Identification of Three Dalbergia Species Based on Differences in Extractive Components
Abstract
:1. Introduction
2. Results and Discussion
2.1. Contents Analysis of Extractives
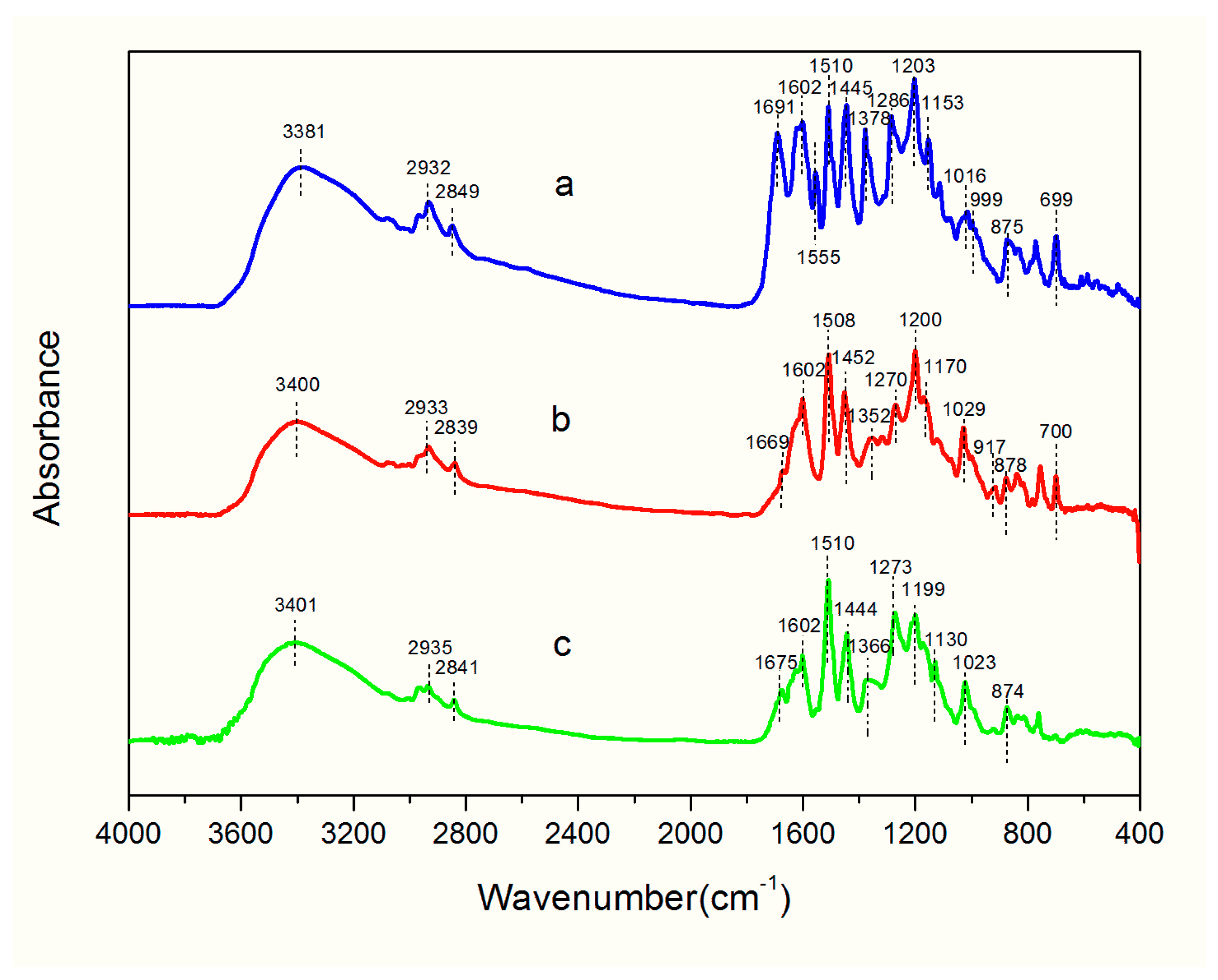
2.2. FTIR Analysis
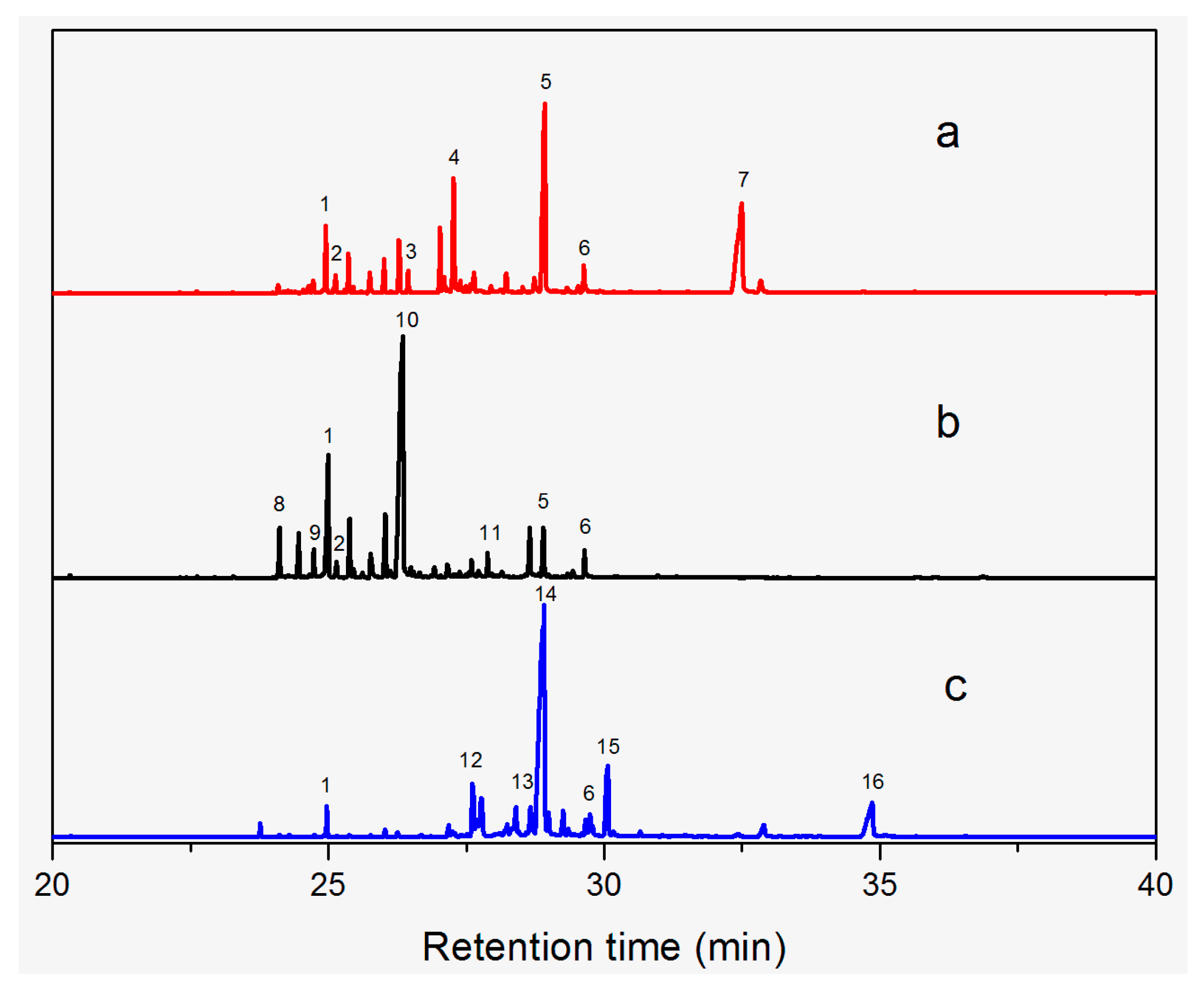
2.3. GC-MS Analysis
3. Materials and Methods
3.1. Wood Samples
3.2. Preparation of Wood Extracts
3.3. FTIR Analysis
3.4. GC-MS Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hillis, W.E. Heartwood and Tree Exudates; Springer: Berlin, Germany, 1987; pp. 76–119. [Google Scholar]
- Roffael, E. Significance of wood extractives for wood bonding. Appl. Microbiol. Biotechnol. 2015, 100, 1589–1596. [Google Scholar] [CrossRef] [PubMed]
- Hillis, W.E. Distribution, properties, and formation of some wood extractives. Wood Sci. Technol. 1971, 5, 272–289. [Google Scholar] [CrossRef]
- Schultz, T.P.; Nicholas, D.D. Naturally durable heartwood evidence for a proposed dual defensive function of the extractives. Phytochemistry 2000, 54, 47–52. [Google Scholar] [CrossRef]
