Binding Performance of Human Intravenous Immunoglobulin and 20(S)-7-Ethylcamptothecin
Abstract
1. Introduction
2. Results and Discussion
2.1. Fluorescence Quenching Study
2.2. Binding Mode Study
2.3. FT-IR Spectroscopy Study
2.4. UV-Vis Spectroscopy Study
2.5. Molecular Docking Study
3. Materials and Methods
3.1. Materials
3.2. Methods
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Herben, V.M.M.; Beijnen, J.H.; Ten Bokkel Huinink, W.W.; Schellens, J.H.M. Clinical pharmacokinetics of camptothecin topoisomerase I inhibitors. Pharm. World Sci. 1998, 20, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Jaxel, C.; Kohn, K.W.; Wani, M.C.; Wall, M.E.; Pommier, Y. Structure-activity study of the actions of camptothecin derivatives on mammalian topoisomerase I. Evidence of a specific receptor site and for a relation to antitumor activity. Cancer Res. 1989, 49, 1465–1469. [Google Scholar] [PubMed]
- Underberg, W.J.M.; Goossen, R.M.J.; Smith, B.R. Equilibrium kinetics of the new experimental anti-tumor compound SK&F 104864-A in aqueous solution. J. Pharm. Biomed. Anal. 1990, 8, 681–683. [Google Scholar] [CrossRef] [PubMed]
- Fassberg, J.; Stella, V.J. A kinetic and mechanistic study of the hydrolysis of camptothecin and some analogues. J. Pharm. Sci. 1992, 81, 676–684. [Google Scholar] [CrossRef] [PubMed]
- Mi, Z.; Burke, T.G. Marked interspecies variations concerning the interactions of camptothecin with serum albumins: A frequency-domain fluorescence spectroscopic study. Biochemistry 1994, 33, 12540–12545. [Google Scholar] [CrossRef] [PubMed]
- Burke, T.G.; Mi, Z. The structural basis of camptothecin interactions with human serum albumin: Impact on drug stability. J. Med. Chem. 1994, 37, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Burke, T.G.; Munshi, C.B.; Mi, Z.; Jiang, Y. The Important Role of Albumin in Determining the Relative Human Blood Stabilities of the Camptothecin Anticancer Drugs. J. Pharm. Sci. 1995, 84, 518–519. [Google Scholar] [CrossRef] [PubMed]
- Zeshaan, A.R.; Eric, H.R. Mechanisms of resistance to topoisomerase I-targeting drugs. Oncogene 2003, 22, 7296–7304. [Google Scholar] [CrossRef]
- Allikmets, R.; Schriml, L.M.; Hutchinson, A.; Romano-Spica, V.; Dean, M. A Human Placenta-specific ATP-Binding Cassette Gene (ABCP) on Chromosome 4q22 That Is Involved in Multidrug Resistance. Cancer Res. 1998, 58, 5337–5339. [Google Scholar] [PubMed]
- Maliepaard, M.; van Gastelen, M.A.; de Jong, L.A.; Pluim, D.; van Waardenburg, R.C.; Ruevekamp-Helmers, M.C.; Floot, B.G.; Schellens, J.H. Overexpression of the BCRP/MXR/ABCP Gene in a Topotecan-selected Ovarian Tumor Cell Line. Cancer Res. 1999, 59, 4559–4563. [Google Scholar] [PubMed]
- Diestra, J.E.; Scheffer, G.L.; Catala, I.; Maliepaard, M.; Schellens, J.H.; Scheper, R.J.; Germa-Lluch, J.R.; Izquierdo, M.A. Frequent expression of the multi-drug resistance-associated protein BCRP/MXR/ABCP/ABCG2 in human tumours detected by the BXP-21 monoclonal antibody in paraffin-embedded material. J. Pathol. 2002, 198, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Özvegy, C.; Litman, T.; Szakács, G.; Nagy, Z.; Bátes, S.; Váradi, A.; Sarkadi, B. Functional Characterization of the Human Multidrug Transporter, ABCG2, Expressed in Insect Cells. Biochem. Biophys. Res. Commun. 2001, 285, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Litman, T.; Druley, T.E.; Stein, W.D.; Ates, S.E. From MDR to MXR: New understanding of multidrug resistance systems, their properties and clinical significance. Cell. Mol. Life Sci. 2001, 58, 931–959. [Google Scholar] [CrossRef] [PubMed]
- Volk, E.L.; Farley, K.M.; Wu, Y.; Li, F.; Robey, R.W.; Schneider, E. Overexpression of Wild-Type Breast Cancer Resistance Protein Mediates Methotrexate Resistance. Cancer Res. 2002, 62, 5035–5040. [Google Scholar] [PubMed]
- Zhou, S.; Schuetz, J.D.; Bunting, K.D.; Colapietro, A.M.; Sampath, J.; Morris, J.J.; Lagutina, I.; Grosveld, G.C.; Osawa, M.; Nakauchi, H.; et al. The ABC transporter Bcrp1/ABCG2 is expressed in a wide variety of stem cells and is a molecular determinant of the side-population phenotype. Nat. Med. 2001, 7, 1028–1034. [Google Scholar] [CrossRef] [PubMed]
- Abbott, B.L.; Colapietro, A.M.; Barnes, Y.; Marini, F.; Andreeff, M.; Sorrentino, B.P. Low levels of ABCG2 expression in adult AML blast samples. Blood 2002, 100, 4594–4601. [Google Scholar] [CrossRef] [PubMed]
- Reid, R.J.; Kauh, E.A.; Bjornsti, M.A. Camptothecin sensitivity is mediated by the pleiotropic drug resistance network in yeast. J. Biol. Chem. 1997, 272, 12091–12099. [Google Scholar] [CrossRef] [PubMed]
- Özvegy-Laczka, C.; Várady, G.; Köblös, G.; Ujhelly, O.; Cervenak, J.; Schuetz, J.D.; Sorrentino, B.P.; Koomen, G.J.; Váradi, A.; Német, K.; et al. Membrane Transport, Structure, Function, and Biogenesis. Function-dependent Conformational Changes of the ABCG2 Multidrug Transporter Modify Its Interaction with a Monoclonal Antibody on the Cell Surface. J. Biol. Chem. 2005, 280, 4219–4227. [Google Scholar] [CrossRef] [PubMed]
- Mechetner, E.B.; Roninson, I.B. Efficient inhibition of P-glycoprotein-mediated multidrug resistance with a monoclonal antibody. Proc. Natl. Acad. Sci. USA 1992, 89, 5824–5828. [Google Scholar] [CrossRef] [PubMed]
- Hamada, H.; Tsuruo, T. Functional role for the 170- to 180-kDa glycoprotein specific to drug-resistant tumor cells as revealed by monoclonal antibodies. Proc. Natl. Acad. Sci. USA 1986, 83, 7785–7789. [Google Scholar] [CrossRef] [PubMed]
