1. Introduction
The seeds of cultivated peanut,
Arachis hypogaea, store proteins, lipids and starch required for energy and growth upon germination. The seed can be harvested to serve as human nutrition, stock feed and biofuels [
1,
2,
3]. Peanuts and other legumes have the ability to fix nitrogen and thus increase the sustainability of agricultural systems [
4]. The pressure for peanut seed as plant-based protein and oilseed source is increasing with a growing world population. Due to its agronomic importance, improvement of peanut seed performance is necessary to meet the demand.
Lipid (oil) is the predominant macro component, and generally increases as the peanut seed matures [
5]. For mature peanuts, total oil was reported to average about 50% on a fresh weight basis [
6]. Oilseeds, such as peanuts, store most of their lipids in small, intracellular organelles commonly called oil bodies [
7,
8]. Triglycerides form the majority of these oil bodies, and the interior triglycerides are encapsulated by a phospholipid bilayer and embedded oleosin protein. The oil bodies provide a stable energy reserve that can be accessed upon germination [
9].
Phytosterols are a special class of structural lipids that provide stability and fluidity in cell membranes including specialized encapsulation of lipids, such as oil bodies [
10]. The primary function of phytosterols are as membrane reinforcers and precursors for brassinosteroids, an important phytohormone in plants. Unlike animal and fungi counterparts with cholesterol and ergosterol as the dominant sterol, the plant usually synthesizes an array of sterols with different alkyl group substitutions at the sterol side chain. In addition to the primary function, the correct sterol composition is necessary for many aspects of plant biology such as embryonic pattern formation, cell division, cell elongation, cell polarity, cellulose accumulation, and interactions with other signaling pathways [
11].
Physiological maturity impacts seed quality through a variety of mechanisms including desiccation tolerance, preparation of storage reserves and establishment of dormancy [
12]. Thus, the impact of seed maturity on germination efficiency is of primary importance to peanut producers. To best assess physiological maturation, a method of classification based on color and morphological differences of the mesocarp was described for determining the developmental stages of fresh peanut [
13]. Maturity determination by this method requires removal of a portion of the exocarp to expose the pod mesocarp. The characteristic darkening of the pericarp is part of a progressive change in colors resulting in mesocarp colors from white (immature) to black (mature) [
14]. The outer layer of the pod can be removed, and seeds sorted by maturity class (white, yellow, orange, brown or black) in a nondestructive manner allowing for physiological and chemical studies pertaining to the stage of development [
12].
Seed maturity proceeds with the thickening of cell walls and addition of oil bodies [
15]. For mature peanuts of the highest classification (black mesocarp), the cytoplasm of the parenchyma cells is essentially full of oil bodies. The specific aim of this project seeks to clarify the role of phytosterols in the formation of oil bodies as the seed matures. To accomplish this aim, seeds were organized into different maturity classes and the phytosterols extracted and identified. It is hypothesized that establishment of a critical mass of phytosterols provides the membrane stability for proliferation of oil bodies during maturation.
2. Results
2.1. Stage of Peanut Maturity and Macronutrient Composition in Different Maturity Classes
Peanut seed development can be classified into seven classes with four incremental stages in each class [
13]. Based on the color of the mesocarp, the last three classes are described and named as “orange”, “brown” or “black”. Pooled samples from each maturity classes were analyzed to determine macronutrient percentages, and results are summarized in
Table 1. The data reported is a check for comparison against the Biochemistry Research Laboratory database of crop samples from 2011–2018. The reported results in
Table 1 are consistent with observed trends in peanut crop analysis conducted by the Biochemistry Research Laboratory at Lubbock Christian University (LCU) during the 2011–2018 CYs. The USDA National Nutrient Database for Standard Reference (Release 27, Basic Report 16087) reports macronutrient composition as 49.2% fat, 25.8% protein and 4.7% sugars. The reported results are within expected ranges as compared to the standard values. Any slight variation could be attributed to a specific variety versus average trends across all market types of cultivated peanut for a specific crop year.