- Umezawa, T. Chemistry of extractives. In Wood and Cellulosic Chemistry; Taylor Francis Inc.: New York, NY, USA, 2000. [Google Scholar]
- Tittikpina, N.K.; Nana, F.; Fontanay, S.; Philippot, S.; Batawila, K.; Akpagana, K. Antibacterial activity and cytotoxicity of pterocarpus erinaceus poir extracts, fractions and isolated compounds. J. Ethnopharmacol. 2018, 212, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Nagawa, C.B.; Böhmdorfer, S.; Rosenau, T. Chemical composition of volatiles extracted from indigenous tree species of Uganda: Composition of bark extracts from Psorospermum febrifugum and Milicia excelsa. Holzforschung 2015, 69, 815–821. [Google Scholar] [CrossRef]
- Seshadri, T.R. Polyphenols of Pterocarpus and Dalbergia woods. Phytochemistry 1972, 11, 881–898. [Google Scholar] [CrossRef]
- Convention on International Trade in Endangered Species of Wild Fauna and Flora. Appendices I, II, and III; Government of South Africa: Johannesburg, South Africa, 24 September 2016.
- Li, Q.; Wu, J.; Wang, Y.; Lian, X.; Wu, F.; Zhou, L. The phylogenetic analysis of Dalbergia (fabaceae: Papilionaceae) based on different DNA barcodes. Holzforschung 2017, 71, 939–949. [Google Scholar] [CrossRef]
- Jenkins, M.; Oldfield, S.; Aylett, T.; Jenkins, M.; Oldfield, S.; Aylett, T. International Trade in African Blackwood; Fauna and Flora International: Cambridge, UK, 2002. [Google Scholar]
- Barrett, M.A.; Brown, J.L.; Morikawa, M.K.; Labat, J.; Yoder, A.D. CITES Designation for endangered rosewood in Madagascar. Science 2010, 328, 1109–1110. [Google Scholar] [CrossRef] [PubMed]
- Hassold, S.; Ii, P.P.L.; Bauert, M.R.; Razafintsalama, A.; Ramamonjisoa, L.; Widmer, A. DNA barcoding of malagasy rosewoods: Towards a molecular identification of cites-listed Dalbergia species. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- APPENDIX. A Summary of the Criteria for the Three CITES Appendices. Available online: https://www.cites.org (accessed on 8 May 2018).