- Mechetner, E.B.; Schott, B.; Morse, B.S.; Stein, W.D.; Druley, T.; Davis, K.A.; Tsuruo, T.; Roninson, I.B. P-glycoprotein function involves conformational transitions detectable by differential immunoreactivity. Proc. Natl. Acad. Sci. USA 1997, 94, 12908–12913. [Google Scholar] [CrossRef] [PubMed]
- Nagy, H.; Goda, K.; Arceci, R.; Cianfriglia, M.; Mechetner, E.; Szabo, G., Jr. P-Glycoprotein conformational changes detected by antibody competition. Eur. J. Biochem. 2001, 268, 2416–2420. [Google Scholar] [CrossRef] [PubMed]
- Ejendal, K.F.K.; Diop, N.K.; Schweiger, L.C.; Hrycyna, C.A. The nature of amino acid 482 of human ABCG2 affects substrate transport and ATP hydrolysis but not substrate binding. Protein Sci. 2006, 15, 1597–1607. [Google Scholar] [CrossRef] [PubMed]
- Urquhart, B.; Ware, J.; Tirona, R.G.; Ho, R.; Leake, B.; Schwarz, U.I.; Zaher, H.; Palandra, J.; Gregor, J.; Dresser, G.K.; et al. Breast cancer resistance protein (ABCG2) and drug disposition: Intestinal expression, polymorphisms and sulfasalazine as an in vivo probe. Pharmacogenet. Genom. 2008, 18, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Shen, M.C. Pharmacological and immunological mechanisms of third generation intravenous immunoglobulin. Chin. J. Blood Transfus. 2001, 14, 325–327. [Google Scholar] [CrossRef]
- Lebing, W.; Remington, K.M.; Schreiner, C.; Paul, H.-I. Properties of a new intravenous immunoglobulin (IGIV-C, 10%) produced by virus inactivation with caprylate and column chromatography. Vox Sang. 2003, 84, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Xing, Y.T.; Li, C.S.; Hu, Y.; Li, T.J.; Zhou, Z.J.; Peng, Y.; Lin, L.Z.; Chen, K.J.; Deng, Z.J.; Li, J.; et al. Characterizations and Analysis of 10% IVIG Prepared by Chromatography. Chin. J. Blood Transfus. 2017, 30, 890–893. [Google Scholar] [CrossRef]
- Melamed, I.R.; Borte, M.; Trawnicek, L.; Kobayashi, A.L.; Kobayashi, R.H.; Knutsen, A.; Gupta, S.; Smits, W.; Pituch-Noworolska, A.; Strach, M.; et al. Pharmacokinetics of a novel human intravenous immunoglobulin 10% in patients with primary immunodeficiency diseases: Analysis of a phase III, multicentre, prospective, open-label study. Eur. J. Pharm. Sci. 2018, 118, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Bayry, J.; Misra, N.; Latry, V.; Prost, F.; Delignat, S.; Lacroix-Desmazes, S.; Kazatchkine, M.D.; Kaveri, S.V. Mechanisms of action of intravenous immunoglobulins in autoimmune and inflammatory diseases. Transfusion Clinique et Biologique 2003, 10, 165–169. [Google Scholar] [CrossRef]
- Shoenfeld, Y.; Fishman, P. Gamma-globulin inhibits tumor spread in mice. Int. Immunol. 1999, 11, 247–252. [Google Scholar] [CrossRef]
- Shoenfeld, Y.; Levy, Y.; Fishman, P. Shrinkage of melanoma metastases following high dose intravenous immunoglobulin treatment. Isr. Med. Assoc. J. 2001, 3, 698–699. [Google Scholar] [PubMed]
- Phuphanich, S.; Brock, C. Neurologic improvement after high-dose intravenous immunoglobulin therapy in patients with paraneoplastic cerebellar degeneration associated with anti-Purkinje cell antibody. J. Neurooncol. 2007, 81, 67–69. [Google Scholar] [CrossRef] [PubMed]
- Rowland, G.F.; Axton, C.A.; Baldwin, R.W.; Brown, J.P.; Corvalan, J.R.F.; Embleton, M.J.; Gore, V.A.; Hellström, I.; Hellström, K.E.; Jacobs, E.; et al. Antitumor properties of vindesine-monoclonal antibody conjugates. Cancer Immunol. Immun. 1985, 19, 1–7. [Google Scholar] [CrossRef]
- Yu, Z.Y.; Tang, Z.Y.; Ma, J.Y. Cytostatic effect of VCR-IgG conjugate to hepatoma. Tumor 1989, 9, 154–156. [Google Scholar]
- Szebeni, J.; Alving, C.R.; Savay, S.; Barenholz, Y.; Priev, A.; Danino, D.; Talmon, Y. Formation of complement-activating particles in aqueous solutions of Taxol: Possible role in hypersensitively reactions. Int. Immunopharmacl. 2001, 1, 721–735. [Google Scholar] [CrossRef]
- Lutz, H.U.; Stammler, P.; Jelezarova, E.; Nater, M.; Spath, P.J. High doses of immunoglobulin G attenuate immune aggregate-mediated complement activation by enhancing physiologic cleavage of C3b in C3bn-IgG complexes. Blood 1996, 88, 184–193. [Google Scholar] [PubMed]
- Lev, S.; Gilburd, B.; Lahat, N.; Shoenfeld, Y. Prevention of tumor spread by matrix metalloproteinase-9 inhibition: Old drugs, new concept. Eur. J. Int. Med. 2002, 13, 101–103. [Google Scholar] [CrossRef]
- Westemark, J.; Kahari, V.M. Regulation of matrix metalloproteinase expression in tumor invasion. FASEB J. 1999, 13, 781–792. [Google Scholar] [CrossRef]
- Strohl, W.R. Current progress in innovative engineered antibodies. Protein Cell 2018, 9, 86–120. [Google Scholar] [CrossRef] [PubMed]
- Strohl, W.R.; Strohl, L.M. Therapeutic Antibody Engineering: Current and Future Advances Driving the Strongest Growth Area in the Pharma Industry; Series in Biomedicine No. 11; Woodhead Publishing: Cambridge, UK, 2012; ISBN 9781907568374. [Google Scholar]
- Tsuchikama, K.; An, Z. Antibody-drug conjugates: Recent advances in conjugation and linker chemistries. Protein Cell 2018, 9, 33–46. [Google Scholar] [CrossRef] [PubMed]
- Beck, A.; Goetsch, L.; Dumontet, C.; Corvaïa, N. Strategies and challenges for the next generation of antibody–drug conjugates. Nat. Rev. Drug Discov. 2017, 16, 315–337. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.C.; Li, Y.J.; Yao, X.J.; Li, Y.Y.; Qi, H.L.; Zhang, K.J.; Lei, R.X.; Liu, J.N. Interaction of intravenous immunoglobulin and three 20(S)-camptothecin analogues: Maintaining higher circulatory levels of the biologically active species. Med. Chem. Res. 2017, 26, 3286–3295. [Google Scholar] [CrossRef]
