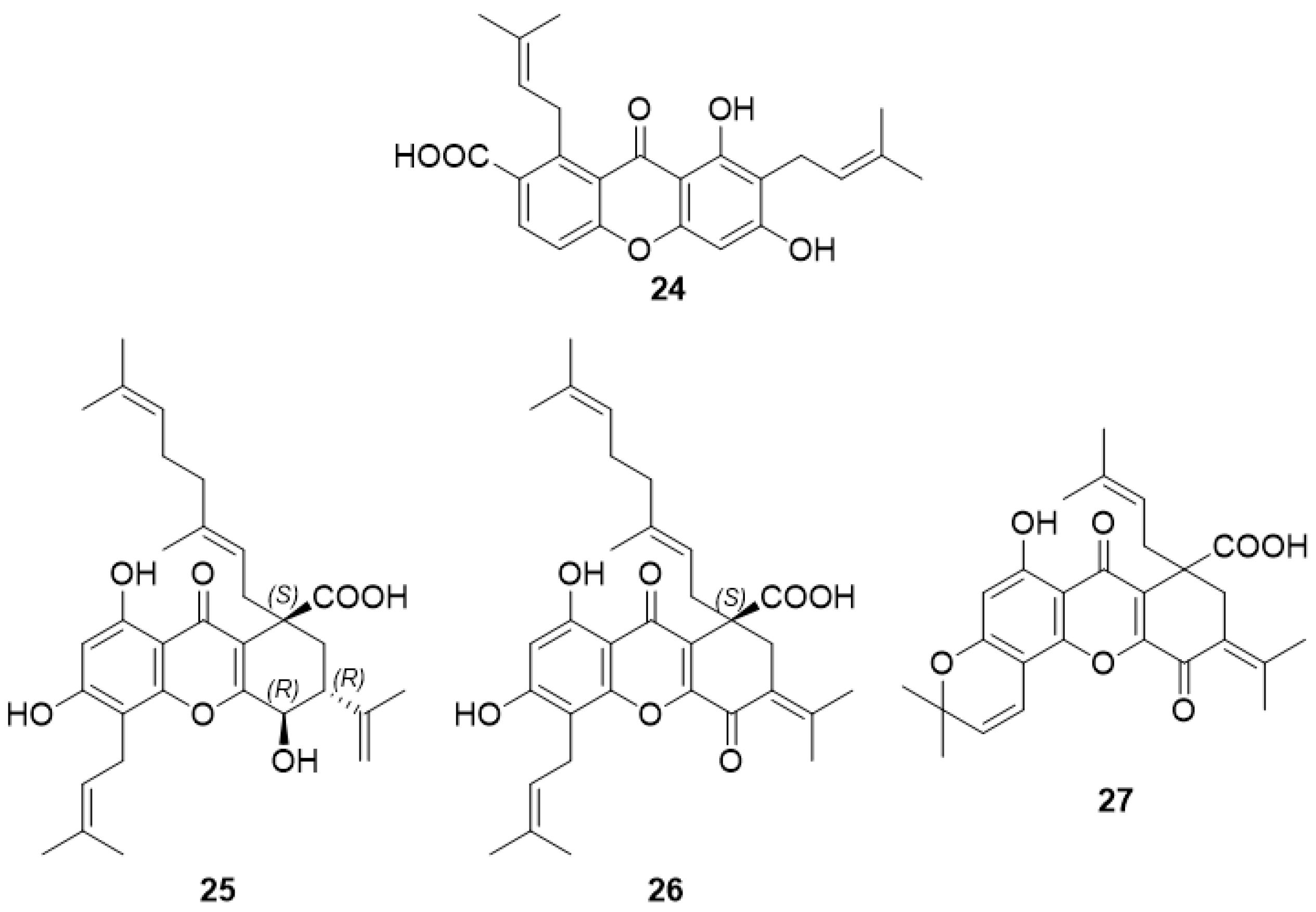
2.2.1. 2,8-Di-(3-Methylbut-2-Enyl)-1,3,8-Trihydroxy-4-Methyl-Xanthone (24)
2.2.2. Oliganthic Acid A (25), Oliganthic Acid B (26), and (±)-Oliganthic Acid C (27)
2.3. Caged Carboxyxanthone Derivatives
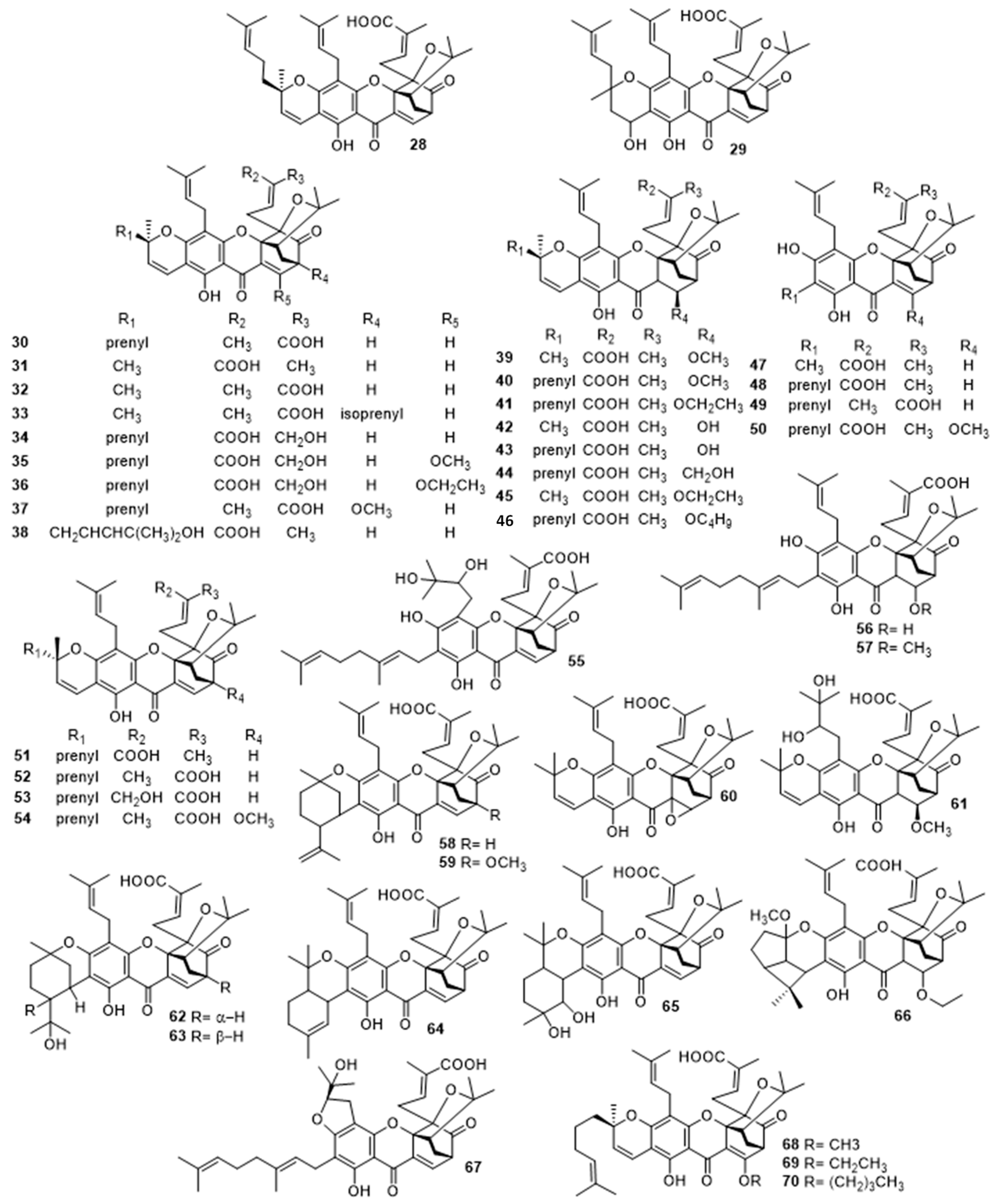
2.3.1. Gambogic Acid (28) and Analogues (29–70)
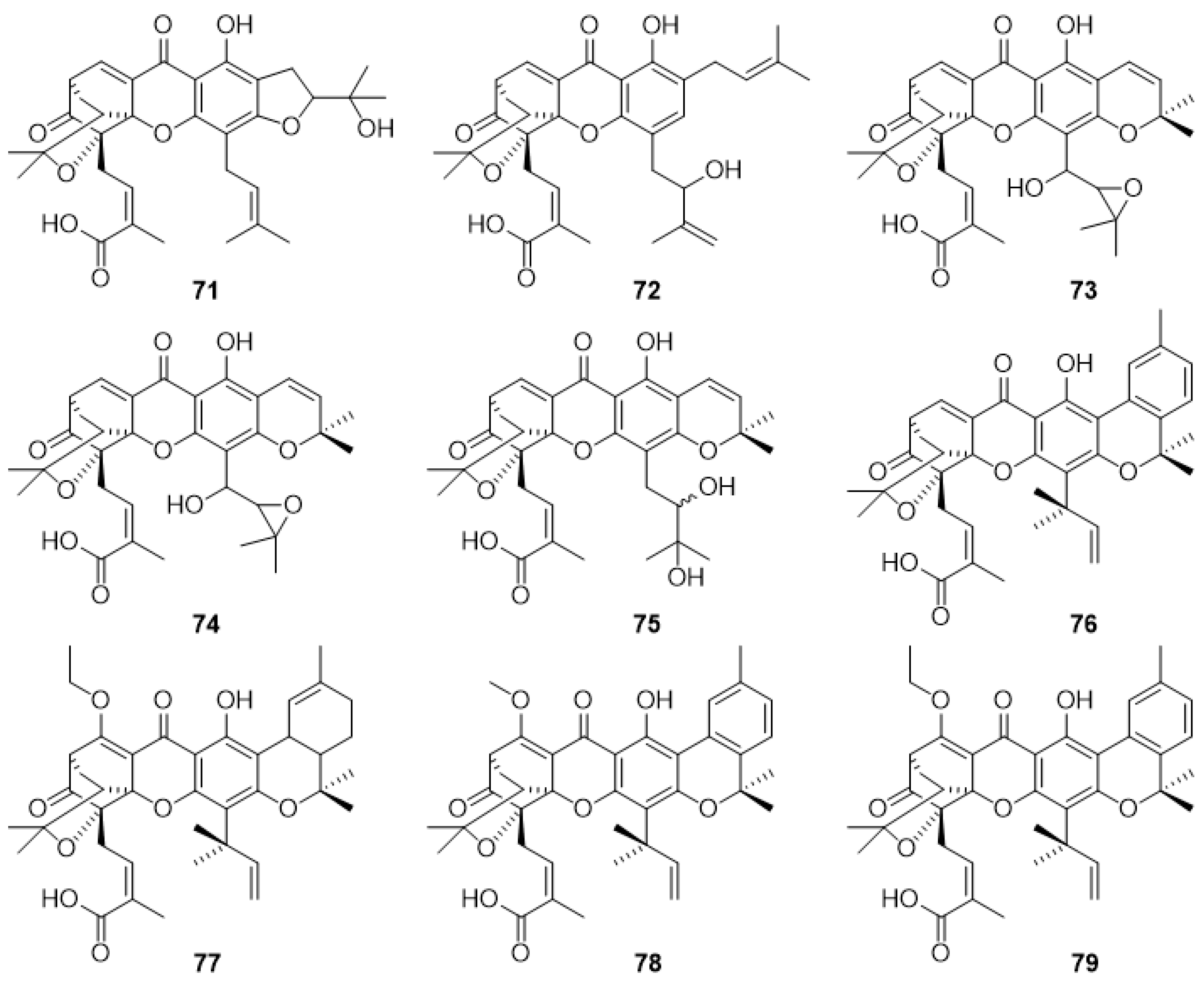
2.3.2. Gaudichaudiic Acids A–I (71–79)
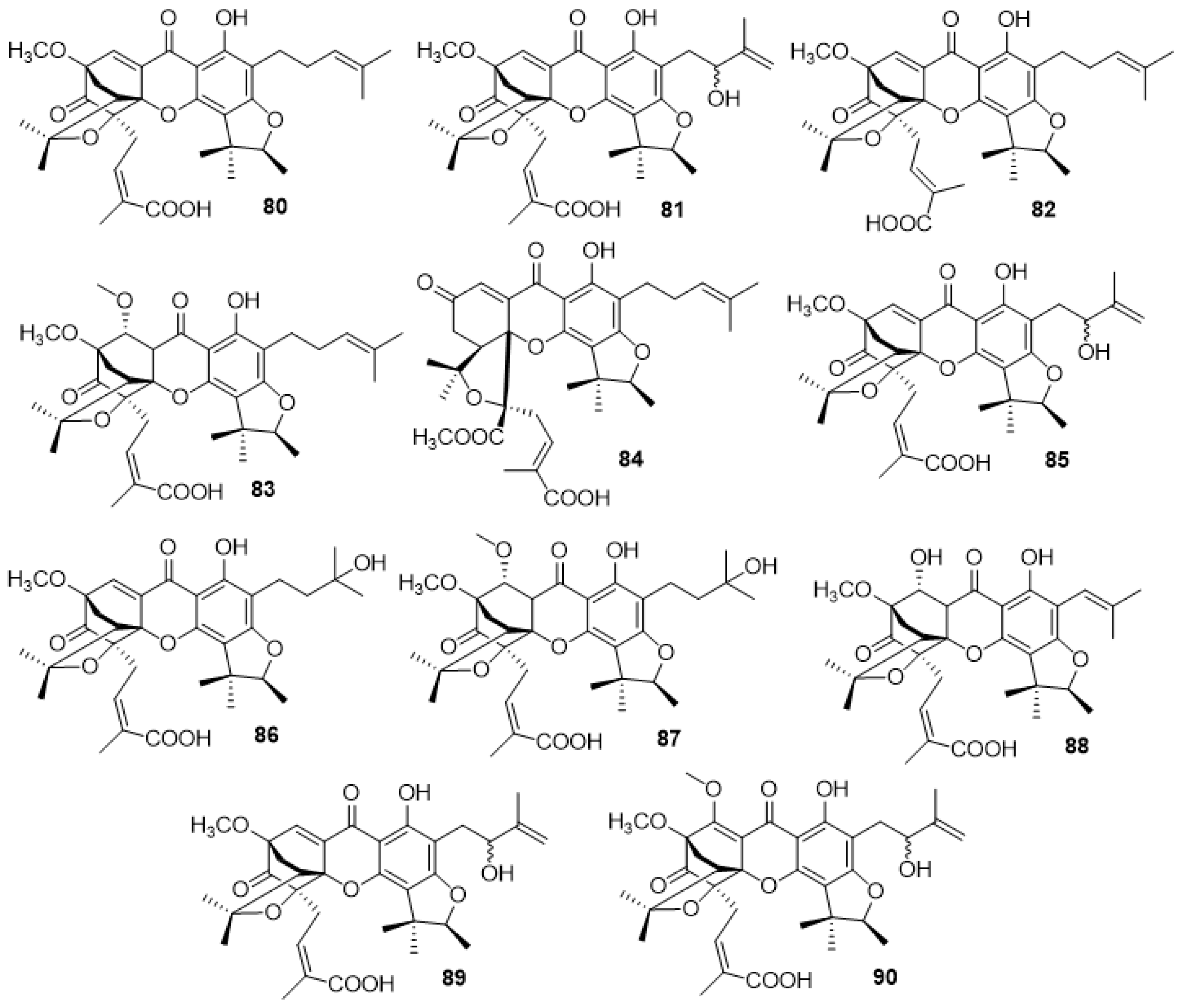
2.3.3. Scortechinones (80–90)
2.4. Carboxyxanthone Derivatives Bound or Fused to Polysubstituted Oxygenated Heterocycles
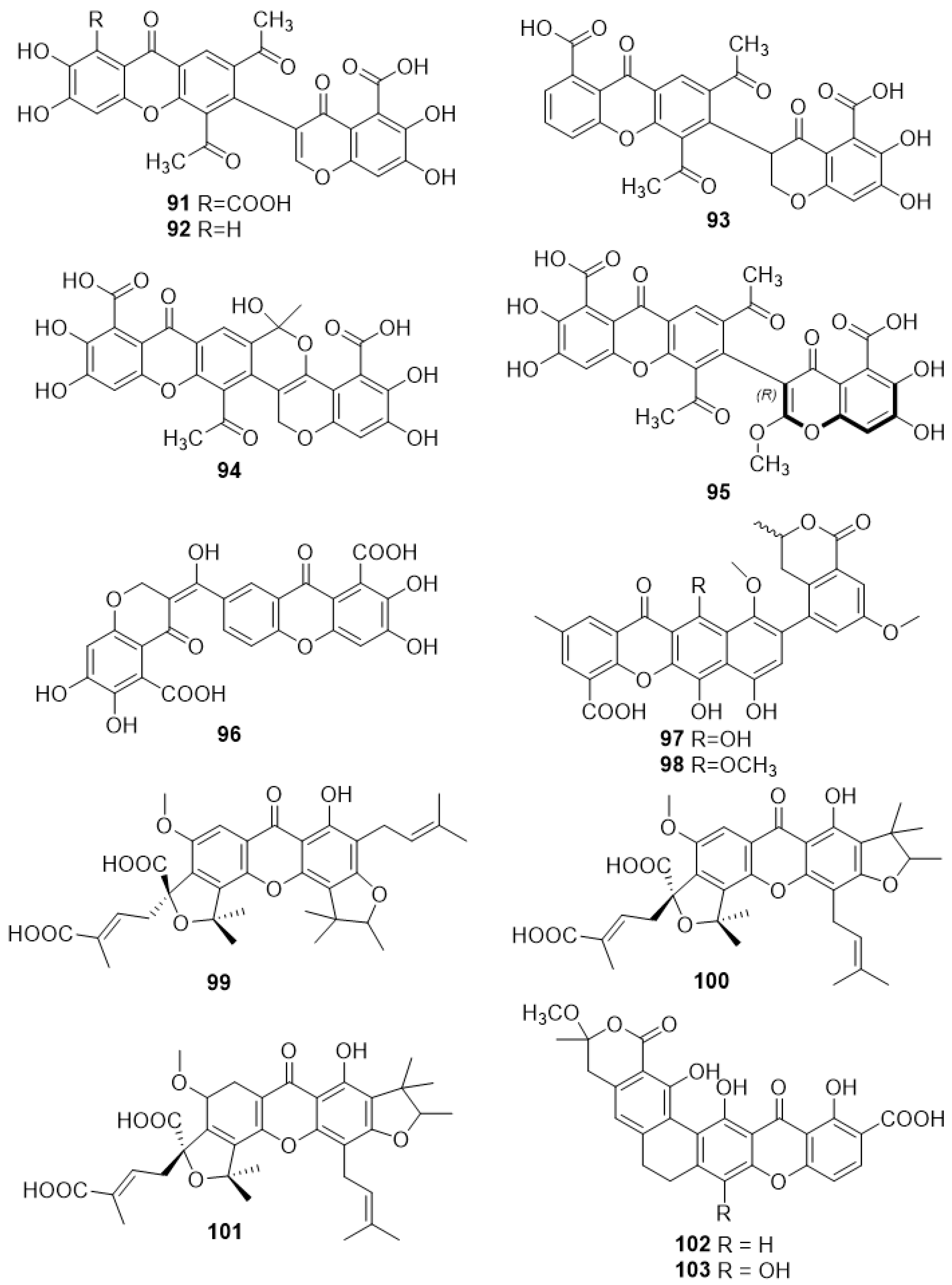
2.4.1. Vinaxanthone 411F (91) and Analogues (92–95)
2.4.2. Xanthofulvin (96)
2.4.3. 6,7,11-Trihydroxy-10-Methoxy-9-(7-Methoxy-3-Methyl-1-Oxoisochroman-5-yl)-2-Methyl-12-Oxo-12H-Benzo[b]Xanthene-4-Carboxylic Acid (97) and 6,7-Dihydroxy-10,11-Dimethoxy-9-(7-Methoxy-3-Methyl-1-Oxoisochroman-5-yl)-2-Methyl-12-Oxo-12H-Benzo[b]Xanthene-4-Carboxylic Acid (98)
2.4.4. Scortechinones V (99), W (100) and X (101)
2.4.5. Dehydrocitreaglycon A (102) and Citreaglycon A (103)
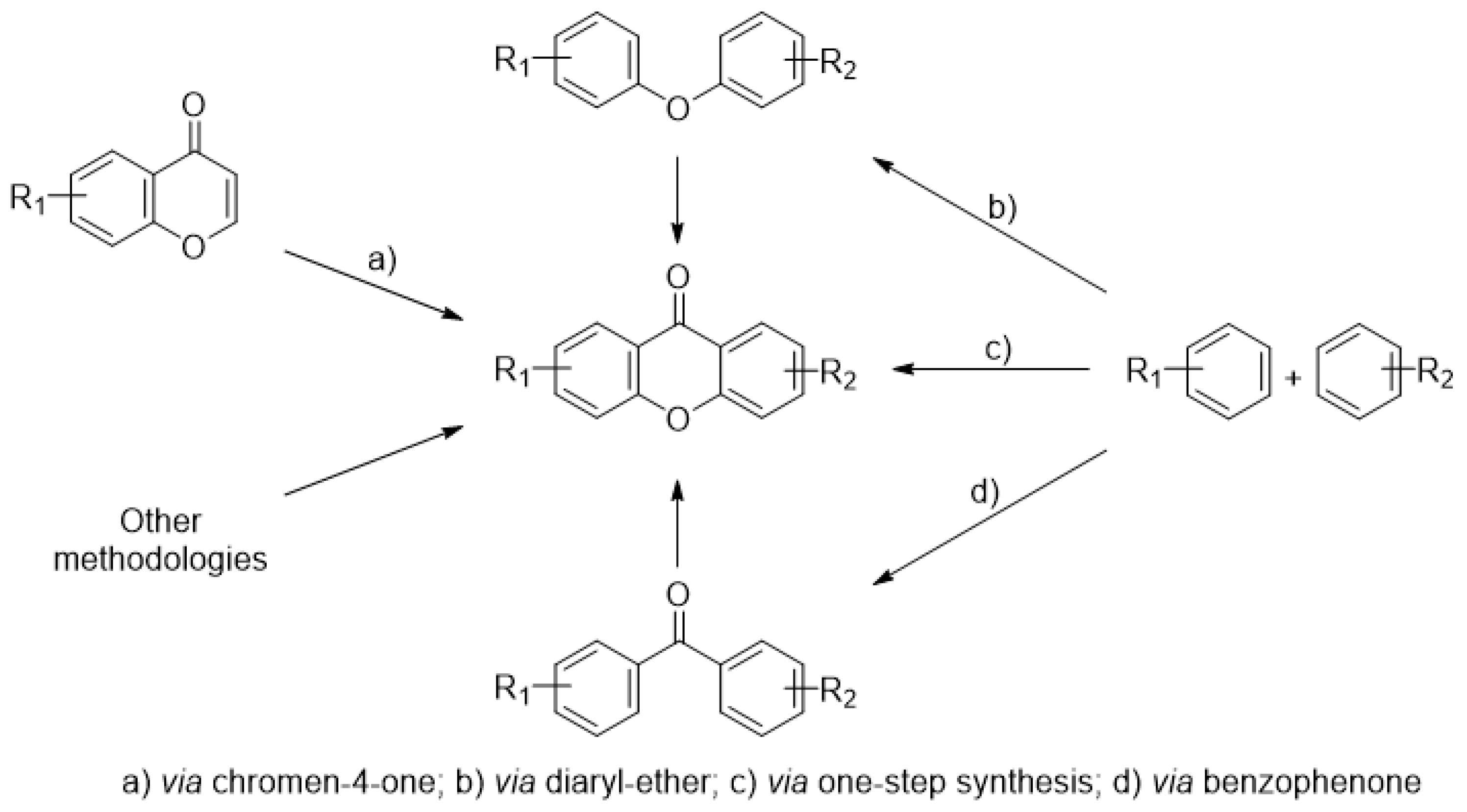
3. Synthetic Carboxyxanthone Derivatives
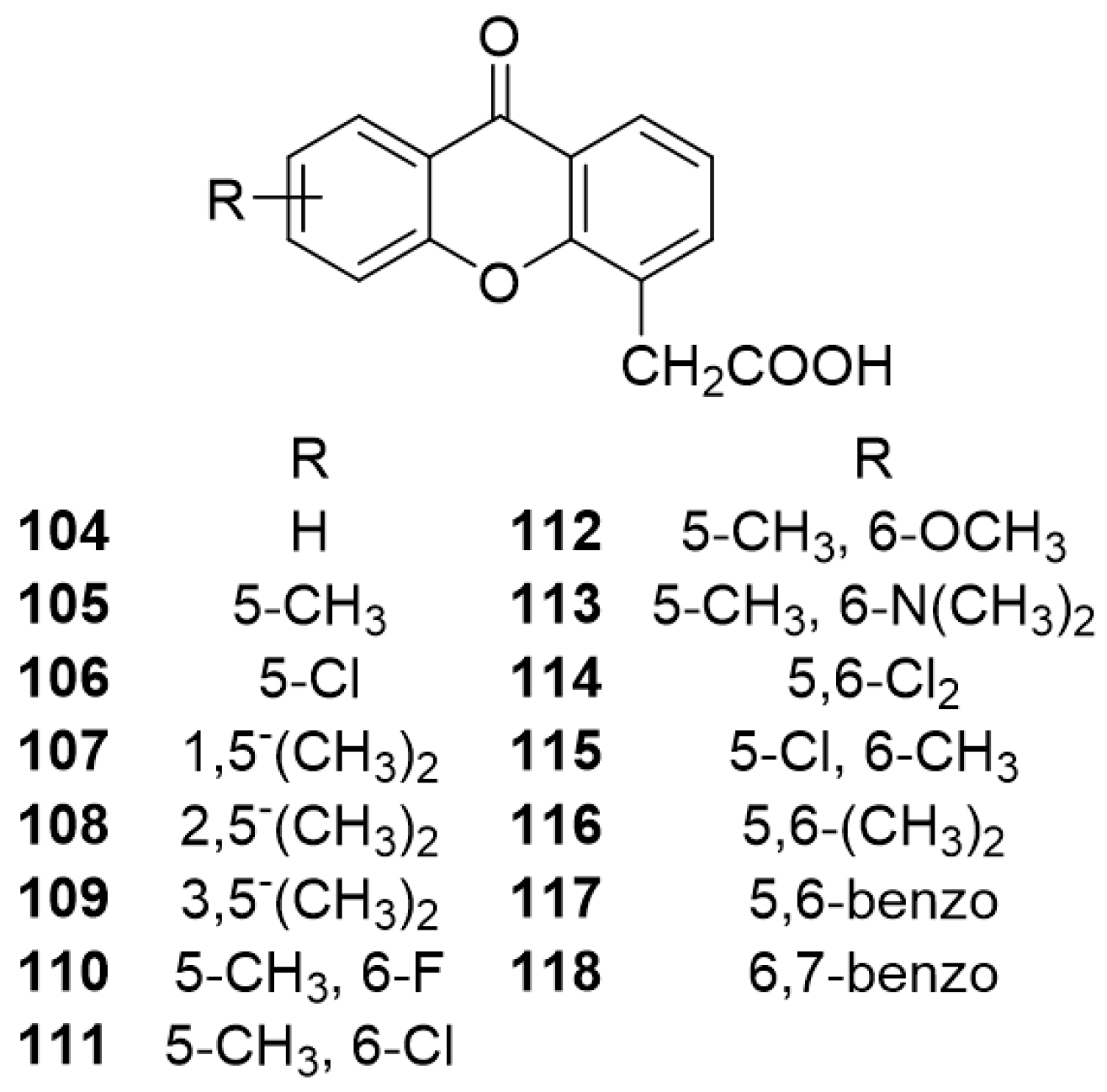
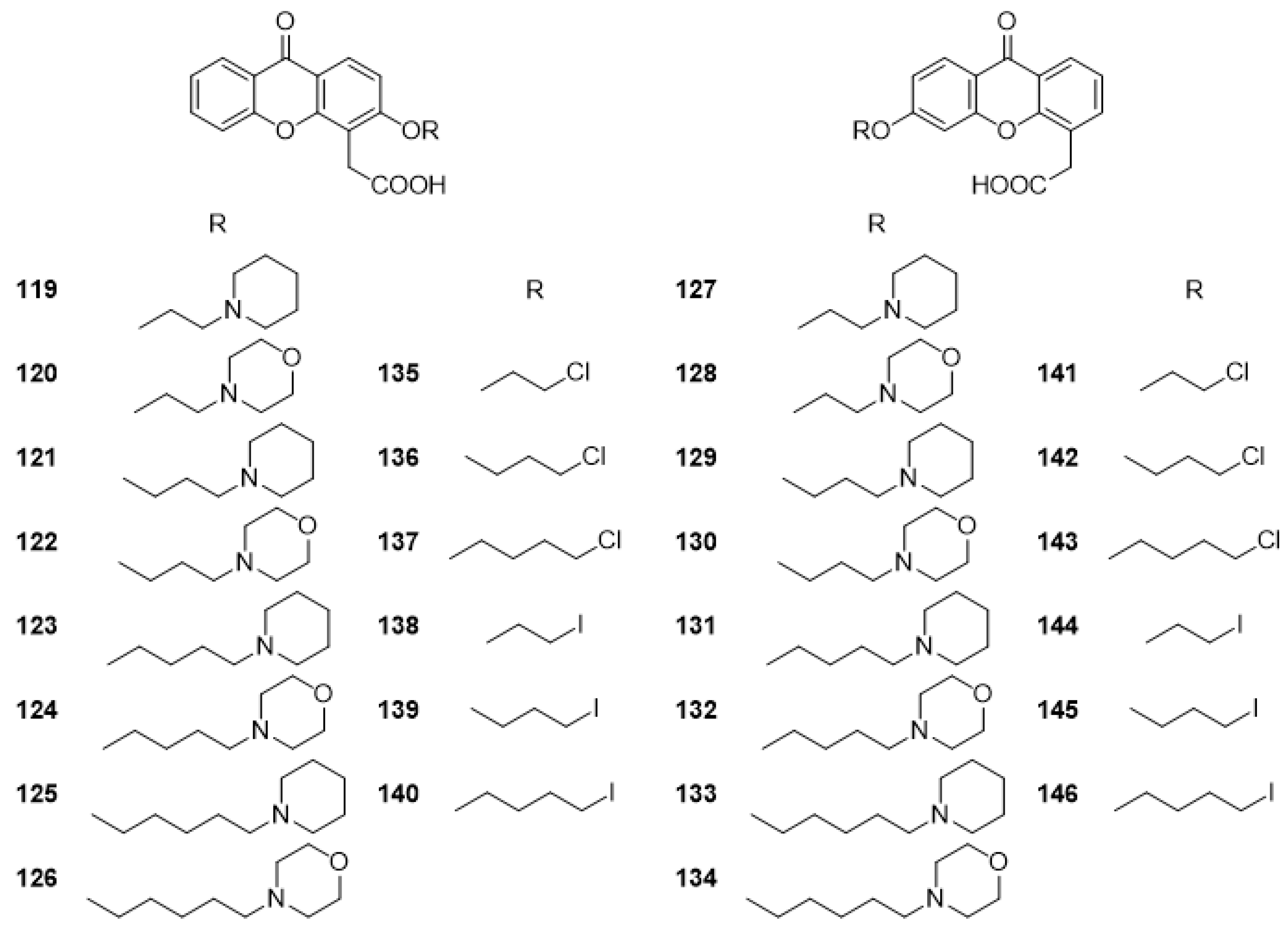
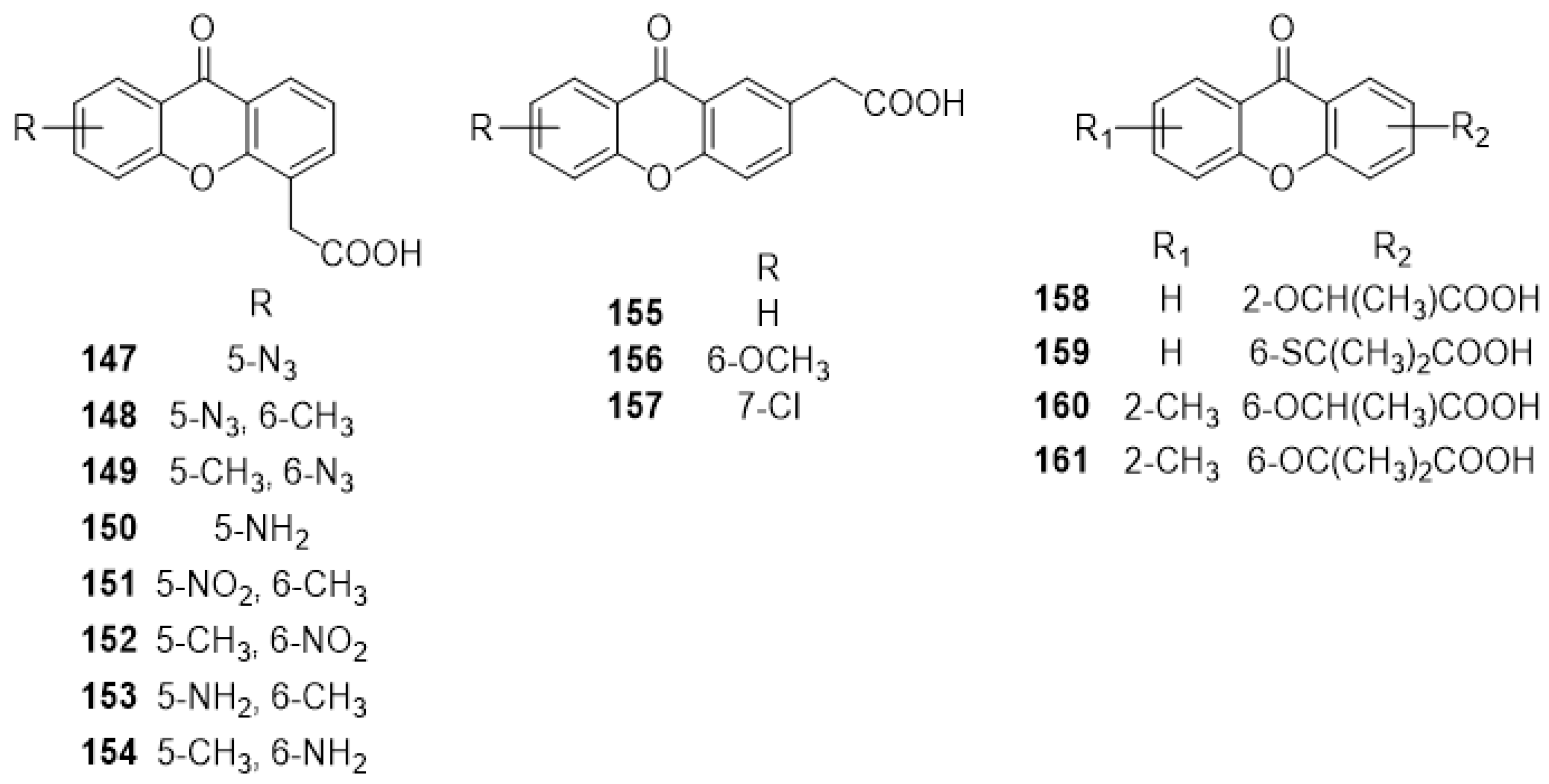
3.1. DMXAA (2), XAA (104) and Analogues (105–161)
3.2. 9-Oxo-9H-Xanthene-2-Carboxylic Acid (162) and Analogues (163–284)
3.2.1. Synthesis
3.2.2. Biological Activities
3.3. Other 9-Oxo-9H-Xanthene Carboxylic Acid Derivatives (285–338)
3.3.1. Synthesis
3.3.2. Biological Activities
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gales, L.; Damas, A.M. Xanthones-a structural perspective. Curr. Med. Chem. 2005, 12, 2499–2515. [Google Scholar] [CrossRef] [PubMed]
- Shagufta; Ahmad, I. Recent insight into the biological activities of synthetic xanthone derivatives. Eur. J. Med. Chem. 2016, 116, 267–280. [Google Scholar] [CrossRef] [PubMed]
- Pinto, M.M.; Sousa, M.E.; Nascimento, M.S. Xanthone derivatives: New insights in biological activities. Curr. Med. Chem. 2005, 12, 2517–2538. [Google Scholar] [CrossRef] [PubMed]
- Wezeman, T.; Brase, S.; Masters, K.S. Xanthone dimers: A compound family which is both common and privileged. Nat. Prod. Rep. 2015, 32, 6–28. [Google Scholar] [CrossRef] [PubMed]
- Na, Y. Recent cancer drug development with xanthone structures. J. Pharm. Pharmacol. 2009, 61, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Muthukrishnan, M.; Basavanag, U.M.V.; Puranik, V.G. The first ionic liquid-promoted Kabbe condensation reaction for an expeditious synthesis of privileged bis-spirochromanone scaffolds. Tetrahedron Lett. 2009, 50, 2643–2648. [Google Scholar] [CrossRef]
- Horton, D.A.; Bourne, G.T.; Smythe, M.L. The combinatorial synthesis of bicyclic privileged structures or privileged substructures. Chem. Rev. 2003, 103, 893–930. [Google Scholar] [CrossRef]
- Masters, K.S.; Brase, S. Xanthones from fungi, lichens, and bacteria: The natural products and their synthesis. Chem. Rev. 2012, 112, 3717–3776. [Google Scholar] [CrossRef]
- Vieira, L.M.; Kijjoa, A. Naturally-occurring xanthones: Recent developments. Curr. Med. Chem. 2005, 12, 2413–2446. [Google Scholar] [CrossRef]
- Pinto, M.M.M.; Castanheiro, R.A.P.; Kijjoa, A. Xanthones from marine-derived microorganisms: Isolation, structure elucidation, and biological activities. In Encyclopedia of Analytical Chemistry; John Wiley & Sons: Hoboken, NJ, USA, 2014; Volume 27, pp. 1–21. [Google Scholar]
- Mayer, A.M.S.; Rodriguez, A.D.; Taglialatela-Scafati, O.; Fusetani, N. Marine pharmacology in 2012–2013: Marine compounds with antibacterial, antidiabetic, antifungal, anti-inflammatory, antiprotozoal, antituberculosis, and antiviral activities; affecting the immune and nervous systems, and other miscellaneous mechanisms of action. Mar. Drugs 2017, 15, 273. [Google Scholar] [CrossRef]
- Gomes, A.S.; Brandao, P.; Fernandes, C.S.G.; da Silva, M.; de Sousa, M.; Pinto, M.M.M. Drug-like Properties and ADME of Xanthone Derivatives: The Antechamber of Clinical Trials. Curr. Med. Chem. 2016, 23, 3654–3686. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.; Soares, J.X.; Cravo, S.; Tiritan, M.E.; Reis, S.; Afonso, C.; Fernandes, C.; Pinto, M.M.M. Lipophilicity assessement in drug discovery: Experimental and theoretical methods applied to xanthone derivatives. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2018, 1072, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Lanzotti, V. Drugs based on natural compounds: Recent achievements and future perspectives. Phytochem. Rev. 2014, 13, 725–726. [Google Scholar] [CrossRef]
- Cragg, G.M.; Newman, D.J. Natural products: A continuing source of novel drug leads. Biochim. Biophys. Acta 2013, 1830, 3670–3695. [Google Scholar] [CrossRef] [PubMed]
- Dias, D.A.; Urban, S.; Roessner, U. A historical overview of natural products in drug discovery. Metabolites 2012, 2, 303–336. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, C.M.G.; Afonso, C.M.M.; Pinto, M.M.M. Routes to Xanthones: An Update on the Synthetic Approaches. Curr. Org. Chem. 2012, 16, 2818–2867. [Google Scholar] [CrossRef]
- Sousa, M.E.; Pinto, M.M. Synthesis of xanthones: An overview. Curr. Med. Chem. 2005, 12, 2447–2479. [Google Scholar] [CrossRef] [PubMed]
- Sathyadevi, P.; Chen, Y.J.; Wu, S.C.; Chen, Y.H.; Wang, Y.M. Reaction-based epoxide fluorescent probe for in vivo visualization of hydrogen sulfide. Biosens. Bioelectron. 2015, 68, 681–687. [Google Scholar] [CrossRef]
- Takashima, I.; Kawagoe, R.; Hamachi, I.; Ojida, A. Development of an AND logic-gate-type fluorescent probe for ratiometric imaging of autolysosome in cell autophagy. Chemistry 2015, 21, 2038–2044. [Google Scholar] [CrossRef]
- Fernandes, C.; Phyo, Y.; Silva, A.S.; Tiritan, M.E.; Kijjoa, A.; Pinto, M.M.M. Chiral stationary phases based on small molecules: An update of the last seventeen years. Sep. Purif. Rev. 2017. [Google Scholar] [CrossRef]
- Fernandes, C.; Tiritan, M.E.; Cravo, S.; Phyo, Y.Z.; Kijjoa, A.; Silva, A.M.S.; Cass, Q.B.; Pinto, M.M.M. New chiral stationary phases based on xanthone derivatives for liquid chromatography. Chirality 2017, 29, 430–442. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, C.; Tiritan, M.E.; Pinto, M.M.M. Chiral derivatives of xanthones: Applications in Medicinal Chemistry and a new approach in Liquid Chromatography. Sci. Chromatogr. 2015, 7, 1–14. [Google Scholar]
- Sousa, E.; Paiva, A.; Nazareth, N.; Gales, L.; Damas, A.M.; Nascimento, M.S.; Pinto, M. Bromoalkoxyxanthones as promising antitumor agents: Synthesis, crystal structure and effect on human tumor cell lines. Eur. J. Med. Chem. 2009, 44, 3830–3835. [Google Scholar] [CrossRef] [PubMed]
- Sousa, E.; Palmeira, A.; Cordeiro, A.S.; Sarmento, B.; Ferreira, D.; Lima, R.T.; Vasconcelos, M.H.; Pinto, M. Bioactive xanthones with effect on P-glycoprotein and prediction of intestinal absorption. Med. Chem. Res. 2013, 22, 2115–2123. [Google Scholar] [CrossRef]
