Structural Diversity of Six Coordination Polymers Based on the Designed X-Shaped Ligand 1,1,1,1-Tetrakis[(3-pyridiniourea)methyl]methane
Abstract
:1. Introduction
2. Results and Discussion
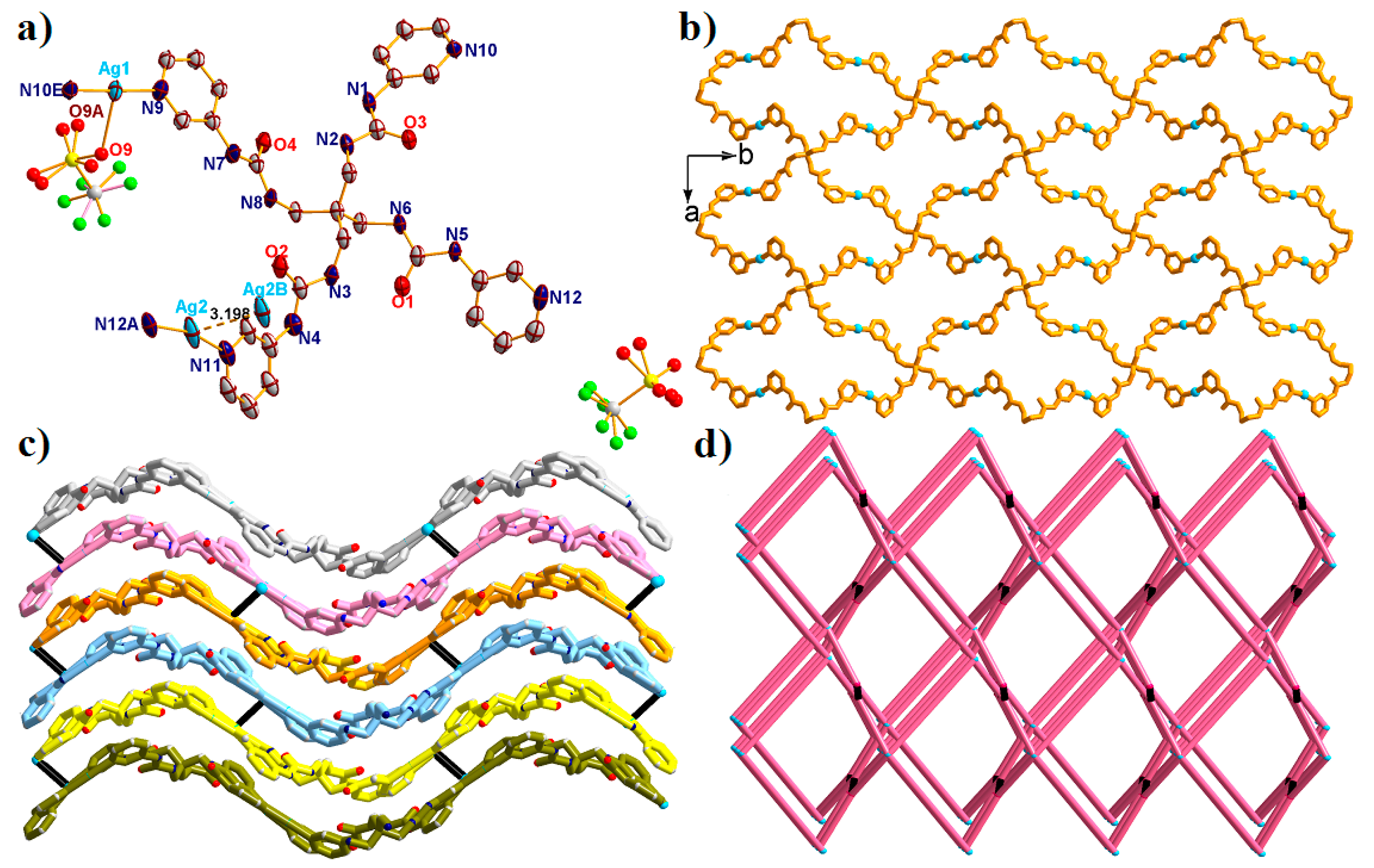
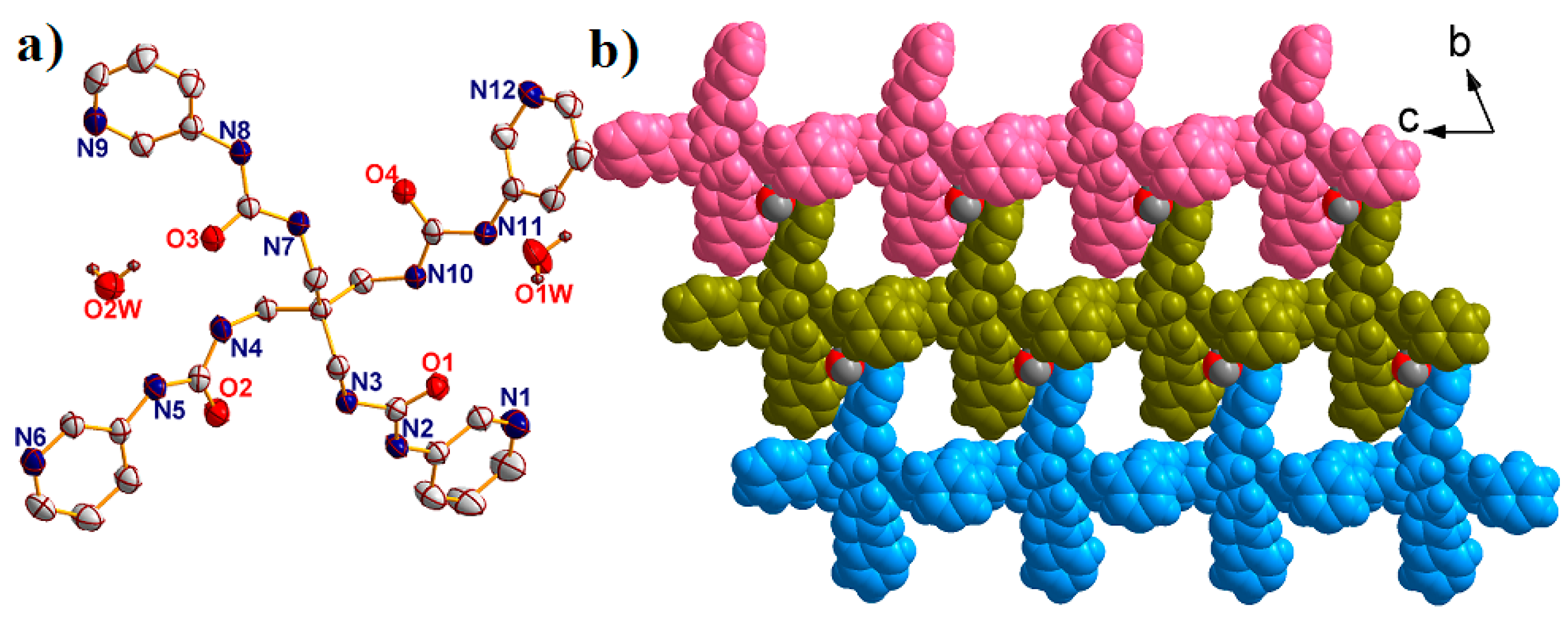
2.1. Structural Description of {[Ag2(L)(CF3SO3)2]·2H2O·DMF}n (1)
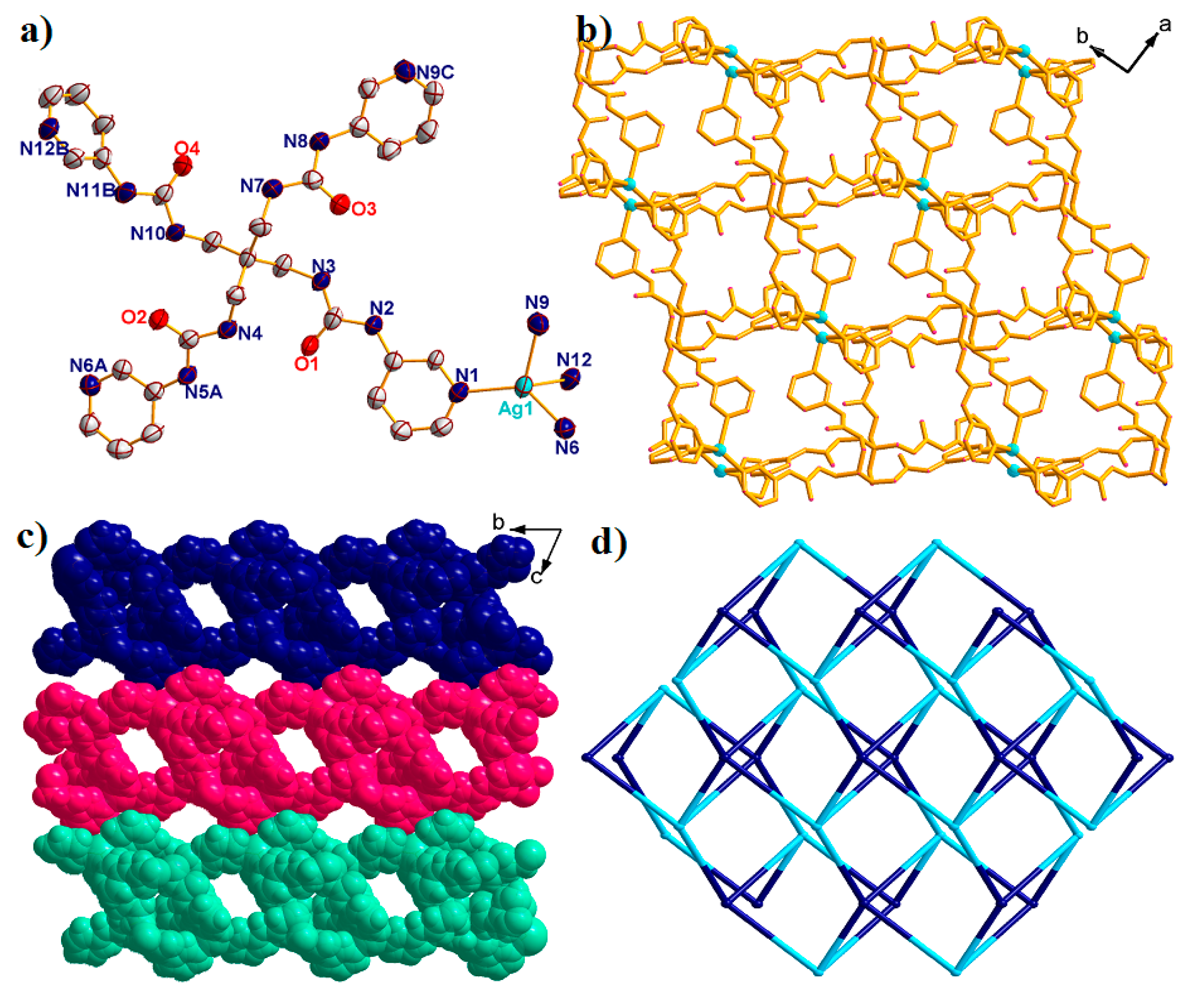
2.2. Structural Description of {[Ag(L)]·SbF6·4DMF·H2O}n (2)
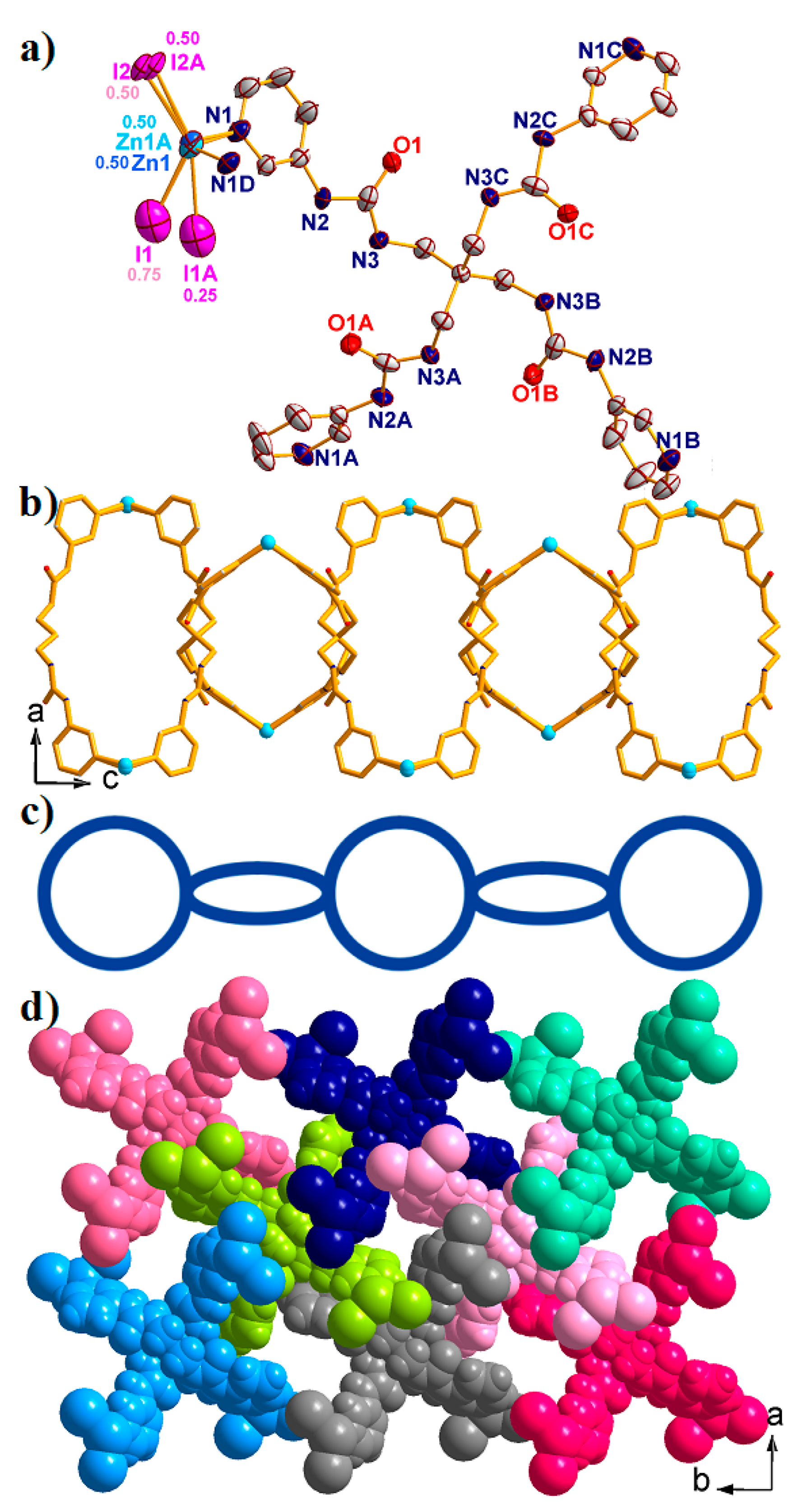
2.3. Structural Description of {[Zn(L)0.5(I)2]·3.75H2O}n (3)
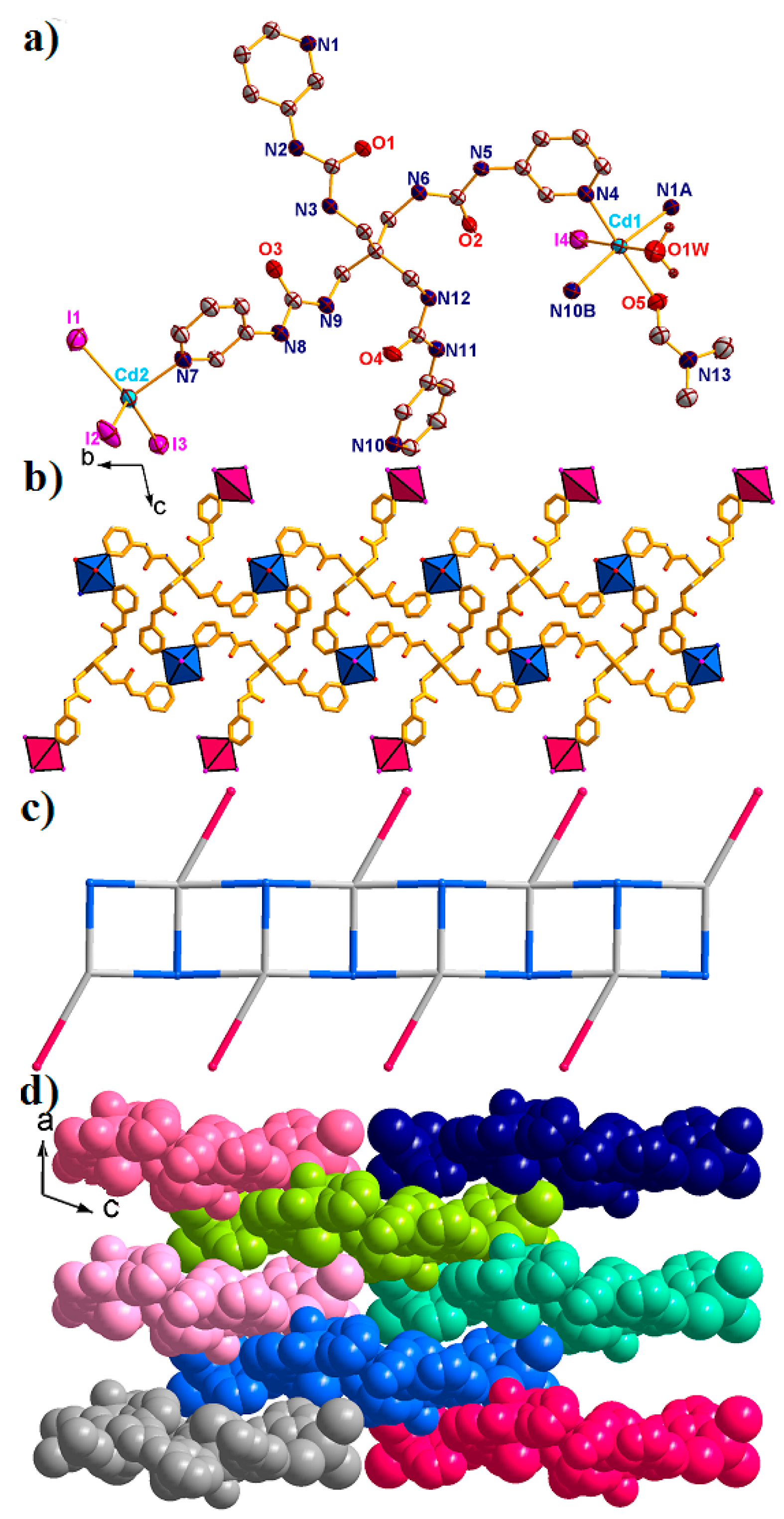
2.4. Structural Description of {[Cd2(L)(I)4(H2O)(DMF)]·4H2O·3DMF}n (4)
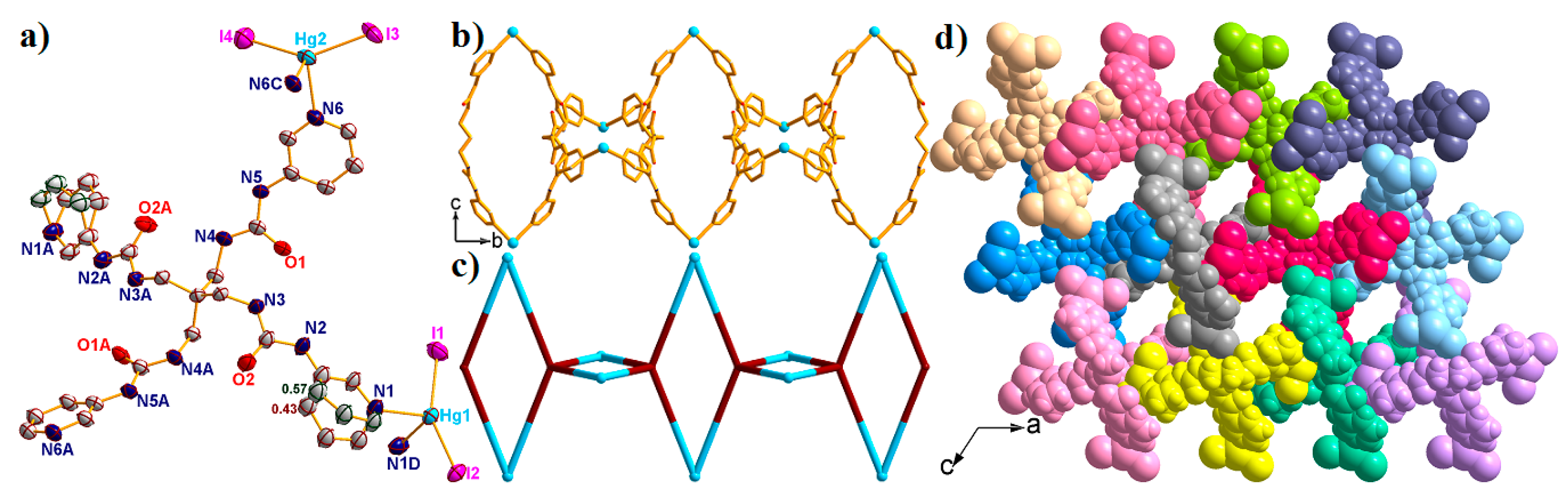
2.5. Structural Description of {[Hg2(L)(I)4]·H2O·4DMF}n (5)
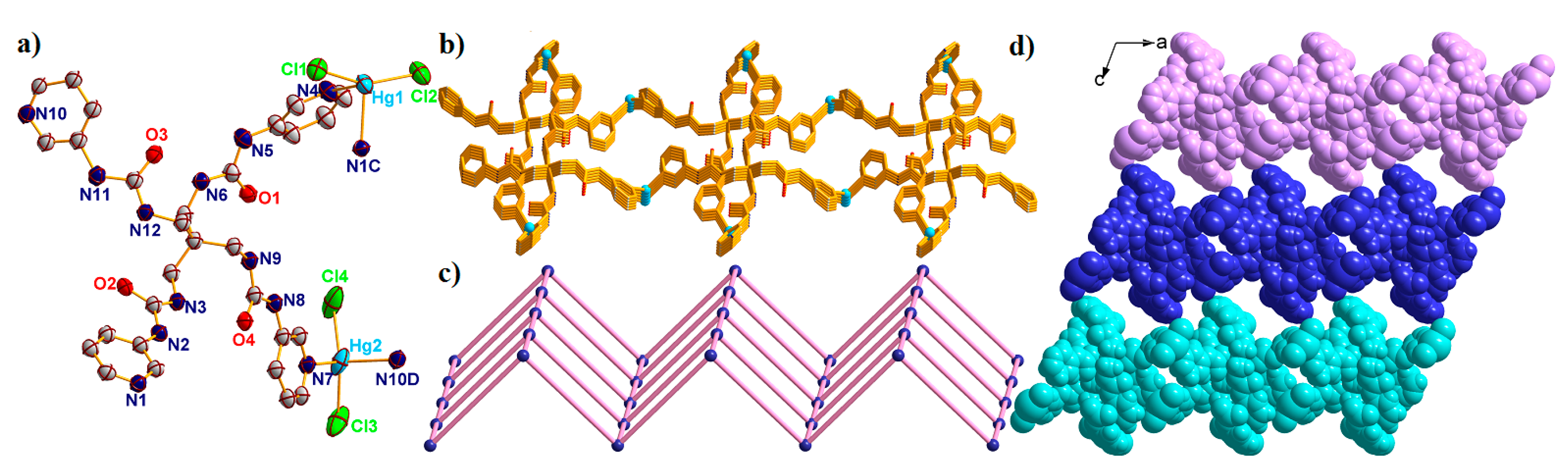
2.6. Structural Description of {[Hg2(L)(Cl)4]·2H2O·3DMF}n (6)
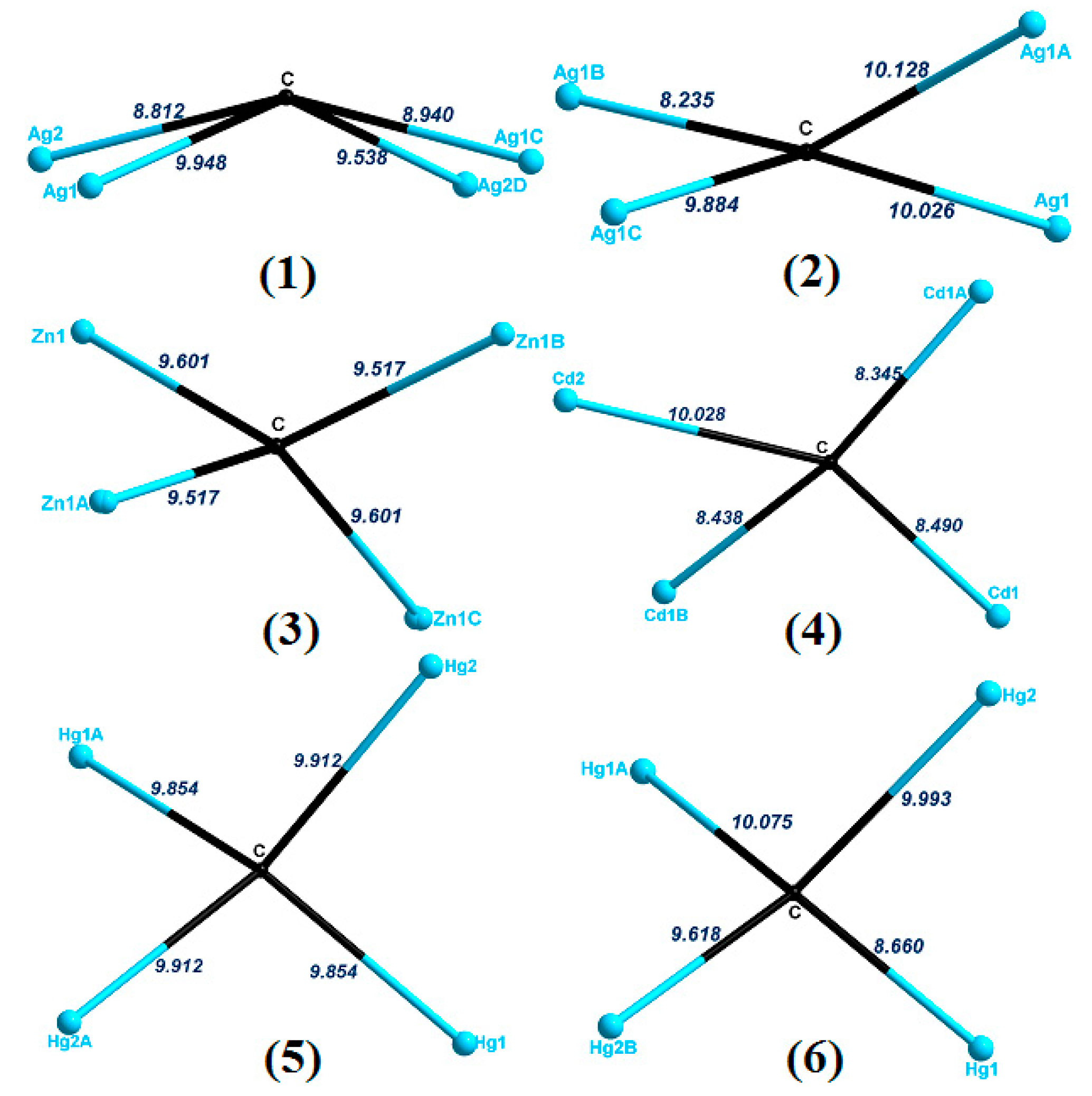
2.7. Structural Comparison
2.8. Powder X-ray Diffraction Analyses (PXRD)