- Liu, Y.C.; Wei, X.X.; Xu, X.Y.; Yao, X.J.; Lei, R.X.; Zheng, X.D.; Liu, J.N. Interaction of IVIG and camptothecin. In Medicine Sciences and Bioengineering; Wang, M., Ed.; Taylor & Francis Group: London, UK, 2015; pp. 551–557. ISBN 978-1-138-02684-1. [Google Scholar]
- Liu, Y.C.; Qi, H.L.; Wei, X.X.; Yao, X.J.; Li, Y.Y.; Zhang, K.J.; Liu, J.N. Interaction of intravenous immunoglobulin and irinotecan hydrochloride. Chem. Res. Appl. 2015, 27, 1841–1848. [Google Scholar]
- Yuan, T.; Weljie, A.M.; Vogel, H.J. Tryptophan Fluorescence Quenching by Methionine and Selenomethionine Residues of Calmodulin: Orientation of Peptide and Protein Binding. Biochemistry 1998, 37, 3187–3195. [Google Scholar] [CrossRef] [PubMed]
- Bagatolli, L.A.; Kivatinitz, S.C.; Fidelio, G.D. Interaction of Small Ligands with Human Serum Albumin IIIA Subdomain. How to Determine the Affinity Constant Using an Easy Steady State Fluorescence Method. J. Pharm. Sci. 1996, 85, 1131–1132. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.M.; Yang, P.; Zhang, L.W. Study on Interaction of Caffeic Acid Series Medicine and Albumin by Fluorescence Method. Chin. Sci. Bull. 1994, 9, 31–36. [Google Scholar]
- Yang, P.; Gao, F. Principles of Bioinorganic Chemistry, 1st ed.; Science Press: Beijing, China, 2002; pp. 487–495. ISBN 7-03-010276-2. [Google Scholar]
- Liu, Y.C.; He, W.Y.; Gao, W.H.; Hu, Z.D.; Chen, X.G. Binding of wogonin to human gammaglobulin. Int. J. Biol. Macromol. 2005, 37, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.C.; Lei, R.X.; Hu, Z.D.; Chen, X.G.; Shen, F.L.; Jing, J. Studies on the Binding of Barbaloin to Human Gamma Globulin. Spectrosc. Lett. 2006, 39, 265–284. [Google Scholar] [CrossRef]
- Hasemann, C.A.; Capra, J.D. Mutational analysis of arsonate binding by a CRIA + antibody. VH and VL junctional diversity are essential for binding activity. J. Biol. Chem. 1991, 266, 7626–7632. [Google Scholar] [PubMed]
- Amit, A.G.; Mariuzza, R.A.; Phillips, S.E.V.; Poljak, R.J. Three-Dimensional Structure of an Antigen-Antibody Complex at 2.8 angstrom Resolution. Science 1986, 233, 747–753. [Google Scholar] [CrossRef] [PubMed]
- Corper, A.L.; Sohi, M.K.; Bonagura, V.R.; Steinitz, M.; Jefferis, R.; Feinstein, A.; Beale, D.; Taussig, M.J.; Sutton, B.J. Structure of human IgM rheumatoid factor Fab bound to its autoantigen IgG Fc reveals a novel topology of antibody-antigen interaction. Nat. Struct. Biol. 1997, 4, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Ouameur, A.A.; Marty, R.; Tajmir-Riahi, H.A. Human Serum Albumin Complexes with Chlorophyll and Chlorophyllin. Biopolymers 2005, 77, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Timasheff, S.N. Thermodynamic of protein interactions. In Proteins of Biological Fluids; Peeters, H., Ed.; Pergamon Press: Oxford, UK, 1972; pp. 511–519. ISBN 978-0-08-018233-9. [Google Scholar]
- Yang, S.Y.; Tsuboi, M. Polarizing microscopy of eyespot of Chlamydomonas: In situobservation of its location, orientation, and multiplication. Biospectros 1999, 5, 93–100. [Google Scholar] [CrossRef]
- Rahmelow, K.; Hubner, W. Secondary structure determination of proteins in aqueous solution by infrared spectroscopy. A comparison of multivariate data analysis. Anal. Biochem. 1996, 241, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.C.; Yang, Z.Y.; Du, J.; Yao, X.J.; Lei, R.X.; Zheng, X.D.; Liu, J.N.; Hu, H.S.; Li, H. Interaction of curcumin with intravenous immunoglobulin: A fluorescence quenching and Fourier transformation infrared spectroscopy study. Immunobiology 2008, 213, 651–661. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.C.; Yang, Z.Y.; Du, J.; Yao, X.J.; Lei, R.X.; Zheng, X.D.; Liu, J.N.; Hu, H.S.; Li, H. Study on the Interactions of Kaempferol and Quercetin with Intravenous Immunoglobulin by Fluorescence Quenching, Fourier Transformation Infrared Spectroscopy and Circular Dichroism Spectroscopy. Chem. Pharm. Bull. 2008, 56, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.Y.; Zhang, L.Y. Immunocytochemistry, 1st ed.; Shanghai Medical University Press: Shanghai, China, 1996; pp. 487–495. ISBN 7-5627-0299-3/R·282. [Google Scholar]
- Dong, A.C.; Huang, P.; Caughey, W.S. Protein secondary structure in water from second-derivative amide I infrared spectra. Biochemistry 1990, 29, 3303–3306. [Google Scholar] [CrossRef] [PubMed]
- Purcell, M.; Neault, J.F.; Tajumir-Riahi, H.A. Interaction of taxol with human serum albumin. Biochim. Biophys. Acta 2000, 1478, 61–68. [Google Scholar] [CrossRef]
- Maestro, version 9.0; Schrödinger, LLC: New York, NY, USA, 2009.
- LigPrep, version 2.3; Schrödinger, LLC: New York, NY, USA, 2009.
- Kaminski, G.A.; Friesner, R.A.; Tirado-Rives, J.; Jorgensen, W.L. Evaluation and reparametrization of the OPLS-AA force field for proteins via comparison with accurate quantum chemical calculations on peptides. J. Phys. Chem. B 2001, 105, 6474–6487. [Google Scholar] [CrossRef]
- Epik, version 2.0; Schrödinger, LLC: New York, NY, USA, 2009.
- Wedemayer, G.J.; Patten, P.A.; Wang, L.H.; Schultz, P.G.; Stevens, R.C. Structural Insights into the Evolution of an Antibody Combining Site. Science 1997, 276, 1665–1669. [Google Scholar] [CrossRef] [PubMed]
- Glide, version 5.5; Schrödinger, LLC: New York, NY, USA, 2009.
