A Comparative Metabolomics Analysis Reveals the Tissue-Specific Phenolic Profiling in Two Acanthopanax Species
Abstract
1. Introduction
2. Results
2.1. Method Validation
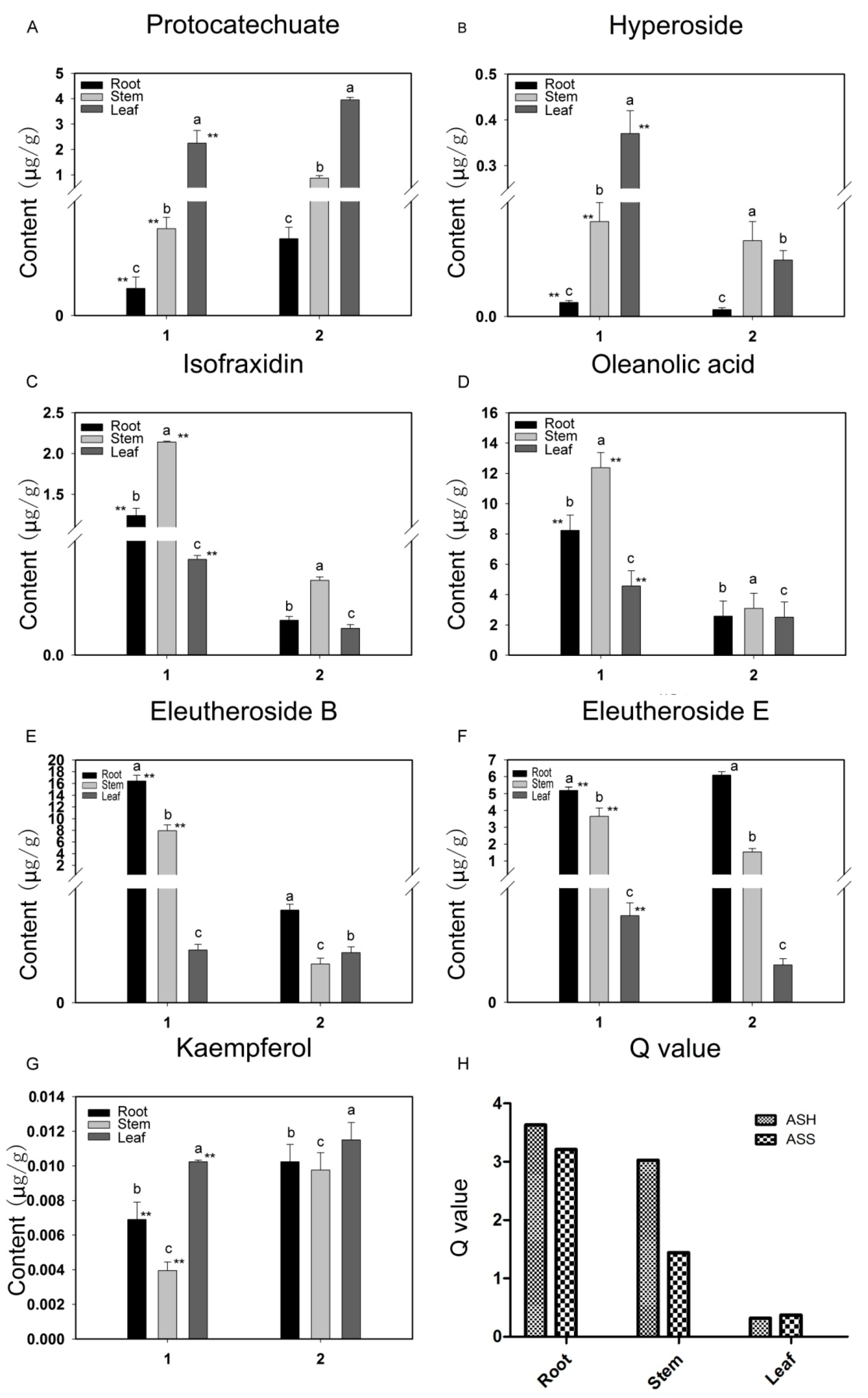
2.2. Accumulation of Pharmaceutically Active Compounds in Two Acanthopanax Species
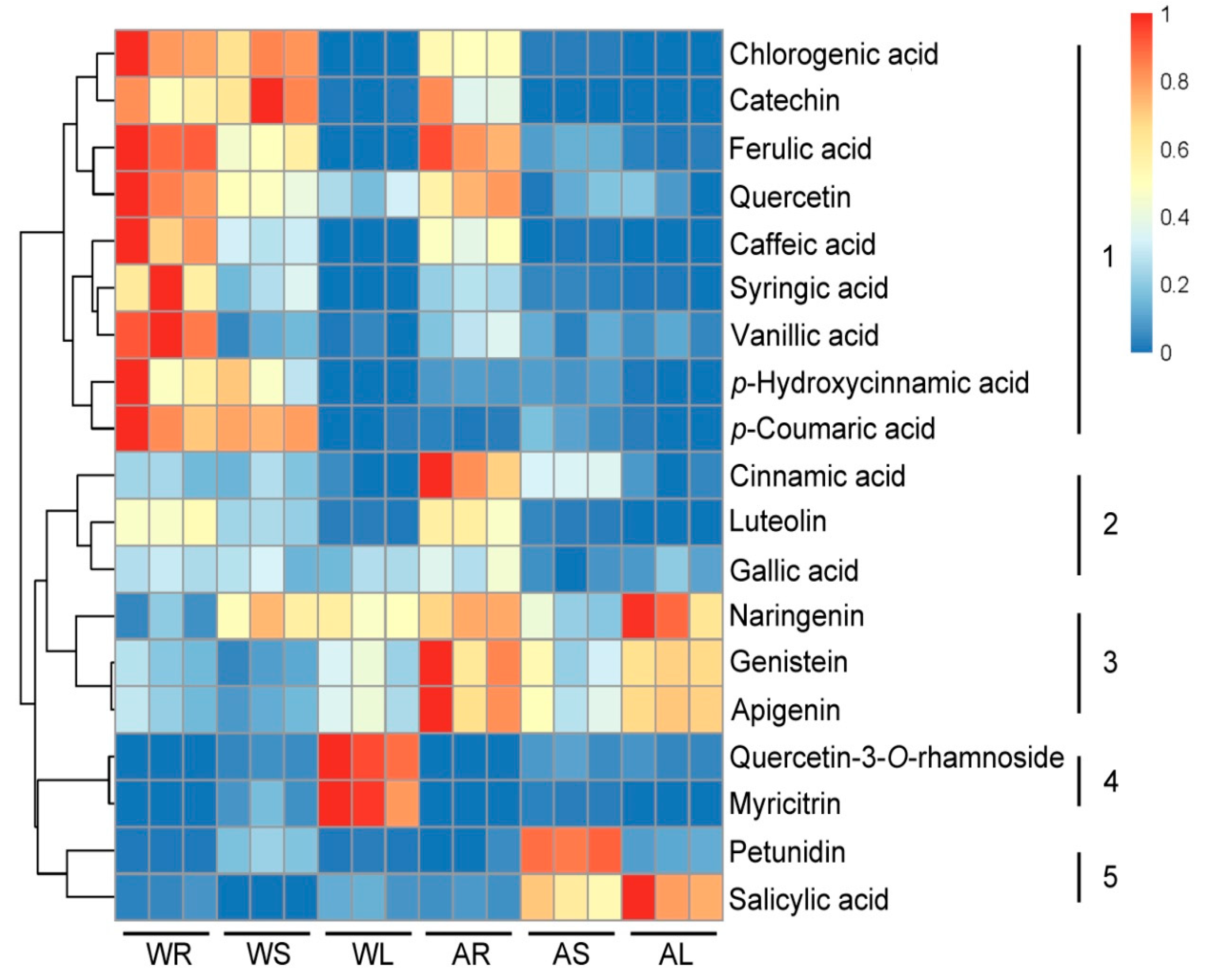
2.3. Targeted Analysis of Phenolic Compounds in Different Tissues of ASH and ASS
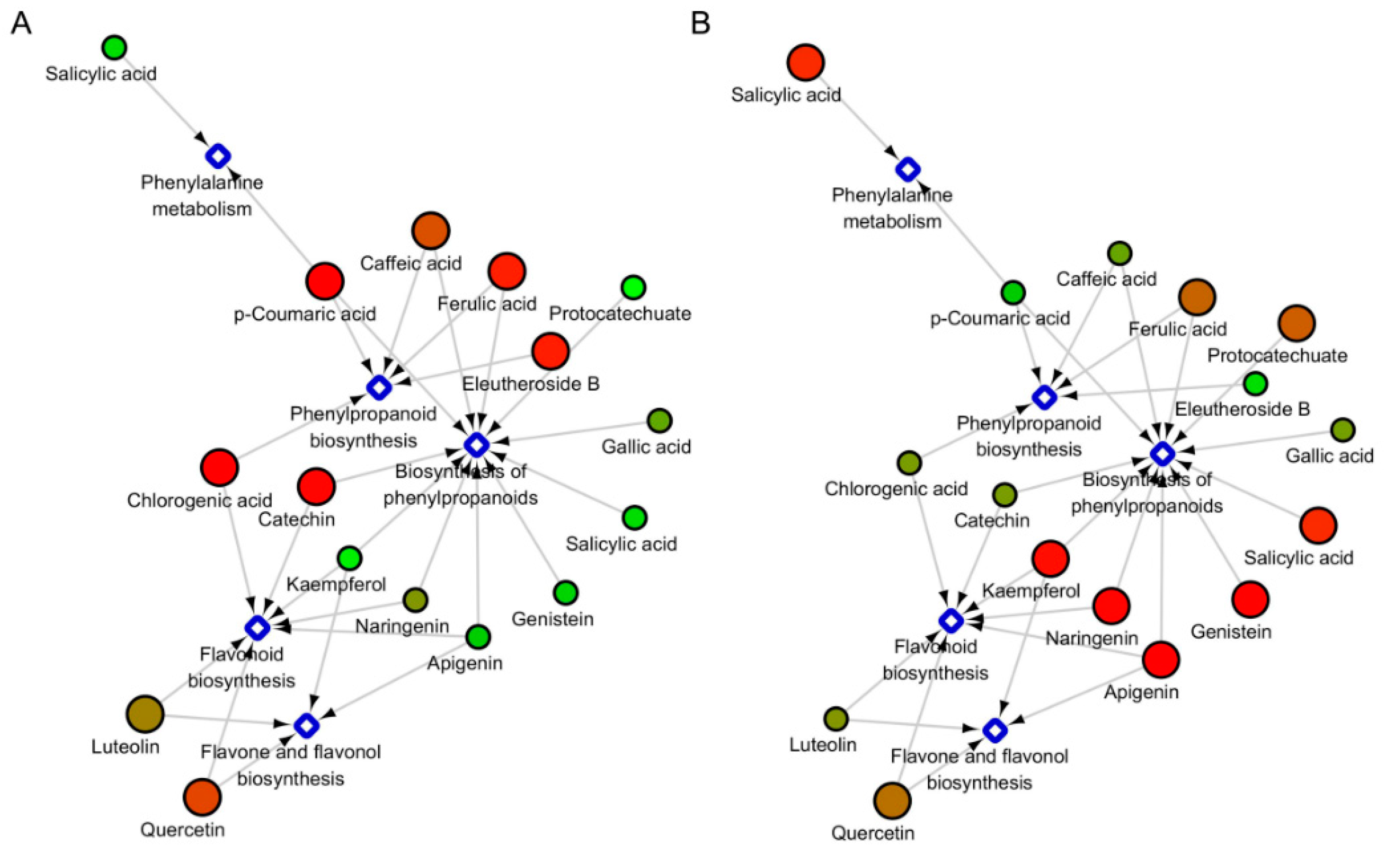
2.4. The Under-Ground and Above-Ground Distribution of Metabolites in the Specific Metabolic Pathways
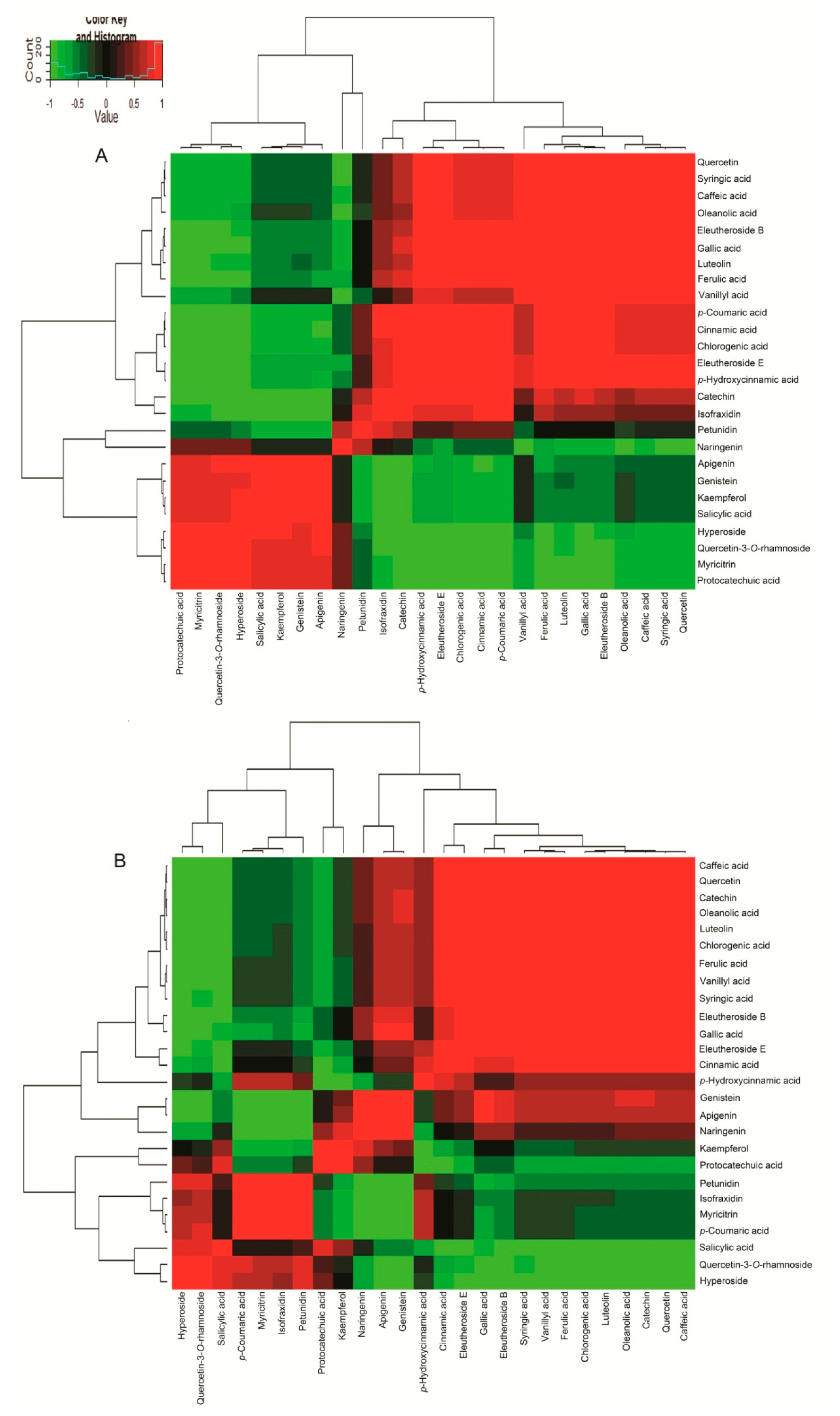
2.5. Correlation Analysis of 7 Active Compounds and 19 Phenols in ASH and ASS
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Sample Preparation
4.3. Instrumentation and Conditions
4.4. Data Processing and Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Li, Q.; Jia, Y.; Xu, L.; Wang, X.; Shen, Z.; Liu, Y.; Bi, K. Simultaneous determination of protocatechuic acid, syringin, chlorogenic acid, caffeic acid, liriodendrin and isofraxidin in Acanthopanax senticosus Harms by HPLC-DAD. Biol. Pharm. Bull. 2006, 29, 532–534. [Google Scholar] [CrossRef] [PubMed]
- Bączek, K.; Przybył, J.L.; Kosakowska, O.; Węglarz, Z. Accumulation of Phenolics in Eleuthero (Eleutherococcus senticosus (Rupr. Et Maxim.) Maxim.) as Affected by Plant Development. Acta Sci. Pol. Technol. Aliment. 2017, 16, 89–99. [Google Scholar] [CrossRef]
- Kim, Y.-H.; Cho, M.; Kim, D.-B.; Shin, G.-H.; Lee, J.-H.; Lee, J.; Park, S.-O.; Lee, S.-J.; Shin, H.; Lee, O.-H. The Antioxidant Activity and Their Major Antioxidant Compounds from Acanthopanax senticosus and A. koreanum. Molecules 2015, 20, 13281–13295. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, Y.; Zhang, A.; Zhang, Y.; Sun, H.; Meng, X.; Yan, G. Application of Ultra-performance liquid chromatography with Time-of-Flight mass spectrometry for the rapid analysis of constituents and metabolites from the extracts of Acanthopanax senticosus harms leaf. Pharmacogn. Mag. 2016, 12, 145. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Wang, Z.; Feng, X.; Deng, X.; Zhu, J. Simultaneous determination and pharmacokinetics of four triterpenoids by ultra high performance liquid chromatography with tandem mass spectrometry after the oral administration of Acanthopanax sessiliflorus (Rupr. et Maxim) Seem extract. J. Sep. Sci. 2016, 39, 2229–2237. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; An, Q.; Qin, F.; Xiong, Z. Simultaneous determination of six constituents in the fruit of Acanthopanax sessiliflorus (Rupr. et Maxim.) Seem. by HPLC–UV. Nat. Prod. Res. 2014, 28, 500–502. [Google Scholar] [CrossRef] [PubMed]
- Lu, F.; Sun, Q.; Bai, Y.; Bao, S.; Li, X.; Yan, G.; Liu, S. Characterization of eleutheroside B metabolites derived from an extract of Acanthopanax senticosus Harms by high-resolution liquid chromatography/quadrupole time-of-flight mass spectrometry and automated data analysis. Biomed. Chromatogr. 2012, 26, 1269–1275. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Zhao, H.; Huang, B.; Zheng, C.; Peng, W.; Qin, L. Acanthopanax senticosus: Review of botany, chemistry and pharmacology. Die Pharmazie 2011, 66, 83–97. [Google Scholar] [PubMed]
- Davydov, M.; Krikorian, A.D. Eleutherococcus senticosus (Rupr. & Maxim.) Maxim. (Araliaceae) as an adaptogen: A closer look. J. Ethnopharmacol. 2000, 72, 345–393. [Google Scholar] [PubMed]
- Sun, H.; Liu, J.; Zhang, A.; Zhang, Y.; Meng, X.; Han, Y.; Zhang, Y.; Wang, X. Characterization of the multiple components of Acanthopanax Senticosus stem by ultra high performance liquid chromatography with quadrupole time-of-flight tandem mass spectrometry. J. Sep. Sci. 2016, 39, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.-P.; An, J.-T.; Wang, R.; Li, Q. Simultaneous Quantification of Five Bioactive Components of Acanthopanax senticosus and Its Extract by Ultra Performance Liquid Chromatography with Electrospray Ionization Time-of-Flight Mass Spectrometry. Molecules 2012, 17, 7903–7913. [Google Scholar] [PubMed]
