Suppressive Effects of EGCG on Cervical Cancer
Abstract
1. Introduction
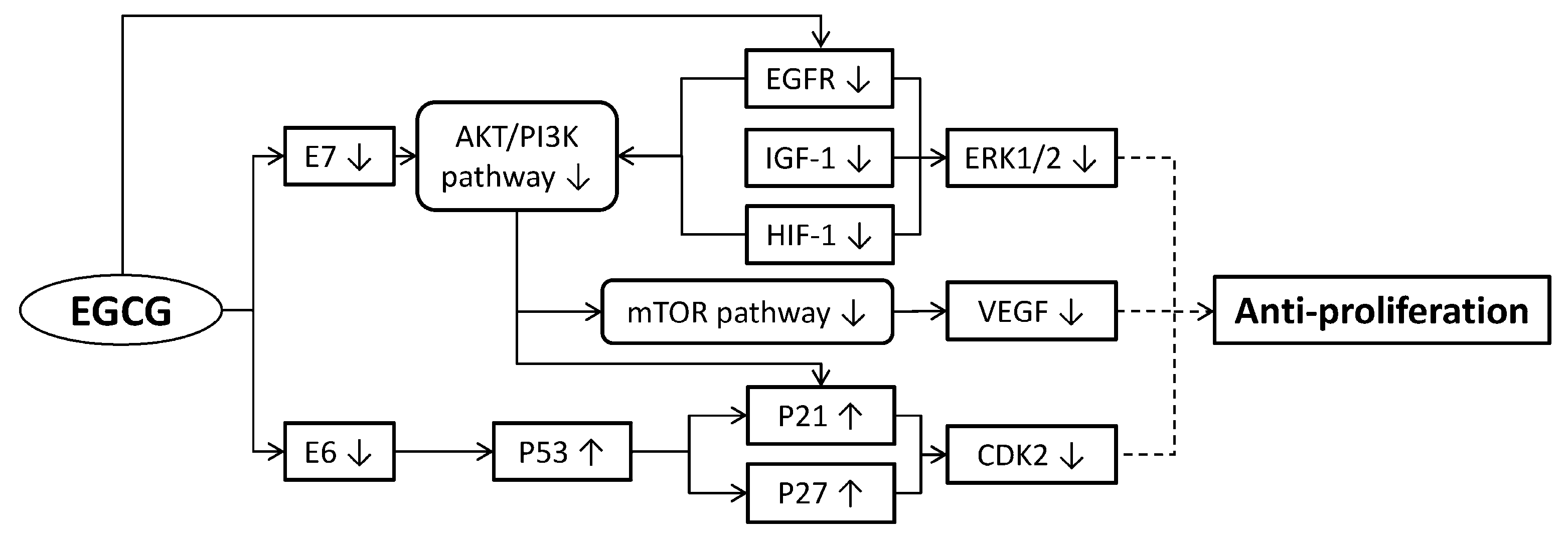
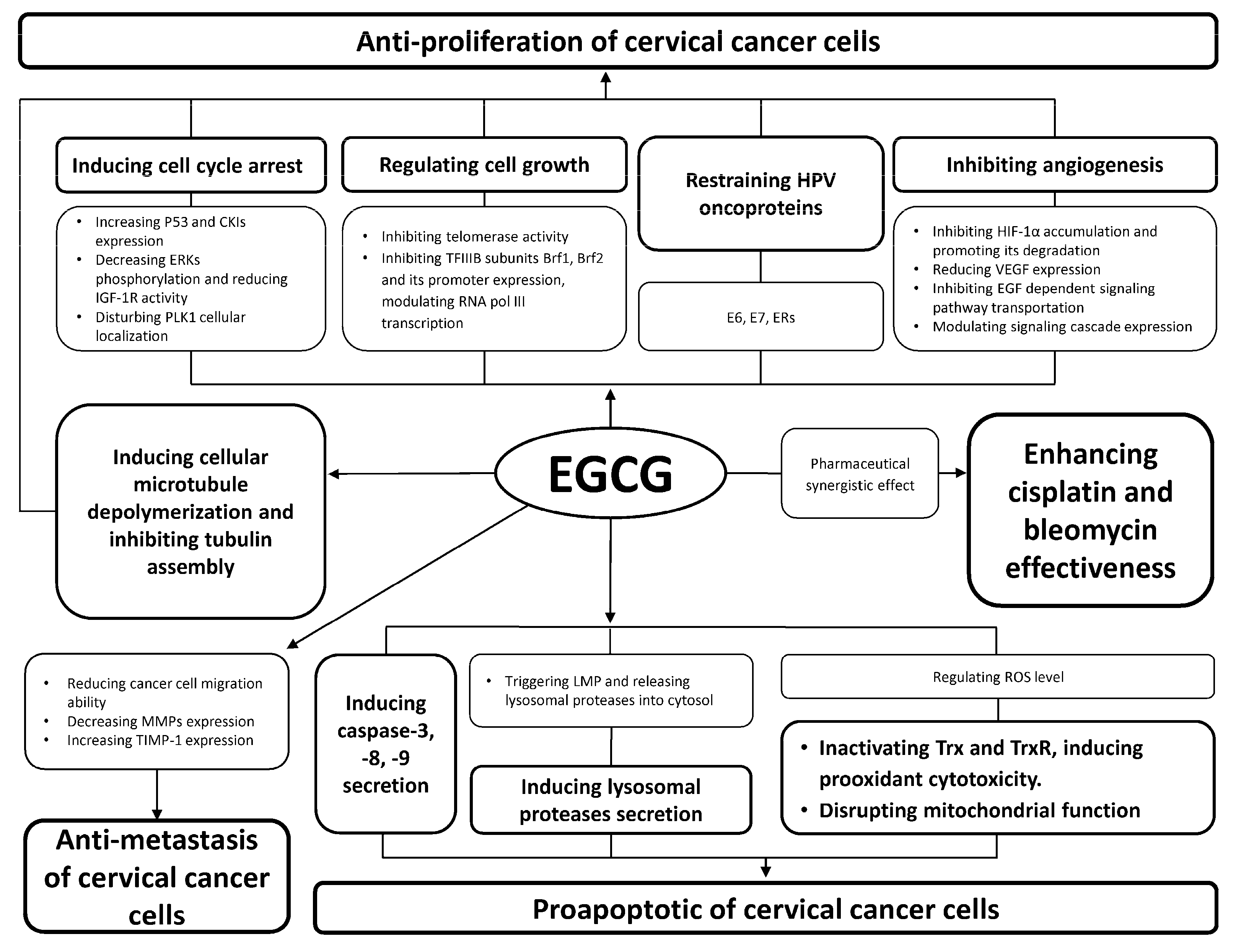
2. Anti-Proliferation of Cervical Cancer Cells
2.1. Inducing Cancer Cell Cycle Arrest
2.2. Regulating Cancer Cell Growth
2.3. Inducing Cellular Microtubule Depolymerization and Inhibiting Tubulin Assembly
2.4. Inhibiting Angiogenesis
2.5. Restraining Human Papillomavirus (HPV) Oncoproteins
3. Anti-Metastasis of Cervical Cancer Cells
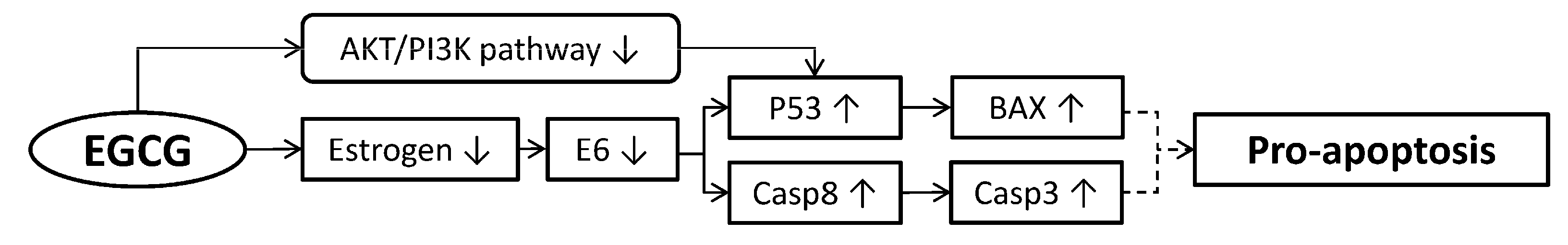
4. Pro-Apoptosis of Cervical Cancer Cells
4.1. Inducing Caspase Secretion
4.2. Reactive Oxygen Species (ROS) Induced Apoptosis of Cancer Cells
4.3. Inducing Lysosomal Proteases Secretion
5. Pharmaceutical Synergistic Effect
6. Ambiguous Potential Functions of (-)-Epigallocatechingallate (EGCG) on Cervical Cancer
7. Further Suggestions
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- World Health Organization. Globocan2012: Estimated Cancer Incidence, Mortality and Prevalence worldwide in 2012. Available online: http://globocan.iarc.fr/Pages/fact_sheets_cancer.aspx (accessed on 12 December 2013).
- Waggoner, S.E. Cervical cancer. Lancet 2003, 361, 2217–2225. [Google Scholar] [CrossRef]
- Moody, C.A.; Laimins, L.A. Human papillomavirus oncoproteins: Pathways to transformation. Nat. Rev. Cancer 2010, 10, 550–560. [Google Scholar] [CrossRef] [PubMed]
- De Sanjose, S.; Quint, W.G.V.; Alemany, L.; Geraets, D.T.; Klaustermeier, J.E.; Lloveras, B.; Tous, S.; Felix, A.; Bravo, L.E.; Shin, H.R.; et al. Human papillomavirus genotype attribution in invasive cervical cancer: A retrospective cross-sectional worldwide study. Lancet Oncol. 2010, 11, 1048–1056. [Google Scholar] [CrossRef]
- Doorbar, J.; Quint, W.; Banks, L.; Bravo, I.G.; Stoler, M.; Broker, T.R.; Stanley, M.A. The biology and life-cycle of human papillomaviruses. Vaccine 2012, 30, F55–F70. [Google Scholar] [CrossRef] [PubMed]
- Bernard, H.U.; Burk, R.D.; Chen, Z.G.; Van Doorslaer, K.; Zur Hausen, H.; Ze Villiers, E.M. Classification of papillomaviruses (pvs) based on 189 pv types and proposal of taxonomic amendments. Virology 2010, 401, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Hirchaud, F.; Hermetet, F.; Ablise, M.; Fauconnet, S.; Vuitton, D.A.; Pretet, J.L.; Mougin, C. Isoliquiritigenin induces caspase-dependent apoptosis via downregulation of hpv16 e6 expression in cervical cancer caski cells. Planta Med. 2013, 79, 1628–1635. [Google Scholar] [PubMed]
- Bosch, F.X.; Burchell, A.N.; Schiffman, M.; Giuliano, A.R.; De Sanjose, S.; Bruni, L.; Tortolero-Luna, G.; Kjaer, S.K.; Munoz, N. Epidemiology and natural history of human papillomavirus infections and type-specific implications in cervical neoplasia. Vaccine 2008, 26, K1–K16. [Google Scholar] [CrossRef] [PubMed]
- Melsheimer, P.; Vinokurova, S.; Wentzensen, N.; Bastert, G.; Doeberitz, M.V. DNA aneuploidy and integration of human papillomavirus type 16 e6/e7 oncogenes in intraepithelial neoplasia and invasive squamous cell carcinoma of the cervix uteri. Clin. Cancer Res. 2004, 10, 3059–3063. [Google Scholar] [CrossRef] [PubMed]
- Ziegert, C.; Wentzensen, N.; Vinokurova, S.; Kisseljov, F.; Einenkel, J.; Hoeckel, M.; Doeberitz, M.V. A comprehensive analysis of hpv integration loci in anogenital lesions combining transcript and genome-based amplification techniques. Oncogene 2003, 22, 3977–3984. [Google Scholar] [CrossRef] [PubMed]
- Wilting, S.M.; Steenbergen, R.D.M.; Tijssen, M.; Van Wieringen, W.N.; Helmerhorst, T.J.M.; Van Kemenade, F.J.; Bleeker, M.C.G.; Van de Wiel, M.A.; Carvalho, B.; Meijer, G.A.; et al. Chromosomal signatures of a subset of high-grade premalignant cervical lesions closely resemble invasive carcinomas. Cancer Res. 2009, 69, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Poljak, M. Prophylactic human papillomavirus vaccination and primary prevention of cervical cancer: Issues and challenges. Clin. Microbiol. Infect. 2012, 18, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Alhamlan, F.S.; Al-Zahrani, A.S.; Almatrrouk, S.A.; Al-Ahdal, M.N. Human papillomaviruses: The cervical cancer saga in developing countries. J. Infect. Dev. Ctries. 2017, 11, 819–825. [Google Scholar] [CrossRef]
- Yuan, C.H.; Filippova, M.; Tungteakkhun, S.S.; Duerksen-Hughes, P.J.; Krstenansky, J.L. Small molecule inhibitors of the hpv16-e6 interaction with caspase 8. Bioorg. Med. Chem. Lett. 2012, 22, 2125–2129. [Google Scholar] [CrossRef] [PubMed]