2.2. Isoprenoids and Phytosterol Composition in Different Tissues
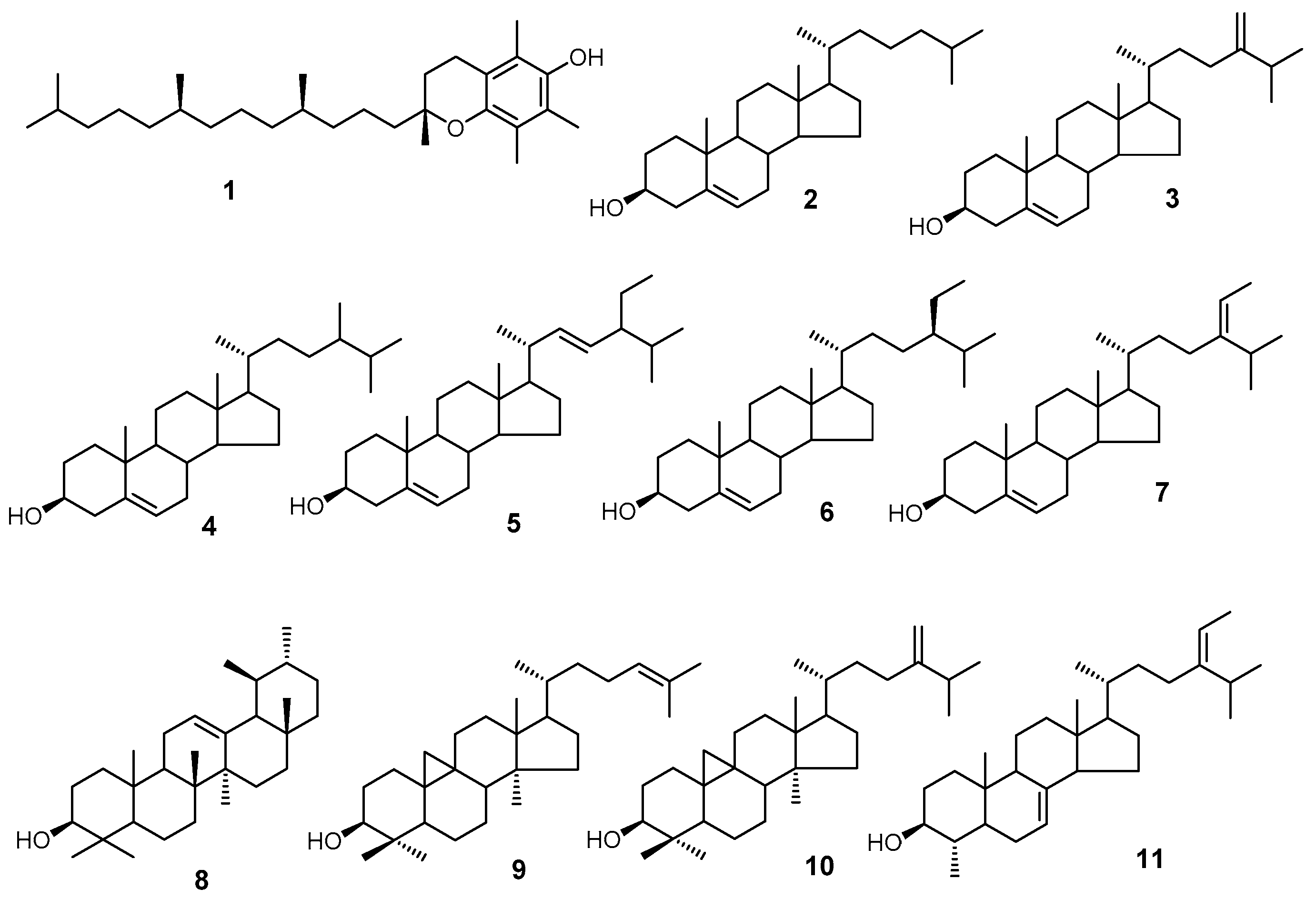
Using GC/MS, we analyzed three classes of peanut samples for their isoprenoid and sterol composition. We have positively identified 11 compounds (
Figure 1) based on the mass spectra and retention time relative to the authentic standards.