- Fujikawa, T.; Soya, H.; Hibasami, H.; Kawashima, H.; Takeda, H.; Nishibe, S.; Nakashima, K. Effect of Acanthopanax senticosus Harms on biogenic monoamine levels in the rat brain. Phytother. Res. 2002, 16, 474–478. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, Y.; Wang, Y.; Abozeid, A.; Zu, Y.-G.; Tang, Z.-H. The integration of GC–MS and LC–MS to assay the metabolomics profiling in Panax ginseng and Panax quinquefolius reveals a tissue- and species-specific connectivity of primary metabolites and ginsenosides accumulation. J. Pharm. Biomed. Anal. 2017, 135, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, Y.; Wang, Y.; Abozeid, A.; Zu, Y.-G.; Zhang, X.-N.; Tang, Z.-H. GC-MS Metabolomic Analysis to Reveal the Metabolites and Biological Pathways Involved in the Developmental Stages and Tissue Response of Panax ginseng. Molecules 2017, 22, 496. [Google Scholar] [CrossRef]
- Ben Brahim, S.; Kelebek, H.; Ammar, S.; Abichou, M.; Bouaziz, M. LC–MS phenolic profiling combined with multivariate analysis as an approach for the characterization of extra virgin olive oils of four rare Tunisian cultivars during ripening. Food Chem. 2017, 229, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sun, Y.; Xi, W.; Shen, Y.; Qiao, L.; Zhong, L.; Ye, X.; Zhou, Z. Phenolic compositions and antioxidant capacities of Chinese wild mandarin (Citrus reticulata Blanco) fruits. Food Chem. 2014, 145, 674–680. [Google Scholar] [CrossRef] [PubMed]
- Mertz, C.; Cheynier, V.; Günata, Z.; Brat, P. Analysis of phenolic compounds in two blackberry species (Rubus glaucus and Rubus adenotrichus) by high-performance liquid chromatography with diode array detection and electrospray ion trap mass spectrometry. J. Agric. Food Chem. 2007, 55, 8616–8624. [Google Scholar] [CrossRef] [PubMed]
- Luengo Escobar, A.; Alberdi, M.; Acevedo, P.; Machado, M.; Nunes-Nesi, A.; Inostroza-Blancheteau, C.; Reyes-Díaz, M. Distinct physiological and metabolic reprogramming by highbush blueberry (Vaccinium corymbosum) cultivars revealed during long-term UV-B. radiation. Physiol. Plant. 2017, 160, 46–64. [Google Scholar] [CrossRef] [PubMed]
- Mirali, M.; Purves, R.W.; Vandenberg, A. Profiling the Phenolic Compounds of the Four Major Seed Coat Types and Their Relation to Color Genes in Lentil. J. Nat. Prod. 2017, 80, 1310–1317. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, Y.; Wang, Y.; Zhang, Z.H.; Zu, Y.G.; Efferth, T.; Tang, Z.H. The Combined Effects of Ethylene and MeJA on Metabolic Profiling of Phenolic Compounds in Catharanthus roseus Revealed by Metabolomics Analysis. Front. Physiol. 2016, 7, 217. [Google Scholar] [CrossRef] [PubMed]