- Vici, P.; Pizzuti, L.; Mariani, L.; Zampa, G.; Santini, D.; Di Lauro, L.; Gamucci, T.; Natoli, C.; Marchetti, P.; Barba, M.; et al. Targeting immune response with therapeutic vaccines in premalignant lesions and cervical cancer: Hope or reality from clinical studies. Expert Rev. Vaccines 2016, 15, 1327–1336. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.R.; Ye, Q.; Jin, J.; Liang, H.; Lu, J.L.; Du, Y.Y.; Dong, J.J. Chemical and instrumental assessment of green tea sensory preference. Int. J. Food Prop. 2008, 11, 258–272. [Google Scholar] [CrossRef]
- Dong, J.J.; Ye, J.H.; Lu, J.L.; Zheng, X.Q.; Liang, Y.R. Isolation of antioxidant catechins from green tea and its decaffeination. Food Bioprod. Process. 2011, 89, 62–66. [Google Scholar] [CrossRef]
- Xiang, L.P.; Wang, A.; Ye, J.H.; Zheng, X.Q.; Polito, C.A.; Lu, J.L.; Li, Q.S.; Liang, Y.R. Suppressive effects of tea catechins on breast cancer. Nutrients 2016, 8, 458. [Google Scholar] [CrossRef] [PubMed]
- Hou, I.C.; Amarnani, S.; Chong, M.T.; Bishayee, A. Green tea and the risk of gastric cancer: Epidemiological evidence. World J. Gastroenterol. 2013, 19, 3713–3722. [Google Scholar] [CrossRef] [PubMed]
- Arts, I.C. A review of the epidemiological evidence on tea, flavonoids, and lung cancer. J. Nutr. 2008, 138, 1561S–1566S. [Google Scholar] [CrossRef] [PubMed]
- Adhami, V.M.; Malik, A.; Zaman, N.; Sarfaraz, S.; Siddiqui, I.A.; Syed, D.N.; Afaq, F.; Pasha, F.S.; Saleem, M.; Mukhtar, H. Combined inhibitory effects of green tea polyphenols and selective cyclooxygenase-2 inhibitors on the growth of human prostate cancer cells both in vitro and in vivo. Clin. Cancer Res. 2007, 13, 1611–1619. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.C.; Chan, W.K.; Lee, T.W.; Lam, W.H.; Wang, X.; Chan, T.H.; Wong, Y.C. Effect of a prodrug of the green tea polyphenol (-)-epigallocatechin-3-gallate on the growth of androgen-independent prostate cancer in vivo. Nutr. Cancer 2008, 60, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Li, Y.; Lin, Q.; Wang, Y.; Sun, H.; Wang, J.; Cui, G.; Cai, L.; Dong, X. Tea polyphenols induced apoptosis of breast cancer cells by suppressing the expression of survivin. Sci. Rep. 2014, 4, 4416. [Google Scholar] [CrossRef] [PubMed]
- Ni, C.X.; Gong, H.; Liu, Y.; Qi, Y.; Jiang, C.L.; Zhang, J.P. Green tea consumption and the risk of liver cancer: A meta-analysis. Nutr. Cancer 2017, 69, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Connors, S.K.; Chornokur, G.; Kumar, N.B. New insights into the mechanisms of green tea catechins in the chemoprevention of prostate cancer. Nutr. Cancer 2012, 64, 4–22. [Google Scholar] [CrossRef] [PubMed]
- Pandey, M.; Shukla, S.; Gupta, S. Promoter demethylation and chromatin remodeling by green tea polyphenols leads to re-expression of GSTP1 in human prostate cancer cells. Int. J. Cancer 2010, 126, 2520–2533. [Google Scholar] [CrossRef] [PubMed]
- Garcia, F.A.R.; Cornelison, T.; Nuno, T.; Greenspan, D.L.; Byron, J.W.; Hsu, C.H.; Alberts, D.S.; Chow, H.H.S. Results of a phase II randomized, double-blind, placebo-controlled trial of polyphenon E in women with persistent high-risk hpv infection and low-grade cervical intraepithelial neoplasia. Gynecol. Oncol. 2014, 132, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Small, W., Jr.; Bacon, M.A.; Bajaj, A.; Chuang, L.T.; Fisher, B.J.; Harkenrider, M.M.; Jhingran, A.; Kitchener, H.C.; Mileshkin, L.R.; Viswanathan, A.N.; et al. Cervical cancer: A global health crisis. Cancer 2017, 123, 2404–2412. [Google Scholar] [CrossRef] [PubMed]
- Zou, C.; Liu, H.; Feugang, J.M.; Hao, Z.; Chow, H.H.; Garcia, F. Green tea compound in chemoprevention of cervical cancer. Int. J. Gynecol. Cancer 2010, 20, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Singh, R.; Bhui, K.; Tyagi, S.; Mahmood, Z.; Shukla, Y. Tea polyphenols induce apoptosis through mitochondrial pathway and by inhibiting nuclear factor-kappaB and Akt activation in human cervical cancer cells. Oncol. Res. 2011, 19, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Ahn, W.S.; Huh, S.W.; Bae, S.M.; Lee, I.P.; Lee, J.M.; Namkoong, S.E.; Kim, C.K.; Sin, J.I. A major constituent of green tea, EGCG, inhibits the growth of a human cervical cancer cell line, caski cells, through apoptosis, G1 arrest, and regulation of gene expression. DNA Cell Biol. 2003, 22, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.F.; Wu, J.H.; Ling, M.T.; Zhao, L.; Zhao, K.N. The role of the PI3K/Akt/mTOR signalling pathway in human cancers induced by infection with human papillomaviruses. Mol. Cancer 2015, 14, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Sah, J.F.; Balasubramanian, S.; Eckert, R.L.; Rorke, E.A. Epigallocatechin-3-gallate inhibits epidermal growth factor receptor signaling pathway. Evidence for direct inhibition of ERK1/2 and Akt kinases. J. Biol. Chem. 2004, 279, 12755–12762. [Google Scholar] [CrossRef] [PubMed]
