Enhancing the Delivery of Chemotherapeutics: Role of Biodegradable Polymeric Nanoparticles
Abstract
1. Introduction
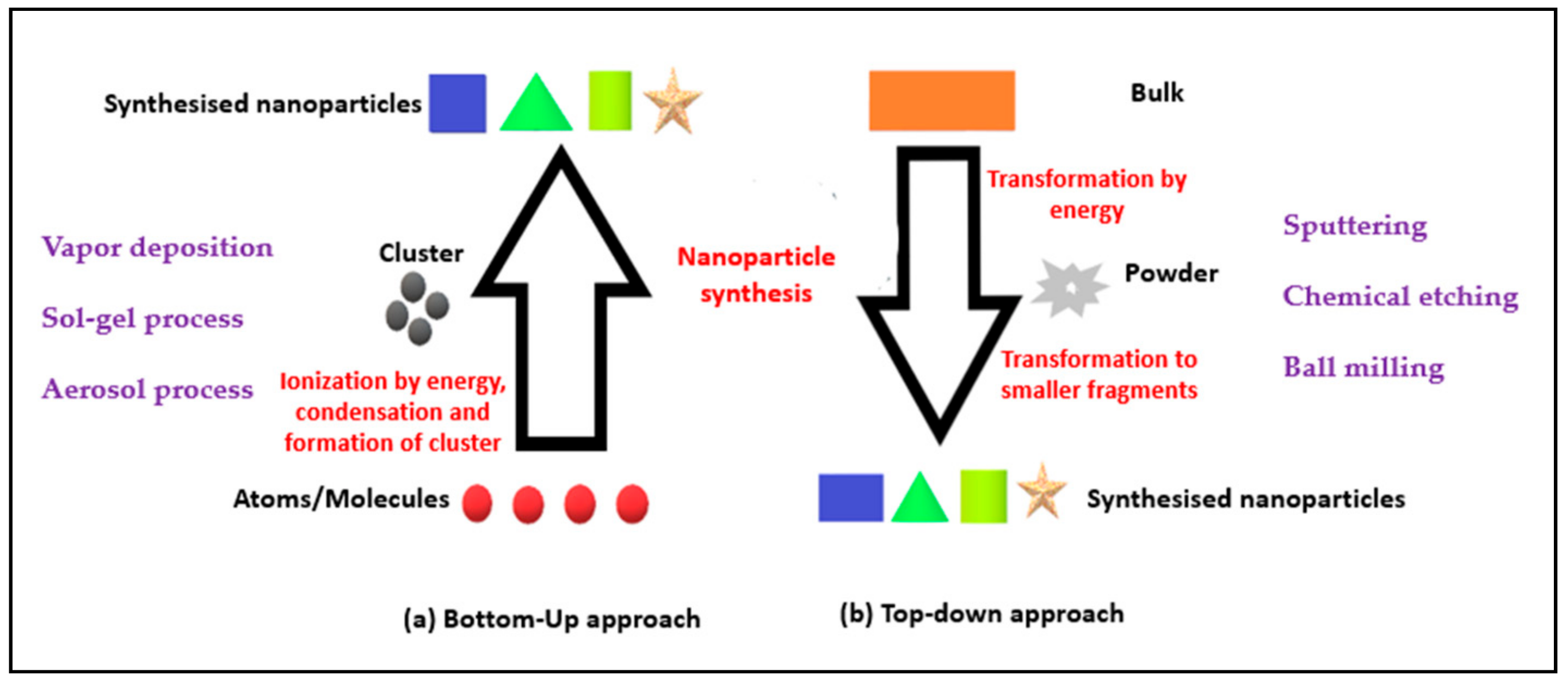
- “Bottom-Up” approach: Involves engineering the materials at atomic or molecular level through a process of assembly or self-assembly [2].
2. Types of Nanoparticles for Drug Delivery
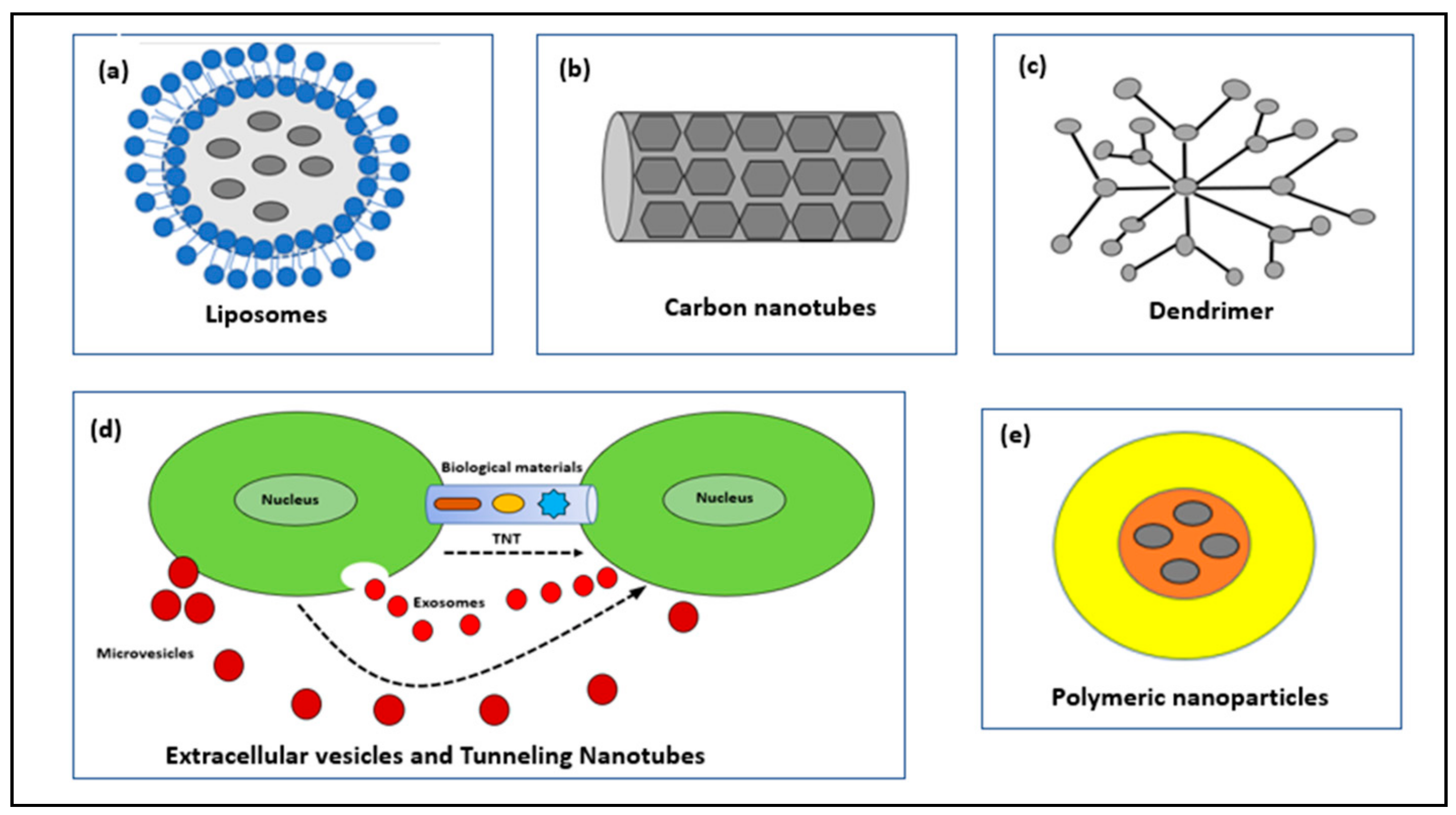
2.1. Liposomes
2.2. Carbon Nanotubes (CNTs)
2.3. Dendrimers
2.4. Extracellular Vesicles (EVs) and Tunneling Nanotubes (TNTs)
2.5. Polymeric Nanoparticles
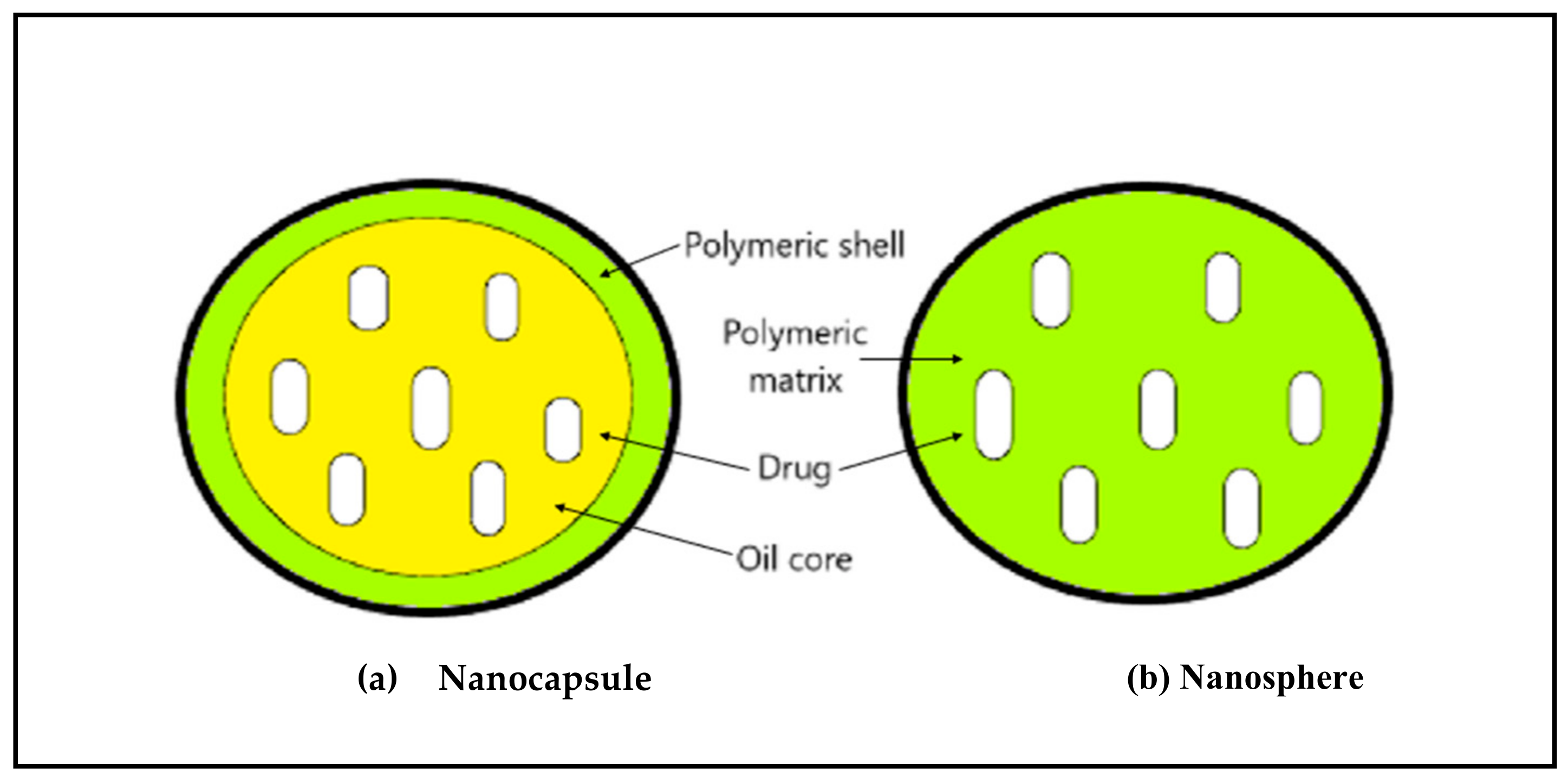
- Nanocapsules: They have an oily core and a polymeric outer surface. The drug can be adsorbed on the surface or encapsulated in the core.
