Inhibitory Effects of Bioassay-Guided Isolation of Anti-Glycation Components from Taraxacum coreanum and Simultaneous Quantification
Abstract
:1. Introduction
2. Results and Discussion
2.1. AGE Formation in BSA-glucose and BSA-MGO Systems, and α-Glucosidase Inhibitory Activities of the Extracts and Fractions from T. coreanum
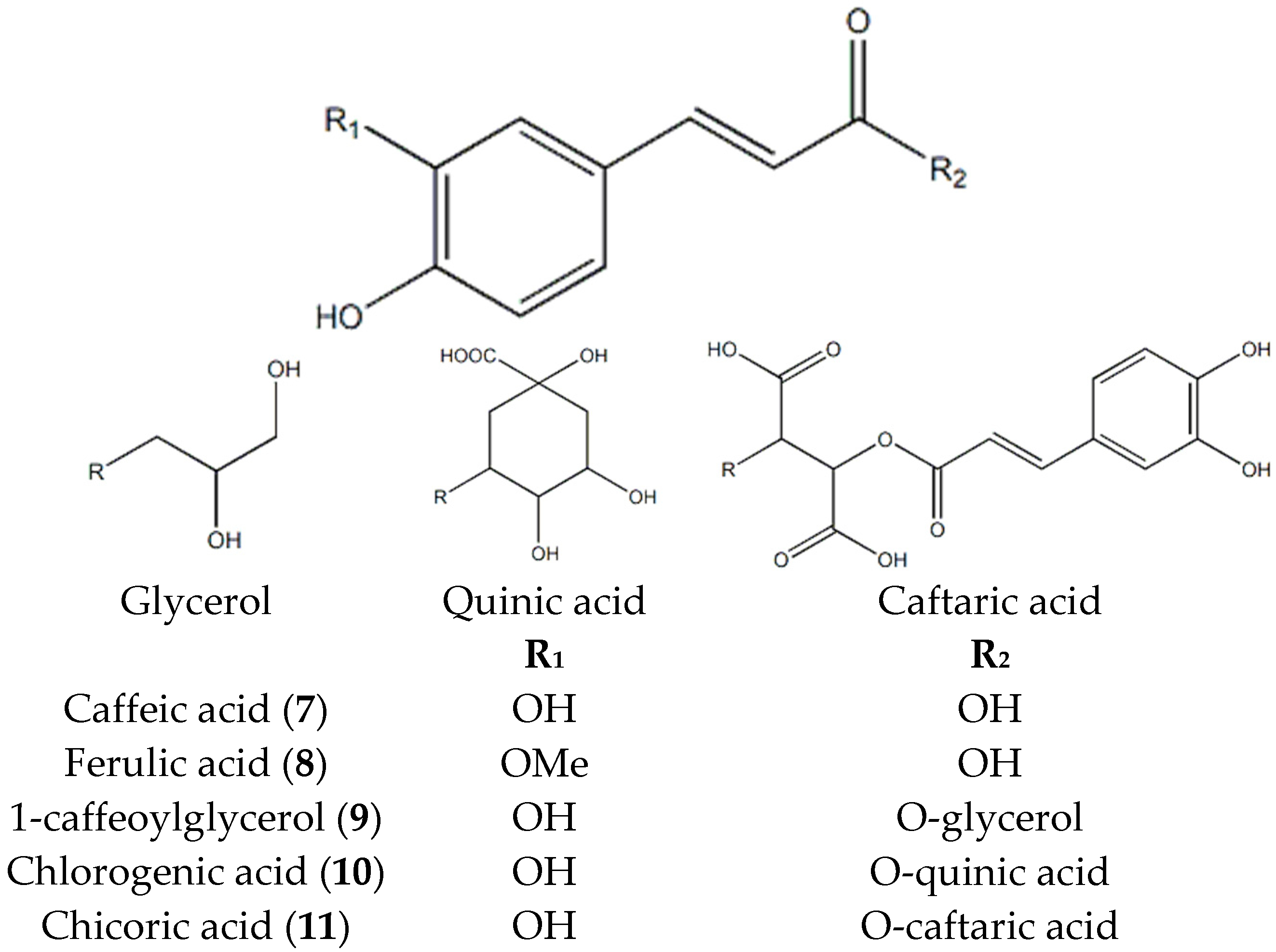
2.2. Identification of Compounds 1–11 Isolated from T. coreanum
2.3. AGE Formation in BSA-glucose and BSA-MGO Systems and α-Glucosidase Inhibitory Activities of Compound 1–11 Isolated from T. coreanum
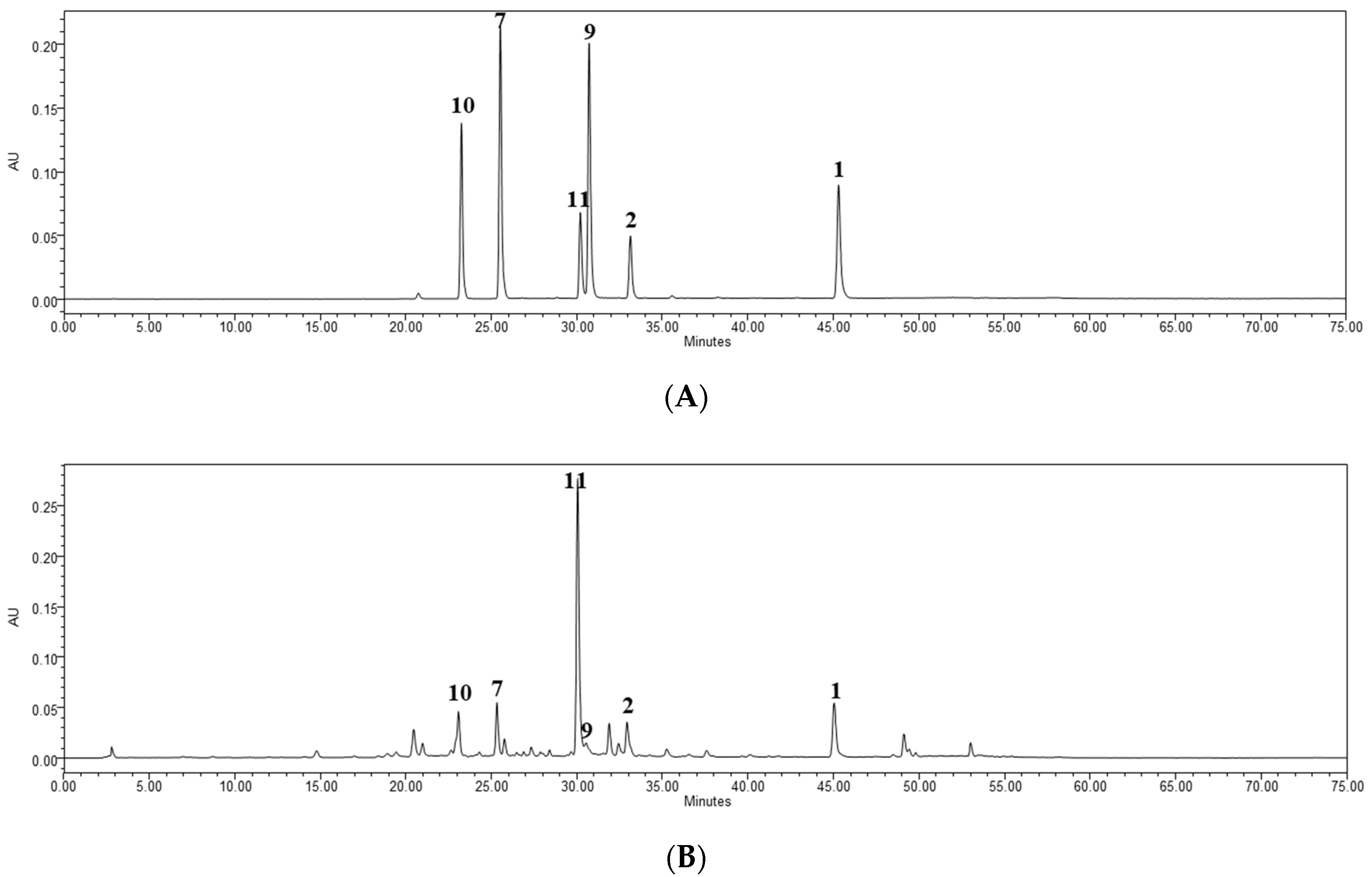
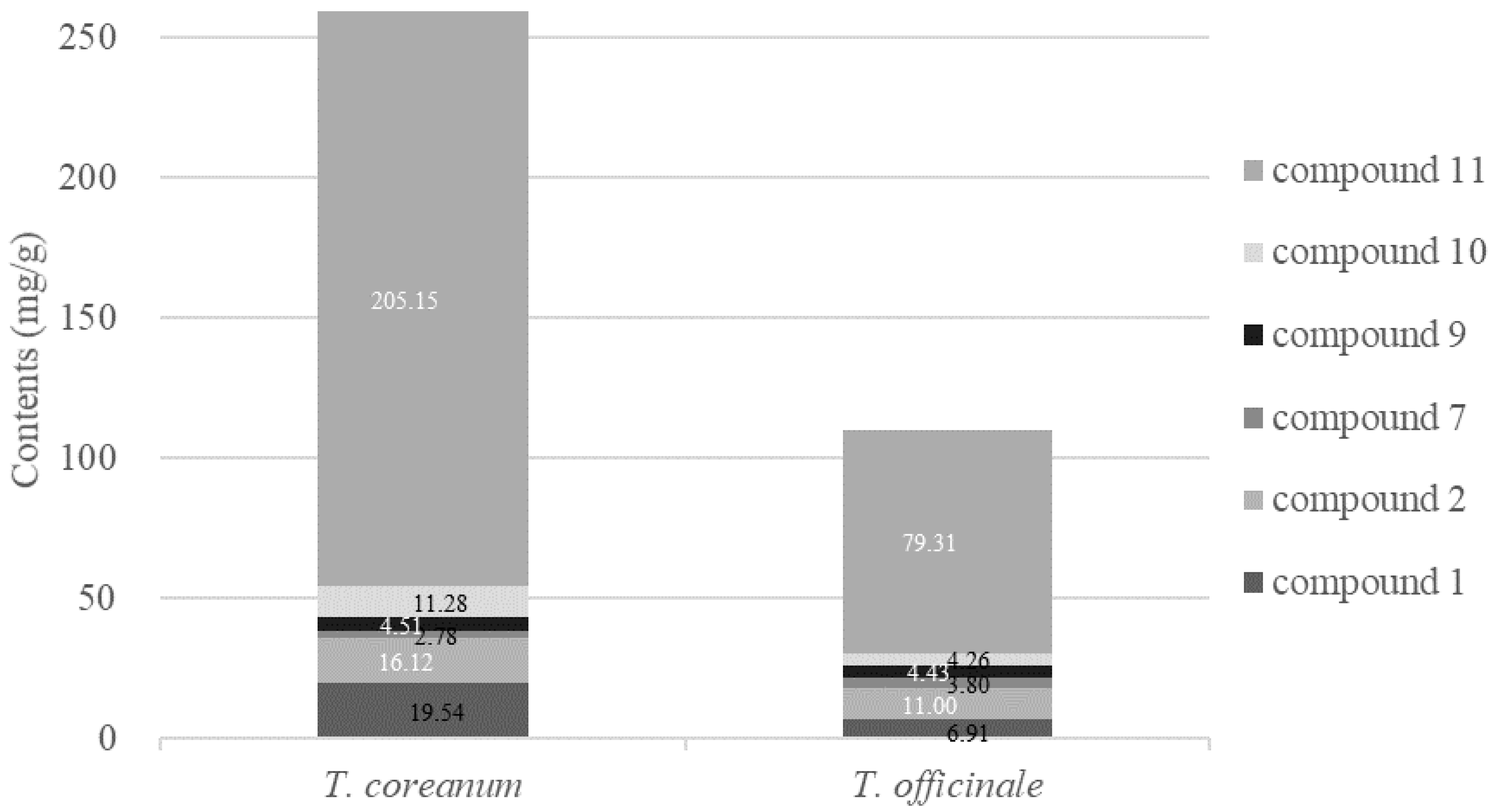
2.4. Quantitative HPLC Analysis of Six Bioactive Compounds
3. Materials and Methods
3.1. Plant Materials
3.2. Instruments and Reagents
3.3. Extraction, Fractionation, and Isolation from T. coreanum
3.4. Identification of Compounds Isolated from T. coreanum
3.5. UHPLC-ESI/LTQ-Orbitrap-HRMS Conditions
3.6. Measurement of BSA-glucose and Fructose Inhibitory Activity
3.7. Measurement of BSA-MGO Inhibitory Activity
3.8. Measurement α-Glucosidase Inhibitory Activity
3.9. Statistical Analysis
3.10. HPLC Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ho, S.-C.; Chang, P.-W.; Tong, H.-T.; Yu, P.-Y. Inhibition of fluorescent advanced glycation end-products and N-Carboxymethyllysine formation by several floral herbal infusions. Int. J. Food Prop. 2013, 17, 617–628. [Google Scholar] [CrossRef]
- Sharma, C.; Kaur, A.; Thind, S.S.; Singh, B.; Raina, S. Advanced glycation End-products (AGEs): An emerging concern for processed food industries. J. Food Sci. Technol. 2015, 52, 7561–7576. [Google Scholar] [CrossRef] [PubMed]
- Semba, R.D.; Nicklett, E.J.; Ferrucci, L. Does accumulation of advanced glycation end products contribute to the aging phenotype? J. Gerontol. A Biol. Sci. Med. Sci. 2010, 65A, 963–975. [Google Scholar] [CrossRef] [PubMed]
- Makita, Z.; Radoff, S.; Rayfield, E.J.; Yang, Z.; Skolnik, E.; Delaney, V.; Friedman, E.A.; Cerami, A.; Vlassara, H. Advanced glycosylation end products in patients with diabetic nephropathy. N. Engl. J. Med. 1991, 325, 836–842. [Google Scholar] [CrossRef] [PubMed]