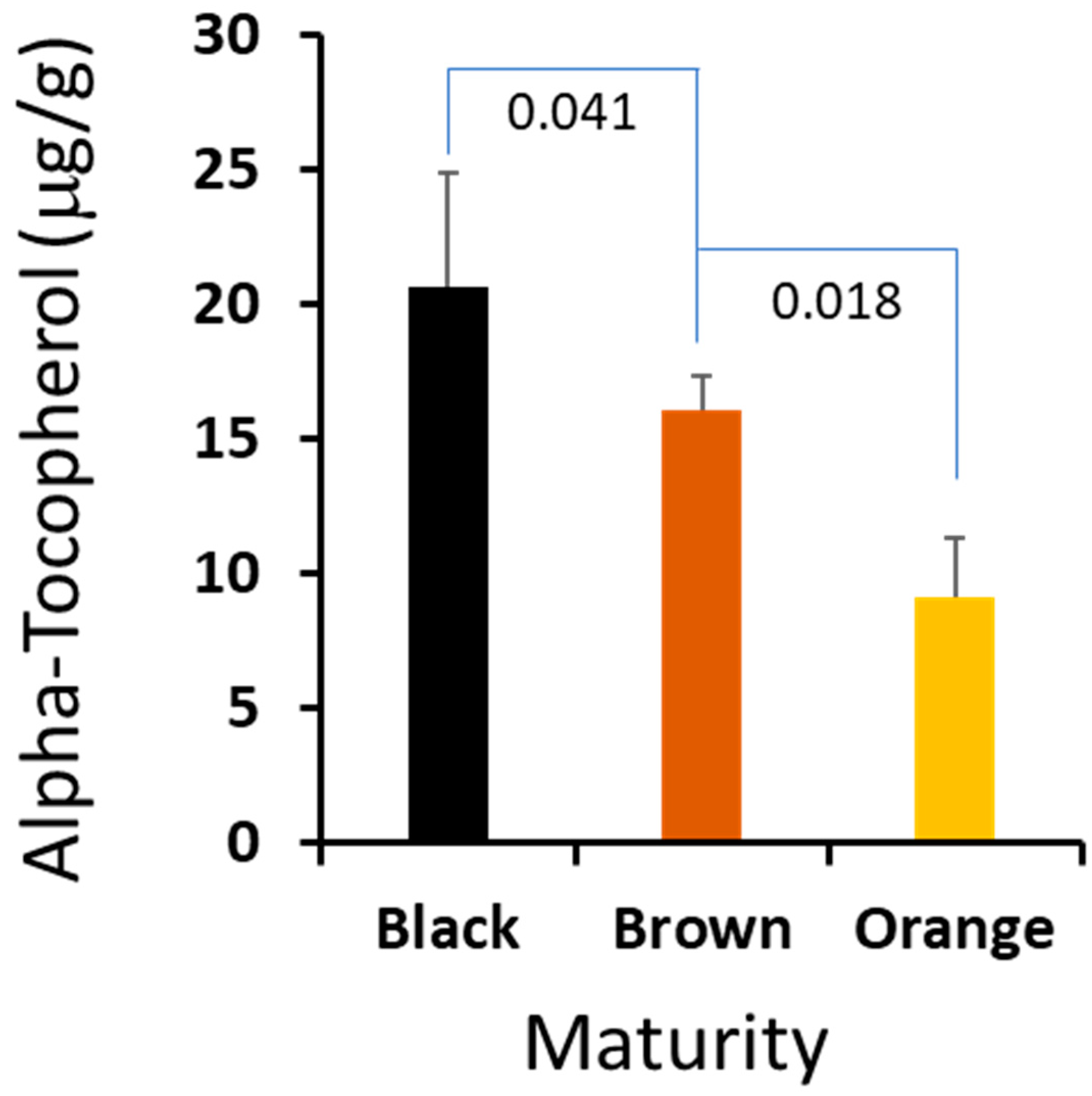
We found that of the three seed component classes analyzed, the kernel and seed coat generally contained the same percentage of α-tocopherol (0.63% and 0.64%, respectively), decreasing slightly with maturity in brown versus black mesocarp classes, 0.69% to 0.63% kernel composition and 0.71% to 0.64% seed coat composition (
Figure 2 and
Table 2,
Table 3 and
Table 4). In contrast, the hearts contained significantly less tocopherol by percentage (
Table 2,
Table 3,
Table 4 and
Table 5) as compared to the kernel and seed coat. Also, the amount of α-tocopherol increased from 0.09% in the orange mesocarp class, to 0.19% in the brown mesocarp class, and 0.24% in the black mesocarp class. Vitamin E comprises eight structurally related molecules including four forms of tocopherols. Peanuts, like many other oilseeds, contain tocopherols [
16]. Within plants, these molecules are found in cell membranes and possess antioxidant activity which protects organelles from reactive oxygen species (ROS) [
17]. Like phytosterols, tocopherols contribute to maintaining membrane fluidity [
17].