- Ye, S.L.; Li, D.; Liu, F.J.; Lei, M.; Jiang, P.; Wang, Z.K.; Zhang, R.; Du, X.; Cao, H.J.; Ma, L.; et al. In vitro evaluation of the biological activities of IgG in seven Chinese intravenous immunoglobulin preparations. J. Pharm. Biomed. Anal. 2018, 151, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Diep, C.; Shih, A.W.; Jamula, E.; Heddle, N.M.; Parvizian, M.; Hillis, C.M. Impact of organizational interventions on reducing inappropriate intravenous immunoglobulin (IVIG) usage: A systematic review and meta-analysis. Transfus. Apher. Sci. 2018, 57, 215–221. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
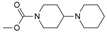 , R3 = H, R4 = CH2CH3; 10-Hydroxycamptothecin: R1 = H, R2 = OH, R3 = H, R4 = H; 7-Ethylcamptothecin: R1 = H, R2 = H, R3 = H, R4 = CH2CH3; SN-38: R1 = H, R2 = OH, R3 = H, R4 = CH2CH3.
, R3 = H, R4 = CH2CH3; 10-Hydroxycamptothecin: R1 = H, R2 = OH, R3 = H, R4 = H; 7-Ethylcamptothecin: R1 = H, R2 = H, R3 = H, R4 = CH2CH3; SN-38: R1 = H, R2 = OH, R3 = H, R4 = CH2CH3.
 , R3 = H, R4 = CH2CH3; 10-Hydroxycamptothecin: R1 = H, R2 = OH, R3 = H, R4 = H; 7-Ethylcamptothecin: R1 = H, R2 = H, R3 = H, R4 = CH2CH3; SN-38: R1 = H, R2 = OH, R3 = H, R4 = CH2CH3.
, R3 = H, R4 = CH2CH3; 10-Hydroxycamptothecin: R1 = H, R2 = OH, R3 = H, R4 = H; 7-Ethylcamptothecin: R1 = H, R2 = H, R3 = H, R4 = CH2CH3; SN-38: R1 = H, R2 = OH, R3 = H, R4 = CH2CH3.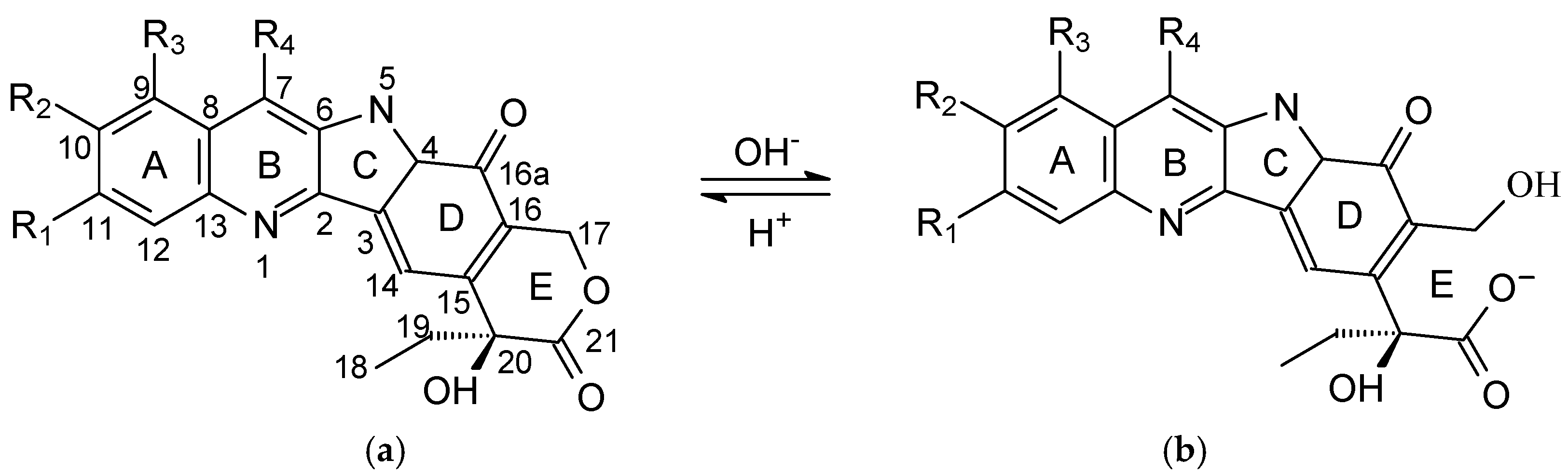
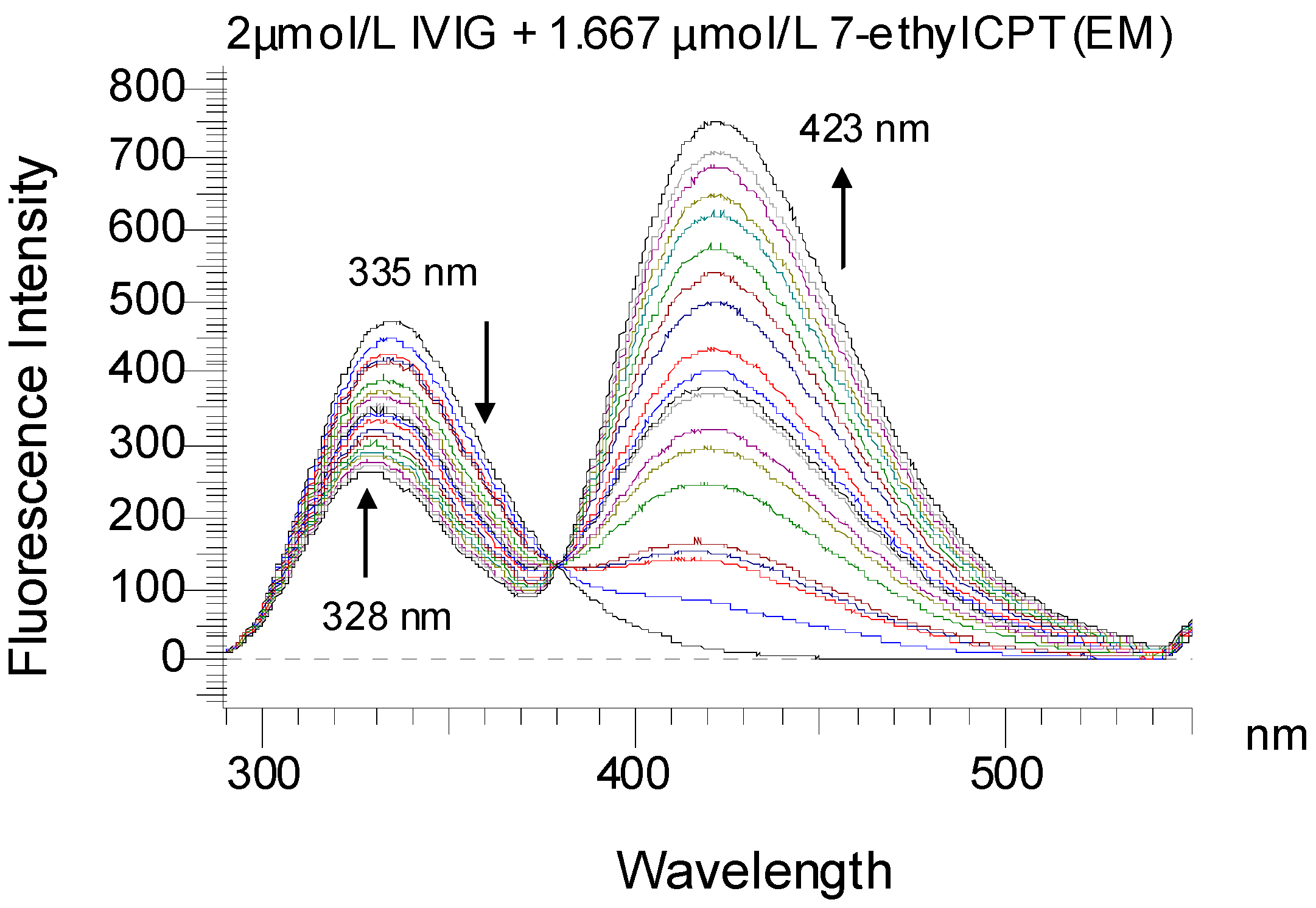
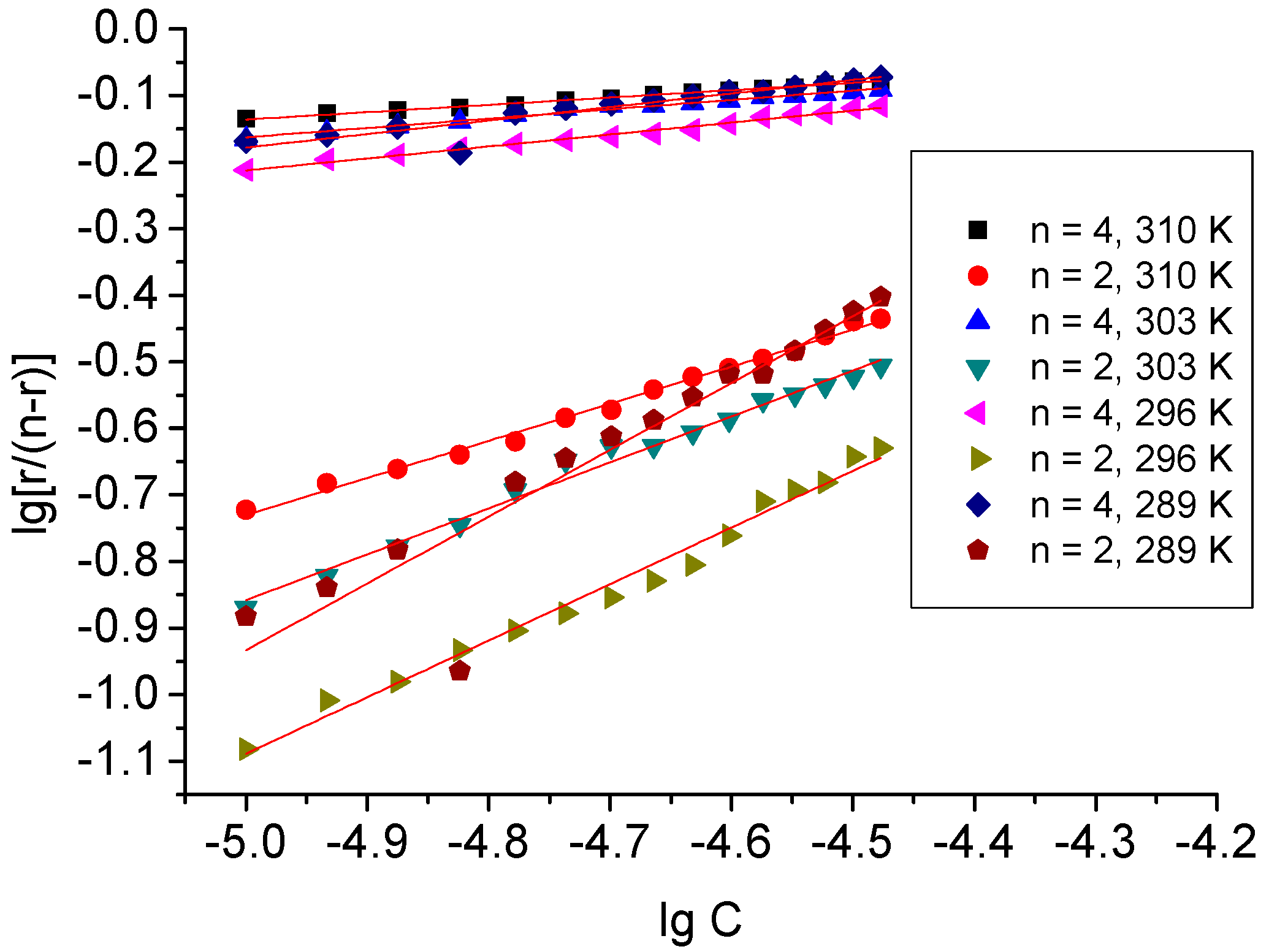
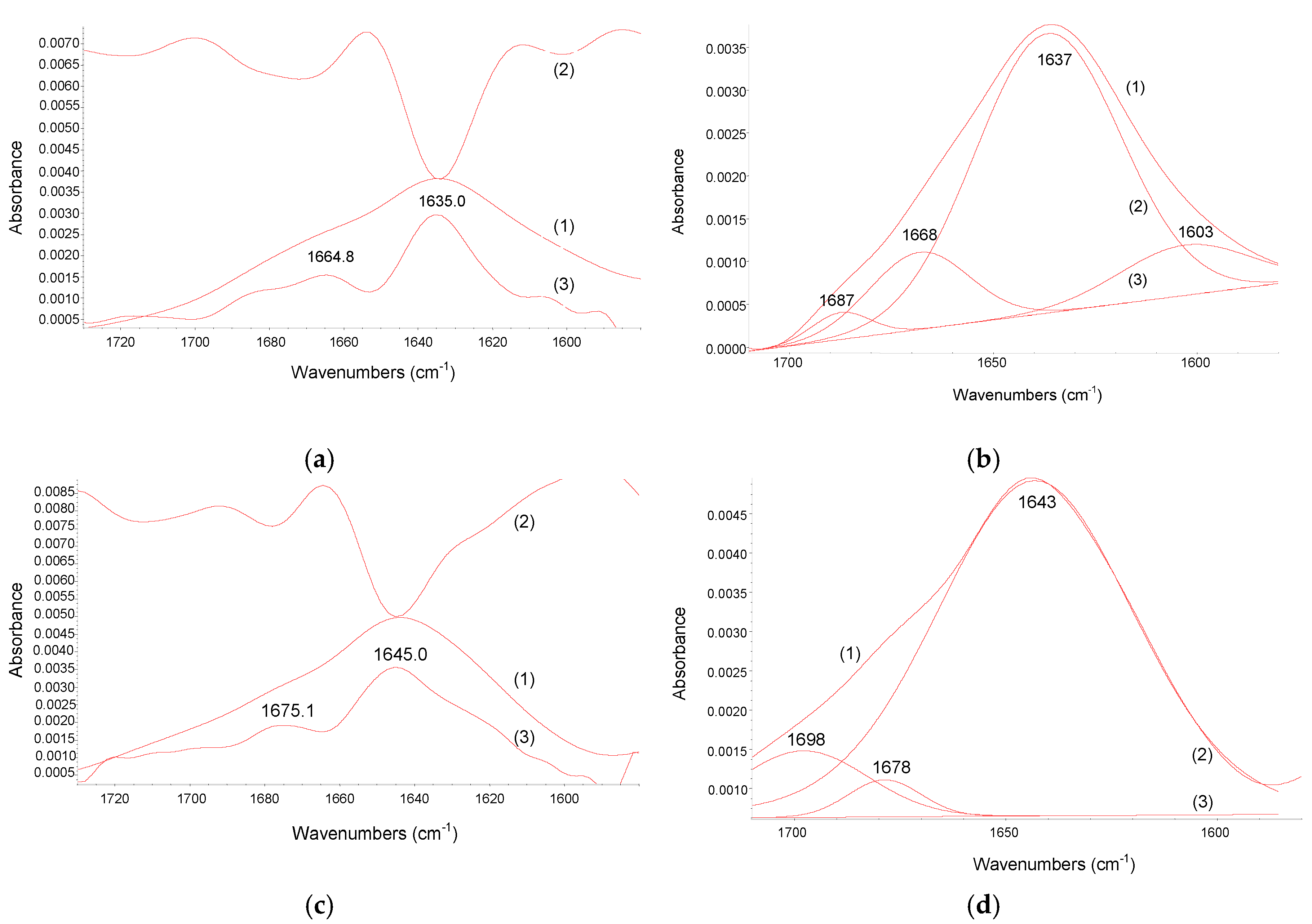