- Valente, T.; Gella, A.; Fernàndez-Busquets, X.; Unzeta, M.; Durany, N. Immunohistochemical analysis of human brain suggests pathological synergism of Alzheimer′s disease and diabetes mellitus. Neurobiol. Dis. 2010, 37, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Van Heerebeek, L.; Hamdani, N.; Handoko, M.L.; Falcao-Pires, I.; Musters, R.J.; Kupreishvili, K.; Ijsselmuiden, A.J.; Schalkwijk, C.G.; Bronzwaer, J.G.; Diamant, M.; et al. Diastolic stiffness of the failing diabetic heart: Importance of fibrosis, advanced glycation end products, and myocyte resting tension. Circulation 2008, 117, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Cristina, D.-A. Carboxymethyl-lysine: Thirty years of investigation in the field of AGE formation. Food Funct. 2016, 7, 46–57. [Google Scholar]
- Uribarri, J.; Del Castillo, M.D.; de la Maza, M.P.; Filip, R.; Gugliucci, A.; Luevano-Contreras, C.; Macías-Cervantes, M.H.; Bastos, D.H.M.; Medrano, A.; Menini, T.; et al. Dietary advanced glycation end products and their role in health and disease. Adv. Nutr. 2015, 6, 461–473. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-H.; Huang, S.-M.; Lin, J.-A.; Yen, G.-C. Inhibition of advanced glycation endproduct formation by foodstuffs. Food Funct. 2011, 2, 224–234. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zheng, T.; Sang, S.; Lv, L. Quercetin inhibits advanced glycation end product formation by trapping methylglyoxal and glyoxal. J. Agric. Food Chem. 2014, 62, 12152–12158. [Google Scholar] [CrossRef] [PubMed]
- Kirchner, A. Der gemeine Lowenzahn, Taraxacum officinale Web. Der Versuch einer monographie in landwirtschaftlicher betrachtung. Zeitschrift fur Acker-und Pflanzenbau. 1955, 99, 488–518. [Google Scholar]
- Faber, K. Dandelion-Taraxacum officinale Weber. Pharmazie 1958, 13, 423–436. [Google Scholar] [PubMed]
- Lee, S.; Choi, M.J.; Choi, J.M.; Lee, S.; Kim, H.Y.; Cho, E.J. Flavonoids from Taraxacum coreanum protect from radical-induced oxidative damage. J. Med. Plant. Res. 2012, 6, 5377–5384. [Google Scholar]