Many phytosterols are present in nature [
18]. In plants, the three biosynthetic end products in the phytosterol pathway are β-sitosterol, campesterol and stigmasterol. In 2004, it was reported that these three sterols account for approximately 95% of total peanut sterols [
18]. Our analysis confirmed that these sterols are major components of total peanut sterols in each component, 82.29% of hearts, 86.39% of kernels and 94.25% seed coats (
Table 2,
Table 3 and
Table 4), but at lower percentages as compared to the 2004 reported values. Other minor component phytosterols were detected (
Table 2,
Table 3 and
Table 4) including those not previously identified most likely due to advances in technology. Squalene and γ-tocopherol as reported by Maguire et al. [
18] were not detected. This discrepancy of results can be explained in addition to sample variations by two possible explanations. Firstly, squalene is the substrate for phytosterol, whereas γ-tocopherol can be converted to α-tocopherol via a methylation reaction. Depending on the harvest time, these two compounds may be converted to their final products and therefore are undetectable. Secondly, in the references, these sterols and tocopherols were determined by an HPLC method. Because of the differences in UV absorption between the two classes of compounds, tocopherol can be easily overestimated. Also, there were no internal standards used in the literature to correct the extraction and HPLC injection errors, as well as a reference for quantitation. Some variation may also be due to varietal differences, as these samples were isolated from Runner-type peanuts as compared to Spanish, Valencia and Virginia market types.
When comparing sterol composition in the different seed components, the relative percentage of stigmasterol should be noted (
Table 2,
Table 3 and
Table 4), heart 2.80%, kernel 11.31% and seed coat 4.09%. This result differs from the reported value of stigmasterol in Runner peanuts as 11.0% [
18]. However, the kernel is the largest component of the seed by mass, and as a result, would more closely reflect data on the entire seed.
2.3. Isoprenoid and Phytosterol Composition in Different Maturity Classes
It is critical for farmers to harvest peanut at the optimized maturity to maximize the crop value, and a suitable biomarker could help farmers to harvest at the right time. We compared the profiles of the sterols and isoprenoids from the three stages, and there are no statistically significant differences among the major sterols. However, we found that α-tocopherol in the peanut hearts changed dramatically crossing the maturation stages. The absolute amount of this important metabolite was increased correspondingly from 0.92 μg/mg in orange to 1.61 μg/mg in brown, and peaked at 2.08 μg/mg in black. Given the fact that vitamin E is a very important metabolite to plant biology and a valuable compound to human health, we think that the content of vitamin E could be used as a biomarker for peanut harvesting. We plan to develop a user-friendly and portable method to determine vitamin E content, which may have important practical value to peanut farmers.
3. Discussion
Peanuts are a nutrient rich plant-based protein source that contain vitamins, minerals, antioxidants and bioactive phytochemicals, leading to the perception that peanuts are a “super food” [
19]. Peanut phytosterols have been shown to help lower LDL cholesterol by competing in the digestive tract with cholesterol and preventing absorption [
20]. One of the major phytosterols, stigmasterol, has been investigated for its pharmacological importance as an antihypercholesterolemic, anti-inflammatory, antioxidant, hypoglycemic and antitumor effector [
21,
22]. In this study, it is reported that the kernel contains a higher percentage of stigmasterol as compared to the heart and seed coat. Different manufacturing processes may remove the seed coat or heart, so it is beneficial to the health of the consumer that the kernel possess relatively high concentrations of stigmasterol.
In addition to potential health benefits, stigmasterol is thought to play a role in temperature stress tolerance in plants [
23]. Drought and extreme heat in the growing season can increase the sensitivity of the plant to opportunistic organisms [
24]. Accumulation of critical phytosterols, such as stigmasterol, during pod development may set the foundation for physiological maturation processes and resistance to stress.