- Cruz, I.; Puthongking, P.; Cravo, S.; Palmeira, A.; Cidade, H.; Pinto, M.; Sousa, E. Xanthone and flavone derivatives as dual agents with acetylcholinesterase inhibition and antioxidant activity as potential anti-alzheimer agents. J. Chem. 2017, 2017, 8587260. [Google Scholar] [CrossRef]
- Neves, M.P.; Cidade, H.; Pinto, M.; Silva, A.M.; Gales, L.; Damas, A.M.; Lima, R.T.; Vasconcelos, M.H.; de Sao Jose Nascimento, M. Prenylated derivatives of baicalein and 3,7-dihydroxyflavone: Synthesis and study of their effects on tumor cell lines growth, cell cycle and apoptosis. Eur. J. Med. Chem. 2011, 46, 2562–2574. [Google Scholar] [CrossRef] [PubMed]
- Paiva, A.M.; Sousa, M.E.; Camoes, A.; Nascimento, M.S.J.; Pinto, M.M.M. Prenylated xanthones: Antiproliferative effects and enhancement of the growth inhibitory action of 4-hydroxytamoxifen in estrogen receptor-positive breast cancer cell line. Med. Chem. Res. 2012, 21, 552–558. [Google Scholar] [CrossRef]
- Azevedo, C.M.; Afonso, C.M.; Soares, J.X.; Reis, S.; Sousa, D.; Lima, R.T.; Vasconcelos, M.H.; Pedro, M.; Barbosa, J.; Gales, L.; et al. Pyranoxanthones: Synthesis, growth inhibitory activity on human tumor cell lines and determination of their lipophilicity in two membrane models. Eur. J. Med. Chem. 2013, 69, 798–816. [Google Scholar] [CrossRef]
- Cidade, H.; Rocha, V.; Palmeira, A.; Marques, C.; Tiritan, M.E.; Ferreira, H.; Lobo, J.S.; Almeida, I.F.; Sousa, M.E.; Pinto, M. In silico and in vitro antioxidant and cytotoxicity evaluation of oxygenated xanthone derivatives. Arab. J. Chem. 2017. [Google Scholar] [CrossRef]
- Sousa, E.P.; Silva, A.M.S.; Pinto, M.M.M.; Pedro, M.M.; Cerqueira, F.A.M.; Nascimento, M.S.J. Isomeric kielcorins and dihydroxyxanthones: Synthesis, structure elucidation, and inhibitory activities of growth of human cancer cell lines and on the proliferation of human lymphocytes in vitro. Hel. Chim. Acta 2002, 85, 2862–2876. [Google Scholar] [CrossRef]
- Correia-Da-Silva, M.; Sousa, E.; Duarte, B.; Marques, F.; Carvalho, F.; Cunha-Ribeiro, L.M.; Pinto, M.M.M. Polysulfated xanthones: Multipathway development of a new generation of dual anticoagulant/antiplatelet agents. J. Med. Chem. 2011, 54, 5373–5384. [Google Scholar] [CrossRef] [PubMed]
- Urbatzka, R.; Freitas, S.; Palmeira, A.; Almeida, T.; Moreira, J.; Azevedo, C.; Afonso, C.; Correia-da-Silva, M.; Sousa, E.; Pinto, M.; et al. Lipid reducing activity and toxicity profiles of a library of polyphenol derivatives. Eur. J. Med. Chem. 2018, 151, 272–284. [Google Scholar] [CrossRef] [PubMed]
- Gales, L.; Sousa, M.E.d.; Pinto, M.M.M.; Kijjoa, A.; Damas, A.M. Naturally occurring 1,2,8-trimethoxyxanthone and biphenyl ether intermediates leading to 1,2-dimethoxyxanthone. Acta Crystallogr. C 2001, 57, 1319–1323. [Google Scholar] [CrossRef] [PubMed]
- Kijjoa, A.; Gonzalez, M.J.; Pinto, M.M.M.; Silva, A.M.S.; Anantachoke, C.; Herz, W. Xanthones from Calophyllum teysmannii var. inophylloide. Phytochemistry 2000, 55, 833–836. [Google Scholar] [CrossRef]
- Rehman, F.; Rustin, G. ASA404: Update on drug development. Expert Opin. Investig. Drugs 2008, 17, 1547–1551. [Google Scholar] [CrossRef] [PubMed]
- Healy, P.C.; Hocking, A.; Tran-Dinh, N.; Pitt, J.I.; Shivas, R.G.; Mitchell, J.K.; Kotiw, M.; Davis, R.A. Xanthones from a microfungus of the genus Xylaria. Phytochemistry 2004, 65, 2373–2378. [Google Scholar] [CrossRef] [PubMed]
- Beattie, K.D.; Ellwood, N.; Kumar, R.; Yang, X.; Healy, P.C.; Choomuenwai, V.; Quinn, R.J.; Elliott, A.G.; Huang, J.X.; Chitty, J.L.; et al. Antibacterial and antifungal screening of natural products sourced from Australian fungi and characterisation of pestalactams D–F. Phytochemistry 2016, 124, 79–85. [Google Scholar] [CrossRef]
- Krick, A.; Kehraus, S.; Gerhäuser, C.; Klimo, K.; Nieger, M.; Maier, A.; Fiebig, H.-H.; Atodiresei, I.; Raabe, G.; Fleischhauer, J.; et al. Potential cancer chemopreventive in vitro activities of monomeric xanthone derivatives from the marine algicolous fungus monodictys putredinis. J. Nat. Prod. 2007, 70, 353–360. [Google Scholar] [CrossRef]
- Shao, C.; Wang, C.; Wei, M.; Gu, Y.; Xia, X.; She, Z.; Lin, Y. Structure elucidation of two new xanthone derivatives from the marine fungus Penicillium sp. (ZZF 32#) from the South China Sea. Magn. Reson. Chem. 2008, 46, 1066–1069. [Google Scholar]
- Sun, R.-R.; Miao, F.-P.; Zhang, J.; Wang, G.; Yin, X.-L.; Ji, N.-Y. Three new xanthone derivatives from an algicolous isolate of Aspergillus wentii. Magn. Reson. Chem. 2013, 51, 65–68. [Google Scholar]
- Sun, Y.L.; Zhang, X.Y.; Zheng, Z.H.; Xu, X.Y.; Qi, S.H. Three new polyketides from marine-derived fungus Penicillium citrinum SCSGAF 0167. Nat. Prod. Res. 2014, 28, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.-T.; Shan, W.-G.; Ying, Y.-M.; Ma, L.-F.; Liu, W.-H.; Zhan, Z.-J. Xanthones with α-glucosidase inhibitory activities fromaspergillus versicolor, a fungal endophyte of huperzia serrata. Helv. Chim. Acta 2015, 98, 148–152. [Google Scholar] [CrossRef]
- Liao, Z.J.; Tian, W.J.; Liu, X.X.; Jiang, X.; Wu, Y.; Lin, T.; Chen, H.F. A New Xanthone from an Endophytic Fungus of Anoectochilus roxburghii. Chem. Nat. Compd. 2018, 54, 267–269. [Google Scholar] [CrossRef]
- Li, J.; Zhang, Y.X.; Chen, L.X.; Dong, Z.H.; Di, X.; Qiu, F. A new xanthone from Penicillium oxalicum. Chem. Nat. Compd. 2010, 46, 216–218. [Google Scholar] [CrossRef]
- Wijeratne, E.M.K.; Turbyville, T.J.; Fritz, A.; Whitesell, L.; Gunatilaka, A.A.L. A new dihydroxanthenone from a plant-associated strain of the fungus Chaetomium globosum demonstrates anticancer activity. Bioorg. Med. Chem. 2006, 14, 7917–7923. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.A.; Pierens, G.K. 1H and 13C NMR assignments for two new xanthones from the endophytic fungus Xylaria sp. FRR 5657. Magn. Reson. Chem. 2006, 44, 966–968. [Google Scholar] [CrossRef] [PubMed]
- Munekata, H. Studies on some new metabolic products of Penicillium. II. J. Biochem. 1953, 40, 451–460. [Google Scholar] [CrossRef]
- Abdissa, N.; Heydenreich, M.; Midiwo, J.O.; Ndakala, A.; Majer, Z.; Neumann, B.; Stammler, H.-G.; Sewald, N.; Yenesew, A. A xanthone and a phenylanthraquinone from the roots of Bulbine frutescens, and the revision of six seco-anthraquinones into xanthones. Phytochem. Lett. 2014, 9, 67–73. [Google Scholar] [CrossRef]
- Singh, O.; Ali, M.; Akhtar, N. New antifungal xanthones from the seeds of Rhus coriaria L. Zeitschrift fur Naturforschung. C J. Biosci. 2011, 66, 17–23. [Google Scholar] [CrossRef]
- Jackson, B.; Locksley, H.D.; Scheinmann, F. Extractives from Guttiferae. Part, V. Scriblitifolic acid, a new xanthone from Calophyllum scriblitifolium Henderson and Wyatt-Smith. J. Chem. Soc. C Org. Chem. 1967, 785–796. [Google Scholar] [CrossRef]
- Kijjoa, A.; Gonzalez, M.J.; Afonso, C.M.; Pinto, M.M.M.; Anantachoke, C.; Herz, W. Xanthones from Calophyllum teysmannii var. inophylloide. Phytochemistry 2000, 53, 1021–1024. [Google Scholar] [CrossRef]