- García-Calderón, M.; Pons-Ferrer, T.; Mrázova, A.; Pal’Ove-Balang, P.; Vilková, M.; Pérez-Delgado, C.M.; Vega, J.M.; Eliášová, A.; Repčák, M.; Márquez, A.J.; et al. Modulation of phenolic metabolism under stress conditions in a Lotus japonicus mutant lacking plastidic glutamine synthetase. Front. Plant Sci. 2015, 6, 760. [Google Scholar] [CrossRef] [PubMed]
- Abaza, L.; Taamalli, A.; Nsir, H.; Zarrouk, M. Olive tree (Olea europeae L.) leaves: Importance and advances in the analysis of phenolic compounds. Antioxidants 2015, 4, 682–698. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Zheng, C.; Huang, J.; You, T. Identification of herb Acanthopanax senticosus (Rupr. Et Maxim.) harms by capillary electrophoresis with electrochemical detection. Anal. Sci. 2007, 23, 705–711. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, Y.; Pan, Y.-J.; Zu, Y.-G.; Tang, Z.-H. Determination of Alkaloids in Catharanthus roseus and Vinca minorby High-Performance Liquid Chromatography–Tandem Mass Spectrometry. Anal. Lett. 2015, 49, 1143–1153. [Google Scholar] [CrossRef]
- Liu, J.; Liu, Y.; Zhang, Z.-H.; Zu, Y.-G.; Tang, Z.-H.; Efferth, T. Correlation of cultivation time of Panax ginseng with metabolic profiles of nine ginsenosides and mRNA expression of genes encoding major biosynthetic enzymes. Acta Physiol. Plant. 2016, 38, 51. [Google Scholar] [CrossRef]
- Abozeid, A.; Liu, J.; Liu, Y.; Wang, H.; Tang, Z. Gas chromatography mass spectrometry–based metabolite profiling of two sweet-clover vetches via multivariate data analyses. Bot. Lett. 2017, 164, 385–391. [Google Scholar] [CrossRef]
- Liu, J.; Liu, Y.; Zhao, L.; Zhang, Z.-H.; Tang, Z.-H. Profiling of ginsenosides in the two medicinal Panax herbs based on ultra-performance liquid chromatography-electrospray ionization–mass spectrometry. Springerplus 2016, 5, 1770. [Google Scholar] [CrossRef] [PubMed]