Tocopherol content can vary with environmental stress and growing location in addition to other factors [
25]. In this study, hearts contained the lowest percentage of α-tocopherol as compared to the other seed components, but the amount increased during maturation. During germination, the stability of the peanut heart, or embryo, is critically important to the development of the seedling and stand establishment under adverse environmental conditions [
17]. α-tocopherol deactivates ROS generated during photosynthesis and is upregulated during stressful events [
17]. Immature peanut seed is less resistant to stress, and as a result, is more likely to be adversely affected during germination. The results suggest that the synthesis and accumulation of α-tocopherol in developing peanut hearts may be vitally important to seedling vigor upon germination.
4. Materials and Methods
4.1. Materials and Reagents
Epicoprostanol, 5β-cholestan-3α-ol, heptane (99%), anhydride pyridine (99.9%), and N,O-bis(trimethylsilyl)trifluoroacetamide (BSTFA) (99.9%) were purchased from Sigma-Aldrich (St. Louis, MO, USA). HPLC-grade n-hexane, HPLC-grade methanol, potassium hydroxide (85%) (KOH), acetone, and HPLC-grade dichloromethane were purchased from Thermo Fisher (Waltham, MA, USA).
4.2. Pod Blasting
Freshly harvested pods, 2017 crop year (CY), from one specific genotype of runner market-type peanuts grown under conventional cultural practices were obtained from the University of Georgia research facility under the direction of Dr. W.D. Branch. Pods were removed from the plant material, and ‘pod blasted’ to reveal the mesocarp. Pod blasting is a process by which in-shell peanut pods are placed in a wire basket and a residential-style pressure washer is used to spray the shell exterior with high-pressure water, removing the outer portion of the peanut hull and exposing the colored mesocarp layer underneath. The blasted pods were separated by color into three different maturity classes, orange, brown and black. After separation, the remainder of the pod outer layer was removed, and the seeds segregated for additional chemical analyses. Upon receipt at the research laboratory at LCU, samples were examined and verified to be A. hypogaea runner type seeds by technicians under the supervision of Dr. Julie Marshall.
4.3. Isolation of Peanut Seed Components
For each maturity class, 10–50 g of redskin kernels were weighed and dried in a forced-air oven (VWR, Radnor, PA, USA) at 130 °C for 45 min. After cooling to ambient temperature, the kernels were manually separated by removing the skin, breaking open the seed, and removing the heart to subdivide the samples into three subsections consisting of seed coats, hearts and kernels. The subsections were scaled at a specific mass for 5 replicates. Each replicate of the heart and kernel contained 50 mg while the seed coat replicates contained 20 mg. Each scaled replicate was placed in a 2 mL microcentrifuge tube with locking lid.
4.4. Fat, Protein and Sugar Analysis
To analyze the seed sample for total fat by organic solvent extraction, the exact mass of 10 g ± 0.1 g sample was recorded and the sample pre-dried in a forced-air oven at 130 °C for 45 min to remove moisture. After cooling to ambient temperature, the dried sample was quantitatively transferred to an explosion-proof blender jar (Eberbach Corporation, Belleville, MI, USA). 60 mL dichloromethane (DCM) was added and the mixture blended at high speed for 1 min. After allowing the blender jar to cool for 30 s before opening, we removed the blender lid and washed down the sides of the blender with DCM in a wash bottle. We replaced the lid and blended at high speed for an additional 1 min. We allowed the blender jar to cool for 30 s before opening and washing down the sides of the blender with DCM a second time to remove all residue. We carefully poured the blender contents into a Büchner funnel vacuum filter apparatus with a Toxicity Characteristics Leaching Procedure (TCLP) glass fiber filter, rinsing the blender jar residue with DCM into the funnel until all residue was removed. We filtered the mixture, and transferred the filtrate from the vacuum flask to a tared stainless beaker, rinsing the vacuum flask with DCM into the stainless beaker to ensure all residue was transferred. We evaporated the solvent in the stainless flask over a steam bath until all solvent had been removed. We monitored evaporation and weighed the beaker/remaining oil as needed by removing the beaker from the steam bath and allowing it to come to ambient temperature. Evaporation was complete when the mass of the oil remaining in the stainless beaker stabilized (≤0.03 g change in mass over a 30-min span on the steam bath). We recorded the final weight of the beaker and oil, and calculated the percent oil using the following formula:
The protein analysis (reference methods AOAC 992.15; AACC 46-30) was conducted by Medallion Labs (Minneapolis, MN, USA) and the sugar analysis (sugar by HPLC) was conducted by North Carolina Extension/North Carolina State University (Raleigh, NC, USA).