- Pollak, M.N.; Schernhammer, E.S.; Hankinson, S.E. Insulin-like growth factors and neoplasia. Nat. rev. Cancer 2004, 4, 505–518. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; He, Z.; Ermakova, S.; Zheng, D.; Tang, F.; Cho, Y.Y.; Zhu, F.; Ma, W.Y.; Sham, Y.; Rogozin, E.A.; et al. Direct inhibition of insulin-like growth factor-I receptor kinase activity by (-)-epigallocatechin-3-gallate regulates cell transformation. Cancer Epidemiol. Biomark. Prev. 2007, 16, 598–605. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarty, S.; Ganguli, A.; Das, A.; Nag, D.; Chakrabarti, G. Epigallocatechin-3-gallate shows anti-proliferative activity in hela cells targeting tubulin-microtubule equilibrium. Chem. Biol. Interact. 2015, 242, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Q.; Erikson, R.L. Polo-like kinase 1 in the life and death of cancer cells. Cell Cycle 2003, 2, 424–425. [Google Scholar] [CrossRef] [PubMed]
- Goh, K.C.; Wang, H.S.; Yu, N.F.; Zhou, Y.F.; Zheng, Y.; Lim, Z.Y.; Sangthongpitag, K.; Fang, L.J.; Du, M.; Wang, X.K.; et al. PLK1 as a potential drug target in cancer therapy. Drug Dev. Res. 2004, 62, 349–361. [Google Scholar] [CrossRef]
- Shan, H.M.; Shi, Y.; Quan, J. Identification of green tea catechins as potent inhibitors of the polo-box domain of polo-like kinase 1. ChemMedChem 2015, 10, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Blackburn, E.H. Telomerases. Annu. Rev. Biochem. 1992, 61, 113–129. [Google Scholar] [CrossRef] [PubMed]
- Shay, J.W. Role of telomeres and telomerase in aging and cancer. Cancer Discov. 2016, 6, 584–593. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, M.; Noguchi, M.; Nakao, Y.; Pater, A.; Iwasaka, T. The tea polyphenol, (-)-epigallocatechin gallate effects on growth, apoptosis, and telomerase activity in cervical cell lines. Gynecol. Oncol. 2004, 92, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, M.; Yokoyama, M.; Watanabe, S.; Uchiyama, M.; Nakao, Y.; Hara, K.; Iwasaka, T. Inhibitory effect of the tea polyphenol, (-)-epigallocatechin gallate, on growth of cervical adenocarcinoma cell lines. Cancer Lett. 2006, 234, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Schramm, L.; Hernandez, N. Recruitment of RNA polymerase III to its target promoters. Gene Dev. 2002, 16, 2593–2620. [Google Scholar] [CrossRef] [PubMed]
- White, R.J. RNA polymerase III transcription and cancer. Oncogene 2004, 23, 3208–3216. [Google Scholar] [CrossRef] [PubMed]
- Mital, R.; Kobayashi, R.; Hernandez, N. RNA polymerase III transcription from the human U6 and adenovirus type 2 VAI promoters has different requirements for human BRF, a subunit of human TFIIIB. Mol. Cell. Biol. 1996, 16, 7031–7042. [Google Scholar] [CrossRef] [PubMed]
- Teichmann, M.; Wang, Z.X.; Roeder, R.G. A stable complex of a novel transcription factor IIB-related factor, human TFIIIB50, and associated proteins mediate selective transcription by RNA polymerase III of genes with upstream promoter elements. Proc. Natl. Acad. Sci. USA 2000, 97, 14200–14205. [Google Scholar] [CrossRef] [PubMed]
- Jacob, J.; Cabarcas, S.; Veras, I.; Zaveri, N.; Schramm, L. The green tea component EGCG inhibits RNA polymerase III transcription. Biochem. Biophys. Res. Commun. 2007, 360, 778–783. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, M.; Noguchi, M.; Nakao, Y.; Ysunaga, M.; Yamasaki, F.; Iwasaka, T. Antiproliferative effects of the major tea polyphenol, (-)-epigallocatechin gallate and retinoic acid in cervical adenocarcinoma. Gynecol. Oncol. 2008, 108, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Kavallaris, M. Microtubules and resistance to tubulin-binding agents. Nat. Rev. Cancer 2010, 10, 194–204. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarty, S.; Das, A.; Bhattacharya, A.; Chakrabarti, G. Theaflavins depolymerize microtubule network through tubulin binding and cause apoptosis of cervical carcinoma hela cells. J. Agric. Food Chem. 2011, 59, 2040–2048. [Google Scholar] [CrossRef] [PubMed]