- Cottiglia, F.; Casu, L.; Bonsignore, L.; Casu, M.; Floris, C.; Sosa, S.; Altinier, G.; Della Loggia, R. Topical anti-inflammatory activity of flavonoids and a new xanthone from Santolina insularis. Zeitschrift fur Naturforschung. C J. Biosci. 2005, 60, 63–66. [Google Scholar] [CrossRef]
- Li, C.-J.; Yang, J.-Z.; Yu, S.-S.; Zhao, C.-Y.; Peng, Y.; Wang, X.-L.; Zhang, D.-M. Glomexanthones A–C, three xanthonolignoid C-glycosides from Polygala glomerata Lour. Fitoterapia 2014, 93, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Gopalakrishnan, G.; Balaganesan, B. Two novel xanthones from Garcinia mangostana. Fitoterapia 2000, 71, 607–609. [Google Scholar] [CrossRef]
- Tang, Y.-X.; Fu, W.-W.; Wu, R.; Tan, H.-S.; Shen, Z.-W.; Xu, H.-X. Bioassay-Guided Isolation of Prenylated Xanthone Derivatives from the Leaves of Garcinia oligantha. J. Nat. Prod. 2016, 79, 1752–1761. [Google Scholar] [CrossRef] [PubMed]
- Lu, G.B.; Yang, X.X.; Huang, Q.S. Isolation and structure of neo-gambogic acid from Gamboge (Garcinia hanburryi). Yao Xue Xue Bao 1984, 19, 636–639. [Google Scholar] [PubMed]
- Lin, L.-J.; Lin, L.-Z.; Pezzuto, J.M.; Cordell, G.A.; Ruangrungsi, N. Isogambogic acid and isomorellinol from Garcinia hanburyi. Magn. Reson. Chem. 1993, 31, 340–347. [Google Scholar] [CrossRef]
- Asano, J.; Chiba, K.; Tada, M.; Yoshii, T. Cytotoxic xanthones from Garcinia hanburyi. Phytochemistry 1996, 41, 815–820. [Google Scholar] [CrossRef]
- Wu, J.; Xu, Y.-J.; Cheng, X.-F.; Harrison, L.J.; Sim, K.-Y.; Goh, S.H. A highly rearranged tetraprenylxanthonoid from Garcinia gaudichaudii (Guttiferae). Tetrahedron Lett. 2001, 42, 727–729. [Google Scholar] [CrossRef]
- Sukpondma, Y.; Rukachaisirikul, V.; Phongpaichit, S. Antibacterial caged-tetraprenylated xanthones from the fruits of Garcinia hanburyi. Chem. Pharm. Bull. 2005, 53, 850–852. [Google Scholar] [CrossRef]
- Reutrakul, V.; Anantachoke, N.; Pohmakotr, M.; Jaipetch, T.; Sophasan, S.; Yoosook, C.; Kasisit, J.; Napaswat, C.; Santisuk, T.; Tuchinda, P. Cytotoxic and Anti-HIV-1 Caged Xanthones from the Resin and Fruits of Garcinia hanburyi. Plant. Med. 2007, 73, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.-B.; Wang, Y.-L.; Yang, L.; Tso, T.-F.; Qiao, C.-F.; Song, J.-Z.; Xu, L.-J.; Chen, S.-L.; Yang, D.-J.; Xu, H.-X. Cytotoxic Polyprenylated Xanthones from the Resin of Garcinia hanburyi. Chem. Pharm. Bull. 2006, 54, 265–267. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.; Yang, L.; Liu, Y.; Wang, Y.; Qiao, C.; Song, J.; Xu, L.; Yang, D.; Chen, S.; Xu, H. Gambogic Acid and Epigambogic Acid, C-2 Epimers with Novel Anticancer Effects from Garcinia hanburyi. Plant. Med. 2006, 72, 281–284. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.-B.; Yang, L.; Wang, Y.-L.; Qiao, C.-F.; Song, J.-Z.; Sun, H.-D.; Xu, H.-X. A Pair of Novel Cytotoxic Polyprenylated Xanthone Epimers from Gamboges. Chem. Biodivers. 2006, 3, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Song, J.-Z.; Yip, Y.-K.; Han, Q.-B.; Qiao, C.-F.; Xu, H.-X. Rapid determination of polyprenylated xanthones in gamboge resin of Garcinia hanburyi by HPLC. J. Sep. Sci. 2007, 30, 304–309. [Google Scholar] [CrossRef] [PubMed]
- Feng, F.; Liu, W.-Y.; Chen, Y.-S.; Guo, Q.-L.; You, Q.-D. Five novel prenylated xanthones from Resina Garciniae. J. Asian Nat. Prod. Res. 2007, 9, 735–741. [Google Scholar] [CrossRef] [PubMed]
- Tao, S.-J.; Guan, S.-H.; Wang, W.; Lu, Z.-Q.; Chen, G.-T.; Sha, N.; Yue, Q.-X.; Liu, X.; Guo, D.-A. Cytotoxic Polyprenylated Xanthones from the Resin of Garcinia hanburyi. J. Nat. Prod. 2009, 72, 117–124. [Google Scholar] [CrossRef]
- Deng, Y.-X.; Pan, S.-L.; Zhao, S.-Y.; Wu, M.-Q.; Sun, Z.-Q.; Chen, X.-H.; Shao, Z.-Y. Cytotoxic alkoxylated xanthones from the resin of Garcinia hanburyi. Fitoterapia 2012, 83, 1548–1552. [Google Scholar] [CrossRef]
- Deng, Y.-X.; Guo, T.; Shao, Z.-Y.; Xie, H.; Pan, S.-L. Three New Xanthones from the Resin of Garcinia hanburyi. Plant. Med. 2013, 79, 792–796. [Google Scholar] [CrossRef]
- Dong, B.; Zheng, Y.-F.; Wen, H.-M.; Wang, X.-Z.; Xiong, H.-W.; Wu, H.; Li, W. Two new xanthone epimers from the processed gamboge. Nat. Prod. Res. 2017, 31, 817–821. [Google Scholar] [CrossRef]
- Chen, Y.; He, S.; Tang, C.; Li, J.; Yang, G. Caged polyprenylated xanthones from the resin of Garcinia hanburyi. Fitoterapia 2016, 109, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Leão, M.; Gomes, S.; Pedraza-Chaverri, J.; Machado, N.; Sousa, E.; Pinto, M.; Inga, A.; Pereira, C.; Saraiva, L. α-Mangostin and gambogic acid as potential inhibitors of the p53–MDM2 interaction revealed by a yeast approach. J. Nat. Prod. 2013, 76, 774–778. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.-B.; Cheung, S.; Tai, J.; Qiao, C.-F.; Song, J.-Z.; Xu, H.-X. Stability and Cytotoxicity of Gambogic Acid and Its Derivative, Gambogoic Acid. Biol. Pharm. Bull. 2005, 28, 2335–2337. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, W.; Ye, C.; Lin, Y.; Cheang, T.Y.; Wang, M.; Zhang, H.; Wang, S.; Zhang, L.; Wang, S. Gambogic acid induces G0/G1 cell cycle arrest and cell migration inhibition via suppressing PDGF receptor beta tyrosine phosphorylation and Rac1 activity in rat aortic smooth muscle cells. J. Atheroscler. Thromb. 2010, 17, 901–913. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.B.; Xu, H.X. Caged Garcinia xanthones: Development since 1937. Curr. Med. Chem. 2009, 16, 3775–3796. [Google Scholar] [CrossRef] [PubMed]
- Chantarasriwong, O.; Batova, A.; Chavasiri, W.; Theodorakis, E.A. Chemistry and Biology of the Caged Garcinia Xanthones. Chem. A Eur. J. 2010, 16, 9944–9962. [Google Scholar] [CrossRef] [PubMed]
- El-Seedi, H.R.; El-Barbary, M.A.; El-Ghorab, D.M.; Bohlin, L.; Borg-Karlson, A.K.; Goransson, U.; Verpoorte, R. Recent insights into the biosynthesis and biological activities of natural xanthones. Curr. Med. Chem. 2010, 17, 854–901. [Google Scholar] [CrossRef]
- Jia, B.; Li, S.; Hu, X.; Zhu, G.; Chen, W. Recent Research on Bioactive Xanthones from Natural Medicine: Garcinia hanburyi. AAPS PharmSciTech 2015, 16, 742–758. [Google Scholar] [CrossRef]
- Chantarasriwong, O.; Althufairi, B.D.; Checchia, N.J.; Theodorakis, E.A. Chapter 4-Caged Garcinia Xanthones: Synthetic Studies and Pharmacophore Evaluation. In Studies in Natural Products Chemistry; Attaur, R., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; Volume 58, pp. 93–131. [Google Scholar]
- Cao, S.G.; Sng, V.H.L.; Wu, X.H.; Sim, K.Y.; Tan, B.H.K.; Pereira, J.T.; Goh, S.H. Novel cytotoxic polyprenylated xanthonoids from Garcinia gaudichaudii (Guttiferae). Tetrahedron 1998, 54, 10915–10924. [Google Scholar] [CrossRef]
- Xu, Y.J.; Yip, S.C.; Kosela, S.; Fitri, E.; Hana, M.; Goh, S.H.; Sim, K.Y. Novel Cytotoxic, Polyprenylated Heptacyclic Xanthonoids from Indonesian Garcinia gaudichaudii (Guttiferae). Org. Lett. 2000, 2, 3945–3948. [Google Scholar] [CrossRef]