- Nanospheres: The core and outer surface are made up of polymeric material and the drug is either retained or adsorbed in this polymeric structure.
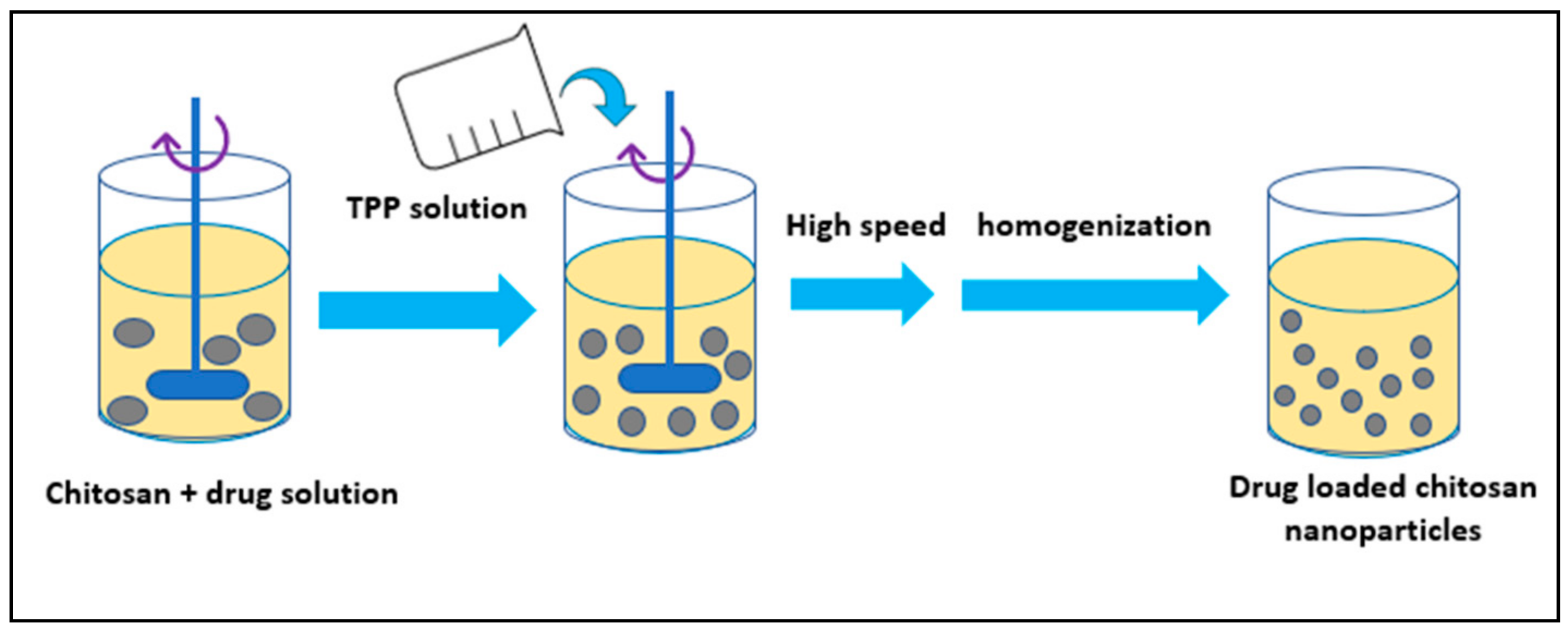
3. Synthesis of Polymeric Nanoparticles
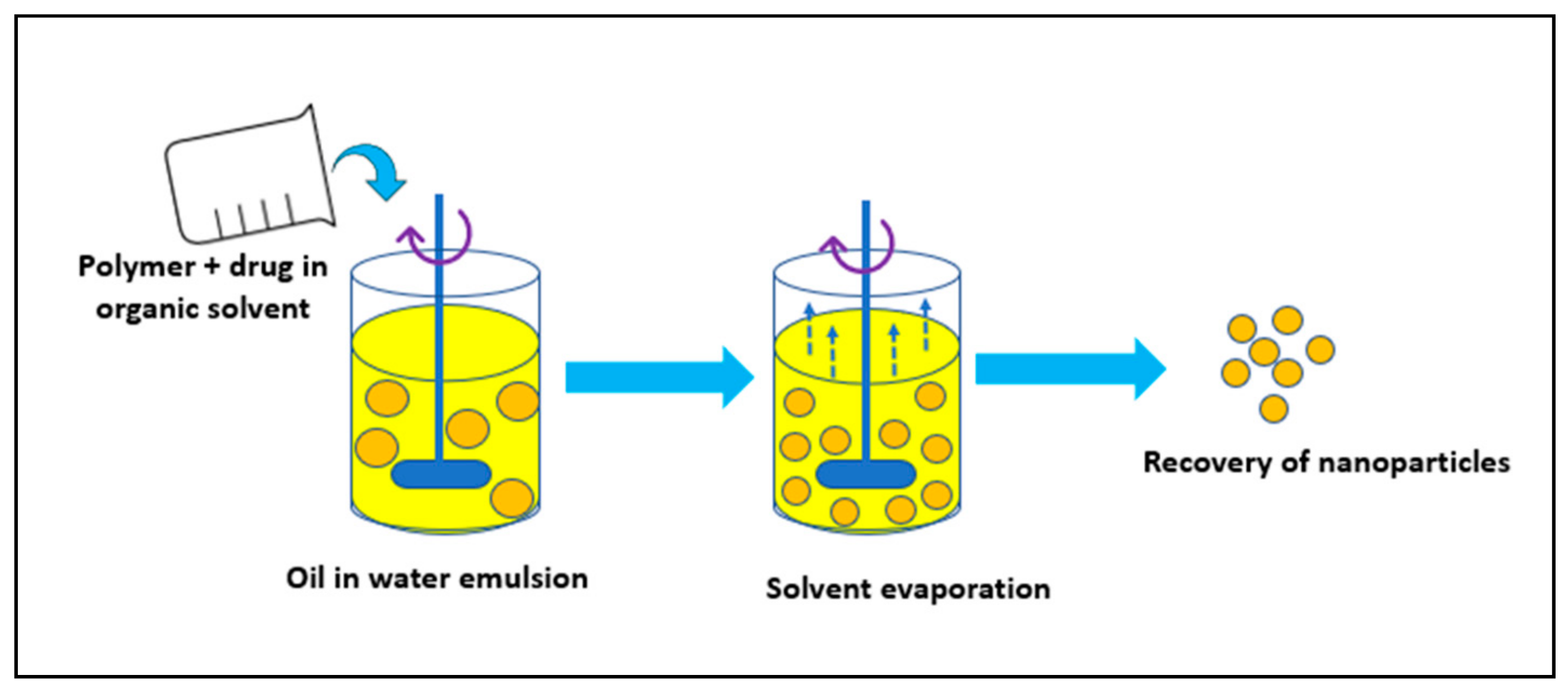
3.1. Solvent Evaporation Method
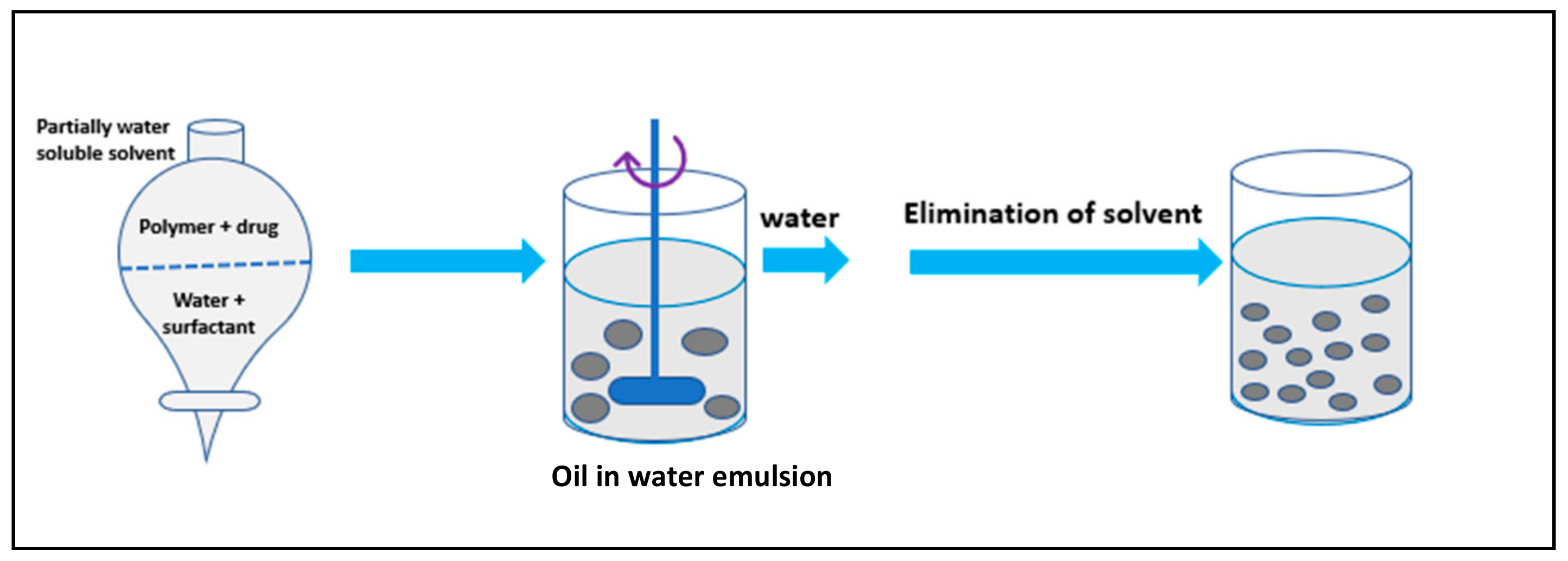
3.2. Spontaneous Emulsification/Solvent Diffusion Method
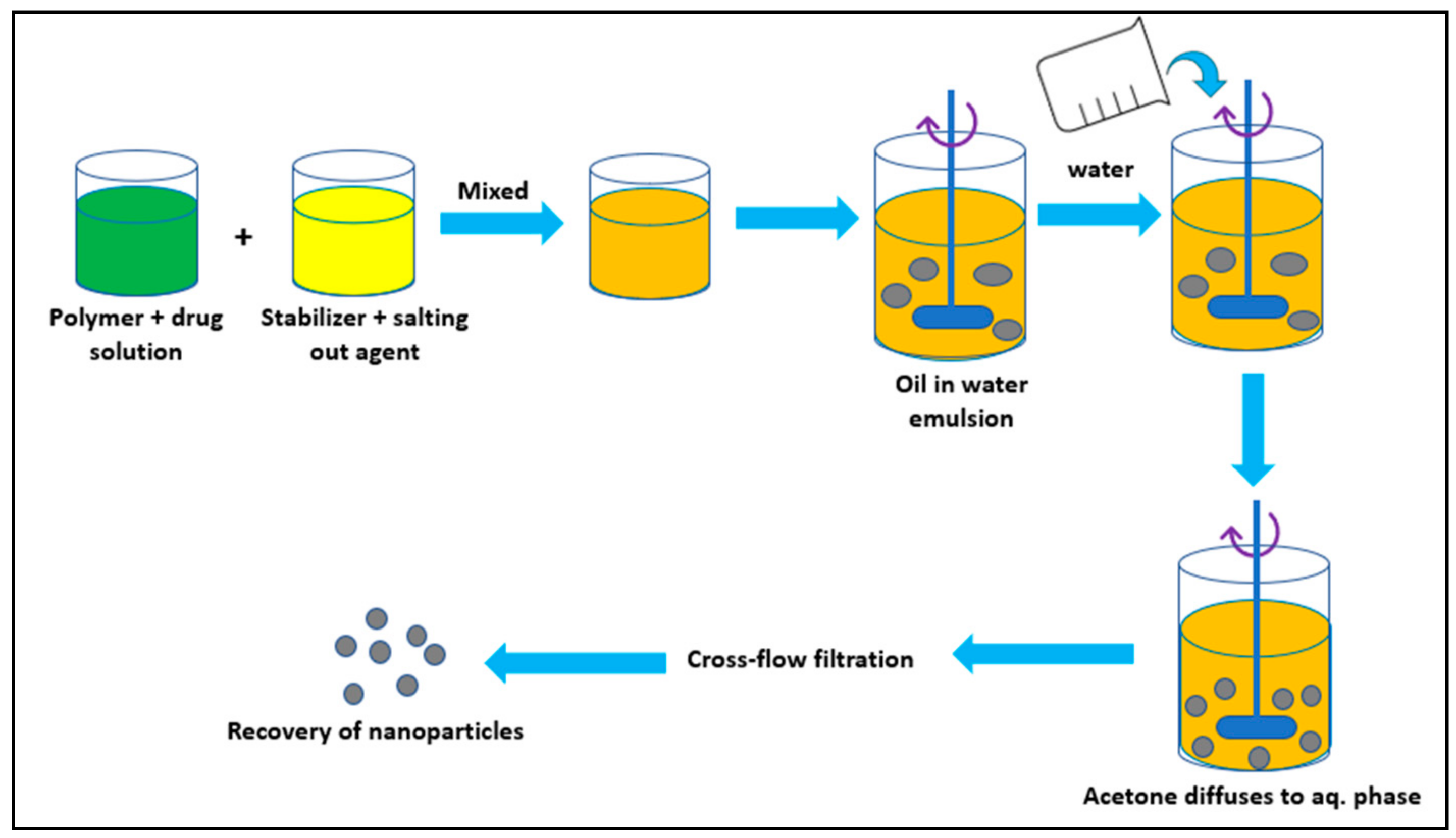
3.3. Salting Out Method
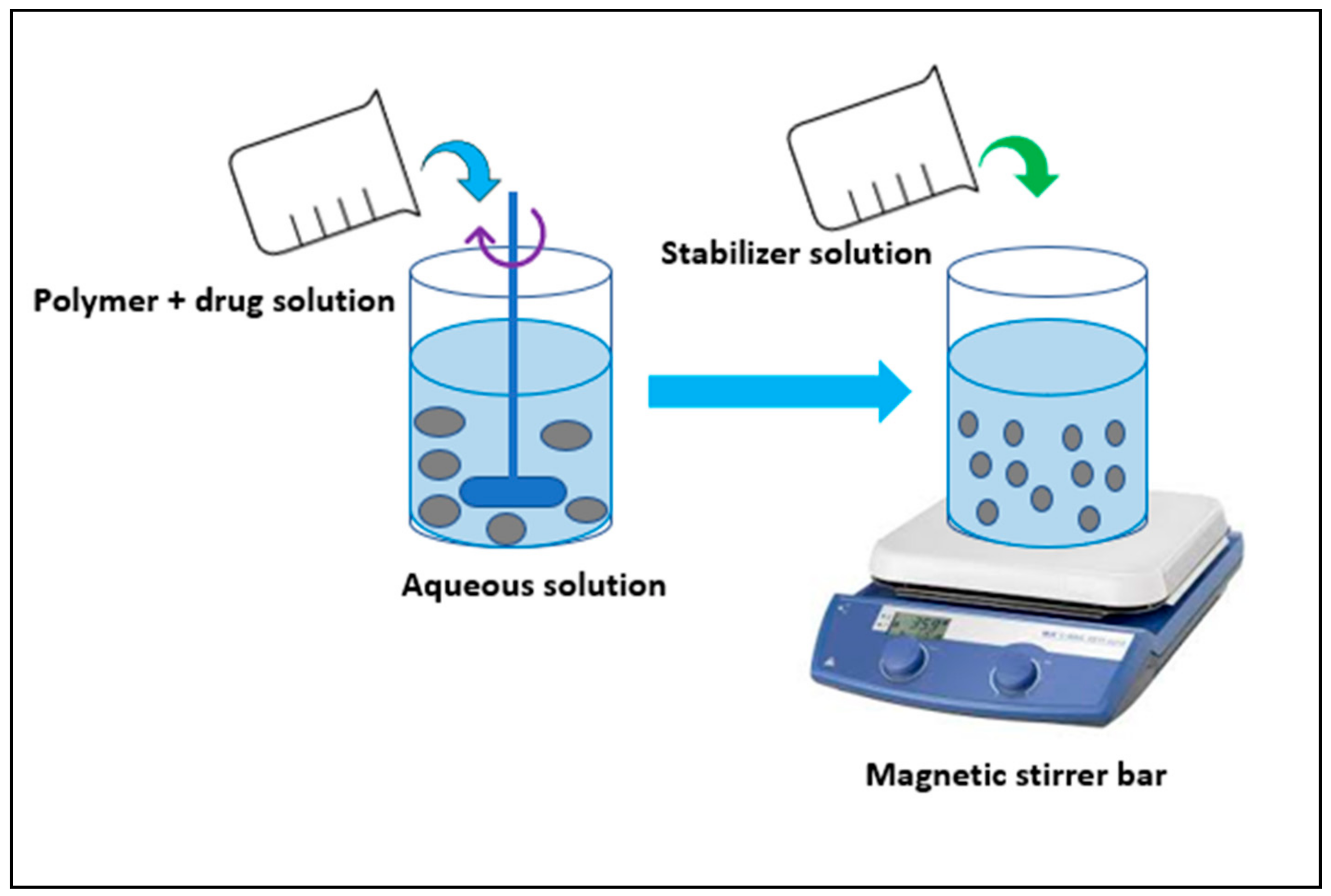
3.4. Nanoprecipitation/Solvent Displacement Method
3.5. Polymerization Methods