3. Experimental Section
3.1.Materials and Methods
3.2. Synthesis of the Ligand and Complexes
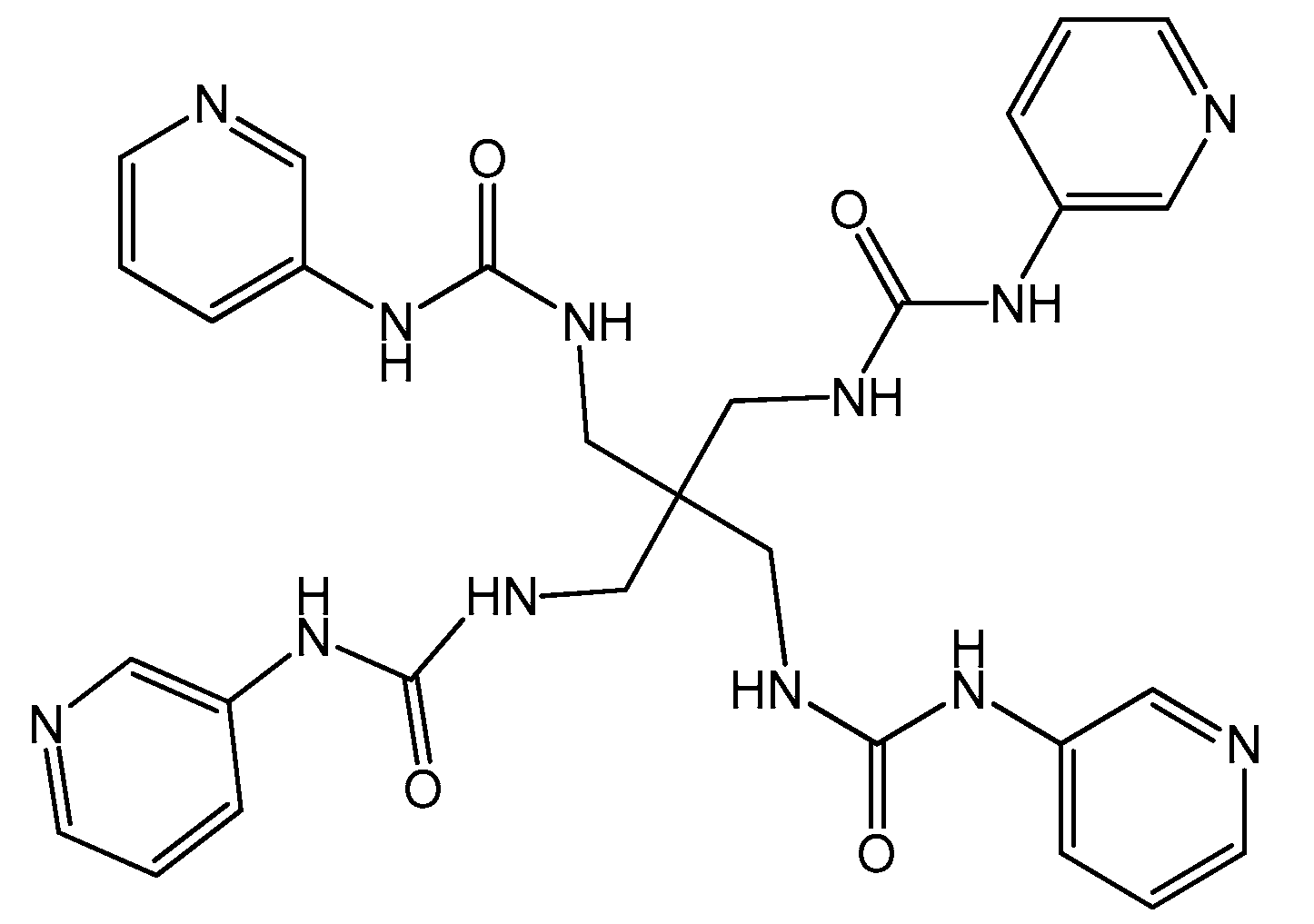
3.2.1. Design of 1,1,1,1-tetrakis[(3-pyridiniourea)methyl]methane (L)
3.2.2. Preparation of {[Ag2(L)(CF3SO3)]·CF3SO3·2H2O·DMF}n (1)
3.2.3. Preparation of {[Ag(L)]·SbF6·4DMF·H2O}n (2)
3.2.4. Preparation of {[Zn(L)0.5(I)2]·3.75H2O}n (3)
3.2.5. Preparation of {[Cd2(L)(I)4(H2O)(DMF)]·4H2O·3DMF}n (4)
3.2.6. Preparation of {[Hg2(L)(I)4]·H2O·4DMF}n (5)
3.2.7. Preparation of {[Hg2(L)(Cl)4]·2H2O·3DMF}n (6)
3.3. X–ray Crystallography
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dhakshinamoorthy, A.; Asiri, A.M.; Garcia, H. Metal-organic frameworks catalyzed C-C and C-heteroatom coupling reactions. Chem. Soc. Rev. 2015, 44, 1922–1947. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Li, N.; Wang, X.; Chang, Z.; Bu, X.H. Host-guest engineering of coordination polymers for highly tunable luminophores based on charge transfer emissions. ACS Appl. Mater. Interfaces 2017, 9, 2662–2668. [Google Scholar] [CrossRef] [PubMed]