- Ji, H.; Liu, N.; Yin, Y.; Wang, X.; Chen, X.; Li, J.; Li, J. Oxytocin inhibits ovarian cancer metastasis by repressing the expression of MMP-2 and VEGF. J. Cancer 2018, 9, 1379–1384. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.Q.; Xu, M.; Zhong, W.T.; Cui, Z.Y.; Liu, F.M.; Zhou, K.Y.; Li, X.Y. EGCG decreases the expression of HIF-1α and VEGF and cell growth in MCF-7 breast cancer cells. J. Buon 2014, 19, 435–439. [Google Scholar] [PubMed]
- Li, X.Y.; Feng, Y.; Liu, J.H.; Feng, X.W.; Zhou, K.Y.; Tang, X.D. Epigallocatechin-3-gallate inhibits IGF-I-stimulated lung cancer angiogenesis through downregulation of HIF-l α and VEGF expression. J. Nutrigenet. Nutr. 2013, 6, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, N.; Gerber, H.P.; LeCouter, J. The biology of VEGF and its receptors. Nat. Med. 2003, 9, 669–676. [Google Scholar] [CrossRef] [PubMed]
- De Francesco, E.M.; Sims, A.H.; Maggiolini, M.; Sotgia, F.; Lisanti, M.P.; Clarke, R.B. GPER mediates the angiocrine actions induced by IGF1 through the HIF-1 α/VEGF pathway in the breast tumor microenvironment. Breast Cancer Res. 2017, 19, 129. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.L.; Jiang, B.H.; Rue, E.A.; Semenza, G.L. Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. Proc. Natl. Acad. Sci. USA 1995, 92, 5510–5514. [Google Scholar] [CrossRef] [PubMed]
- Carmeliet, P.; Dor, Y.; Herbert, J.M.; Fukumura, D.; Brusselmans, K.; Dewerchin, M.; Neeman, M.; Bono, F.; Abramovitch, R.; Maxwell, P.; et al. Role of HIF-1α in hypoxia-mediated apoptosis, cell proliferation and tumour angiogenesis. Nature 1998, 394, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Krishnamachary, B.; Berg-Dixon, S.; Kelly, B.; Agani, F.; Feldser, D.; Ferreira, G.; Iyer, N.; LaRusch, J.; Pak, B.; Taghavi, P.; et al. Regulation of colon carcinoma cell invasion by hypoxia-inducible factor 1. Cancer Res. 2003, 63, 1138–1143. [Google Scholar] [PubMed]
- Frezza, M.; Schmitt, S.; Dou, Q.P. Targeting the ubiquitin-proteasome pathway: An emerging concept in cancer therapy. Curr. Top. Med. Chem. 2011, 11, 2888–2905. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Tang, X.; Lu, Q.; Zhang, Z.; Rao, J.; Le, A.D. Green tea extract and (-)-epigallocatechin-3-gallate inhibit hypoxia- and serum-induced HIF-1α protein accumulation and VEGF expression in human cervical carcinoma and hepatoma cells. Mol. Cancer Ther. 2006, 5, 1227–1238. [Google Scholar] [CrossRef] [PubMed]
- Joyce, J.A. Therapeutic targeting of the tumor microenvironment. Cancer Cell 2005, 7, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Park, C.C.; Bissell, M.J.; Barcellos-Hoff, M.H. The influence of the microenvironment on the malignant phenotype. Mol. Med. Today 2000, 6, 324–329. [Google Scholar] [CrossRef]
- Wilson, K.E.; Li, Z.Q.; Kara, M.; Gardner, K.L.; Roberts, D.D. β(1) integrin- and proteoglycan-mediated stimulation of T lymphoma cell adhesion and mitogen-activated protein kinase signaling by thrombospondin-1 and thrombospondin-1 peptides. J. Immunol. 1999, 163, 3621–3628. [Google Scholar] [PubMed]
- Kleine-Lowinski, K.; Gillitzer, R.; Kuhne-Heid, R.; Rosl, F. Monocyte-chemo-attractant-protein-1 (MCP-1)-gene expression in cervical intra-epithelial neoplasias and cervical carcinomas. Int. J. Cancer 1999, 82, 6–11. [Google Scholar] [CrossRef]
- Cui, X.D.; Lee, M.J.; Yu, G.R.; Kim, I.H.; Yu, H.C.; Song, E.Y.; Kim, D.G. EFNA1 ligand and its receptor EphA2: Potential biomarkers for hepatocellular carcinoma. Int. J. Cancer 2010, 126, 940–949. [Google Scholar] [CrossRef] [PubMed]
- Le Jan, S.; Amy, C.; Cazes, A.; Monnot, C.; Lamande, N.; Favier, J.; Philippe, J.; Sibony, M.; Gasc, J.M.; Corvol, P.; et al. Angiopoietin-like 4 is a proangiogenic factor produced during ischemia and in conventional renal cell carcinoma. Am. J. Pathol. 2003, 162, 1521–1528. [Google Scholar] [CrossRef]
- Kaido, T.; Bandu, M.T.; Maury, C.; Ferrantini, M.; Belardelli, F.; Gresser, I. IFN-α(1) gene transfection completely abolishes the tumorigenicity of murine B16 melanoma-cells in allogeneic DBA/2 mice and decreases their tumorigenicity in syngeneic C57BL/6 mice. Int. J. Cancer 1995, 60, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Sidky, Y.A.; Borden, E.C. Inhibition of angiogenesis by interferons-effects on tumor-induced and lymphocyte-induced vascular-responses. Cancer Res. 1987, 47, 5155–5161. [Google Scholar] [PubMed]