- Lee, M.-H.; Kang, H.; Lee, K.; Yang, G.; Ham, I.; Bu, Y.; Kim, H.; Choi, H.-Y. The aerial part of Taraxacum coreanum extract has an anti-inflammatory effect on peritoneal macrophages in vitro and increases survival in a mouse model of septic shock. J. Ethnopharmacol. 2013, 146, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Paul, N.C.; Kim, W.K.; Woo, S.K.; Park, M.S.; Yu, S.H. Diversity of endophytic fungi associated with Taraxacum coreanum and their antifungal activity. Mycobiology 2006, 34, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.-H.; Shin, K.-M.; Kim, S.-Y.; Hong, J.-P.; Lee, Y.S.; Kim, H.J.; Park, H.-J.; Lee, K.-T. Taraxinic Acid, a hydrolysate of sesquiterpene lactone glycoside from the Taraxacum coreanum NAKAI, induces the differentiation of human acute promyelocytic leukemia HL-60 Cells. Biol. Pharm. Bull. 2002, 25, 1446–1450. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Han, S.; Kim, H.M.; Lee, J.M.; Mok, S.-Y.; Lee, S. Isolation and identification of phytochemical constituents from Taraxacum coreanum. J. Korean Soc. Appl. Biol. Chem. 2011, 54, 73–78. [Google Scholar] [CrossRef]
- Im, D.Y.; Lee, K.I. Nitric oxide production inhibitory and scavenging activity and tyrosinase inhibitory activity of extracts from Taraxacum officinale and Taraxacum coreanum. Korean J. Med. Crop. Sci. 2011, 19, 362–367. [Google Scholar] [CrossRef]
- Lee, H.H.; Lee, S.Y. Cytotoxic and antioxidant effects of Taraxacum coreanum Nakai. and T. officinale WEB. extracts. Korean J. Med. Crop. Sci. 2008, 16, 79–85. [Google Scholar]
- Oh, C.H.; Shin, N.S.; Kwan, J.; Lee, K.G.; Lee, S.R.; Lee, C.H. Antidiabetic Effects of water extract from Taraxacum coreanum Nakai in type II diabetic mice. Korean J. Orient. Physiol. Pathol. 2012, 26, 707–713. [Google Scholar]
- Szawara-Nowak, D.; Koutsidis, G.; Wiczkowski, W.; Zieliñski, H. Evaluation of the in vitro inhibitory effects of buckwheat enhanced wheat bread extracts on the formation of advanced glycation end-products (AGEs). LWT-Food Sci. Technol. 2014, 58, 327–334. [Google Scholar] [CrossRef]