- Rukachaisirikul, V.; Kaewnok, W.; Koysomboon, S.; Phongpaichit, S.; Taylor, W.C. Caged-tetraprenylated xanthones from Garcinia scortechinii. Tetrahedron 2000, 56, 8539–8543. [Google Scholar] [CrossRef]
- Rukachaisirikul, V.; Painuphong, P.; Sukpondma, Y.; Koysomboon, S.; Sawangchote, P.; Taylor, W.C. Caged-Triprenylated and -Tetraprenylated Xanthones from the Latex of Garcinia scortechinii. J. Nat. Prod. 2003, 66, 933–938. [Google Scholar] [CrossRef] [PubMed]
- Rukachaisirikul, V.; Phainuphong, P.; Sukpondma, Y.; Phongpaichit, S.; Taylor, W.C. Antibacterial caged-tetraprenylated xanthones from the stem bark of Garcinia scortechinii. Plant. Med. 2005, 71, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Sukpondma, Y.; Rukachaisirikul, V.; Phongpaichit, S. Xanthone and sesquiterpene derivatives from the fruits of Garcinia scortechinii. J. Nat. Prod. 2005, 68, 1010–1017. [Google Scholar] [CrossRef] [PubMed]
- Aoki, M.; Itezono, Y.; Shirai, H.; Nakayama, N.; Sakai, A.; Tanaka, Y.; Yamaguchi, A.; Shimma, N.; Yokose, K.; Seto, H. Structure of a novel phospholipase C inhibitor, vinaxanthone (Ro 09-1450), produced by penicillium vinaceum. Tetrahedron Lett. 1991, 32, 4737–4740. [Google Scholar] [CrossRef]
- Gammon, G.; Chandler, G.; Depledge, P.; Elcock, C.; Wrigley, S.; Moore, J.; Cammarota, G.; Sinigaglia, F.; Moore, M. A fungal metabolite which inhibits the interaction of CD4 with major histocompatibility complex-encoded class II molecules. Eur. J. Immunol. 1994, 24, 991–998. [Google Scholar] [CrossRef] [PubMed]
- Wrigley, S.K.; Latif, M.A.; Gibson, T.M.; Chicarelli-Robinson, M.I.; Williams, D.H. Structure elucidation of xanthone derivatives with CD4-binding activity from Penicillium glabrum (Wehmer) Westling. Pure Appl. Chem. 1994, 66, 2383. [Google Scholar] [CrossRef]
- Řezanka, T.; Řezanka, P.; Sigler, K. A Biaryl Xanthone Derivative Having Axial Chirality from Penicillium vinaceum. J. Nat. Prod. 2008, 71, 820–823. [Google Scholar] [CrossRef]
- Zheng, C.J.; Sohn, M.J.; Kim, W.G. Vinaxanthone, a new FabI inhibitor from Penicillium sp. J. Antimicrob. Chemother. 2009, 63, 949–953. [Google Scholar] [CrossRef]
- Roche, H.-L. Xanthofulvin as an inhibitor of chitin synthase and its potential as an antifungal. Expert Opin. Ther. Pat. 1993, 3, 1801–1802. [Google Scholar]
- Kumagai, K.; Hosotani, N.; Kikuchi, K.; Kimura, T.; Saji, I. Xanthofulvin, a novel semaphorin inhibitor produced by a strain of Penicillium. J. Antibiot. 2003, 56, 610–616. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, K.; Kishino, A.; Konishi, O.; Kumagai, K.; Hosotani, N.; Saji, I.; Nakayama, C.; Kimura, T. In Vitro and in Vivo Characterization of a Novel Semaphorin 3A Inhibitor, SM-216289 or Xanthofulvin. J. Biol. Chem. 2003, 278, 42985–42991. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, S.; Iwanami, A.; Nakamura, M.; Kishino, A.; Kikuchi, K.; Shibata, S.; Okano, H.J.; Ikegami, T.; Moriya, A.; Konishi, O.; et al. A selective Sema3A inhibitor enhances regenerative responses and functional recovery of the injured spinal cord. Nat. Med. 2006, 12, 1380–1389. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.; Jeelani, G.; Masuda, Y.; Sakai, K.; Tsukui, K.; Waluyo, D.; Tarwadi; Watanabe, Y.; Nonaka, K.; Matsumoto, A.; Omura, S.; et al. Identification of natural inhibitors of Entamoeba histolytica cysteine synthase from microbial secondary metabolites. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Chin, M.R.; Zlotkowski, K.; Han, M.; Patel, S.; Eliasen, A.M.; Axelrod, A.; Siegel, D. Expedited Access to Vinaxanthone and Chemically Edited Derivatives Possessing Neuronal Regenerative Effects through Ynone Coupling Reactions. ACS Chem. Neurosci. 2015, 6, 542–550. [Google Scholar] [CrossRef] [PubMed]
- Omolo, J.J.; Maharaj, V.; Naidoo, D.; Klimkait, T.; Malebo, H.M.; Mtullu, S.; Lyaruu, H.V.; de Koning, C.B. Bioassay-guided investigation of the Tanzanian plant Pyrenacantha kaurabassana for potential anti-HIV-active compounds. J. Nat. Prod. 2012, 75, 1712–1716. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.-L.; Xu, Y.; Han, Z.; Li, Y.-X.; Lu, L.; Lai, P.-Y.; Zhong, J.-L.; Guo, X.-R.; Zhang, X.-X.; Qian, P.-Y. Four New Antibacterial Xanthones from the Marine-Derived Actinomycetes Streptomyces caelestis. Mar. Drugs 2012, 10, 2571. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, L.; Li, Z.; Wang, Y.; Tian, L.; Pei, Y.; Hua, H. A new sulfo-xanthone from the marine-derived fungus Penicillium sacculum. Chem. Nat. Compd. 2012, 48, 771–773. [Google Scholar] [CrossRef]
- Michael, A. On the action of aromatic oxy-acids on phenols. Amer. Chem. J. 1883, 5, 81–97. [Google Scholar]
- v. Kostanecki, S. Über das Gentisin. Monatshefte für chemie und verwandte teile anderer wissenschaften 1891, 12, 205–210. [Google Scholar] [CrossRef]
- Barbero, N.; SanMartin, R.; Dominguez, E. An efficient copper-catalytic system for performing intramolecular O-arylation reactions in aqueous media. New synthesis of xanthones. Green Chem. 2009, 11, 830–836. [Google Scholar] [CrossRef]
- Genovese, S.; Fiorito, S.; Specchiulli, M.C.; Taddeo, V.A.; Epifano, F. Microwave-assisted synthesis of xanthones promoted by ytterbium triflate. Tetrahedron Lett. 2015, 56, 847–850. [Google Scholar] [CrossRef]
- Li, J.; Jin, C.; Su, W.K. Microwave-assisted, yb(otf)(3)/tfoh cocatalyzed synthesis of xanthones and thioxanthones by intramolecular friedel-crafts reaction under solvent-free conditions. Heterocycles 2011, 83, 855–866. [Google Scholar] [CrossRef]
- Menendez, C.A.; Nador, F.; Radivoy, G.; Gerbino, D.C. One-step synthesis of xanthones catalyzed by a highly efficient copper-based magnetically recoverable nanocatalyst. Org. Lett. 2014, 16, 2846–2849. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Shi, R.; Gan, P.; Liu, C.; Ding, A.; Wang, Q.; Lei, A. Palladium-catalyzed oxidative double C-H functionalization/carbonylation for the synthesis of xanthones. Angew. Chem. Int. Ed. Engl. 2012, 51, 5204–5207. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.J.; Yang, L.; Wu, Y.; Du, J.Y.; Mao, Y.L.; Wang, X.; Luan, S.J.; Lei, Y.H.; Li, X.; Sun, H.P.; et al. Microwave-assisted transition-metal-free intramolecular Ullmann-type O-arylation in water for the synthesis of xanthones and azaxanthones. Tetrahedron Lett. 2014, 55, 4883–4887. [Google Scholar] [CrossRef]
- Zhang, Z.H.; Wang, H.J.; Ren, X.Q.; Zhang, Y.Y. A facile and efficient method for synthesis of xanthone derivatives catalyzed by HBF4/SiO2 under solvent-free conditions. Monatsh. Chem. 2009, 140, 1481–1483. [Google Scholar] [CrossRef]
- Castanheiro, R.A.P.; Pinto, M.M.M.; Cravo, S.M.M.; Pinto, D.C.G.A.; Silva, A.M.S.; Kijjoa, A. Improved methodologies for synthesis of prenylated xanthones by microwave irradiation and combination of heterogeneous catalysis (K10 clay) with microwave irradiation. Tetrahedron 2009, 65, 3848–3857. [Google Scholar] [CrossRef]