3.6. Nanoparticles Developed from Hydrophilic Polymers
4. Chemotherapy and Its Limitations
5. Polymeric Nanoparticles in Cancer Treatment
6. Poly-d,l-lactide-co-glycolide (PLGA)
6.1. Cisplatin
6.2. Curcumin
6.3. Docetaxel
6.4. 9-Nitrocamptothecin (9-NC)
6.5. Paclitaxel
6.6. Rose Bengal
7. Poly-ε-caprolactone (PCL)
7.1. Docetaxel
7.2. Vinblastine
7.3. Tamoxifen
7.4. Taxol
8. Gelatin
Paclitaxel
9. Poly-alkyl-cyano-acrylates (PAC)
Ftorafur
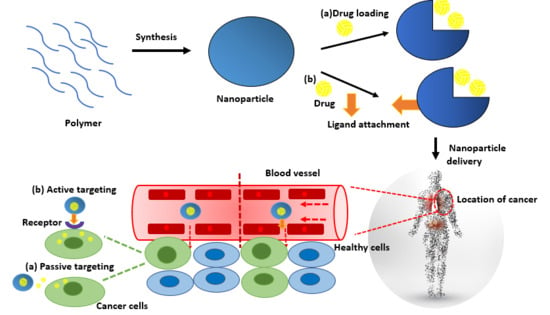
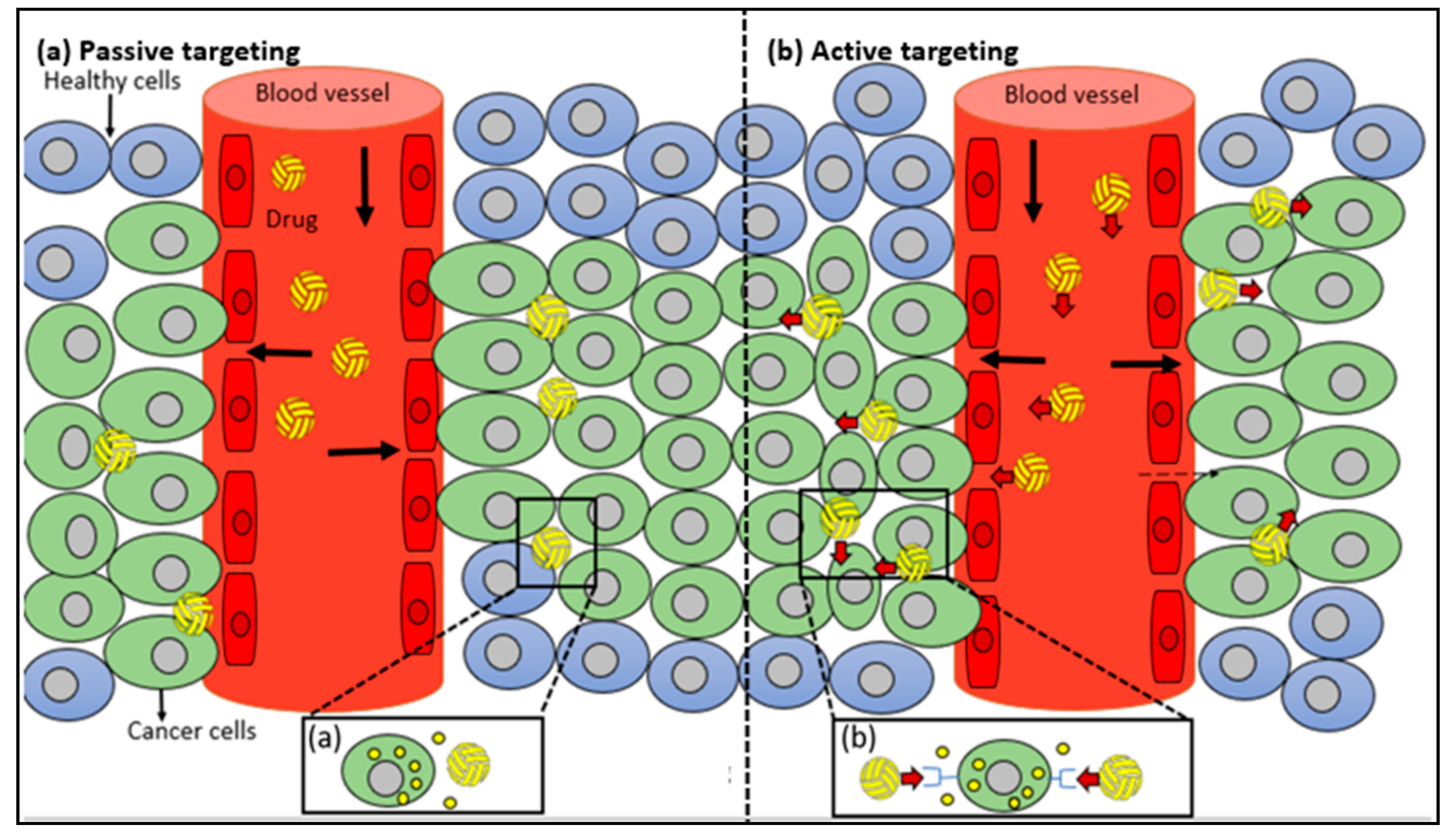
10. Targeted Delivery of Nanoparticles
- 1
- Reaching the site of action after crossing all the biological barriers while retaining their activity with minimum loss of volume.
- 2
- Attacking the tumor cells with minimal cytotoxic effect on the healthy neighboring cells or the tissues.
10.1. Passive Targeting
10.2. Active Targeting
- 1
- High concentration of drug can be transported to the site of action.
- 2
- The activity of the drug can be affected by conjugating ligand with the drug which is not the case in active targeting using ligand tagged nanocarriers.
- 3
- Numerous ligands can be attached on the surface of nanocarriers increasing chance to pass the target site.