- Yang, J.Y.; Park, J.H.; Chung, N.; Lee, H.S. Inhibitory potential of constituents from Osmanthus fragrans and structural analogues against advanced glycation end products, α-Amylase, α-Glucosidase, and oxidative stress. Sci. Rep. 2017, 7, 45746. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-C.; Lee, B.-H.; Lai, Y.-J. Antioxidation and antiglycation of Fagopyrum tataricum ethanol extract. J. Food Sci. Technol. 2015, 52, 1110–1116. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-Y.; Lim, S.-H.; Prak, Y.H.; Ham, H.J.; Lee, K.-J.; Park, D.S.; Kim, K.-H.; Kim, S. Screening of α-Amylase, α-Glucosidase and Lipase inhibitory activity with Gangwon-do wild plants extracts. J. Korean Soc. Food Sci. Nutr. 2011, 40, 308–315. [Google Scholar] [CrossRef]
- Lee, S.; Han, S.; Kim, H.M.; Lee, J.M.; Kim, J.; Park, C.-G.; Lee, S. Simultaneous determination of luteolin and luteoloside in dandelions using HPLC. Hort. Environ. Biotechnol. 2011, 52, 536–540. [Google Scholar] [CrossRef]
- Kassem, M.; Mosharrafa, S.A.; Saleh, N.A.; Abdel-Wahabb, S.M. Two new flavonoids from Retama raetam. Fitoterapia 2000, 71, 649–654. [Google Scholar] [CrossRef]
- Shalaby, N.M.; Maghraby, A.S.; el-Hagrassy, A.M. Effect of Daucus carota var. boissieri extracts on immune response of schistosoma mansoni infected mice. Folia Microbiol. 1999, 44, 441–448. [Google Scholar] [CrossRef]
- Gluchoff-Fiasson, K.; Favre-Bonvin, J.; Fiasson, J.L. Glycosides and acylated glycosides of isoetin from European species of Hypochoeris. Phytochemistry 1991, 30, 1670–1675. [Google Scholar] [CrossRef]
- Shi, S.Y.; Zhou, C.X.; Xu, Y.; Tao, Q.F.; Bai, H.; Lu, F.S.; Lin, W.Y.; Chen, H.Y.; Zheng, W.; Wang, L.W.; et al. Studies on chemical constituents from herbs of Taraxacum mongolicum. Zhongguo Zhong Yao Za Zhi 2008, 33, 1147–1157. [Google Scholar] [PubMed]
- Bi, Y.-F.; Zhieng, X.-K.; Feng, W.-S.; Shi, S.-P. Isolation and structural identification of chemical constituents from Selaginella tamariscina (Beauv.) Spring. Yao Xue Xue Bao = Acta Pharm. Sinica 2004, 39, 41–45. [Google Scholar]