- Jovanovic, M.; Stefanoska, I.; Radojcic, L.; Vicovac, L. Interleukin-8 (CXCL8) stimulates trophoblast cell migration and invasion by increasing levels of matrix metalloproteinase (MMP)2 and MMP9 and integrins α(5) and β(1). Reproduction 2010, 139, 789–798. [Google Scholar] [CrossRef] [PubMed]
- Tudoran, O.; Soritau, O.; Balacescu, O.; Balacescu, L.; Braicu, C.; Rus, M.; Gherman, C.; Virag, P.; Irimie, F.; Berindan-Neagoe, I. Early transcriptional pattern of angiogenesis induced by EGCG treatment in cervical tumour cells. J. Cell. Mol. Med. 2012, 16, 520–530. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, S.L.; Stremlau, M.; He, X.; Basile, J.R.; Munger, K. Degradation of the retinoblastoma tumor suppressor by the human papillomavirus type 16 E7 oncoprotein is important for functional inactivation and is separable from proteasomal degradation of E7. J. Virol. 2001, 75, 7583–7591. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, M.; Tsutsumi, K.; Pater, A.; Pater, M.M. Human papillomavirus 18-immortalized endocervical cells with in-vitro cytokeratin expression characteristics of adenocarcinoma. Obstet. Gynecol. 1994, 83, 197–204. [Google Scholar] [PubMed]
- Kuhn, D.J.; Burns, A.C.; Kazi, A.; Dou, Q.P. Direct inhibition of the ubiquitin-proteasome pathway by ester bond-containing green tea polyphenols is associated with increased expression of sterol regulatory element-binding protein 2 and LDL receptor. Biochim. Biophys. Acta 2004, 1682, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Sampath, A.; Raychaudhuri, P.; Bagchi, S. Both Rb and E7 are regulated by the ubiquitin proteasome pathway in HPV-containing cervical tumor cells. Oncogene 2001, 20, 4740–4749. [Google Scholar] [CrossRef] [PubMed]
- Bonfili, L.; Cuccioloni, M.; Mozzicafreddo, M.; Cecarini, V.; Angeletti, M.; Eleuteri, A.M. Identification of an EGCG oxidation derivative with proteasome modulatory activity. Biochimie 2011, 93, 931–940. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Y.Y.; Cao, J.Y.; Xie, L.Q.; Shi, X.L. Cell growth inhibition and gene expression regulation by (-)-epigallocatechin-3-gallate in human cervical cancer cells. Arch. Pharm. Res. 2009, 32, 1309–1315. [Google Scholar] [CrossRef] [PubMed]
- Giannelli, G.; Antonaci, S. MMP and TIMP assay in cancer: Biological and clinical significance. Int. J. Cancer 2005, 116, 1002–1003. [Google Scholar] [CrossRef]
- GomisRuth, F.X.; Maskos, K.; Betz, M.; Bergner, A.; Huber, R.; Suzuki, K.; Yoshida, N.; Nagase, H.; Brew, K.; Bourenkov, G.P.; et al. Mechanism of inhibition of the human matrix metalloproteinase stromelysin-1 by TIMP-1. Nature 1997, 389, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Sharma, C.; Nusri, Q.E.A.; Begum, S.; Javed, E.; Rizvi, T.A.; Hussain, A. (-)-Epigallocatechin-3-gallate induces apoptosis and inhibits invasion and migration of human cervical cancer cells. Asian Pac. J. Cancer 2012, 13, 4815–4822. [Google Scholar] [CrossRef]
- Roomi, M.W.; Monterrey, J.C.; Kalinovsky, T.; Rath, M.; Niedzwiecki, A. In vitro modulation of MMP-2 and MMP-9 in human cervical and ovarian cancer cell lines by cytokines, inducers and inhibitors. Oncol. Rep. 2010, 23, 605–614. [Google Scholar] [PubMed]
- Siddiqui, F.A.; Naim, M.; Islam, N. Apoptotic effect of green tea polyphenol (EGCG) on cervical carcinoma cells. Diagn. Cytopathol. 2011, 39, 500–504. [Google Scholar] [CrossRef] [PubMed]
- Al-Hazzani, A.A.; Alshatwi, A.A. Catechin hydrate inhibits proliferation and mediates apoptosis of siha human cervical cancer cells. Food Chem. Toxicol. 2011, 49, 3281–3286. [Google Scholar] [CrossRef] [PubMed]
- Finkel, T. Oxidant signals and oxidative stress. Curr. Opin. Cell Biol. 2003, 15, 247–254. [Google Scholar] [CrossRef]
- Beckman, K.B.; Ames, B.N. Oxidative decay of DNA. J. Biol. Chem. 1997, 272, 19633–19636. [Google Scholar] [CrossRef] [PubMed]
- Azam, S.; Hadi, N.; Khan, N.U.; Hadi, S.M. Prooxidant property of green tea polyphenols epicatechin and epigallocatechin-3-gallate: Implications for anticancer properties. Toxicol. In Vitro 2004, 18, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Li, G.X.; Chen, Y.K.; Hou, Z.; Xiao, H.; Jin, H.Y.; Lu, G.; Lee, M.J.; Liu, B.; Guan, F.; Yang, Z.H.; et al. Pro-oxidative activities and dose-response relationship of (-)-epigallocatechin-3-gallate in the inhibition of lung cancer cell growth: A comparative study in vivo and in vitro. Carcinogenesis 2010, 31, 902–910. [Google Scholar] [CrossRef] [PubMed]