4.5. Preparation of Nonsaponifiable Fraction (NSF)
We prepared 10% KOH/methanolic solution by dissolving 50 g KOH in 50 mL deionized water and bringing to volume of 500 mL with methanol. We prepared internal standard by mixing the epicoprostanol 5β-cholestan-3α-ol with heptane to a final concentration of 1mg/mL. After adding 25 µL of internal standard solution and 1 mL 10% methanolic KOH to each tube, the samples were saponified at 80 °C for 2 h using a Thermomixer (Eppendorf, Hamburg, Germany) with constant shaking at 500 rpm. Once cooled to ambient temperature, the nonsaponifiable fraction (NSF) containing free sterols was extracted with 1 mL of n-hexane. The hexane was pooled in a 1.5 mL microcentrifuge tube and the hexane was removed by evaporation in a fume hood overnight. To the residual, 20 µL of acetone was added to dissolve the sterols. After solvation, the compounds were converted to their trimethylsilyl ester by adding 10 μL of BSTFA and 10 μL of pyridine as catalyst. The derivatization mixtures were kept at room temperature for 30 min before GC/MS analysis.
4.6. GC/MS Analysis
2 µL of derivatization mixture was injected into an Agilent GC/MS (Agilent 6890 BC coupled with 5973 mass-selective detector (MSD)) (Agilent, Santa Clara, CA, USA). The GC was equipped with an Agilent DB-5Ms+DG narrow-bore capillary column (30 m × 0.25 mm × 0.25 µm with 10 m Duraguard). The injection mode was splitless, with helium carrier gas at a constant flow of 1.2 mL/min. The GC oven was initialed at 170 °C, held for 1 min, the temperature was ramped to 280 °C at 40 °C/min and held at 280 °C for 25 min. The MSD was in electron ionization (EI) mode, scan range was from 50–550 amu, temperature of the ion source was 230 °C, the quadrupole temperature was 150 °C, and the interface was 280 °C.
The GC/MS data was processed with ChemStation software (Version E.02.02.1431, Agilent, Santa Clara, CA, USA) and Automated Mass Spectral Deconvolution & Identification System (AMDIS) (National Institute of Standards and Technology, United States Department of Commerce, Washington, DC, USA). The sterol peaks were deconvoluted using AMDIS after baseline correction and identified by their relative retention time to cholesterol and comparison to the mass spectra from commercial mass database (NIST08 mass spectral library,
http://nistmassspeclibrary.com/). The GC peak representing the sterol amount generated from total ion current (TIC) was integrated using the software default parameters [
26,
27].
5. Conclusions
Physiological maturity of A. hypogaea seeds is impactful to germination efficiency, and consequently, is of economic importance to the peanut industry. Immature seeds have reduced germination frequency, are more susceptible to disease, and require more financial inputs during manufacturing processes which are dependent on fat content or roasting performance. This study seeks to characterize phytosterol and isoprenoid content in seed components as a function of physiological maturation indicated by mesocarp color class.
Peanut hearts contained the highest percentage of phytosterols and isoprenoids by mass as compared to the other seed components. The stability of the peanut heart, or embryo, is critically important to the development and vigor of the seedling during germination and plant development. Tocopherols provide plants with antioxidant capacity and provide fluidity and flexibility to cell membranes. α-tocopherol concentration in the hearts changed dramatically across maturation classes, providing a possible causal agent for poor stress adaptability in immature seeds. Given the economic importance of seed efficiency and performance in all sectors of the peanut industry, α-tocopherol content in the seed embryo could be utilized as a biomarker to optimize harvest timing and profitability. In the subsequent study, we will investigate more details to evaluate using α-tocopherol as a biomarker for peanut harvesting, and will develop a sensitive and portable α-tocopherol detection method for peanut farmers.