- Ghosh, C.K. Synthesis of xanthones from chromones. J. Indian Chem. Soc. 2013, 90, 1721–1736. [Google Scholar]
- Baguley, B.C.; Siemann, D.W. Temporal aspects of the action of ASA404 (vadimezan; DMXAA). Expert Opin. Invest. Drug. 2010, 19, 1413–1425. [Google Scholar] [CrossRef]
- Head, M.; Jameson, M.B. The development of the tumor vascular-disrupting agent ASA404 (vadimezan, DMXAA): Current status and future opportunities. Expert Opin. Invest. Drug. 2010, 19, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Daei Farshchi Adli, A.; Jahanban-Esfahlan, R.; Seidi, K.; Samandari-Rad, S.; Zarghami, N. An overview on Vadimezan (DMXAA): The vascular disrupting agent. Chem. Biol. Drug Des. 2018, 91, 996–1006. [Google Scholar] [CrossRef] [PubMed]
- Ching, L.M. ASA404. Vascular-disrupting agent, oncolytic. Drugs Future 2008, 33, 561–569. [Google Scholar] [CrossRef]
- McKeage, M.J.; Kelland, L.R. 5,6-Dimethylxanthenone-4-acetic acid (DMXAA): Clinical potential in combination with taxane-based chemotherapy. Am. J. Cancer 2006, 5, 155–162. [Google Scholar] [CrossRef]
- Baguley, B.C.; Wilson, W.R. Potential of DMXAA combination therapy for solid tumors. Expert Rev. Antican. 2002, 2, 593–603. [Google Scholar] [CrossRef] [PubMed]
- Baguley, B.C.; McKeage, M.J. ASA404: A tumor vascular-disrupting agent with broad potential for cancer therapy. Future Oncol. 2010, 6, 1537–1543. [Google Scholar] [CrossRef] [PubMed]
- Baguley, B.C. Antivascular therapy of cancer: DMXAA. Lancet Oncol. 2003, 4, 141–148. [Google Scholar] [CrossRef]
- McKeage, M. Clinical trials of vascular disrupting agents in advanced non-small-cell lung cancer. Clin. Lung Cancer 2011, 12, 143–147. [Google Scholar] [CrossRef]
- Zhou, S.; Kestell, P.; Baguley, B.C.; Paxton, J.W. 5,6-Dimethylxanthenone-4-acetic acid (DMXAA): A new biological response modifier for cancer therapy. Investig. New Drug. 2002, 20, 281–295. [Google Scholar] [CrossRef]
- Ching, L.M.; Zwain, S.; Baguley, B.C. Relationship between tumour endothelial cell apoptosis and tumour blood flow shutdown following treatment with the antivascular agent DMXAA in mice. Br. J. Cancer 2004, 90, 906–910. [Google Scholar] [CrossRef]
- Woon, S.T.; Hung, S.S.C.; Wu, D.C.F.; Schooltink, M.A.; Sutherland, R.; Baguley, B.C.; Chen, Q.; Chamley, L.W.; Ching, L.M. NF-κB-independent induction of endothelial cell apoptosis by the vascular disrupting agent DMXAA. Anticancer Res. 2007, 27, 327–334. [Google Scholar] [PubMed]
- Ching, L.M.; Cao, Z.; Kieda, C.; Zwain, S.; Jameson, M.B.; Baguley, B.C. Induction of endothelial cell apoptosis by the antivascular agent 5,6-dimethylxanthenone-4-acetic acid. Br. J. Cancer 2002, 86, 1937–1942. [Google Scholar] [CrossRef] [PubMed]
- Bellnier, D.A.; Gollnick, S.O.; Camacho, S.H.; Greco, W.R.; Cheney, R.T. Treatment with the Tumor Necrosis Factor-α-Inducing Drug 5,6-Dimethylxanthenone-4-Acetic Acid Enhances the Antitumor Activity of the Photodynamic Therapy of RIF-1 Mouse Tumors. Cancer Res. 2003, 63, 7584–7590. [Google Scholar] [PubMed]
- Ching, L.M.; Goldsmith, D.; Joseph, W.R.; Körner, H.; Sedgwick, J.D.; Baguley, B.C. Induction of intratumoral tumor necrosis factor (TNF) synthesis and hemorrhagic necrosis by 5,6-dimethylxanthenone-4-acetic acid (DMXAA) in TNF knockout mice. Cancer Res. 1999, 59, 3304–3307. [Google Scholar] [PubMed]
- Philpott, M.; Baguley, B.C.; Ching, L.M. Induction of tumour necrosis factor-α by single and repeated doses of the antitumour agent 5,6-dimethylxanthenone-4-acetic acid. Cancer Chemoth. Pharmacol. 1995, 36, 143–148. [Google Scholar] [CrossRef]
- Cao, Z.; Baguley, B.C.; Ching, L.M. Interferon-inducible protein 10 induction and inhibition of angiogenesis in vivo by the antitumor agent 5,6-dimethylxanthenone-4-acetic acid (DMXAA). Cancer Research 2001, 61, 1517–1521. [Google Scholar] [PubMed]
- Baguley, B.C.; Ching, L.M. DMXAA: An antivascular agent with multiple host responses. Int. J. Radiat. Oncol. Biol. Phys. 2002, 54, 1503–1511. [Google Scholar] [CrossRef]
- Thomsen, L.L.; Baguley, B.C.; Wilson, W.R. Nitric oxide: Its production in host-cell-infiltrated EMT6 spheroids and its role in tumour cell killing by flavone-8-acetic acid and 5,6-dimethylxanthenone-4-acetic acid. Cancer Chemoth. Pharmacol. 1992, 31, 151–155. [Google Scholar] [CrossRef]
- Thomsen, L.L.; Ching, L.M.; Joseph, W.R.; Baguley, B.C.; Gavin, J.B. Nitric oxide production in endotoxin-resistant C3H/HeJ mice stimulated with flavone-8-acetic acid and xanthenone-4-acetic acid analogues. Biochem. Pharm. 1992, 43, 2401–2406. [Google Scholar] [CrossRef]
- Baguley, B.C.; Zhuang, L.; Kestell, P. Increased plasma serotonin following treatment with flavone-8-acetic acid, 5,6-dimethylxanthenone-4-acetic acid, vinblastine, and colchicine: Relation to vascular effects. Oncol. Res. 1997, 9, 55–60. [Google Scholar]
- Baguley, B.C.; Cole, G.; Thomsen, L.L.; Zhuang, L. Serotonin involvement in the antitumour and host effects of flavone-8-acetic acid and 5,6-dimethylxanthenone-4-acetic acid. Cancer Chemoth. Pharm. 1993, 33, 77–81. [Google Scholar] [CrossRef]
- Philpott, M.; Ching, L.M.; Baguley, B.C. The antitumour agent 5,6-dimethylxanthenone-4-acetic acid acts in vitro on human mononuclear cells as a co-stimulator with other inducers of tumour necrosis factor. Eur. J. Cancer 2001, 37, 1930–1937. [Google Scholar] [CrossRef]
- Woon, S.T.; Zwain, S.; Schooltink, M.A.; Newth, A.L.; Baguley, B.C.; Ching, L.M. NF-kappa B activation in vivo in both host and tumour cells by the antivascular agent 5,6-dimethylxanthenone-4-acetic acid (DMXAA). Eur. J. Cancer 2003, 39, 1176–1183. [Google Scholar] [CrossRef]
- Shirey, K.A.; Nhu, Q.M.; Yim, K.C.; Roberts, Z.J.; Teijaro, J.R.; Farber, D.L.; Blanco, J.C.; Vogel, S.N. The anti-tumor agent, 5,6-dimethylxanthenone-4-acetic acid (DMXAA), induces IFN-β-mediated antiviral activity in vitro and in vivo. J. Leukocyte Biol. 2011, 89, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.H.; Zhang, Y.; Shen, J.; Zhang, S.; Chen, L.; Gu, J.; Mruk, J.S.; Cheng, G.; Zhu, L.; Kunapuli, S.P.; et al. Tumor vascular disrupting agent 5,6-dimethylxanthenone-4-acetic acid inhibits platelet activation and thrombosis via inhibition of thromboxane A2 signaling and phosphodiesterase. J. Thromb. Haemost. 2013, 11, 1855–1866. [Google Scholar] [PubMed]
- Hida, T.; Tamiya, M.; Nishio, M.; Yamamoto, N.; Hirashima, T.; Horai, T.; Tanii, H.; Shi, M.M.; Kobayashi, K.; Horio, Y. Phase I study of intravenous ASA404 (vadimezan) administered in combination with paclitaxel and carboplatin in Japanese patients with non-small cell lung cancer. Cancer Sci. 2011, 102, 845–851. [Google Scholar] [CrossRef] [PubMed]
- McKeage, M.J.; Reck, M.; Jameson, M.B.; Rosenthal, M.A.; Gibbs, D.; Mainwaring, P.N.; Freitag, L.; Sullivan, R.; Von Pawel, J. Phase II study of ASA404 (vadimezan, 5,6-dimethylxanthenone-4-acetic acid/DMXAA) 1800 mg/m2 combined with carboplatin and paclitaxel in previously untreated advanced non-small cell lung cancer. Lung Cancer 2009, 65, 192–197. [Google Scholar] [CrossRef] [PubMed]