- Forino, M.; Tartaglione, L.; Dell’Aversano, C.; Ciminiello, P. NMR-based identification of the phenolic profile of fruits of Lycium barbarum (goji berries). Isolation and structural determination of a novel N-feruloyl tyramine dimer as the most abundant antioxidant polyphenol of goji berries. Food Chem. 2016, 194, 1254–1259. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.H.; Zhao, B.T.; Lee, J.H.; Kim, Y.H.; Min, B.S.; Woo, M.H. Isolation of benzoic and cinnamic acid derivatives from the grains of Sorghum bicolor and their inhibition of lipopolysaccharide-induced nitric oxide production in RAW 264.7 cells. Food Chem. 2015, 168, 512–519. [Google Scholar] [CrossRef] [PubMed]
- Iwahashi, H.; Morishita, H.; Osaka, N.; Kido, R. 3-O-feruloyl-4-O-caffeoylquinic acid from coffee beans. Phytochemistry 1985, 24, 630–632. [Google Scholar] [CrossRef]
- Schmidt, B.; Jaroszewski, J.W.; Bro, R.; Witt, M.; Staerk, D. Combining PARAFAC analysis of HPLC-PDA profiles and structural characterization using HPLC-PDA-SPE-NMR-MS experiments: Commercial preparations of St. John’s Wort. Anal. Chem. 2008, 80, 1978–1987. [Google Scholar] [CrossRef] [PubMed]
- Cheminata, A.; Zawatzky, R.; Becker, H.; Brouillard, R. Caffeoyl conjugates from Echinacea species: Structures and biological activity. Phytochemistry 1988, 27, 2787–2794. [Google Scholar] [CrossRef]
- Crascì, L.; Lauro, M.R.; Puglisi, G.; Panico, A. Natural antioxidant polyphenols on inflammation management: Anti-glycation activity vs metalloproteinases inhibition. Crit. Rev. Food Sci. Nutr. 2018, 58, 893–904. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.K.; Bang, H.J.; Oh, J.B.; Whang, W.K. Bioassay-guided isolated compounds from Morinda officinalis inhibit alzheimer′s disease pathologies. Molecules 2017, 22, 1638. [Google Scholar]
- Ou, J.; Huang, J.; Wang, M.; Ou, S. Effect of rosmarinic acid and carnosic acid on AGEs formation in vitro. Food Chem. 2017, 221, 1057–1061. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |

| Sample | IC50 a (μg/mL) | ||
|---|---|---|---|
| BSA/Glucose | BSA/MGO | α-Glucosidase | |