11. Polymeric Nanoparticles Related Toxicity Issues
12. Conclusions and Outlook
Funding
Acknowledgments
Conflicts of Interest
References
- Safar, J.; Zarnegar, Z. Advanced drug delivery systems: Nanotechnology of health design A review. J. Saudi Chem. Soc. 2012, 18, 85–99. [Google Scholar] [CrossRef]
- Sanchez, F.; Sobolev, K. Nanotechnology in concrete—A review. Constr. Build. Mater. 2010, 24, 2060–2071. [Google Scholar] [CrossRef]
- Pacheco-Torgal, F.; Jalali, S. Nanotechnology: Advantages and drawbacks in the field of construction and building materials. Constr. Build. Mater. 2010, 25, 582–590. [Google Scholar] [CrossRef]
- Amin, M.; Abu el-Hassan, K. Effect of using different types of nano materials on mechanical properties of high strength concrete. Constr. Build. Mater. 2014, 80, 116–124. [Google Scholar] [CrossRef]
- Gutiérrez, H.R.; Kim, J.U.; Kim, P.J.; Eklund, C.P. Thermal Conversion of Bundled Carbon Nanotubes into Graphitic Ribbons. Nano Lett. 2005, 5, 2195–2201. [Google Scholar] [CrossRef] [PubMed]
- Muhd Norhasri, M.S.; Hamidah, M.S.; Mohd Fadzil, A. Applications of using nano material in concrete: A review. Constr. Build. Mater. 2016, 133, 91–97. [Google Scholar] [CrossRef]
- Cevc, G.; Vierl, U. Nanotechnology and the transdermal route: A state of the art review and critical appraisal. J. Control. Release 2010, 141, 277–299. [Google Scholar] [CrossRef] [PubMed]
- Ryman, B. Potential applications of Liposomes to therapy. Ann. N. Y. Acad. Sci. 1978, 308, 281–307. [Google Scholar] [CrossRef] [PubMed]
- Lasic, D.D.; Frederik, P.M.; Stuart, M.C.; Barenholz, Y.; McIntosh, T.J. Gelation of liposome interior. A novel method for drug encapsulation. FEBS Lett. 1992, 312, 255–258. [Google Scholar] [CrossRef]
- Bangham, A.D. Surrogate cells or trojan horses. The discovery of liposomes. BioEssays 1995, 17, 1081–1088. [Google Scholar] [CrossRef] [PubMed]
- Papahadjopoulos, D. Liposomes and their uses in biology and medicine. Ann. N. Y. Acad. Sci. 1978, 308, 1–441. [Google Scholar] [CrossRef]
- Markman, M. Pegylated liposomal doxorubicin in the treatment of cancers of the breast and ovary. Expert Opin. Pharmacother. 2016, 7, 1469–1474. [Google Scholar] [CrossRef] [PubMed]
- Rivera, E. Current status of liposomal anthracycline therapy in metastatic breast cancer. Clin. Breast Cancer 2003, 4, 76–83. [Google Scholar] [CrossRef]
- Rosenthal, E.; Poizot-Martin, I.; Saint-Marc, T.; Spano, J.P.; Cacoub, P. Phase IV study of liposomal daunorubicin (DaunoXome) in AIDS-related Kaposi sarcoma. Am. J. Clin. Nutr. 2002, 25, 57–59. [Google Scholar] [CrossRef]
- Cho, K.; Wang, X.; Nie, S.; Chen, Z.; Shin, D.M. Therapeutic nanoparticles for drug delivery in cancer. Clin. Cancer Res. 2008, 14, 1310–1316. [Google Scholar] [CrossRef] [PubMed]
- Kango, S.; Kalia, S.; Celli, A.; Njuguna, J.; Habibi, Y.; Kumar, R. Surface modification of inorganic nanoparticles for development of organic–inorganic nanocomposites—A review. Prog. Polym. Sci. 2013, 38, 1232–1261. [Google Scholar] [CrossRef]
- Kagan, V.E.; Tyurina, Y.Y.; Tyurin, V.A.; Konduru, N.V.; Potapovich, A.I.; Osipov, A.N.; Kisin, E.R.; Schwegler-Berry, D.; Mercer, R.; Castranova, V.; et al. Direct and indirect effects of single walled carbon nanotubes on RAW 264.7 macrophages: Role of iron. Toxicol. Lett. 2006, 165, 88–100. [Google Scholar] [CrossRef] [PubMed]
- Porter, A.E. Direct imaging of single-walled carbon nanotubes in cells. Nat. Nanotechnol. 2007, 2, 713–717. [Google Scholar] [CrossRef] [PubMed]
- Manna, S.K. Single-walled carbon nanotube induces oxidative stress and activates nuclear transcription factor-kappaB in human keratinocytes. Nanotechnol. Lett. 2005, 5, 1676–1684. [Google Scholar]
- Tomalia, D.A.; Naylor, M.A.; Goddord, A.W., III. Starburst Dendrimers: Molecular-Level Control of Size, Shape, Surface Chemistry, Topology, and Flexibility from Atoms to Macroscopic Matter. Angew. Chem. Int. 1990, 29, 138–175. [Google Scholar] [CrossRef]
- Faraji, A.H.; Wipf, P. Nanoparticles in cellular drug delivery. Bioorg. Med. Chem. 2009, 17, 2950–2962. [Google Scholar] [CrossRef] [PubMed]
- Svenson, S.; Tomalia, D.A. Dendrimers in biomedical applications-reflections on the field. Adv. Drug Deliv. Rev. 2005, 57, 2106–2129. [Google Scholar] [CrossRef] [PubMed]
- Nawaz, M.; Fatima, F. Extracellular Vesicles, Tunneling Nanotubes, and Cellular Interplay: Synergies and Missing Links. Front. Mol. Biosci. 2017, 4, 50. [Google Scholar] [CrossRef] [PubMed]
- Nawaz, M.; Camussi, G.; Valadi, H.; Nazarenko, I.; Ekstrom, K.; Wang, X.; Principe, S.; Shah, N.; Ashraf, N.M.; Fatima, F.; et al. The emerging role of extracellular vesicles as biomarkers for urogenital cancers. Nat. Rev. Urol. 2014, 11, 688–701. [Google Scholar] [CrossRef] [PubMed]
- Rustom, A.; Saffrich, R.; Markovic, I.; Walther, P.; Gerder, H.H. Nanotubular highways for intercellular organelle transport. Science 2004, 303, 1007–1010. [Google Scholar] [CrossRef] [PubMed]
- Austefjord, M.W.; Gerdes, H.H.; Wang, X. Tunneling nanotubes: Diversity in morphology and structure. Commun. Integr. Biol. 2014, 7, e27934. [Google Scholar] [CrossRef] [PubMed]
- Benard, M.; Schapman, D.; Lebon, A.; Monterroso, B.; Bellenger, M.; Le Foll, F.; Pasquier, J.; Vaudry, H.; VAudry, D.; Galas, L. Structural and functional analysis of tunneling nanotubes (TnTs) using gCW STED and gconfocal approaches. Biol. Cell 2015, 107, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Bonifácio, B.V.; da Silva, P.B.; dos Santos Ramos, M.A.; Silveira Negri, K.M.; Bauab, T.M.; Chorilli, M. Nanotechnology-based drug delivery systems and herbal medicines: A review. Int. J. Nanomed. 2014, 9, 1–15. [Google Scholar]
- Hu, L.; Han, S.; Parveen, S.; Yuan, Y.; Zhang, L.; Xu, G. Highly sensitive fluorescent detection of trypsin based on BSA-stabilized gold nanoclusters. Biosens. Bioelectron. 2012, 32, 297–299. [Google Scholar] [CrossRef] [PubMed]
- Zensi, A.; Begley, D.; Pontikis, C.; Legros, C.; Mihoreanu, L.; Wagner, S.; Büchel, C.; Kreuter, J. Albumin nanoparticles targeted with Apo E enter the CNS by transcytosis and are delivered to neurons. Off. J. Control. Release Soc. 2009, 137, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Discher, E.D.; Eisenberg, A. Polymer vesicles. Science 2002, 297, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Letchford, K.; Burt, H. A review of the formation and classification of amphiphilic block copolymer nanoparticulate structures: Micelles, nanospheres, nanocapsules and polymers. Eur. J. Pharm. Biopharm. 2007, 65, 259–269. [Google Scholar] [CrossRef] [PubMed]
- Vila, A.; Gill, H.; McCallion, O.; Alonso, M.J. Transport of PLA-PEG particles across the nasal mucosa: Effect of particle size and PEG coating density. J. Control. Release 2004, 98, 231–244. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.P.; Zeng, Q.H.; Lu, G.Q.; Yu, A.B. Inorganic nanoparticles as carriers for efficient cellular delivery. Chem. Eng. Sci. 2006, 61, 1027–1040. [Google Scholar] [CrossRef]
- Ensign, M.L.; Cone, R.; Hanes, J. Oral drug delivery with polymeric nanoparticles: The gastrointestinal mucus barriers. Adv. Drug Deliv. Rev. 2012, 64, 557–570. [Google Scholar] [CrossRef] [PubMed]
- Coulembier, O.; Degee, P.; Hedrick, L.J.; Dubois, P. From controlled ring-opening polymerization to biodegradable aliphatic polyester: Especially poly(beta-malic acid) derivatives. Prog. Polym. Sci. 2006, 31, 723–747. [Google Scholar] [CrossRef]
- Soppimath, S.K.; Aminabhavi, M.T.; Kulkarni, R.A.; Rudzinski, E.W. Biodegradable polymeric nanoparticles as drug delivery devices. J. Control. Release 2001, 70, 1–20. [Google Scholar] [CrossRef]
- Niwa, T.; Takeuchi, H.; Hino, T.; Kunou, N.; Kawashima, Y. Preparations of biodegradable nanospheres of water-soluble and insoluble drugs with d,l-lactide/glycolide copolymer by a novel spontaneous emulsification solvent diffusion method and the drug release behavior. J. Control. Release 1993, 25, 89–98. [Google Scholar] [CrossRef]
- Wehrle, P.; Magenheim, B.; Benita, S. The Influence of process parameters on the PLA nanoparticle size distribution evaluated by means of factorial design. Eur. J. Pharm. Biopharm. 1995, 41, 19–26. [Google Scholar]
- Murakami, H.; Yoshino, H.; Mizobe, M.; Kobayashi, M.; Takeuchi, H.; Kawashima, Y. Preparation of poly(d,l-lactide-co-glycolide) latex for surface modifying material by a double coacervation method. Proc. Int. Symp. Control. Release Bioact. Mater. 1996, 23, 361–362. [Google Scholar]
- Bodmeier, R.; McGinity, W. Solvent selection in the preparation of poly(d,l-lactide) microspheres prepared by the solvent evaporation method. Int. J. Pharm. 1998, 43, 179–186. [Google Scholar] [CrossRef]
- Guo, C.; Yang, C.; Li, Q.; Li, Q.-L.; Xi, Y.-W.; Liu, W.-N.; Zhai, G.-X. Development of a quercetin-loaded nanostructured lipid carrier formulation for topical delivery. Int. J. Pharm. 2012, 430, 292–298. [Google Scholar]
- Wu, T.; Yen, F.-L.; Lin, L.-T.; Tsai, T.-R.; Lin, C.-C.; Cham, T.-M. Preparation, physicochemical characterization, and antioxidant effects of quercetin nanoparticles. Int. J. Pharm. 2008, 346, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Min, K.H.; Park, K.; Kim, Y.-S.; Bae, S.M.; Lee, S.; Jo, H.G.; Park, R.-W.; Kim, I.-S.; Jeong, S.Y.; Kim, K.; et al. Hydrophobically modified glycol chitosan nanoparticles-encapsulated camptothecin enhance the drug stability and tumor targeting in cancer therapy. J. Control. Release 2008, 127, 208–218. [Google Scholar] [CrossRef] [PubMed]
- Yen, F.Y.; Wu, T.H.; Lin, L.T.; Cham, T.M.; Lin, C.C. Naringenin-loaded nanoparticles improve the physicochemical properties and the hepatoprotective effects of naringenin in orally-administered rats with CCl4-induced acute liver failure. Pharm. Res. 2009, 26, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Vauthier, C.; Dubernet, C.; Fattal, E.; Pinto-Alphandary, H.; Couvreur, P. Poly(alkylcyanoacrylates) as biodegradable materials for biomedical applications. Adv. Drug Deliv. Rev. 2003, 55, 519–548. [Google Scholar] [CrossRef]
- Calvo, P.; Sanchez, A.; Martinez, J.; Lopez, I.M.; Calonge, M.; Pastor, C.J.; Alonso, J.M. Polyester Nanocapsules as New Topical Ocular Delivery Systems for Cyclosporin A. Pharm. Res. 1996, 13, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Mao, H.Q.; Roy, K.; Troung-Le, V.L.; Janes, K.A.; Lin, K.Y.; Wang, Y.; August, J.T.; leong, K.W. Chitosan-DNA nanoparticles as gene carriers: Synthesis, characterization and transfection efficiency. J. Control. Release 2001, 70, 399–421. [Google Scholar] [CrossRef]
- Cooper, G.M. The Cell: A Molecular Approach, 2nd ed.; Sunderland (MA), Sinauer Associates: New York, NY, USA, 2000. [Google Scholar]
- Albert, B.; Johnson, A.; Lewis, J.; Raff, M.; Roberts, K.; Walter, P. Molecular Biology of the Cell, 4th ed.; Garland Science: New York, NY, USA, 2002. [Google Scholar]
- Cancer Facts & Figures; American Cancer Society. 2018. Available online: https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2017.html (accessed on 15 April 2018).