Stigmasterol, one of the three end products in the phytosterol pathway in A. hypogaea, is a significant component by percentage of seeds. The seed coat, or skin, is known to be a major contributor of antioxidant capacity and other health benefits. The relatively high concentration of stigmasterol may contribute to the health benefits reported by the addition of peanut skins to various manufactured goods. Also, stigmasterol biosynthesis is important in plant physiology to deal with low and high temperature stresses, and plays a key role in innate immunity to combat biotic stresses. Since the main function of the seed coat is to protect the embryo, high stigmasterol content may be important for seed storage and germination.
Author Contributions
The author contributions are as follows: conceptualization, W.Z. and J.A.M.; methodology, W.Z., W.D.B., L.G. and J.A.M.; formal analysis, W.Z., L.G. and J.A.M.; resources, W.D.B. and L.G.; data curation, W.Z. and L.G.; writing-original draft preparation, W.Z., L.G. and J.A.M.; writing-review and editing, L.G. and J.A.M.; project administration, J.A.M.; and funding acquisition, J.A.M.
Funding
This research was partially funded by The Welch Foundation, grant number BV-0043.
Acknowledgments
The authors would like to acknowledge Chris Cobos for technical support, Joshua Thomas and Melanie McGilton for literature research and manuscript preparation.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Duranti, M.; Gius, C. Legume seeds: Protein content and nutritional value. Field Crop. Res. 1997, 53, 31–45. [Google Scholar] [CrossRef]
- Djemel, N.; Guedon, D.; Lechevalier, A.; Salon, C.; Miquel, M.; Prosperi, J.M.; Rochat, C.; Boutin, J.P. Development and composition of the seeds of nine genotypes of the Medicago truncatula species complex. Plant Physiol. Biochem. 2005, 43, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Gallardo, K.; Thompson, R.; Burstin, J. Reserve accumulation in legume seeds. Comptes. Rendus. Biol. 2008, 331, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Siddique, K.H.; Johansen, C.; Turner, N.C.; Jeuffroy, M.H.; Hashem, A.; Sakar, D.; Gan, Y.; Alghamdi, S.S. Innovations in agronomy for food legumes. A review. Agron. Sustain. Dev. 2012, 32, 45–64. [Google Scholar] [CrossRef]
- Patee, H.E.; Johns, E.B.; Singleton, J.A.; Sanders, T.H. Composition Changes of Peanut Fruit Parts During Maturation. Peanut. Sci. 1974, 1, 57–62. [Google Scholar] [CrossRef]
- Patee, H.E.; Salunkhe, D.K.; Sathe, S.K.; Reddy, N.R.; Ory, R.L. Legume lipids. CRC Crit. Rev. Food Sci. Nutr. 1983, 17, 97–139. [Google Scholar] [CrossRef]
- Young, C.T.; Schadel, W.E. Microstructure of Peanut Seed: A Review. Food Struct. 1990, 9, 317–328. [Google Scholar]
- Huang, A.H.C. Oil Bodies and Oleosins in Seeds. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1992, 43, 177–200. [Google Scholar] [CrossRef]
- Tzen, J.T.C.; Cao, Y.Z.; Laurent, P.; Ratnayake, C.; Huang, A.H.C. Lipids, Proteins, and Structure of Seed Oil Bodies from Diverse Species. Plant Physiol. 1993, 101, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Dufourc, E.J. Sterols and membrane dynamics. J. Chem. Biol. 2008, 1, 63–77. [Google Scholar] [CrossRef] [PubMed]
- Boutté, Y.; Grebe, M. Cellular processes relying on sterol function in plants. Cur. Opin. Plant Biol. 2009, 12, 705–713. [Google Scholar] [CrossRef] [PubMed]