| Ext. | 360.41 ± 23.58 *** | 311.33 ± 14.86 *** | 1623.08 ± 184.40 *** |
| Hx fr. | ND d | 529.24 ± 4.75 * | 1270.70 ± 72.58 *** |
| DCM fr. | 297.10 ± 5.26 ** | 247.47 ± 23.87 *** | 2041.44 ± 469.46 ** |
| EA fr. | 119.47 ± 12.06 ** | 127.31 ± 5.51 *** | 554.53 ± 36.46 *** |
| BuOH fr. | 333.71 ± 27.02 ** | 171.40 ± 69.28 * | ND d |
| Water fr. | 435.60 ± 59.57 ** | 213.96 ± 3.38 *** | ND d |
| T. officinale | 201.25 ± 33.99 *** | 105.37 ± 24.42 ** | 2195.89 ± 267.35 ** |
| T. coreanum | 183.97 ± 28.52 ** | 98.82 ± 2.51 * | 444.97 ± 55.86 ** |
| AMG b | 218.57 ± 24.38 *** | 67.51 ± 5.27 ** | - |
| Acarbose c | - | - | 116.86 ± 7.23 ** |
| Compound No. | Rt (min) | Formula | Mass Mode | Theoretical Mass | Observed Mass | Ass Error (Da) | Mass Accuracy (ppm) |
|---|---|---|---|---|---|---|---|
| 1 | 6.86 | C15H10O6 | Positive | 287.0550 | 287.0546 | 0.0004 | 1.4 |
| 2 | 5.89 | C21H20O11 | Positive | 449.1078 | 449.1076 | 0.0002 | 0.4 |
| 3 | 6.36 | C21H20O11 | Positive | 449.1078 | 449.1076 | 0.0002 | 0.4 |
| 4 | 5.86 | C27H30O15 | Positive | 595.1657 | 595.1650 | 0.0007 | 1.2 |
| 5 | 5.01 | C26H28O16 | Positive | 597.1450 | 597.1448 | 0.0002 | 0.3 |
| 6 | 5.26 | C26H28O16 | Positive | 597.1450 | 597.1443 | 0.0007 | 1.2 |
| 7 | 5.01 | C26H28O16 | Positive | 597.1450 | 597.1448 | 0.0002 | 0.3 |
| 8 | 4.30 | C9H8O4 | Negative | 179.0339 | 179.0340 | 0.0001 | 0.6 |
| 9 | 5.78 | C12H14O6 | Negative | 193.0495 | 193.0497 | 0.0002 | 1.0 |
| 10 | 4.87 | C10H10O4 | Negative | 253.0707 | 253.0714 | 0.0007 | 1.4 |
| 11 | 4.13 | C16H18O9 | Negative | 353.0867 | 353.0875 | 0.0008 | 2.3 |
| Compound | IC50 a (μM) | ||
|---|---|---|---|
| BSA/Glucose | BSA/MGO | α-Glucosidase | |
| 1 | 236.48 ± 9.11 ** | 66.11 ± 17.06 ** | ND |
| 2 | 122.81 ± 2.02 *** | 107.83 ± 8.14 ** | 1455.95 ± 126.32 *** |
| 3 | 423.30 ± 18.04 ** | 90.81 ± 21.74 ** | 598.24 ± 146.52 * |
| 4 | 253.31 ± 5.10 ** | 135.65 ± 2.64 ** | 670.50 ± 50.83 *** |
| 5 | 268.18 ± 3.41 ** | 129.79 ± 28.27 ** | ND d |
| 6 | 238.05 ± 13.82 *** | 148.37 ± 36.29 ** | ND d |
| 7 | 324.21 ± 8.29 ** | 79.65 ± 23.45 *** | 5134.55 ± 803.54 ** |