- He, C.B.; Liu, M.; Lin, W.B. Nanomedicine applications of hybrid nanomaterials built from metal-ligand coordination bonds: Nanoscale metal-organic frameworks and nanoscale coordination polymers. Chem. Rev. 2015, 115, 11079–11108. [Google Scholar] [CrossRef] [PubMed]
- Schoedel, A.; Li, M.; Li, D.; O’Keeffe, M.; Yaghi, O.M. Structures of metal-organic frameworks with rod secondary building units. Chem. Rev. 2016, 116, 12466–12535. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.F.; Liu, Q.Y.; Wang, Y.L.; Yin, S.G. A water-stable anionic metal-organic framework constructed from columnar zinc-adeninate units for highly selective light hydrocarbon separation and efficient separation of organic dyes. Inorg. Chem. 2017, 56, 2919–2925. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Patil, R.S.; Li, T.; Barnes, C.L.; Atwood, J.L. Self-assembly of magnesium-seamed hexameric pyrogallol[4]arene nanocapsules. Chem. Commun. 2017, 53, 4312–4314. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Zhang, C.L.; Ju, Z.M.; Zheng, H.G. Solvent-induced synthesis of cobalt(II) coordination polymers based on a rigid ligand and flexible carboxylic acid ligands: Syntheses, structures and magnetic properties. Dalton Trans. 2015, 44, 6926–6935. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.C.; Han, Q.; Chen, L.J.; Zhao, J.W. A brief review of the crucial progress on heterometallic polyoxotungstates in the past decade. CrystEngComm 2016, 18, 842–862. [Google Scholar] [CrossRef]
- Zhang, J.W.; Kan, X.M.; Li, X.L.; Luan, J.; Wang, X.L. Transition metal carboxylate coordination polymers with amide-bridged polypyridine co-ligands: Assemblies and properties. CrystEngComm 2015, 17, 3887–3907. [Google Scholar] [CrossRef]