- Ventura, L.; Doná, M.; Macovei, A.; Carbonera, D.; Buttafava, A.; Mondoni, A.; Rossi, G.; Balestrazzi, A. Understanding the molecular pathways associated with seed vigor. Plant Physiol. Biochem. 2012, 60, 196–206. [Google Scholar] [CrossRef]
- Williams, E.J.; Drexler, J.S. A Non-Destructive Method for Determining Peanut Pod Maturity. Peanut Sci. 1981, 8, 134–141. [Google Scholar] [CrossRef]
- Drexler, J.E.; Williams, E.J. A Non-Destructive Method of Peanut Pod Maturity Classification; American Peanut Research and Education Association: Tifton, GA, USA, 1979. [Google Scholar]
- Young, C.T.; Pattee, H.E.; Schadel, W.E.; Sanders, T.H. Microstructure of Peanut (Arachis hypogaea L. cv. ‘NC 7’) Cotyledons During Development. LWT-Food Sci. Technol. 2004, 37, 439–445. [Google Scholar] [CrossRef]
- Carrin, M.E.; Carelli, A.A. Peanut Oil: Compositional Data. Eur. J. Lipid Sci. Technol. 2010, 112, 697–707. [Google Scholar] [CrossRef]
- Munné-Bosch, S.; Alegre, L. The Function of Tocopherols and Tocotrienols in Plants. Crit. Rev. Plant Sci. 2002, 21, 31–57. [Google Scholar] [CrossRef]
- Maguire, L.; O’Sullivan, S.; Galvin, K.; O’Connor, T.; O’Brien, N. Fatty acid profile, tocopherol, squalene and phytosterol content of walnuts, almonds, peanuts, hazelnuts and the macadamia nut. Int. J. Food Sci. Nutr. 2004, 55, 171–178. [Google Scholar] [CrossRef]
- U.S. Department of Agriculture. USDA National Nutrient Database for Standard Reference. Available online: https://www.ars.usda.gov/nea/bhnrc/ndl (accessed on 8 October 2018).
- Jang, M.; Cai, L.; Udeani, G.O.; Slowing, K.V.; Thomas, C.F.; Beecher, C.W.W.; Fong, H.H.; Farnsworth, N.R.; Kinghorn, A.D.; Mehta, R.G.; et al. Cancer Chemopreventive Activity of Resveratrol, A Natural Product Derived From Grapes. Science 2008, 57, 130–139. [Google Scholar] [CrossRef]
- Kaur, N.; Chaudhary, J.; Jain, A.; Kishore, L. Stigmasterol: A comprehensive review. Int. J. Pharm. Sci. Res. 2011, 2, 2259. [Google Scholar] [CrossRef]
- Burg, V.K.; Grimm, H.S.; Rothhaar, T.L.; Grösgen, S.; Hundsdörfer, B.; Haupenthal, V.J.; Zimmer, V.C.; Mett, J.; Weingärtner, O.; Laufs, U.; et al. Plant Sterols the Better Cholesterol in Alzheimer’s Disease? A Mechanistical Study. J. Neurosci. 2013, 33, 16072–16087. [Google Scholar] [CrossRef] [PubMed]
- Senthil-Kumar, M.; Wang, K.; Mysore, K.S. AtCYP710A1 gene-mediated stigmasterol production plays a role in imparting temperature stress tolerance in Arabidopsis thaliana. Plant Signal. Behav. 2013, 8, e23142. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Senthil-Kumar, M.; Ryu, C.M.; Kang, L.; Mysore, K.S. Phytosterols Play a Key Role in Plant Innate Immunity against Bacterial Pathogens by Regulating Nutrient Efflux into the Apoplast. Plant Physiol. 2012. [Google Scholar] [CrossRef] [PubMed]
- Hashim, I.; Koehler, P.; Eitenmiller, R. Tocopherols in Runner and Virginia Peanut Cultivars at Various Maturity Stages. J. Am. Oil Chem. Soc. 1993, 70, 633–635. [Google Scholar] [CrossRef]
- Raffaella, I.; Cardenia, V.; Rodriguez-Estrada, M. Analysis of phytosterols and phytostanols in enriched dairy products by Fast gas chromatography with mass spectrometry. J. Sep. Sci. 2014, 37, 291–2919. [Google Scholar] [CrossRef]
- Menéndez-Carreño, M.; Knol, D.; Janssen, H.G. Development and validation of methodologies for the quantification of phytosterols and phytosterol oxidation products in cooked and baked food products. J. Chromatogr. A 2016, 1428, 316–325. [Google Scholar] [CrossRef] [PubMed]
| Sample Availability: Not available. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).