- Huilan, Y.; Junjun, D.; Zhen, H.; Chengxin, P.; Shilei, L.; Yu, X. Synthesis and analysis of phosphorylated nonapeptide adducts by LC/Q-TOF MS. Phosphorus Sulfur Silicon Relat. Elem. 2015, 191, 111–116. [Google Scholar] [CrossRef]
- Noh, E.; Yoon, C.-Y.; Lee, J.H.; Lee, J.-M.; Baek, S.-Y.; Oh, H.B.; Do, J.-A. A Liquid Chromatography-Quadrupole-Time of Flight Mass Spectrometry (LC-Q-TOF MS) Study for Analyzing 35 Corticosteroid Compounds: Elucidation of MS/MS Fragmentation Pathways. Bull. Korean Chem. Soc. 2016, 37, 1029–1038. [Google Scholar] [CrossRef]
- Ren, Q.; Ding, L.; Sun, S.-S.; Wang, H.-Y.; Qu, L. Chemical identification and quality evaluation of Lycopus lucidus Turcz by UHPLC-Q-TOF-MS and HPLC-MS/MS and hierarchical clustering analysis. Biomed. Chromatogr. 2017, 31, e3867. [Google Scholar] [CrossRef] [PubMed]
- Xing, T.T.; Zhao, X.J.; Zhang, Y.D.; Li, Y.F. Fast Separation and Sensitive Quantitation of Polymethoxylated Flavonoids in the Peels of Citrus Using UPLC-Q.-TOF-MS. J. Agric. Food Chem. 2017, 65, 2615–2627. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Zhang, A.; Sun, H.; Zhang, Y.; Meng, X.; Yan, G.; Liu, L.; Wang, X. High-throughput ultra high performance liquid chromatography combined with mass spectrometry approach for the rapid analysis and characterization of multiple constituents of the fruit of Acanthopanax senticosus (Rupr. et Maxim.) Harms. J. Sep. Sci. 2017, 40, 2178–2187. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.S.; Linh, P.T.; Cai, X.F.; Kim, H.S.; Lee, J.J.; Kim, Y.H. Quantitative determination of eleutheroside B and E from Acanthopanax species by high performance liquid chromatography. Arch. Pharm. Res. 2001, 24, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.-J.; Liu, J.; Guo, X.-R.; Zu, Y.-G.; Tang, Z.-H. Gene transcript profiles of the TIA biosynthetic pathway in response to ethylene and copper reveal their interactive role in modulating TIA biosynthesis in Catharanthus roseus. Protoplasma 2014, 252, 813–824. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Xing, J.; Liu, S.; Song, F.; Cai, Z.; Pi, Z.; Liu, Z.; Liu, S. Screening and Determination for Potential α-Glucosidase Inhibitors from Leaves of Acanthopanax senticosus Harms by Using UF-LC/MS and ESI-MSn. Phytochem. Anal. 2012, 23, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.; Hu, F.; Zhao, J.; Liu, X.; Li, Y. Determination of eleutheroside E and eleutheroside B in rat plasma and tissue by high-performance liquid chromatography using solid-phase extraction and photodiode array detection. Eur. J. Pharm. Biopharm. 2006, 62, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Jun, W.J.; Seong, H.S.; Chun, H.; Lim, E.J.; Kim, K.I.; Cho, H.Y. Determination of antioxidative potentials of Acanthopanax sessiliflorus (Rupr. & Maxim.) Seem. in differentiated HL-60 cells. Food Chem. 2007, 105, 1557–1563. [Google Scholar]
- Ali, A.H.; Zou, X.; Lu, J.; Abed, S.M.; Yao, Y.; Tao, G.; Jin, Q.; Wang, X. Identification of phospholipids classes and molecular species in different types of egg yolk by using UPLC-Q-TOF-MS. Food Chem. 2017, 221, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Cunha, A.G.; Brito, E.S.; Moura, C.F.H.; Ribeiro, P.R.V.; Miranda, M.R.A. UPLC–qTOF-MS/MS-based phenolic profile and their biosynthetic enzyme activity used to discriminate between cashew apple (Anacardium occidentale L.) maturation stages. J. Chromatogr. B 2017, 1051, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Lu, X.; Guo, X.; Guo, Q.; Li, D. Metabolomics Characterization of Two Apocynaceae Plants, Catharanthus roseus and Vinca minor, Using GC-MS and LC-MS Methods in Combination. Molecules 2017, 22, 997. [Google Scholar] [CrossRef] [PubMed]
- Park, H.W.; Cho, S.Y.; Kim, H.H.; Yun, B.S.; Kim, J.U.; Lee, S.J.; Park, J. Enantioselective induction of SIRT1 gene by syringaresinol from Panax ginseng berry and Acanthopanax senticosus Harms stem. Bioorg. Med. Chem. Lett. 2015, 25, 307–309. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, D.R.A.; Praseptiangga, D.; Van de Walle, D.; Dewettinck, K. Interaction between natural antioxidants derived from cinnamon and cocoa in binary and complex mixtures. Food Chem. 2017, 231, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, J.; Wang, Y.; Abozeid, A.; Tian, D.-M.; Zhang, X.-N.; Tang, Z.-H. The Different Resistance of Two Astragalus Plants to UV-B Stress is Tightly Associated with the Organ-specific Isoflavone Metabolism. Photochem. Photobiol. 