- Lu, C.H.; Cecconello, A.; Willner, I. Recent advances in the synthesis and functions of reconfigurable interlocked DNA nanostructures. J. Am. Chem. Soc. 2016, 138, 5172–5185. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.J.; Ju, H.; Kim, S.; Park, K.M.; Lee, S.S. Anion-directed coordination networks of a flexible s-pivot ligand and anion exchange in the solid state. Cryst. Growth Des. 2015, 15, 5427–5436. [Google Scholar] [CrossRef]
- Wright, J.S.; Metherell, A.J.; Cullen, W.M.; Piper, J.R.; Dawson, R. Highly selective CO2 vs. N2 adsorption in the cavity of a molecular coordination cage. Chem. Commun. 2017, 53, 4398–4401. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.L.; Yu, C.X.; Ma, F.J.; Li, Y.R.; Han, J.J.; Lin, L.; Ma, L.F. Structural diversity and photocatalytic properties of Cd(II) coordination polymers constructed by a flexible V-shaped bipyridyl benzene ligand and dicarboxylate derivatives. Dalton Trans. 2015, 44, 1636–1645. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.D.; Steed, J.W. Gels with sense: Supramolecular materials that respond to heat, light and sound. Chem. Soc. Rev. 2016, 45, 6546–6596. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.; Mason, S.A.; Mossou, E.; Lamming, G.; Probert, M.R.; Steed, J.W. Neutron diffraction studies on guest-induced distortions in urea inclusion compounds. Cryst. Growth Des. 2016, 16, 7175–7185. [Google Scholar] [CrossRef]
- Radu, C.; Aurelien, B.; Bruce, A.M. Selectivity principles in anion separation by crystallization of hydrogen-bonding capsules. J. Am. Chem. Soc. 2010, 132, 7177–7185. [Google Scholar]
- Tehrani, A.A.; Esrafili, L.; Abedi, S.; Morsali, A.; Carlucci, L.; Proserpio, D.M.; Wang, J.; Junk, P.C.; Liu, T. Urea metal-organic frameworks for nitro-substituted compounds sensing. Inorg. Chem. 2017, 56, 1446–1454. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.W.; Huang, X.J.; Zhao, Q.L.; Li, S.G.; Wu, B. Hydrogen-bonded 1D nanotubes and 2D layers of group 12 metal complexes with a pyridylurea. CrystEngComm 2012, 14, 5446–5453. [Google Scholar] [CrossRef]
- Zhang, Q.L.; Huang, Y.L.; Xu, H.; Tu, B.; Zhu, B.X. Structural diversity of a series of urea-based coordination complexes: From water tapes, water-sulfate chains, to supramolecular structures. J. Coord. Chem. 2017, 70, 156–167. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [Google Scholar]
- SMART. Saint and SADABS; Bruker AXS Inc.: Madison, WI, USA, 1998. [Google Scholar]
- Blatov, V.A.; O’Keeffe, M.; Proserpio, D.M. Vertex-, face-, point-, Schläfli-, and Delaney-symbols in nets, polyhedra and tilings: Recommended terminology. CrystEngComm 2010, 12, 44–48. [Google Scholar] [CrossRef]
- Blatov, V.A.; Shevchenko, A.P.; Serezhkin, V.N. TOPOS3.2: A new version of the program package for multipurpose crystal-chemical analysis. J. Appl. Crystallogr. 2000, 33, 1193. [Google Scholar] [CrossRef]
- The Network Topology Was Evaluated by the Program “TOPOS–4.0”. Available online: http://www.topos.ssu.samara.ru (accessed on 5 September 2018).