- McKeage, M.J.; Von Pawel, J.; Reck, M.; Jameson, M.B.; Rosenthal, M.A.; Sullivan, R.; Gibbs, D.; Mainwaring, P.N.; Serke, M.; Lafitte, J.J.; et al. Randomised phase II study of ASA404 combined with carboplatin and paclitaxel in previously untreated advanced non-small cell lung cancer. Br. J. Cancer 2008, 99, 2006–2012. [Google Scholar] [CrossRef]
- Pili, R.; Rosenthal, M.A.; Mainwaring, P.N.; Van Hazel, G.; Srinivas, S.; Dreicer, R.; Goel, S.; Leach, J.; Wong, S.; Clingan, P. Phase II study on the addition of ASA404 (vadimezan; 5,6- dimethylxanthenone-4-acetic acid) to docetaxel in CRMPC. Clin. Cancer Res. 2010, 16, 2906–2914. [Google Scholar] [CrossRef]
- Früh, M.; Cathomas, R.; Siano, M.; Tscherry, G.; Zippelius, A.; Mamot, C.; Erdmann, A.; Krasniqi, F.; Rauch, D.; Simcock, M.; et al. Carboplatin and paclitaxel plus ASA404 as first-line chemotherapy for extensive-stage small-cell lung cancer: A multicenter single arm phase II trial (SAKK 15/08). Clin. Lung Cancer 2013, 14, 34–39. [Google Scholar] [CrossRef]
- Lara, P.N., Jr.; Douillard, J.Y.; Nakagawa, K.; Von Pawel, J.; McKeage, M.J.; Albert, I.; Losonczy, G.; Reck, M.; Heo, D.S.; Fan, X.; et al. Randomized phase III placebo-controlled trial of carboplatin and paclitaxel with or without the vascular disrupting agent vadimezan (ASA404) in advanced non-small-cell lung cancer. J. Clin. Oncol. 2011, 29, 2965–2971. [Google Scholar] [CrossRef] [PubMed]
- Rewcastle, G.W.; Atwell, G.J.; Zhuang, L.; Baguley, B.C.; Denny, W.A. Potential antitumor agents. 61. Structure-activity relationships for in vivo colon 38 activity among disubstituted 9-oxo-9H-xanthene-4-acetic acids. J. Med. Chem. 1991, 34, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Atwell, G.J.; Yang, S.; Denny, W.A. An improved synthesis of 5,6-dimethylxanthenone-4-acetic acid (DMXAA). Eur. J. Med. Chem. 2002, 37, 825–828. [Google Scholar] [CrossRef]
- Yang, S.; Denny, W.A. A new short synthesis of 5,6-dimethylxanthenone-4-acetic acid (ASA404, DMXAA). Tetrahedron Lett. 2009, 50, 3945–3947. [Google Scholar] [CrossRef]
- Gobbi, S.; Belluti, F.; Bisi, A.; Piazzi, L.; Rampa, A.; Zampiron, A.; Barbera, M.; Caputo, A.; Carrara, M. New derivatives of xanthenone-4-acetic acid: Synthesis, pharmacological profile and effect on TNF-alpha and NO production by human immune cells. Bioorg. Med. Chem. 2006, 14, 4101–4109. [Google Scholar] [CrossRef] [PubMed]
- Palmer, B.D.; Henare, K.; Woon, S.T.; Sutherland, R.; Reddy, C.; Wang, L.C.; Kieda, C.; Ching, L.M. Synthesis and biological activity of azido analogues of 5,6-dimethylxanthenone-4-acetic acid for use in photoaffinity labeling. J. Med. Chem. 2007, 50, 3757–3764. [Google Scholar] [CrossRef] [PubMed]
- Marona, H.; Pȩkala, E.; Gunia, A.; Czuba, Z.; Szneler, E.; Sadowski, T.; Król, W. The influence of some xanthone derivatives on the activity of J-774A.1 cells. Sci. Pharm. 2009, 77, 743–754. [Google Scholar] [CrossRef]
- Liu, J.; Zhou, F.; Zhang, L.; Wang, H.; Zhang, J.; Zhang, C.; Jiang, Z.; Li, Y.; Liu, Z.; Chen, H. DMXAA-pyranoxanthone hybrids enhance inhibition activities against human cancer cells with multi-target functions. Eur. J. Med. Chem. 2018, 143, 1768–1778. [Google Scholar] [CrossRef]
- Anschütz, R.; Stoltenhoff, W.; Voeller, F. Über zwei gemischte Anhydro-monoxybenzoesäuren und ihre Umwandlung in Xanthon-carbonsäuren. Ber. Dtsch. Chem. Ges. A B Ser. 1925, 58, 1736–1741. [Google Scholar] [CrossRef]
- El-Abbady, A.M.; Ayoub, S.; Baddar, F.G. 517. β-Aroylpropionic acids. Part XVI. The conversion of γ-oxo-γ-2-xanthenylbutyric acid into 2,3-benzoxanthone. J. Chem. Soc. 1960, 0, 2556–2559. [Google Scholar] [CrossRef]
- Graham, R.; Lewis, J.R. A convenient synthesis of xanthone 2-carboxylic acids. Chem. Industr. 1977, 19, 798. [Google Scholar]
- Pickert, M.; Frahm, A.W. Substituted xanthones as antimycobacterial agents*, Part 1: Synthesis and assignment of 1H/13C NMR chemical shifts. Arch. Pharm. 1998, 331, 177–192. [Google Scholar] [CrossRef]
- Pfister, J.R.; Ferraresi, R.W.; Harrison, I.T.; Rooks, W.H.; Roszkowski, A.P.; Van Horn, A.; Fried, J.H. Xanthone-2-carboxylic acids, a new series of antiallergic substances. J. Med. Chem. 1972, 15, 1032–1035. [Google Scholar] [CrossRef] [PubMed]
- Jones, W.D.; Albrecht, W.L.; Munro, N.L.; Stewart, K.T. Antiallergic agents. Xanthone-2,7-dicarboxylic Acid Derivatives. J. Med. Chem. 1977, 20, 594–595. [Google Scholar] [CrossRef] [PubMed]
- Bristol, J.A.; Alekel, R.; Fukunaga, J.Y.; Steinman, M. Antiallergic activity of some 9H-xanthen-9-one-2-carboxylic acids. J. Med. Chem. 1978, 21, 1327–1330. [Google Scholar] [CrossRef]
- Pfister, J.R.; Ferraresi, R.W.; Harrison, I.T.; Rooks, W.H.; Fried, J.H. Synthesis and antiallergic activity of some mono- and disubstituted xanthone-2-carboxylic acids. J. Med. Chem. 1978, 21, 669–672. [Google Scholar] [CrossRef] [PubMed]
- Barnes, A.C.; Hairsine, P.W.; Matharu, S.S.; Ramm, P.J.; Taylor, J.B. Pharmacologically active sulfoximides: 5-hexyl-7-(S-methylsulfonimidoyl)xanthone-2-carboxylic acid, a potent antiallergic agent. J. Med. Chem. 1979, 22, 418–424. [Google Scholar] [CrossRef]
- Barnes, A.C.; Hairsine, P.W.; Kay, D.P.; Ramm, P.J.; Taylor, J.B. Thermal decomposition of a sulfoximide in the presence of a carboxylic acid; an interesting rearrangement. J. Heterocycl. Chem. 1979, 16, 1089–1091. [Google Scholar] [CrossRef]
- Pfister, J.R.; Wymann, W.E.; Mahoney, J.M.; Waterbury, L.D. Synthesis and aldose reductase inhibitory activity of 7-sulfamoylxanthone-2-carboxylic acids. J. Med. Chem. 1980, 23, 1264–1267. [Google Scholar] [CrossRef]
- Pfister, J.R. Application of the smiles rearrangement to the synthesis of 5,7-disubstituted xanthone-2-carboxylic acids. J. Heterocycl. Chem. 1982, 19, 1255–1256. [Google Scholar] [CrossRef]
- Jackson, W.T.; Boyd, R.J.; Froelich, L.L.; Gapinski, D.M.; Mallett, B.E.; Sawyer, J.S. Design, synthesis, and pharmacological evaluation of potent xanthone dicarboxylic acid leukotriene B4 receptor antagonists. J. Med. Chem. 1993, 36, 1726–1734. [Google Scholar] [CrossRef] [PubMed]
- Sawyer, J.S.; Baldwin, R.F.; Sofia, M.J.; Floreancig, P.; Marder, P.; Saussy, D.L. Jr.; Froelich, L.L.; Silbaugh, S.A.; Stengel, P.W.; Cockerham, S.L.; et al. Biphenylyl-substituted xanthones: Highly potent leukotriene B4 receptor antagonists. J. Med. Chem. 1993, 36, 3982–3984. [Google Scholar] [CrossRef] [PubMed]
- Sawyer, J.S.; Schmittling, E.A.; Bach, N.J.; Baker, S.R.; Froelich, L.L.; Saussy Jr, D.L.; Marder, P.; Jackson, W.T. Structural analogues of LY292728, a highly potent xanthone dicarboxylic acid leukotriene B4 receptor antagonist: Spatial positioning of the secondary acid group. Bioorg. Med. Chem. Lett. 1994, 4, 2077–2082. [Google Scholar] [CrossRef]