| 8 | 306.99 ± 10.16 ** | 151.67 ± 65.36 ** | 2951.13 ± 3.94 * |
| 9 | 704.86 ± 167.43 * | 140.72 ± 67.78 ** | ND d |
| 10 | 83.62 ± 55.49 *** | 138.18 ± 1.91 ** | 1148.67 ± 162.05 ** |
| 11 | 64.70 ± 16.73 ** | 141.21 ± 8.76 ** | 639.25 ± 12.51 *** |
| AMG b | 601.53 ± 50.35 *** | 295.21 ± 42.67 *** | - |
| Acarbose c | - | - | 355.86 ± 17.25 *** |
| Solvent Composition | Compound 1 (mg/g) | Compound 2 (mg/g) | Compound 7 (mg/g) | Compound 9 (mg/g) | Compound 10 (mg/g) | Compound 11 (mg/g) |
|---|---|---|---|---|---|---|
| 30% MeOH 60 min | 0.31 ± 0.07 | 0.49 ± 0.04 | 0.47 ± 0.13 | 4.58 ± 0.77 | 3.64 ± 0.35 | 131.98 ± 9.02 |
| 50% MeOH 60 min | 0.17 ± 0.05 | 0.65 ± 0.09 | 0.36 ± 0.09 | 4.66 ± 0.45 | 4.81 ± 0.45 | 181.58 ± 9.80 |
| 70% MeOH 60 min | 0.21 ± 0.03 | 0.61 ± 0.11 | 0.46 ± 0.04 | 4.71 ± 0.35 | 4.66 ± 0.72 | 173.37 ± 11.15 |
| 100% MeOH 60 min | 0.20 ± 0.02 | 0.32 ± 0.02 | 0.23 ± 0.02 | 4.53 ± 0.10 | 1.59 ± 0.19 | 46.06 ± 4.44 |
| 30% EtOH 60 min | 0.27 ± 0.04 | 0.73 ± 0.06 | 0.60 ± 0.06 | 4.62 ± 0.37 | 4.35 ± 0.31 | 161.46 ± 5.19 |
| 50% EtOH 60 min | 0.39 ± 0.09 | 0.76 ± 0.07 | 0.47 ± 0.06 | 4.69 ± 0.45 | 5.40 ± 0.19 | 204.98 ± 12.60 |
| 70% EtOH 60 min | 0.20 ± 0.07 | 0.68 ± 0.09 | 0.46 ± 0.01 | 4.68 ± 0.75 | 5.23 ± 0.26 | 203.60 ± 0.98 |
| 100% EtOH 60 min | 0.16 ± 0.04 | 0.30 ± 0.04 | 0.35 ± 0.05 | 4.39 ± 0.65 | 0.90 ± 0.33 | 33.65 ± 4.45 |
| 70% MeOH 30 min | 0.19 ± 0.05 | 0.61 ± 0.04 | 0.50 ± 0.04 | 4.77 ± 0.41 | 5.15 ± 0.44 | 206.12 ± 3.14 |
| 70% MeOH 60 min | 0.41 ± 0.10 | 0.79 ± 0.09 | 0.49 ± 0.09 | 4.75 ± 0.43 | 5.19 ± 0.49 | 210.91 ± 2.24 |
| 70% MeOH 90 min | 0.24 ± 0.01 | 0.75 ± 0.11 | 0.48 ± 0.12 | 4.76 ± 0.39 | 4.99 ± 0.30 | 208.72 ± 1.15 |
| 70% MeOH 120 min | 0.27 ± 0.03 | 0.73 ± 0.21 | 0.60 ± 0.14 | 4.62 ± 0.45 | 4.35 ± 0.41 | 161.46 ± 0.95 |
| Sample | Contents (mg/g) | |||||
|---|---|---|---|---|---|---|
| 1 | 2 | 7 | 9 | 10 | 11 | |
| Gyeongsangnam-do, Sancheong | 83.33 ± 0.14 | 28.30 ± 0.09 | 4.08 ± 0.09 | 4.59 ± 0.15 | 12.82 ± 0.21 | 562.36 ± 6.21 |
| Gyeongsangbuk-do, Yeongcheon | 41.37 ± 0.09 | 44.10 ± 0.11 | 1.23 ± 0.05 | 4.45 ± 0.09 | 20.32 ± 0.66 | 215.27 ± 0.2.11 |