2018, 94, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Trantas, E.A.; Koffas, M.A.; Xu, P.; Ververidis, F. When plants produce not enough or at all: Metabolic engineering of flavonoids in microbial hosts. Front. Plant Sci. 2015, 6, 7. [Google Scholar] [CrossRef] [PubMed]
- Pei, K.; Ou, J.; Huang, J.; Ou, S. p-Coumaric acid and its conjugates: Dietary sources, pharmacokinetic properties and biological activities. J. Sci. Food Agric. 2016, 96, 2952–2962. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Salcedo, E.M.; Mena, P.; García-Viguera, C.; Hernández, F.; Martínez, J.J. (Poly)phenolic compounds and antioxidant activity of white (Morus alba) and black (Morus nigra) mulberry leaves: Their potential for new products rich in phytochemicals. J. Funct. Foods 2015, 18, 1039–1046. [Google Scholar] [CrossRef]
- An, C.; Mou, Z. Salicylic Acid and its Function in Plant Immunity. J. Integr. Plant Biol. 2011, 53, 412–428. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Mazarei, M.; Zhao, N.; Zhu, J.J.; Zhuang, X.; Liu, W.; Pantalone, V.R.; Arelli, P.R.; Stewart, C.N.; Chen, F. Overexpression of a soybean salicylic acid methyltransferase gene confers resistance to soybean cyst nematode. Plant Biotechnol. J. 2013, 11, 1135–1145. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, N.R.; Verpoorte, R. Phenolic compounds in Catharanthus roseus. Phytochem. Rev. 2007, 6, 243–258. [Google Scholar] [CrossRef]
- Mouradov, A.; Spangenberg, G. Flavonoids: A metabolic network mediating plants adaptation to their real estate. Front. Plant Sci. 2014, 5, 620. [Google Scholar] [CrossRef] [PubMed]
- Cheynier, V.; Comte, G.; Davies, K.M.; Lattanzio, V.; Martens, S. Plant phenolics: Recent advances on their biosynthesis, genetics, and ecophysiology. Plant Physiol. Biochem. 2013, 72, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Park, S.Y.; Lim, S.-H.; Yeo, Y.; Cho, H.S.; Ha, S.-H. Comparative metabolic profiling of pigmented rice (Oryza sativa L.) cultivars reveals primary metabolites are correlated with secondary metabolites. J. Cereal Sci. 2013, 57, 14–20. [Google Scholar] [CrossRef]