- Fonteneau, N.; Martin, P.; Mondon, M.; Ficheux, H.; Gesson, J.P. Synthesis of quinone and xanthone analogs of rhein. Tetrahedron 2001, 57, 9131–9135. [Google Scholar] [CrossRef]
- Fernandes, C.; Masawang, K.; Tiritan, M.E.; Sousa, E.; de Lima, V.; Afonso, C.; Bousbaa, H.; Sudprasert, W.; Pedro, M.; Pinto, M.M. New chiral derivatives of xanthones: Synthesis and investigation of enantioselectivity as inhibitors of growth of human tumor cell lines. Bioorg. Med. Chem. 2014, 22, 1049–1062. [Google Scholar] [CrossRef] [PubMed]
- Hernández, J.V.; Muñiz, F.M.; Oliva, A.I.; Simón, L.; Pérez, E.; Morán, J.N.R. A xanthone-based neutral receptor for zwitterionic amino acids. Tetrahedron. Lett. 2003, 44, 6983–6985. [Google Scholar] [CrossRef]
- Fernandes, C.; Oliveira, L.; Tiritan, M.E.; Leitao, L.; Pozzi, A.; Noronha-Matos, J.B.; Correia-de-Sa, P.; Pinto, M.M. Synthesis of new chiral xanthone derivatives acting as nerve conduction blockers in the rat sciatic nerve. Eur. J. Med. Chem 2012, 55, 1–11. [Google Scholar] [CrossRef]
- Carraro, M.L.; Palmeira, A.; Tiritan, M.E.; Fernandes, C.; Pinto, M.M.M. Resolution, determination of enantiomeric purity and chiral recognition mechanism of new xanthone derivatives on (S,S)-whelk-O1 stationary phase. Chirality 2017, 29, 247–256. [Google Scholar] [CrossRef]
- Fernandes, C.; Brandao, P.; Santos, A.; Tiritan, M.E.; Afonso, C.; Cass, Q.B.; Pinto, M.M. Resolution and determination of enantiomeric purity of new chiral derivatives of xanthones using polysaccharide-based stationary phases. J. Chromatogr. A 2012, 1269, 143–153. [Google Scholar] [CrossRef]
- Fernandes, C.; Tiritan, M.E.; Cass, Q.; Kairys, V.; Fernandes, M.X.; Pinto, M. Enantioseparation and chiral recognition mechanism of new chiral derivatives of xanthones on macrocyclic antibiotic stationary phases. J. Chromatogr. A 2012, 1241, 60–68. [Google Scholar] [CrossRef]
- Fernandes, C.; Palmeira, A.; Ramos, II.; Carneiro, C.; Afonso, C.; Tiritan, M.E.; Cidade, H.; Pinto, P.; Saraiva, M.; Reis, S.; Pinto, M.M.M. Chiral derivatives of xanthones: Investigation of the effect of enantioselectivity on inhibition of cyclooxygenases (COX-1 and COX-2) and binding interaction with human serum albumin. Pharmaceuticals 2017, 10, 50. [Google Scholar] [CrossRef] [PubMed]
- Sato, H.; Dan, T.; Onuma, E.; Tanaka, H.; Koga, H. Studies on uricosuric diuretics. I. Syntheses and activities of xanthonyloxyacetic acids and dihydrofuroxanthone-2-carboxylic acids. Chem. Pharm. Bull. 1990, 38, 1266–1277. [Google Scholar] [CrossRef] [PubMed]
- Zelaszczyk, D.; Lipkowska, A.; Szkaradek, N.; Słoczyńska, K.; Gunia-Krzyżak, A.; Librowski, T.; Marona, H. Synthesis and preliminary anti-inflammatory evaluation of xanthone derivatives. Heterocycl. Commun. 2018, 24, 231–236. [Google Scholar] [CrossRef]

| Comp. | R1 | R2 | R3 | R4 | R5 | R6 | R7 | REF |
|---|---|---|---|---|---|---|---|---|
| 162 | H | H | H | H | H | H | H | [151,152,153,154] |
| 163 | OMe | H | H | H | H | H | H | [155] |
| 164 | H | OMe | H | H | H | H | H | [155] |
| 165 | H | H | OMe | H | H | H | H | [155] |
| 166 | H | H | H | H | H | H | OMe | [155,169] |
| 167 | H | H | H | OMe | H | H | H | [155] |
| 168 | H | H | H | H | H | OMe | H | [155] |
| 169 | H | H | H | H | OMe | H | H | [155,163,167,169,172] |
| 170 | H | H | H | H | H | Me | H | [155] |
| 171 | H | H | H | H | H | C2H5 | H | [155] |
| 172 | H | H | H | H | H | C3H7 | H | [155] |
| 173 | H | H | H | H | H | i-C3H7 | H | [155] |
| 174 | H | H | H | H | H | sec-C4H9 | H | [155] |
| 175 | H | H | H | H | H | C5H11 | H | [155] |
| 176 | H | H | H | i-C3H7 | H | H | H | [155] |
| 177 | H | H | H | H | H | F | H | [155] |
| 178 | H | H | H | H | H | Cl | H | [153,155] |
| 179 | H | H | H | H | H | OH | H | [155] |
| 180 | H | H | H | H | H | OC2H5 | H | [155] |
| 181 | H | H | H | H | H | OC3H7 | H | [155] |
| 182 | H | H | H | H | H | i-OC3H7 | H | [155] |
| 183 | H | H | H | H | H | OC4H9 | H | [155] |
| 184 | H | H | H | i-OC3H7 | H | H | H | [155] |
| 185 | H | H | H | H | H | COOH | H | [155,156] |
| 186 | H | H | H | H | H | OCH2CH(OH)CH2SPh | H | [157] |
| 187 | H | H | H | H | H | OCH2CH(OH)CH2S(4-F-Ph) | H | [157] |
| 188 | H | H | H | H | H | OCH2CH(OH)CH2S(4-Cl-Ph) | H | [157] |
| 189 | H | H | H | H | H | OCH2CH(OH)CH2S(3,4-Cl2-Ph) | H | [157] |
| 190 | H | H | H | H | H | OCH2CH(OH)CH2S(4-Br-Ph) | H | [157] |
| 191 | H | H | H | H | H | OCH2CH(OH)CH2S(4-OCH3-Ph) | H | [157] |
| 192 | H | H | H | H | H | OCH2CH(OH)CH2SCH3 | H | [157] |
| 193 | H | H | H | H | H | OCH2CH(OH)CH2SC2H4OH | H | [157] |
| 194 | H | H | H | H | H | OCH2CH(OH)CH2SCH(CH3)2 | H | [157] |
| 195 | H | H | H | H | H | OCH2CH(OH)CH2SC(CH3)3 | H | [157] |
| 196 | H | H | H | H | H | OCH2CH(OH)CH2SC6H11 | H | [157] |
| 197 a | H | H | H | H | H | OCH2CH(OH)CH2S(1-adm) | H | [157] |
| 198 | H | H | H | H | H | OCH2CH(OH)CH2SC7H15 | H | [157] |
| 199 | H | H | H | H | H | OCH2CH(OH)CH2OH | H | [157,161] |
| 200 | H | H | H | H | H | OCH2CH(OH)CH2OCH3 | H | [157] |
| 201 | H | H | H | H | H | OCH2CH(OH)CH2OC2H4OH | H | [157] |
| 202 | H | H | H | H | H | OCH2CH(OH)CH2OC2H4OCH3 | H | [157] |
| 203 | H | H | H | H | H | OCH2CH(OH)CH2OCH2OF3 | H | [157] |
| 204 | H | H | H | H | H | OCH2CH(OH)CH2SOC6H5 | H | [157] |
| 205 | H | H | H | H | H | OCH2CH(OH)CH2SOCH3 | H | [157] |
| 206 | H | H | H | H | H | COCH3 | H | [158] |
| 207 | H | H | H | H | H | COC2H5 | H | [158] |
| 208 | H | H | H | H | H | i-COC3H7 | H | [158] |
| 209 b | H | H | H | H | H | COC3H5 | H | [158] |
| 210 c | H | H | H | H | H | COC5H9 | H | [158] |
| 211 | H | H | H | H | H | SH | H | [158] |
| 212 | H | H | H | SOCH3 | H | H | H | [158] |
| 213 | H | H | H | i-SOC3H7 | H | H | H | [158] |
| 214 | H | H | H | SCH3 | H | H | H | [158] |
| 215 | H | H | H | i-SC3H7 | H | H | H | [158] |
| 216 | H | H | H | SO2CH3 | H | H | H | [158] |
| 217 | H | H | H | OMe | H | OMe | H | [158] |
| 218 | H | H | H | H | OMe | OMe | H | [158] |
| 219 c | H | H | H | H | OMe | H | OMe | [158] |
| 220 | H | H | H | Me | H | Me | H | [158] |
| 221 | H | H | H | H | Me | Me | H | [158] |
| 222 | H | H | H | H | H | Me | Me | [158] |
| 223 | H | H | H | OMe | H | SCH3 | H | [158] |
| 224 | H | H | H | OEt | H | SOCH3 | H | [158] |
| 225 | H | H | H | OC3H7 | H | SOCH3 | H | [158] |
| 226 | H | H | H | i-OC3H7 | H | SOCH3 | H | [158] |
| 227 | H | H | H | OC4H9 | H | SOCH3 | H | [158] |
| 228 | H | H | H | OC5H11 | H | SOCH3 | H | [158,159] |
| 229 | H | H | H | i-OC5H11 | H | SOCH3 | H | [158] |
| 230 | H | H | H | OC5H9 | H | SOCH3 | H | [158] |
| 231 | H | H | H | OC8H17 | H | SOCH3 | H | [158] |
| 232 | H | H | H | H | H | SCH3 | H | [159] |
| 233 | H | H | H | C6H13 | H | SCH3 | H | [159,160] |
| 234 | H | H | H | OC5H11 | H | SCH3 | H | [159] |
| 235 | H | H | H | H | H | SOCH3 | H | [159,161] |
| 236 | H | H | H | C6H13 | H | SOCH3 | H | [159] |
| 237 | H | H | H | H | H | SO(=NH)CH3 | H | [159] |
| 238 | H | H | H | C6H13 | H | SO(=NH)CH3 | H | [159,160] |