- Richter, H.G.; Krause, V.J.; Muche, C. Wood Structure and physico-chemical properties compared with other central American species of Dalbergia. IAWA J. 1996, 17, 327–341. [Google Scholar] [CrossRef]
- Gasson, P.; Miller, R.; Stekel, D.J.; Whinder, F.; Zieminska, K. Wood identification of Dalbergia nigra (Cites Appendix I) using quantitative wood anatomy, principal components analysis and nacve bayes classification. Ann. Bot. 2010, 10545–10546. [Google Scholar]
- Wu, C.C.; Chu, F.H.; Ho, C.K.; Sung, C.H.; Chang, S.H. Comparative analysis of the complete chloroplast genomic sequence and chemical components of cinnamomum micranthum and cinnamomum kanehirae. Holzforschung 2017, 71, 189–197. [Google Scholar] [CrossRef]
- Kite, G.C.; Green, P.W.C.; Veitch, N.C.; Groves, M.C.; Gasson, P.E.; Simmonds, M.S.J. Dalnigrin, a neoflavonoid marker for the identification of brazilian rosewood (Dalbergia nigra) in cites enforcement. Phytochemistry 2010, 71, 1122–1131. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhang, F.; Huang, A.; Zhou, Q. Distinction of four Dalbergia species by FTIR, 2nd derivative IR, and 2D-IR spectroscopy of their ethanol-benzene extractives. Holzforschung 2016, 70, 503–510. [Google Scholar] [CrossRef]
- Vidaković, V.; Novaković, M.; Popović, Z.; Janković, M.; Matić, R.; Tešević, V. Significance of diarylheptanoids for chemotaxonomical distinguishing between Alnus glutinosa and Alnus incana. Holzforschung 2017, 72, 9–16. [Google Scholar] [CrossRef]
- Huang, A.; Zhou, Q.; Liu, J.; Fei, B.; Sun, S. Distinction of three wood species by Fourier transform infrared spectroscopy and two-dimensional correlation IR spectroscopy. J. Mol. Struct. 2008, 883, 160–166. [Google Scholar] [CrossRef]
- Ma, F.; Huang, A. Rapid identification and quantification three chicken-wing woods of Millettia leucantha, Millettia laurentii and Cassia siamea by FT-IR and 2DCOS-IR. J. Mol. Struct. 2018, 1166, 164–168. [Google Scholar] [CrossRef]
- Luo, S.; Wu, Y.; Huang, J. Thermal and Chemical Properties of Benzene/Alcohol Extractives from Two Species of Redwood. In Proceedings of the 2012 International Conference on Biobase Material Science and Engineering (BMSE), Changsha, China, 21–23 October 2012; pp. 156–160. [Google Scholar]
- Zhang, F.; Xu, C.; Li, M.; Chen, X.; Zhou, Q.; Huang, A. Identification of Dalbergia cochinchinensis (cites appendix ii) from other three Dalbergia, species using ft-ir and 2d correlation ir spectroscopy. Wood Sci. Technol. 2016, 50, 693–704. [Google Scholar] [CrossRef]
- Wang, F.; Huang, A.; Yin, X.; Wang, W.; Chen, J. Multilevel profiling and identification of Dalbergia odorifera and Dalbergia stevensonii by FTIR, NMR and GC/MS. Chin. Chem. Lett. 2017, 29, 1395–1398. [Google Scholar] [CrossRef]
- Iordache, A.; Culea, M.; Gherman, C. Characterization of some plant extracts by GC-MS. Nucl. Instrum. Methods Phys. Res. B 2009, 267, 338–342. [Google Scholar] [CrossRef]
- Mangindaan, B.; Matsushita, Y.; Aoki, D.; Yagami, S.; Kawamura, F.; Fukushima, K. Analysis of distribution of wood extractives in gmelina arborea by gas chromatography and time-of-flight secondary ion mass spectrometry. Holzforschung 2017, 71, 299–305. [Google Scholar] [CrossRef]
- Liu, R.; Wang, C.; Huang, A.; Lv, B. Characterization of Odors of Wood by Gas Chromatography-Olfactometry with Removal of Extractives as Attempt to Control Indoor Air Quality. Molecules 2018, 23, 203. [Google Scholar] [CrossRef] [PubMed]