- Antoni, S.; Ferlay, J.; Soerjomataram, I.; Znaor, A.; Jemal, A.; Bray, F. Bladder cancer incidence and mortality: A global overview and recent trends. Eur. Urol. 2017, 71, 96–108. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.A.; von Eschenbach, A.C.; Wender, R.; Levin, B.; Byers, T.; Rothenberger, D.; Saslow, D. American Cancer Society guidelines for the early detection of cancer: Update of early detection guidelines for prostate, colorectal, and endometrial cancers: Also: Update 2001-Testing for early lung cancer detection. CA Cancer J. Clin. 2001, 51, 38–75. [Google Scholar] [CrossRef] [PubMed]
- Lane, B.R.; Canter, D.J.; Rini, B.I.; Uzzo, R.S.; Rosenber, S.A. Cancer: Principles and Practice of Oncology, 10th ed.; Pili, R., Kauffman, E., Rodriguez, R., Eds.; Ch 82-Cancer of the Kidney; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2015. [Google Scholar]
- National Cancer Institute (NCI). 2018. Available online: https://www.cancer.gov/types/skin (accessed on 15 April 2018).
- Centers for Disease Control and Prevention 2018. Available online: https://www.cdc.gov/cancer/prostate/index.htm (accessed on 15 April 2018).
- Feng, S.S.; Chein, S. Chemotherapeutic engineering: Application and further development of chemical engineering principles for chemotherapy of cancer and other diseases. Chem. Eng. Sci. 2003, 58, 4087–4114. [Google Scholar] [CrossRef]
- Feng, S.S. Interpretation of mechanochemical properties of lipid monolayers at the air-water or oil-water interface and bilayer vesicles from equation of state or pressure-area measurement. Langmuir 1999, 15, 998–1010. [Google Scholar] [CrossRef]
- Steichen, D.S.; Moore, C.M.; Peppas, A.N. A review of current nanoparticle and targeting moieties for the delivery of cancer therapeutics. Eur. J. Pharm. Sci. 2013, 14, 416–427. [Google Scholar] [CrossRef] [PubMed]
- Minotti, G.; Menna, P.; Salvatorelli, E.; Cairo, G.; Gianni, L. Anthracyclines: Molecular Advances and Pharmacologic Developments in Antitumor Activity and Cardiotoxicity. Pharmacol. Rev. 2004, 56, 185–229. [Google Scholar] [CrossRef] [PubMed]
- Weiss, L.M.; Jaffe, E.S.; Liu, X.F.; Chen, Y.Y.; Shibata, D.; Medeiros, L.J. Detection and localization of Epstein-Barr viral genomes in angioimmunoblastic lymphadenopathy and angioimmunoblastic lymphadenopathy-like lymphoma. Blood 1992, 79, 1789–1795. [Google Scholar] [PubMed]
- Rowinsky, E. The development and clinical utility of taxane class of antimicrotubule chemotherapy agents. Ann. Rev. 1997, 48, 353–374. [Google Scholar] [CrossRef] [PubMed]
- Boulikas, T.; Vougiouka, M. Cisplatin and platinum drugs at the molecular level. Oncol. Rev. 2003, 10, 1663–1682. [Google Scholar] [CrossRef]
- Hurley, H.L. DNA and its associated processes as targets for cancer therapy. Nat. Rev. Cancer 2002, 2, 188–200. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H. SMANCS and polymer-conjugated macromolecular drugs: Advantages in cancer chemotherapy. Adv. Drug Deliv. Rev. 2001, 46, 169–185. [Google Scholar] [CrossRef]
- Vauthier, C.; Dubernet, C.; Chauvierre, C.; Brigger, I.; Couvreur, P. Drug delivery to resistant tumors: The potential of poly(alkyl cyanoacrylate) nanoparticles. J. Control. Release 2009, 3, 151–160. [Google Scholar] [CrossRef]
- Miller, D.W.; Batrakova, E.V.; Kabanov, A.V. Inhibition of multidrug resistance-associated protein (MRP) functional activity with pluronic block copolymers. Pharm. Res. 1999, 16, 396–401. [Google Scholar] [CrossRef] [PubMed]
- Mamot, C.; Drummond, D.C.; Hong, K.; Kirpotin, D.B.; Park, J.W. Liposome-based approaches to overcome anticancer drug resistance. Drug Resist. Updates 2003, 6, 271–279. [Google Scholar] [CrossRef]
- Soma, C.E.; Dubernet, C.; Bentolila, D.; Benita, S.; Couvreur, P. Reversion of multidrug resistance by co-encapsulation of doxorubicin and cyclosporin A in polyalkylcyanoacrylate nanoparticles. Biomaterials 2000, 21, 1–7. [Google Scholar] [CrossRef]
- Bonduelle, S.; Foucher, C.; Leroux, J.-C.; Chouinard, F.; Caieux, C.; Lenaerts, V. Association of cyclosporin to isohexylcyanoacrylate nanospheres and subsequent release in human plasma in vitro. J. Microencapsul. 1992, 9, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Kumari, A.; Yadav, K.S.; Yadav, C.S. Biodegradable polymeric nanoparticles based drug delivery systems. Colloids Surf. B Biointerfaces 2010, 75, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Carrasquillo, K.G.; Stanley, A.M.; Aponte-Carro, J.C.; De Jésus, P.; Costantino, H.R.; Bosques, C.J.; Griebenow, K. Non-aqueous encapsulation of excipient-stabilized spray-freeze dried BSA into poly(lactide-co-glycolide) microspheres results in release of native protein. J. Control. Release 2001, 76, 199–208. [Google Scholar] [CrossRef]
- RaviKumar, M.N.; Mohapatra, S.S.; Kong, X.; Jena, P.K.; Bakowsky, U.; Lehrd, C.M. Cationic poly(lactide-co-glycolide) nanoparticles as efficient in vivo gene transfection agents. J. Nanosci. Nanotechnol. 2004, 4, 990–994. [Google Scholar] [CrossRef]
- Hedberg, E.L.; Kroese-Deutman, H.C.; Shih, C.K.; Crowther, R.S.; Carney, D.H.; Mikos, A.G.; Jansen, J.A. In vivo degradation of porous poly(propylenefumarate)/poly(dl-lactic-co-glycolic acid) composite scaffolds. Biomaterials 2005, 26, 4616–4623. [Google Scholar] [CrossRef] [PubMed]
- Patil, D.S.; Papadimitrakopoulos, F.; Burgess, J.D. Dexamethasone-loaded poly(lactic-co-glycolic) acid microspheres/poly(vinyl alcohol) hydrogel composite coatings for inflammation control. Diabetes Technol. Ther. 2004, 6, 887–897. [Google Scholar] [CrossRef] [PubMed]
- Avgoustakis, K.; Beletsi, A.; Panagi, Z.; Lepetsanis, K.P.; Karydas, G.A.; Lthakissios, S.D. PLGA-mPEG nanoparticles of cisplatin: In vitro nanoparticle degradation, in vitro drug release and in vivo drug residence in blood properties. J. Control. Release 2002, 79, 123–135. [Google Scholar] [CrossRef]