| 239 | H | H | H | OC5H11 | H | SO(=NH)CH3 | H | [159] |
| 240 | H | H | H | H | H | SO(=NCONH2)CH3 | H | [159] |
| 241 | H | H | H | C6H13 | H | SO(=NCONH2)CH3 | H | [159] |
| 242 | H | H | H | H | H | SO(=NCOPh)CH3 | H | [159] |
| 243 | H | H | H | H | H | SO(=NCOCH3)CH3 | H | [159] |
| 244 | H | H | H | H | H | SO(=NCOOC2H5)CH3 | H | [159] |
| 245 d | H | H | H | H | H | SO(=N-Tos)CH3 | H | [159] |
| 246 | H | H | H | H | H | 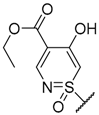 | H | [159] |
| 247 d | H | H | H | H | H | S(=N-Tos)CH3 | H | [159] |
| 248 | H | H | H | H | H | SO2Cl | H | [161] |
| 249 | H | H | H | H | H | SO3H | H | [161] |
| 250 | H | H | H | H | H | SO2NH2 | H | [161] |
| 251 | H | H | H | H | H | SO2NHCH3 | H | [161] |
| 252 | H | H | H | H | H | SO2NH(CH3)2 | H | [161] |
| 253 | H | H | H | H | H | SO2NH(CH3)C2H5 | H | [161] |
| 254 | H | H | H | H | H | SO2NH-i-C3H8 | H | [161] |
| 255 | H | H | H | H | H | SO2NH(CH3)-i-C3H8 | H | [161] |
| 256 | H | H | H | H | H | SO2NH(CH3)-i-C4H9 | H | [161] |
| 257 e | H | H | H | H | H | SO2-pyrr | H | [161] |
| 258 f | H | H | H | H | H | SO2-morp | H | [161] |
| 259 | H | H | H | H | H | SO2NHC2H4OH | H | [161] |
| 260 | H | H | H | H | H | SO2NH(CH3)C2H4OH | H | [161] |
| 261 | H | H | H | H | H | SO2NH(C2H4OH)2 | H | [161] |
| 262 | H | H | H | H | H | SC2H4OH | H | [161] |
| 263 | H | H | H | H | H | SOC2H4OH | H | [161] |
| 264 | H | H | H | H | H | SO2C2H4OH | H | [161] |
| 265 | H | H | H | H | H | SOC2H4OCH3 | H | [161] |
| 266 | H | H | H | H | H | CH(OH)CH3 | H | [161] |
| 267 | H | H | H | H | H | CH(OCH3)CH3 | H | [161] |
| 268 | H | H | H | i-C3H8 | H | i-C3H8 | H | [162] |
| 269 | H | H | H | t-C4H9 | H | t-C4H9 | H | [162] |
| 270 | H | H | H | H | OC10H21 | C2H4COOH | H | [163] |
| 271 | H | H | H | C2H4COOH | OC10H21 | H | H | [163] |
| 272 | H | H | H | C2H4COOH | OC4H8CH=CH(4-OMe-Ph) | H | H | [163] |
| 273 | H | H | H | C2H4COOH | OC3H6O(4-COCH3-2-Et-5-OH-Ph) | H | H | [163,164] |
| 274 | H | H | H | C2H4COOH | OC3H6O(5-Et-4′-F-2-OH-1,1′-Ph2) | H | H | [164,165] |
| 275 | H | H | H | COOH | H | H | H | [154] |
| 276 | H | H | H | COOH | H | NO2 | H | [154] |
| 277 | H | H | H | H | H | NO2 | H | [154] |
| 278 | H | H | NO2 | H | H | NO2 | H | [154] |
| 279 | H | H | NO2 | COOH | H | NO2 | H | [154] |
| 280 | H | H | H | H | H | NH2 | H | [154] |
| 281 | H | H | OCOCH3 | H | H | H | H | [166] |
| 282 | H | H | OCOCH3 | OCOCH3 | H | H | H | [166] |
| 283 | H | H | OH | OH | H | H | H | [166] |
| 284 | H | H | NH2 | NO2 | H | tert-Butyl | H | [168] |
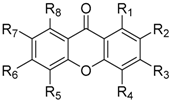
| Comp. | R1 | R2 | R3 | R4 | R5 | R6 | R7 | R8 | REF |
|---|---|---|---|---|---|---|---|---|---|
| 285 | COOH | H | H | H | H | H | H | H | [151,154] |
| 286 | H | H | COOH | H | H | H | H | H | [151,154] |
| 287 | H | H | H | COOH | H | H | H | H | [151,154] |
| 288 | H | COC2H4COOH | H | H | H | H | H | H | [152] |
| 289 | H | H | OCH2COOH | H | H | H | H | H | [169,174] |
| 290 | H | H | OCH2COOH | H | H | H | H | F | [167,174] |
| 291 | H | H | OCH2COOH | Cl | H | H | H | F | [174] |
| 292 | H | H | OCH2COOH | H | H | H | H | F | [174] |
| 293 | H | H | OCH2COOH | Me | H | H | H | F | [174] |
| 294 | H | H | OCH2COOH | Cl | H | H | H | Cl | [174] |
| 295 | H | H | OCH2COOH | Cl | H | H | Cl | H | [174] |
| 296 | H | H | OCH2COOH | Cl | H | Cl | H | H | [174] |
| 297 | H | H | OCH2COOH | Cl | Cl | H | H | H | [174] |
| 298 | Cl | Cl | OCH2COOH | H | H | H | H | H | [174] |
| 299 | H | Cl | OCH2COOH | Cl | H | H | H | H | [174] |
| 300 | Cl | H | OCH2COOH | H | H | H | H | H | [174] |
| 301 | H | Cl | OCH2COOH | H | H | H | H | H | [174] |
| 302 | H | H | OCH2COOH | Cl | H | H | H | H | [174] |
| 303 | Me | H | OCH2COOH | H | H | H | H | H | [174] |
| 304 | H | Me | OCH2COOH | H | H | H | H | H | [174] |
| 305 | H | H | OCH2COOH | Me | H | H | H | H | [174] |
| 306 | H | Br | OCH2COOH | H | H | H | H | H | [174] |
| 307 | H | H | OCH(COOH)CH2 | H | H | H | H | [174] | |
| 308 | H | H | OCH(COOH)CH2 | H | H | H | F | [174] | |
| 309 | H | H | OCH(COOH)CH2 | H | H | H | Cl | [174] | |
| 310 | H | Cl | OCH(COOH)CH2 | H | H | H | H | [174] | |
| 311 | Cl | H | OCH(COOH)CH2 | H | H | H | H | [174] | |
| 312 | H | Me | OCH(COOH)CH2 | H | H | H | H | [174] | |
| 313 | Me | H | OCH(COOH)CH2 | H | H | H | H | [174] | |
| 314 | Br | H | OCH(COOH)CH2 | H | H | H | H | [174] | |
| 315 | Cl | Me | OCH(COOH)CH2 | H | H | H | H | [174] | |
| 316 | H | CH2CH(COOH)O | Cl | H | H | H | F | [174] | |
| 317 | H | CH2CH(COOH)O | Me | H | H | H | F | [174] | |
| 318 | H | CH2CH(COOH)O | Cl | H | H | H | Cl | [174] | |
| 319 | H | CH2CH(COOH)O | Cl | H | H | H | H | [174] | |
| 320 | H | CH2CH(COOH)O | Me | H | H | H | H | [174] | |
| 321 | H | H | H | COOH | H | OC10H21 | C2H4COOH | H | [163] |
| 322 | H | H | H | COOH | C2H4COOH | OC10H21 | H | H | [163] |
| 323 | H | H | H | H | C2H4COOH | OC3H6O-(5-Et-4′-F-2-OH-1,1′-Ph2) | H | H | [164] |
| 324 | COOH | H | H | H | H | H | NO2 | H | [154] |
| 325 | H | H | COOH | H | H | H | NO2 | H | [154] |
| 326 | H | H | H | COOH | H | H | NO2 | H | [154] |
| 327 | H | H | COOH | COOH | H | H | NO2 | H | [154] |
| 328 | COOH | NO2 | H | H | H | H | NO2 | H | [154] |
| 329 | H | NO2 | COOH | H | H | H | NO2 | H | [154] |
| 330 | H | NO2 | H | COOH | H | H | NO2 | H | [154] |
| 331 | H | NO2 | COOH | COOH | H | H | NO2 | H | [154] |
| 332 | H | H | COOH | H | H | H | NH2 | H | [154] |
| 333 | H | H | OC(CH3)2COOH | H | CH3 | H | H | H | [175] |
| 334 | H | H | OCH2COOH | H | H | H | CH3 | H | [175] |
| 335 | H | H | OCH2COOH | H | CH3 | H | H | H | [175] |
| 336 | H | H | OCH(CH3)COOH | H | H | H | CH3 | H | [175] |
| 337 | H | H | OC(CH3)2COOH | H | H | H | CH3 | H | [175] |
| 338 | H | H | H | OCH(CH3)COOH | H | Cl | H | H | [175] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ribeiro, J.; Veloso, C.; Fernandes, C.; Tiritan, M.E.; Pinto, M.M.M. Carboxyxanthones: Bioactive Agents and Molecular Scaffold for Synthesis of Analogues and Derivatives. Molecules 2019, 24, 180. https://doi.org/10.3390/molecules24010180
Ribeiro J, Veloso C, Fernandes C, Tiritan ME, Pinto MMM. Carboxyxanthones: Bioactive Agents and Molecular Scaffold for Synthesis of Analogues and Derivatives. Molecules. 2019; 24(1):180. https://doi.org/10.3390/molecules24010180
Chicago/Turabian StyleRibeiro, João, Cláudia Veloso, Carla Fernandes, Maria Elizabeth Tiritan, and Madalena M. M. Pinto. 2019. "Carboxyxanthones: Bioactive Agents and Molecular Scaffold for Synthesis of Analogues and Derivatives" Molecules 24, no. 1: 180. https://doi.org/10.3390/molecules24010180
APA StyleRibeiro, J., Veloso, C., Fernandes, C., Tiritan, M. E., & Pinto, M. M. M. (2019). Carboxyxanthones: Bioactive Agents and Molecular Scaffold for Synthesis of Analogues and Derivatives. Molecules, 24(1), 180. https://doi.org/10.3390/molecules24010180