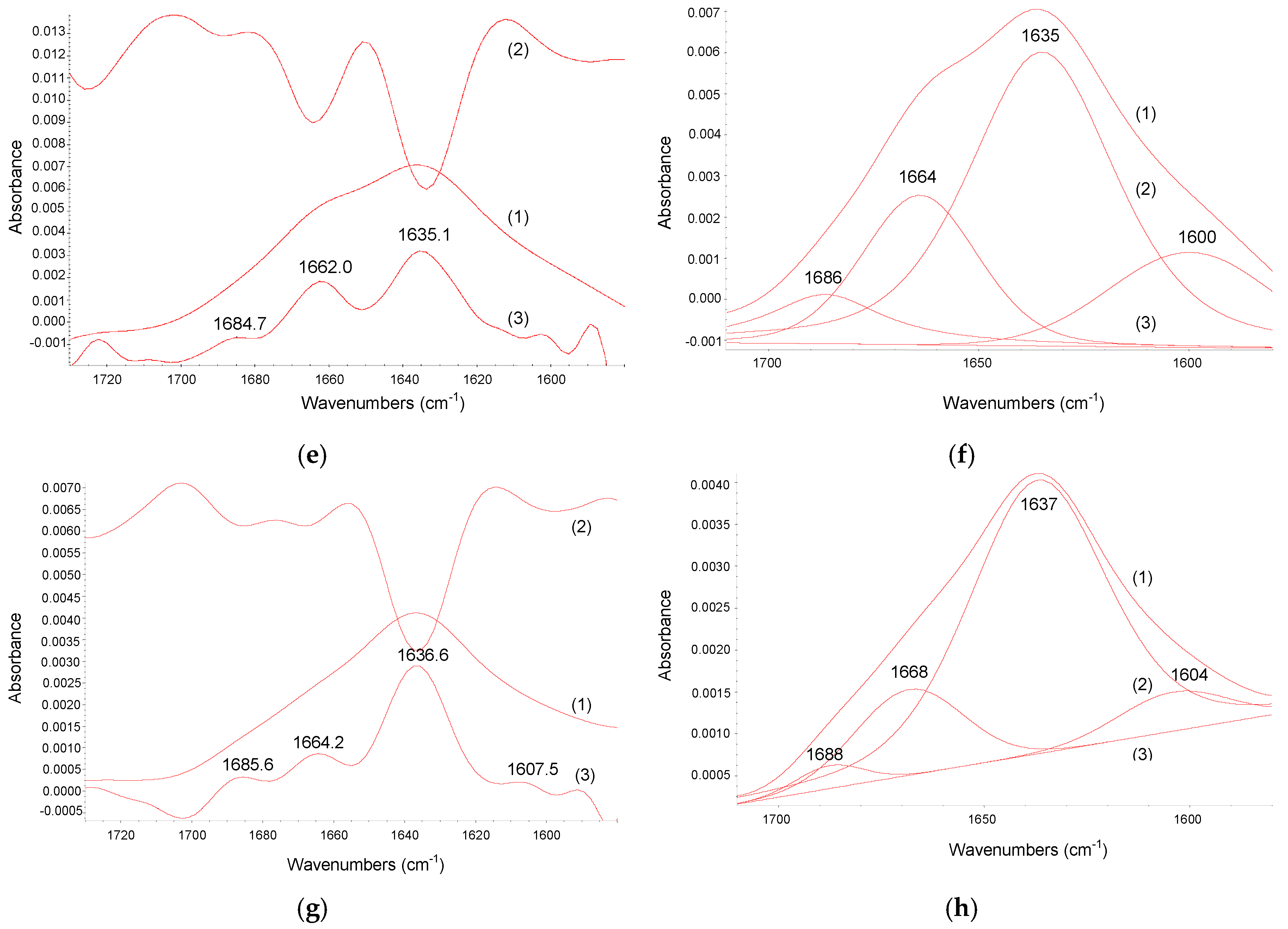
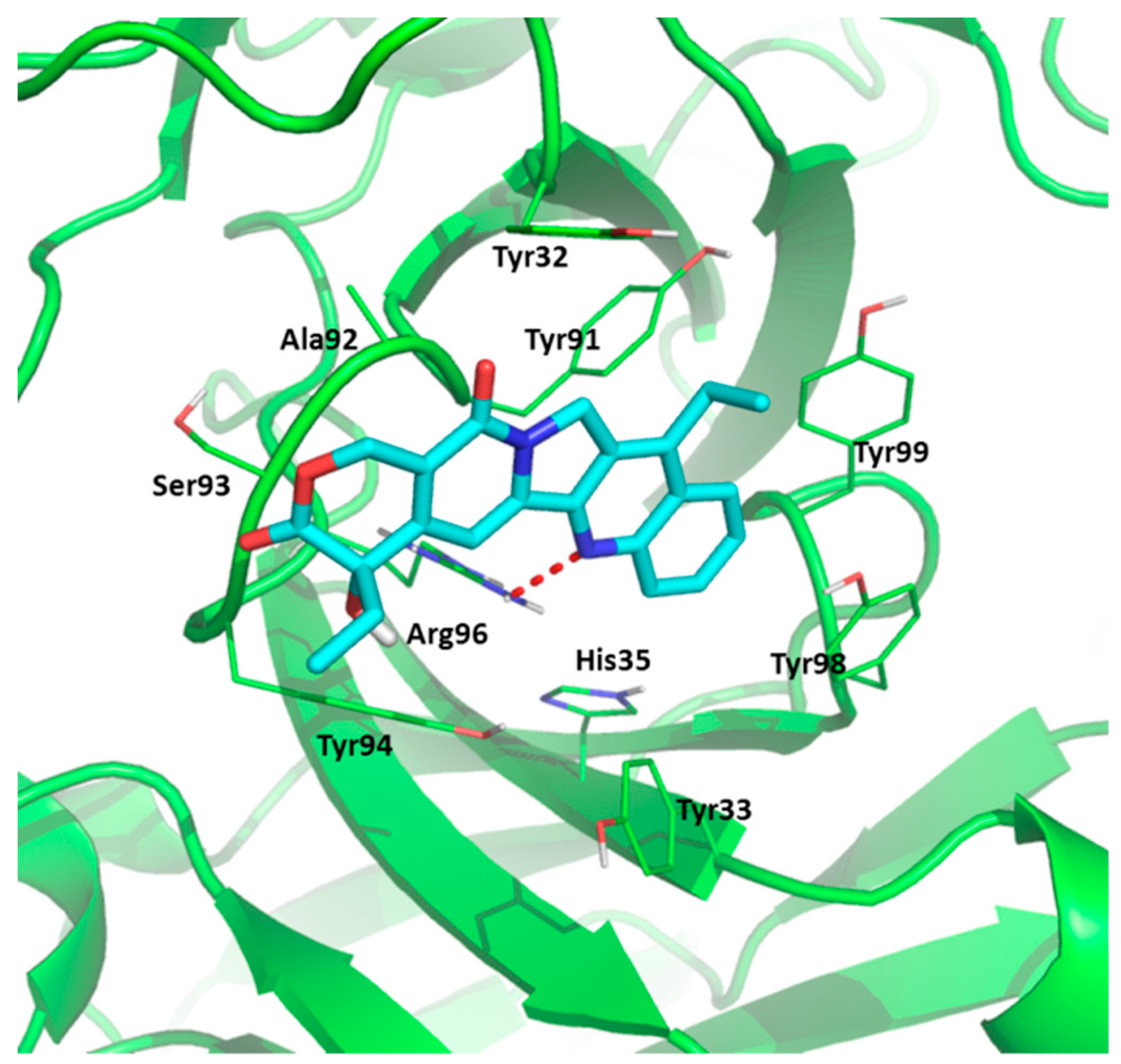
| Temperature K | Ko × 104/M−1 | α | Ko × 104/M−1 | α | KSV × 104/M−1 | Kq × 1012/M−1·s−1 |
|---|---|---|---|---|---|---|
| n = 2 | n = 4 | n = 2, n = 4 | ||||
| CPT | ||||||
| 289 | 0.2636 | 0.4529 | 0.04152 | 0.4391 | 4.988 | 4.988 |
| 296 | 0.8933 | 0.5784 | 0.1968 | 0.5544 | 6.737 | 6.737 |
| 303 | 4.135 | 0.9097 | 1.645 | 0.8830 | 11.74 | 11.74 |
| 310 | 9.683 | 1.373 | 5.341 | 1.343 | 14.67 | 14.67 |
| Topotecan chloride | ||||||
| 289 | 1.452 | 1.158 | 0.6501 | 1.099 | 4.743 | 4.743 |
| 296 | 1.528 | 1.101 | 0.6324 | 1.032 | 3.930 | 3.930 |
| 303 | 0.9268 | 0.8889 | 0.3119 | 0.8311 | 3.499 | 3.499 |
| 310 | 0.8570 | 0.7064 | 0.2028 | 0.6483 | 3.479 | 3.479 |
| Irinotecan chloride | ||||||
| 289 | 1.500 | 1.061 | 1.790 | 0.2290 | 4.514 | 4.514 |
| 296 | 1.122 | 0.8402 | 1.371 | 0.1779 | 4.497 | 4.497 |
| 303 | 1.052 | 0.8245 | 1.306 | 0.1752 | 3.623 | 3.623 |
| 310 | 0.9247 | 0.8708 | 1.167 | 0.1873 | 3.376 | 3.376 |
| 10-Hydroxycamptothecin | ||||||
| 289 | 0.9805 | 0.9365 | 0.4065 | 1.118 | 3.155 | 3.155 |
| 296 | 0.8630 | 1.157 | 0.3477 | 0.8823 | 2.410 | 2.410 |
| 303 | 0.8587 | 0.9614 | 0.3347 | 0.9164 | 2.134 | 2.134 |
| 310 | 0.6337 | 0.8893 | 0.2304 | 0.8485 | 1.979 | 1.979 |
| 7-Ethylcamptothecin | ||||||
| 289 | 1.301 | 1.351 | 0.6763 | 1.300 | 2.744 | 2.744 |
| 296 | 0.6293 | 1.010 | 0.2702 | 0.9769 | 1.888 | 1.888 |
| 303 | 0.8375 | 1.087 | 0.3764 | 1.048 | 1.768 | 1.768 |
| 310 | 0.8472 | 1.161 | 0.4072 | 1.125 | 1.552 | 1.552 |
| SN-38 | ||||||