- Ranjan, P.A.; Mukherjee, A.; Helson, L.; Vishwanantha, K.J. Scale up, optimization and stability analysis of Curcumin C3 complex-loaded nanoparticles for cancer therapy. J. Nanobiotechnol. 2012, 10, 38. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili, F.; Ghahremani, M.H.; Ostad, S.N.; Atyabi, F.; Seyedabadi, M.; Malekshahi, M.R.; Amini, M.; Dinarvand, R. Folate-receptor-targeted delivery of docetaxel nanoparticles prepared by PLGA–PEG–folate conjugate. J. Drug Target 2008, 16, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Redhead, M.H.; Davis, S.S.; Illum, L. Drug delivery in poly(lactide-co-glycolide) nanoparticles surface modified with poloxamer 407 and poloxamine 908: In vitro characterisation and in vivo evaluation. J. Control. Release 2001, 70, 353–363. [Google Scholar] [CrossRef]
- Zheng, D.; Li, X.; Xu, H.; Lu, X.; Hu, Y.; Fan, W. Study on docetaxel-loaded nanoparticles with high antitumor efficacy against malignant melanoma. Acta Biochim. Biophys. Sin. 2009, 41, 578–587. [Google Scholar] [CrossRef] [PubMed]
- Prabu, P.; Chaudhari, A.A.; Dharmaraj, N.; Khil, M.S.; Park, S.Y.; Kim, H.Y. Preparation, characterization, in-vitro drug release and cellular uptake of poly(caprolactone) grafted dextran copolymeric nanoparticles loaded with anticancer drug. J. Biomed. Mater. Res. 2008, 90, 1128–1136. [Google Scholar]
- Shenoy, B.D.; Amiji, M.M. Poly(ethylene oxide)-modified poly(epsiloncaprolactone) nanoparticles for targeted delivery of tamoxifen in breast cancer. Int. J. Pharm. 2005, 293, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Wang, Y.N.; Fan, Y.G.; Ma, J.B. Synthesis of a biodegradable tadpole-shaped polymer via the coupling reaction of polylactide onto mono(6-(2-aminoethyl)amino-6-deoxy)-beta-cyclodextrin and its properties as the new carrier of protein delivery system. J. Control. Release 2005, 107, 158–173. [Google Scholar] [CrossRef] [PubMed]
- Zambaux, M.F.; Bonneaux, F.; Gref, R.; Dellacherie, E.; Vigneron, C. Preparation and characterization of protein C-loaded PLA nanoparticles. J. Control. Release 1999, 60, 179–188. [Google Scholar] [CrossRef]
- Oppenhiem, C.R. Paclitaxel loaded gelatin nanoparticles for intravesical bladder cancer therapy. Int. J. Pharm. 1981, 8, 217. [Google Scholar]
- Lu, Z.; Yeh, T.K.; Tsai, M.; Au, J.L.; Wientjes, M.G. Paclitaxel-loaded gelatin nanoparticles for intravesical bladder cancer therapy. Clin. Cancer Res. 2004, 10, 7677–7684. [Google Scholar] [CrossRef] [PubMed]
- Reis, P.C.; Neufeld, R.J.; Ribeiro, A.J.; Veiga, F. Nanoencapsulation I. Methods for preparation of drug loaded polymeric nanoparticles. Nanomedicine 2006, 2, 8–21. [Google Scholar] [CrossRef] [PubMed]
- Davis, S. Drug delivery systems. Interdiscip. Sci. Rev. 2000, 25, 175–183. [Google Scholar] [CrossRef]
- Arias, J.L.; Gallardo, V.; Ruiz, M.A.; Delgado, A.V. Ftorafur loading and controlled release from poly(ethyl-2-cyanoacrylate) and poly(butylcyanoacrylate) nanospheres. Int. J. Pharm. 2007, 337, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Carmeliet, P.; Jain, R.K. Angiogenesis in cancer and other diseases. Nature 2000, 40, 7249–7257. [Google Scholar] [CrossRef] [PubMed]
- Yatvin, M.B.; Kreutz, W.; Horwitz, B.A.; Shinitzky, M. pH-sensitive liposomes: Possible clinical implications. Science 1980, 210, 1253–1255. [Google Scholar] [CrossRef] [PubMed]
- Bianco, A.; Kostarelos, K.; Partidos, C.D.; Prato, M. Biomedical applications of functionalised carbon nanotubes. Chem. Commun. 2005, 5, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Farokhzad, C.O.; Langer, R. Impact of Nanotechnology on Drug Delivery. ACS Nano 2009, 3, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Farokhzad, O.C.; Karp, J.M.; Langer, R. Nanoparticle-Aptamer Bioconjugates for Cancer Targeting. Expert Opin. Drug Deliv. 2006, 3, 311–324. [Google Scholar] [CrossRef] [PubMed]
- Koo, O.M.; Rubinstein, I.; Onyksel, H. Role of nanotechnology in targeted drug delivery and imaging: A concise review. Nanomed. Nanotechnol. Biol. Med. 2005, 1, 193–212. [Google Scholar] [CrossRef] [PubMed]
- Missailidis, S.; Thomaidou, D.; Borbas, K.E.; Price, M.R. Selection of aptamers with high affinity and high specificity against C595, an anti-MUC1 IgG3 monoclonal antibody, for antibody targeting. J. Immunol. Methods 2005, 296, 45–62. [Google Scholar] [CrossRef] [PubMed]
- Farokhzad, O.C.; Jon, S.; Khademhosseini, A.; Tran, T.N.; Lavan, D.A.; Langer, R. Nanoparticle-Aptamer Bioconjugates. Cancer Res. 2004, 64, 7668–7672. [Google Scholar] [CrossRef] [PubMed]
- Gabizon, A.; Horowitz, T.A.; Goren, D.; Tzemach, D.; Shmeeda, H.; Zalipsky, S. In Vivo Fate of Folate-Targeted Polyethylene-Glycol Liposomes in Tumor-Bearing Mice. Clin. Cancer Res. 2003, 9, 6551–6559. [Google Scholar] [PubMed]
- Medina, O.P.; Kairemo, K.; Valtanen, H.; Kangasniemi, A.; Kaukinen, S.; Ahonen, I.; Permi, P.; Annila, A.; Sneck, M.; Holopainen, M.J.; et al. Radionuclide Imaging of Tumor Xenografts in Mice Using a Gelatinase-targeting Peptide. Anticancer Res. 2005, 25, 33–42. [Google Scholar] [PubMed]
- Dharap, S.S. Tumor-specific targeting of an anticancer drug delivery system by LHRH peptide. Proc. Natl. Acad. Sci. USA 2003, 102, 12962–12967. [Google Scholar] [CrossRef] [PubMed]
- Mitra, A.; Mulholland, J.; Nan, A.; McNeil, E.; Ghandehari, H.; Line, R.B. Targeting tumor angiogenic vasculature using polymer–RGD conjugates. J. Control. Release 2005, 102, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Temming, K.; Schiffelers, M.R.; Molema, G.; Kok, J.R. RGD-based strategies for selective delivery of therapeutics and imaging agents to the tumour vasculature. Drug Resist. Updates 2005, 8, 381–402. [Google Scholar] [CrossRef] [PubMed]
- Adabi, M. Biocompatibility and nanostructured materials: Applications in nanomedicine. Artif. Cells Nanomed. Biotechnol. 2016, 45, 833–842. [Google Scholar] [CrossRef] [PubMed]
- Naahidi, S.; Mousa, J.; Faramarz, E.; Kevin, R.; Khademhosseini, A.; Chen, P. Biocompatibility of engineered nanoparticles for drug delivery. J. Control. Release 2013, 166, 182–194. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Singh, H.N. Nanotechnology and Health Safety-Toxicity and Risk Assessments of Nanostructured Materials on Human Health. J. Nanosci. Nanotechnol. 2007, 23, 3048–3070. [Google Scholar] [CrossRef]