Sample Availability: Samples of the compounds are not available from the authors. |
| Metal Salts | τ4 Parameter a | Average M-Ccentral (Å) | M-L Motifs | Final Structure | |
|---|---|---|---|---|---|
| 1 | AgCF3SO3 | 0.44 (7) | 9.310 | 2D [Ag2L]n sheet | 3D (3,4)-connected {6·82}{64·82}-tcj net |
| 2 | AgSbF6 | 0.49 (7) | 9.568 | 2D [AgL]n sheet | 2D 4-connected {43·63} sheet |
| 3 | ZnI2 | 0.67 (0) | 9.559 | 1D [Zn2L]n loop chain | 1D polymeric loop chain |
| 4 | CdI2 | 0.36 (0) | 8.825 | 1D [Cd2L]n fishbone chain | 1D polymeric fishbone chain |
| 5 | HgI2 | 0.65 (5) | 9.883 | 1D [Hg2L]n loop chain | 1D polymeric loop chain |
| 6 | HgCl2 | 0.78 (1) | 9.587 | 2D [Hg2L]n bilayer | 2D 4-connected{44·62}-sql sheet |
| Complex | Ligand | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|---|
| Empirical formula | C29H36N12O6 | C34H43Ag2F6N13O13S2 | C79H117Ag2Sb2F12N31O17 | C58H94I8N24O23Zn4 | C41H70Cd2I4N16O13 | C41H60Hg2I4N16O8 | C38H58Cl4Hg2N15O9 |
| Formula weight | 648.70 | 1235.67 | 2460.28 | 2772.25 | 1727.53 | 1813.82 | 1411.97 |
| Crystal system | Triclinic | Monoclinic | Triclinic | Tetragonal | Triclinic | Monoclinic | Monoclinic |
| Space group | P-1 | P21/c | P-1 | I4/m | P-1 | C2/m | P21 |
| a (Å) | 8.7220(10) | 18.667(3) | 10.9801(8) | 17.902(9) | 11.842(16) | 26.3943(10) | 17.782(8) |
| b (Å) | 14.1492(14) | 29.678(5) | 16.3332(12) | 17.902(9) | 16.48(2) | 15.4834(6) | 8.948(4) |
| c (Å) | 14.7268(17) | 8.6711(15) | 17.1655(12) | 15.312(16) | 17.71(2) | 16.8250(8) | 19.152(9) |
| α (°) | 64.258(3) | 90 | 66.498(2) | 90 | 101.147(17) | 90 | 90 |
| β (°) | 77.246(3) | 102.504(4) | 81.929(2) | 90.00 | 108.606(16) | 123.338(2) | 110.646(7) |
| γ (°) | 72.356(6) | 90 | 87.312(2) | 90 | 92.633(18) | 90 | 90 |
| V (Å3) | 1551.6(3) | 4689.8(14) | 2795.0(3) | 4907(6) | 3191(7) | 5744.4(4) | 2852(2) |
| Z | 2 | 4 | 1 | 2 | 2 | 2 | 2 |
| Dcalcd (Mg/m3) | 1.388 | 1.750 | 1.462 | 1.876 | 1.798 | 1.759 | 1.644 |
| μ (mmÿ1) | 0.101 | 1.022 | 0.913 | 3.555 | 2.665 | 7.523 | 5.623 |
| F (000) | 684 | 2488 | 1248 | 2676 | 1680 | 2776 | 1382 |
| Rint | 0.0570 | 0.1292 | 0.0799 | 0.0479 | 0.0486 | 0.0508 | 0.0831 |
| Final R indices a | 0.0479 (0.1111) | 0.0971 (0.1915) | 0.0803 (0.2284) | 0.1259 (0.2454) | 0.0587 (0.1492) | 0.0382 (0.1026) | 0.0695 (0.1486) |
| R indices (all data) a | 0.0802 (0.1366) | 0.1999 (0.2349) | 0.1363 (0.2734) | 0.1427 (0.2540) | 0.1172 (0.1492) | 0.0453 (0.1065) | 0.1624 (0.1729) |
| Gof | 1.025 | 1.072 | 1.048 | 1.007 | 1.069 | 1.042 | 1.036 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Q.-L.; Yu, Q.; Xie, H.-F.; Tu, B.; Xu, H.; Huang, Y.-L.; Yang, X.-S. Structural Diversity of Six Coordination Polymers Based on the Designed X-Shaped Ligand 1,1,1,1-Tetrakis[(3-pyridiniourea)methyl]methane. Molecules 2018, 23, 2292. https://doi.org/10.3390/molecules23092292
Zhang Q-L, Yu Q, Xie H-F, Tu B, Xu H, Huang Y-L, Yang X-S. Structural Diversity of Six Coordination Polymers Based on the Designed X-Shaped Ligand 1,1,1,1-Tetrakis[(3-pyridiniourea)methyl]methane. Molecules. 2018; 23(9):2292. https://doi.org/10.3390/molecules23092292
Chicago/Turabian StyleZhang, Qi-Long, Qing Yu, Hai-Fang Xie, Bo Tu, Hong Xu, Ya-Li Huang, and Xiao-Sheng Yang. 2018. "Structural Diversity of Six Coordination Polymers Based on the Designed X-Shaped Ligand 1,1,1,1-Tetrakis[(3-pyridiniourea)methyl]methane" Molecules 23, no. 9: 2292. https://doi.org/10.3390/molecules23092292
APA StyleZhang, Q.-L., Yu, Q., Xie, H.-F., Tu, B., Xu, H., Huang, Y.-L., & Yang, X.-S. (2018). Structural Diversity of Six Coordination Polymers Based on the Designed X-Shaped Ligand 1,1,1,1-Tetrakis[(3-pyridiniourea)methyl]methane. Molecules, 23(9), 2292. https://doi.org/10.3390/molecules23092292