| 289 | 1.183 | 1.185 | 0.7992 | 1.284 | 1.923 | 1.923 |
| 296 | 1.088 | 1.308 | 0.5671 | 1.269 | 1.907 | 1.907 |
| 303 | 0.4756 | 0.8135 | 0.1633 | 0.7832 | 1.308 | 1.308 |
| 310 | 0.2731 | 0.8007 | 0.0973 | 0.7793 | 0.8997 | 0.8997 |
| Temperature K | ΔGo/KJ·mol−1 | ΔHo/KJ·mol−1 | ΔSo/J·mol−1·K−1 | ΔGo/KJ·mol−1 | ΔHo/KJ·mol−1 | ΔSo/J·mol−1·K−1 |
|---|---|---|---|---|---|---|
| n = 2 | n = 4 | |||||
| CPT, binding mode: hydrophobic interaction | ||||||
| 289 | −13.39 | 131.5 | 501.4 | −8.953 | 177.8 | 645.9 |
| 296 | −16.72 | −13.00 | ||||
| 303 | −20.98 | −18.66 | ||||
| 310 | −23.66 | −22.12 | ||||
| Topotecan, binding mode: Van der Waals force, hydrogen bonding | ||||||
| 289 | −23.03 | −31.68 | −27.49 | −21.10 | −44.49 | −79.66 |
| 296 | −23.71 | −21.54 | ||||
| 303 | −23.01 | −20.27 | ||||
| 310 | −23.34 | −19.63 | ||||
| Irinotecan, binding mode: electrostatic interaction | ||||||
| 289 | −23.12 | −16.21 | 23.43 | −23.54 | −14.26 | 31.70 |
| 296 | −22.96 | −23.45 | ||||
| 303 | −23.34 | −23.89 | ||||
| 310 | −23.55 | −24.15 | ||||
| 10-hydroxycamptothecin, binding mode: electrostatic interaction | ||||||
| 289 | −22.08 | −13.90 | 28.53 | −19.97 | −18.42 | 5.646 |
| 296 | −22.30 | −20.07 | ||||
| 303 | −22.82 | −20.44 | ||||
| 310 | −22.56 | −19.95 | ||||
| 7-ethylcamptothecin, binding mode: hydrophobic interaction | ||||||
| 289 | −22.76 | 16.31 | 128.2 | −21.19 | 22.45 | 141.9 |
| 296 | −21.53 | −19.45 | ||||
| 303 | −22.76 | −20.74 | ||||
| 310 | −23.31 | −21.42 | ||||
| SN-38, binding mode: Van der Waals force, hydrogen bonding | ||||||