- Pan, R.; Guo, F.; Lu, H.; Feng, W.; Liang, Y. Development of the chromatographic fingerprint of scutellaria barbata D. Don by GC-MS combined with chemometrics methods. J. Pharm. Biomed. Anal. 2011, 55, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, M.P.; Watson, P.A.; Breuil, C. Gas chromatography-mass spectrometry method for the simultaneous determination of wood extractive compounds in quaking aspen. J. Chromatogr. A 2001, 922, 225–233. [Google Scholar] [CrossRef]
- Vaysi, R. Identification and comparison of chemical components in wood and bark of planted eldar pine tree by GC/MS methods. Pro Ligno 2014, 10, 21–25. [Google Scholar]
- Xu, B.; Zhu, T.; Li, J.; Liu, S. Identification of wood between Phoebe zhennan and Machilus pingii using the gas chromatography-mass spectrometry direct injection technique. Eur. J. Mass Spectrom. 2013, 19, 187–193. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, H.; Sun, S.; Huang, L.; Noda, I. Differentiation of Rhizoma Curcumas longae and Radix Curcumae by a multistep infrared macro-fingerprint method. Anal. Lett. 2013, 46, 2597–2609. [Google Scholar] [CrossRef]
- Xu, R.; Sun, S.; Zhu, W. Multi-step infrared macro-fingerprint features of ethanol extracts from different Cistanche species in China combined with HPLC fingerprint. J. Mol. Struct. 2014, 1069, 236–244. [Google Scholar] [CrossRef]
- Liu, Y.; Li, J.; Fan, G.; Sun, S.; Zhang, Y.; Zhang, Y. Identification of the traditional tibetan medicine “shaji” and their different extracts through tri-step infrared spectroscopy. J. Mol. Struct. 2016, 1124, 180–187. [Google Scholar] [CrossRef]
- Sun, S.; Zhou, Q.; Chen, J. Infrared Spectroscopy for Complex. Mixtures: Applications in Food and Traditional Chinese Medicine; Chemical Industry Press: Beijing, China, 2011; pp. 58–61. [Google Scholar]
- Jiang, S.; Ge, S.; Wang, M.; Peng, W. Molecules and functions of rosewood: Dalbergia stevenson. Arab. J. Chem. 2017. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |

| Species | Yield of Extractives (%) | |||
|---|---|---|---|---|
| Ethanol/Water (9:1, v/v) | Benzene/Ethanol (2:1, v/v) | Ethyl Acetate | n-Hexane | |
| Dalbergia cultrate | 11.6 | 8.6 | 6.2 | - |
| Dalbergia latifolia | 10.1 | 9.65 | 6.8 | - |
| Dalbergia melanoxylon | 16.65 | 16.05 | 13.0 | - |
| Wavenumbers(cm−1) | |||
|---|---|---|---|
| D. Cultrata | D. Latifolia | D. Melanoxylon | Band Assignments |
| 3381 | 3400 | 3401 | O-H stretch; N-H stretch |
| 2932 | 2933 | 2935 | C-H stretch: CH2 |
| 2849 | 2839 | 2841 | C-H stretch:CH3 |
| 1691 | 1669 | 1675 | C=O stretch; C=N stretch |
| 1602 | 1602 | 1602 | C=C stretching of aromatic skeleton |
| 1555 | - | - | C=C stretching of aromatic skeleton |
| 1510 | 1508 | 1510 | C-C stretch bands within ring skeleton |
| - | 1452 | - | Skeletal C-C stretching; CH3 symmetrical bending vibrations ;CH2 scissoring |
| 1445 | 1444 | Aromatic stretching(flavonoids); CH3 symmetrical bending vibrations | |
| 1378 | - | - | CH3 asymmetrical bending vibrations |
| 1366 | CH3 symmetrical bending; in-plane C-OH bending rical bending | ||
| - | 1352 | CH3 symmetrical bending; in-plane C-OH bending C-N-C asymmrtric vibration of aromatic compounds | |
| - | 1318 | - | in-plane C-OH bending; C-O stretching |
| 1286 | - | - | C-O-C stretching (flavonoids) |
| - | 1270 | 1273 | C-O vibration |
| 1203 | 1200 | 1199 | C-O-C stretching |
| - | 1170 | - | C-O stretching C-O-C stretching or frame vibration (flavonoids) |
| 1153 | - | - | C-O-C stretching(flavonoids); in-plane C-OH bending C-O-C stretching or frame vibration (flavonoids) |
| - | - | 1130 | C-OH stretching |