- Krstic, M.; Stojadinovic, M.; Smiljanic, K.; Stanic-Vucinic, D.; Velickovic, T.C. The anti-cancer activity of green tea, coffee and cocoa extracts on human cervical adenocarcinoma hela cells depends on both pro-oxidant and anti-proliferative activities of polyphenols. RSC Adv. 2015, 5, 3260–3268. [Google Scholar] [CrossRef]
- Zhang, H.; Cao, D.; Cui, W.; Ji, M.; Qian, X.; Zhong, L. Molecular bases of thioredoxin and thioredoxin reductase-mediated prooxidant actions of (-)-epigallocatechin-3-gallate. Free Radic. Biol. Med. 2010, 49, 2010–2018. [Google Scholar] [CrossRef] [PubMed]
- Zorov, D.B.; Juhaszova, M.; Sollott, S.J. Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol. Rev. 2014, 94, 909–950. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.M.; Bae, Y.S.; Lee, S.Y. Molecular ordering of ROS production, mitochondrial changes, and caspase activation during sodium salicylate-induced apoptosis. Free Radic. Biol. Med. 2003, 34, 434–442. [Google Scholar] [CrossRef]
- Singh, M.; Tyagi, S.; Bhui, K.; Prasad, S.; Shukla, Y. Regulation of cell growth through cell cycle arrest and apoptosis in HPV 16 positive human cervical cancer cells by tea polyphenols. Investig. New Drug. 2010, 28, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Luzio, J.P.; Pryor, P.R.; Bright, N.A. Lysosomes: Fusion and function. Nat. Rev. Cell Biol. 2007, 8, 622–632. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yang, N.D.; Zhou, F.; Shen, T.; Duan, T.; Zhou, J.; Shi, Y.; Zhu, X.Q.; Shen, H.M. (-)-Epigallocatechin-3-gallate induces non-apoptotic cell death in human cancer cells via ROS-mediated lysosomal membrane permeabilization. PLoS ONE 2012, 7, e46749. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.P.; Au-Yeung, S.C.F.; To, K.K.W. Platinum-based anticancer agents: Innovative design strategies and biological perspectives. Med. Res. Rev. 2003, 23, 633–655. [Google Scholar] [CrossRef] [PubMed]
- Arany, I.; Safirstein, R.L. Cisplatin nephrotoxicity. Semin. Nephrol. 2003, 23, 460–464. [Google Scholar] [CrossRef]
- Matsushima, H.; Yonemura, K.; Ohishi, K.; Hishida, A. The role of oxygen free radicals in cisplatin-induced acute renal failure in rats. J. Lab. Clin. Med. 1998, 131, 518–526. [Google Scholar] [CrossRef]
- Kilic, U.; Sahin, K.; Tuzcu, M.; Basak, N.; Orhan, C.; Elibol-Can, B.; Kilic, E.; Sahin, F.; Kucuk, O. Enhancement of cisplatin sensitivity in human cervical cancer: Epigallocatechin-3-gallate. Front. Nutr. 2014, 1, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Bhui, K.; Singh, R.; Shukla, Y. Tea polyphenols enhance cisplatin chemosensitivity in cervical cancer cells via induction of apoptosis. Life Sci. 2013, 93, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Bhatnagar, P.; Srivastava, A.K.; Kumar, P.; Shukla, Y.; Gupta, K.C. Enhancement of cancer chemosensitization potential of cisplatin by tea polyphenols poly(lactide-co-glycolide) nanoparticles. J. Biomed. Nanotechnol. 2011, 7, 202. [Google Scholar] [CrossRef] [PubMed]
- Alshatwi, A.A.; Athinarayanan, J.; Subbarayan, P.V. Green synthesis of platinum nanoparticles that induce cell death and G2/m-phase cell cycle arrest in human cervical cancer cells. J. Mater. Sci. Mater. Med. 2015, 26, 7. [Google Scholar] [CrossRef] [PubMed]
- Burger, R.M.; Peisach, J.; Horwitz, S.B. Activated bleomycin-a transient complex of drug, iron, and oxygen that degrades DNA. J. Biol. Chem. 1981, 256, 1636–1644. [Google Scholar]
- Sleijfer, S. Bleomycin-induced pneumonitis. Chest 2001, 120, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Hay, J.; Shahzeidi, S.; Laurent, G. Mechanisms of bleomycin-induced lung damage. Arch. Toxicol. 1991, 65, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Vorechovsky, I.; Munzarova, M.; Lokaj, J. Increased bleomycin-induced chromosome-damage in lymphocytes of patients with common variable immunodeficiency indicates an involvement of chromosomal instability in their cancer predisposition. Cancer Immunol. Immunothera. 1989, 29, 303–306. [Google Scholar] [CrossRef]