| 289 | −22.53 | −55.35 | −112.1 | −21.59 | −80.35 | −202.1 |
| 296 | −22.87 | −21.27 | ||||
| 303 | −21.33 | −18.64 | ||||
| 310 | −20.39 | −17.73 | ||||
| Amide I components (%) | pH 4.0 | pH 7.4 | ||||
|---|---|---|---|---|---|---|
| IVIG | +4.0 µM Analogues | +8.0 µM Analogues | IVIG | +4.0 µM Analogues | +8.0 µM Analogues | |
| CPT | ||||||
| β-anti | 5.10 | 2.79 | 10.24 | 3.61 | 6.16 | 4.85 |
| Turn | 8.28 | 13.72 | 3.46 | 24.88 | 3.68 | 10.46 |
| β-sheet | 40.04 | 11.09 | little | 39.49 | 23.73 | little |
| Remainders | 46.58 | 72.40 | ~86.30 | 32.04 | 66.43 | ~84.69 |
| Topotecan | ||||||
| β-anti | 5.10 | little | 8.93 | 3.61 | 4.45 | little |
| Turn | 8.28 | ~32.55 | 32.66 | 24.88 | 31.03 | ~18.28 |
| β-sheet | 40.04 | 15.72 | 3.22 | 39.49 | 24.26 | 27.73 |
| Remainders | 46.58 | 51.73 | 55.19 | 32.04 | 40.26 | 53.99 |
| Irinotecan | ||||||
| β-anti | 5.10 | little | little | 3.61 | 6.16 | little |
| Turn | 8.28 | ~4.97 | ~6.78 | 24.88 | 29.19 | ~14.81 |
| β-sheet | 40.04 | little | little | 39.49 | 30.50 | little |
| Remainders | 46.58 | ~95.03 | ~93.22 | 32.04 | 40.31 | ~85.19 |
| 10-Hydroxycamptothecin | ||||||
| β-anti | 5.10 | 10.24 | 6.17 | 3.61 | little | little |
| Turn | 8.28 | 3.46 | little | 24.88 | ~16.00 | ~33.71 |
| β-sheet | 40.04 | little | 33.36 | 39.49 | little | little |
| Remainders | 46.58 | ~86.31 | ~60.47 | 32.04 | ~84.00 | ~66.29 |
| 7-Ethylcamptothecin | ||||||
| β-anti | 5.10 | little | little | 3.61 | 7.68 | 2.76 |
| Turn | 8.28 | 1.36 | ~16.93 | 24.88 | 21.28 | 16.64 |