- Li, X.; Wang, L.; Fan, Y.; Feng, Q.; Cui, F.Z. Biocompatibility and toxicity of nanoparticles and nanotubes. J. Nanomater. 2012, 2012, 6. [Google Scholar] [CrossRef]
- Brocchini, S.; Duncan, R. Pendent drugs, release from polymers. Encycl. Control. Drug Deliv. 1999, 2, 786–816. [Google Scholar]
- Shiah, J.G.; Sun, Y.; Kopečková, P.; Peterson, C.M.; Straight, R.C.; Kopeček, J. Combination chemotherapy and photodynamic therapy of targetable N-(2-hydroxypropyl) methacrylamide copolymer–doxorubicin/mesochlorin e6-OV-TL 16 antibody immunoconjugates. J. Control. Release 2001, 74, 249–253. [Google Scholar] [CrossRef]
- Cardoso, P.B.; Machado, T.O.; Feuser, P.E.; Sayer, C.; Meier, M.A.; Araujo, P.H. Biocompatible Polymeric Nanoparticles from Castor Oil Derivatives via Thiol-Ene Miniemulsion Polymerization. Eur. J. Lipid Sci. Technol. 2018, 120, 1700212. [Google Scholar] [CrossRef]
Sample Availability: Since this is a review, no samples are available. |
| Synthetic Biodegradable Polymers | Natural Biodegradable Polymers | ||
|---|---|---|---|
| Polyesters | Polyoxalates | Starch | Chitosan |
| Polyorthoesters | Polyiminocarbonates | Hyaluronic acid | Dextran |
| Polyanhydrides | Polyurethanes | Heparin | |
| Polydioxanones | Polyphosphazenes | Gelatin | |
| Poly(a-cyanoacrylates) | Albumin | ||
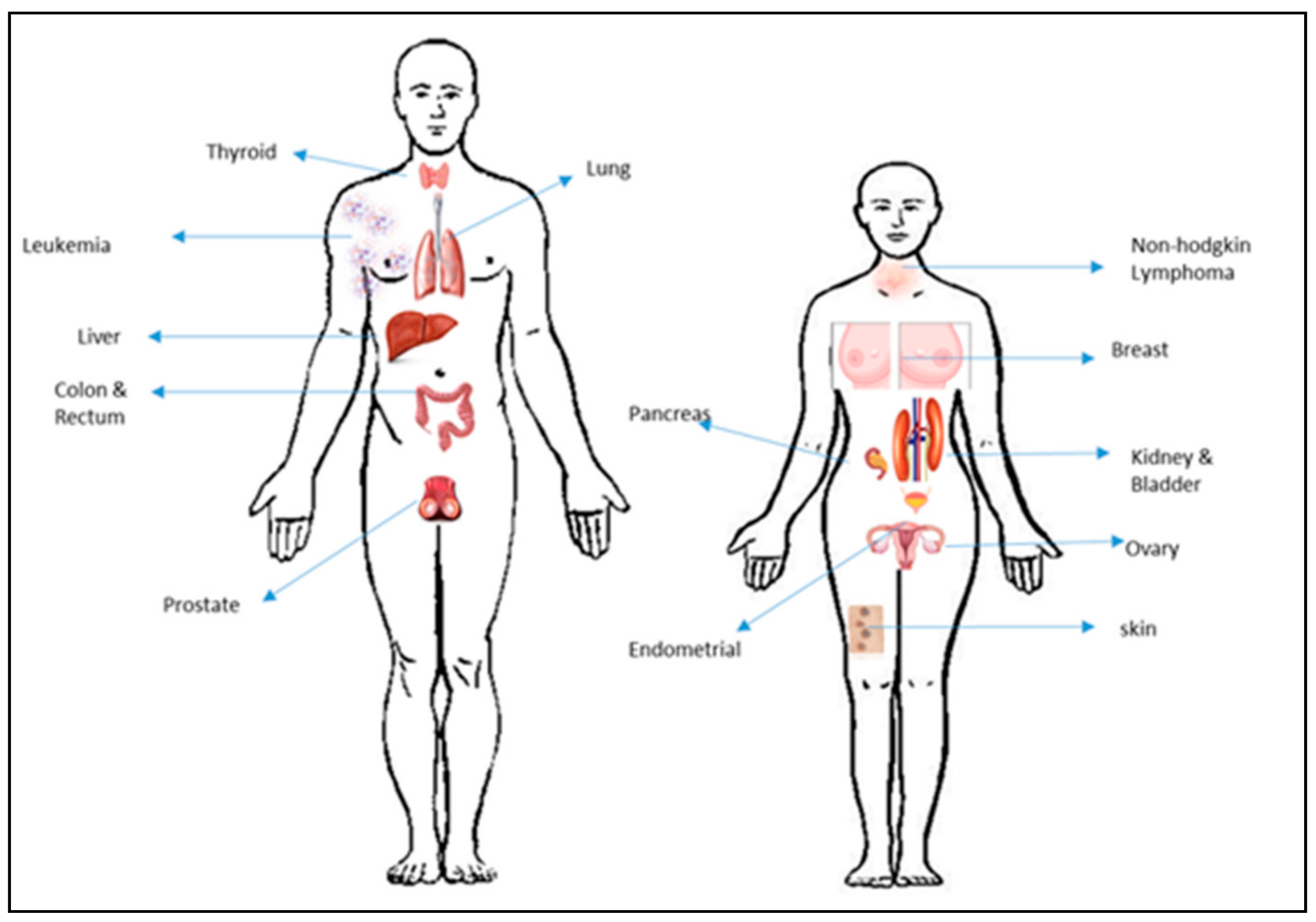
| Most Prevalent Cancers | Cause | Most Common Type | Estimated Death | References |
|---|---|---|---|---|
| Bladder | Smoking, Exposition to certain chemicals, chronic bladder infections, Abnormal cell growth in the muscular sac that stores urine, urothelium, infection with Schistosoma haematobium | Urothelial carcinoma, squamous cell carcinoma, adenocarcinoma, Superficial bladder cancer, Invasive bladder cancer | 17,240 | [51,52] |
| Breast (Men and Female) | Malignant tumor in the breast, gene mutation, family history | Ductal carcinoma in situ (DCIS), Invasive Ductal Carcinoma (IDC), Mammogram, Lumpectomy, Mastectomy | 268,670 | [51] |
| Colon and Rectal | Genetic mutation, an inherited or acquire mutation to the APC gene. | More than 95% of colon cancer can be classified as adenocarcinomas. | 50,630 | [51] |
| Endometrial | Increasing age, unopposed estrogen therapy, late menopause, tamoxifen therapy, nulliparity, infertility or failure to ovulate, obesity, hypertension, diabetes, and HNPCC. | Adenocarcinoma, Carcinosarcoma, Squamous cell carcinoma, Undifferentiated carcinoma, Small cell carcinoma, Transitional carcinoma | 11,350 | [53] |
| Kidney (Renal Cell and Renal Pelvis) | Smoking, obesity, Workplace exposures, Family history of kidney cancer, High blood pressure, Certain medicines | Renal cell carcinoma (RCC), Clear cell renal cell carcinoma, Papillary renal cell carcinoma, Chromophobe renal cell carcinoma, transitional cell carcinomas, Wilms tumors, and renal sarcomas. | 14,970 | [51,54] |
| Leukemia (All Types) | DNA of immature blood cells, mainly white cells, becomes damaged. | Acute lymphoblastic leukemia, Acute myeloid leukemia, Chronic lymphocytic leukemia (CLL), Chronic myeloid leukemia (CML), Chronic myelomonocytic leukemia (CMML), Leukemia in children | 24,370 | [51] |
| Liver and Intrahepatic Bile Duct | Alcohol, age, smoking, genetic, hepatitis, obesity, cirrhosis, gender | Hepatocellular carcinoma, Hepatoblastoma, Hepatocellular carcinoma | 30,200 | [51] |
| Lung (Including Bronchus) | Smoking tobacco, second hand smoke, genetic undergoing radiation therapy and environmental exposure | Small Cell Lung Cancer (SCLC) Non-Small Cell Lung Cancer (NSCLC) | 154,050 | [51,55] |
| Skin | Exposure to Ultraviolet (UV) light, tanning beds or sunlamps. | Melanoma, Basal, Squamous and Merkel Cell Carcinoma, Epidermoid cysts | 9320 | [51,55] |
| Non-Hodgkin Lymphoma | Infection-fighting cells of the immune system, called lymphocytes, immune deficiency. | Hodgkin lymphoma and non-Hodgkin lymphoma (NHL), Skin lymphoma, pediatric lymphoma, AIDS-related lymphoma, Waldenstrom macroglobulinemia (WM) | 19,910 | [51] |
| Pancreatic | DNA mutations, Diabetes, smoking, pancreatitis, smoking, Obesity | Exocrine cancers, Pancreatic adenocarcinoma, endocrine | 44,330 | [51] |
| Prostate | Oncogenes change or mutation in the DNA, Age, growth of abnormal cells, which may invader healthy cells in the body. | Acinar, ductal adenocarcinoma, ductal, urothelial, squamous cell cancer, Small all prostate cancer | 29,430 | [56] |
| Thyroid | Radiation, low iodine consumptions, family history, gender, age, hereditary conditions, DNA mutations | papillary, follicular, medullary, and anaplastic thyroid cancer. | 2060 | [51] |
| Targeting Ligands | Targets | References |
|---|---|---|
| Aptamers | Antibodies, cell surface receptors, enzymes, small organic molecules, peptides, proteins | [96,97] |
| Folate | Folate receptor | [98] |
| Gelatinase inhibitor peptide CTTHWGFTLC | Matrix Metalloprotease-2 and Matrix Metalloprotease-9 gelatinase | [99] |
| Luteinizing hormone-releasing hormone | Luteinizing hormone-releasing hormone receptor | [100,101] |
| RGD peptide | Integrin | [102] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahlawat, J.; Henriquez, G.; Narayan, M. Enhancing the Delivery of Chemotherapeutics: Role of Biodegradable Polymeric Nanoparticles. Molecules 2018, 23, 2157. https://doi.org/10.3390/molecules23092157
Ahlawat J, Henriquez G, Narayan M. Enhancing the Delivery of Chemotherapeutics: Role of Biodegradable Polymeric Nanoparticles. Molecules. 2018; 23(9):2157. https://doi.org/10.3390/molecules23092157
Chicago/Turabian StyleAhlawat, Jyoti, Gabriela Henriquez, and Mahesh Narayan. 2018. "Enhancing the Delivery of Chemotherapeutics: Role of Biodegradable Polymeric Nanoparticles" Molecules 23, no. 9: 2157. https://doi.org/10.3390/molecules23092157
APA StyleAhlawat, J., Henriquez, G., & Narayan, M. (2018). Enhancing the Delivery of Chemotherapeutics: Role of Biodegradable Polymeric Nanoparticles. Molecules, 23(9), 2157. https://doi.org/10.3390/molecules23092157