| Gyeonggi-do, Gimpo | 5.08 ± 0.04 | 6.40 ± 0.04 | 1.17 ± 0.01 | 4.39 ± 0.11 | 2.02 ± 0.04 | 46.33 ± 0.96 |
| Chungcheongnam-do, Taean | 7.05 ± 0.02 | 15.87 ± 0.12 | 5.19 ± 0.05 | 4.47 ± 0.19 | 2.75 ± 0.04 | 106.10 ± 2.64 |
| Jeollabuk-do, Jeongeup | 7.10 ± 0.05 | 13.24 ± 0.07 | 3.69 ± 0.01 | 4.62 ± 0.16 | 10.53 ± 0.02 | 230.07 ± 2.98 |
| Gangwon-do, Yanggu | 1.23 ± 0.02 | 1.38 ± 0.09 | 1.52 ± 0.01 | 4.48 ± 0.05 | 4.54 ± 0.04 | 70.16 ± 1.65 |
| Gangwon-do, Yanggu | 1.06 ± 0.01 | 2.03 ± 0.09 | 2.80 ± 0.03 | 4.54 ± 0.04 | 24.11 ± 0.47 | 230.27 ± 3.11 |
| Gyeongsangbuk-do, Bonghwa | 10.10 ± 0.08 | 17.66 ± 0.09 | 2.57 ± 0.09 | 4.56 ± 0.04 | 13.14 ± 0.69 | 180.62 ± 2.64 |
| Compound Number | Rt (min) | Regression Equation | r2 | Linear Range (µg/mL) | LOD (µg/mL) | LOQ (µg/mL) |
|---|---|---|---|---|---|---|
| 1 | 0.9999 | 0.5–50 | 0.12 | 0.36 | ||
| 2 | 0.9999 | 0.5–50 | 0.24 | 0.73 | ||
| 7 | 0.9999 | 0.5–50 | 0.22 | 0.67 | ||
| 9 | 0.9728 | 0.5–50 | 0.27 | 0.82 | ||
| 10 | 0.9998 | 0.5–50 | 0.13 | 0.40 | ||
| 11 | 0.9999 | 0.5–50 | 0.31 | 0.95 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, K.H.; Whang, W.K. Inhibitory Effects of Bioassay-Guided Isolation of Anti-Glycation Components from Taraxacum coreanum and Simultaneous Quantification. Molecules 2018, 23, 2148. https://doi.org/10.3390/molecules23092148
Lee KH, Whang WK. Inhibitory Effects of Bioassay-Guided Isolation of Anti-Glycation Components from Taraxacum coreanum and Simultaneous Quantification. Molecules. 2018; 23(9):2148. https://doi.org/10.3390/molecules23092148
Chicago/Turabian StyleLee, Kang Hee, and Wan Kyunn Whang. 2018. "Inhibitory Effects of Bioassay-Guided Isolation of Anti-Glycation Components from Taraxacum coreanum and Simultaneous Quantification" Molecules 23, no. 9: 2148. https://doi.org/10.3390/molecules23092148
APA StyleLee, K. H., & Whang, W. K. (2018). Inhibitory Effects of Bioassay-Guided Isolation of Anti-Glycation Components from Taraxacum coreanum and Simultaneous Quantification. Molecules, 23(9), 2148. https://doi.org/10.3390/molecules23092148