| 1114 | - | - | C-OH stretching |
| 1016 | 1029 | 1023 | C-O stretching |
| 999 | - | - | C-H out-of-plane bend C-O stretching |
| - | 917 | - | C-H out-of-plane bend |
| 875 | 878 | 874 | C-O stretching |
| - | 839 | 836 | C-H stretching out of plane of aromatic ring; C-N wagging |
| 699 | 700 | C-H stretching out of plane of aromatic ring; C-S stretching | |
| ID | RT(min) | Compounds | Molecular Structure | Releative Content (%) * | ||
|---|---|---|---|---|---|---|
| D. Cultrate | D. Latifolia | D. Melanoxylon | ||||
| 1 | 24.97 | Phenol,4-methyl-2-[5-(2-thienyl)pyrazol-3-yl]- | 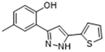 | 4.76 | 12.41 (0.7) | 1.47 |
| 6 | 29.66 | 13-Docosenamide, (Z)- |  | 2.08 (0.2) | 2.17 (0.2) | 1.26 (0.1) |
| 2 | 25.13 | Naphtho[2,3-b]furan-4,9-dione, 2-isopropyl- | 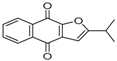 | 1.54 | 1.36 (0.2) | - |
| 5 | 28.90 | 1-Thioflavone, 7-methoxy- | 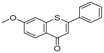 | 25.55 (0.2) | 4.23 | - |
| 3 | 26.44 | 3,3′,4,4′-Tetramethoxystilbene | 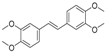 | 1.49 | - | - |
| 4 | 27.26 | 3,7,3′,4′-Tetrahydroxyflavone | 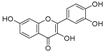 | 10.78 (0.1) | - | - |
| 7 | 32.49 | Parietin | 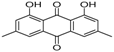 | 24.81 (0.4) | - | - |
| 8 | 24.11 | 1,7,7-Trimethyl-3-phenethylidenebicyclo[2.2.1]heptan-2-one | 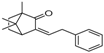 | - | 3.28 | - |
| 9 | 24.73 | Phenol, 4,4′-methylenebis[2,6-dimethyl- |  | - | 1.60 | - |
| 10 | 26.32 | 1,1′-Biphenyl, 4,2′,3′,4′-tetramethoxy-5′-methyl-6-methylaminomethyl- | 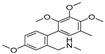 | - | 29.75 (0.8) | - |
| 11 | 27.88 | (4-Methylsulfanylphenyl)carbamic acid, 2,6-dimethoxyphenyl ester | 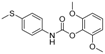 | - | 1.38 | - |
| 12 | 27.76 | 10,11-Dihydro-10-hydroxy-2,3- dimethoxydibenz(b,f)oxepin | 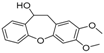 | - | - | 4.57 |
| 13 | 28.66 | 2-(4-Methoxy-2,5-dimethyl-phenyl)-9-methyl-2H-benzo[g]indazole |  | - | - | 3.14 |
| 14 | 28.88 | 10,11-Dihydro-10-hydroxy-2,3,6-trimethoxydibenz(b,f)oxepin | 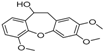 | - | - | 37.30 (0.8) |
| 15 | 30.06 | 10,11-Dihydro-2,3,6-trimethoxydibenz(b,f)oxepin-10-one | 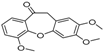 | - | - | 8.32 (0.2) |
| 16 | 34.86 | Pilloin |  | - | - | 7.77 (0.1) |
| Latin Name | Trade Name | Place of Origin |
|---|---|---|
| Dalbergia cultrate Benth. | Burmese blackwood | Laos |
| Dalbergia latifolia Roxb. | Indian rosewood | Indonesia |
| Dalbergia melanoxylon (Guill. & Perr.) | African blackwood | Mozambique |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yin, X.; Huang, A.; Zhang, S.; Liu, R.; Ma, F. Identification of Three Dalbergia Species Based on Differences in Extractive Components. Molecules 2018, 23, 2163. https://doi.org/10.3390/molecules23092163
Yin X, Huang A, Zhang S, Liu R, Ma F. Identification of Three Dalbergia Species Based on Differences in Extractive Components. Molecules. 2018; 23(9):2163. https://doi.org/10.3390/molecules23092163
Chicago/Turabian StyleYin, Xiaoqian, Anmin Huang, Shifeng Zhang, Ru Liu, and Fang Ma. 2018. "Identification of Three Dalbergia Species Based on Differences in Extractive Components" Molecules 23, no. 9: 2163. https://doi.org/10.3390/molecules23092163
APA StyleYin, X., Huang, A., Zhang, S., Liu, R., & Ma, F. (2018). Identification of Three Dalbergia Species Based on Differences in Extractive Components. Molecules, 23(9), 2163. https://doi.org/10.3390/molecules23092163