- Alshatwi, A.A.; Periasamy, V.S.; Athinarayanan, J.; Elango, R. Synergistic anticancer activity of dietary tea polyphenols and bleomycin hydrochloride in human cervical cancer cell: Caspase-dependent and independent apoptotic pathways. Chem. Biol. Interact. 2016, 247, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Tallen, G.; Riabowol, K. Keep-ING balance: Tumor suppression by epigenetic regulation. FEBS Lett. 2014, 588, 2728–2742. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.; Hussain, A.; Sundaram, M.K.; Alalami, U.; Gunasekera, D.; Ramesh, L.; Hamza, A.; Quraishi, U. (-)-Epigallocatechin-3-gallate reverses the expression of various tumor-suppressor genes by inhibiting DNA methyltransferases and histone deacetylases in human cervical cancer cells. Oncol. Rep. 2015, 33, 1976–1984. [Google Scholar] [CrossRef] [PubMed]
- Virksaite, A.; Bakutyte, S.; Navakauskiene, R. Assessment of apoptosis and senescence in acute myeloid leukemia NB-4 cells treated with epigenetic modifiers EGCG and BIX-01294. Eur. J Cancer 2016, 69, S85. [Google Scholar] [CrossRef]
- Subramaniam, D.; Thombre, R.; Dhar, A.; Anant, S. DNA methyltransferases: A novel target for prevention and therapy. Front. Oncol. 2014, 4, 80. [Google Scholar] [CrossRef] [PubMed]
- Sobel, M.E. Differential expression of the 67 kDa laminin receptor in cancer. Semin. Cancer Biol. 1993, 4, 311–317. [Google Scholar] [PubMed]
- Kumazoe, M.; Sugihara, K.; Tsukamoto, S.; Huang, Y.H.; Tsurudome, Y.; Suzuki, T.; Suemasu, Y.; Ueda, N.; Yamashita, S.; Kim, Y.; et al. 67-kDa laminin receptor increases cGMP to induce cancer-selective apoptosis. J. Clin. Investig. 2013, 123, 787–799. [Google Scholar] [CrossRef] [PubMed]
- Gundimeda, U.; McNeill, T.H.; Fan, T.K.; Deng, R.; Rayudu, D.; Chen, Z.; Cadenas, E.; Gopalakrishna, R. Green tea catechins potentiate the neuritogenic action of brain-derived neurotrophic factor: Role of 67-kDa laminin receptor and hydrogen peroxide. Biochem. Biophys. Res. Commun. 2014, 445, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Umeda, D.; Tachibana, H.; Yamada, K. Epigallocatechin-3-o-gallate disrupts stress fibers and the contractile ring by reducing myosin regulatory light chain phosphorylation mediated through the target molecule 67 kDa laminin receptor. Biochem. Biophys. Res. Commun. 2005, 333, 628–635. [Google Scholar] [CrossRef] [PubMed]
- Umeda, D.; Yano, S.; Yamada, K.; Tachibana, H. Green tea polyphenol epigallocatechin-3-gallate signaling pathway through 67-kDa laminin receptor. J. Biol. Chem. 2008, 283, 3050–3058. [Google Scholar] [CrossRef] [PubMed]
- Tsukamoto, S.; Yamashita, S.; Kim, Y.H.; Kumazoe, M.; Huang, Y.; Yamada, K.; Tachibana, H. Oxygen partial pressure modulates 67-kDa laminin receptor expression, leading to altered activity of the green tea polyphenol, EGCG. FEBS Lett. 2012, 586, 3441–3447. [Google Scholar] [CrossRef] [PubMed]
- Moradzadeh, M.; Hosseini, A.; Erfanian, S.; Rezaei, H. Epigallocatechin-3-gallate promotes apoptosis in human breast cancer T47D cells through down-regulation of PI3K/AKT and telomerase. Pharmacol. Rep. 2017, 69, 924–928. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zuo, J.; Wang, G.D. Epigallocatechin-3-gallate suppresses cell proliferation and promotes apoptosis in Ec9706 and Eca109 esophageal carcinoma cells. Oncol. Lett. 2017, 14, 4391–4395. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, L.; Ma, H.W. Label-free real-time investigation of the effect of telomerase inhibitors based on quartz crystal microbalance measurement. Sens. Actuators B Chem. 2017, 239, 943–950. [Google Scholar] [CrossRef]
- Stern, J.L.; Theodorescu, D.; Vogelstein, B.; Papadopoulos, N.; Cech, T.R. Mutation of the TERT promoter, switch to active chromatin, and monoallelic TERT expression in multiple cancers. Genes Dev. 2015, 29, 2219–2224. [Google Scholar] [CrossRef] [PubMed]
- Circu, M.; Cardelli, J.; Barr, M.P.; O’Byrne, K.; Mills, G.; El-Osta, H. Modulating lysosomal function through lysosome membrane permeabilization or autophagy suppression restores sensitivity to cisplatin in refractory non-small-cell lung cancer cells (vol 12, e0184922, 2017). PLoS ONE 2018, 13, e0197016. [Google Scholar] [CrossRef] [PubMed]
- Kaminskas, E.; Farrell, A.T.; Wang, Y.C.; Sridhara, R.; Pazdur, R. FDA drug approval summary: Azacitidine (5-azacytidine, vidazaTM) for injectable suspension. Oncologist 2005, 10, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Hu, T.; Hang, C.Y.; Yang, R.; Li, X.; Chen, Z.L.; Mei, Y.D.; Zhang, Q.H.; Huang, K.C.; Xiang, Q.Y.; et al. Case-control study of diet in patients with cervical cancer or precancerosis in Wufeng, a high incidence region in China. Asian Pac. J. Cancer Prev. 2012, 13, 5299–5302. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |

| Ingredient | Drug | Cell Line | Cytotoxic Action | Reference |
|---|---|---|---|---|
| EGCG | cisplatin | HeLa | Attenuated the toxicity and enhanced the sensitivity of cisplatin, decreased cellular survival and induced apoptosis, regulated NF-kB p65, Akt and mTOR pathways | Kilic et al. (2014) Singh et al. (2011) |
| retinoic acid | HEN | Prevented carcinogenesis and induced apoptosis, inhibited telomerase activity | Yokoyama et al. (2008) | |
| Tea polyphenols | platinum | SiHa | Induced G2/M phase cell cycle arrest, increased subG0 cell death phase and inhibited proliferation | Alshatwi et al. (2015) |
| bleomycin hydrochloride | SiHa | Enhanced the therapeutic properties of bleomycin (BLM), activated caspase-3, -8, -9, upregulated Bcl-2 and P53 expression and induced apoptosis | Alshatwi et al. (2016) | |
| cisplatin | HeLa, SiHa | Increased the chemosensitivity and minimized the toxicity of cisplatin | Singh et al. (2013) |
| Cell Line | Function | Cytotoxic Action | Reference |
|---|---|---|---|
| HeLa | Anti-proliferation | Induced G1 phase cell cycle arrest and apoptosis, inhibited EGFR signaling pathway | Sah et al. (2004) |
| Induced G2/M phase cell cycle arrest and apoptosis, depolymerized microtubule | Chakrabarty et al. (2011) | ||
| Reduced IGF-1R activity and inhibited proliferation of cells | Li et al. (2007) | ||
| Inhibited Akt and NF-kB activation, inhibited cell growth | Singh et al. (2011) | ||
| Inhibited the expression of Brf1, Brf2 and its promoter, inhibited RNA polIII transcription | Jacob et al. (2007) | ||
| Depolymerized cellular microtubule | Chakrabarty et al. (2015) | ||
| Inhibited HIF-1α protein accumulation, decreased VEGF expression, blocked P3K/Akt, ERK1/2 signaling pathway | Zhang et al. (2006) | ||
| Inhibited HPV E6, E7, ERα, and aromatase expression | Qiao et al. (2009) | ||
| Inhibited proteasome functionality, induced apoptosis | Bonfili et al. (2011) | ||
| Reduced enzymatic activity of DNMT and HDAC, inhibited carcinogenesis | Khan et al. (2015) | ||
| Anti-metastasis | Inhibited invasion and migration, decreased MMP-9 and TIMP-1 expression | Sharma et al. (2012) | |
| Reduced proliferation, adhesion, invasion of tumor cell, exhibited anti-angiogenesis effect | Tudoran et al. (2012) | ||
| Inhibited invasion and migration, decreased MMP-2, -9 expression | Roomi et al. (2010) | ||
| Pro-apoptosis | Inactivated Trx/TrxR, induced prooxidant cytotoxicity and apoptosis | Zhang et al. (2010) | |
| Induced LMP secretion | Zhang et al. (2012) | ||
| Induced formation of intracellular ROS | Krstic et al. (2015) | ||
| Caski | Anti-proliferation | Induced G1 phase cell cycle arrest and apoptosis, regulated gene expression | Ahn et al. (2003) |
| Induced G1 phase cell cycle arrest and apoptosis, inhibited EGFR signaling pathway | Sah et al. (2004) | ||
| Inhibited HPV E6/7, ERα, and aromatase expression | Qiao et al. (2009) | ||
| SiHa | Anti-proliferation | Induced G1 phase cell cycle arrest and apoptosis, inhibited EGFR signaling pathway | Sah et al. (2004) |
| Pro-apoptosis | Increased caspase-3, -8, -9 secretion and inhibited cell growth | Al-Hazzani et al. (2011) | |
| OMC-1 | Anti-proliferation | Inhibited telomerase activity, induced cell cycle dysregulation and apoptosis | Noguchi et al. 2006) |
| HEN, HEC | Anti-proliferation | Inhibited telomerase activity and cell growth, induced apoptosis | Yokoyama et al. (2004) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.-Q.; Lu, J.-L.; Liang, Y.-R.; Li, Q.-S. Suppressive Effects of EGCG on Cervical Cancer. Molecules 2018, 23, 2334. https://doi.org/10.3390/molecules23092334
Wang Y-Q, Lu J-L, Liang Y-R, Li Q-S. Suppressive Effects of EGCG on Cervical Cancer. Molecules. 2018; 23(9):2334. https://doi.org/10.3390/molecules23092334
Chicago/Turabian StyleWang, Ying-Qi, Jian-Liang Lu, Yue-Rong Liang, and Qing-Sheng Li. 2018. "Suppressive Effects of EGCG on Cervical Cancer" Molecules 23, no. 9: 2334. https://doi.org/10.3390/molecules23092334
APA StyleWang, Y.-Q., Lu, J.-L., Liang, Y.-R., & Li, Q.-S. (2018). Suppressive Effects of EGCG on Cervical Cancer. Molecules, 23(9), 2334. https://doi.org/10.3390/molecules23092334