| β-sheet | 40.04 | 29.98 | 26.30 | 39.49 | 15.82 | 7.57 |
| Remainders | 46.58 | ~68.66 | 56.77 | 32.04 | 55.22 | 73.03 |
| SN-38 | ||||||
| β-anti | 5.10 | 6.84 | little | 3.61 | 11.79 | little |
| Turn | 8.28 | 35.40 | ~30.23 | 24.88 | 5.19 | ~24.50 |
| β-sheet | 40.04 | 1.51 | little | 39.49 | 15.37 | 32.25 |
| Remainders | 46.58 | 56.25 | ~69.77 | 32.04 | 67.64 | 43.25 |
| Compounds | Ratios of Lactone to Total for Three CPTs at pH 7.40 and 310 K | |
|---|---|---|
| SN-38 | 90.40% | 86.47~87.35% |
| 10-Hydroxycamptothecin | 86.68% | 56.56~68.58% |
| Topotecan | 34.47% | 29.76~32.71% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.-C.; Li, Y.-Y.; Yao, X.-J.; Qi, H.-L.; Wei, X.-X.; Liu, J.-N. Binding Performance of Human Intravenous Immunoglobulin and 20(S)-7-Ethylcamptothecin. Molecules 2018, 23, 2389. https://doi.org/10.3390/molecules23092389
Liu Y-C, Li Y-Y, Yao X-J, Qi H-L, Wei X-X, Liu J-N. Binding Performance of Human Intravenous Immunoglobulin and 20(S)-7-Ethylcamptothecin. Molecules. 2018; 23(9):2389. https://doi.org/10.3390/molecules23092389
Chicago/Turabian StyleLiu, Yong-Chun, Ying-Ying Li, Xiao-Jun Yao, Hui-Li Qi, Xiao-Xia Wei, and Jian-Ning Liu. 2018. "Binding Performance of Human Intravenous Immunoglobulin and 20(S)-7-Ethylcamptothecin" Molecules 23, no. 9: 2389. https://doi.org/10.3390/molecules23092389
APA StyleLiu, Y.-C., Li, Y.-Y., Yao, X.-J., Qi, H.-L., Wei, X.-X., & Liu, J.-N. (2018). Binding Performance of Human Intravenous Immunoglobulin and 20(S)-7-Ethylcamptothecin. Molecules, 23(9), 2389. https://doi.org/10.3390/molecules23092389