- Murphy, A.; Peer, W.A.; Taiz, L. Regulation of auxin transport by aminopeptidases and endogenous flavonoids. Planta 2000, 211, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Watkins, J.M.; Hechler, P.J.; Muday, G.K. Ethylene-Induced Flavonol Accumulation in Guard Cells Suppresses Reactive Oxygen Species and Moderates Stomatal Aperture. Plant Physiol. 2014, 164, 1707–1717. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds in the methanol extract from Acanthopanax senticosus (Rupr. Maxim.) Harms (ASH) and Acanthopanax sessiliflorus (Rupr. Maxim.) Seem are available from the authors. |
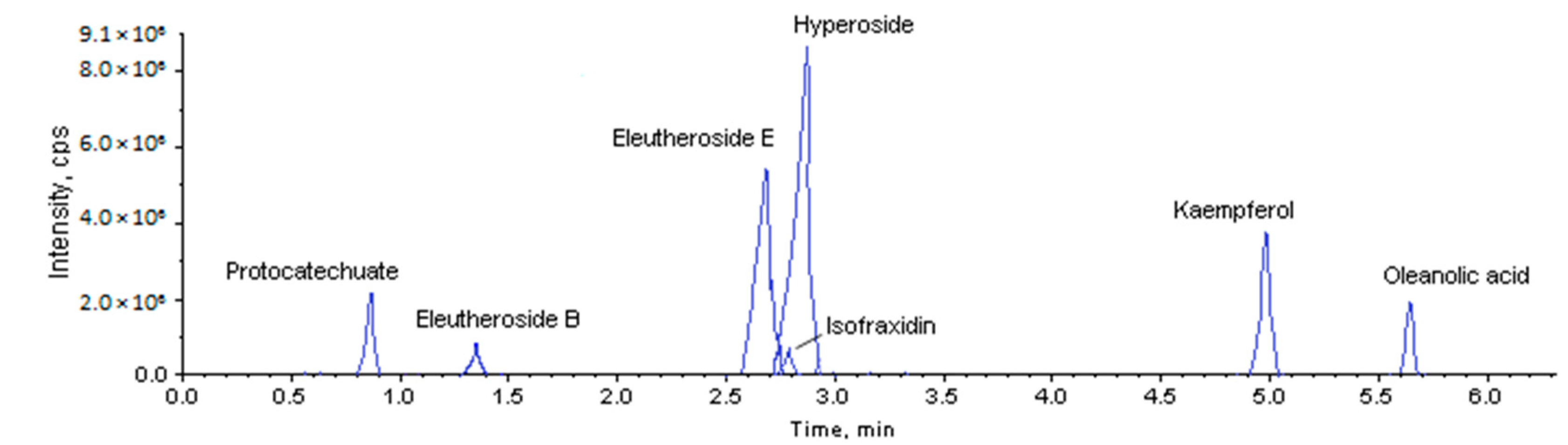
| Compounds | m/z | DP | CE | CXP |
|---|---|---|---|---|
| Oleanolic acid | 479.3→435.5 | 70 | 28 | 17 |
| Kaempferol | 287.1→153 | 70 | 51 | 10 |
| Isofraxidin | 223.1→206.3 | 60 | 40 | 9 |
| Eleutheroside B | 394.8→231.8 | 70 | 40 | 17 |
| Eleutheroside E | 765.3→603.1 | 70 | 62 | 23 |
| Hyperoside | 487→324 | 70 | 43 | 17 |
| Protocatechuate | 155→92.9 | 70 | 21 | 17 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, K.-X.; Liu, J.; Liu, Y.; Guo, X.-R.; Mu, L.-Q.; Hu, X.-H.; Tang, Z.-H. A Comparative Metabolomics Analysis Reveals the Tissue-Specific Phenolic Profiling in Two Acanthopanax Species. Molecules 2018, 23, 2078. https://doi.org/10.3390/molecules23082078
Wu K-X, Liu J, Liu Y, Guo X-R, Mu L-Q, Hu X-H, Tang Z-H. A Comparative Metabolomics Analysis Reveals the Tissue-Specific Phenolic Profiling in Two Acanthopanax Species. Molecules. 2018; 23(8):2078. https://doi.org/10.3390/molecules23082078
Chicago/Turabian StyleWu, Ke-Xin, Jia Liu, Yang Liu, Xiao-Rui Guo, Li-Qiang Mu, Xiao-Hang Hu, and Zhong-Hua Tang. 2018. "A Comparative Metabolomics Analysis Reveals the Tissue-Specific Phenolic Profiling in Two Acanthopanax Species" Molecules 23, no. 8: 2078. https://doi.org/10.3390/molecules23082078
APA StyleWu, K.-X., Liu, J., Liu, Y., Guo, X.-R., Mu, L.-Q., Hu, X.-H., & Tang, Z.-H. (2018). A Comparative Metabolomics Analysis Reveals the Tissue-Specific Phenolic Profiling in Two Acanthopanax Species. Molecules, 23(8), 2078. https://doi.org/10.3390/molecules23082078






